Abstract
Acrylonitrile, an industrial chemical, is a multisite carcinogen in rats and mice, producing tumors in four tissues with barrier function, that is, brain, forestomach, Zymbal’s gland, and Harderian gland. To assess mechanism(s) of action (MoA) for induction of neoplasia and to evaluate whether the findings in rodents are indicative of human hazard, data on the potential key effects produced by acrylonitrile in the four rodent target tissues of carcinogenicity were evaluated. A notable finding was depletion of glutathione in various organs, including two target tissues, the brain, and forestomach, suggesting that this effect could be a critical initiating event. An additional combination of oxidative DNA damage and cytotoxic effects of acrylonitrile and its metabolites, cyanide, and 2-cyanoethylene oxide, could initiate pro-inflammatory signaling and sustained cell and tissue injury, leading to compensatory cell proliferation and neoplastic development. The
Keywords
Introduction
Acrylonitrile, also known as vinyl cyanide, is an organic compound widely used as a monomer in industry for the synthesis of polymers and resins.1,2 Human exposure to acrylonitrile occurs mainly through inhalation in occupational settings or through cigarette smoke, exposures by oral and dermal routes are uncommon.1,2 Small amounts of acrylonitrile can be also released from the combustion of plant matter (e.g., biomass). 3
The carcinogenicity of acrylonitrile in rodents has been extensively investigated and large-scale epidemiological studies have been conducted. While in humans most epidemiological studies produced no consistent findings of increased cancer risk associated with acrylonitrile exposure,4–12 in rodent carcinogenicity bioassays, acrylonitrile produced tumors in multiple sites, including the brain, mammary gland, and Zymbal’s (auditory sebaceous tissue) gland of rats, forestomach of rats and mice, and Harderian (secretory orbital tissue) gland of mice. Increases in brain tumors observed in several acrylonitrile bioassays in male and female rats of two strains,13,14 although not in mice,15,16 led to a concern that acrylonitrile could be a central nervous system carcinogen in humans.
Previously, Meek et al. 17 reviewed the evidence relating to key events in acrylonitrile rodent brain carcinogenicity and whether the mechanisms of acrylonitrile carcinogenicity in rodents are plausible in humans. They concluded that the data available at that time were not sufficient to support a consensus view on a plausible mode of action for acrylonitrile-induced rat brain tumors. The subsequent finding that these tumors in rats were microglial/histiocytic in origin (i.e., malignant microgliomas), 18 as well as the availability of new mechanistic studies,19–24 prompted the present re-evaluation of potential mechanism(s) of action (MoA(s)) of acrylonitrile and relevance to humans.
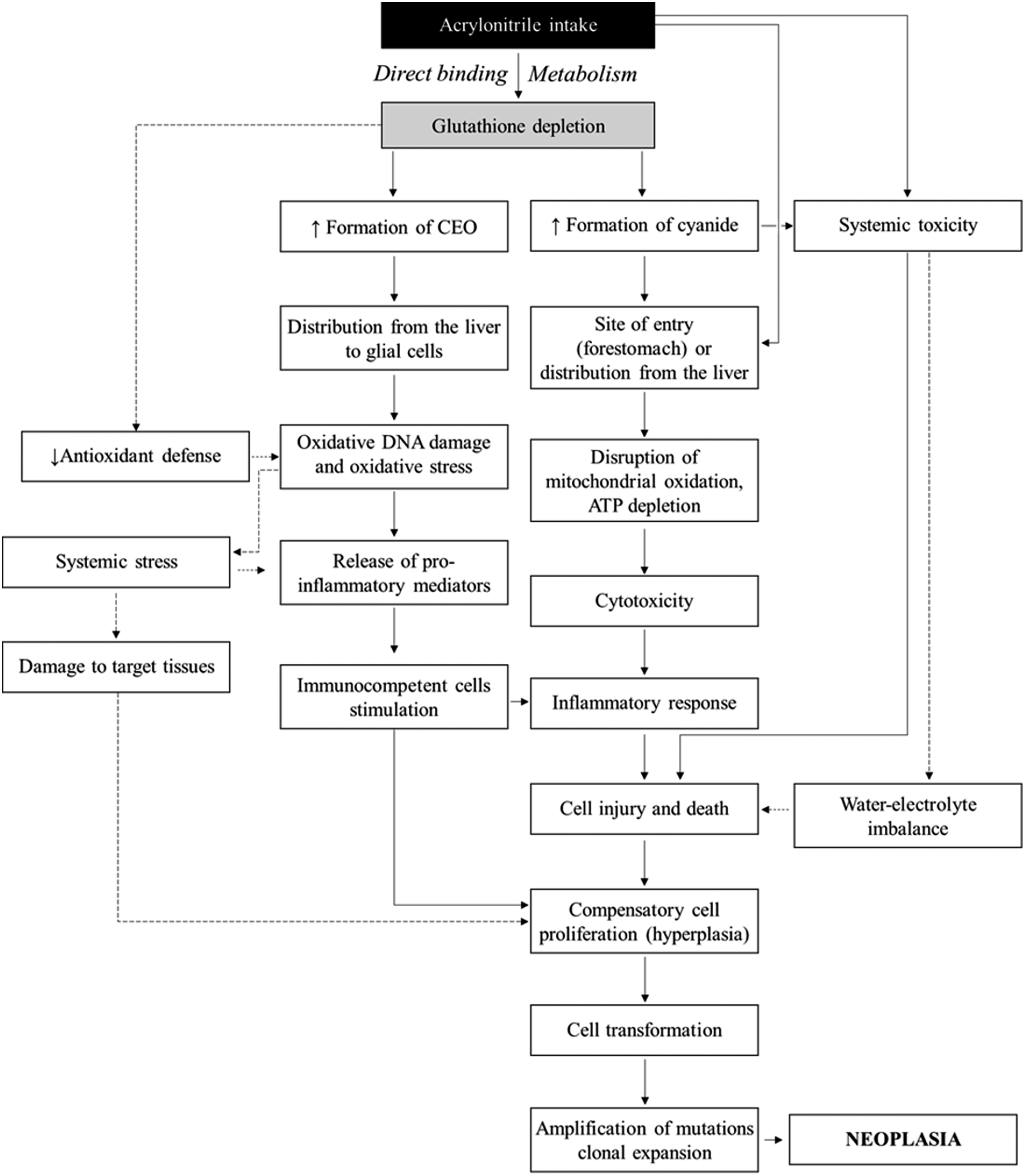
This review of available data was undertaken to establish MoA(s) for the principal rodent neoplasia findings in four target tissues. The assessment leads to the hypothesis that the MoA for tumor increases elicited by acrylonitrile in forestomach, brain, Zymbal’s gland, and Harderian gland could, in part, involve cytotoxic effects to target tissues by the parent compound and its metabolites, cyanide, and 2-cyanoethyleneoxide (CEO) due to rapid depletion of glutathione (GSH) in target organs and liver. In the brain, cytotoxicity was accompanied by oxidative stress. Those effects are postulated to initiate a cascade of events leading to neoplasms in the four vulnerable barrier tissues, which are susceptible to tumorigenesis. 25 It is possible, however, that more than one MoA can be involved in carcinogenicity produced by acrylonitrile in rodents, and definitive mechanistic data for a MoA were found to be lacking.
Methods
Literature search
A search of the published literature was conducted for combinations of the following terms: acrylonitrile, vinyl cyanide, toxicity, single-dose, repeat dose, acute, chronic, long-term, reproductive, developmental, carcinogenicity, tumorigenicity, cancer, neoplasia, tumor, malignancy, forestomach, brain, Zymbal’s gland, Harderian gland, epidemiologic, biomonitoring, genotoxicity, 1. Pub Med (https://www.ncbi.nlm.nih.gov/pubmed) is a free source premier database for biomedical literature indexed in the National Library of Medicine MEDLINE database. It contains over 30 million citations from over 5000 current biomedical journals. Coverage range: 1950s to present. 2. EMBASE (https://www.embase.com) is a comprehensive index of the biomedical and pharmacological literature. It includes citations from over 8400 journals, 1000 conferences, and 70 countries. Coverage range: 1947 to present. 3. Web of Science (http://apps.webofknowledge.com) provides access to current and retrospective abstracts and cited references found in 3700 journals covering more than 100 disciplines. Coverage range: 1996 to present. 4. Science Direct (http://www.sciencedirect.com) is a web collection of 161 full-text scientific journals and e-books. Coverage range: 1995 to present. 5. ChemIDplus database (https://chem.nlm.nih.gov) provides access to the structure and properties for chemical substances cited in National Library of Medicine database. The database contains more than 400,000 chemical records.
In addition, safety profiles for acrylonitrile prepared by regulatory organizations including European Chemicals Agency, 3 European Chemicals Bureau, 1 IARC,2,26,27 Occupational Safety and Health Administration, 28 and US EPA29,30 were reviewed. Publications written in languages other than English were excluded.
MoA analyses
To address some concerns of the robustness of the IARC approach, 32 a weight of evidence (WoE) evaluation for the key events of hypothesized MoA(s) based on evolved Bradford Hill considerations 33 (Table 1) was also applied.
Dose-response assessment for carcinogenicity findings
In order to characterize carcinogenic potency of acrylonitrile, tumor findings in target organs in rodent chronic bioassays were analyzed using the Benchmark dose (BMD) approach. BMD modeling was performed using the US EPA BMDS software version 3.0, according to the guidance in the software manual. A multistage-cancer model was used to calculate the BMD10 (indicative of a 10% increase in tumor incidence over the control) for each data set. This model was selected for the present analyses since it also calculates a cancer slope factor (CSF). This cancer potency factor is an upper-bound estimate of the increase in cancer risk after a lifetime exposure of 70 years to a chemical by ingestion or inhalation. The fit of the model is given in Supplemental Figures 1–4. The BMDL and BMDU refer to the lower and upper bounds of a two-sided 95% confidence interval for calculated BMD10.
Results
Summary of acrylonitrile toxicity and carcinogenicity in humans
Humans exhibit acute toxicity to acrylonitrile. Thus, in workers, acute inhalational and/or dermal exposure to acrylonitrile at various concentrations above 5 ppm (10.9 mg/m3) was reported to result in irritation of eyes and nose, cyanosis, and neurological symptoms (headache, dizziness, fatigue, nausea, and vomiting).2,3 These symptoms were reversible. Cases of occupational contact dermatitis have been reported after dermal exposure to acrylonitrile. 34
A considerable body of epidemiological data has not revealed evidence of human cancer risk from acrylonitrile exposure.5–12 While a recent retrospective study by the US National Cancer Institute on mortality of workers exposed to acrylonitrile reported a statistically significant (
The International Agency for Research on Cancer (IARC) conducted several evaluations of acrylonitrile. In the first evaluation, IARC concluded that acrylonitrile “
Experimental carcinogenicity data and summary of neoplastic findings
Acrylonitrile was reported to produce tumors in multiple sites in rats and mice in a dose-related fashion in several chronic rodent studies, conducted with various protocols, utilizing several modes of acrylonitrile administration. Six of these studies of greater than 18 months in duration have been validated and accepted by regulatory agencies, and hence, were selected as the dataset for the current analyses. Five out of six studies were conducted in rats, with acrylonitrile administered in drinking water,14,37,38 by oral gavage 37 or inhalation. 39 One study was conducted in mice dosed with acrylonitrile by oral gavage.15,16 Findings in several other chronic bioassays conducted with acrylonitrile13,40,41 are also briefly discussed.
Systemic toxicity-associated findings in rodent carcinogenicity studies of acrylonitrile.
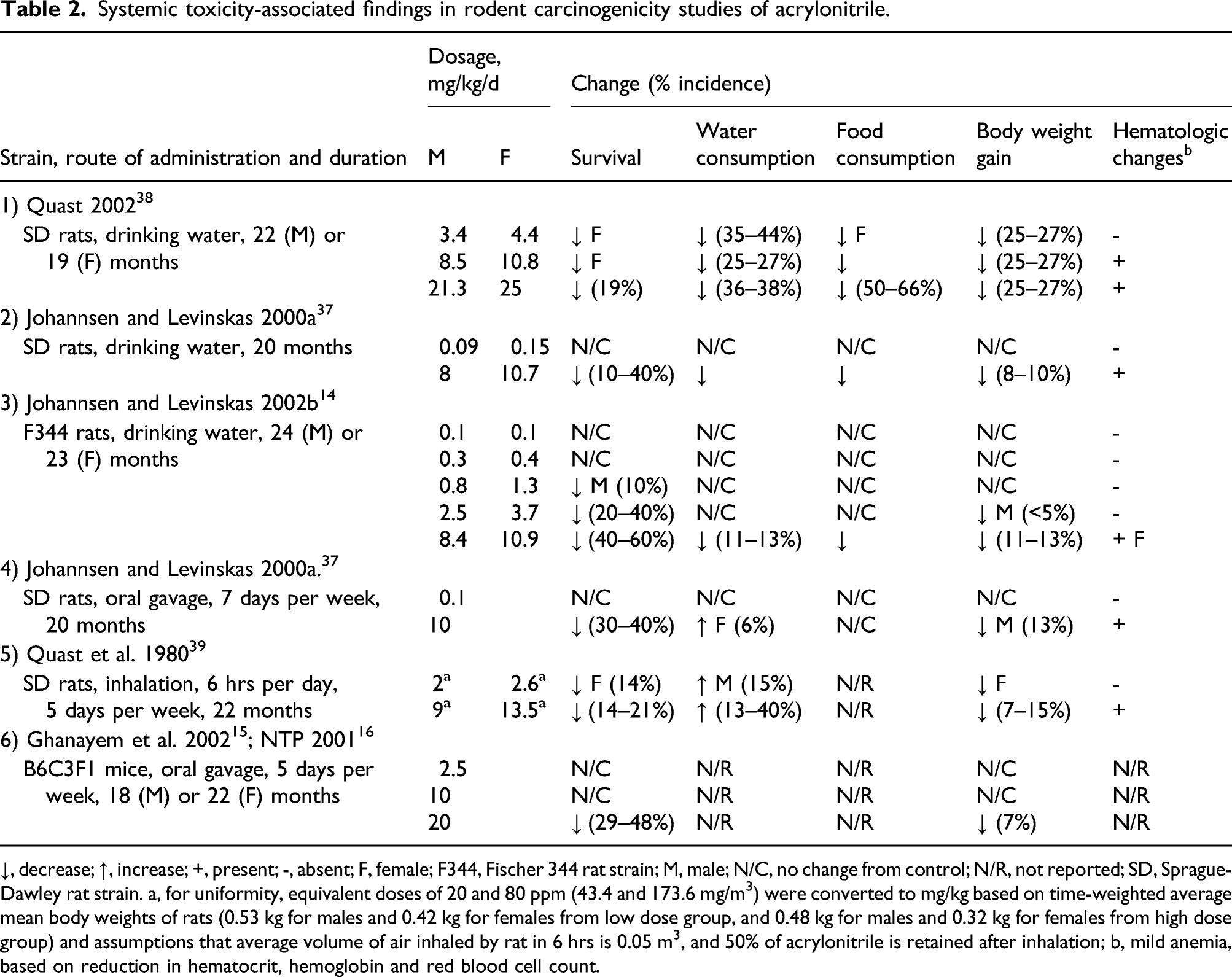
↓, decrease; ↑, increase; +, present; -, absent; F, female; F344, Fischer 344 rat strain; M, male; N/C, no change from control; N/R, not reported; SD, Sprague-Dawley rat strain. a, for uniformity, equivalent doses of 20 and 80 ppm (43.4 and 173.6 mg/m3) were converted to mg/kg based on time-weighted average mean body weights of rats (0.53 kg for males and 0.42 kg for females from low dose group, and 0.48 kg for males and 0.32 kg for females from high dose group) and assumptions that average volume of air inhaled by rat in 6 hrs is 0.05 m3, and 50% of acrylonitrile is retained after inhalation; b, mild anemia, based on reduction in hematocrit, hemoglobin and red blood cell count.
Of the three modes of acrylonitrile administration to rats, intake in drinking water in both SD and F344 strains most severely impacted survival, especially in female rats, which were dosed with higher doses (up to 17% greater) compared to males (Table 2). An increase in mortality rates began at 10 and 16 months at 300 ppm and at 12 and 20 months at 100 ppm in both female and male rats, respectively, and at 18 months in both sexes at 35 ppm in the drinking water. 38 The remainder of acrylonitrile-dosed rats of both sexes were terminated at 22 months. Reduction in survival was similar for the same oral dosages between the SD and F344 strains of rats.14,37 With gavage administration, increased mortality was evident in rats of both sexes at 8.5 (male)/10.8 (female) mg/kg/day beginning at 14 months, until study termination at 20 months. 37 In B6C3F1 mice, which also were dosed with acrylonitrile by oral gavage,15,16 the high dose (14.3 mg/kg/day) produced significant reduction in survival in both sexes (males greater than females), after approximately 1 year of dosing. In the rat inhalation study, mortality was increased at 80 ppm, starting at 6 months in males, and 10 months in females, until termination of the study at 22 months. 39
Percent incidence of pertinent non-neoplastic and neoplastic findings in Sprague-Dawley rats dosed with acrylonitrile in drinking water for 2 years.
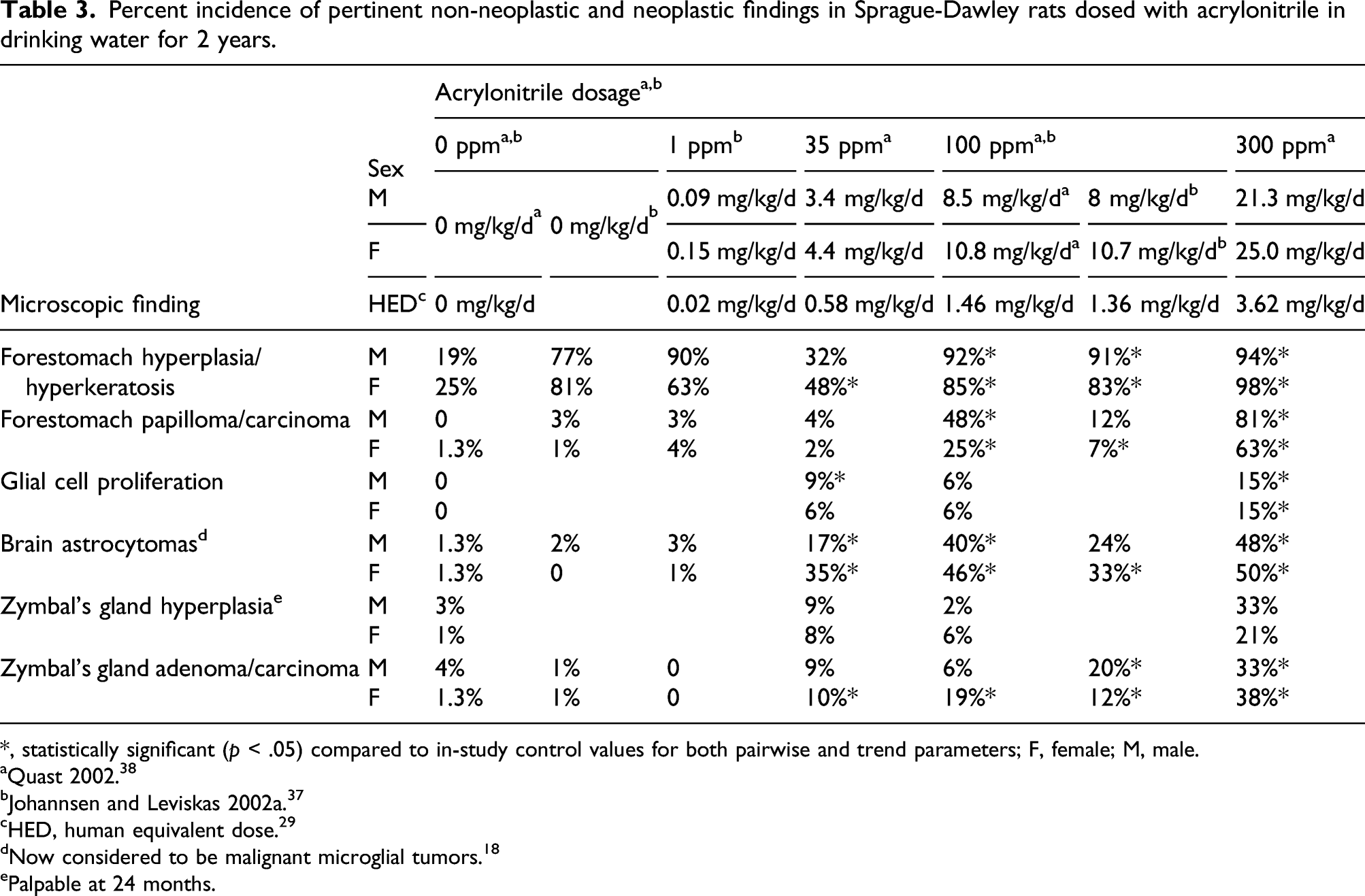
*, statistically significant (
aQuast 2002. 38
bJohannsen and Leviskas 2002a. 37
cHED, human equivalent dose. 29
dNow considered to be malignant microglial tumors. 18
ePalpable at 24 months.
Percent incidence of pertinent non-neoplastic and neoplastic findings in Fischer 344 rats dosed with acrylonitrile in drinking water for 2 years.
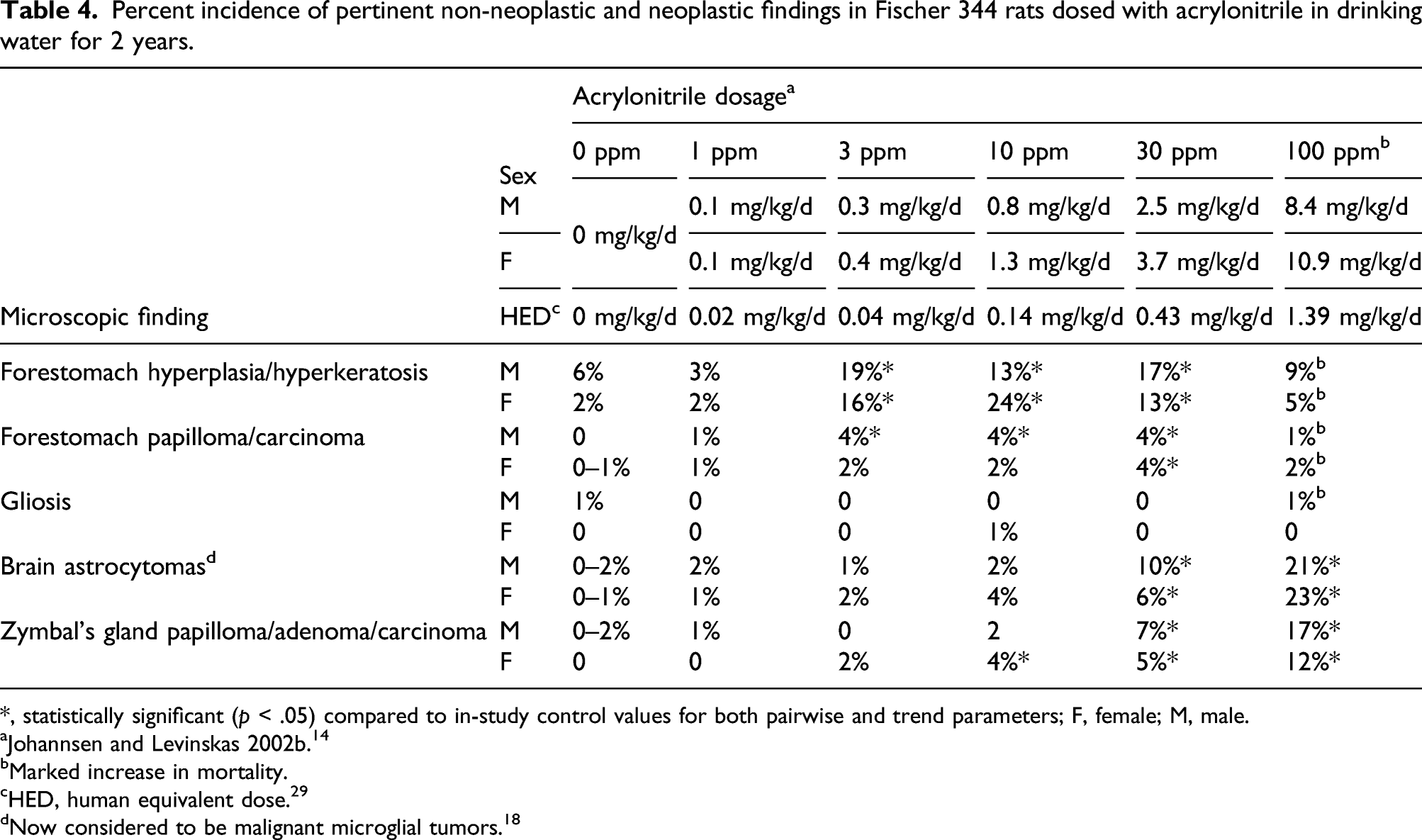
*, statistically significant (
aJohannsen and Levinskas 2002b. 14
bMarked increase in mortality.
cHED, human equivalent dose. 29
dNow considered to be malignant microglial tumors. 18
Percent incidence of pertinent neoplastic findings in Sprague-Dawley rats dosed with acrylonitrile by oral gavage a and by inhalation b for 2 years.
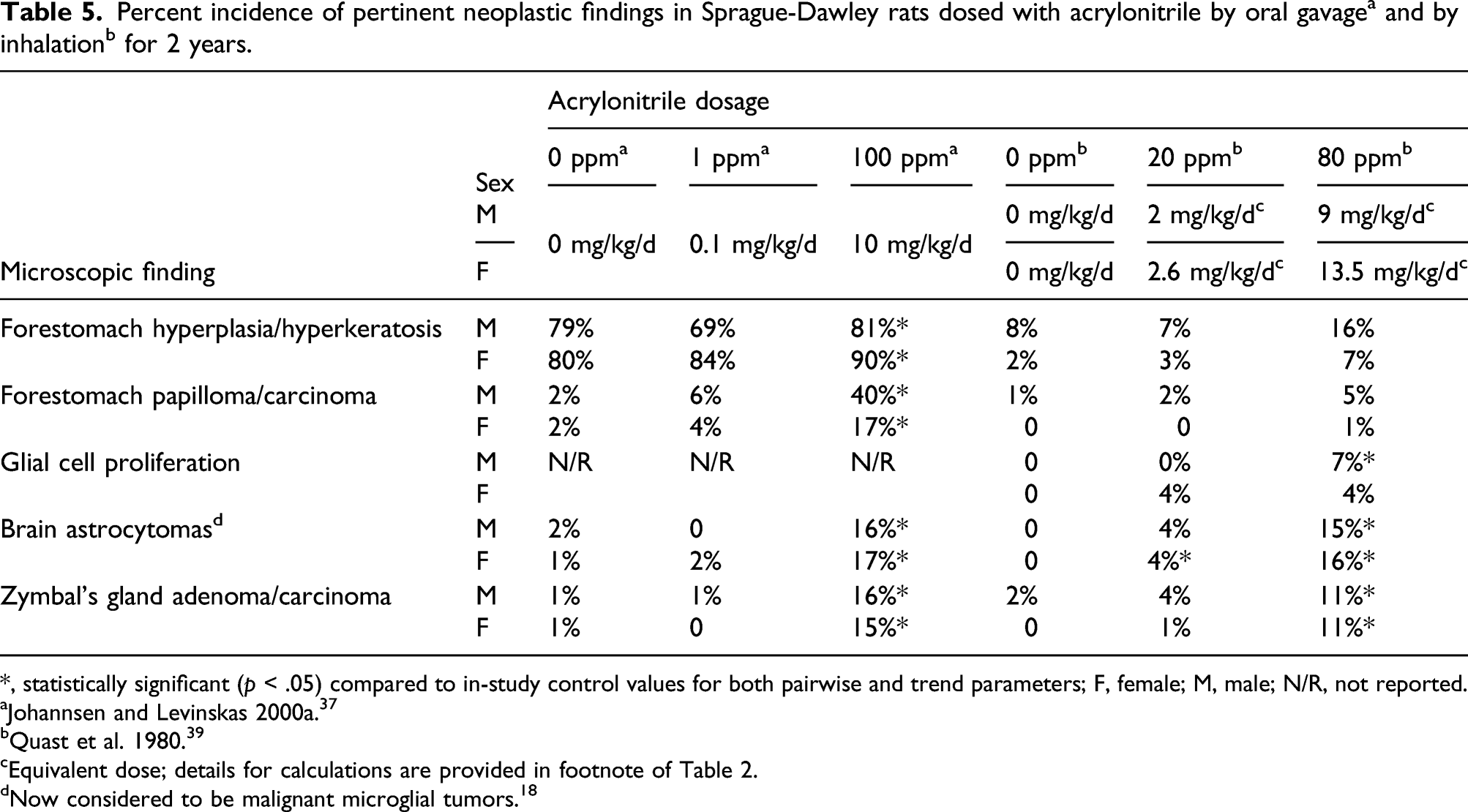
*, statistically significant (
aJohannsen and Levinskas 2000a. 37
bQuast et al. 1980. 39
cEquivalent dose; details for calculations are provided in footnote of Table 2.
dNow considered to be malignant microglial tumors. 18
Percent incidence of pertinent non-neoplastic and neoplastic findings in B6C3F1 mice dosed with acrylonitrile and delivered by oral gavage for 2 years.
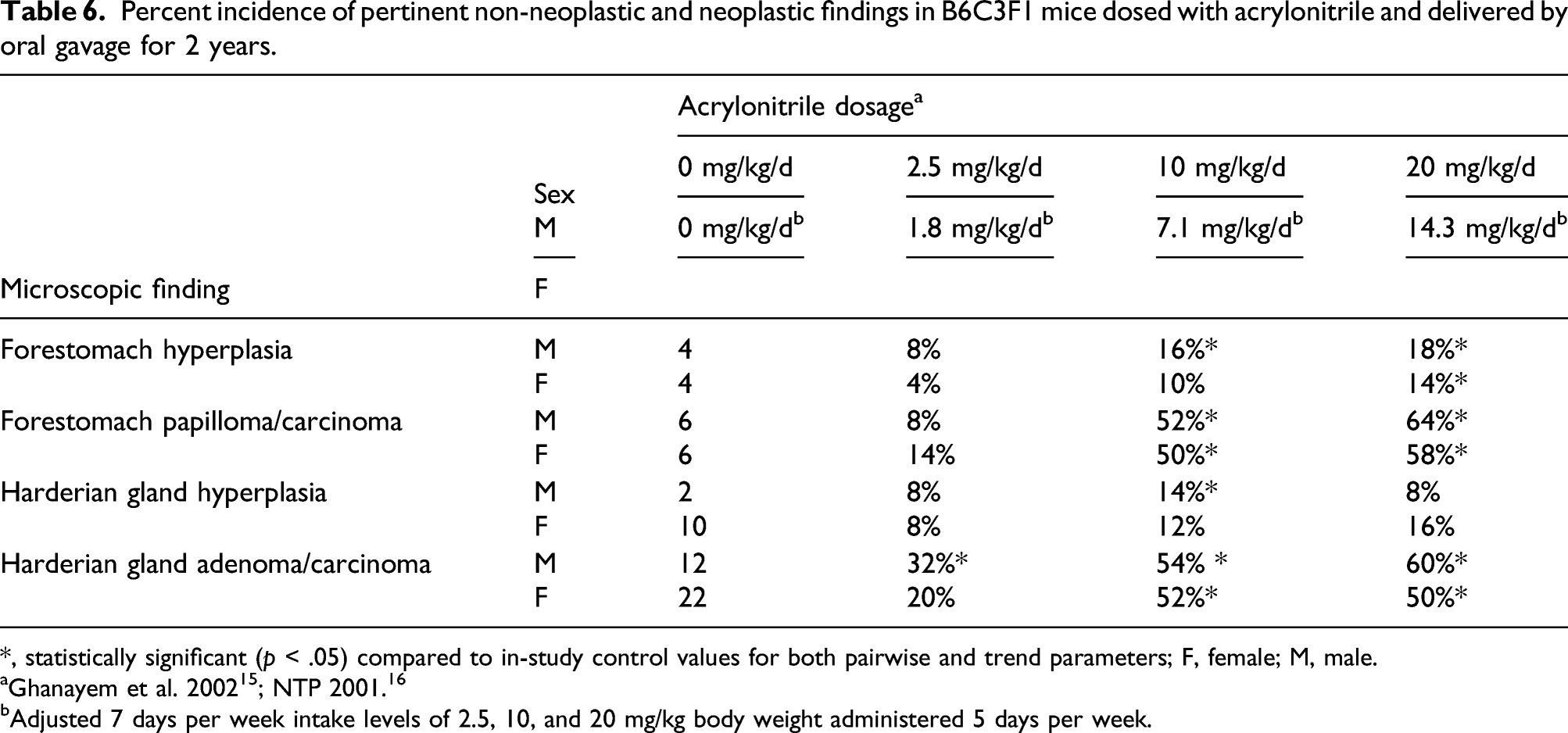
*, statistically significant (
bAdjusted 7 days per week intake levels of 2.5, 10, and 20 mg/kg body weight administered 5 days per week.
Studies in rats
Thresholds for neoplasms produced by acrylonitrile in rodent carcinogenicity bioassays.
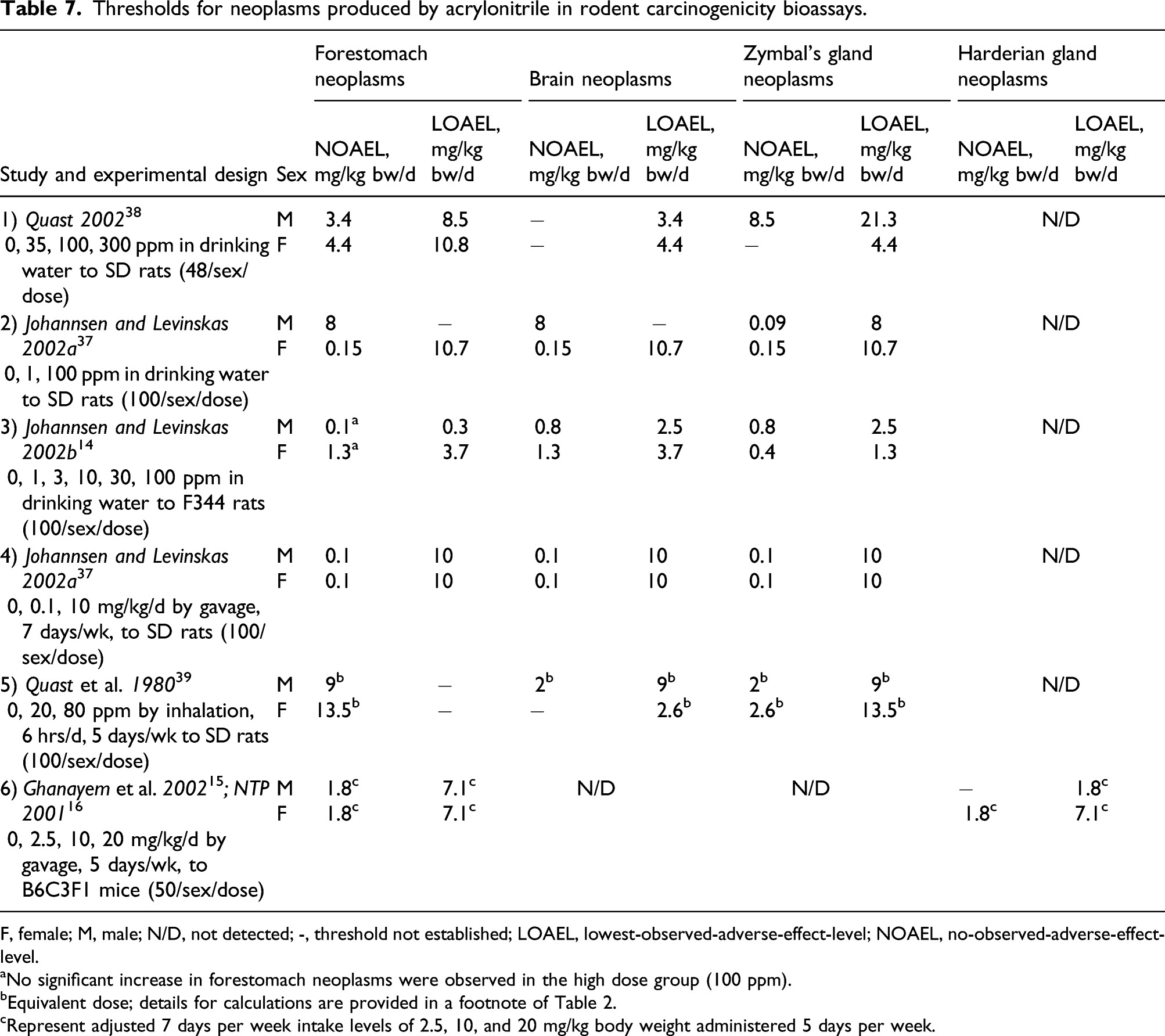
F, female; M, male; N/D, not detected; -, threshold not established; LOAEL, lowest-observed-adverse-effect-level; NOAEL, no-observed-adverse-effect-level.
aNo significant increase in forestomach neoplasms were observed in the high dose group (100 ppm).
bEquivalent dose; details for calculations are provided in a footnote of Table 2.
cRepresent adjusted 7 days per week intake levels of 2.5, 10, and 20 mg/kg body weight administered 5 days per week.
Statistically significant increases in the incidences of squamous carcinomas of Zymbal’s gland and a trend towards an increase of forestomach papillomas were also observed in SD rats that received 500 ppm of acrylonitrile in drinking water. 40 The small group size (20 rats per group) limited the power of the study. At 500 ppm, rats exhibited higher mortality rates, a trend towards a decrease in water consumption, and a slower increase in body weight gain. No tumor increases in other organs were reported.
F344 rats were more susceptible to acrylonitrile tumorigenicity compared to SD rats. Thus, acrylonitrile administered in drinking water produced a significant increase in the incidences of forestomach neoplasia in F344 males at 3 ppm and above (with exception of 100 ppm), and in F344 females at 30 ppm. 14 Brain tumors in both sexes and Zymbal’s gland tumors in males were significantly induced by 30 ppm and above, Zymbal’s gland tumors in females were induced by 10 ppm and above (Tables 4 and 7). The low survival of rats in the highest dose group (100 ppm) after 14 months of the study possibly affected statistical analyses.
In a study to investigate the nature of the brain tumors, F344 rats of both sexes were administered acrylonitrile in drinking water at 100 and 500 ppm for 12–18 months. 13 At both dose levels, acrylonitrile produced an increase in incidences of brain tumors, primarily observed in the cortex. While cells adjacent to the lesions were reactive astrocytes, negative staining of the tumors for glial fibrillary acidic protein (GFAP) microscopical, as well as ultrastructural evidence of no glial filaments, indicated that the neoplastic cells were different in origin (i.e., not astrocytes). An increase in incidences of Zymbal’s gland tumors and forestomach and subcutaneous papillomas was also observed in acrylonitrile-dosed rats. In addition to carcinogenicity, rats that received acrylonitrile exhibited signs of systemic and neurotoxicity from 12 to 18 months (i.e., until termination).
Similar to the outcomes described above for F344 rats, acrylonitrile administered by oral gavage to SD rats at 100 ppm (equivalent to male and female doses of 8 and 10.7 mg/kg of acrylonitrile in drinking water per day, respectively) produced a significant increase in tumor incidences in three target organs (forestomach, brain, and Zymbal’s gland) in rats in both sexes (Tables 5 and 7). 37
In an inhalation study of acrylonitrile in SD rats, no increase in forestomach tumor incidences was observed, while brain and Zymbal’s gland neoplasms were present in both sexes at the highest dose of 80 ppm (equivalent dose of 173.6 mg/m3 per day). 39 Brain tumors were also observed at 20 ppm in female rats (43.4 mg/m3 per day) (Tables 5 and 7). The authors also reported inflammatory changes in the respiratory epithelium of the nasal turbinates at both dose levels, with less severe changes observed at 20 ppm. These findings were considered to result from irritation produced by acrylonitrile. 39 Acute suppurative pneumonia observed in the rats administered 80 ppm of acrylonitrile was interpreted to be related to stress from acrylonitrile exposure.
In a study in SD rats, no increase in tumor incidences was observed after oral gavage with acrylonitrile at 5 mg/kg bodyweight for 52 weeks. 41 In contrast, in inhalation studies, statistically significant, but not dose-related, increases in the total number of animals with tumors were observed in rats of both sexes exposed to acrylonitrile at the dose range from 5 to 40 ppm for 52 weeks. Slight, not significant increases were observed in the incidences of “encephalic glyomas,” although, the numbers were within the historical control range. When administered at 60 ppm for 104 weeks to breeders and offspring rats, acrylonitrile produced an increase in “encephalic glyomas” (oligodendrogliomas) in all dosed groups. In addition, a significant increase in incidences of Zymbal’s gland carcinomas was observed in male offspring.
Study in mice
In a 2-year carcinogenicity study in B6C3F1 mice administered acrylonitrile by gavage, increases in the incidences of neoplasia in two target organs, forestomach and Harderian gland, were observed in both sexes at 10 and 20 mg/kg body weight per day.15,16 In males, the percent incidences of Harderian gland adenoma and carcinoma were significantly increased at all tested dose levels, from 2.5 to 20 mg/kg/day of acrylonitrile, and in females at 10 and 20 mg/kg/day (Tables 6 and 7). Noteworthy, in both sexes, over 90% of all tumors of the Harderian gland were benign adenomas. Increased incidences of combined alveolar/bronchiolar adenomas and carcinomas, as well as benign/malignant granulosa cell tumors of the ovary, were reported only in female mice from a mid-dose group (10 mg/kg/day). These findings were considered equivocal due to a lack of dose-response.
Summary of carcinogenicity findings with regard to potential MoA
The difference in the target organs of carcinogenicity between rats and mice denotes inter-rodent variations, supporting the concept that acrylonitrile-induced neoplasia is “species-specific,” with exception of the forestomach which was a common tumor site in both species with oral administration. Overall, responses were similar between the sexes, although, in a few studies, female rats were more susceptible to brain or Zymbal’s gland carcinogenicity (Table 7). Target organs of acrylonitrile carcinogenicity are the sites of spontaneous background neoplasms, 25 indicating the susceptibility of these tissues to tumor development.
Rates of “spontaneous” forestomach squamous cell carcinomas or papilloma’s range between 0.08 and 0.25% in F344 and SD rats and between 1 and 2% in B6C3F1 mice.42–44 Forestomach squamous cell neoplasia is the sixth most commonly induced neoplasm in NTP rodent carcinogenicity assays and is similar in histological appearance in all rodent species and strains.45,46 Two recognized key events involved in the development of forestomach neoplasms are DNA damage and chronic irritation. 46 Moreover, an association between early epithelial cell proliferation and hyperkeratosis and development of forestomach neoplasm was described for several carcinogens, especially following chronic administration by gavage. 47
The incidence of “spontaneous” brain glial neoplasms in rodents is low, that is, 1–2%, although, with advancing age, rats develop these neoplasms more commonly than mice.18,48 In the F344 rat, their incidence is lower than that of the SD strain.18,48 While previous studies reported that “astrocytomas” have been the most commonly identified brain tumors by conventional histopathology analyses, 49 based on immunohistochemical analyses, commonly observed background glial brain tumor in rats were primarily oligodendroglioma. 18 Staining characteristics of other, less common spontaneous tumors previously diagnosed as astrocytomas suggested origin from monocytic cells, and thus they were diagnosed as malignant microglial tumors. 18 Genotoxicity is an accepted MoA for several compounds that induce brain tumors in rodents, including ethyl-N-nitrosourea, N-methylnitrosurea, and several electrophilic epoxide or epoxide-forming compounds, including ethylene oxide and acrylamide.50,51
Spontaneous neoplasms of Zymbal’s auditory gland in rats are not common, even in old age.45,52 Thus, the historical incidences of Zymbal’s gland tumors in rodents are reported as ranging from 0.4 to 2%.42–44,53 Many Zymbal’s gland neoplasms, however, are missed because either they are not large enough at necropsy or the tissue is not on the standard protocol organ list. NTP data suggest that chemicals that produce Zymbal’s gland tumors are strongly associated with tumor development in the skin and in preputial glands in male and clitoris and mammary glands in female rats. 54 This holocrine auditory integumental accessory tissue is described in rats but is in a vestigial form in mice. To date, no key events in Zymbal’s gland neoplasia have been reported. Non-neoplastic changes observed in Zymbal’s gland, including inflammatory and degenerative changes, are histologically similar to those seen in the mammary gland. 55
The incidence of spontaneous neoplasms of the ocular orbit Harderian gland increases with age, especially in the mouse. 52 Thus, the rate of “spontaneous” carcinomas or adenomas in B6C3F1 mice is over 12% in males and 7.4% in females, while in F344 and SD rats it does not exceed 0.3%.42–44 Harderian gland neoplasms are suggested to result from genotoxicity, 52 but no direct measure of DNA damage in this gland has been reported for any chemical, and no key events are known.
Dose-response assessment of carcinogenic effects
In all six rodent bioassays, tumors were observed only after at least 12 months of acrylonitrile administration. Delayed occurrence of tumors might suggest that acrylonitrile has at most weak genotoxic activity, as suggested by ethylene oxide.51,56 Thus, the existence of thresholds for acrylonitrile tumorigenicity is plausible.57,58
Overall, in rats, the lowest-observed-adverse-effect-level (LOAEL) for neoplasia induction by acrylonitrile in target organs was at least 0.3 mg/kg/day, and the lowest NOAEL was 0.09 mg/kg (Table 7). In mice, the LOAEL was at least 1.8 mg/kg/day, and the NOAEL was below this concentration (Table 7). According to the Carcinogenic Potency Database (CPDB, https://toxnet.nlm.nih.gov/cpdb/chempages/ACRYLONITRILE.html), median tumor doses (TD50) for acrylonitrile are estimated to be 16.9 and 6.32 mg/kg per body weight per day, for rat and mouse, respectively.
BMD modeling showed that overall, the potency of acrylonitrile was consistent for different tumor types across studies with different routes of acrylonitrile administration (Figure 1). Male rats and mice in most cases were more susceptible to carcinogenic effects of acrylonitrile, compared to females. Acrylonitrile was more potent in mice compared to rats. Analyzing each tumor type separately, the lowest BMDL for the brain tumor incidence in rats of both sexes was ∼2 mg/kg body weight (Figure 1a). BMDs for forestomach tumors could not be computed in F344 rats of both sexes from the drinking water study, and in female SD rats from the inhalation study. The failure to estimate BMDs for these groups could be due to the fact that maximum tumor increase in the dosed groups was smaller than specified benchmark response of 10% (Suppl. Figures 2 and 3). Based on the remaining data, the lowest BMDLs for forestomach tumors were 1.7 mg/kg in rats and ∼1 mg/kg in mice (Figure 1b and d). The lowest BMDL for the Zymbal’s gland tumors in rats was 2.7 mg/kg, and 1.2 mg/kg for Harderian gland tumors in mice (Figure 1c and d). Comparison of benchmark doses (BMDL10) for various tumor types produced by acrylonitrile in rats (a–c) and mice (d). BMD10 is a dose estimated to produce a 10% increase in tumor incidences compared to a corresponding control group. Upper and lower bars indicate respective upper and lower bounds of two-sided 95% confidence interval for each calculated BMD10 value. BMDL, the lower limit of BMD; BMDU, the upper limit of BMD. CSF, cancer slope factor, is a plausible upper-bound estimate associated with increased cancer risk after a lifetime exposure to a chemical, presented as risk per mg/kg per day. Proposed mechanisms of action (MoA) of acrylonitrile associated neoplasia. Solid arrows indicate direct key event relationships; broken arrows indicate indirect key event relationships. CEO, 2-cyanoethyleneoxide.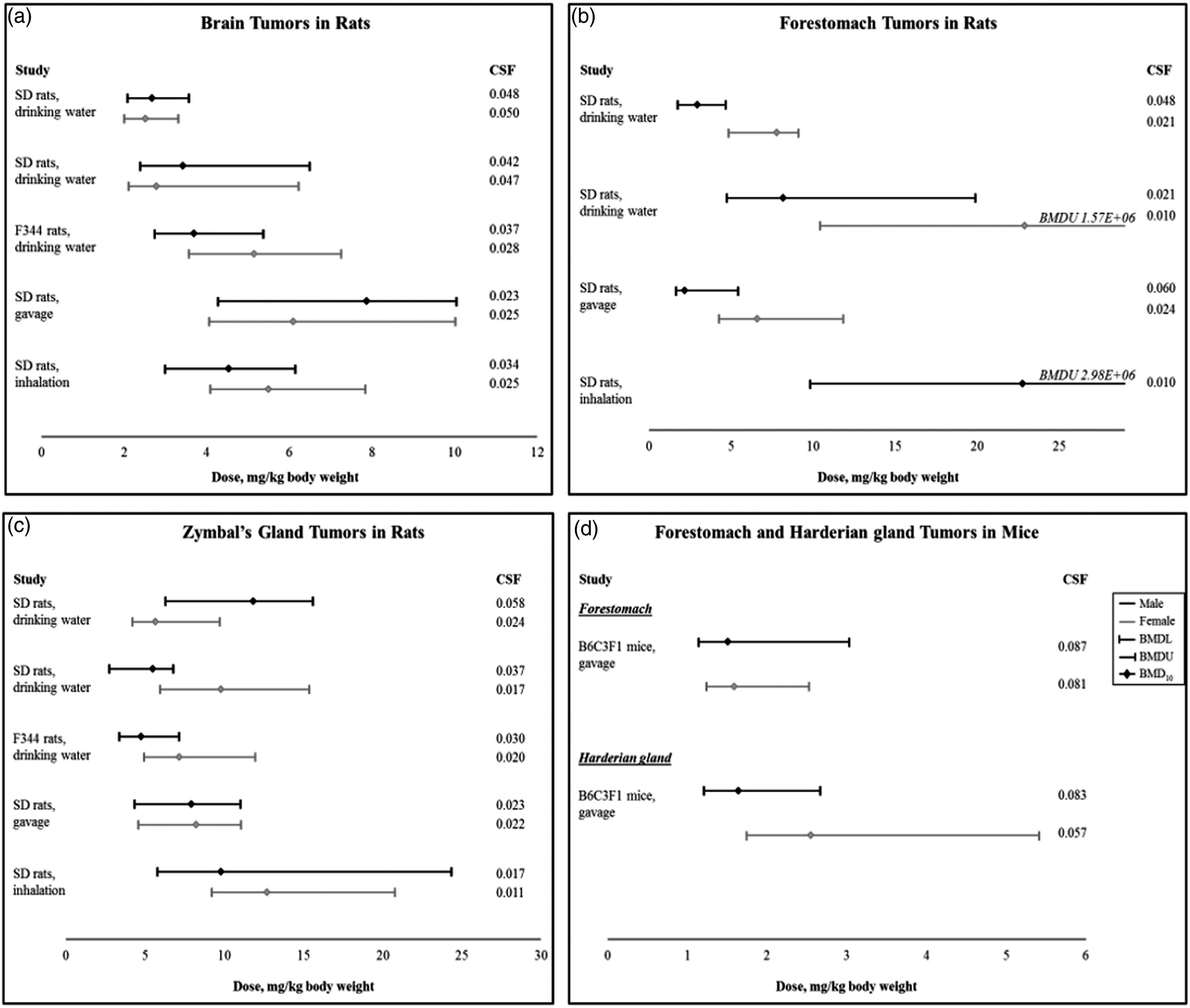
CSF for all tumor types was estimated to range from 0.01 to 0.06 per mg/kg per day based on the findings in the rat studies, and from 0.06 to 0.09 per mg/kg per day based on the study conducted in mice. An oral multi-tumor Slope Factor of 0.03 per mg/kg per day was derived from a geometric mean for all rat studies with acrylonitrile administration in drinking water. In order to estimate human risk from inhalation exposure, inhalation cancer unit risk factors (CSF expressed as μg/m3) were also calculated based on the tumor incidences in the inhalation study in rats (Quast et al. 1980) using the concentration in μg/m3 (43.4 × 10−3 and 173.6 × 10−3 μg/m3). The geometric mean for all tumors in both sexes of rats was estimated to be 1.02 × 10−6. Individual values for each tumor type and sex are provided in Supplemental Table 1.
US EPA (2006) estimated an inhalation unit risk for acrylonitrile of 6.8 × 10−5 per μg/m 3 , and that inhalation of 1 μg/m3 acrylonitrile in the air over the lifetime of an individual would result in not greater than one-in-ten thousand increased risk of developing cancer. This estimate is adjusted for smoking and continuous lifetime equivalent of occupational exposure, and thus, is different from the calculations above.
Previous cancer dose-response modeling of acrylonitrile cancer potency, based on the findings in rodent carcinogenicity, PBPK prediction for internal peak dose of CEO in the brain, and mechanistic studies, established that oral doses below 0.009 mg/kg/day and air concentrations below 0.1 mg/m3 are not expected to pose an exposure risk to humans. 59
Review of key characteristics of acrylonitrile relevant to its mechanism of action
Electrophilicity and metabolic activation
Chemical characteristics and reactivity data
Acrylonitrile (CAS: 107-13-1) is a 3-carbon alkene, with a nitrile substituent group (Figure 2). It is a direct alkylating compound, which is highly reactive due to the presence of an olefinic group (double bond between two carbons) and nitrile group (cyano radical). The olefinic group can react with many substances, such as ammonia, halogens, alcohols, and can undergo polymerization, oxidation, and hydrogenation. The nitrile group can also undergo transformation including hydrolysis, esterification, and reduction. In addition, the cyano radical can react with various nucleophiles in cyanoethylation reactions. Thus, acrylonitrile and its metabolite, CEO, have the structure of an electrophile.
Chemicokinetic characteristics
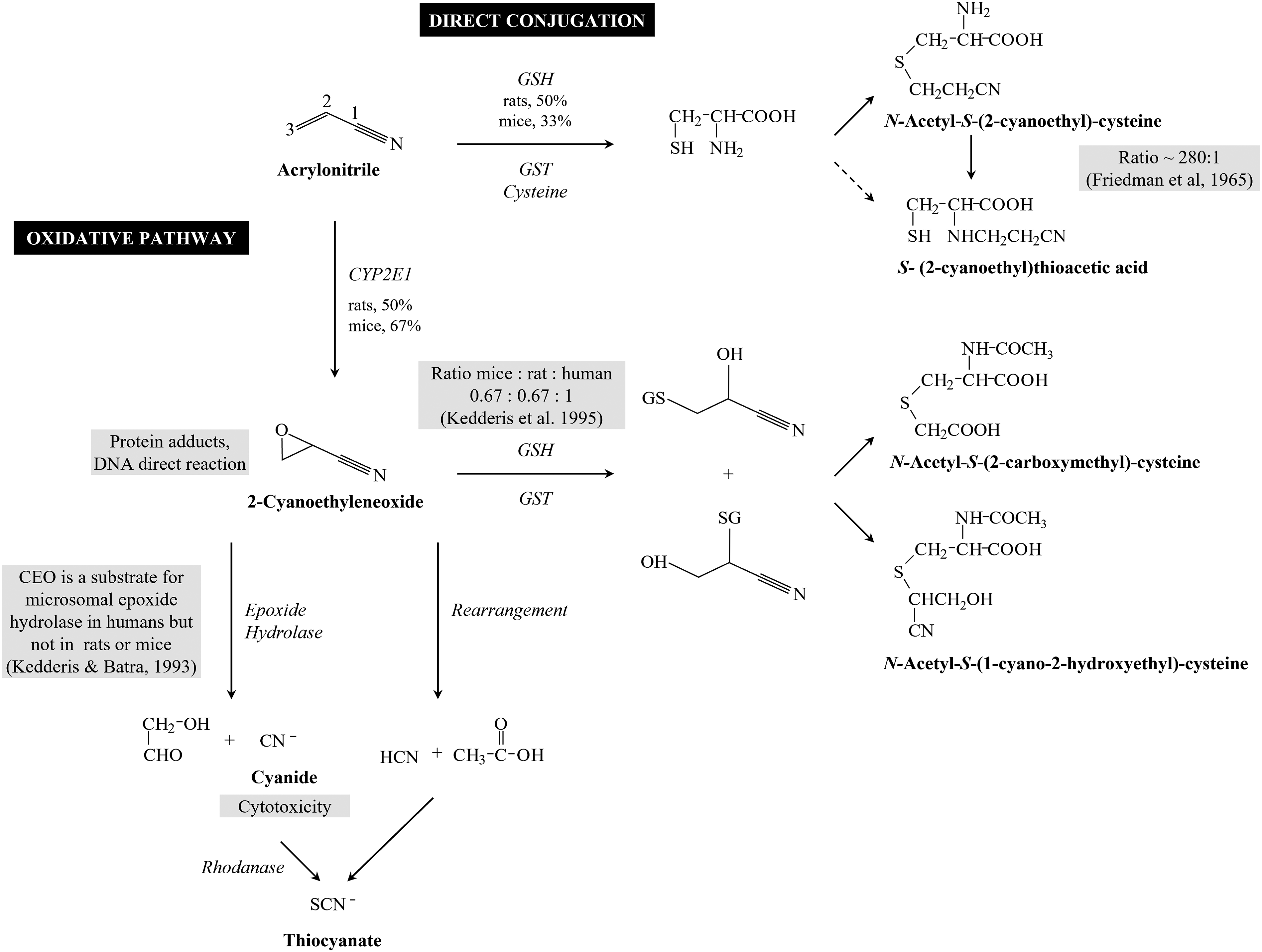
Acrylonitrile is rapidly absorbed after either enteral or parenteral administration and distributed to all tissues in rats and mice.60,61 It is mainly metabolized in the liver by two major pathways; one involves a direct conjugation with reduced glutathione, while the other entails oxidation by cytochrome P-450 to an epoxide intermediate, CEO (Figure 2).15,62,63 Acrylonitrile and CEO are chemically reactive, although both are relatively stable, with acrylonitrile having a half-life of 8 hrs, and CEO of approximately 1.5 hrs.62,63
An alternative route of acrylonitrile metabolism, epoxidation to CEO, is mediated primarily by CYP2E1 in rodents and humans (Figure 2). 70 This reaction occurs mostly in the liver, although lung tissue may also contribute to the metabolism of inhaled acrylonitrile. 71 Mice biotransform acrylonitrile in greater proportion via the CYP2E1-mediated oxidative pathway compared to rats.72–74 Thus, the rate of CEO formation in liver microsome preparations is 501 pmol/min-mg protein in humans compared to 667 in rats, and 2801 in mouse. 75 However, CEO concentrations in blood and brain of rats are higher (about 428 pmol/ml in blood and 433 pmol/g in brain) than in mice (about 380 pmol/ml in blood and 351 pmol/g in brain).61, 75
CEO can be further oxidized to cyanide (Figure 2), mostly via epoxide hydrolase or a cytochrome P-450-dependent mixed-function oxidase system in hepatic microsomes.
76
In contrast to humans, in rat and mouse liver cytosol or microsomes, epoxide hydrolase is inactive to CEO substrate.
63
In rats and mice, inhibition of CYP2E1 enzyme prevents an increase in levels of cyanide and significantly decreases the toxicity of acrylonitrile.77–79 Higher levels of oxidative metabolism of CEO in humans compared to rodents could result in different manifestations of acrylonitrile toxicity, and CYP2E1 polymorphism in humans might correlate with interindividual susceptibility to cyanide toxicity.51,80 Lactoperoxidase found in lacrimal, salivary, mammary glands, and airways was also demonstrated to oxidize acrylonitrile to cyanide
CEO is also detoxified by conjugation mediated by GST with consequent formation of mercapturic acids (Figure 2). In addition, reaction of CEO with GSH can lead to production of cyanide. 82
Rats and mice exhibit dose-dependent increase in cyanide levels following administration of acrylonitrile, with highest levels recorded in blood and liver, followed by kidney and brain. 83 Higher levels of cyanide were observed in the tissues of rats compared to those in mice after oral administration of acrylonitrile at a dose equivalent to the LD50 (46.5 mg/kg for rats and 13.5 mg/kg for mice). After 1 h of administration, respective cyanide concentrations in blood, liver, and brain were 120 nmol/ml, 110 nmol/g, and 38 nmol/g in rats compared to 92 nmol/ml, 85, and 19 nmol/g in mice. However, mice metabolized acrylonitrile to cyanide faster compared to rats. Thus, a time-course study conducted in male rats and mice administered an equivalent of 50% of the oral LD50 dose (46.5 mg/kg for rats and 13.5 mg/kg for mice) demonstrated that in rats, maximum concentration of cyanide in blood (∼110 nmol/ml) is achieved 3 hrs after administration, while in mice blood cyanide concentration is the highest (∼57 nmol/ml) at 1 h. 83
High uptake of radioactivity after oral and intravenous administration of [1-14C]-acrylonitrile was observed in various tissues, including blood, liver, kidney, lung, adrenal cortex, and stomach mucosa. 84 However, irreversible binding of radioactivity to proteins, detected after intraperitoneal administration of [2,3-14C]-acrylonitrile to rats, 85 makes it difficult to determine whether these findings are indicative of accumulation of free acrylonitrile, its metabolites or cyanoethylated proteins.
[1-14C]-acrylonitrile administered to rats by the oral, inhalation, or intravenous routes produces recoverable radioactivity mainly in the urine (about 65% after oral or inhalation) followed by carcass (about 10% after oral or inhalation), feces (about 3% after oral or inhalation), and 14CO2 (about 3% after oral, or inhalation).86,87 At least 95% of acrylonitrile is absorbed within the first 72 hrs. 87 In mice, similar data were obtained. 88 In general, radioactivity is detected in several tissues, regardless of the route of administration, dose level, or duration of exposure. The stomach, red blood cells, and skin showed the greatest levels of acrylonitrile distribution, followed by lungs, liver, kidneys, intestines, skeletal muscle, heart, spleen, brain, thymus, and testes.61,75,87,89
At 48 hrs after oral dosing, 61% of a dose of radioactive acrylonitrile is excreted by rats, with 53% in feces, 4% in urine, and 4% as exhaled CO2. With intravenous administration, elimination over 48 hrs is 21% in feces, 8% in urine, and 2% in expired air. 89 Kedderis et al. 88 estimated that 3–5% of acrylonitrile doses (0.09–28.8 mg/kg orally) are excreted in feces of rats, while 2–8% of the dose (0.09–10 mg/kg orally) is recovered from the feces of mice. Similarly, 72 hrs following the same oral doses as above, 73–99% of the radioactive dose was excreted in the urine, while 3–5% was found in feces. In mice, receiving orally the same range of doses as above, 83–94% of the dose is excreted in the urine and 2–8% in feces. Excretion is not dose-dependent in either species. Analysis of urine metabolites after oral administration of [2,3-14C]-CEO in rodents revealed that approximately 55% of the CEO metabolites are excreted in the urine in rats and 75% in mice. 88 In humans, acrylonitrile and its metabolites thiocyanate and mercapturic acid are mainly eliminated in urine. 1
Thus, chemical kinetic data reveals high bioavailability of acrylonitrile, with widespread distribution throughout the body. It is likely that the reactive metabolite CEO and the toxic metabolite cyanide form predominantly in the liver (which, however, is not a target organ for carcinogenicity), but may also have widespread distribution throughout the body in the circulation, with potential exposure of target organs. Nevertheless, CEO is cleared faster by the mouse liver, and consequently, the rat has higher blood and brain CEO levels compared to respective mouse levels. Overall, the elimination of acrylonitrile is mostly complete, without any accumulation. Lack of accumulation is supported by a study showing that 28-day dosing of rats did not affect the binding of subsequently administered radiolabeled acrylonitrile in brain. 24
Review of animal toxicity data
Acute toxicity
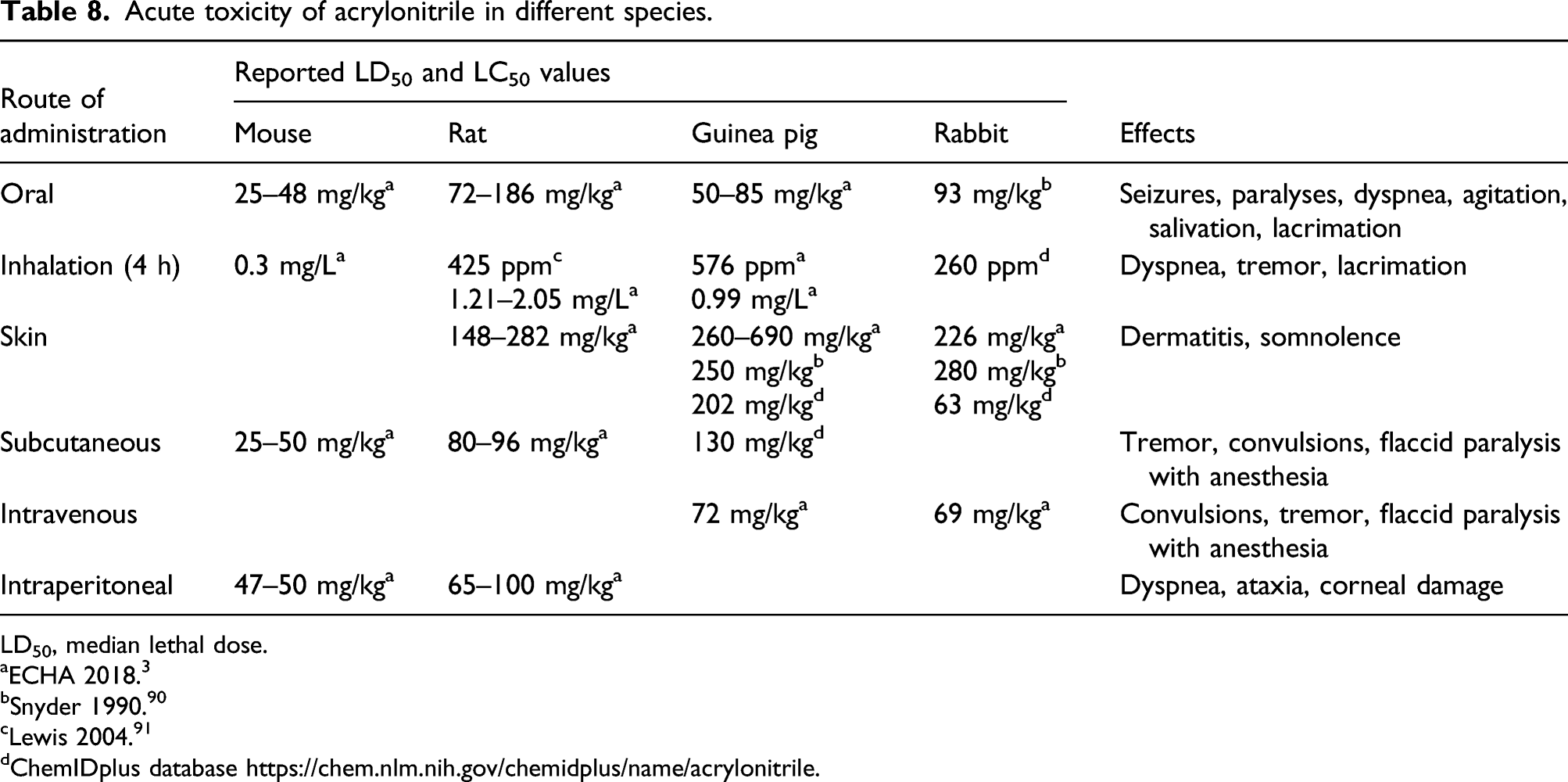
Acute toxicity of acrylonitrile in different species.
LD50, median lethal dose.
aECHA 2018. 3
bSnyder 1990. 90
cLewis 2004. 91
dChemIDplus database https://chem.nlm.nih.gov/chemidplus/name/acrylonitrile.
Signs of neurotoxicity were commonly observed in the majority of acute toxicity studies. They occurred in stages, starting with agitation and lacrimation in the excitatory phase, salivation, lacrimation, urination, and defecation during a tranquil phase, which was followed by seizures (convulsive phase), paralysis (paralytic phase), and death. 3 Edema of the brain and lungs, as well as bleeding in the gastrointestinal tract and hemorrhagic necrosis of the adrenals, were also observed. A single dose of acrylonitrile at 20, 40, or 80 mg/kg body weight administered to male Sprague-Dawley (SD) rats by gavage or subcutaneous injection also produced neurotoxic effects, cholinomimetic in nature. 92 The signs included salivation, lacrimation, miosis, vasodilation, and diarrhea. Pre-dosing with atropine protected rats from the neurotoxicity, suggesting possible involvement of the cholinergic system in acute toxicity produced by acrylonitrile.
A single oral administration of acrylonitrile by gavage at LD50 levels to male SD rats (93 mg/kg) and male Albino-Swiss mice (27 mg/kg) resulted in different symptoms of toxicity in both species. 83 Specifically, while rats developed cholinomimetic signs of toxicity (salivation, lacrimation, diarrhea, wheezing, peripheral vasodilation), mice developed CNS disturbances, such as depression, agitation, convulsion, and asphyxia.
The acute toxicity of acrylonitrile has been proposed to result from its bioactivation by cytochrome P450 enzymes leading to formation of cyanide (Figure 2).77,93 Thus, in wild-type mice (mixed 129/Sv and C57BL strains) of both sexes dosed at up to 40 mg/kg of acrylonitrile by gavage in a single dose, increase in lethality was associated with dose-dependent increase of cyanide levels in blood (in males up to 120 nmol/ml and in females up to 40 nmol/ml one hour after exposure) and brain (in males up to 60 nmol/ml and in females up to 10 nmol/ml one hour after exposure). In contrast, CYP2E1-deficient mice did not exhibit significant signs of toxicity, and cyanide levels in their blood and brain tissue were comparable to that of the control group.77,93 Ahmed and Patel 83 suggested a role for cyanide formation in acrylonitrile-produced toxicity in rodents which is species dependent. In mice, which metabolize acrylonitrile faster, cyanide formation is the major mechanism of toxicity, whereas in rats, it plays a minor role.
Others suggest that binding of acrylonitrile to cysteine residues on proteins can lead to inhibition of glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
94
Thus,
Depletion of GSH is likely to exacerbate toxicity of acrylonitrile. Thus, a single subcutaneous dose of acrylonitrile administered to male SD rats at 30 or 50 mg/kg body weight produced focal erosions of glandular stomach mucosa, which became more severe with time and were associated with hepatic and gastric glutathione depletion (25% decrease in liver and 50% decrease in glandular stomach). 98 In this study, rats which were pretreated with sulfhydryl-containing compounds, as well as atropine, were protected against necrosis and gastric erosions, suggesting that acrylonitrile alters cholinergic receptors by depleting endogenous sulfhydryl groups. A similar effect on GSH depletion was observed in male SD rats administered a single oral dose of 23 mg/kg of acrylonitrile, which decreased gastric glutathione levels by 50%, 4 hrs after administration, and 46 mg/kg of acrylonitrile further decreased the glutathione level to 27% of the control group. 99
Szabo and colleagues 100 reported that intravenous administration of a single dose of acrylonitrile at 150 mg/kg to female SD rats resulted in rapid depletion (80–90%) of glutathione in the liver, lung, and kidney in the absence of cell damage in these organs, while in a target tissue for acrylonitrile carcinogenicity, brain, GSH depletion was more gradual (approximately 30% depletion at the highest dose) but correlated with mortality. GSH levels in the liver at lower dose levels of 10 and 50 mg/kg were decreased by approximately 25% and 60%, respectively. Similar findings were obtained in the rat study with a single subcutaneous dose of acrylonitrile. 101 Thus, 20 mg/kg of acrylonitrile, a dose which did not produce toxicity in acute toxicity studies, produced a 50% decrease in GSH levels in the liver of male SD rats, while the LD10 dose of 50 mg/kg depleted GSH by up to 85%. The two highest doses, 80 mg/kg (equivalent of the LD50 dose) and 115 mg/kg (equivalent of the LD90 dose) almost entirely depleted liver GSH within 30 and 15 min, respectively.
While the brain was more resistant to GSH depletion, a dose-dependent decrease was demonstrated at doses that exhibit toxicity (50, 80, and 115 mg/kg), with 10% depletion at the highest dose. Importantly, GSH levels showed little recovery in the brain, unlike in non-target tissues, liver, and kidney. GSH depletion in the forestomach (another target organ for acrylonitrile carcinogenicity) and glandular stomach was about 40% at the highest dose and occurred at a slower rate compared to other tissues; similar to the brain, no GSH recovery was observed in these tissues. 101 The GSH depletion was accompanied by a dose-dependent increase in cyanide levels, at the highest dose reaching approximately 160 nmol/ml in the blood and over 20 nmol/g in the brain. Toxic levels of cyanide persisted in the blood and brain for 2 hours after exposure to 50 and 80 mg/kg of acrylonitrile and remained at the same level after 4 hrs from exposure to 115 mg/kg of acrylonitrile. Thus, the authors concluded that an increase in cyanide levels result from inability of the liver to detoxify acrylonitrile due to depletion of GSH (Figure 2). 101
Based on the outcomes of dermal and ocular irritation studies, in which acrylonitrile administration to rabbits at 0.5 mL (skin) or 0.1 mL (eye) produced erythema, edema, corneal opacity, and severe conjunctival irritation, acrylonitrile is classified as a skin and eye irritant.1,3 In addition, the guinea pig maximization test provided clear evidence of skin sensitization following acrylonitrile exposure at 0.2, 0.5 and 1%. 3
Subacute and subchronic repeat-dose toxicity
Repeat-dose subacute or subchronic administration of acrylonitrile in drinking water or by gavage to rats or mice often produced changes in the forestomach and adrenal gland, as well as signs of general intoxication and neurotoxicity, while inhalation exposure produced changes in the nasal mucosa of rats. Findings in repeat-dose drinking water or gavage studies in rodents were consistent with findings in studies with single-dose administration of acrylonitrile.
In female SD rats administered acrylonitrile at 0.05% and 0.2% in drinking water or by gavage for 7, 21, or 60 days, acrylonitrile toxicity was observed in the gastrointestinal tract. 102 This effect was absent at lower doses of 0.0001% (1 ppm), 0.002%, and 0.01%. Specifically, hemorrhagic necrosis and cell proliferation (hyperplasia) of mucosa at the junction between the forestomach and glandular stomach was recorded in rats that received 0.05% and 0.2% of acrylonitrile for 21 and 60 days. These concentrations also caused decreases in food and water intake, the latter possibly representing an effect of poor palatability. These effects led to decreases in body weight gain. In this study, atrophy of adrenal gland, in particular zona fasciculata, was observed in rats that received 0.05% and 0.2% acrylonitrile for 7 and 21 days, in contrast, after 60 days of dosing adrenal hyperplasia occurred and was especially prominent in groups that received acrylonitrile by gavage. Concurrent dose- and time-dependent decreases in circulating cortisone levels were also reported.
Dose-dependent increase in hyperplasia of forestomach mucosa, which was accompanied by an increase in apoptosis, was also reported in male Fischer 344 (F344) rats after daily administration of acrylonitrile by gavage at 0.22 or 0.43 mmol/kg for 6 weeks 103
Male SD rats that were administered acrylonitrile either by oral gavage at 12.5, 25, and 50 mg/kg for 5 days per week for 12 weeks or through inhalation exposure to 25, 50, and 100 ppm of acrylonitrile for 6 hrs daily, 5 days per week for 24 weeks, exhibited signs of neurotoxicity, 104 which were similar to the findings in the single-dose study by Ghanayem et al. 92 These signs, including salivation, lacrimation, miosis, and diarrhea, resembled acute acetylcholine toxicity. These effects became more pronounced with time, suggesting sensitization to acrylonitrile. In addition, a decrease in body weight gain was observed in groups dosed with 25 and 50 mg/kg of acrylonitrile in the oral study and in the high dose group (100 ppm) in the inhalation study. 104
A few general toxic effects were reported in a 90-day study in SD rats administered acrylonitrile in drinking water at 35, 85, 210, and 500 ppm. 105 These included decreased water consumption accompanied by decreased body weight gain and an increase in relative liver weight at 210 and 500 ppm groups, in rats of both sexes. A no-adverse-effect level (NOAEL) of 85 ppm (8–10 mg/kg body weight) was established in this study.
Daily administration of up to 60 mg/kg body weight of acrylonitrile to B6C3F1 mice by gavage 5 days per week for 14 weeks resulted in high mortality in both sexes. At 40 mg/kg/day, survival and mean body weight gain of female mice were significantly higher compared to that of males, suggesting that male mice are more sensitive to acrylonitrile toxicity than females. 16 In female mice, acrylonitrile at 40 mg/kg/day produced mild chronic inflammation and hyperplastic changes in the forestomach, as well as mild hematologic effects (lymphocytopenia and hemolytic anemia), which, with exception of hemolytic anemia, also occurred in males administered 20 mg/kg/day. Hematologic findings were suggested to be a result of cyanide formation or binding of acrylonitrile to hemoglobin. 16 The dosage of 5 mg/kg/day was considered to be a NOAEL. Another 13-week study with acrylonitrile administered to B6C3F1 mice by gavage 5 days per week yielded a NOAEL of 12 mg/kg/day (the highest tested dose in the study), since no dose-related effects were observed. 3
A decrease in body weight gain, signs of neurotoxicity (hyperexcitability, excessive salivation), as well as thickening of the non-glandular stomach (forestomach) were observed in pregnant SD rats, which received acrylonitrile at 65 mg/kg body weight per day by oral gavage from day 6 to day 15 of gestation. 106 In contrast, no forestomach thickening was reported in pregnant rats after inhalation with up to 80 ppm of acrylonitrile for the same duration (equivalent to 23 mg/kg body weight per day of acrylonitrile by gavage).
In studies with acrylonitrile administered to SD rats by inhalation at concentrations of 5, 15, and 45 ppm (two-generation study) and 90 ppm (one-generation study) 6 hrs/day, 5 days/week, signs of systemic toxicity were observed in adult rats in two generations at 45 and 90 ppm in both sexes. 107 The NOAEL for parental systemic toxicity was established as 15 ppm. In addition, acrylonitrile-related microscopic nasal lesions predominantly in rostral epithelium, including respiratory/transitional epithelial hyperplasia, subacute inflammation, squamous metaplasia, and/or degeneration of the olfactory epithelium, were observed in some animals at 5 and 45 ppm of acrylonitrile.
In a three-generation study with acrylonitrile administered in drinking water, the parental generation of SD rats of both sexes that received 100 and 500 ppm of acrylonitrile (corresponding to approximately 11 and 37 mg/kg, respectively, for males and 20 and 40 mg/kg per day for the females, respectively) exhibited decreased water intake and, at 500 ppm, food intake, as well as a decrease in body weight gain compared to the control groups.
108
The F1 offspring mortality was increased at 500 ppm. No signs of neurotoxicity were observed in the study. Increases in the incidences of astrocytoma and Zymbal’s gland tumors were observed in adult females in each generation after almost a year of dosing. Tumor rates in F2 generation were lower compared to F1 and comparable to F0, suggesting minimal
Genotoxicity and binding to macromolecules
DNA-reactivity and mutagenicity in the target tissues represent a key MoA for a certain type of chemical carcinogen referred to as DNA-reactive or genotoxic.109,110 Thus, an important question in assessing the MoA of acrylonitrile is whether its electrophilic structure can lead to covalent binding to DNA, resulting in genotoxicity (Table 1).1,17,31
Binding to macromolecules
Some data suggest that acrylonitrile can form DNA adducts in several organs (e.g., brain, liver), but evidence of covalent binding to DNA is uncertain. 1 Alkylation of DNA was mainly due to DNA binding of the reactive metabolite CEO which is faster compared to acrylonitrile.111, 112
Irreversible binding of 2,3-14C-2-acrylonitrile and CEO to calf thymus DNA and microsomal protein was demonstrated
In studies by Farooqui and Ahmed,117,118 DNA adduct levels were almost 10 times higher in the brain (56 in 106 normal nucleotides) than in the liver. DNA adduct levels were relatively stable increasing over the first 24 hrs and remained approximately the same at 48 hrs, the last time point studied. The authors commented that the radioactivity was found in peaks “corresponding to standard DNA nucleosides deoxyadenosine and deoxyguanosine,” which would be consistent with metabolic incorporation of 14C rather than DNA binding. High levels of DNA adducts in the brain of male F344 rats were also detected after single administration of 4 mg/kg of [2,3-14C] acrylonitrile either orally or by inhalation to rats with depleted glutathione.66,119 However, when samples were digested with RNase, the residual radioactivity was at or below the detection limit of about 3 adducts per 106 normal nucleotides.
The conflicting evidence for DNA adduct formation in the brain was reviewed by Whysner et al.116,120 with the conclusion that reported “DNA adducts” were most likely a result of contamination by acrylonitrile bound to residual protein.
Abdel-Rahman and coauthors 121 reported very rapid DNA binding of 2,3-14C-acrylonitrile in gastric glandular tissues after a single oral dose, with adduct levels in the order of 1 adduct in 103 nucleotides, which, however, declined after 15 min. Such high levels of adducts are unusual for any chemical. The authors stated that DNA was free from protein based on 260/280 ratios, which, however, is very insensitive for protein contamination of nucleic acids. If this radioactivity represented DNA adducts, the vast majority are rapidly repaired. Similar results were obtained by the authors in the lungs 122 and testes, 123 which were not target tissues of neoplasia in rodents. In these studies, no attempt was made to investigate metabolic incorporation of 14C into the normal DNA nucleotides. Radioactivity from 2,3-14C-acrylonitrile was incorporated into normal RNA bases in the liver of Wistar rats with higher incorporation at 72 hrs compared to 24 hrs. In contrast, little or no radioactivity was incorporated into DNA nucleotides and the amount of adducts was too low to allow for identification, however, the authors presumed that acrylonitrile, either itself or its metabolites, can alkylate nucleic acids.124,125 Regardless, such DNA-binding was much lower when compared to vinyl halides.
Williams et al. 24 assessed acrylonitrile-DNA adduct formation in SD and F344 rat brain tissues after a single oral gavage dose of 11 mg/kg body weight 14C-acrylonitrile, which in some groups was preceded by administration of acrylonitrile in drinking water at 100 ppm for 27 days. Binding of 2,3-14C-acrylonitrile to purified DNA was measured using accelerator mass spectrometry, which is orders of magnitude more sensitive than liquid scintillation counting. 126 Association of 14C-acrylonitrile with DNA in the brain was detected, but possible binding represented less than 15 adducts per 108 nucleotides, which is considered to be of questionable biological significance. 58 Moreover, despite extensive purification, protein contamination of DNA was considered still possible. 24 The absence of DNA adducts in acrylonitrile-dosed groups was confirmed with 32P-nucleotide post labeling, which can detect one adduct in 1010 nucleotides. 127
Evidence exists that binding of acrylonitrile to protein is much higher than to nucleic acids and that thiols are a major target, with GSH playing a strong protective role.
128
The reaction does not appear to be enhanced by metabolic activation.
129
Both acrylonitrile and CEO react rapidly with GSH under physiological conditions (e.g., the temperature of 37°C) due to high reactivity of thiol groups.63,64 However, CEO reacts with GSH less rapidly compared to acrylonitrile. Thus,
A protein site identified as particularly reactive was the cysteine 86 moiety of rat GST. 67 The levels of protein binding in the brain, kidney, liver, and stomach of male SD rats were about 1 adduct per 103 amino acids after a single oral administration of 2,3-14C-acrylonitrile.117,118 Similar levels were found in RNA from the brain, liver, and stomach. Hemoglobin N-terminal valine adducts of acrylonitrile, while constituting only a small percentage of total hemoglobin binding, have been proposed as a marker of both active 131 and passive 132 smoking or accidental exposure to acrylonitrile. 133 Benz et al. 128 reported that in rats after subcutaneous administration of 2,3-14C-acrylonitrile, N-(2-cyanoethyl)valin adduct formation, which represented only 0.2% of total acrylonitrile globin adducts, had a linear function at dose levels between 0.02 to 0.95 mmol/kg. In contrast, at higher doses (above 1 mmol/kg), which are known to produce toxicity, a sharp decrease in hemoglobin binding was reported. Similarly, a dose-related increase in N-(2-cyanoethyl)valin hemoglobin adducts was reported in male F344 rats exposed to several concentrations of acrylonitrile in drinking water ranging from 3 to 300 ppm for 105 days. 134 At dosages above 10 ppm, levels of adducts increased sublinearly, indicating saturation of metabolic pathway and elimination of acrylonitrile. In addition, these globin adducts were also detected in smokers consuming 10 to 20 cigarettes per day, whereas in non-smokers they were below detection limit. 134
In vitro genotoxicity
Summary of findings for acrylonitrile in short term
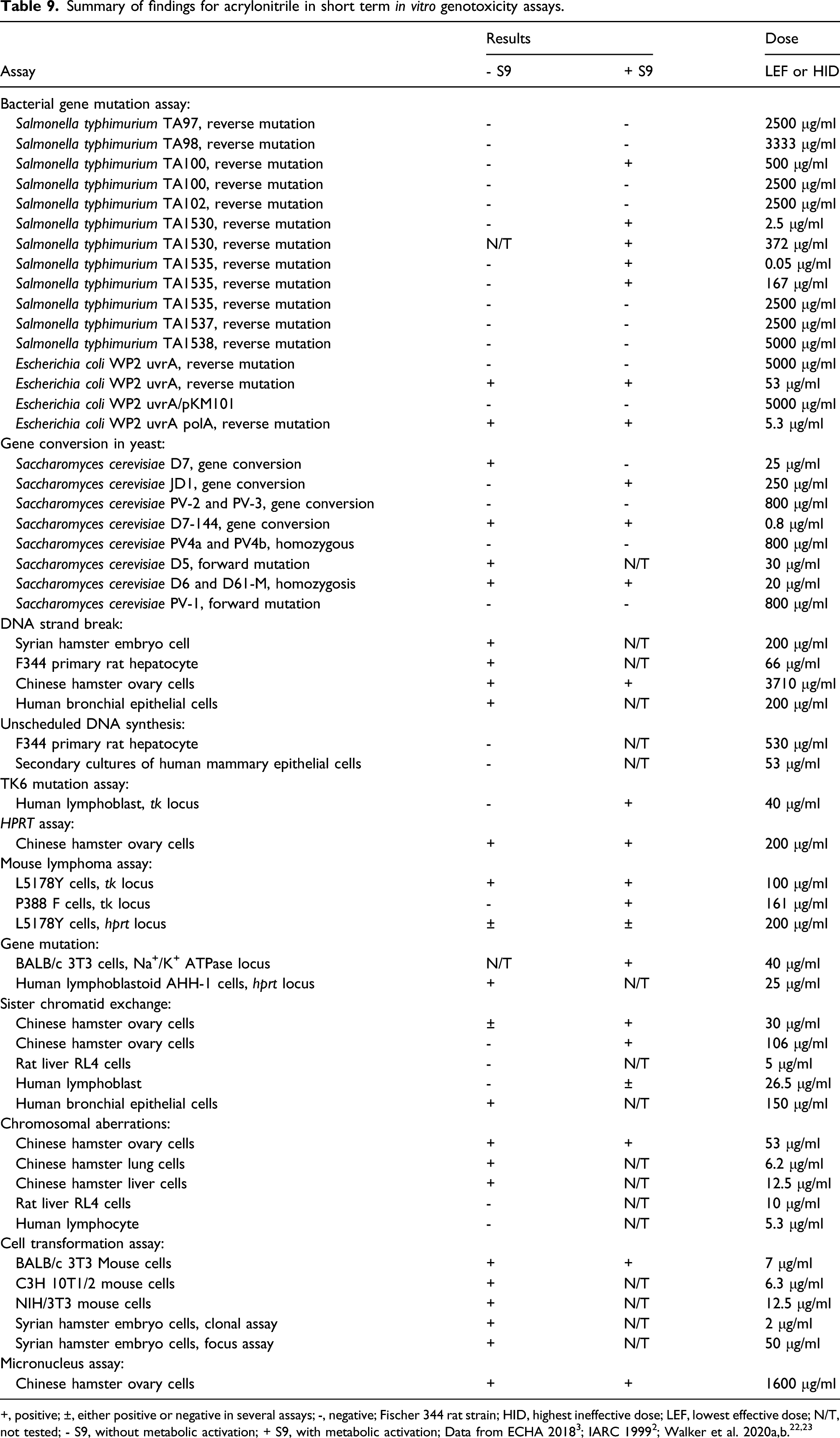
+, positive; ±, either positive or negative in several assays; -, negative; Fischer 344 rat strain; HID, highest ineffective dose; LEF, lowest effective dose; N/T, not tested; - S9, without metabolic activation; + S9, with metabolic activation; Data from ECHA 2018 3 ; IARC 1999 2 ; Walker et al. 2020a,b.22,23
In the majority of the assays, positive responses were observed only at very high dose levels (3 or 4 times higher than the LOAELs) of acrylonitrile which were associated with cytotoxic effects, indicating its weak mutagenic potential. The induction or enhancement of genotoxicity after metabolic activation, suggests that conversion to CEO may be responsible for the
In addition, it is possible that oxidative stress could contribute to positive results in some of the assays. Thus, L5178Y cells utilized by the mouse lymphoma assay are known to be sensitive to oxidative damage due to decreased antioxidant defenses. 137
In vivo genotoxicity
Summary of findings for acrylonitrile in short term

+, positive; ±, either positive or negative in several assays; -, negative; HID, highest ineffective dose; Fischer 344 rat strain; LEF, lowest effective dose; SD, Sprague-Dawley rat strain; Data from ECHA 2018 3 ; IARC 1999 2 ; Walker et al. 2020a,b.22,23
The lack of in vivo genotoxicity can be explained by differences in metabolic activation of acrylonitrile and detoxication/elimination capacities of live animals compared to
Mutagenic effects evident by dose-related elevations in
No direct production of DNA strand breaks was detected in the target tissues, brain, and Zymbal’s gland, of SD and F344 rats and Harderian gland of wild-type B6C3F1 or CYP2E-1-null mice using a conventional comet assay.23,24 Positive comet assay results were reported in forestomach and liver in wild-type B6C3F1 but not CYP2E-1-null mice, after administration of acrylonitrile at 20 mg/kg by gavage 5 days/week for 6 weeks, indicating the significance of the oxidative pathway in producing DNA damage by acrylonitrile. 23 The authors noted that the nature of DNA damage detected by comet assay was difficult to determine, since the assay does not differentiate between single-strand breaks and excision-repair and alkali-labile sites (including those formed by repair of N-7-guanine DNA adducts).
Toxicity of acrylonitrile can also affect the outcomes of genotoxicity testing. Thus, while acrylonitrile produced DNA-strand breaks in the livers of male SD rats, no increase in DNA damage was detected in the glandular stomach (non-target tissue), where mild histopathological changes were found compared to remarkable necrosis of squamous cells and ulceration that were observed in forestomach (target tissue) at 62.5 mg/kg administered orally as 3 doses. 138
Non-genotoxic mechanisms: induction of oxidative stress and inflammation
Oxidative stress has been implicated as a key mechanism of action of carcinogens.3,139–142 However, only a few reports focus on the effect of oxidative damage specifically on microglial cell,19–21 which recently were identified as cells of origin for acrylonitrile-induced tumors. 18 In contrast to astrocytes, microglia, which is a counterpart to macrophages, can proliferate, migrate, and change functional status depending upon the location in the CNS.143,144 Thus, response of microglia to oxidative stress or other effects of acrylonitrile, CEO, and cyanide should be investigated further.
Increased levels of reactive oxygen species and 8-deoxyoxoguanine adducts in rat glial DITNC1 cell line (astrocytes), but not in primary rat hepatocytes after incubation with acrylonitrile at v 2.5, 5, and 10 m
In the study by Jacob and Ahmed 148 incubation of normal human astrocytes with up to 50 μM of acrylonitrile for 12 hrs did not produce any alterations. However, at higher dosages, 200 and 400 μM, acrylonitrile significantly affected viability of these cells, decreasing it up to 60%, with concomitant redox imbalance (depletion of GSH levels up to 35% lower than respective control) and increase in the formation of reactive species.
In primary glial cells of rats, induction of oxidative damage was also accompanied by significant depletion of GSH (93% decrease compared to the respective control) and enhancement of lipid peroxidation after incubation with 1.0 m
Incubation of cultured rat astrocytes for up to 14 weeks with 0.25 m
Caito et al.
19
compared the responses of rat astrocytes and microglia to acrylonitrile dosing
Evidence of oxidative DNA damage in rodents was mainly observed in a target tissue for carcinogenicity, brain, while in other target organs, Zymbal’s gland, Harderian gland, and in cells from a non-target organ, liver, it was not detected.23,24,145 These findings can be attributed to the lower antioxidant capacity of rat brain compared to the liver. Difference in expression of GST isoforms between the brain and other tissues149,150 might also play a role in the formation of oxidative stress predominantly in the brain. Such selectivity can indicate the importance of oxidative damage in the development of brain neoplasia, while in other target organs other mechanisms may be involved. For example, damage produced by acrylonitrile in the forestomach was suggested to involve activation of neutrophils and an increase in myeloperoxidase activity, which subsequently stimulated acrylonitrile biotransformation to cyanide. 151 Also, depletion of GSH in combination with release of tissue-damaging radicals from neutrophils can exacerbate tissue damage.
In addition to cytotoxicity, the accumulation of reactive oxygen species can induce an inflammatory response in both astrocytes and microglia, activating expression of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and p53 transcription factors in primary microglia and astrocytes exposed to 0.1, 0.5, or 1 m
Primary glial cells isolated from cortical tissue of neonatal C57BL/6J mice were resistant to acrylonitrile-induced oxidative stress, which could contribute to species-specific response to chronic acrylonitrile administration. 21
Mice were more resistant to chronic administration of acrylonitrile, showing a decrease in survival only at dose levels close to the LD50. Moreover, the brain glial tumors observed in rats of both sexes, were absent in mice, which were administered higher (up to 3 times) doses of acrylonitrile. This could be explained by inter-species differences in dynamic properties of the organ. Glial cells in mice were more resistant than in rats to oxidative damage produced by acrylonitrile, possibly since mice clear CEO faster,72–74 thereby reducing CEO concentration in the brain by half.61,75 Moreover, in rats, CEO concentration in the brain was almost double that of blood. This may explain, in part, the lack of brain neoplasia in mice, which in general are less susceptible to brain neoplasia. 48
In cell suspensions prepared from the spleens of C57/Bl6 female mice, acrylonitrile inhibited proliferation of T- and B-lymphocytes, with a greater effect on the B-cells, while not affecting their viability. 153 According to the authors this might create a favorable condition for carcinogenicity. In an earlier study by Hamada et al. 154 acrylonitrile also produced an immunosuppressive effect in CD-1 mice, decreasing the number of IgA producing cells in the intestines and a significant depletion of spleen lymphocyte population, which could be a contributing factor to the carcinogenicity of acrylonitrile in the forestomach.
Some epigenetic effects could also contribute to acrylonitrile carcinogenicity.
8
Thus, in rat DITNC1 cell line, acrylonitrile produced a dose-dependent inhibition of gap junctional intercellular communication in following 2-hour treatment with 0.10 mmol/L and 1 mmol/L, which was absent in primary cultured rat hepatocytes.
155
This effect was alleviated by co-incubation of astrocytes with vitamin E or 2-oxothiazolidine-4-carboxylic acid, which is a precursor to glutathione synthesis. Li and colleagues
156
using Jurkat cell lines reported that acrylonitrile can produce immunotoxicity by damaging separation of lipid raft, structures of cell membrane present in leukocytes which are involved in cell signal transduction and restraining Ras-Raf-MAPK-ERK signaling pathways. While this evidence is compelling, data supporting an epigenetic MoA, in particular
Discussion
Suggested Mechanisms of Acrylonitrile Carcinogenicity
The reviewed information available in the literature supports the conclusion that acrylonitrile is a highly toxic carcinogen with a complex MoA for tumorigenicity which may involve multiple mechanisms. The biological significance of DNA binding of acrylonitrile for the carcinogenic MoA of the compound is questionable, since, in contrast to
Regarding brain and forestomach tumorigenicity, neurotoxicity and ulcers of forestomach mucosa were commonly observed effects of acute and subchronic toxicity of acrylonitrile, associated with depletion of glutathione. Moreover, oxidation of acrylonitrile can lead to production of a reactive metabolite CEO, and a cytotoxic metabolite cyanide, both of which were associated with adverse effects found in various studies. In addition, acrylonitrile, cyanide, and CEO can penetrate the blood-brain barrier, accumulating in the target tissue.
The four target sites of acrylonitrile carcinogenicity in the six bioassays were in tissues, which are located at the interface of different microenvironments and provide a barrier protection from injury or stress. The absence of response of other barrier tissues (e.g., lung) could reflect chemical kinetic (e.g., different metabolism of acrylonitrile) and dynamic factors and inherent tumor susceptibility. All of the target tissues are composed of parenchymal and non-parenchymal cellular elements. The predominant cellular component of the induced brain neoplasia was a non-parenchymal component (microglia), whereas in the other three tissues the predominant cellular component was parenchymal. When the integrity of a tissue is compromised, for example, injury to the forestomach wall, the parenchymal cellular elements respond predominantly with regenerative compensatory proliferation to correct the tissue perturbation. The integument-associated tissues undergo similar processes, which protect the auditory (Zymbal’s gland) or ocular orbital (Harderian gland) regions. In the blood-brain barrier tissue (region), the microglia, which is a non-parenchymal cellular component derived from macrophage precursor, plays a predominant role in response to stress and injury to neural cells produced by neurotoxins.143,144 Unlike astrocytes, which previously were considered to be the target cells of acrylonitrile carcinogenicity, 18 microglia cells can readily undergo proliferation in response to pro-inflammatory signaling or, in case of depletion, recolonization.144,157 Microglial cells activated in response to neurotoxins by classical M1 activation, release more pro-inflammatory mediators and reactive oxygen species, exacerbating tissue damage.157,158
Hyperplastic changes in the Harderian gland were observed in mice after administration of various chemicals.159–161 Since this gland is also considered to be a site of immune response, among other functions, 162 it is possible that cell proliferation observed in the gland could be a result of immune response activation.
As evident from available extensive chemokinetic data, the rodent target organs of acrylonitrile carcinogenicity were exposed to acrylonitrile and CEO in a dose-proportional manner. However, the mouse eliminated acrylonitrile and CEO faster, resulting in levels of CEO which were higher in rat blood and brain compared to respective mouse levels. This could contribute to the lack of brain tumors in mice.
Tissue- and species-specific induction of oxidative stress and subsequent inflammation is a likely MoA for brain tumor development in rats, while in the forestomach, direct cytotoxicity of acrylonitrile and its metabolite cyanide together with immunosuppression might be more relevant (Figure 3). For the Zymbal’s and Harderian glands, mechanistic studies investigating a potential acrylonitrile MoA are lacking, although absence of direct and oxidative DNA damage in the Zymbal’s gland after acrylonitrile administration was reported. 24 Sustained stress and cytotoxicity produced by acrylonitrile possibly can be involved in carcinogenicity in these two tissues.
Depletion of GSH after prolonged exposure to a high concentration of acrylonitrile is a potential initiating event in acrylonitrile MoA (Figure 3). Decrease of the detoxication route and the presence of an oxidation route of acrylonitrile metabolism permits increased formation of two metabolites, reactive CEO, as well as cytotoxic cyanide. This could lead to accumulation of oxygen-derived free radicals and mitochondrial damage leading to ATP depletion, as shown with long-term exposure of rat astrocytes and glial cells to acrylonitrile.95,96
Also, oxidative stress is exacerbated by depletion of GSH, which has antioxidant properties. Cytotoxicity and increased oxidative stress may contribute to tissue injury, particularly in the brain, which lacks a lymphatic vascular system. Production of pro-inflammatory mediators induces inflammatory conditions, which can lead to further cell and tissue injury and death or apoptosis of contiguous cells. Subsequently, local tissue immunocytes (e.g., microglia and other histiocytes) are recruited. The extent of immunocyte recruitment differs from site to site. In the case of microglia, immunocyte involvement is significant. In response to the injury, cells undergo compensatory regenerative cell proliferation (hyperplasia), which was observed in all target organs of carcinogenicity with chronic dosing (Tables 3–7). Such prolonged administration of an injurious agent leads to cell proliferation, and in turn, proliferating cells can transform and progress to neoplasia, as reported with numerous other chemical carcinogens. 163 To support this hypothesis, time course studies are needed.
It is possible that in addition to direct and indirect cytotoxicity of acrylonitrile and oxidative stress, neoplastic conversion of the target cells can develop through the induction of cell proliferation signals leading to increased DNA synthesis, in combination with inhibition of an apoptosis signal, as reported by Pu et al. 152 Again, mechanistic studies are required to support this hypothesis.
A direct local injurious effect of acrylonitrile dosing may occur in the forestomach barrier site (the portal of entry of acrylonitrile in five of the six bioassays), where neoplasia developed in both rodent species of both sexes in a similar dose and time-related fashion. Injury was evident by formation of erosions of the forestomach as well as hyperplasia/hyperkeratosis of forestomach and glandular stomach mucosa, which were also observed after a shorter duration of administration of acrylonitrile. In the other systemically accessible sites, the species differences in the permeability of the tissue and the speed of acrylonitrile clearance may have influenced development of the neoplastic response, that is, the absence of brain and Zymbal’s gland tumors in mice and Harderian gland tumors in rats.
The combination of the different sustained stresses, which occurred across species, strains, and sexes, was exacerbated in some studies by a significant chronic reduction in water intake. This was pronounced in all drinking water studies in rats (Table 2), likely resulting from poor palatability. Reduced food consumption and consequent body weight gain reduction in a dose and time-dependent fashion contributed to compromised survival. It is possible that water-electrolyte imbalance induced by hypohydration and malnutrition could have a co-carcinogenic effect, although, specific evidence supporting this hypothesis is lacking. In the oral gavage and inhalation studies, water intake was increased, indicating better tolerability. Nevertheless, the authors reported that acrylonitrile produced kidney toxicity, which could have exacerbated water-electrolyte imbalance.
The above suggested MoAs are based on extensive data, but a definitive MoA for acrylonitrile carcinogenicity in the four primary target tissues has not been established. For Zymbal’s and Harderian glands, evidence to conduct a WoE evaluation is limited or missing. The postulated MoAs should be further investigated by short-term
Weight of evidence evaluation for acrylonitrile MoA
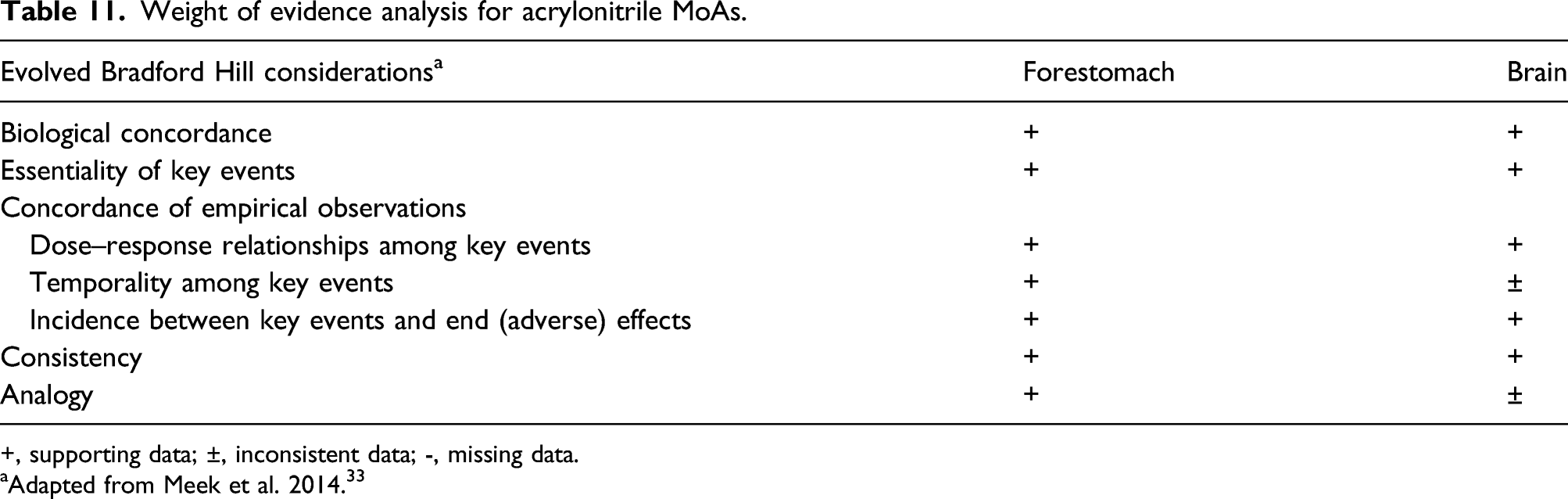
The US EPA/IPCS animal MoA guidance
33
was applied to two of the target tissues of acrylonitrile carcinogenicity, forestomach and brain, to evaluate weight of evidence for MoAs of acrylonitrile carcinogenicity suggested above (Table 11). 1. Biological concordance: The hypothesized MoA does not conflict with the broader biological knowledge. Although, the contribution of GSH depletion to chemical-mediated carcinogenicity is not established, it is known that GSH plays an important role in the detoxication of xenobiotics and protects tissues from oxidative stress. On the other hand, sustained cytotoxicity that was associated with glutathione depletion by acrylonitrile in gastric tissue and brain and resulting compensatory cell proliferation is a well-established MoA for many chemical carcinogens.
163
2. Essentiality of key events: Cytotoxicity of acrylonitrile was prevented by depletion of CYP2E1 enzyme and by supplementation of sulfhydryl compounds. Thus, no toxicity was observed in CYP2E1 knockout mice administered acrylonitrile, while in rats, induction of CYP2E1 enzyme exacerbated acrylonitrile toxicity. Depletion of GSH showed strong correlation with accumulation of cyanide in various tissues. In addition, GSH depletion correlated with forestomach mucosa toxicity and oxidative DNA damage in brain. 3. Concordance of empirical observations among key events (dose-response, temporality, incidence): Reduction of GSH levels after acrylonitrile administration was shown to be dose- and time-dependent. Both target organs for acrylonitrile carcinogenicity, forestomach and brain, demonstrated limited capacity to recover depleted GSH. Mice, which metabolize acrylonitrile faster, developed forestomach tumors at lower doses compared to rats. Progression from cytotoxicity to hyperplastic changes of the glandular mucosa of the stomach that correlated with GSH depletion was observed in many subchronic and chronic studies at dosages comparable to those at which tumors or acute toxicity signs were observed. In the brain, cytotoxicity caused by oxidative damage which correlated with GSH depletion was also observed in a dose-dependent manner. However, in the chronic carcinogenicity bioassay, a temporal relationship between glial cell proliferation and incidences of brain tumors was not clearly defined. 4. Consistency: Toxicity of acrylonitrile to the gastrointestinal tract and central nervous system was reported in multiple acute, subacute, subchronic, and chronic studies in rodents. Evidence of oxidative stress after acrylonitrile exposure was demonstrated in the brain of rats in several studies. 5. Analogy: Regarding the forestomach neoplasms, a variety of other chemicals that produce cytotoxicity in the forestomach have produced an increase in neoplasia in that tissue, that is, butylated hydroxyanisole, ethyl acrylate, and sesamol.46,164,165 Concerning the brain tumors, no other tumorigen for this tissue has been associated with oxidative DNA damage. Nevertheless, oxidative DNA damage has been found in other target tissues, for example, lung or liver, for other carcinogens, for example, pentachlorophenol, arsenic, phenobarbital.166–168 In addition, oxidative damage has been implicated in production of neurotoxicity by several chemotherapeutic agents, such as cisplatin.
169
Thus, the suggested MoA would be anticipated in rodents. Weight of evidence analysis for acrylonitrile MoAs. +, supporting data; ±, inconsistent data; -, missing data. aAdapted from Meek et al. 2014.
33
Human biological plausibility
Summary of evidence for key characteristics of carcinogen based on mechanistic data for acrylonitrile described in literature.
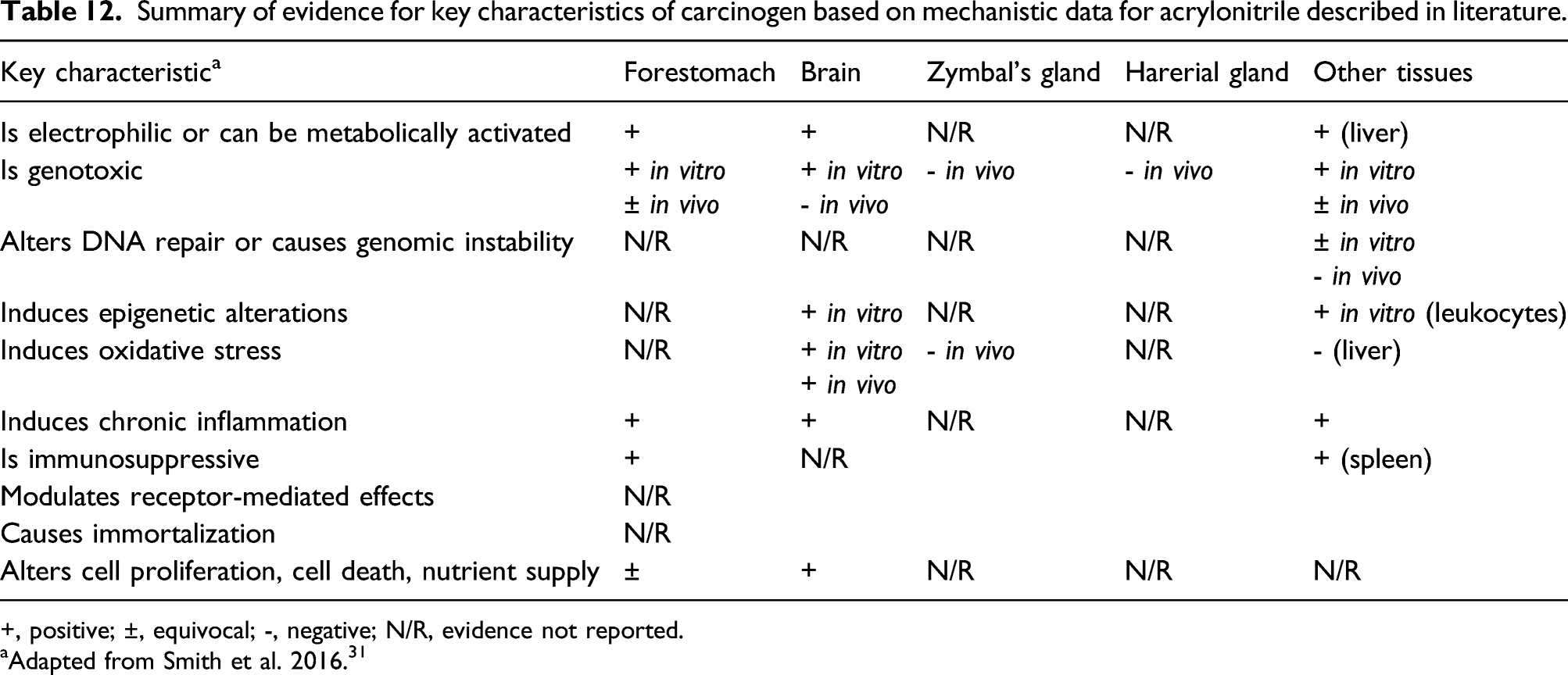
+, positive; ±, equivocal; -, negative; N/R, evidence not reported.
aAdapted from Smith et al. 2016. 31
Symptoms of acrylonitrile toxicity in workers were associated with high blood levels of cyanide ranging from 1.9 mg/L to 4.3 mg/L, suggesting that oxidative metabolism of acrylonitrile in humans is more active compared to rodents. 170 However, under environmentally-relevant low-dose exposures, acrylonitrile metabolism to CEO is limited by liver blood flow and alveolar ventilation rate rather than intrinsic rate of metabolism. 171 Another important species difference in the metabolism of acrylonitrile is the presence of genetic polymorphism in human population. Investigation of hemoglobin adducts associated with acrylonitrile exposure suggested that in humans GSTP1 is more important for detoxication of acrylonitrile compared to GSTM1 subunit which is selective for acrylonitrile binding in rats,67,69 and potential role of variation in GST genotypes might play a role in susceptibility of humans to acrylonitrile toxicity. 68 Specifically, polymorphism of the GSTP1 and GSTT1, which is associated with deficient conjugation, might play a role. No significant influences of CYP2E1 polymorphism were found on acrylonitrile-specific hemoglobin adduct levels in humans, except for a trend pointing to higher levels of adducts with the A-316G mutations, nevertheless, slower metabolism of acrylonitrile via CYP2E1-mediated pathway could be associated with higher adduct levels. 80
Three of the target tissues, that is, forestomach and Harderian gland (both present in rats and mice) and Zymbal’s gland (present and described commonly in rats, but only in a vestigial form in mice),are absent in humans. Of these three target tissues, applying data on rodent forestomach tumors (occurring in both sexes of rats and mice) to the prediction of cancer risk in humans has been a particularly controversial aspect of interspecies extrapolation. 46 Since the forestomach is lined with squamous epithelium similar to the pharynx and esophagus, some argue that forestomach tumors in rodents may be relevant to human hazard.164,172 It is generally accepted that genotoxic chemicals that elicit forestomach neoplasia in the absence of forestomach irritation and inflammation are considered to be more likely relevant to human risk.46,173 Acrylonitrile does not have this property. Additionally, the most relevant exposure route in humans is inhalation and not the alimentary route. No increase in forestomach tumors was observed in the carcinogenicity bioassay with acrylonitrile administration by inhalation, 39 indicating a role for direct contact.
In contrast to observations of acrylonitrile-associated brain tumors in several rat bioassays,13,14 none of several epidemiological studies reported increases in brain tumors in populations exposed to acrylonitrile in occupational settings.5,6,10–12 The acrylonitrile-associated brain tumors in rats were initially diagnosed by conventional histopathology as gliomas derived from astrocytes (astrocytomas). Re-examination of these tumors utilizing immunohistochemical markers revealed that they were primarily composed of microglia, 18 components of glia that function as macrophages and are important for the maintenance of the blood-brain barrier. In humans, the most common malignant primary brain tumor is glioblastoma, which is primarily derived from the astrocytic lineage.174–176 While microglia represent a major non-neoplastic component of gliomas, 158 presently, only two cases of true microglioma in humans have been reported.174,177 This difference in cellular susceptibility may explain the absence of central nervous system carcinogenicity of acrylonitrile in humans. 178 Thus, in an evaluation of carcinogenic risk to humans, IARC 27 found that the evidence for the carcinogenicity of acrylonitrile was very limited, with brain tumors being highly infrequent in the epidemiological studies. In addition, the human blood-brain barrier offers more effective protection from toxins compared to either rats or mice.179,180
No macroscopic or histologic changes in the lungs were observed in rats in a 2-year inhalation study of acrylonitrile, although degenerative and inflammatory changes in the nasal passages were observed. 39 As mentioned above, a slight increase in combined incidences of alveolar/bronchiolar adenoma or carcinoma that may have been related to acrylonitrile exposure was observed only in female mice and was not dose dependent.15,16 In humans, while some studies detected an association between acrylonitrile exposure and increased risk of lung cancer in humans, 35 others have shown that these findings are unsupported based on the absence of dose-response and lack of proper adjustments for confounding factors, such as smoking. 4
Currently, the occupational exposure levels to acrylonitrile are estimated to be below 2 ppm.7,28 Assuming that the highest allowed occupational exposure to acrylonitrile is 2 ppm or 4.3 mg/m3 for 8-hours over a 40-hour work week,28,30 an average of 261 workdays per year and work duration of 10 years, an average lifetime daily exposure to acrylonitrile is estimated to be 146.4 μg/m3 (lifetime daily exposure = 4.3 mg/m3 x (8/24) hrs x (261/365) days x (10/70) years). Using the inhalation cancer unit risk factor of 1.02 × 10−6 per μg/m3 calculated above based on the animal data, an estimated excess lifetime cancer risk from acrylonitrile exposure is 1.49 × 10−4 (risk = 146.4 μg/m3 x (1.02 × 10−6 per μg/m3)) or approximately 0.015%, which is negligible. Even when calculations are corrected for a 40-year career-long occupational exposure, the lifetime cancer risk does not exceed 0.06%.
In conclusion, the suggested MoA of acrylonitrile carcinogenicity in rodents is consistent with direct and indirect (due to oxidative damage) cytotoxicity, and compensatory cell proliferation, although weak, likely indirect, mutagenicity cannot be ruled out. Overall, the relevance to humans of findings with acrylonitrile in rodent studies is questionable and requires further dose-effect and mechanistic investigation. Combined with the largely negative epidemiology in humans and margin of exposure, human risk is unlikely.
Supplemental Material
sj-pdf-1-tor-10.1177_23978473211055363 – Supplemental Material for Acrylonitrile induction of rodent neoplasia: Potential mechanism of action and relevance to humans
Supplemental Material, sj-pdf-1-tor-10.1177_23978473211055363 for Acrylonitrile induction of rodent neoplasia: Potential mechanism of action and relevance to humans by Tetyana Kobets, Michael J Iatropoulos and Gary M Williams in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the Global Acrylonitrile Product Stewardship. The authors certify that their freedom to design, conduct, interpret, and publish research was not compromised by the sponsor.
Supplemental material
Supplemental material for this article is available online.
Nomenclature
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

