Abstract
Electronic nicotine delivery systems (ENDSs; e.g. e-cigarettes) are being developed as potentially reduced-risk alternatives to the continued use of conventional tobacco products. They typically comprise a device that heats an e-liquid to generate inhalable vapor. E-liquids and ENDS-derived vapor have been the focus of toxicological assessment; in particular, their DNA-damaging potential has been investigated with varying outcomes and conclusions. In vitro genetic toxicology assays have formed a part of these assessments. However, they are susceptible to producing misleading or false positive results, especially under extreme conditions. In the present study, we evaluated a series of six neat (non-vaporized) non-flavored e-liquids (NFEL-A to F) in a flow cytometry version of the in vitro micronucleus assay in order to characterize their baseline effects on Chinese hamster ovary cells under hazard identification conditions. The NFELs induced cytotoxicity universally despite differing in propylene glycol (PG), vegetable glycerin (VG), and nicotine content. In addition, significant genotoxic responses were also detected with the PG-predominant e-liquids NFEL-A, D, and F but not with NFEL-B, C, or E, which contained higher proportions of VG. All six NFELs induced extreme cell culture conditions (i.e. increases in pH and osmolality) at the concentrations assessed. They also exhibited nonbiologically relevant effects on the mechanistic endpoints (i.e. cell cycle and phosphorylated histones H2AX and H3). In conclusion, although the PG component of the NFELs drove micronucleus formation in the assay, data on the complementary mechanistic endpoints suggest that this apparent DNA damage is potentially misleading and of negligible biological relevance as a risk for DNA integrity. In future assessments, any adverse changes (such as signatures of micronuclei induction, G2M arrest, and increases in γH2AX) relative to this reference data set might indicate a possible genotoxic hazard and would prompt further investigations for exploring the extent of risk.
Introduction
Electronic nicotine delivery systems (ENDSs; e.g. e-cigarettes) are being developed as potentially reduced-risk alternatives to the continued use of conventional tobacco products. 1,2 They consist of a battery-powered device that heats an “e-liquid” contained inside an atomizer; this interaction leads to the generation of inhalable vapor upon puffing by the user. 3 E-liquids can contain various levels of nicotine, flavoring agents, and excipients such as propylene glycol (PG) and vegetable glycerin (VG). 4 Because of the recent rise in the use of ENDS among the global population, the toxicity of ENDS-derived vapors has been the focus of much attention. 5,6 In particular, their DNA-damaging potential and, by inference, tumor-initiating capacity have been investigated both in vitro and in vivo. These studies have reported varying findings and drawn dichotomous conclusions (both genotoxic and non-genotoxic); but, in the case of positive results, the most frequently cited genotoxic mechanism is related to oxidative stress. 7 –13 However, certain groups of flavor chemicals used in e-liquids as well as in food and other consumer products (e.g. α,β-unsaturated aldehydes and alicyclic or heterocyclic alpha-ketoenols) possess genotoxic profiles in vitro; these may, at least in part, be responsible for the observed positive effects. 14 But, the risk of these agents to human health, albeit when used in accordance with their specified intended purpose (i.e. to be ingested), has been judged by several authoritative bodies to be of negligible concern, potentially because of the low levels of exposure to these agents, their efficient detoxification (e.g. through β-oxidation), or their presumably thresholded mode-of-action (MoA; e.g. reactive oxygen species formation). 14 –16
Besides being overly sensitive relative to in vivo models, in vitro genetic toxicology assays are also susceptible to producing false results, especially false positive (genotoxic) ones, under “extreme” conditions. For example, in the mouse lymphoma assay, induction of high levels of cytotoxicity by chemicals such as 2,4-dichlorophenol and phthalic anhydride can lead to apparent mutation of the thymidine kinase gene. 17 In assays that measure clastogenicity (e.g. the in vitro micronucleus assay), the genotoxic effects observed in the presence of appreciable cytotoxicity can be misinterpreted as being biologically relevant, although efforts have been made to refine cytotoxicity limits and indices in order to minimize the likelihood of this scenario. 18 Furthermore, a plethora of other factors, including high test concentrations (>10 mM), liver microsome activation systems, pH, osmolality, and lysosomal nuclease release, has been shown to possess the potential to confound the performance and predictivity of in vitro assays that detect genetic damage. 19,20 Interestingly, the chemical foundations of e-liquids, that is, PG, VG, and nicotine, have the potential to induce extreme culture conditions such as increases in osmolality and pH, which may lead to assay interference. 21,22 These changes are predicted to be particularly extensive when mixtures of these agents are assessed in a hazard identification context, that is, at concentrations that produce cytotoxicity up to the limits of the assay in question.
With the goal of understanding the effects of neat (non-vaporized) non-flavored e-liquids (NFELs) in the in vitro micronucleus assay vis-à-vis the potential extreme culture conditions they induce, we conducted the flow cytometry version of the assay in Chinese hamster ovary (CHO) cells using six NFEL variants in general accordance with the recommendations set out in Organisation for Economic Co-operation and Development (OECD) Test Guideline 487. 23 We sought to establish this “baseline” of effects that could serve as a reference point for assessing flavored e-liquids and, ultimately, the collected vapor of aerosolized e-liquids using the same approach. Based on the findings of the in vitro micronucleus assay, we performed additional mechanistic experiments to facilitate further comprehension of the various cellular responses to NFELs, given the importance of these types of data to provide biological context.
Materials and methods
Chemicals
The NFELs were manufactured by Philip Morris International R&D (Switzerland) using the following chemical components: PG (BASF, Germany; CAS 57-55-6), VG (Emery Oleochemicals, Malaysia; CAS 56-81-5), nicotine (-/L enantiomer; Siegfried, Switzerland; CAS 54-11-5), and water (Merck, Switzerland). NFEL composition is specified in Table 1. All other chemicals were obtained from Sigma-Aldrich (Switzerland) unless otherwise specified.
Chemical composition of the NFELs.
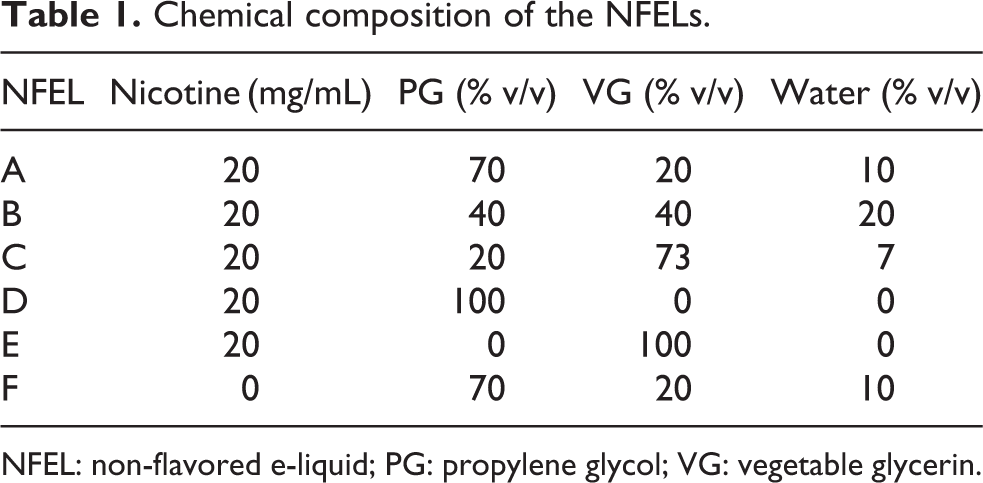
NFEL: non-flavored e-liquid; PG: propylene glycol; VG: vegetable glycerin.
Cell culture
The CHO-Wolff–Bloom–Litton (WBL) cell line was obtained from Merck Research Laboratories (West Point, Pennsylvania, USA) and certified mycoplasma-free. 24 Briefly, cells were routinely cultured in a humidified incubator at 37°C in an atmosphere of 5% carbon dioxide (CO2) and used between passages 2 and 5 in a given assay. McCoy’s 5A + GlutaMAX™ medium (Thermo Fisher Scientific, Switzerland) supplemented with fetal bovine serum (10% v/v; Thermo Fisher Scientific), penicillin (100 U/mL), and streptomycin (100 μg/mL) was used for culture maintenance as well as the in vitro micronucleus assay.
In vitro micronucleus assay
On two-three independent test occasions, CHO-WBL cells were seeded (4500 cells/well) into transparent, flat-bottom 96-well plates (Nunc™, Thermo Fisher Scientific) and cultivated for 24 h. Duplicate cultures were then exposed to a range of NFEL concentrations (1.6–4.0% v/v) as well as the positive controls methyl methanesulfonate (MMS; 15 and 20 µg/mL) and colchicine (0.15 and 0.2 µg/mL) for 24 h in the absence of S9 at 37°C and 5% CO2 in a humidified environment. Note that only long-term treatment, and not short-term treatment ±S9, was carried out in this study as it was deemed the most optimal condition in which to study genotoxicity vis-à-vis extreme culture conditions. After this period (and also immediately prior to treatment; see below for more information), nuclei species—including 2n–4n nuclei, apoptotic nuclei, hypodiploid nuclei (HDN), polyploid nuclei, and micronuclei—were harvested using the In Vitro MicroFlow® Kit (Litron Laboratories, Rochester, New York, USA) in general accordance with the manufacturer’s instructions. 25 This approach elucidates cell cycle changes and, in parallel, omits fragmented DNA from apoptotic cells from micronuclei enumeration (which may confound the analysis). 26 A minor adaptation of the manufacturer’s instructions was implemented in order to permit the measurement of an OECD Test Guideline 487-recommended cytotoxicity index (i.e. relative population doubling (RPD)) rather than other non-recommended indices such as relative survival. Ten thousand 6-µm Cell Sorting Set-up Beads (Thermo Fisher Scientific) were added to each well and used in conjunction with other flow cytometry acquisition data ((nuclei events × 10,000)/bead events) to determine the absolute number of nuclei/well both immediately prior to and following treatment. These values were ultimately used to produce the RPD index of cytotoxicity applied in this study. A FACSCanto II flow cytometer (BD Biosciences, San Jose, California, USA) equipped with FACSDiva software (v8.01, BD Biosciences) was used to enumerate the frequency of each nuclei species. Concentration-response data were analyzed statistically through: (1) the Dunnett’s pairwise test; (2) the Mann–Kendall trend test; and (3) a comparison against the 95% upper control limit of the laboratory’s historical solvent-treated control database (SAS® Enterprise Guide® v6.1, USA).
Assessment of extreme culture conditions
The pH and osmolality of each concentration of the NFEL dosing medium used in the in vitro micronucleus assay were measured using a pH meter (S40 SevenMulti™; Mettler Toledo, Switzerland) and osmometer (OsmoPRO® Multi-Sample Micro-Osmometer; Advanced Instruments, Norwood, Massachusetts, USA), respectively.
DNA damage response
In order to investigate the effects of NFELs on DNA damage response (DDR) pathways, CHO-WBL cells were seeded, cultured, and exposed to NFELs as in the in vitro micronucleus assay. However, nuclei were harvested at 4- and 24-h time-points using the MultiFlow™ DNA Damage Kit—p53, γH2AX, and Phospho-Histone H3 (Litron Laboratories) and analyzed in accordance with the manufacturer’s instructions. It should be noted that the p53 antibody contained in the kit is specific for the human p53 protein; thus, p53-related data were not considered in this study. Briefly, following aspiration of the cell culture medium at the two time-points, Accutase® was added to each well (25 µL), and the plates were incubated for 10 min at room temperature to induce cell detachment. A 50-µL solution containing all kit components and fetal bovine serum (1.9% v/v) was prepared and applied to each well, and the plates were incubated for 30 min at room temperature in the dark in order to allow nuclei liberation and labeling. Nuclei were transferred to transparent U-bottom 96-well plates (Nunc™, Thermo Fisher Scientific), and the FACSCanto II system was used to quantify and extract median allophycocyanin fluorescence values (γH2AX-associated) and determine the proportion of cells expressing phosphoserine 10 -H3 (pH3) from the total nuclei population. Although evaluation criteria for DDR endpoints in CHO cells have not yet been developed, we used data from two previous studies to guide our interpretation (biologically relevant or not) of the current data set. 27,28
To investigate the capacity of nicotine specifically to modulate the DDR to genotoxic agents, MultiFlow™ analysis was performed as described earlier except for the use of CHO-WBL cells co-exposed to nicotine (concentration range: 50–800 µg/mL (0.31–4.94 mM)) and either MMS (25, 30, and 35 µg/mL; 4- and 24-h time-points) or vinblastine (VIN; 0.2, 0.4, and 0.6 µg/mL; 4-h time-point). The t-test (GraphPad Prism® v5.04, GraphPad Software Inc., La Jolla, California, USA) was then used to compare statistically genotoxin-induced changes in γH2AX-, pH3-, and G2M-accumulated cells in the presence and absence of nicotine.
Results
Significant micronuclei and HDN formation in response to three NFELs but not the others
At the concentration ranges employed in the study, all six NFELs induced a full spectrum of cytotoxicity from minimal up to and just beyond the maximum recommended level for the in vitro micronucleus assay (RPD 50–40%; Figure 1(a)). While all six NFELs induced cytotoxicity, only NFEL-A, NFEL-D, and NFEL-F induced marked concentration-dependent increases in both micronuclei and HDN endpoints. On the basis of the statistical analysis, the latter three were classified as genotoxic in the study (Figure 1(b) and (c)). In contrast, although subtle concentration-dependent increases in micronuclei were observed in response to NFEL-B, NFEL-C, and NFEL-E, these increases were ultimately determined to reach nonsignificant genotoxic levels in accordance with the evaluation criteria recommended in OECD Test Guideline 487 (Figure 1(b)). However, despite inducing appreciable cytotoxicity, NFEL-exposed cells exhibited negligible cell cycle effects relative to non-treated controls; in contrast, cells exposed to the positive controls (MMS and colchicine) showed G2M phase accumulation and polyploid nuclei (see the Online Supplementary Figure A.1 to A.7).
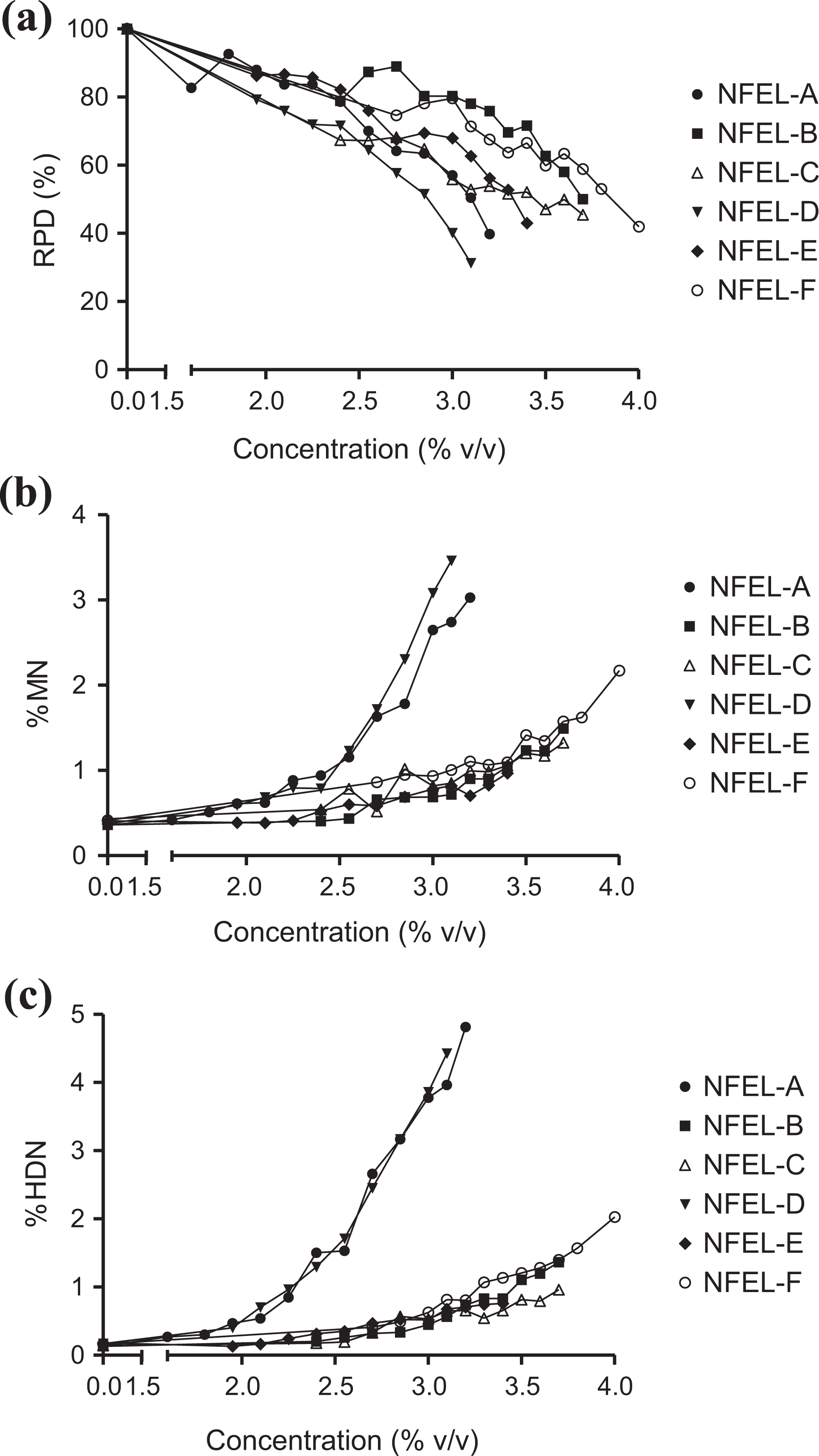
NFEL-induced effects in CHO-WBL cells following 24 h exposure, as measured by the flow cytometry-based in vitro micronucleus assay. (a) RPD, (b) MN, and (c) HDN. Note: Error bars have been omitted from the graphs in order to improve legibility; however, levels of variability across the data set were <100% when calculated via the relative standard deviation metric. NFEL: non-flavored e-liquid; CHO-WBL: Chinese hamster ovary–Wolff–Bloom–Litton; RPD: relative population doubling; MN: micronuclei; HDN: hypodiploid nuclei.
Induction of extreme culture conditions by NFELs
Concentration-dependent increases in pH and osmolality were observed across the different concentrations of NFEL dosing medium. All NFELs, including NFEL-F (nicotine-free), generally increased the pH by approximately one unit relative to non-treated cell culture medium (average: pH 7.48; Figure 2(a)). Similarly, all six NFELs increased osmolality to maximal levels between 741 mOsm/kg and 1015 mOsm/kg H2O compared with an average of 298 mOsm/kg H2O for non-treated cell culture medium (Figure 2(b)).
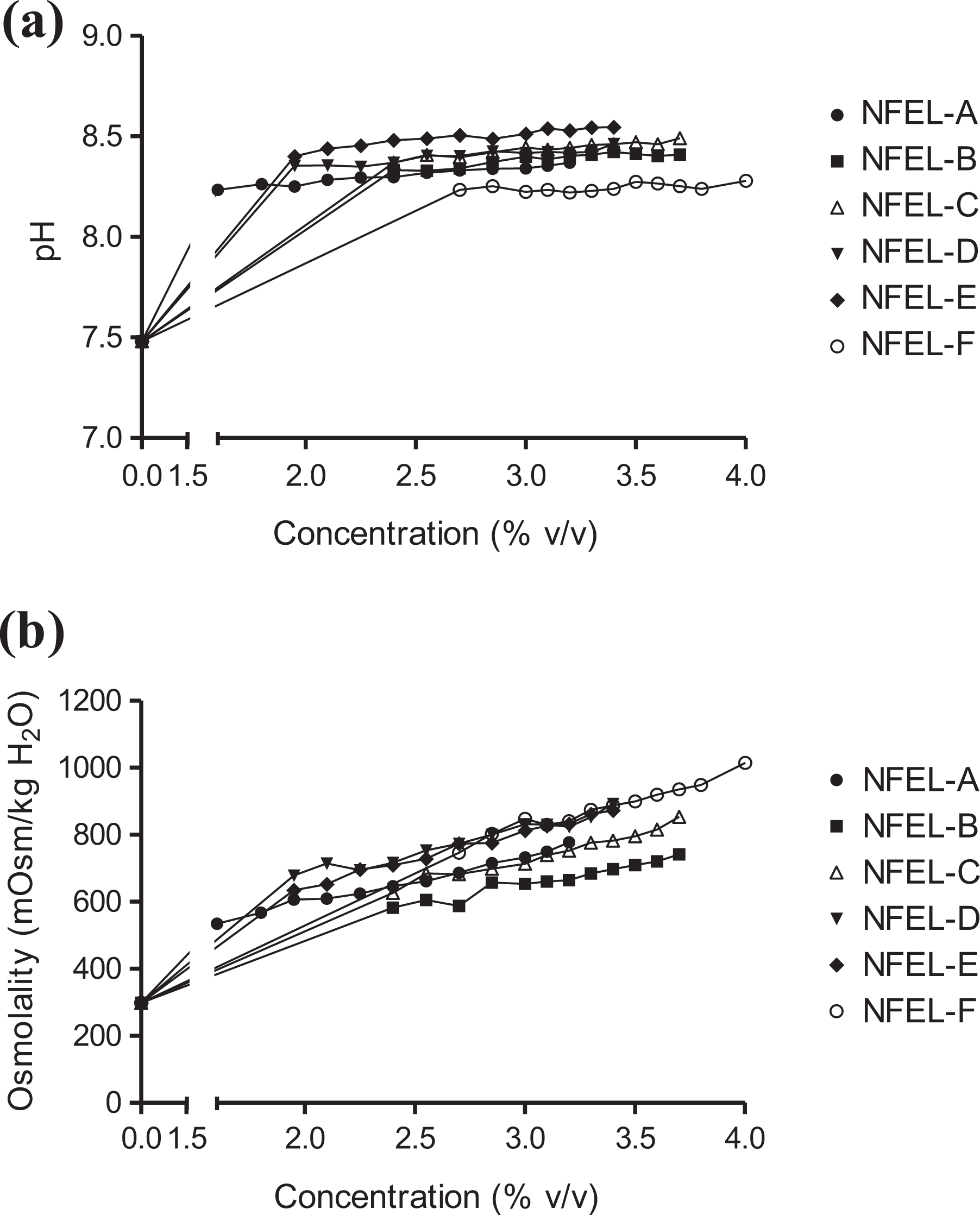
NFEL-induced changes in cell culture conditions. (a) pH and (b) osmolality. Note: Error bars have been omitted from the graphs in order to improve legibility; however, levels of variability across the data set were <13% when calculated via the relative standard deviation metric. NFEL: non-flavored e-liquid.
DDR pathway activation by NFELs
CHO-WBL cells exposed to the same NFEL concentrations as those in the in vitro micnucleus assay were subjected to a MultiFlow™ assay at two time-points: 4- and 24-h. At both time-points, ostensibly nonbiologically relevant changes (direction and/or magnitude) were detected in γH2AX and pH3 levels in response to NFEL-A, NFEL-B, NFEL-C, NFEL-D, and NFEL-F (see the Online Supplementary Figure B.1 to B.4 and B.6). In response to NFEL-E, γH2AX concentrations at the 4-h time-point were elevated up to a biologically relevant level (average: 1.59-fold increase); however, the levels at the 24-h time-point were similar to those in non-treated controls (see the Online Supplementary Figure B.5). The positive controls, on the other hand, induced changes (at both time-points) in γH2AX and pH3 levels that were biologically relevant and commensurate with their genotoxic MoA (see the Online Supplementary Figure B.7 and B.8).
Continued γH2AX and pH3 expression as well as G2M arrest in cells co-exposed to nicotine and genotoxins
To investigate the capacity of nicotine to mask the DDR induced by genotoxic agents, cells co-exposed to nicotine and MMS or VIN were assessed in the MultiFlow™ assay. Cells exposed to MMS or VIN alone expressed biologically relevant levels of γH2AX (4- and 24-h time-points) and pH3 (4-h time-point), respectively, as expected (Figure 3(a) to (c)). Although some sporadic statistically significant decreases were detected, cells co-exposed to MMS and a range of nicotine concentrations (50–800 µg/mL) continued to express biologically relevant levels of γH2AX at both time-points, relative to the corresponding no-nicotine controls (Figure 3(a) and (b)). pH3 expression was also induced after 4 h in the presence of VIN and 50–400 µg/mL nicotine; however, at the two higher nicotine concentrations (600 and 800 µg/mL), pH3 levels were both reproducibly and statistically significantly suppressed relative to those observed in cells exposed only to VIN (Figure 3(c)). In addition, MMS- and VIN-treated cells and the corresponding co-exposed cultures showed similar cell cycle arrest profiles (i.e. G2M accumulation) at the two time-points (4-h: VIN; 24-h: MMS) despite a number of statistically significant differences (Figure 4(a) and (b)).
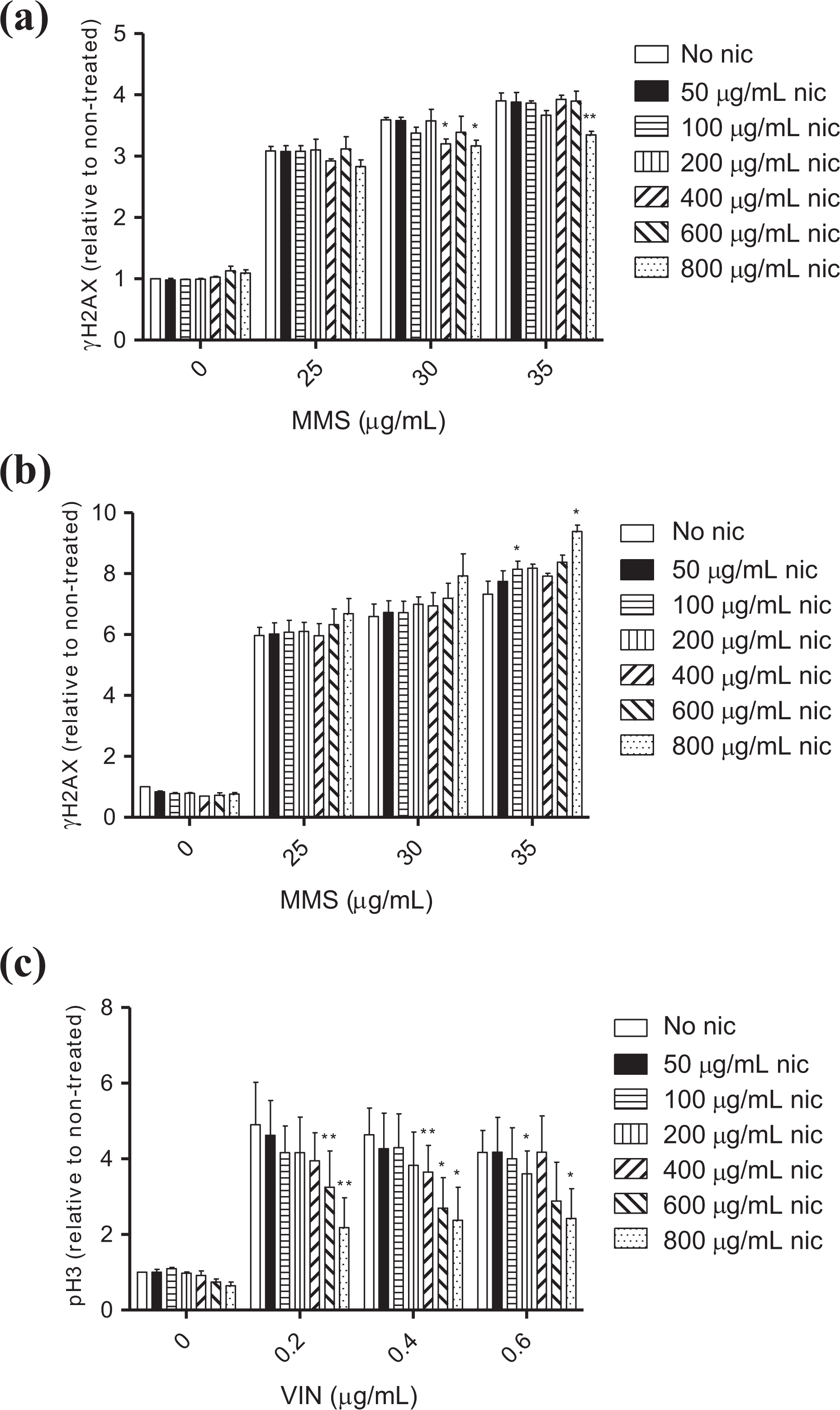
Effects of nicotine on genotoxin-induced DNA damage response signaling (relative change compared to controls ± standard error). MMS-induced changes in γH2AX levels at the (a) 4-h and (b) 24-h time-points. VIN-induced changes in pH3 levels at the (c) 4-h time-point. *p < 0.05 and **p < 0.01—statistically significant difference relative to its respective no-nicotine control (t-test). nic: nicotine; MMS: methyl methanesulfonate; VIN: vinblastine.
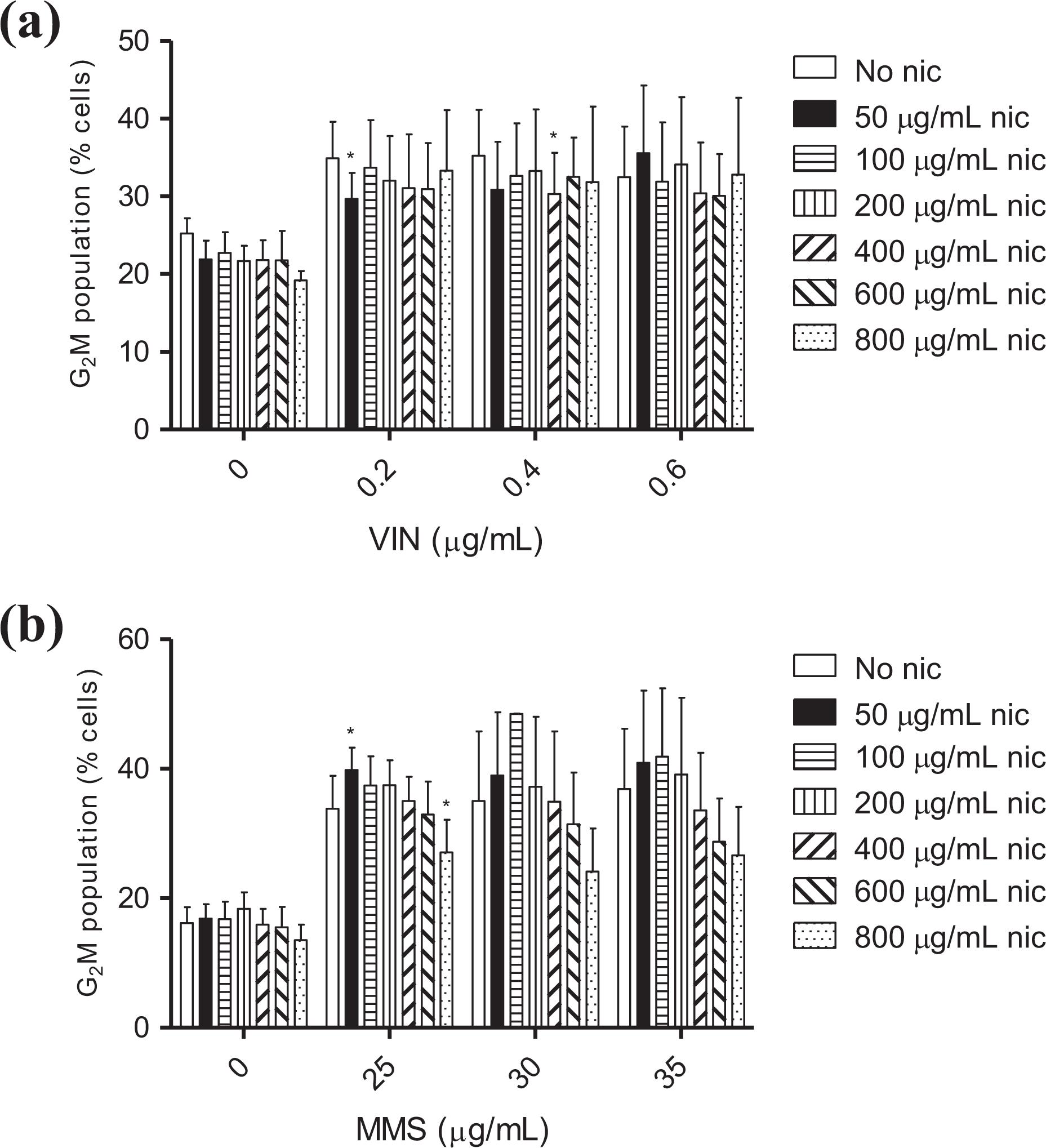
Effects of nicotine on genotoxin-induced cell cycle arrest (relative change compared to controls ± standard error). VIN- and MMS-induced G2M arrest at the (a) 4-h and (b) 24-h time-points, respectively. *p < 0.05—statistically significant difference relative to its respective no-nicotine control (t-test). nic: nicotine; MMS: methyl methanesulfonate; VIN: vinblastine.
Discussion
We studied the effects of NFELs in the in vitro micronucleus assay in order to establish their baseline characteristics. The NFELs assessed in this study reflected some of the diversity found in commercially available e-liquid base formulations. 29 Importantly, this study also took into consideration the capacity of NFELs to induce extreme culture conditions in terms of changes to cell culture medium pH and osmolality. As expected under the hazard identification testing approach, NFEL-induced cytotoxicity was universally observed up to the limits of the assay. In addition, significant genotoxic responses were also detected with NFEL-A, NFEL-D, and NFEL-F; these NFELs induced concentration-dependent increases in both micronuclei and HDN endpoints, a profile that is consistent with an aneugenic MoA. 30 Furthermore, as NFEL-A, NFEL-D, and NFEL-F contained at least 70% v/v PG, we deduced that the genotoxic responses in the assay were likely driven by the PG component in the mixtures. In contrast, the NFELs that did not induce significant genotoxicity were composed of only 40% v/v PG or were predominantly VG-based. Although it has a “generally recognized as safe” status and is non-mutagenic in the Ames test, 31,32 PG induces apparent genotoxicity at high concentrations in mammalian assays in vitro. Ishidate et al. attributed its effects on chromosomal aberrations in hamster lung fibroblasts to an indirect MoA through extreme changes in culture medium osmolality. 21 While Aye et al. made no mention of the MoA, they found that PG induced DNA damage in CHO-K1 cells in comet and micronucleus assays. 33 In the present study, all six NFELs induced concentration-dependent increases in the osmolality and pH of the dosing medium. Thus, although the induction of extreme culture conditions by the PG-predominant NFELs may contribute to their apparent genotoxic effects, it was not the changes in osmolality (or pH) per se that drove these responses; if that were the case, these changes would have been observed for all NFELs. Importantly, these observations confirm the previous findings on aliphatic alcohols, although it is still not clear precisely which MoA drives PG-induced DNA breakage in chromosome damage assays. 21,34 In addition, while these three NFELs induced apparent aneugen-like increases in micronuclei and HDN populations, the cell cycle profiles derived concurrently from the assay did not corroborate these data. In higher content assays, genuine genotoxicity induced by mutagens, clastogens, and aneugens typically manifests in a signature of DNA damage and G2M cell cycle arrest, or even endoreduplicated (8n/polyploid) nuclear DNA. 25,35 Significantly, while the positive control chemicals (MMS and colchicine) used here demonstrated these effects, none of the NFELs induced this type of profile, even at high levels of cytotoxicity. Thus, it is interesting to speculate that the PG-predominate NFELs induce a form of DNA fragmentation, possibly through cell death, which manages to escape detection as such and, hence, why G2M cell cycle arrest is not triggered. 36
To understand further the NFEL-induced effects in the in vitro micronucleus assay as well as their potential MoA, we probed two of the key phosphorylated proteins involved in the cell’s DDR to genotoxic stress. Using the MultiFlow™ assay, we assessed γH2AX (a biomarker of clastogenicity) and pH3 (a biomarker of microtubule-mediated aneugenicity) endpoints at two time-points: 4- and 24-h post-NFEL exposure. Although both biomarkers exhibited changes, neither the direction nor magnitude of change was consistent with the biologically relevant effects of true genotoxins. 27,28,35 Only with NFEL-E was there an elevation in γH2AX level (up to 1.59-fold) at the 4-h time-point. However, it returned to a control-like level at 24 h; therefore, the early response was not considered to be meaningful in the context of this study, especially also given its lack of micronuclei effects. Thus, the results of these follow-up DDR-related experiments—like the cell cycle data—contradict the in vitro micronucleus assay findings and might indicate that the increases in micronuclei and HDN observed here in response to NFEL-A, NFEL-D, and NFEL-F are not biologically relevant forms of genotoxicity. Interestingly, a recent comprehensive in vitro assessment of the genotoxic hazard potential of nicotine made similar observations on the same endpoints. 37
Although DDR biomarkers such as γH2AX and pH3 are being increasingly used in genetic toxicology assessments, the reliability of these endpoints is entirely dependent on the fidelity of the signaling pathways that underpin them. 28,36,38 –42 In the present study, owing to the contradictory nature of the In vitro MicroFlow™ and MultiFlow™ findings, we investigated whether the pharmacologically active NFEL component nicotine could modulate DDR signaling pathways and mask cellular responses to genotoxic stress. Essentially, we attempted to understand if the use of DDR endpoints and cell cycle changes for e-liquid genotoxicity assessment is scientifically justifiable as, for example, nicotine has been reported to override cell cycle checkpoints in response to DNA damage. 43 Importantly, CHO-WBL cells co-exposed to nicotine and MMS continued to show γH2AX expression and G2M arrest, indicating that nicotine does not significantly interfere with the underlying signaling pathways of these cellular responses. Cells exposed to both nicotine and the tubulin poison VIN also continued to express pH3 and exhibit G2M arrest; however, the levels of pH3 were markedly decreased after exposure to the two highest concentrations of nicotine (600 and 800 µg/mL), possibly because of nonspecific effects on the kinases responsible for H3 phosphorylation. These data suggest that nicotine can suppress H3 phosphorylation at high concentrations. However, at the lowest effective concentrations of NFEL-A and NFEL-D (i.e. approximately 2.2% v/v) at which induced micronuclei consistently start to diverge from background levels, the nominal concentration of nicotine is estimated to be 440 µg/mL. Importantly, according to our data, nicotine is not anticipated to interfere with H3 signaling at this concentration. These conclusions on nicotine are further supported by our results which reveal that the nicotine-free mixture NFEL-F induced cellular responses consistent with its nicotine-containing counterpart NFEL-A, albeit with reduced potency. Thus, taking all the MultiFlow™ data together, the negligible effects on γH2AX and H3 phosphorylation observed here are likely to be an accurate representation of (the lack of) DDR to NFELs. Similarly, the absence of cell cycle changes following NFEL exposure indicates that this response is also genuine, as nicotine was found not to perturb cell cycling notably during induction of genotoxic stress by MMS and VIN.
A deeper analysis of the published concentration–response genotoxicity data on PG also revealed quantitative support for the micronucleus data generated on the PG-predominant NFELs in this study. For NFEL-A, NFEL-D, and NFEL-F, the nominal levels of PG calculated to be present in the dosing medium at the lowest effective concentrations are in the range of 152–544 mM. Therefore, the responses they induced are wholly consistent with those observed in two previous studies that reported PG genotoxicity at concentrations between 206 mM and 420 mM. 21,33 While these concentrations far surpass the concentrations limits (1 or 10 mM) applied when evaluating individual pharmaceuticals or chemicals, 44 –46 it is important to recognize that NFELs and e-liquids, in general, are chemical mixtures, and recommended concentration limits for such items are not so well defined; testing to the limits of cytotoxicity (or solubility) is, therefore, implied. However, we have shown that by complementing a conventional genotoxicity endpoint like micronucleus with MoA endpoints (such as DDR signaling proteins and cell cycle status), the data can be potentially better understood and rationalized, even in a limited cell line like CHO-WBL. Our recent findings (including the work of Smart et al. 37 ) show that, in the absence of changes in cellular DNA content (e.g. G2M accumulation and endoreduplicated DNA), increases in the HDN endpoint alongside those in micronuclei might be indicative of a misleading (false positive) genotoxic response, at least in this cell model. Therefore, results demonstrating such a profile should raise fewer concerns than those exhibiting the full signature of effects. Importantly, this more holistic, biology-centric approach to genotoxicity assessment could be another strategy for reducing the number of artifactual positive results generated in the standard battery of in vitro genetic toxicology assays and, consequently, avoiding unnecessary follow-up in vivo tests. 47 –49
Conclusions
The present study has characterized the effects of NFELs in the CHO-WBL cell-based in vitro micronucleus assay when assessed under hazard identification conditions. The PG component of the NFELs was found to drive micronucleus formation in the assay. However, the data on the complementary endpoints (i.e. DDR and cell cycle analyses) suggest that this apparent DNA damage is potentially misleading and of negligible biological relevance as a risk for DNA integrity as none of these fundamental biological responses to genotoxic stress was triggered (summarized in Figure 5). In future e-liquid-related genotoxicity assessments, any adverse changes—such as signatures of micronuclei and HDN induction, G2M arrest, endoreduplicated DNA, and increases in γH2AX or pH3 levels—relative to this reference data set may indicate the presence of a possible genotoxic hazard and would prompt further investigations for exploring the extent of risk.
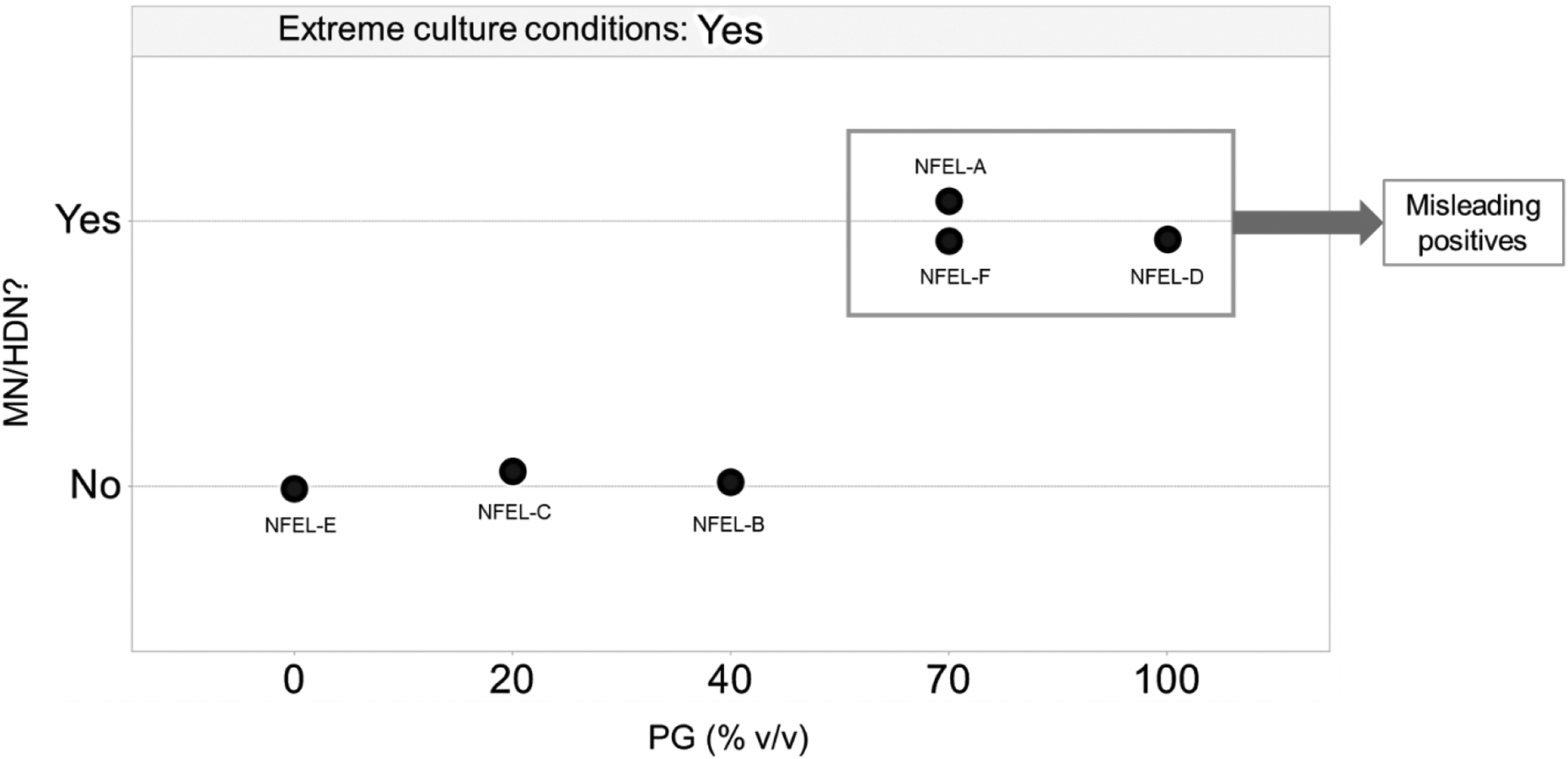
Summary of NFEL-induced effects in this study; although extreme culture conditions were induced by all NFELs, PG-predominate mixtures induced increases in MN and HDN that were likely to be nonbiologically relevant, as revealed by insignificant changes in cell cycle and DNA damage response endpoints (not represented in the figure). NFEL: non-flavored e-liquid; MN: micronuclei; HDN: hypodiploid nuclei; PG: propylene glycol.
Supplemental material
7_Supplementary_Figures - Baseline effects of non-flavored e-liquids in the in vitro micronucleus assay
7_Supplementary_Figures for Baseline effects of non-flavored e-liquids in the in vitro micronucleus assay by Daniel J Smart, Fabian R Helbling, Damian McHugh and Patrick Vanscheeuwijck in Toxicology Research and Application
Footnotes
Acknowledgement
The authors would like to thank Ms Sindhoora Bhargavi Gopala Reddy for scientific editing of the manuscript.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Philip Morris International is the employer of all the authors (at the time of publication).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Philip Morris International and is the sole source of funding and sponsor of this research.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
