Abstract
The aim of this study is to evaluate the cytotoxic and the genotoxic effects of triflumuron (TFM) on human colon carcinoma cells (HCT-116). Indeed, TFM is used to protect vegetables, fruits, and domestic animals against a large spectrum of parasites causing animal and human disorders. However, studies revealing its toxicity and its mode of action in mammalian systems remain very limited. We monitored our work with the cytotoxicity assay starting with the cell viability test, the ROS generation, the malondialdehyde (MDA) production, the DNA fragmentation, and the measurement of some antioxidant enzymes activities such as catalase, superoxide dismutase, and the glutathione S-transferase. Also, we measured the mitochondrial transmembrane potential. We showed that TFM induced a dose-dependent cell death. This decrease in cell viability was accompanied by a significant reduction in the mitochondrial membrane potential. We also have shown that TFM induced oxidative stress as revealed by the generation of reactive oxygen species, the increase of the MDA levels, and the activation of the antioxidant enzymes. Moreover, our results indicated that TFM induced DNA damage in HCT-116 cells as monitored by the comet assay. We demonstrate, for the first time, the cytotoxic and the genotoxic potentials of TFM on human cultured cells.
Introduction
Excessive use of pesticides in the agricultural sector can cause chemical contamination leading to the loss of plant and animal lives. 1 Indeed, pesticides can pose problems for human health since pesticide residues are frequently detected in different compartments of the environment (surface water, groundwater, soil, etc.) and in products intended for human and animal food. 2,3
Numerous epidemiological studies suggest that pesticides may be involved in the induction of cancer, neurodegenerative diseases, fertility, and reproductive disorders. 3 –7 That is why it is necessary to look for new products with higher selectivity for the target organisms and lower toxicity for human and for the environment.
Insect growth regulators (IGR) are among these new compounds. They act on the growth and development of insects. In fact, IGR were introduced following the appearance of insect’s resistance to pyrethroids and organophosphate insecticides. 8 Among the IGR, the class of benzoylureas (disrupting molt) interferes with the insect’s exoskeleton causing then its death. 9
Benzoylurea insecticides are widely used for crop protection, especially fruits and vegetables and could be classified such as insecticides of fourth generation having high selectivity, low acute toxicity for mammals, and high biological activity. 10 In this context, we focused our interest in this study on triflumuron (TFM), a benzoylurea insecticide that is used worldwide. TFM shows interesting properties such as high selectivity, high biological activity, and low toxicity for animals. 11 TFM is used to protect crops (apples, tomatoes, trees, cotton, and potato) and domestic animals against a broad spectrum of parasites. 12
Regarding the molecular level, TFM acts by inhibiting the synthesis of chitin which is a linear polymer composed of
Particularly, TFM was used to control
Like all other pesticides, TFM is known for its toxic effects to mammals and especially to aquatic organisms and invertebrates. For example, in rats, dogs, and rabbits the administration of these compounds causes spasms, skin, and respiratory irritation resulting in sneezing and in several eye damages. 17,18 Repeated administration of TFM is also known to cause hemolytic anemia 19 and reproductive toxicity. 20
Also, benzoylurea insecticides are known by their ovicidal activity. Indeed, the treatment of insects of the genus
Moreover, oral administration of TFM to male Wistar rats for 28 days resulted in elevated hemoglobin accompanied by an increase in the number of reticulocytes. 19
In human, the ingestion of an unspecified BPU caused toxic methemoglobinemia. The hospitalized patient manifested cyanosis, low oxygen saturation, and chocolate-brown colored blood, the methemoglobin concentration was 59.3%. Following treatment with ascorbic acid, there was evidence of hemolysis. 24
Moreover, those symptoms were confirmed in humans. Recently, Ning et al. 25 reported that in Hep G2 cells, TFM was able to promote the metastasis of human liver cancer cells and that hypoxiainducible factor-1a is potentially responsible for these changes.
Thus, we aimed to evaluate the cytotoxicity and the genotoxicity induced by this insecticide on human colon carcinoma cells (HCT-116), being one of the target cells exposed to this pesticide following the ingestion of contaminated products.
Materials and methods
Chemicals
TFM was supplied by Sigma-Aldrich (St Louis, Missouri, USA). 3-[4,5-Dimethylthiazol-2-yl]-2,5-diphenyltetrazolium bromide (MTT), cell culture medium (RPMI 1640), fetal calf serum, phosphate-buffered saline (PBS), trypsin-EDTA, penicillin and streptomycin mixture, and
Cell culture and treatment
HCT-116 were cultured in RPMI, supplemented with 10% featal bovin serum (FBS), 1%
Cell toxicity assay (MTT assay)
The MTT assay (a tetrazolium salt reduction assay) provides sensitive measurements of the normal metabolic status of cells, particularly that of the mitochondrion, where measurements reflect early cellular redox changes. 26 HCT-116 cells (2.5 × 104 cells/well in 96-well plates) were incubated at 37°C for 24 h with different concentrations of TFM (100, 200, 400, 600, 800 and 1000 µM). A negative control containing untreated cells was also evaluated. After treatment, the plates were incubated with the MTT solution (final concentration of 0.5 mg/mL) for 3 h. The dark-blue formazan crystals formed in intact cells were dissolved with DMSO, and the absorbance at 570 nm was measured with a spectrophotometer microplate reader (Bioteck, Elx 800, France). The results were expressed as the percentage of MTT reduction relative to the absorbance’s measured from negative control cells. All assays were performed in triplicate.
FDA/BET marking
The fluorescent probe fluorescein diacetate (FDA) and the ethidium bromide (BET) were used to assess cell mortality. FDA/BET labeling allowed the distinction between live and dead cells. Indeed FDA and BET are two chloroforms that enter into the cells except that for the FDA it will be converted and cleaved only by esterases of living cells. To do this, cells were seeded in six-well plates (7.5 × 10 6 cell/well) and treated with the different concentrations of TFM (100, 200, 400, 600, 800 and 1000 µM). After 24 hexposure, cells were trypsinized and centrifuged (5 min at 3000 r/min). The pellet was resuspended in 100 µL of the medium. Thus, 10 µL FDA (10 µg/mL) and 10 µL BET (100 µg/mL) were added to the mixture. Then the contents are spread on a slide and cells were visualized using a fluorescence microscope, and on 100 cells, we count the number of living and dead cells.
ROS determination and oxidative stress status
Reactive oxygen species (ROS) are markers of oxidative status. Indeed, in case of cellular stress, it has an increased production of these ROS that interact with DNA, lipids, and proteins. The intracellular amounts of ROS were measured by a fluorometric assay with DCFH-DA used extensively to monitor oxidation in biological systems as a well-established compound to detect and quantify intracellular compounds produced such as superoxide radical, hydroxyl radical, and hydrogen peroxide (H2O2).
27,28
The conversion of the nonfluorescent (DCFH-DA) to the highly fluorescent 2,7-dichlorofluorescein (DCF) product (
HCT-116 cells were seeded on 24-well culture plates (Polylabo, Strasbourg, France) at 105 cells/well for 24 h of incubation. After cells were incubated with different concentrations of TFM, for 24 h at 37°C. H2O2 (20 µM) was used as a positive control. After incubation, cells were treated with 20-µM DCFH-DA. Intracellular production of ROS was measured after 30-min incubation at 37°C by fluorometric detection of DCF oxidation on a fluorimeter (Biotek FL 800×) with an excitation wavelength of 485 nm and emission wavelength of 522 nm. The DCF fluorescence intensity is proportional to the amount of ROS formed intracellularly.
Lipid peroxidation measurement
The measurement of lipid peroxidation was determined according to Ohkawa et al. 30 by the measurement of the level of malondialdehyde (MDA) which is an ultimate fragment of the degradation of the polyunsaturated fatty acids of the lipidic membrane.
To do this, cells were plated in six-well plates at 7.5 × 105 cells/well and treated with the different concentrations of TFM for 24 h of incubation. A positive control using H2O2 (20 µM) has been realized. Then, cells were collected in coded test tubes. Samples were mixed with 0.1 mL of 1.15% potassium chloride, 0.2 mL of 8.1% sodium-dodecylSulfate (SDS), 1.5 mL of 20% acetic acid adjusted to pH 3.5, and 1.5 mL of 0.8% thiobarbituric acid. The obtained mixture was vortexed and heated at 95°C for 2 h. After cooling to room temperature, a volume of 5 mL of mixture of
Protein extraction
To obtain cell lysates, HCT-116 cells were seeded in six-well plates (106 cells/well) and then treated with TFM for 24 h. Thus, cells were recovered in PBS, centrifuged and collected in cold lysis buffer solution for 30 min. A centrifugation makes it possible to obtain the protein extracts. Protein concentrations were determined in cell lysates using Protein Bio-Rad (France) assay. 31
Measurement of SOD activity
Superoxide dismutases (SODs) are ubiquitous metalloenzymes that catalyze the disproportionation of superoxide ions into H2O2 and molecular oxygen. The measurement of SOD activity was carried out according to the method described by Markland and Marklund, 32 by following the autoxidation and illumination of pyrogallol at 440 nm for 3 min. One unit of SOD activity was calculated as the amount of protein that caused 50% pyrogallol autoxidation inhibition. The SOD activity is expressed as U/mg protein.
Measurement of CAT
Catalases (CATs) are enzymes that intervene in the defense of the cell against oxidative stress by eliminating oxygen species (H2O2). To measure the activity of this enzyme, 780 µL of phosphate-buffered solution, 20 µL of each sample, and 200 µL of H2O2 (the substrate of the enzyme) were placed in a quartz cuvette. Then, the optical density was measured at 240 nm for an interval of time of 1 min. 33 The activity of CAT was calculated using the molar extinction coefficient (0.04 mM−1 cm−1). The results were expressed as mmol/min/mg protein.
Measurement of GST
The activity of this enzyme was determined by the formation of a chromophore, the 1-glutathione-2,4-dinitrobenzene from 1-Cl-2,4 dinitrobenzene (CDNB), which was followed spectrophotometrically at 340 nm. To do this, in a cuve, a mixture of 830 µL of phosphate buffer, 50 µL of CDNB, 100 mL of glutathione reduced (glutathion [GSH]), and 20 µL of each sample were realized. The absorbance was determined at 340 nm for an interval of 1 min. 34
MMP assay
The uptake of the cationic fluorescent dye rhodamine 123 has been investigated to estimate the mitochondrial membrane potential (MMP). 35 In a typical experiment, cells seeded in 96-well culture plates were treated for 24 h with TFM (100, 200, 400, 600, 800 and 1000 µM) then rinsed with PBS and 100 µL of rhodamine 123 (1 µM) in PBS was added on the plates. Cells were incubated (37°C, 5% CO2) for 15 min. Next, the PBS solution containing un-absorbed rhodamine 123 was removed and replaced by fresh PBS. The uptake rhodamine 123 was measured by fluorimetric detection with an excitation wavelength of 488 nm and an emitting wavelength of 525 nm. The results were expressed as the percentage of uptaken rhodamine fluorescence relative to the fluorescence measured from negative control cells.
DNA damage assessed by the comet assay
To measure DNA damage, caused by a toxic agent, in individual mammalian cells, the comet test was considered. For this, six-well plates were used to seed HCT-116 cells at 7.5 × 105 cells/well. After 24 h of incubation, cells were cultured in the presence of TFM (100, 200, 400, 600, 800 and 1000 µM) and H2O2 (20 µM) which served as a positive control. Cells already recovered in PBS were mixed with low melting agarose, and the mixture was then spread on a microscope slides covered with normal agarose. Thus, the slides were put in an alkaline lysis buffer solution for 1 h at 4° C. Then, electrophoresis step (30 min, at 25 V, 300 mA) was done before neutralizing slides using a Tris buffer solution for 15 min. After drying the slides, a staining with ethidium bromide (20 µg/mL) allowed to quantify the DNA damage using a fluorescence microscope. The experiment was done in triplicate. The damage is represented by an increase of DNA fragments that have migrated out of the cell nucleus during electrophoresis and formed an image of a “comet” tail. A total of 100 comets on each slide were visually scored according to the intensity of fluorescence in the tail and classified by one of five classes as described by Collins et al. 36 The total score was evaluated according to the following equation:
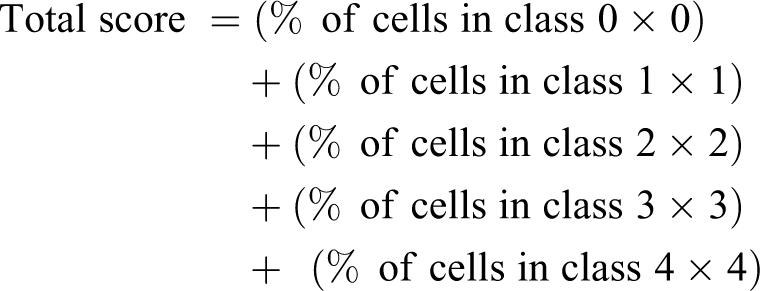
Statistical analysis
Each experiment was done three times separately and data were expressed as the mean ± standard deviation of the means. One-way ANOVA was used to assess differences among the groups followed by Dunnett’s post hoc test. When two groups were compared, differences were assessed by Student’s
Results
TFM induces cell death in HCT-116
After treating HCT-116 cells with the increasing concentrations of TFM (100, 200, 400, 600, 800 and 1000 µM) for 24 h, the MTT test which is a cell viability test has been carried out. We showed that TFM significantly decreased cell viability (
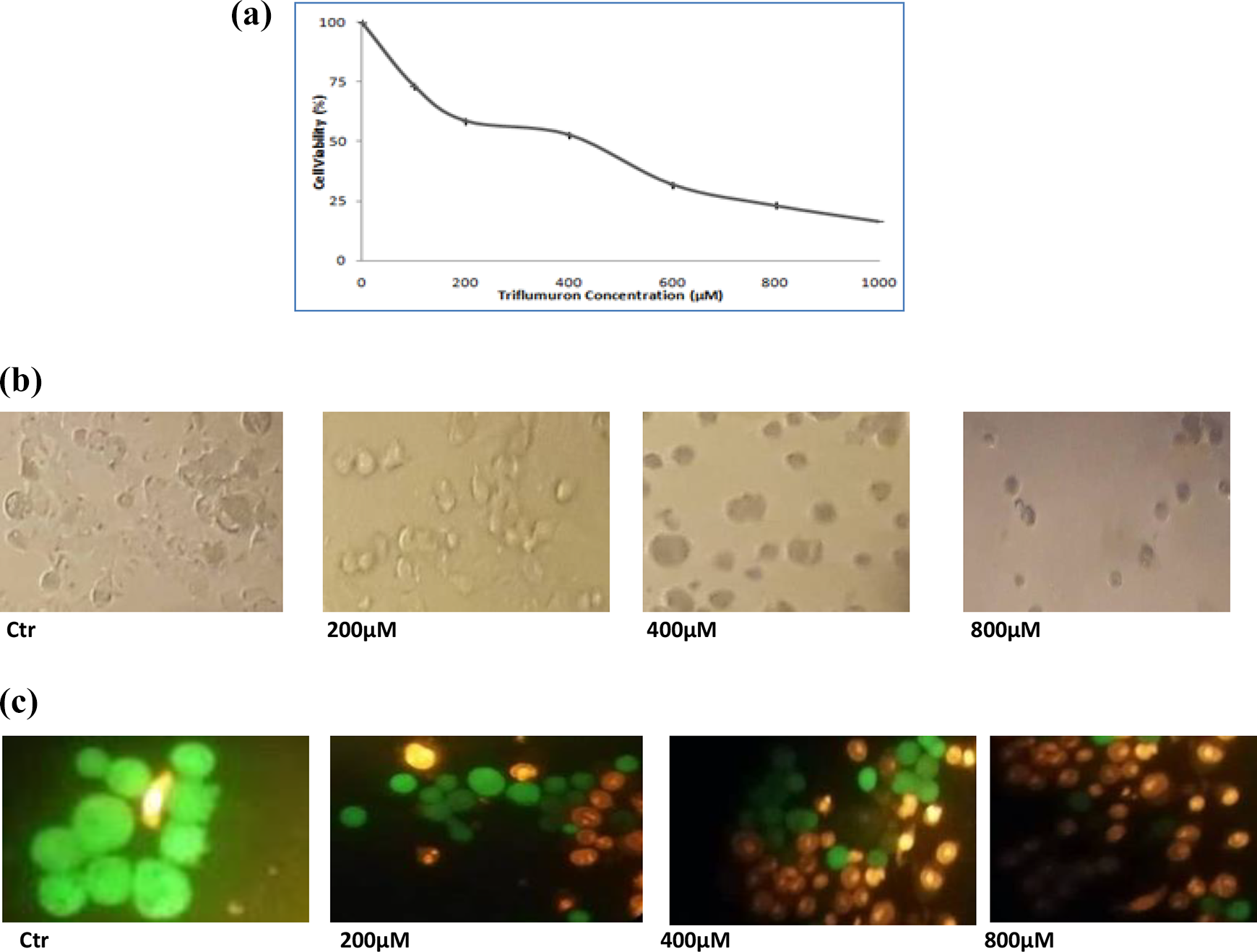
Cytotoxic effect of triflumuron on HCT-116 cells after 24-h treatment. Cells were treated with different concentrations. Cell viability was determined using the MTT assay and expressed as percentages of viability (a). Data are expressed as the mean ± SD of three independent experiments. Values are significantly different (
Using another cell viability test, FDA/BET labeling, our results were presented in Figures 1(b) and 2(c). Thus, after cells exposure to different TFM concentrations (200, 400 and 800 µM), cell morphology was changed, visualized with an optical microscope (Figure 1(b)). After mixing cells with the fluorochromes, living cells appeared in green, while dead ones appeared in orange. Finally, cells were visualized using a fluorescence microscope (Figure 1(c)). In both cases, cells were photographed using a digital camera (original magnification 200×).
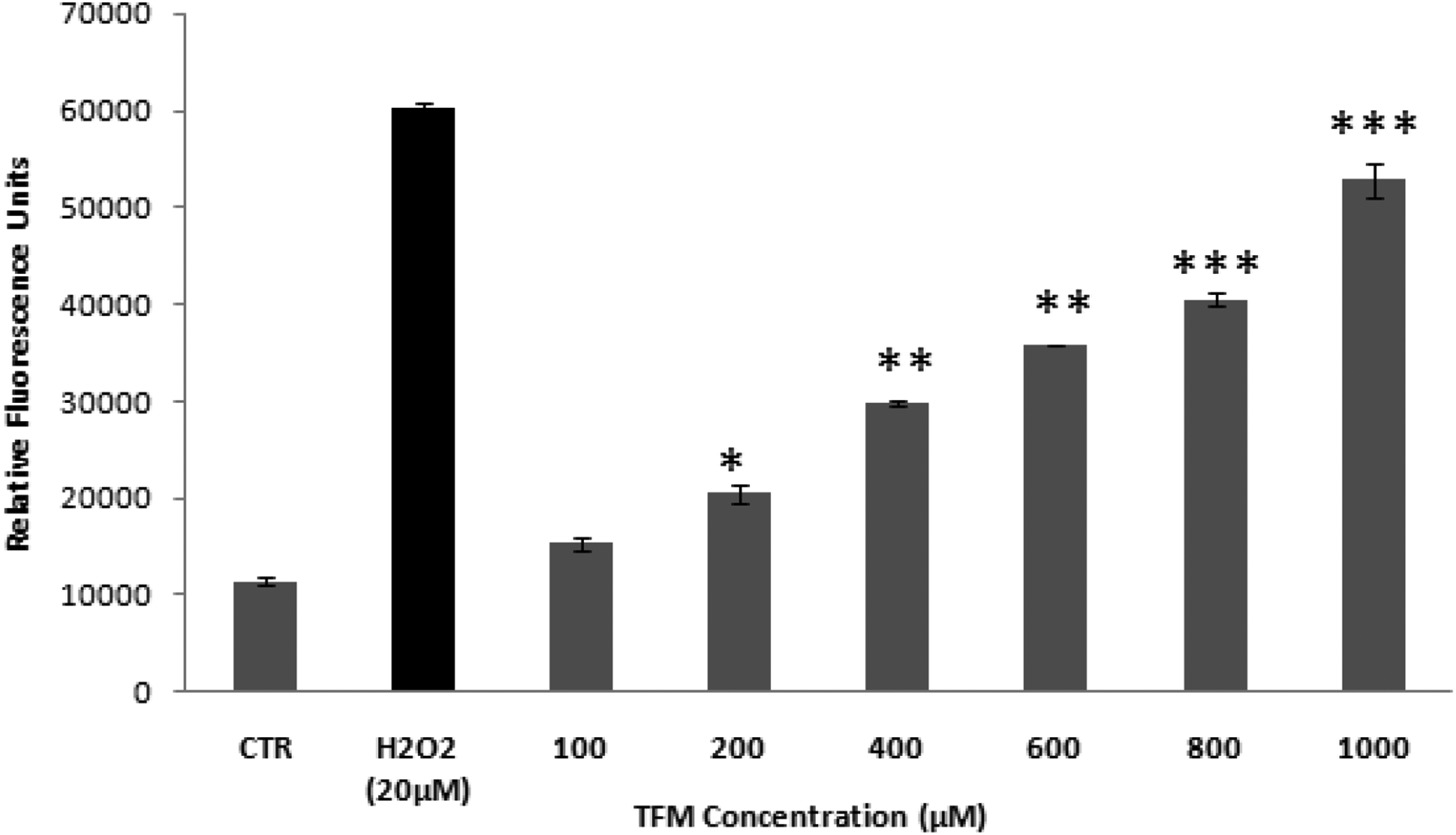
Levels of relative fluorescent DCF production after exposure of HCT-116 cells to different triflumuron concentrations for 24 h. H2O2 (20 µM) was used as a positive control. Data are expressed as the mean ± SD of three independent experiments. Values are significantly different (
Measurement of ROS production
The concentration range already used was tested to verify the proportion of the oxidative stress induced by TFM in the HCT-116 cells. Thus, the generation of ROS was measured via the production of fluorescent DCF. Our results showed that TFM is able to increase the level of ROS in the cellular model used in a dose-dependent manner (Figure 2).
Induction of lipid peroxidation
After exposure of HCT-116 cells to different concentrations of TFM for 24 h, the MDA assay was performed. Our results showed that TFM is able to induce lipid peroxidation. Indeed, the level of MDA increased from 0.057 ± 0.0069 µmol MDA/mg of protein in the untreated cells to 0.198 ± 0.0023 µmol MDA/mg of proteins in cells treated with the highest concentration of TFM (Figure 3).
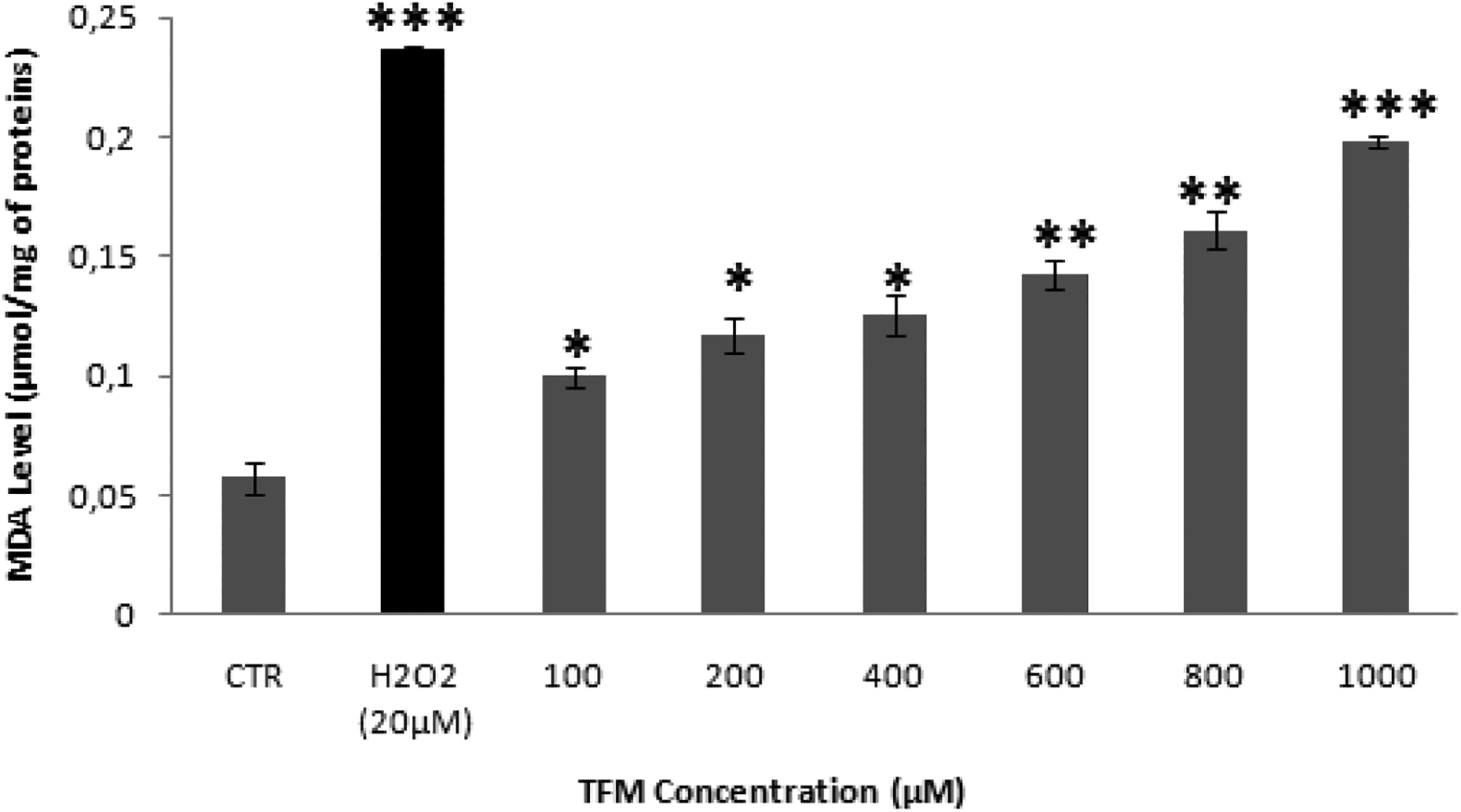
Induction of lipid peroxidation in HCT-116 cells, after 24 h of incubation with triflumuron measured by the production of MDA. H2O2 (20 µM) was used as a positive control. Data are expressed as the mean ± SD of three independent experiments. Values are significantly different (
Effect of TFM on antioxidant enzymes activities
To evaluate the effect of TFM on the activities of antioxidant enzymes, we measured the activity of CAT, SOD, and glutathione S-transferase (GST). Our results showed that TFM is able to increase the activities of these enzymes in HCT-116 cells, in a dose-dependent manner. Indeed, the activities of these enzymes passed from 113.80 ± 4.28 (USOD/mg of proteins), 64.26 ± 3.58 (mmol/min/mg of proteins), and 0.271 ± 0.0036 (µmol/min/mg of proteins) in the untreated cells to 303.10 ± 6.03 (USOD/ mg of proteins), 201.64 ± 3.23 (mmol/min/mg of proteins), and 1.48 ± 0.18 (µmol/min/mg of proteins) in cells treated with TFM at 1000 µM, respectively, for SOD, CAT, and GST (Figure 4(a), (b) and (c)).
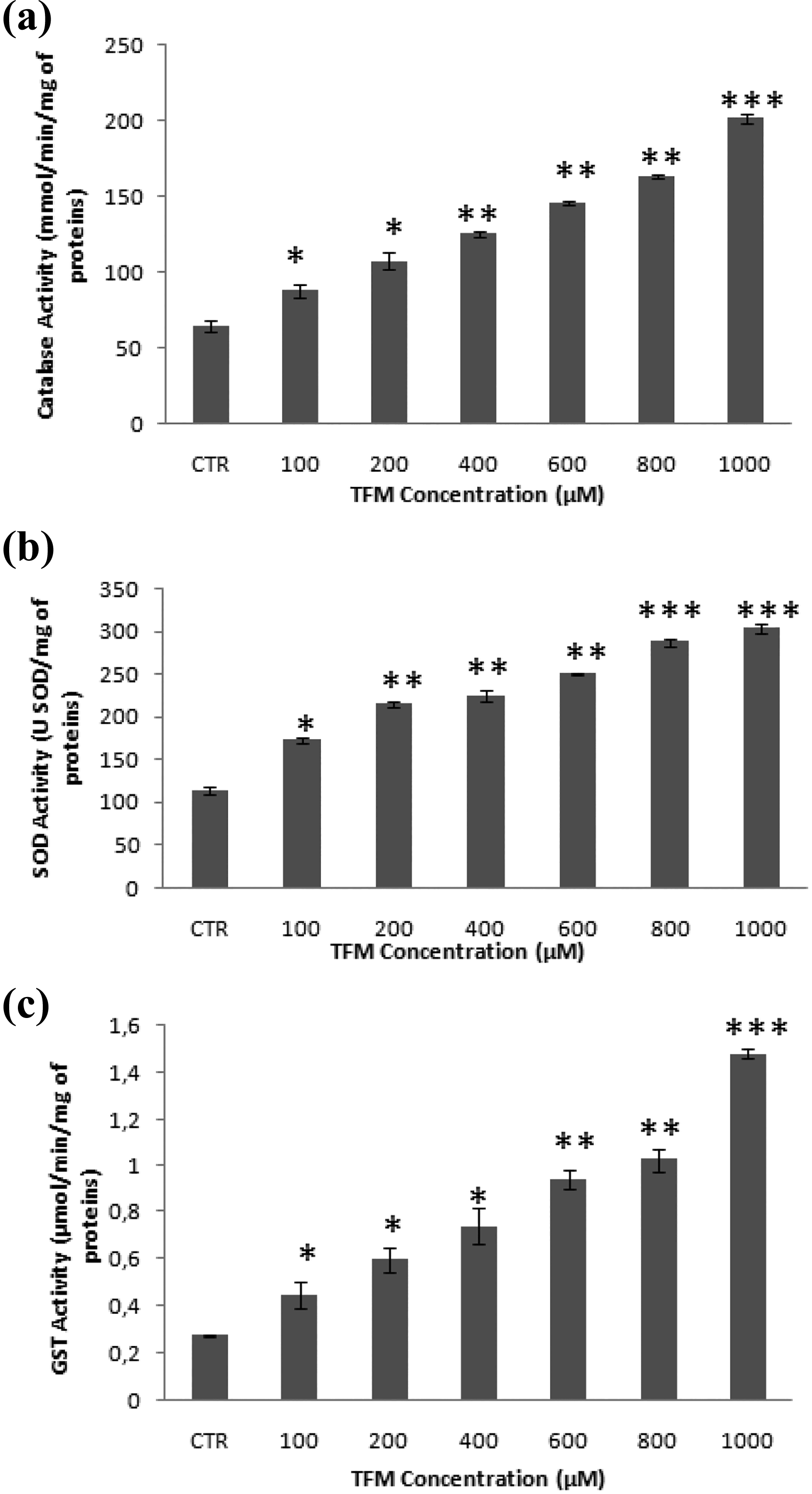
Effects of triflumuron on antioxydant enzyme activities such as catalase (a), superoxide dismutase (b) and glutathion S-transferase (c), after incubation of HCT-116 cells with the tested concentrations of TFM for 24 h. Data are expressed as the mean ± SD of three independent experiments. Values are significantly different (
Measurement of mitochondrial transmembrane potential (ΔΨm)
The measurement of mitochondrial transmembrane potential (ΔΨm) after cell exposure to TFM was carried out using rhodamine 123. Indeed, ΔΨm is proportional to uptaken rhodamine 123. 37 Results of this test are indicated in Figure 5, where it has been found that the tested concentrations have significantly decreased this potential, indicating then the depolarization of mitochondria.
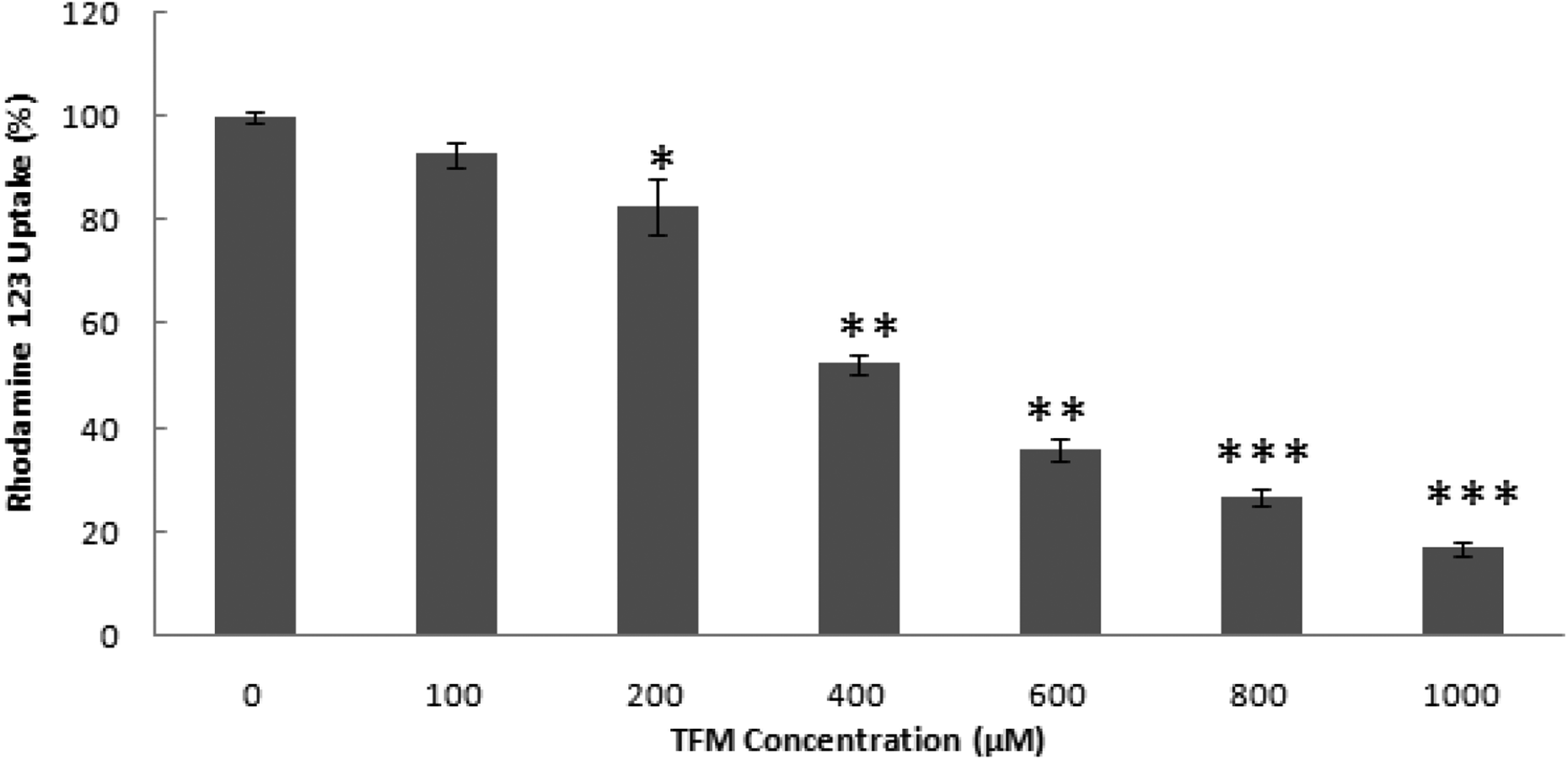
Triflumuron induces a loss of mitochondrial transmembrane potential on HCT-116. Cells were treated with TFM at the indicated concentrations for 24 h. Data are expressed as the mean ± SD of three independent experiments. Values are significantly different (
TFM induces DNA damage
DNA damages were analyzed using the alkaline comet assay. After treatment of HCT-116 cells with different concentrations of TFM for 24 h, we noticed a marked increase in the DNA damages. These damages are classified into five classes from 0 to 5 (Figure 6(a)). Indeed, as shown in Figure 6(b), the total score passed from 53 ± 8.48 in the control cells to 264 ± 12.72 when cells were treated with TFM at 1000 µM. H2O2 (20 µM)-treated cells (positive control) induced 298.4 ± 14.24 of the total score of DNA damage. Moreover, after 24 h of TFM cell exposure to 200, 400, 600 and 1000 µM. These damages are classified into five classes from 0 to 4 (Figure 6(b)).
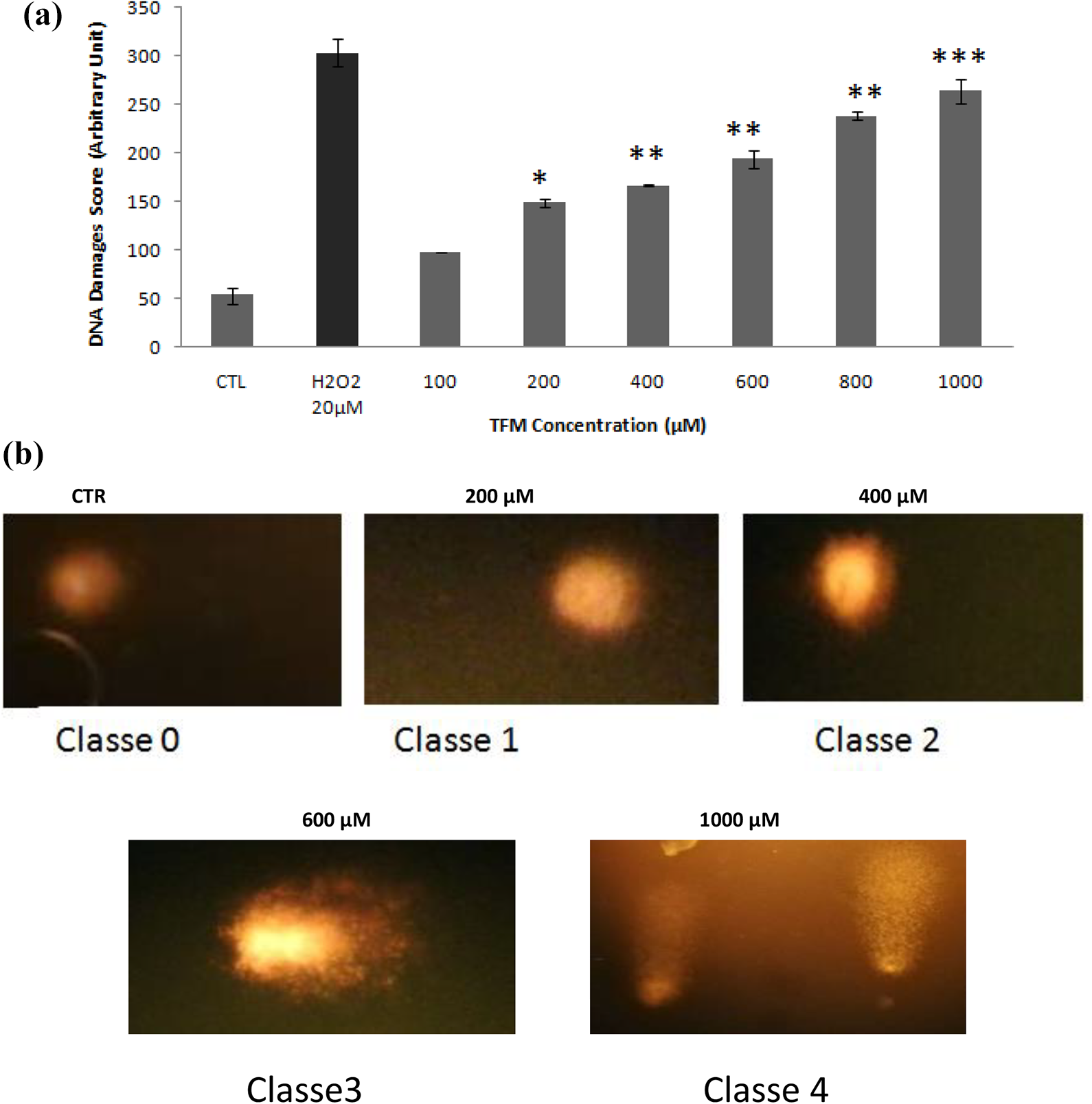
(a) Induction of DNA damages in HCT-116 cells, following treatment with triflumuron at 100, 200, 400, 600, 800, and 1000 μM. H2O2 (20 μM) was used as a positive control. DNA stand breaks were detected by the standard comet assay. Data are expressed as the mean ± SD of three independent experiments. Values are significantly different (
Discussion
Pesticides are molecules used to protect humans and their environment against attacks caused by harmful vectors. 10 Due to their increased use in many areas such as agriculture, industry, and traditional medicine, pesticides cause adverse effects on human health and threaten ecosystems. 38 In this context, several studies have revealed the role of pesticides in the occurrence of numerous pathologies, namely cancer, neurodegenerative, and cardiac disorders. 4,39,40 In this article, we investigated the mechanisms by which TFM, a benzoylurea insecticide belonging to the IGRs, was used. 18 TFM inhibits the synthesis of chitin, the major constituent of the insect cuticle causing consequently its death. 35 However, studies highlighting the toxicity mechanism of TFM remain very limited.
The aim of the present study is to evaluate the mechanism of action of TFM-induced cytotoxicity in cultured HCT-116 cells which are intestinal human cells that represent the first barrier to meet for any toxic substance.
Our results indicated that exposure of HCT-116 cells to increased concentrations of TFM induces a dose-dependent cell death as revealed by MTT test. Moreover, cells labeling with FDA/BET showed that by increasing TFM concentrations, cell morphology was changed and cell mortality was increased, which is in agreement with the results founded using the MTT assay (data not shown).
In fact, the tested concentrations represent 0.035, 0.071, 0.14, 0.21, 0.28, and 0.35 mg/mL, respectively, for 100, 200, 400, 600, 800, and 100 0 µM, of the environmental doses detected in TFM residues. TFM was detected in crops with values of 0.04 mg/kg and 0.03 mg/kg respectively in pear and wool. 41,42 Moreover, TFM was detected in tomatoes with value ranged from 0.5 mg/kg to 2.1 mg/kg. 43
These results support the toxicity of benzoylurea pesticides. Indeed, another study demonstrated that Lufenuron, belonging to this family, causes toxic effects on aquatic species where hemorrhages were observed in eyes, fins, and lids of fish exposed to this pesticide. Moreover, the histological analyses revealed that the gills structure have changed. 44
Besides, we found that TFM-induced cell death was associated with a significant decrease in the MMP after 24 h of treatment, which suggest the involvement of mitochondrial pathway in cell apoptosis caused by TFM since the mitochondrial membrane depolarization is able to cause the release of cytochrome
Moreover, oxidative stress has been proposed as a mechanism linking to pesticides exposure, increasing the risk for the development of diseases such as cancer and neurodegenerative diseases. In addition to the increase in the production of free radicals, exposure to pesticides can also affect antioxidant capacity and defense mechanisms, as well as increase the lipid peroxidation. 47,48 In agreement, we observed, in HCT-116 cells, that TFM induces the production of ROS and stimulates the activities of antioxidant enzymes such as CAT, SOD, and GST. Moreover, the level of MDA, one of the major oxidation products of peroxidized polyunsaturated fatty acids, was greatly increased following cell treatment with TFM. These results confirm the ability of benzoylphenyl urea pesticides to induce oxidative stress in mammalian systems. In this context, diflubenzuron (DFB) was described as an inductor of lipid peroxydation in Caco-2 cells. The same study showed that DFB was able to increase CAT, SOD, and Gpx activities. 49,50 DFB was also found cytotoxic toward Balb/c 3T3 cells leading to cell transforming activities. 49 In addition, DFB was found to be cytotoxic by decreasing cell viability in CHOK1 cells 49 and Hep G2 cells 51 using the MTT assay and the neutral red test. Besides, Lufenuron was also able to increase the levels of MDA generation and to disrupt the antioxidant parameters in the liver of exposed mice. 52 Our findings are in agreement with other research studies which showed that chlorfluazuron, a benzoylureas insecticide, induced cytotoxicity revealed via the MTT test in renal (HEK 293) and hepatic (Hep G2) human cells. 53 In addition, our previous study showed that TFM induced toxicity on a murine model after a short exposure. 54 In this study, we demonstrated that TFM is able to increase the levels of both carbonyl proteins and MDA as well as it altered the enzyme activities of antioxidant systems. 54 Similarly, the genotoxic effect of TFM was demonstrated in the bone marrow cells of male Balb/C mice. 55 Also, cytoxicity and genotoxicity of TFM on hepatic and renal human cells were well clarified. 56
Pesticides can be at the origin of genotoxic alterations. If a xenobiotic reacts with nuclear DNA, it may cause mutagenic and carcinogenic effects to the exposed organisms. Interestingly, our study clearly indicates that TFM induces significant DNA damage in HCT-116 cells. Therefore, the TFM-induced cell death in intestinal cells in this study can be due to the increase in oxidative stress accompanied by mitochondrial dysfunction and DNA fragmentation.
In conclusion, TFM is considered as a potential tool for the control of disease vectors in public health and agriculture. However, its uses should be controlled in order to avoid the health problems that may arise from unconscious human uses.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
This article does not contain any studies on human participants or animals performed by any of the authors.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by “Le Ministère Tunisien de l’Enseignement Supérieur et de la Recherche Scientifique.”
