Abstract
Human Milk Oligosaccharides (HMOs) are the third most abundant, solid component of human milk after lactose and fat. As novel processes are developed to cost-effectively produce commercial volumes of these oligosaccharides, they are becoming more common components of infant formulas worldwide. The study evaluated the safety of a novel mixture of HMOs in a neonatal piglet model with the objective of identifying potential effects during the sensitive, preweaning developmental stage of life. The mixture of HMOs (HMO MIX 1) was composed of 2′-fucosyllactose (2′-FL), 3-fucosyllactose (3-FL), lacto-N-tetraose (LNT), 3′-sialyllactose (3′-SL), and 6′-sialyllactose (6′-SL), and was administered to 2-day old piglets at either 5.75 or 8.0 g/L for a period of 21 days. Piglets in the 5.75 and 8.0 g/L HMO MIX 1 dosing groups did not exhibit differences in body weight, food consumption, or feed efficiency. Analysis of clinical chemistry parameters on Study Day 7 and Study Day 21 did not demonstrate any effects that could be attributed to HMO MIX 1, nor were there any findings in organ weight, macroscopic, or microscopic inspection of tissues that could be attributed to this oligosaccharide blend. Therefore, since administration of HMO MIX 1 in a liquid diet up to 8.0 g/L resulted in no toxicologically-relevant effects in comparison with animals fed a control diet, this study supports the safety of this ingredient for addition to infant formula products.
Introduction
Human Milk Oligosaccharides (HMOs) are the third most abundant solid component of human breast milk after lactose and fat, 1 with total HMO concentrations reaching up to 20 g/L. 2 HMOs are a diverse class of compounds, with over 100 unique structures present in human milk, 3 among these are neutral HMOs including 2′-fucosyllactose (2′-FL), 3-fucosyllactose (3-FL), and lacto-N-tetraose (LNT); and acidic HMOs including 3′-sialyllactose (3′-SL) and 6′-sialyllactose (6′-SL). Despite being one of the primary components of human milk, challenges in the commercial production of these oligosaccharides has limited investigations into the benefits of HMOs until recently. However, recent studies have shown prebiotic and immune benefits to infants. 3 –5
Upon the development of safe, cost-effective, large-scale processes to manufacture HMOs via fermentation, manufacturers began introducing HMOs into US infant formula products beginning with 2′-FL in 2016. With expanding evidence of the potential benefits of these oligosaccharides, 6 commercial-scale production of additional HMOs has begun.
This study focuses on the safety of a novel mixture of five human milk oligosaccharides (HMO MIX 1), containing 2′-FL, 3-FL, LNT, 3′-SL, and 6′-SL. HMO MIX 1 had previously been evaluated in a sub-chronic 90-day rat study in which HMO MIX 1 was administered at 10% of the diet, bacterial reverse mutation assay, and an in vitro micronucleus assay. 7 In these previous studies, HMO MIX 1 was demonstrated to be non-genotoxic and produced no adverse effects in animals at 10% of the diet, for a calculated dose of 5.67 g/kg and 6.97 g/kg in male and female rats, respectively. Other safety studies have been conducted on the individual HMOs in this novel mixture: 2′-FL, 8 –10 3-FL, 11 LNT, 12 3′-SL, 13,14 and 6′-SL, 15,16 including previous studies in neonatal piglets for 2′-FL 17 and 3′-SL. 18,19 These previous studies and the long history of exposure via human milk at the concentrations intended to be added to infant formula strongly support the safety of these oligosaccharides to human infants.
Standard testing paradigms, such as a sub-chronic rat study, may not adequately capture all potential effects of a food substance in young infants. This concept was discussed in a 1971 report by the Joint FAO/WHO Expert Committee on Food Additives (JECFA), 20 and confirmed in more recent assessments including a workshop held in 1998 by the International Life Sciences Institute (ILSI) 21 and guidance published in 2017 by the European Food Safety Authority (EFSA). 22 These reports identified that unique physiological, developmental, and nutritional aspects of infants during the first several months of life may not be fully addressed by standard food safety models such as a sub-chronic study conducted in rats.
In the past decade, the neonatal piglet model has been recognized by multiple risk assessment agencies, including EFSA 22 and JECFA, 23 as an appropriate model to assess the safety of food substances in young infants, including infants during the sensitive preweaning stage of life (under 12 weeks of age). Neonatal piglets have an advantage over other models through many nutritional and physiological aspects including similarities to young infants in terms of: expression of digestive enzymes, nutrient absorption, timing of gut closure, microbial populations, and gut transit time. 24 –26 Since 2014, at least four infant formula food additives (carrageenan, OSA-modified starch, xanthan gum, and gellan gum) have received a positive safety evaluation by JECFA 23,27,28 in which a neonatal piglet study was provided to support the safety determination.
Consideration of the safety of a substance added to infant formula should consider the totality of data, 29 including the additional types of data referenced by JECFA 23 and EFSA 22 such as toxicokinetics, the overall toxicological profile of the substance, and other human data available through clinical trials or post-marketing surveillance. However, the neonatal piglet model remains a useful tool in evaluating potential effects during the particularly sensitive pre-weaning developmental life stage. The data presented in this study demonstrate that this novel mixture of human milk oligosaccharides (HMOs) is well-tolerated by neonatal piglets up to 8.0 g/L, as-consumed.
Materials and methods
Animals and study site
The study was conducted by Charles River Laboratories (CRL), Mattawan, Michigan in accordance with Good Laboratory Practice (GLP) Regulations following the United States Food and Drug Administration. A total of 18 male and 18 female Domestic Yorkshire Crossbred Swine were received 2 days after birth (designated as Study Day 1) from Bailey Terra Nova Farms, Schoolcraft, Michigan. The piglets were allowed to nurse from the sow for at least 48 hours prior to receipt to allow them to consume maternal colostrum for its beneficial effects during the neonatal period.
All piglets placed on the study were examined by veterinary staff prior to the study and found to be in good health. Findings that would have led to the exclusion of an animal from the study include: abnormal lung sounds, significantly lower weight, reduced physical condition, enlarged joints or navel, severe watery diarrhea, or obvious congenital defects. As these piglets were sourced from a commercial farm and not a germ-free animal facility, minor scabbed areas, heart murmurs, and some soft feces were considered normal for piglets of this age and strain. Prior to receipt at the laboratory, the piglets were given injections of an iron supplement and a broad-spectrum antibiotic, followed by an additional iron supplement approximately 1 week after receipt. Six male and six female piglets were assigned to each of three treatment groups: control, 5.75 g/L of HMO MIX 1, and 8.0 g/L HMO MIX 1. The piglets were assigned to study groups upon receipt, and no formal randomization of animals was conducted. Animals were single housed in stainless steel cages with plastic coated flooring.
Diets
The diets used in this study included a control diet, containing no HMO Mix 1, and two test diets containing HMO MIX 1 7 at a total HMO concentration in the diet as fed of 5.75 or 8.0 g/L. A total oligosaccharide concentration of 8.0 g/L corresponds to the concentration of oligosaccharides (fructo-oligosaccharides and galacto-oligosaccharides) that can be added to infant formula per multiple global regulations, including those in the European Union and Australia/New Zealand. HMO MIX 1 was obtained from Jennewein Biotechnologie (Rheinbreitbach, Germany). The composition of the HMO MIX 1 used in this study was (percentage of dry weight): 2′-FL (49.1%), 3-FL (10.4%), LNT (19.9%), 3′-SL (3.5%), and 6′-SL (4.2%). The concentration of total HMOs in the HMO MIX 1 was 87.2%, with other carbohydrates (such as lactose) composing the majority of the remaining test article.
The test vehicle for all three diets was Land O’Lakes ProNurse Specialty Milk Replacer, reconstituted with deionized water. The reconstituted control diet had a caloric density of approximately 600 kcal/L. With a caloric density of HMO Mix 1 of approximately 2 kcal/g, the addition of 5.75 and 8.0 g/L of HMO Mix 1 did not significantly impact the caloric density of the test diets. Formulations were prepared daily and stored refrigerated prior to administration to the animals. Test diets were allowed to warm to room temperature for at least 30 minutes prior to administration. Experimental diets were introduced on the day of the animals’ arrival to the testing facility, which was designated as Study Day 1. The diets were administered orally via a feeding bowl (approximately 1500 mL) filled by hand six times per day for 21 days.
Study endpoints
The following endpoints were evaluated in this study: mortality, clinical observations, body weight, food consumption, food efficiency, compound consumption, clinical pathology parameters (hematology, coagulation, clinical chemistry, and urinalysis), gross necropsy findings, organ weights, and histopathologic examinations. Body weights and food consumption were measured daily, cage side observations were conducted twice daily, and detailed clinical observations were conducted twice weekly as well as at unscheduled intervals as needed. Clinical pathology evaluations were conducted on animals on Study Days 7 and 21, except for urinalysis which was conducted on Day 22 (at necropsy). All analyses, including the preparation of samples, were conducted per the testing facility Standard Operating Procedures, aligned with Good Laboratory Practice.
The clinical chemistry parameters measured were: alkaline phosphatase, total bilirubin (with direct bilirubin if total bilirubin exceeds 1 mg/dl), aspartate aminotransferase, alanine aminotransferase, gamma glutamyl transferase, sorbitol dehydrogenase, urea nitrogen, creatinine, total protein, glutamate dehydrogenase, albumin, globulin and albumin/globulin ratio, glucose, total cholesterol, triglycerides, electrolytes (sodium potassium, chloride), calcium, phosphorus, and LDL. The hematology parameters measured were: leukocyte count (total and absolute differential), erythrocyte count, hemoglobin, hematocrit, mean corpuscular hemoglobin, mean corpuscular volume, mean corpuscular hemoglobin concentration (calculated), absolute reticulocytes, platelet count, red blood cell distribution width, and blood smear (preserve and stain). Coagulation parameters measured were: activated partial thromboplastin time, Prothrombin Time, and Fibrinogen. Urinalysis parameters were: volume, color and appearance, protein, glucose, bilirubin, ketones, blood, urobilinogen, specific gravity, and pH.
Microscopic evaluation was conducted on: aorta, sternum, brain (cerebrum, midbrain, cerebellum, medulla/pons), epididymis, esophagus, eye, gallbladder, adrenal gland, Harderian gland, mammary gland, parathyroid gland, pituitary gland, prostate gland, seminal vesicle, thyroid gland, gut-associated lymphoid tissue, heart, kidney, large intestine (cecum, colon, rectum), larynx, liver, lung, lymph node (mandibular, mesenteric), skeletal muscle, nerves (optic, sciatic), ovary, pancreas, skin, small intestine (duodenum, jejunum, ileum), spinal cord, spleen, stomach, testis, thymus, tongue, trachea, urinary bladder, uterus/cervix, and vagina. These tissues were also examined macroscopically, and macroscopic inspection also included nasal cavity, bone marrow smear, lumbar ganglion, salivary glands (sublinghual, parotid), tibial nerve, oviduct, and ureter.
Statistical analysis
For all endpoints/parameters, the normality of the residuals and homogeneity of variances were tested to determine if the data were approximately normal or if a log transformation was required. Levene’s test 30 was used to assess homogeneity of group variances and Shapiro-Wilk’s test 31 was used to test the normality of the residuals. For the raw data, if Levene’s test was not significant (p ≥ 0.01) and Shapiro-Wilk’s test was not significant (p ≥ 0.01), then a normal distribution was used. If either the Levene’s or Shapiro-Wilk’s test were significant, normality and homogeneity of variances were tested with a log transformation used on the data. For log transformed data, if Levene’s test was not significant (p ≥ 0.01) and Shapiro-Wilk’s test was not significant (p ≥ 0.01), then a log normal distribution was used. If either the Levene’s or Shapiro-Wilk’s test was significant, then a rank transformation was used on the data.
For raw or log transformed data, a one-way analysis of variance was used to test each endpoint for the effects of treatment. If the treatment effect was significant (p < 0.05), linear contrasts were constructed for a Dunnett’s pair-wise comparison of treatment groups as described above. For rank transformed data, a Kruskal-Wallis test was used to test each endpoint for the effects of treatment. If the treatment effect was significant (p < 0.05), a non-parametric Dunn’s pair-wise comparison test of each treatment group with the control group was performed.
Results
Study completion
All of the animals, with the exception of one male piglet, survived until the scheduled study termination on Study Day 22. On Study Day 7, one male piglet in the 8.0 g/L dosing group was euthanized for humane reasons per facility procedures. Clinical and veterinary observations of the animal that was euthanized included yellow discolored feces, thin body condition, unkempt appearance, generalized muscle wasting, and lateral recumbency. Analysis was performed on this animal including measurements of body weight and food consumption, clinical pathology parameters, and necropsy.
Growth and food consumption
There were no statistically significant differences in body weight (Figure 1) or food consumption between the control group and either of the treatment groups for either male or female animals when compared within each gender. Food efficiency was statistically lower in the female piglets in the 5.75 g/L dosing group compared to controls on Study Days 18–19 (11.0% vs. 18.1% in controls). However, this decrease in food efficiency was not seen on any other study days and was also not seen in the 8.0 g/L dosing group.
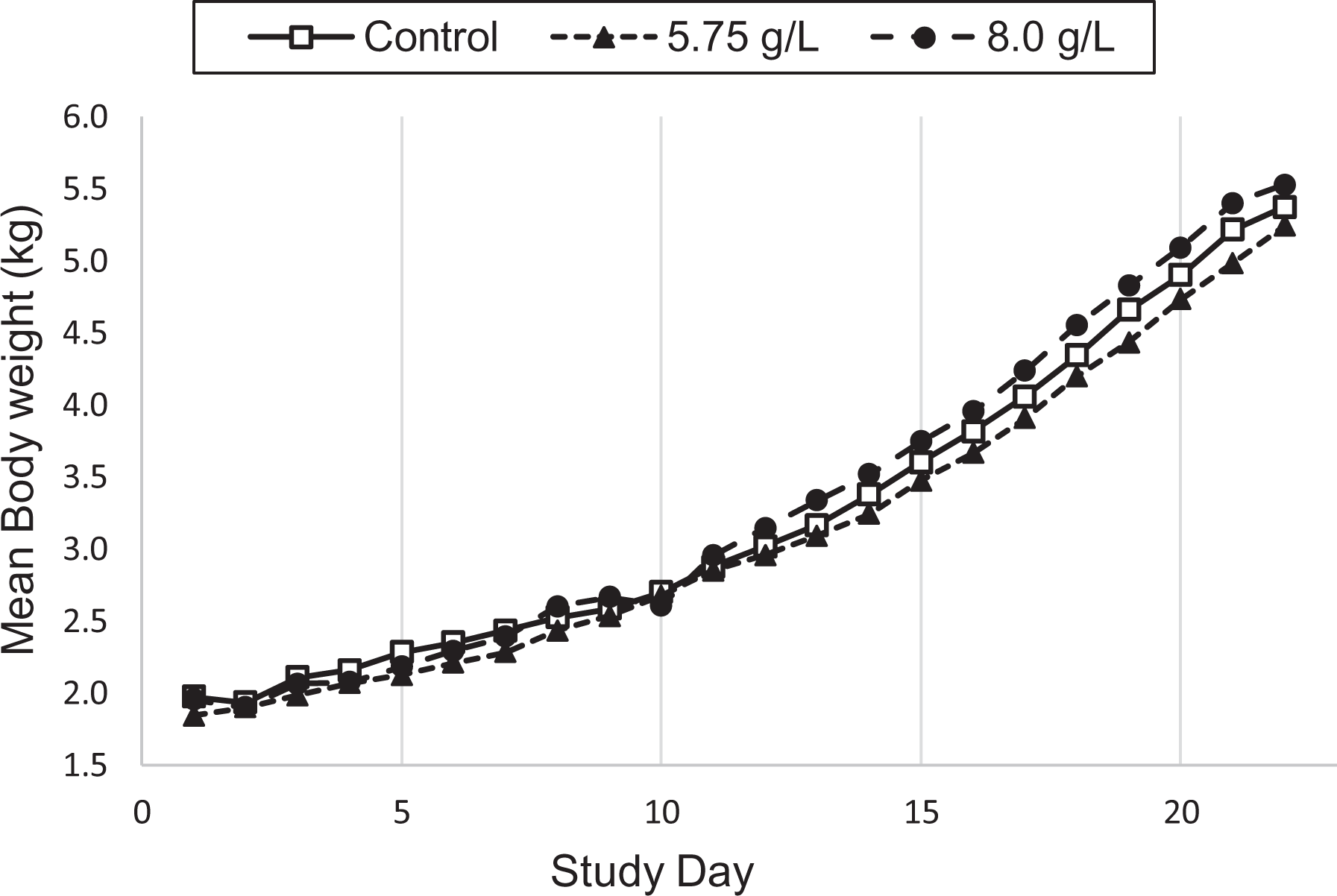
Mean body weight values (kg) in neonatal farm piglets. Values for the control, 5.75 g/L, and 8.0 g/L dosing groups represent the mean body weight of male and female piglets combined. Body weights for the piglet in the 8.0 g/L dosing group that was euthanized on Study Day 7 were excluded from the calculation.
The calculated consumption of the HMO MIX 1 ingredient was calculated for males as 2556.2 ± 180.2 and 3576.4 ± 257.4 mg/kg body weight in the 5.75 and 8.0 g/L dosing groups, respectively, and in females as 2603.9 ± 77.9 and 3659.8 ± 133.8 mg/kg body weight/day, respectively. Thus, HMO MIX 1 consumption in the high dose groups (approximately 1.4-fold greater) was comparable to the difference in formulation concentration.
Clinical and veterinary observations
Animals in all treatment groups exhibited general deviations, including occasional gastrointestinal signs (e.g. watery or discolored feces), physical signs (e.g. skin abrasions), or other signs (e.g. swelling of the tongue, eyelid swelling). However, the observations of animals in the 5.75 and 8.0 g/L dosing groups were similar to those in the control group, seen infrequently, and/or considered common in neonatal piglets.
Of particular note is the administration of additional antibiotics to animals that exhibited signs of a mild bacterial infection of the gastrointestinal tract as evidenced by yellow/watery feces. Piglets with a fecal score indicative of this infection were administered a systemic antibiotic (injectable oxytetracycline) for a total of 3 days. The distribution of piglets across the treatment groups that required additional antibiotic treatment was distributed evenly across all groups: 5 in the control group, 4 in the 5.75 g/L dosing group, and 5 in the 8.0 g/L dosing group. All antibiotic treatments were completed by Study Day 10.
Clinical pathology
Per the protocol, clinical chemistry samples were taken from all animals on Study Day 7 and 21. On Study Day 7, several animals (3 in the control group, 2 in the 5.75 g/L group, 1 in the 8.0 g/L group) had lower serum sodium and/or chloride concentrations that were likely secondary to electrolyte loss associated with the watery feces that was observed in these animals. These effects were seen in all three treatment groups, were not dose-dependent, and/or the signs resolved in all three treatment groups by Study Day 21.
In the study there were no statistically significant differences between treatment groups in either hematology or urinalysis parameters. While there were significant differences in several coagulation and clinical chemistry parameters (Table 1), the differences were not considered treatment-related as they were small in magnitude, were not dose-dependent, were not seen in both sexes, resolved between Study Day 7 and 21, and/or changes were consistent with historical control data.
Statistically significant clinical pathology parameters.
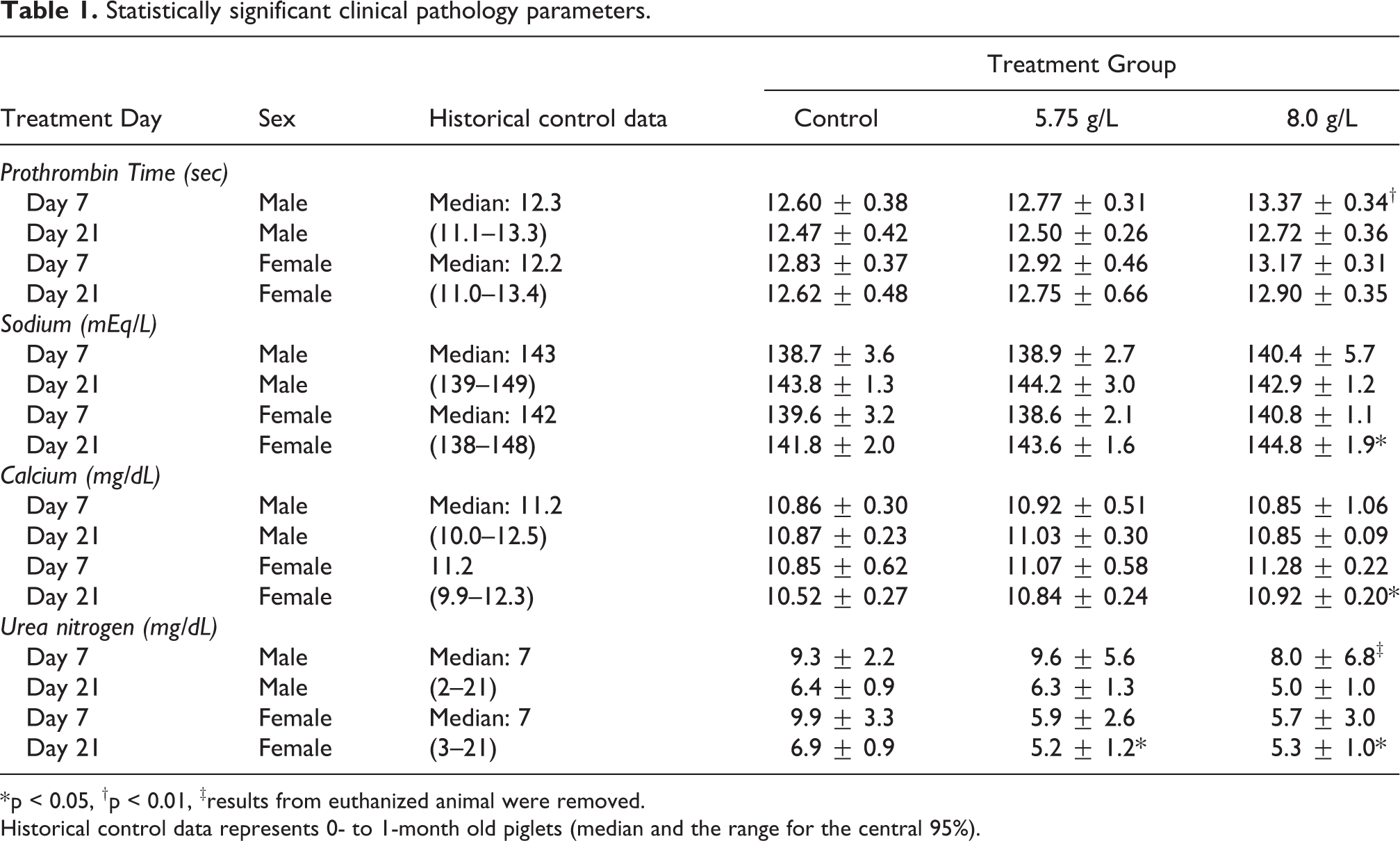
* p < 0.05, †p < 0.01, ‡results from euthanized animal were removed.
Historical control data represents 0- to 1-month old piglets (median and the range for the central 95%).
Clinical chemistry analysis was performed on the one male piglet in the 8.0 g/L dosing group that was euthanized on Study Day 7. On Study Day 7, this piglet had higher potassium, chloride, phosphorus, alkaline phosphatase (ALP), urea nitrogen, creatine, total protein, albumin, globulin, triglyceride, cholesterol, and LDL cholesterol measurements and a lower aspartate aminotransferase (AST) measurement than male piglets in the control group, the 5.75 g/L dosing group, and the remaining five piglets from the 8.0 g/L dosing group.
Postmortem study evaluations
The only statistically significant differences in absolute or relative organ weights was an increase in large intestine (cecum and colon) weight/body weight ratio in male piglets in the 8.0 g/L dosing group (Table 3). This effect was not significant in the female piglets, but a trend toward increasing cecum and colon weight was observed in the female piglets, as well as in the male piglets in the 5.75 g/L dosing group. There was not a significant increase in rectum to body weight ratio in male piglets. There were no microscopic observations that were correlated to the changes in large intestine weights.
Clinical chemistry parameters for euthanized piglet.
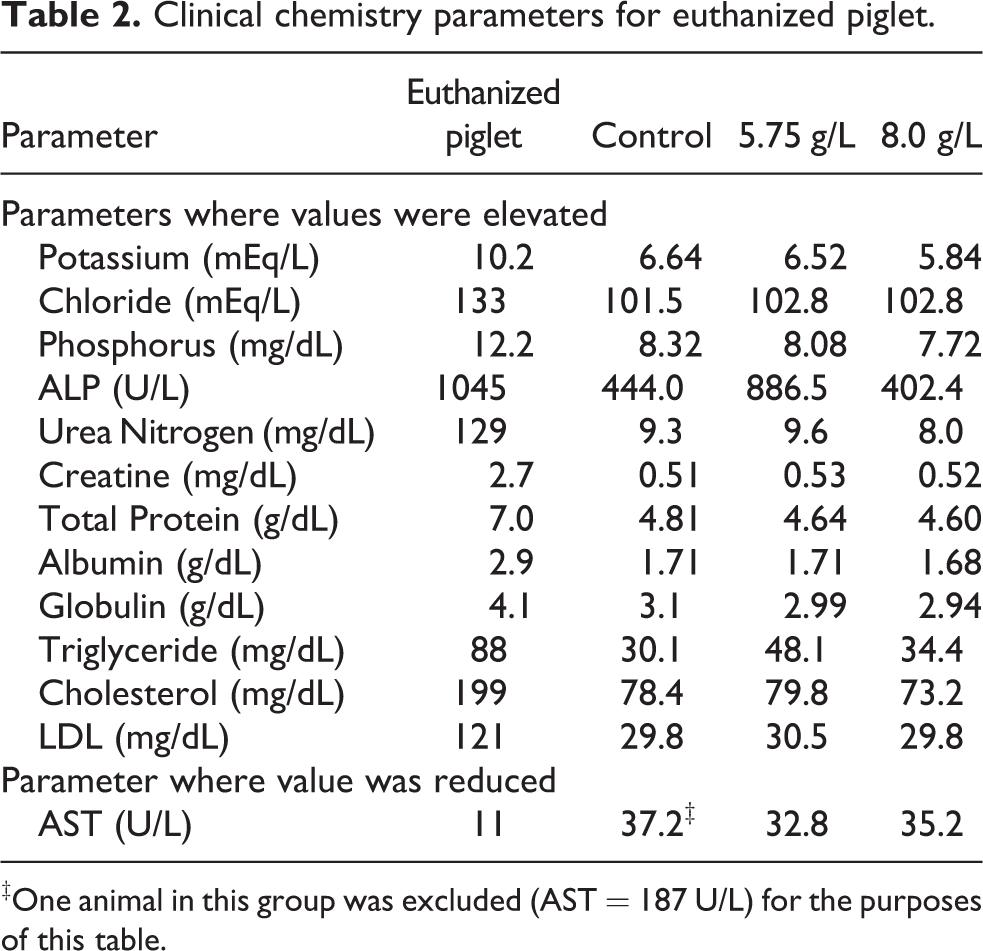
‡One animal in this group was excluded (AST = 187 U/L) for the purposes of this table.
Intestinal weight ratios.
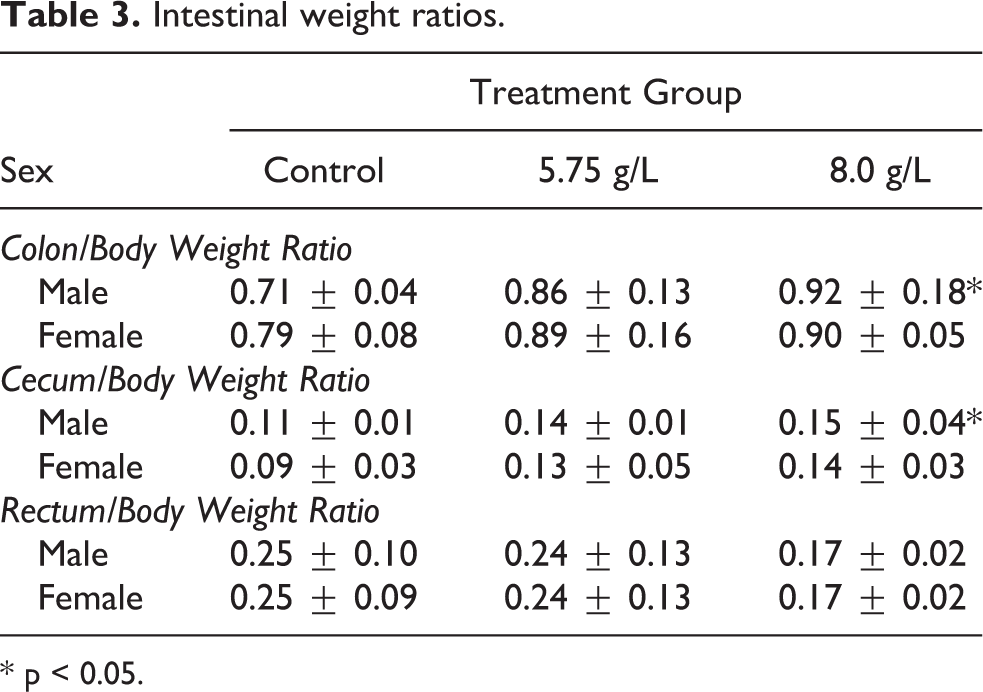
* p < 0.05.
Macroscopic evaluation of tissues at the termination of the study (Study Day 22) identified some observations that were incidental and/or consistent with those typically observed in piglets of this age: kidney cysts in 5 male piglets (2 control, 2 in the 5.75 g/L group, 1 in the 8.0 g/L group), a nodule in the kidney of one of the female piglets in the control group, clear fluid in the scrotum of one of the male piglets in the 5.75 g/L group, and red discoloration in the ileum of one of the male piglets in the control group.
Additionally, the microscopic observations in the tissues at the termination of the study (Study Day 22) were low in incidence, lacked a dose response, occurred in control animals, or were otherwise consistent with observations in piglets of this age. The most common microscopic observations were polyarteritis (inflammation of small to medium sized arteries) in a variety of tissues, which was observed in 6 animals in the control group (3 male, 3 female), 3 animals in the 5.75 g/L group (3 female), and 5 animals in the 8.0 g/L group (3 male, 2 female). The inflammation was typically of minimal severity (scored 1 on a 5-point scale, 72.2% of the observations), with a small number of moderate severity (4.3% scored 3 on a 5-point scale), and none scoring as more than moderate severity. The polyarteritis was distributed throughout a variety of tissues (Supplemental Table 1). Of the 14 animals where polyarteritis was observed, 8 animals had polyarteritis in more than 10 organs (5 in the control group, 3 in the 8.0 g/L group), while the other animals (1, 3, and 5 animals in the control, 5.75 g/L, 8.0 g/L groups, respectively) had polyarteritis in less than 10 different organs.
The other microscopic findings were distributed throughout different organs (Supplemental Table 2), were primarily of a minimal or mild degree of severity (1 or 2 on a 5-point scale) and were distributed between the sexes and the treatment groups. There were no findings determined to be a 4 or 5 on a 5-point scale of severity, and the findings that were determined to be of moderate severity were eosinophilic infiltration of the mesenteric lymph node that was observed in one male animal in the control group and two male animals in the 8.0 g/L treatment group.
Microscopic inspection of the tissues from the piglet in the 8.0 g/L dosing group that was euthanized on Study Day 7 revealed gastrointestinal inflammation, gram-negative bacilli, mucosal gland dilation, and goblet cell hypertrophy/hyperplasia with increased mucous. Other findings included fat atrophy, lymphoid depletion and decreased bone marrow cellularity. These observations were unique to this animal and were not present in animals of the other treatment groups, or other animals in the 8.0 g/L dosing group. Additionally, this piglet exhibited values for a number of clinical chemistry parameters that were also inconsistent with all of the other animals in the study (Table 2).
Discussion
With the exception of a single male piglet in the 8.0 g/L dosing group, all of the piglets administered HMO MIX 1 at either 5.75 g/L or 8.0 g/L survived through study termination at Study Day 22. All differences in clinical chemistry parameters, including those that were statistically significant (Table 1), were small in magnitude, inconsistent in direction of response, did not show dose responsiveness, or were consistent with findings commonly observed in farm piglets under similar study conditions. Similarly, with the exception of large intestine weight in male piglets (Table 3), all pathological endpoints including organ weights, and macroscopic and microscopic evaluation of tissues demonstrated no adverse effects that could be contributed to HMO MIX 1. Therefore, considered collectively this study demonstrated that HMO MIX 1 was administered to neonatal piglets up to 8.0 g/L without adverse effects.
HMO MIX 1 did significantly increase large intestine weights (cecum and colon) in male piglets. In the previous neonatal piglet study in which 2′-FL 17 was administered at concentrations up to 2.0 g/L, it should be noted that while there were not statistically-significant differences in cecum and colon weights, there was a trend toward increasing large intestine to body weight ratio (Table 4). However, multiple studies have demonstrated a similar effect with other oligosaccharides in both rats and pigs, 32 –35 therefore this result is not unexpected. Since other oligosaccharides have been shown to impart this effect, together with the lack of other indices (such as microscopic observations) in this study, the increase in large intestine (colon and cecum) weight is not considered adverse, but rather an adaptive response to exposure to oligosaccharides.
Intestinal weight ratios from Hanlon and Thorsrud. 17
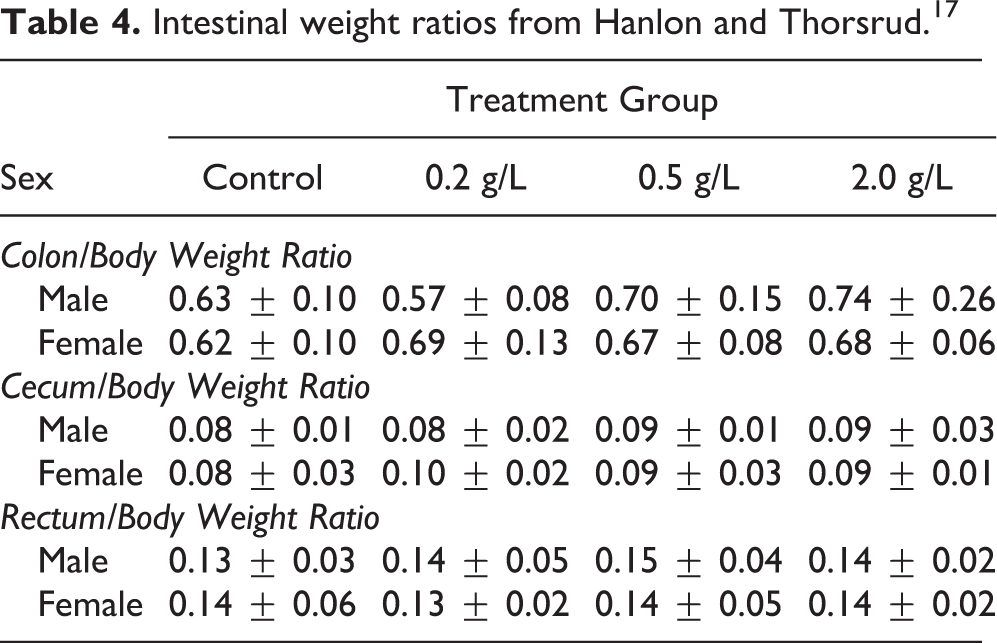
The need to euthanize one male piglet in the 8.0 g/L dosing group on Study Day 7 for humane reasons was unexpected. Clinical chemistry analysis of the piglet (Table 2) demonstrated that this individual piglet was exceptional compared to the other piglets in the control and 5.75 g/L dosing groups, but also in comparison with the other male piglets in the 8.0 g/L dosing group. Microscopic inspection of the tissues from this animal were consistent with the causes of mortality frequently observed in pre-weaned piglets, 36 and these effects were also not seen in any of the other animals in either the control, 5.75 g/L, or 8.0 g/L treatment groups. Therefore, it was concluded that the effects in this animal were unrelated to the administration of HMO MIX 1.
All piglets were examined by a staff veterinarian prior to their inclusion in this study and were found to be in good health and were continually monitored during the study. Unlike rodent models, where purpose-bred animals are typically sourced from germ-free facilities, the neonatal piglet model necessitates sourcing animals from a local farm facility. The farm is a certified food production facility, but the animal conditions are notably different than what a purpose-bred research animal facility can provide. Neonatal piglets are a sensitive laboratory model, and transportation of the 2-day old animals is a significant concern for their welfare, presenting logistical challenges to developing a centralized purpose-bred facility for research piglets.
The analysis of the samples from the euthanized piglet suggests that the decrease in physical condition that led to the need for humane euthanasia on Study Day 7 was due to a bacterial infection that was likely obtained at the farm prior to enrollment on the study. A fecal culture from this animal identified that E. coli was present in this animal. Veterinary observations of yellow, discolored feces occurred in all three treatment groups, including the control group, and antibiotics were administered to these animals presenting with these clinical signs. The distribution of these clinical signs supports the conclusion that the unscheduled death of the male piglet in the 8.0 g/L dosing group was not related to HMO MIX 1, but rather due to an underlying infection that was distributed evenly between the animals in all dosing groups.
In this study, administration of HMO MIX 1 to neonatal farm piglets up to 8.0 g/L was not associated with any adverse outcomes. The lack of adverse effects in genotoxicity and sub-chronic studies conducted in rat with HMO Mix 1, 7 the safety of the individual oligosaccharides demonstrated with other safety studies, 8 –19 and the results of this study support the safety of this ingredient in infant formulas, including those consumed during the sensitive period of human development below 12 weeks of age.
Supplemental material
Supplemental Material, Supplemental_Tables - A safety evaluation of mixed human milk oligosaccharides in neonatal farm piglets
Supplemental Material, Supplemental_Tables for A safety evaluation of mixed human milk oligosaccharides in neonatal farm piglets by Paul R Hanlon in Toxicology Research and Application
Footnotes
Acknowledgments
Thanks to Jeff Walburn, Shelley Beazley, and Laura Cregar of Charles River Laboratories and Jessica Deis of Abbott Nutrition for critically reading and providing suggestions to improve this review paper.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Paul Hanlon is a full-time employee of Abbott Nutrition.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
