Abstract
Assessing the risk of chemical mixtures is an intricate process that should integrate published laboratory data; comparisons with the composition, toxicity, and functionality of similar mixtures; complete analytical characterization of the mixture components; and in silico modeling. Various tiered assessment protocols have been proposed to address this need, and these protocols may be adapted on a case-by-case basis for both mixture-based and component-based evaluations. Emerging technologies have enabled rapid mixture testing in alternative animal models, such as human organotypic cultures and zebrafish. In addition, quantitative modeling that uses systems toxicology approaches can identify exposure-induced cellular and molecular alterations that would not be detected by standard toxicology assays. This review summarizes the approaches to risk assessment of complex chemical mixtures as presented at the Eighth International Congress of the Asian Society of Toxicology, June 2018.
Keywords
Strategies for the assessment of potential health impact of exposure to chemical mixtures
Humans are typically exposed to multiple chemicals rather than to single compounds, and various sources contribute to both simultaneous and sequential exposure to mixtures. The effects of these mixtures, however, are largely unknown. Chemical risk assessment is commonly conducted with isolated compounds without considering the potential interaction effects of mixtures of compounds. These effects can occur even when each compound is present at concentrations below the no-observed-adverse-effect level. 1,2
Assessing the risk of mixtures requires a proper risk assessment framework. Potential toxicity of a new mixture could be derived from data of similar mixtures if dose–response and mode-of-action data on the whole mixture are available. However, if such data are not available, component-based approaches can be leveraged. For a chemical mixture, the potential interactions between components have been traditionally described as additive, synergistic, antagonistic, potentiating, inhibiting, or coalitive. These specifications, however, are typically applied to interactions between two chemicals. Therefore, as most mixtures contain multiple constituents, quantifying these interactions in terms of risk assessment is not an easy task. 3
When sufficient safety information of a mixture is available from the literature, additional toxicology testing may not be necessary. Nevertheless, the characteristics of each compound in a mixture can also vary considerably. Compounds such as food ingredients or natural extracts may differ by species, variant, part, growing/synthesis conditions, batch, pollution, location, harvest time and stage, handling practices, and storage conditions. For the evaluation of a compound with unknown toxicity, it is possible to establish a “chemical bridge” to existing toxicology data of a similar compound. If the two compounds share similar chemical compositions, manufacturing processes, and physicochemical properties, one may be able to forgo toxicity testing on the new compound. The sources of the two compounds may still vary naturally or be purified differently; however, this variability may be similar or within the variability observed between batches of the same chemical. Moreover, if the composition and functionality of the two compounds are equivalent, their differences may not be of toxicological concern. 4
The scarcity of data on the toxicology of whole extracts or individual components of natural products (botanicals) makes safety determination challenging. A strategic framework for assessing the safety of natural products has been proposed by Booth and colleagues, who used cranberry leaf extract as a case study. 5 Although cranberry juice and cranberry extract are used widely, there is limited work on the chemistry of cranberry leaf. Because the leaf may have a different chemical profile than the fruit, an independent toxicity evaluation on the cranberry leaf is needed. The framework comprised various stages (Figure 1). 5 Booth and colleagues demonstrated that using appropriate criteria, the cranberry leaf extract could be established as a food-grade raw material for use in manufacturing for use in food. Although the authors “were not aware of any publicly available toxicological databases allowing them to predict synergistic, additive, or antagonistic effects of naturally-occurring compounds found in a mixture,” such analytical evaluation can suggest the appropriate limits of detection at which naturally occurring toxins are out of concern. 5 The European Food Safety Authority recently published a draft guidance 6 on “harmonized methodologies for human health, animal health and ecological risk assessment of combined exposure to multiple chemicals.” The harmonized framework is shaped based on the risk assessment phases (i.e. problem formulation, exposure assessment, hazard identification and characterization, and risk characterization, including uncertainty analysis). Booth and colleagues further specified that a set formula for dealing with safety evaluation of natural products does not exist and that each new submission must be dealt with on a case-by-case basis. 5
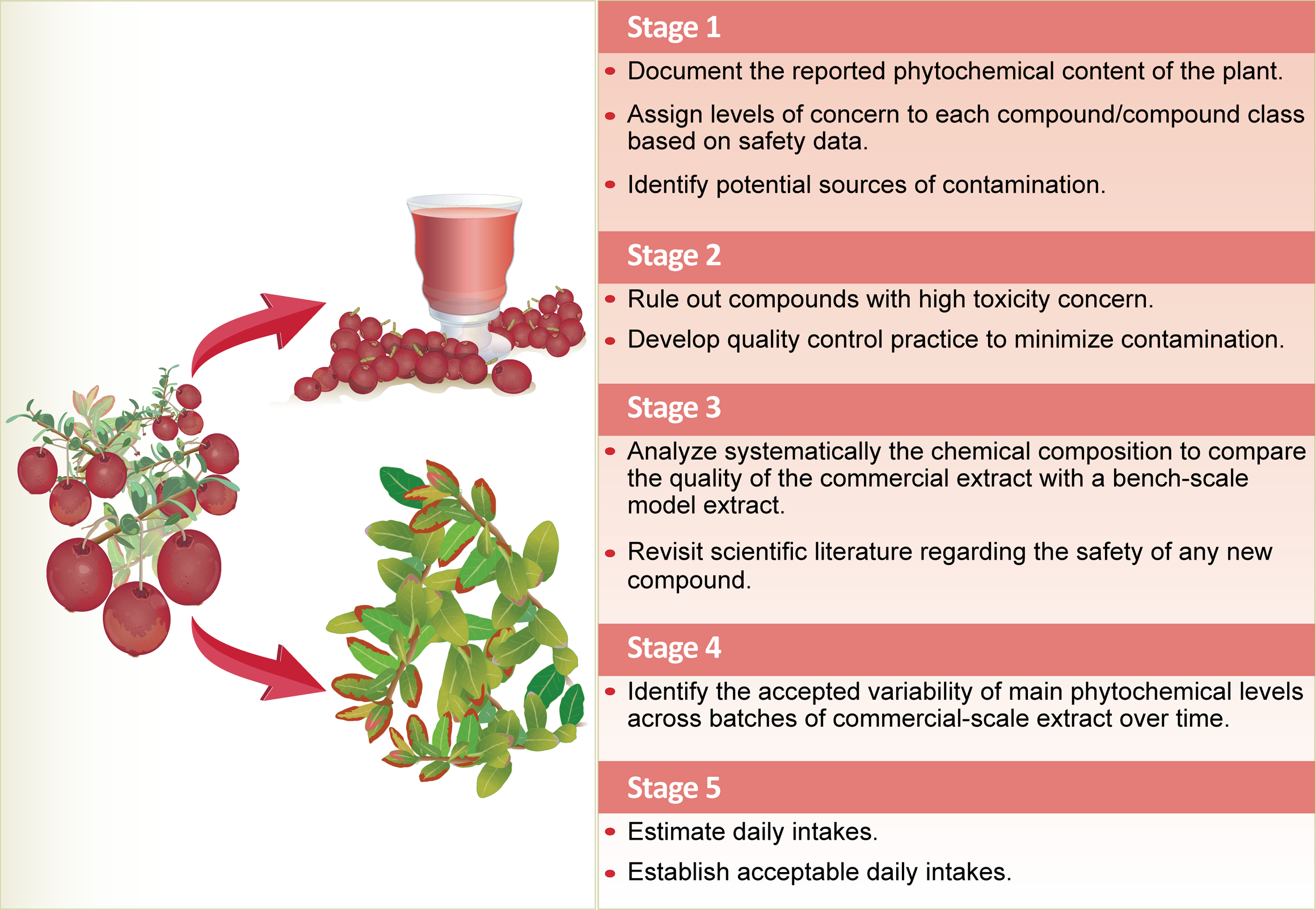
A strategic framework for the safety assessment of a cranberry leaf aqueous extract. There is limited work on the chemistry of cranberry leaf, although cranberry juice and cranberry extract are widely used. Because the leaf may have a different chemical profile than the fruit, an independent toxicity evaluation on the cranberry leaf is needed. The framework proposed by Booth and colleagues 5 comprised various stages of assessment.
In addition, Roe and colleagues had also proposed an industry strategy for the evaluation of the safety of botanicals as dietary supplements. 7 A part of the strategy was an in silico decision tree to address toxicity data gaps and to confirm the safe use of a botanical ingredient. When safety data are insufficient, one can address the data gap by utilizing both significant-human-use information and phytochemical constituent-based safety assessments to resolve toxicity end points of concern. By identifying and quantifying botanical constituents, food intake levels of the individual chemical constituent can thus be assessed using an in silico toxicology assessment tool for hazard identification. This decision tree approach can also bridge safety assessment between botanicals prepared using different methods (preparation and extractions). The decision tree uses multiple databases and information on structure–activity relationships to characterize the risks from individual constituents, establishing exposure thresholds for human use.
Evaluating mixture risk is a multistep process that includes mining published data and characterizing the mixture in the laboratory as well as in silico hazard analysis and modeling. Finally, the nature of the assessment may require the use of different approaches that should be determined on a case-by-case basis, which is particularly pertinent for the safety assessment of botanicals.
Systems toxicology approaches for chemical hazard assessment in zebrafish
Traditionally used by developmental biologists, zebrafish have gained recognition in the field of toxicology over the last few decades.
8,9
This is largely due to some unique characteristics that this model possesses. The eggs are fertilized and undergo development and maturation
The above characteristics make zebrafish ideally suited for high-throughput screening, which can be performed in multiwell plates integrating automated exposure and screening, 10 not unlike cell culture assays. Additionally, as only independently feeding animals are subject to regulation for animal experimentation in the European Union, early zebrafish embryos do not fall into the regulatory frameworks. 11 Unlike cultured cells, however, zebrafish embryos are intact vertebrates possessing functioning vertebrate organs, allowing the study of the cardiovascular, digestive, and central nervous systems. Owing to the organism’s suitability for genetic manipulation, numerous zebrafish transgenic reporter lines are available for rapid organ toxicity screening. 12,13 Additionally, toxicant absorption, distribution, metabolism, and excretion can be investigated in zebrafish, because the larvae possess functional skin, gills, kidneys, livers, and guts. 14 Seventy percent of human genes have zebrafish orthologs, 15 and the hematopoietic, 16 cardiovascular, 17 and pancreatic 18 physiologies are similar between humans and zebrafish. Pharmacology also appears to be largely conserved for diverse classes of drugs, 19,20 suggesting that toxic effects exerted by chemicals in humans can be studied in fish.
The Organization for Economic Co-operation and Development has published a guideline for determining the acute toxicity of chemicals in embryonic stages of fish 21 ; lethal and sublethal morphological end points are used to calculate lethal and effective concentrations for a given chemical. This has prompted major advances in the automation of methods such as embryo manipulation, 22 imaging, 23 and behavioral measurements, 20 facilitating phenotype discovery and high-throughput toxicity testing in an in vivo vertebrate system.
Assessing chemical mixtures in the developing fish and identifying transcriptional markers are relatively straightforward 24 and may help identify synergistic toxic effects that are difficult to foresee 25 or detect drivers of toxicity in the mixture. 26 Many zebrafish screens rely on phenotypical observations to infer toxicity, but, as with any phenotype-based screening, the mechanism of action is difficult to decipher from such data. Matching phenotypes between chemicals with a known mechanism of action and chemicals or mixtures with no known targets can be used to infer the mechanism. 27 Phenotype matching between known genetic mutations and chemicals has also been used successfully to identify chemical targets. 28 This approach, however, is labor-intensive, because it relies heavily on data generated from mutant or chemically exposed fish. Cellular and behavioral phenotypes must be defined in sufficient detail to capture subtle chemical toxicities.
Transcriptomic approaches have become reliable and economically viable as part of a chemical toxicity analysis, 29 but they present their own challenges. Protein activity, such as folding, localization, and posttranslational modifications in the presence of binding partners, is not captured in a transcriptomic experiment. Therefore, a toxicity assessment based on transcriptomic data alone may result in a significant number of false negatives. Proteomics and metabolomics address this issue to an extent, although these methods are relatively challenging, less reliable, and more expensive than transcriptomic approaches, with limited coverage of the proteome 30 or metabolome. 31 Developments in systems toxicology may address these limitations (Figure 2). Systems toxicology measures molecular changes within organ systems, using computational analyses to identify causal molecular events that lead to adverse outcomes. 32 This approach relies on a causal biological network, a collection of computable statements curated from scientific literature that describe molecular pathways within organ systems. 33 Biological entities, including mRNA, proteins, protein activities, biological processes, and diseases, are represented by nodes within the network. The nodes are connected by directed relationships, such as upregulation or inhibition, giving the network a cause-and-effect topology. Nodes within the network are then assigned information about downstream transcript abundance. Downstream transcripts are mRNAs that are known to be regulated by the corresponding upstream node. This information is either manually curated from literature or gathered from publicly available transcriptomic data sets. Transcriptomic data from organisms exposed to chemical mixtures can be used next to infer whether the upstream node was activated or inhibited. By performing this analysis for all scorable nodes within a biological network, it is possible to calculate the perturbation of the network by a chemical or a chemical mixture and to decipher the molecular pathways that lead to adverse outcomes. 34 If the same pathways are found in mammals, mechanisms of toxicity can be extrapolated to higher vertebrates, including humans (Figure 2).
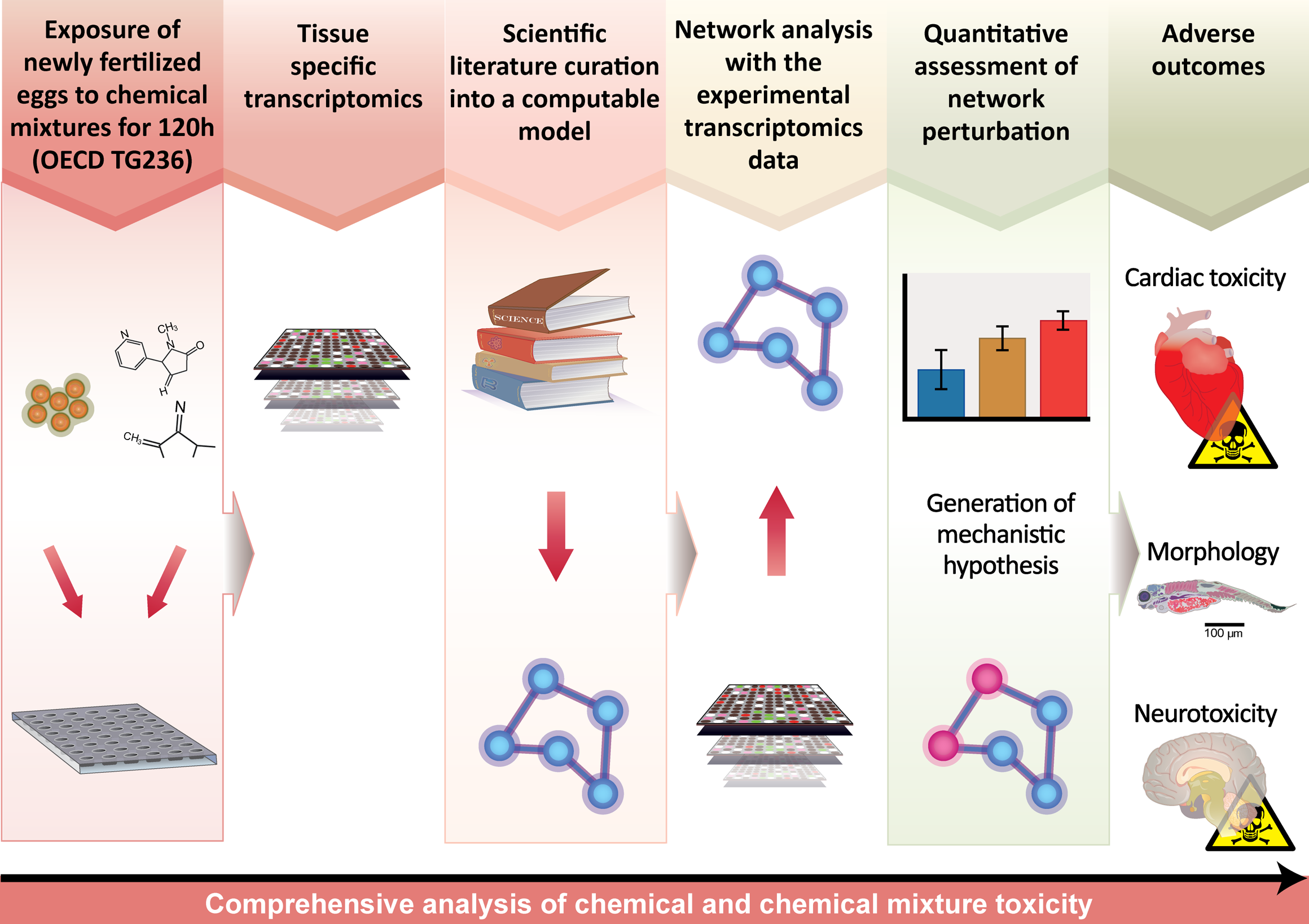
A systems toxicology approach to analyzing chemical and chemical mixture toxicity in zebrafish. Organ-specific transcriptomic analysis following exposure generates data for computations of biological network impact. The results are used to generate mechanistic hypotheses. Together with apical end points, this approach can be used to decipher the chains of molecular events leading to adverse outcomes.
The advantages of a network-based approach are as follows. (1) Multiple biological outcomes and entities, from RNA to protein to pathology, may be measured during a single transcriptomic experiment without the need for proteomics or metabolomics. (2) The assumption that mRNA abundance corresponds to protein activity is avoided. (3) Networks are organ system-specific, allowing for the assessment of toxicity for distinct organs. (4) Because networks are directed, it is easy to visualize a chain of molecular events that lead to adverse outcomes. (5) Perturbation of the entire network can be quantified, giving a numerical measure of toxicity. (6) Networks permit the analysis of the effects of chemical mixtures, which may perturb multiple pathways.
Zebrafish studies in environmental toxicology, 35 drug discovery, 36 and drug toxicity 37 have demonstrated that this organism is a valuable tool in biomedical research. The assessment of the mechanism of action of chemicals, chemical mixtures, and medicines afforded by the network biology approach may lead to more accurate environmental and human risk assessment and faster discoveries of safer pharmaceuticals. The next step toward this goal will be to build zebrafish-specific biological networks that describe pathways leading to organ toxicity.
In vitro approaches for assessment of complex aerosol mixtures
Smoking causes serious conditions such as lung cancer and chronic obstructive pulmonary disease. Philip Morris International (PMI) is developing novel products with the potential to reduce individual risk and population harm in comparison with smoking cigarettes and is conducting extensive and rigorous mechanistic studies to assess their biological impact vis-à-vis that of reference cigarettes. Reducing harm requires the availability of reduced-risk products that are accepted by users and can be accomplished by replacing cigarettes with such products (the effects of which on health should be as close to smoking cessation as possible).
The desire to move away from animal testing has been the impetus for developing advanced cell culture models, although findings from in vitro studies may not be interpreted or extrapolated in a straightforward manner in the context of in vivo effects. Thus, relevant biological test systems are needed to facilitate an accurate identification of biomarkers of exposure, response, and disease. For testing inhaled mixtures, in vitro studies may be conducted using submerged cultures or air–liquid interface (ALI) cultures. Although easier to handle, submerged cultures are not suitable for toxicity testing of inhaled compounds. ALI cultures enable direct exposure of cells and tissues to inhalable gases and aerosols, unlike testing protocols in two-dimensional submerged airway cultures, where liquid fractions or extracts of the smoke or aerosol of interest must be generated and applied. These fractions do not typically represent the entire chemical composition of the smoke or aerosol.
Various exposure systems are available for testing inhaled aerosols, including those manufactured by Vitrocell Systems, Borgwaldt, and Cultex Laboratories. 38 These systems must include modules that ensure the functionality of all steps in the process: generation of smoke or aerosol, although it can be accomplished manually, should be consistent; dilution of the smoke or aerosol must be fast and reproducible; and dose monitoring should be accurate. The last of these typically employs time-of-flight mass spectrometry for the gas phase and photometry, and microbalances for the particle phase, although the concentrations of deposited compounds can be monitored further. The aerosols must also be characterized for accurate computation of aerosol deposition rate, the mixing efficiency of the dilution system, the aerosol’s stability in the dilution system, and the influence of operating conditions and physical mechanisms. Finally, auxiliary equipment can be incorporated to control vacuum and dilution air flow rates, temperature, and humidity. 39
More than 6000 constituents have been identified in cigarette smoke, and many of these are formed during the combustion process. 40 Some constituents are harmful or potentially harmful, and it is unknown which of them are responsible for tobacco-related disease. 41 The lower temperatures and lack of combustion in heat-not-burn tobacco products reduce the number of constituents in the aerosol, and nicotine is transferred by distillation.
PMI has carried out a series of in vitro studies using human organotypic epithelium cultures (nasal, buccal, and bronchial) to assess a heat-not-burn tobacco product, the Tobacco Heating System (THS) 2.2, in comparison with a reference cigarette. 42 The Vitrocell® Exposure System (Vitrocell Systems GmbH, Waldkirch, Germany) was used to deliver aerosols to ALI cultures of human primary epithelial cells, and end points of interest were captured after the exposure by measuring mediators secreted into the basolateral medium and by histological analysis. The biological impacts of exposure to THS 2.2 aerosol were compared with those of exposure to reference cigarette smoke for culture morphology, secretion of inflammatory mediators, and differential gene expression. At similar nicotine concentrations in the THS 2.2 aerosol and reference cigarette smoke, the average reduction in the formation of harmful and potentially harmful constituents in THS 2.2 aerosol compared with constituents in reference cigarette smoke was 95%. 41 Pronounced alterations in culture morphology were seen following exposure to reference cigarette smoke but not to THS 2.2 aerosol (Figure 3). Levels of pro-inflammatory mediators, such as VEGFA, TIMP1, MMP-1, CXCL8, CXCL10, IL-6, IL1β, and CSF2, were much lower in cultures exposed to THS 2.2 aerosol than in cultures exposed to cigarette smoke. The THS 2.2 aerosol exposure elicited differential regulation of fewer genes than did the cigarette smoke exposure, and this differential regulation was mostly limited to the earliest postexposure time points assessed, suggesting a lower and more transient response to the exposure. 42 –44
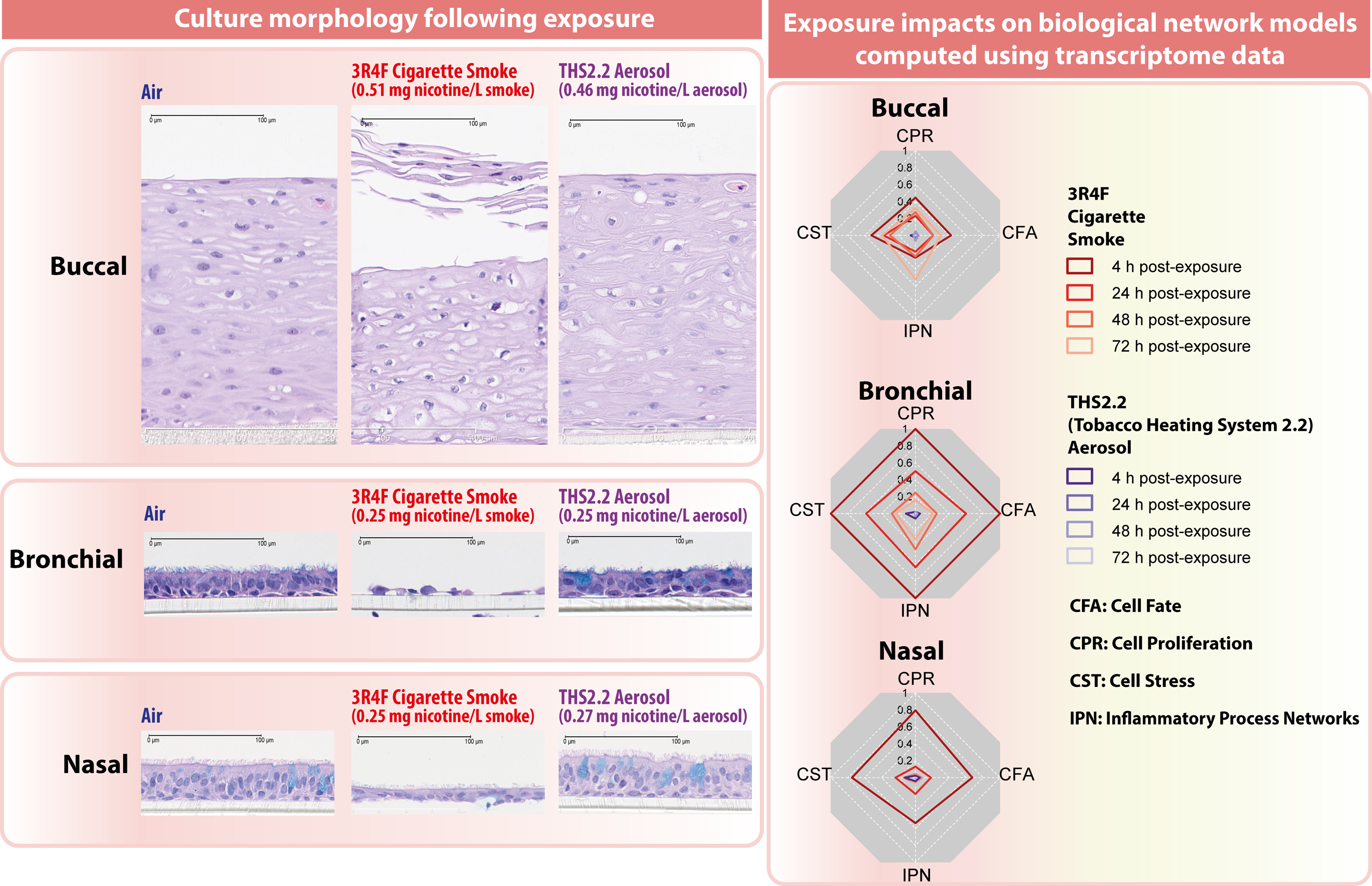
Systems toxicology approach for an in vitro assessment of cigarette smoke and THS 2.2 aerosol exposures. Representative images showing the histology sections of human organotypic buccal, bronchial, and nasal cultures following exposure to cigarette smoke or THS 2.2 aerosol (left panel). Biological impact scores shown as radar charts reflecting the perturbation scores of biological network model families (cell fate, cell proliferation, cell stress, and inflammatory process network) following exposure to cigarette smoke or THS 2.2 aerosol (right panel). Adapted from a previous publication. 42 THS: Tobacco Heating System.
These in vitro studies were conducted following a systems toxicology approach, allowing us to detect the underlying perturbation of cellular and molecular signals that would not be evident morphologically. Systems toxicology integrates traditional laboratory testing with quantitative modeling of large data sets to identify the biological networks affected by an exposure and the molecular pathways involved and to infer the adverse outcomes. 43 The approach involves a robust experimental design to generate reproducible empirical global gene expression data and to identify and quantify the biological networks perturbed by the exposure (Figure 3). In all cultures tested (nasal, buccal, and bronchial), similar molecular entities and networks were perturbed; these were the networks governing cell fate, cell proliferation, cell stress, and inflammatory processes. The xenobiotic metabolism response was elicited by exposure to cigarette smoke but had only a minor and mainly transient impact on cultures exposed to THS 2.2 aerosol. 42 –44
A similar approach to that used for THS 2.2 could be leveraged for assessing electronic cigarettes (e-cigarettes), one type of electronic nicotine delivery system. The popularity of e-cigarettes has grown tremendously in recent years, but assessing the potential toxicity of e-cigarettes presents multiple challenges involving a lack of standards in (1) analytical methods, (2) the testing of flavoring compounds, (3) aerosol generation protocols, and (4) selecting the chemicals to monitor. 45,46 Multiple in vitro studies have compared biological responses in human bronchial epithelial cultures exposed to reference cigarette smoke and to e-cigarette vapor and found reduced cytotoxicity in human cell cultures exposed to e-cigarette vapor 47,48 as well as a reduced impact of gene expression 49 ; these limited effects are believed to result from the lower temperatures and lack of combustion. 50 In comparison with cigarette smoke, e-cigarette vapor has been shown to elicit only limited mutagenicity, 51 although it can induce measurable levels of oxidative stress and inflammatory mediators, 48 and the composition of added flavoring compounds may modulate toxic responses. 52 Nevertheless, doses that are physiologically relevant and reflect real-world use should be assessed to better predict the potential toxicity of e-cigarette use. Furthermore, in the context of harm reduction, e-cigarette assessment should include a comparison with a combustible tobacco product. 53
PMI has established the INTERVALS platform (http://intervals.science) to demonstrate the scientific rigor, thoroughness, precision, and transparency required to assess the inhalation toxicology of potential reduced-risk products in support of designing a smoke-free future. The platform promotes independent, third-party analyses and collaborations by sharing protocols, data, and tools. Raw data, reports, and detailed laboratory protocols from preclinical and clinical studies of potential reduced-risk products are shared freely. With the rapid evolution of heated tobacco products and e-cigarettes, collaborative efforts between the scientific community, industry, and regulatory stakeholders are needed. Such efforts will also facilitate the adoption of 21st-century approaches for toxicity testing.
New tiered risk assessment approaches of mixtures
The challenges in assessing the risk of exposure to mixtures involve consideration of aggregate risk (exposure to a single chemical from multiple sources), cumulative risk (exposure to multiple agents), and the interaction of exposures to multiple chemicals that, in combination, generate risk other than the sum of their single-agent exposure risks. Assessing whole mixtures involves toxicity profiles, epidemiological investigations, and generating hazard quotients.
In cases where the mixture’s mode of action is unknown, information on the mode of action of each constituent is gathered, along with consideration of structure–activity similarities and possible interactions. The US Environmental Protection Agency (EPA) (2000) 54 and European Union (2011) 55 have issued guidance documents on evaluating chemical mixtures. Both texts emphasize development of an assessment on the basis of whether information is available on the mixture of concern. In the EPA guidance, this question is extended to mixtures that are sufficiently similar. If information is not available on the mixture or a sufficiently similar mixture, both documents emphasize constituent-based assessment. When evaluating individual components, both guidance documents consider whether chemical interactions are suspected between constituents. If they are, a case-by-case assessment that is based on the observed synergism or antagonism is generally recommended. If interactions are not suspected, an additivity assessment is conducted by either dose addition, if the modes of action are similar, or by response addition, if the modes of action are dissimilar.
Data-driven approaches to mixture assessment leverage information on exposure, dose–response, and mode of action. When this information is available for the mixture being studied, the mixture is considered as a whole, and toxicity values, such as cancer slope factors, minimum risk levels, and reference doses and concentrations, can be estimated. When information on the mixture, or in the case of EPA a similar mixture, is not available, a component-based approach is used instead, and the toxic effects and mode of action of each component of the mixture are compared.
These approaches are based on tiered assessments that consider exposure and hazard in an integrative and iterative manner in each tier. Meek and colleagues 56 have proposed a four-tier framework that can be used in both mixture- and component-based investigations. Each of the four tiers proposed includes exposure assessment, hazard evaluation, and risk characterization, and each tier provides a more refined and specific assessment than the preceding tier (Figure 4). Information from different levels of biological organization can be incorporated into the model as well as analyses of factors such as mode of action and species concordance. 57
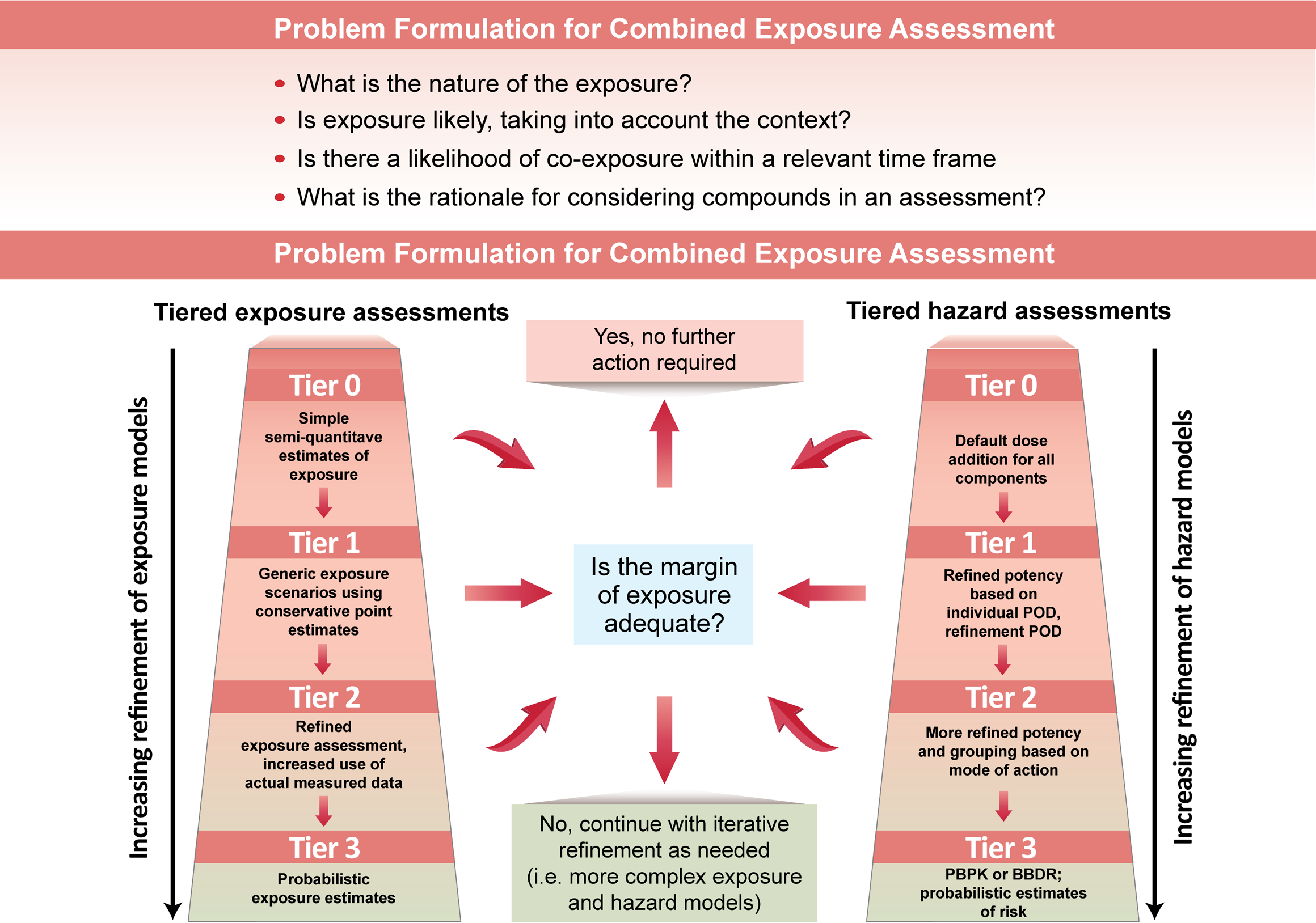
Tiered framework for exposure assessment, hazard evaluation, and risk characterization. A conceptual representation of a tiered framework, reproduced from a previous publication. 57
Another multistep approach to mixture evaluation is the dose additivity combined prediction model proposed by Hertzberg and colleagues. 58 If the mixture’s components are known to be toxicologically similar, if their dose–response curves are geometrically congruent, and if the predicted mixture response follows that of each component, the mixture is treated as dose-additive and can be represented as a linear combination of its constituents’ doses. To evaluate dose additivity in a specific mixture in this four-step approach, the assessment considers how well the model fits the data on constituent chemicals (step 1) and the data on the mixture as a whole (step 2), the agreement between the model and the mixture data (step 3), and the consistency between the model and other mixture models (step 4). 58
High-throughput screening can provide information on biological activity that, when combined with chemical characterization, enables the implementation of similarity approaches and prototype mixtures for hazard assessment. 59 Because in vivo testing in mammals is not logistically feasible, high-throughput testing in alternative model organisms, such as zebrafish, could be integrated into risk assessment.
Both in vitro and in silico comparative approaches begin with analytical characterization of mixture composition and toxicity. Each identified constituent is processed through a decision tree to close safety gaps, inform supportable exposure levels, and determine the need for safety studies. The variables involved, biological activity, and mode of action can be estimated. New methods offer a significant improvement in assessing the hazards of chemical mixtures. Coupled with appropriate exposure determination, these in vitro modeling tools can promote a credible estimate to the in vivo toxicity outcome.
Summary
Evaluating the risk from chemical mixtures is a multifaceted task that presents multiple challenges and requires an appropriate assessment framework. Depending on the available data, whole mixture approaches or component-based approaches, or a combination of both, can be used. In silico modeling and decision trees can facilitate the risk assessment of mixtures. In vivo testing using animal models is not always logistically feasible, but high-throughput testing in alternative models, such as organotypic in vitro models and zebrafish, can be integrated into the assessment. Apart from classical toxicology testing methods, risk assessment can further leverage omics approaches such as transcriptomics, proteomics, and metabolomics to reveal the mode of action. In addition, a network-based systems toxicology approach, which utilizes quantitative modeling of data sets to identify the biological networks perturbed by a given exposure and the underlying molecular pathways involved, will facilitate the biological interpretation of omics data. Finally, various tiered assessment strategies have been proposed that incorporate laboratory data and computational modeling for robust investigation into the risk posed by chemical mixtures. These emerging tools and protocols will lead to a significant improvement in the hazard assessment of complex mixtures and should continue to promote credible protection of public health.
Footnotes
Authors’ note
Some of the information in this review was previously presented at the Eighth International Congress of the Asian Society of Toxicology (ASIATOX) in Pattaya, Thailand, on 18 June 2018.
Acknowledgements
The authors thank Dean Meyer, PhD, of Edanz Medical Writing for providing medical writing support, which was funded by Philip Morris International.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JH, AI, RL are employees of Philip Morris International.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
