Abstract
The microalgae
Introduction
The phycobiliproteins are a group of light-capturing proteins associated with the photosynthetic apparatus of several types of microalgae. Due to their role in passing on light energy to the cells’ photosystems, phycobiliproteins are deeply coloured, fluorescent and water soluble. They are composed of an apoprotein to which is bound the bilin chromophore, phycocyanobilin. Often the most abundant phycobiliprotein is C-phycocyanin, a blue phycobiliprotein responsible for giving the characteristic colour to blue–green microalgae such as
In addition to its natural role in photosynthesis, C-phycocyanin is now used as a blue colouring agent for foods and presents a natural alternative to blue dyes of chemical origin such as E131, E132 and E133. Extracts rich in C-phycocyanin from
Whilst a suitable replacement for synthetic blues in many applications, the low stability of
There is, therefore, an opportunity to develop phycocyanins from other organisms where the technical characteristics of the pigmented proteins are better and where different types of growth system can be exploited to improve safety, consistency and productivity. A group of organisms that are actively being investigated is the cyanidiophyceae. These eukaryotic algae are members of the red algae (rhodophytes), which are generally characterized by the presence of a phycoerythrin, a red phycobiliprotein closely related to phycocyanin. However, the cyanidiophyceae is a green–blue colour similar to that of the cyanobacteria due to the presence of significant quantities of C-phycocyanin. 18,19 The apoproteins of C-phycocyanin from cyanobacteria and those from cyanidiophyceae are structurally very similar but have minor differences in primary amino acids structures resulting in them having different isoelectric points and stabilities. 17
The microalga
Besides a technically superior phycocyanin for colourant purposes,
To date, no risk assessment of
Material and methods
The bacterial reverse mutation test and the in vitro micronucleus test were conducted by Citoxlab France (Evreux, France). The 90-day oral toxicity study was conducted by Phycher Bio Développement (Martillac, France).
Test item details
The test item used for the studies was an extract prepared from the dried biomass of a unicellular red microalga,
The reference item used in this study was a phycocyanin extract of
Gs-PC and Sp-PC are completely soluble in water. Before use, both extracts were suspended in analytical grade water or water for injections for the in vivo and the in vitro studies, respectively. For the in vivo study, the dosing formulations were prepared freshly each day at different concentrations to allow a constant volume of administration of 10 mL/kg. All concentrations of the Gs-PC formulations were subjected for verifications twice, in the first and in the last week of the in vivo study.
Bacterial reverse mutation test
The study design was based on the Organisation for Economic Co-operation and Development (OECD) guidelines
After 48–72 h of incubation at 37°C, the number of revertant colonies was scored for each strain and for each dose level using an automatic counter (Sorcerer Automatic Colony Counter, Perceptive Instruments Ltd, UK). The observation of a decrease in the number of revertant colonies and/or thinning of the bacterial lawn was used to assess the toxicity of Gs-PC. Criteria for a positive result was a reproducible 2-fold increase (for the TA 98, TA 100 and TA 102 strains) or 3-fold increase (for the TA 1535 and TA 1537 strains) in the mean number of revertant colonies as compared to the vehicle control and/or the evidence of a dose relationship.
In vitro micronucleus test in L5178Y TK+/− mouse lymphoma cells
The study design was based on OECD guideline
Cells treatment
The day of experiment, cells were counted and suspended at 3.105 cells/mL in 24-well culture plates and then exposed to increasing concentrations of Gs-PC (500, 1000 and 2000 µg/mL) or positive control in the absence or in the presence of S9 mix. When cells were treated without S9 mix, they were exposed according to two protocols durations as follows: 3 h treatment with Gs-PC and 24 h recovery period, or 24 h treatment with no recovery period. When cells were treated with S9 mix, they were exposed for 3 h with Gs-PC followed by 24 h of recovery period. The positive controls that were used were mitomycin C (1 µg/mL), colchicine (0.5 µg/mL) or cyclophosphamide (6 µg/mL). At the end of the treatment protocol, the cytotoxic and the cytogenetic effects of Gs-PC were determined.
Assessment of cytotoxicity
At the end of the treatment period, the cytotoxic effect of Gs-PC was determined by the measurement of population doubling (PD). Briefly, the cells were collected and counted, and the PD was determined as follows:
PD was then expressed as a percentage of control as follows: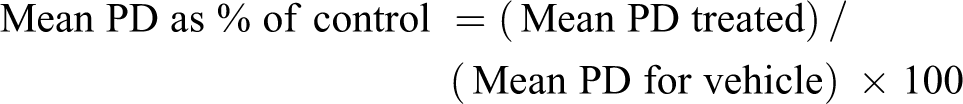
The cytotoxicity induced by treatments was calculated by the decrease in the PD when compared to the control as follows: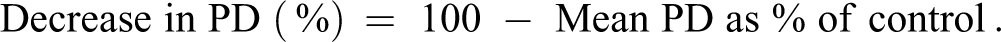
Cytogenetic experiment
At the end of the treatment period, the cytogenetic effect of Gs-PC was determined by the count of micronuclei. Briefly, the cells were washed and suspended at 04°C overnight in 49.5% culture medium (containing 10% of inactivated horse serum), 50% phosphate-buffered saline and 0.5% of pluronic acid. The cells were thereafter spread on glass slides, air-dried and stained with 5% Giemsa for 15 min. All slides were blind coded and the number of micronuclei was assessed in 2000 mononucleated cells according to the recommendations of Miller et al. 27 To consider a positive result for the test, the evaluation criteria were a statistically significant dose-dependent increase in the frequency of micronucleated cells, the frequency of micronucleated cells of each replicate was to be above the corresponding vehicle historical range and a statistically significant difference as compared to the vehicle for at least one dose of the test item. A negative response was considered when none of these above-mentioned criteria for a positive response were met.
Repeated dose 90-day oral toxicity study
The assessment of toxicological profile of Gs-PC and Sp-PC was assessed in rats over a period of oral administration of 90 days. This study was performed in compliance with the OECD guidelines n°408 under the recommendations of the Committee for the Purpose of Control and Supervision of Experiments on Animals guidelines for laboratory animal facility and was approved by Institutional Animals Ethics Committee (Form B O8:18488).
Animals
Male and female Wistar rats of 7–8 weeks and weighting from 228 to 293 g for males and 168 to 211 g for females were obtained from Hylasco Biotechnology Pvt. Ltd (Telangana, India). Animals were acclimatized for a period of 7 days before experiment and housed in the experimental animal facility of INTOX PVT, Ltd (Maharashtra, India). Groups of two or three animals of similar sex were housed in the same cage (1 218 cm2) with stainless steel grill tops, facilities for food and water bottle and with bedding of clean and sterilized corn-cob. Rat pellets were provided by M/s Altromin Spezialfutter GmBH & Co. KG (Germany) and water filtered and subjected to UV irradiation was provided
Experimental design
Groups of 10 male and 10 female rats were administered with Gs-PC or Sp-PC by daily oral gavage for a period of 90 consecutive days, along with the control group of rats. Three levels of dose (250, 2500 and 4000 mg/kg bw/day that represent 82.5, 825 and 1320 mg/kg bw/day of phycocyanin, respectively) were chosen on the basis of a preliminary 14-day dose finding study for Gs-PC, and one dose (2012 mg/kg bw/day that represent 1327 mg/kg bw/day of phycocyanin) was chosen for Sp-PC to reach the same level of phycocyanin as for the highest dose of Gs-PC. Additional groups of five rats per sex for the vehicle and high-dose group of Gs-PC were further kept for observations for a post-treatment period of 28 days to allow the evaluation of the persistence, the reversibility or delayed occurrence of toxic effects. Vehicle for Gs-PC or Sp-PC was analytical grade water and each dose was prepared in vehicle as described above at the dose volume of 10 mL/kg. As Gs-PC formed very viscous dose formulation at the highest dose, each dose was divided into two parts and orally administered in the morning and in the afternoon to rats at an interval made about 4–6 h. To the same manner Sp-PC was administered by gavage to each rat twice a day at approximately the same time with the same procedure as per Gs-PC. The design of the groups is indicated in Table 1.
Design of the 90-day repeated dose oral toxicity study.
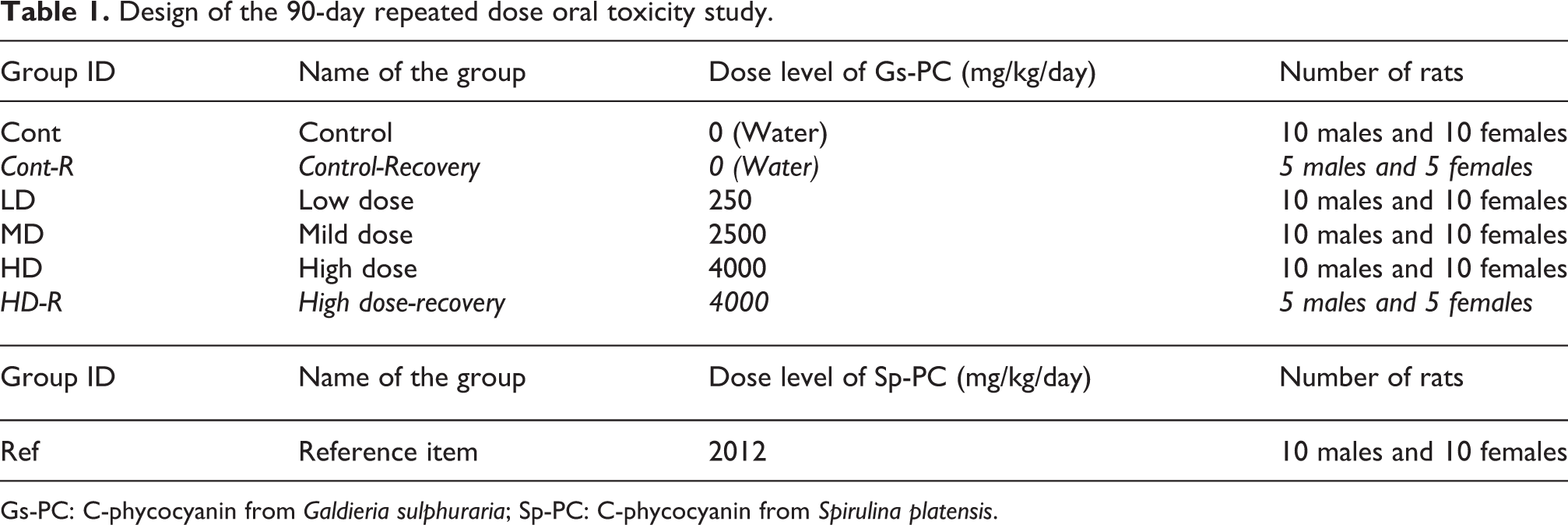
Gs-PC: C-phycocyanin from
Observations
The body weight of each rat was recorded before initiation of the treatment, weekly thereafter and before euthanasia for necroscopy. Body weights of recovery groups were recorded weekly during the post-treatment period and before euthanasia for necroscopy. The quantity of food consumed was recorded each week for all the groups.
Each animal was subjected to clinical examinations once daily during the treatment and the recovery periods. All signs of illness, together with behavioural changes were recorded. Detailed clinical examinations outside the cage in a standard arena were performed for each rat before initiation of the treatment and weekly thereafter during the treatment and recovery periods. The recorded signs included changes in skin, fur, eyes and mucous membranes, occurrence of secretions and excretions and autonomic activities such as lacrimation, piloerection, pupil size and unusual respiratory pattern. Changes in gait, posture, presence of abnormal movements or behaviours and response to handling were also noted. In the last week of treatment, animals were subjected to neurological examination according to Kulig et al. for assessment of sensory reactivity, grip strength and motor activity. 28
Haematological and biochemical examinations
At the end of the treatment and recovery periods, blood samples were performed under light carbon dioxide anaesthesia from the orbital plexus of each animal. Samples were collected in potassium ethylenediaminetetra-acetic acid collection tubes for haematology and heparin for clinical chemistry. In addition, urine samples from all animals were collected in the last week of treatment and recovery periods.
Complete blood count and haematological parameters’ measurements for each animal were performed using ADVIA 2120i Haematology System (Siemens Healthcare Diagnostic Ltd. Camberley, UK) and Abbott Cell Dyn 3700 (Abbott Park, Illinois, USA). Coagulation parameters were measured using Semi Automated Coagulation Analyser Start 4® (Diagnostica Stago, France). Urinalysis was performed for each animals using Multistix® 10SG manufactured by Siemens Healthcare Diagnostics Pty. Ltd. (Bayswater Victoria, Austria). Plasma samples for clinical chemistry parameters were analysed by commercially available diagnostic kits manufactured by Siemens Healthcare Diagnostics Ltd. (Camberley, UK). The hormones analysis for triiodothyronine (T3), thyroxine (T4) and thyroid-stimulating hormone (TSH) were carried out using competitive enzyme-linked immunosorbent assay (ELISA) and Sandwich ELISA kits, respectively, and measured with a Thermo Scientific Multiskan GO ELISA plate reader (Thermo Fisher Scientific Corporation, Waltham, USA).
Necroscopy examinations
At the termination of the treatment and the recovery periods, all rats were sacrificed by exsanguination under CO2 anaesthesia and complete necroscopy was carried out. The following organs were dissected free from connective tissues and were weighed: kidneys, liver, adrenals, testes, epididymis, uterus, thymus, spleen, brain, ovaries, heart, thyroid gland, pituitary gland, prostate and seminal vesicles with coagulating glands as a whole complex. Tissues were embedded in paraffin wax, sectioned and stained with haematoxylin and eosin. A microscopic examination was performed on selected tissues (and on any macroscopic lesions) from control and high-dose groups. Examinations were not extended to the lower dose groups and recovery groups in the absence of any treatment-related histopathological findings in the high-dose group.
Statistical analysis
Results are expressed as a mean ± standard deviation. Body weight, haematological parameters, clinical chemistry parameters and organ weights were analysed using parametric one-way analysis of variance followed by Dunnett
For cytogenetic experiments, the frequency of micronucleated cells in treated cultures was compared to that of the vehicle control culture using
Results
In the following, results from the toxicological in vitro and in vivo studies are presented and started by the description of the results obtained in bacteria.
Bacterial reverse mutation test
As the mean number of revertants for the vehicle and positive controls met the acceptance criteria, the study was considered to be valid. Gs-PC was freely soluble and no precipitate was observed in plates cultures at any concentrations used. At concentration up to 5000 µg/plate, Gs-PC did not induce any decrease in the number of revertant colonies or a thinning of the bacterial lawn (except for the TA 98 strain in the presence of S9 mix, not shown) and was further considered to not be severely toxic (Table 2). No noteworthy toxicity was shown at the highest dose level selected of 5000 µg/mL according to the criteria specified in the international guidelines. A colouration of agar was observed in the plates for concentrations of Gs-PC above 2500 µg/plate, but this colouration did not prevent scoring. Gs-PC did not increase the number of revertant colonies in any bacterial strains at any dose level either with or without S9 mix as compared to the vehicle (Table 2).
Bacterial reverse mutation test on Gs-PC.
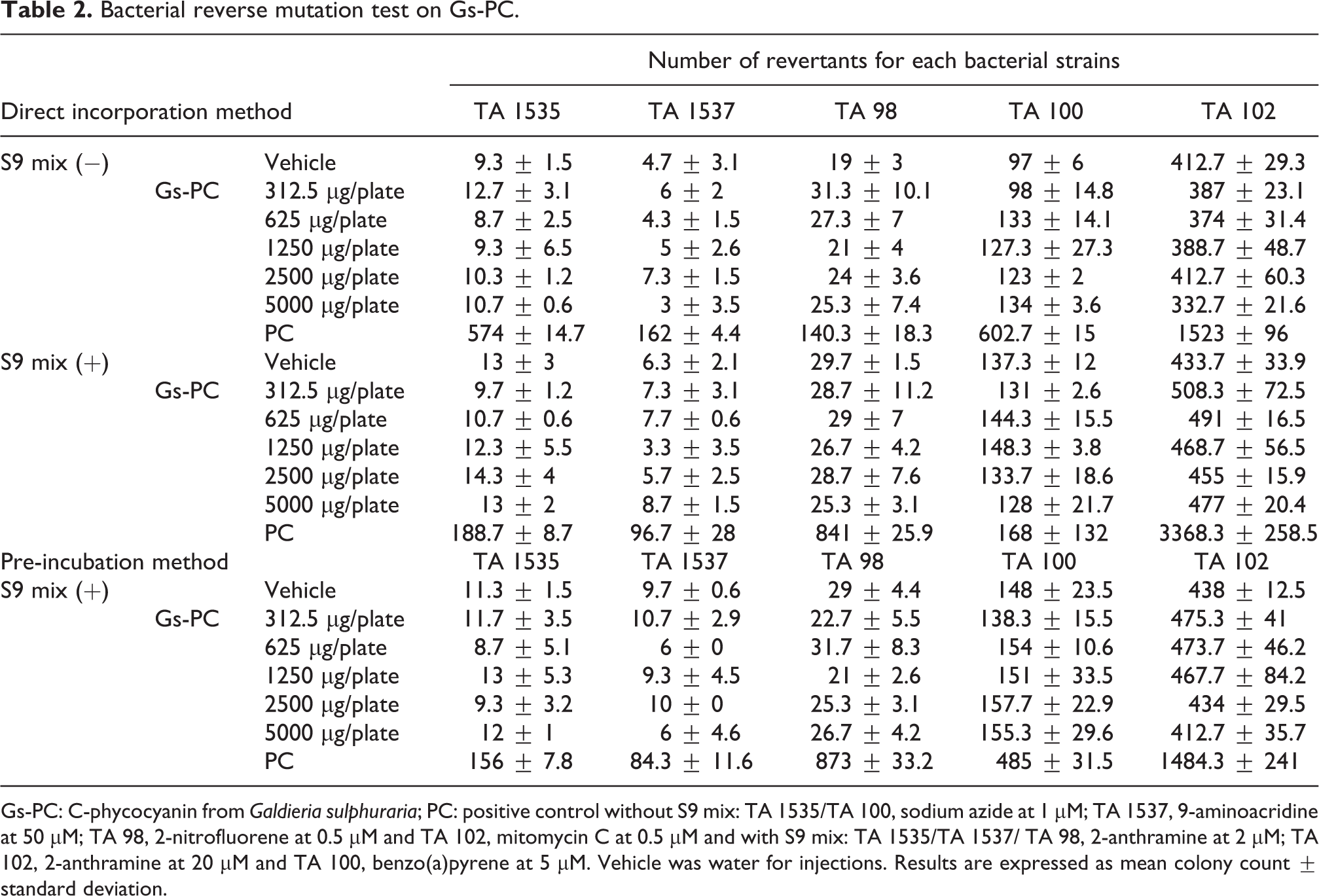
Gs-PC: C-phycocyanin from
In vitro micronucleus test in L5178Y TK+/− mouse lymphoma cells
In a preliminary study, several doses up to 2000 µg/mL were assessed to estimate the cytotoxicity of GS-PC by the measurement of PD (see ‘Material and methods’ section). At any dose tested, no decrease in PD was observed (not shown). Therefore, Gs-PC was considered to be non-cytotoxic for the doses used. As recommended to criteria specified in the international guidelines, the highest dose level of Gs-PC to test its genotoxic potential was set to 2000 µg/mL. At this dosage, the pH of the medium remained around 7.4 as for the osmolality which was measured to 306 mOsm/kg. Those values were close to those measured for the control conditions (pH: 7.4 and osmolality: 300 mOsm/kg). A blue colouration of the culture medium was observed at doses above 200 µg/mL. At the end of the treatment period, no precipitate that could lead to a misreading was observed in the culture medium either in the absence or in the presence of S9.
During the experiment PD of the control vehicle in the absence or in the presence of S9 was greater than 1, thus indicating that cells have undergone mitosis. The mean frequency of micronucleated cells for vehicle was comprised between 2‰ and 4‰ and is consistent with the control historical data of the laboratory. As presented in Table 3, the frequency of micronucleated cells treated with positive controls were significantly increased as compared to the respective control in each type of experiments either in the absence or in the presence of S9. Therefore, the study met the acceptance criteria specified by the international guidelines.
In vitro micronucleus test on Gs-PC in L5178Y TK+/− mouse lymphoma cells.
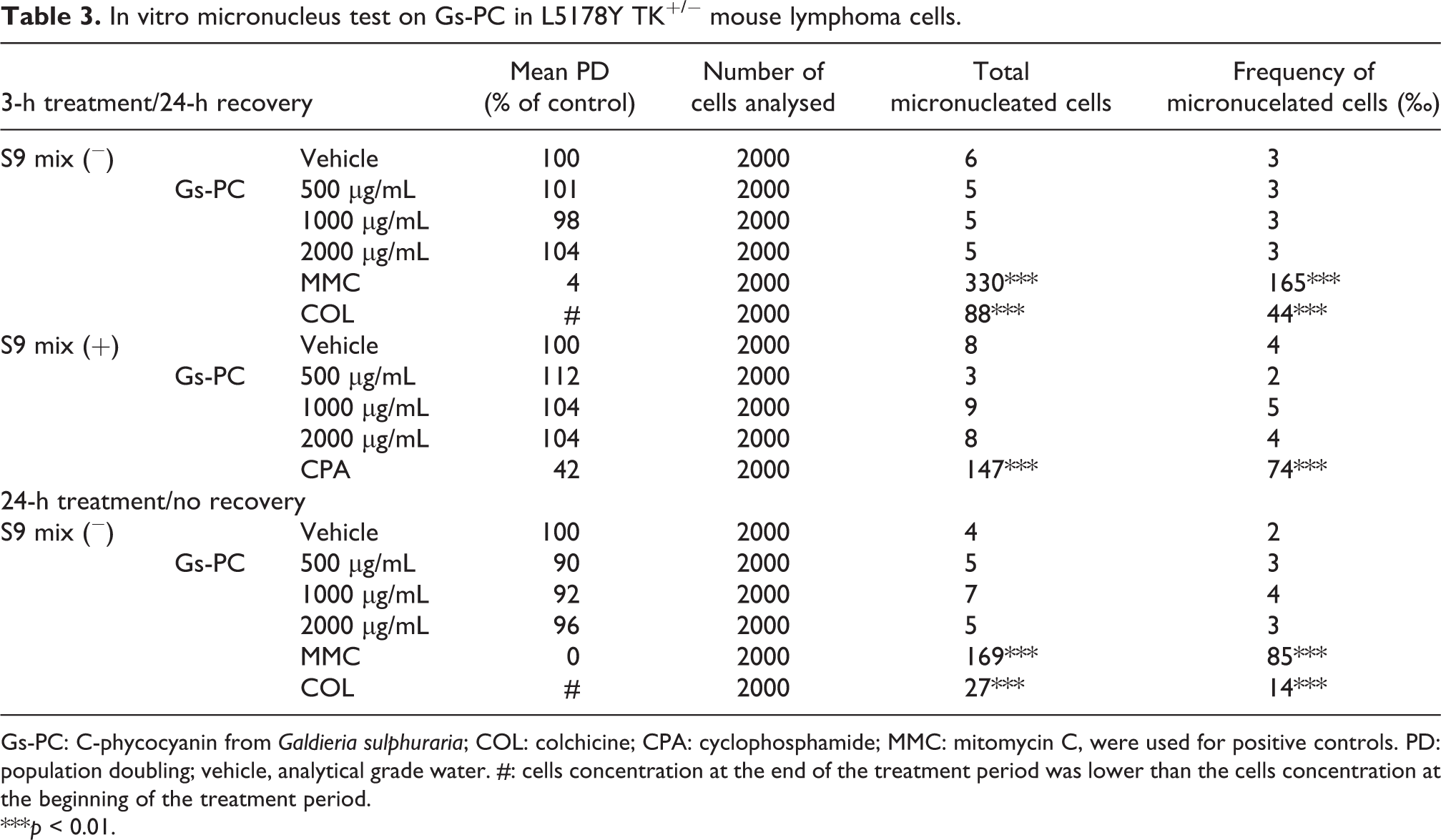
Gs-PC: C-phycocyanin from
***
In the 3-h treatment/24-h recovery experiment, the mean PD for Gs-PC for all the concentrations tested was not statistically different from those observed within their respective vehicle groups. In addition, the frequencies of micronucleated cells, whatever the concentrations of Gs-PC used, were not different from those observed for their respective vehicle control group (Table 3). Same results were observed either in the absence or the presence of S9. In the 24-h treatment/no recovery experiment, the mean PD for Gs-PC for all the concentrations tested were not statistically different as compared to the vehicle control group. As reported in Table 3, the frequencies of micronucleated cells, whatever the concentration of Gs-PC used, were not statistically different from the frequency recorded for the vehicle group.
Repeated dose 90-day oral toxicity study
A preliminary dose-finding study was performed for the rationale selection of doses for the further 90-day oral toxicity study. Male and female rats were treated by oral gavage at the dose of 250, 2500 and 4000 mg/kg bw/day with Gs-PC, for 14 consecutive days. Animals were examined daily for signs of toxicity, morbidity and mortality. At day 15, animals were sacrificed and subjected to a detailed necropsy. During experiment, no mortality, no clinical abnormality, no effect on body weight, no effect on food consumption, no effect on organ weight and no gross pathological change were observed in rats treated with 250, 2500 and 4000 mg/kg bw/day. Thus the above-mentioned doses were selected for the 90-day oral toxicity study and results are presented in the following.
Mortality
No incidence of any mortality amongst the rats treated with Gs-PC or Sp-PC was observed throughout the study (treatment and recovery periods), whatever the dose.
Clinical signs
The general daily and detailed weekly clinical examinations did not reveal any incidence of clinical abnormality throughout the study (treatment and recovery periods), whatever the dose or the type of extract.
Ophthalmoscopy
At the beginning (day 0) and at the end (day 89) of the study, no incidence of ocular abnormality was observed for the ophthalmologic examinations, whatever the dose or the type of extract.
Neurological examination
The neurological examinations assessed by functional observations were conducted in the last week of the study and did not reveal any remarkable incidence of neurological abnormality, whatever the dose or the type of extract.
Body weights and food consumption
The body weights for male and female rats of the treated groups, whatever the dose or the type of extract used, were not statistically different between groups and also as compared to the control group throughout the study (treatment and recovery periods). Those results were reported on Figures 1(a) and 2(a) for males and females, respectively. However, statistically significant changes were recorded in body weight gains in male rats compared to the vehicle control group during the third (mild-dose group lower), the fourth (mild-dose group higher), the fifth (low-dose, mild-dose and high-dose groups higher) and the sixth week (mild-dose and high-dose groups lower; not shown). Even if those changes were statistically significant, they were not considered to be related to the treatment by GS-PC as they were no further noticed in subsequent weeks. During the recovery period, the body weight gains by male and female rats were found to be not statistically significant as compared to those of the control group of rats.
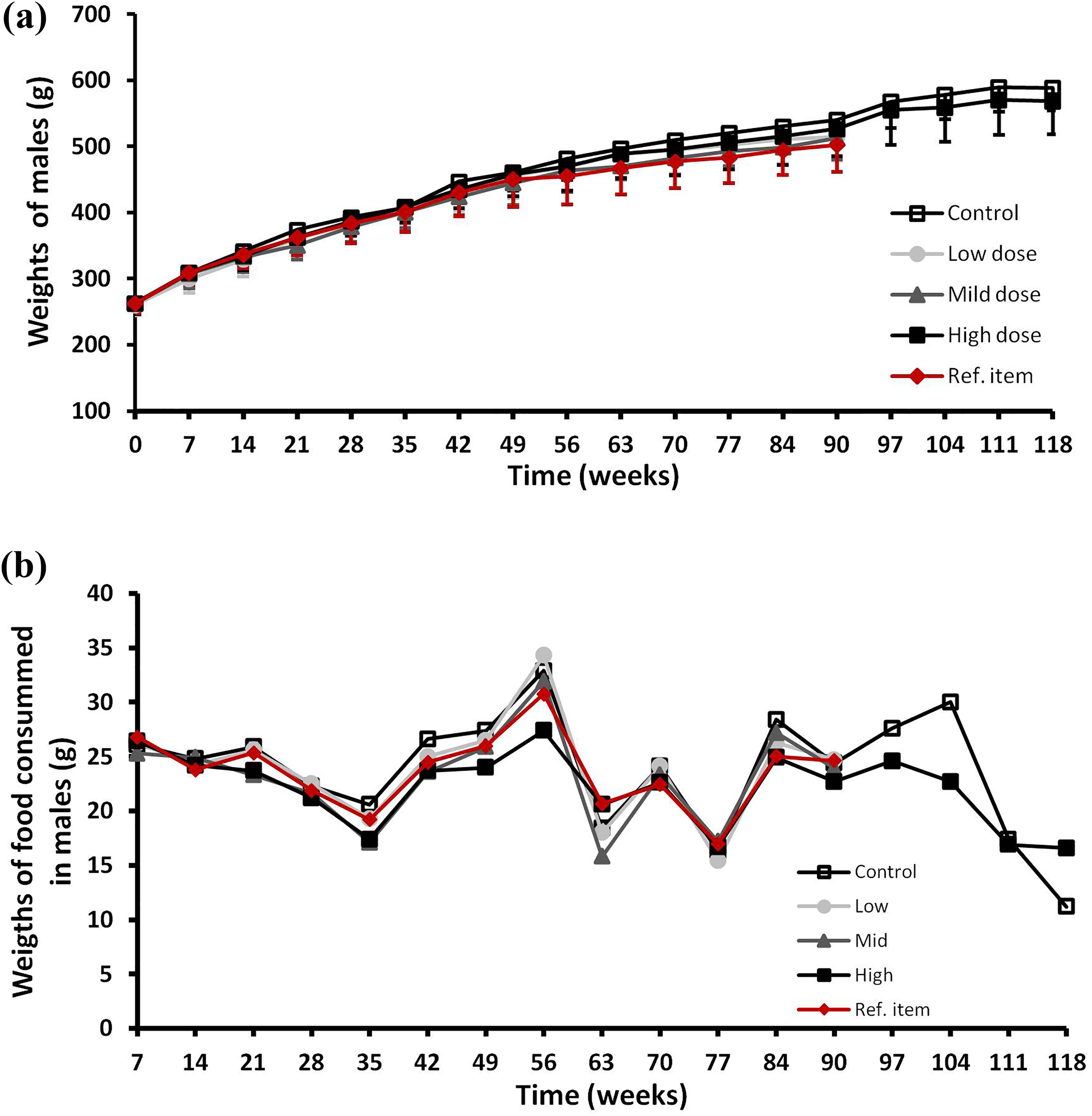
(a) Body weights and (b) food consumption over the treatment period (main and recovery periods) for male rats. Male rats were given orally and daily increasing dosing of Gs-PC (low, 250 mg/kg bw/day; mild, 2500 mg/kg bw/day and high, 4000 mg/kg bw/day) during 13 weeks and body weights (a) and food consumption (b) were recorded once a week, every week. The body weights and the food consumption were also recorded during four subsequent weeks (recovery groups) after the end of the 13-week treatment period for the control and the high-dose groups. Control group was treated with analytical grade water given orally and daily. Ref. item group was treated with Sp-PC given orally and daily at the dose of 2012 mg/kg bw/day.
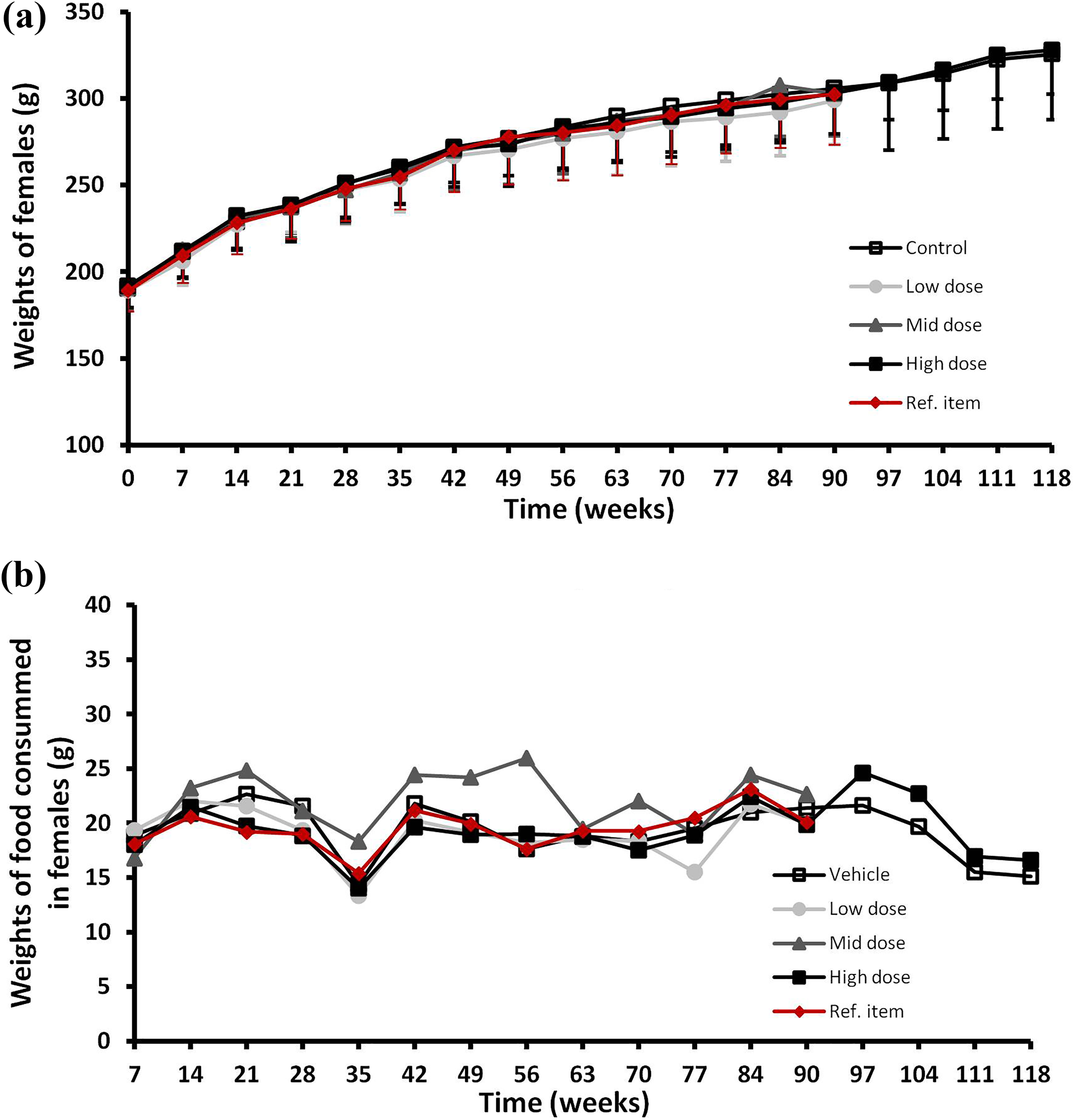
(a) Body weights and (b) food consumption over the treatment period (main and recovery periods) for female rats. Female rats were given orally and daily increasing dosing of Gs-PC (low, 250 mg/kg bw/day; mild, 2500 mg/kg bw/day and high, 4000 mg/kg bw/day) during 13 weeks and body weights (a) and food consumption (b) were recorded once a week, every week. The body weights and the food consumption were also recorded during four subsequent weeks (recovery groups) after the end of the 13 weeks treatment period for the control and the high-dose groups. Control group was treated with analytical grade water given orally and daily. Ref. item group was treated with Sp-PC given orally and daily at the dose of 2012 mg/kg bw/day.
The daily food consumptions by male and female rats of the treated groups at the different dose levels were not statistically different from those of the control group rats throughout the study and also during the recovery period. Those results were reported in Figures 1(b) and 2(b) for males and females, respectively.
Haematological parameters
The haematological parameters measured at the end of the study are resumed in Tables 4 and 5 for male and female rats, respectively. For male rats, a statistically significant increase in the packed cells volume for the mild-dose group was recorded as compared to the vehicle control group (Table 4). In addition, statistically significant decreases in the reticulocytes count for the mild dose, in the mean corpuscular haemoglobin concentration for the mild and high doses and for the Sp-PC-treated groups were recorded as compared to the vehicle control group (Table 4). Those differences were not further observed in the recovery groups (Table S2 in the Online supplementary data). For all other haematological parameters measured in male rats, no difference was observed between the Gs-PC- or the Sp-PC-treated groups, whatever the doses, as compared to the vehicle control group.
Haematological data for male rats at the end of the 90-day toxicology study.
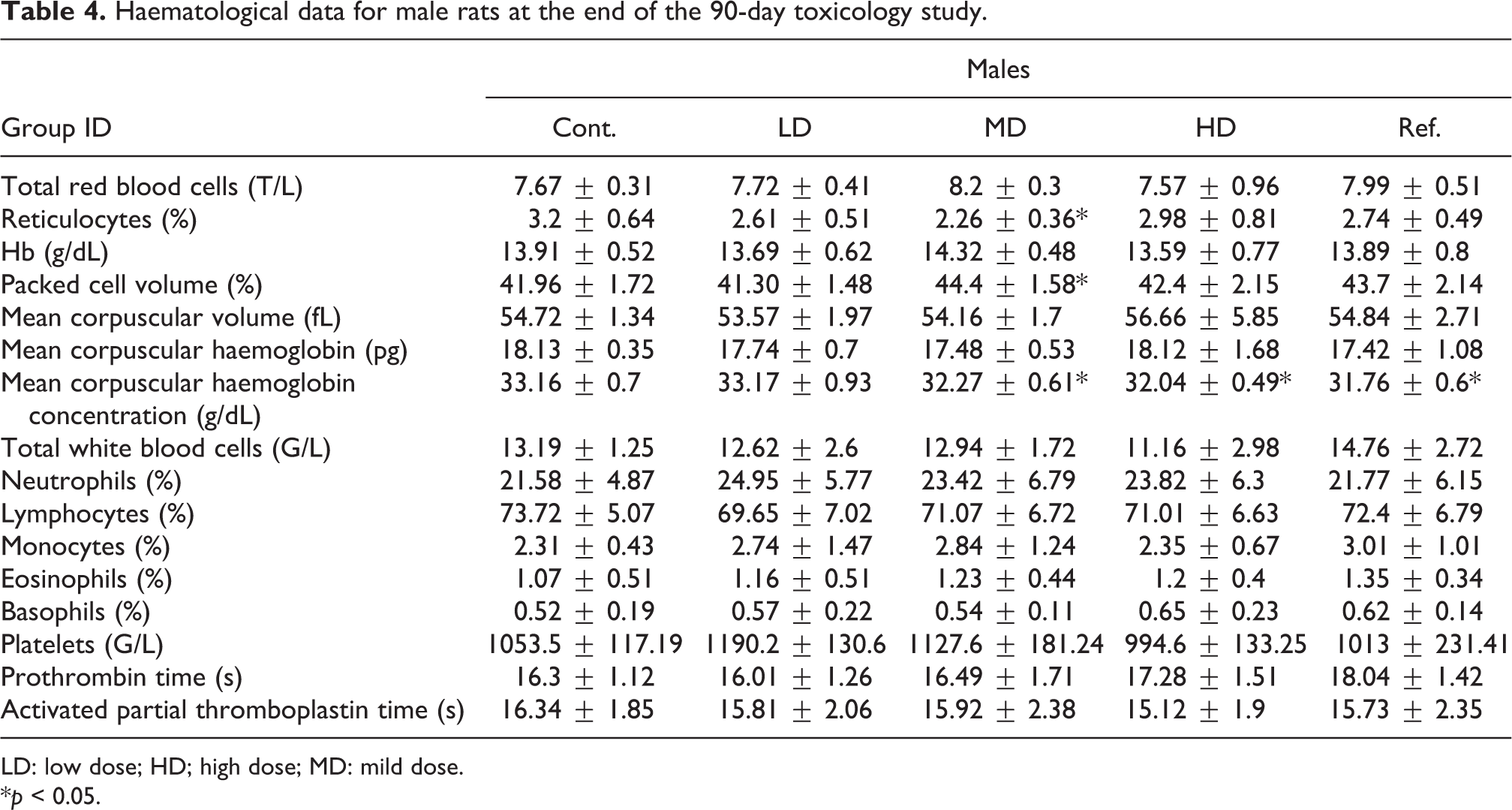
LD: low dose; HD; high dose; MD: mild dose.
*
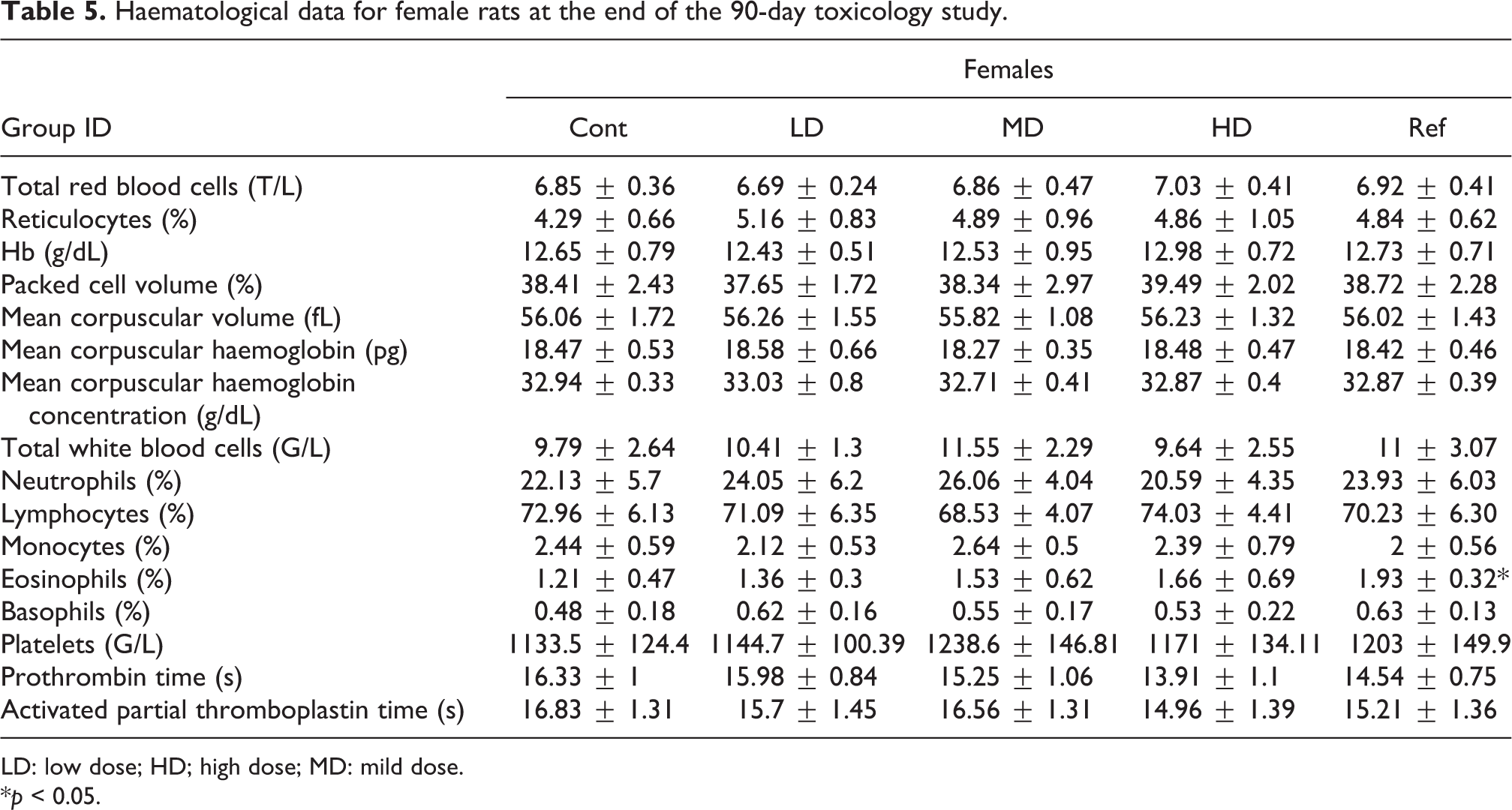
Haematological data for female rats at the end of the 90-day toxicology study.
LD: low dose; HD; high dose; MD: mild dose.
*
For female rats, a statistically significant increase in the eosinophil count was recorded for the Sp-PC-treated group as compared to the vehicle control group (Table 5). No difference was observed between the Gs-PC-treated groups, whatever the doses, as compared to the vehicle control group for haematological parameters (Table 5 and Table S3 in the Online supplementary data for recovery groups).
Clinical chemistry parameters
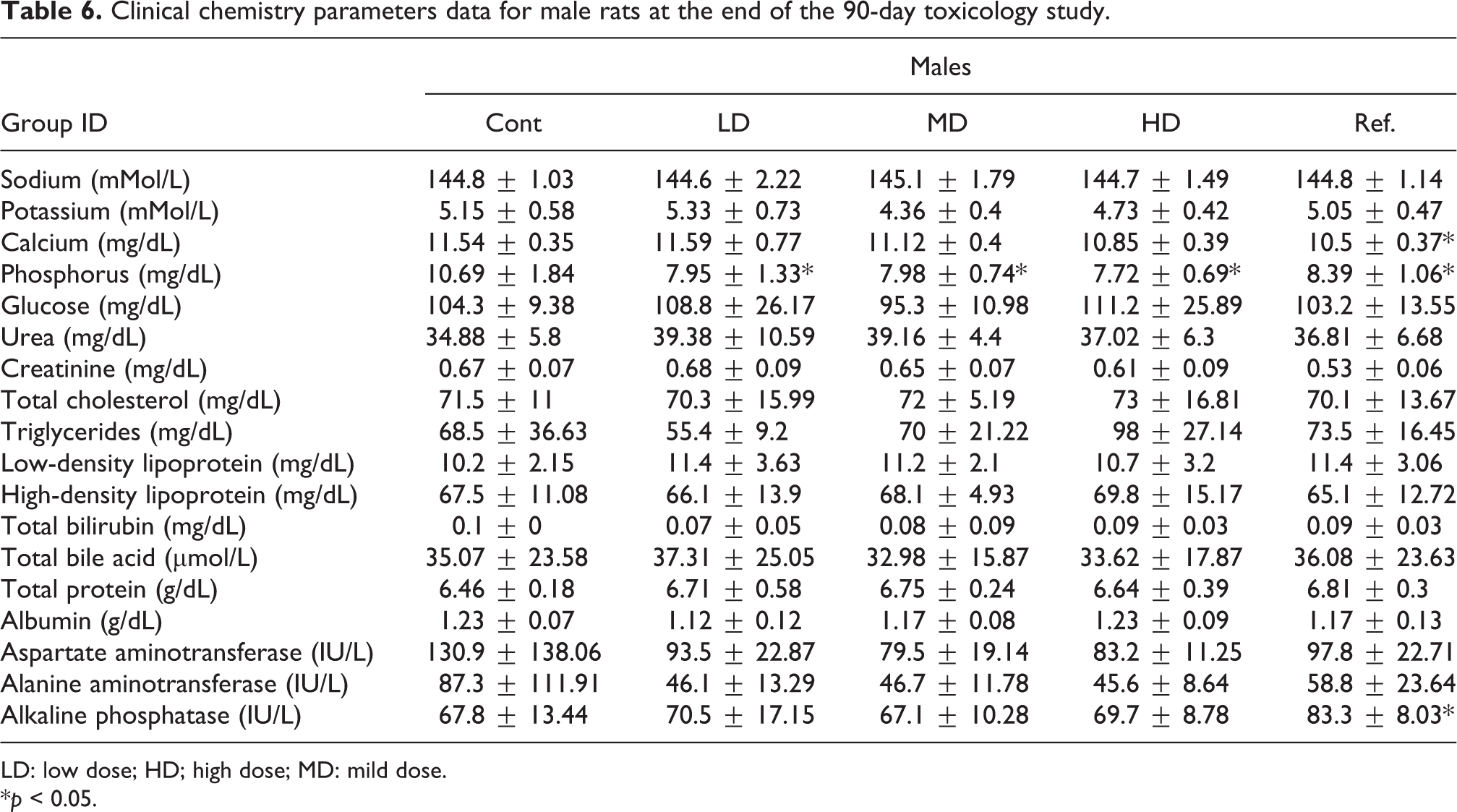
The clinical chemistry parameters measured for male and female rats at the end of the study are resumed in Tables 6 and 7, respectively. For male rats, statistically significant decreases in the blood phosphorus concentration for all the Gs-PC and for the Sp-PC-treated groups were recorded as compared to the vehicle control group (Table 6). At the end of the recovery period, a statistically significant decrease in blood calcium and triglycerides concentrations was recorded for the high-dose recovery group of Gs-PC as compared to their respective vehicle control recovery group (Table S4 in the Online supplementary data).
Clinical chemistry parameters data for male rats at the end of the 90-day toxicology study.
LD: low dose; HD; high dose; MD: mild dose.
*
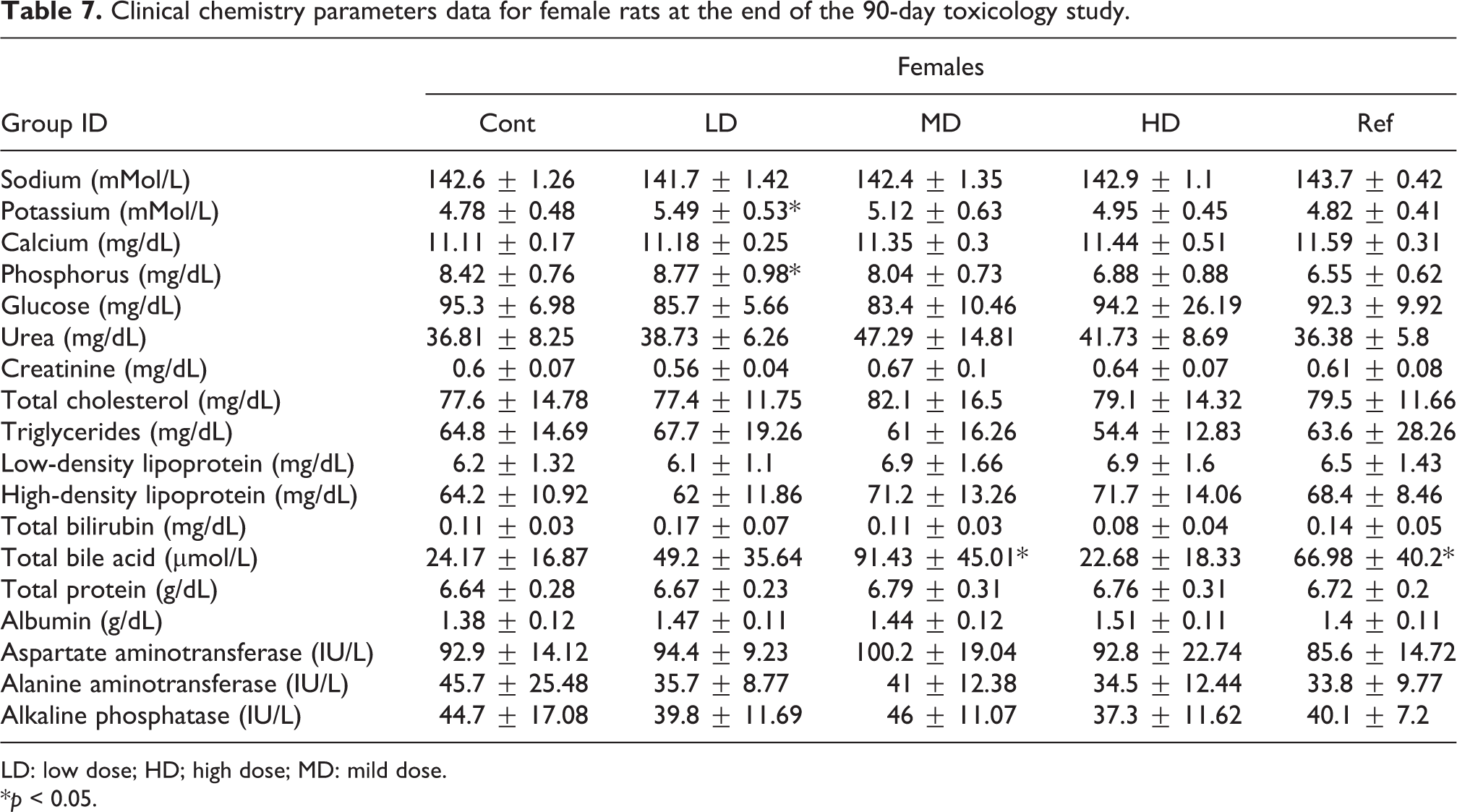
Clinical chemistry parameters data for female rats at the end of the 90-day toxicology study.
LD: low dose; HD; high dose; MD: mild dose.
*
Finally, a statistically significant increase in alkaline phosphatase and decrease in calcium concentrations were recorded for the Sp-PC males-treated group as compared to the vehicle control group (Table 6).
For female rats, statistically significant increases in the blood concentration of potassium and phosphorus were recorded for the low-dose-treated animals as compared to the vehicle control group (Table 7). In addition, statistically increases in blood total bile acid concentration were measured in the mild dose and in the Sp-PC-treated groups as compared to the vehicle-treated group (Table 7). No difference was observed between the high-dose recovery group and the control recovery group (Table S5).
Finally, for all other clinical chemistry parameters measured in male and female rats, no difference was observed between the Gs-PC- and the Sp-PC-treated groups, whatever the doses, as compared to the vehicle control group.
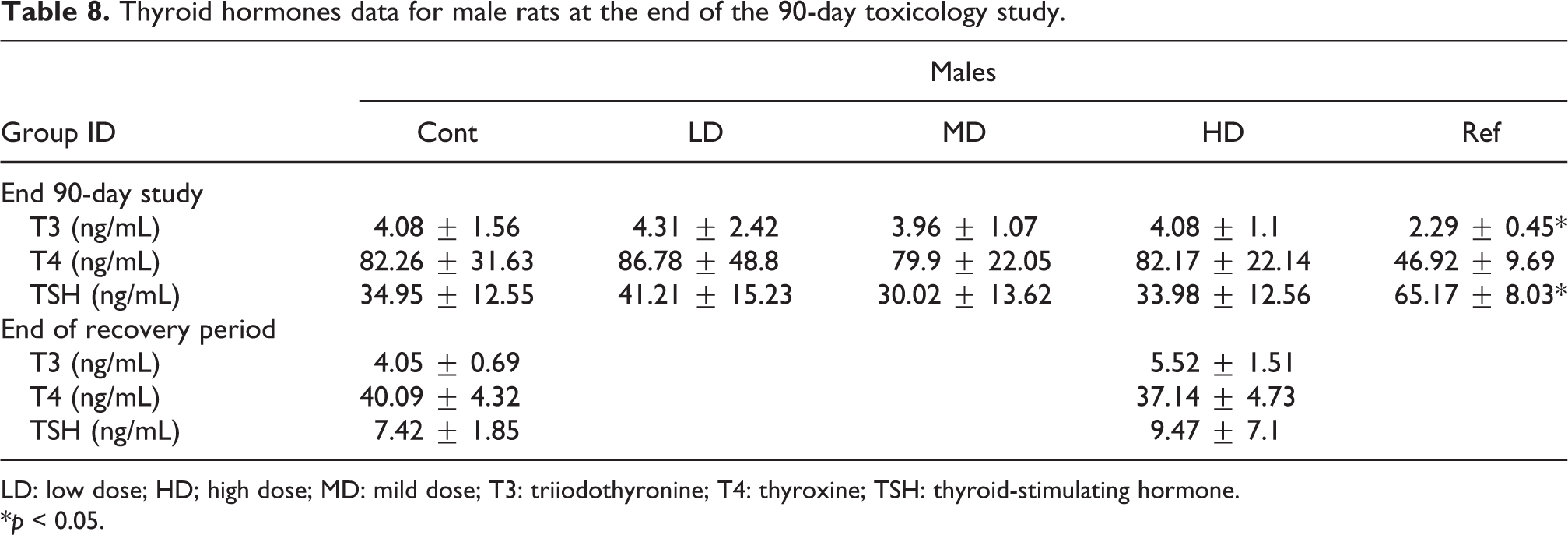
A statistically significant decrease in triiodothyronine (T3) and increase in TSH concentrations were recorded the SP-PC-treated group as compared to the vehicle control group for males (Table 8). The Gs-PC extract did not significantly modify the circulating serum levels of T3, thyroxine (T4) and TSH in female and male rats from 250 and up to 4000 mg/kg bw/day as compared to the vehicle control group (Tables 8 and 9).
Thyroid hormones data for male rats at the end of the 90-day toxicology study.
LD: low dose; HD; high dose; MD: mild dose; T3: triiodothyronine; T4: thyroxine; TSH: thyroid-stimulating hormone.
*
Thyroid hormones data for female rats at the end of the 90-day toxicology study.
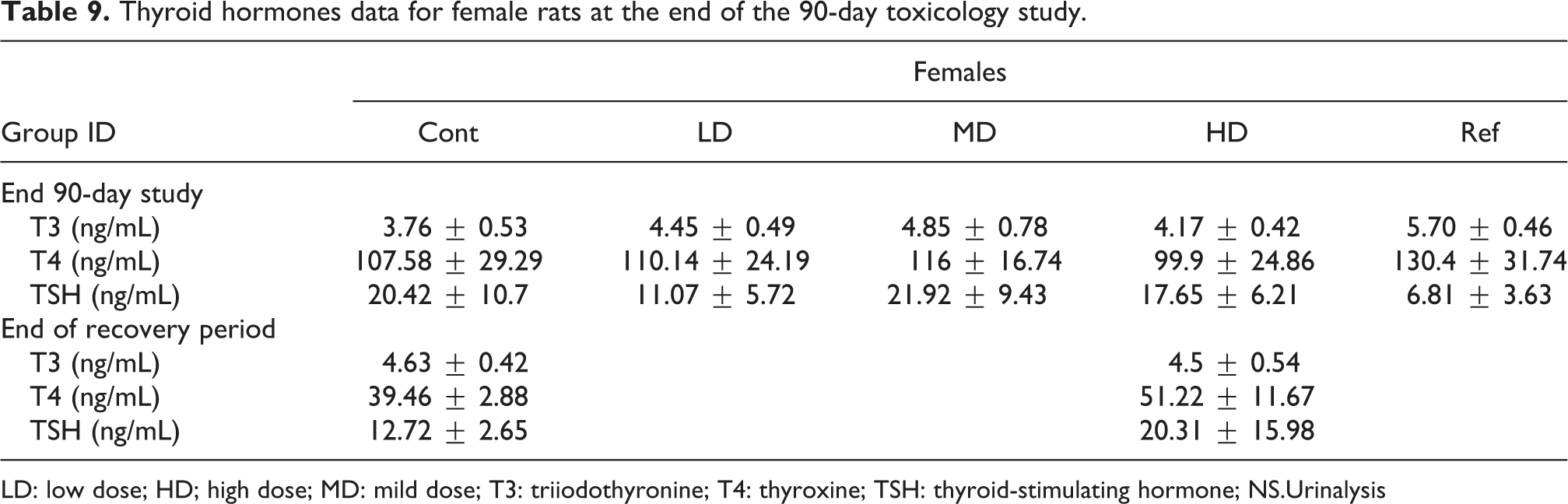
LD: low dose; HD; high dose; MD: mild dose; T3: triiodothyronine; T4: thyroxine; TSH: thyroid-stimulating hormone; NS.Urinalysis
Data from urinalysis at the end of the treatment period were not different between control vehicle group and all other groups treated with increasing doses of Gs-PC or with Sp-PC. In addition, there was no difference between the vehicle control and the high-dose-treated group at the end of the recovery period (not shown). Those results did not indicate any abnormality due to treatment with Gs-PC.
Organ weights
Statistically significant decrease in the weight of pituitary gland in males for the high-dose group was measured as compared to the control group (0.013 ± 0.002 g vs. 0.015 ± 0.001 g for the high-dose group and the control group, respectively, Table S6), but those values felt within the historical control range of the laboratory (0.009–0.026 g). Statistically significant increases in the weight of kidneys in females for the mild dose of Gs-PC and the Sp-PC-treated groups were recorded as compared to their respective vehicle control group at the end of the study (2.03 ± 0.22 g; 2.01 ± 0.21 g and 1.79 ± 0.14 g for the mild-dose Gs-PC, the Sp-PC and the control groups, respectively, Table S7). Nevertheless, those values felt within the historical control range of the laboratory (1.19–2.12 g). A statistically significant increase in the weight of thymus for Sp-PC-treated females group was recorded as compared to the vehicle control group (0.38 ± 0.05 g and 0.29 ± 0.05 g for the Sp-PC and the control groups, respectively, Table S7), but those values felt within the historical control range of the laboratory (0.29–0.5 g). Finally, a statistically significant increase in the weight of ovary in females for Sp-PC-treated group was recorded as compared to the vehicle control group (0.16 ± 0.01 g and 0.14 ± 0.01 g for the Sp-PC and the control groups, respectively, Table S7), but again those values felt within the historical control range of the laboratory (0.061–0.183 g). The weights for the other organs collected (see methods) did not statistically differ from those measured in their respective vehicle control group (Tables S6 and S7).
Gross pathology and histopathology
At the end of the study, rat tissues were collected; Gs-PC and Sp-PC at dose level up to 4000 mg/kg bw/day and 2012 mg/kg bw/day, respectively, did not induce any gross pathological alterations at the detailed necropsy examination.
The histopathological examinations of the tissues of male and female rats from the control group and those treated at the high dose of Gs-PC did not reveal any significant treatment-related alterations. Nevertheless, some various histopathological changes were noticed in several organs but those changes were considered as incidental. They are detailed in the following and in Tables S8 and S9 in the Online supplementary data for male and female rats, respectively. In the vehicle control group and in the high-dose group, a duct hyperplasia associated to a mononuclear cell infiltration, a focal necrosis and a cytoplasmic vacuolization in liver were observed. Cystic glands in glandular stomach, focal mineralization and focal tubular dilation and unilateral dilation of pelvis in kidneys and focal increased vacuolization in cortex in adrenals were also noticed. Focal interstitial cell infiltration and accumulation of foam cells in lungs, lymphoid hyperplasia in rectum and colon, an increased lymphocyte in paracortex in mesenteric lymph node, a focal myocardial infiltrate associated to a fibrosis in heart and a focal interstitial lymphocytic aggregation in prostate were also noticed. Finally, pituitary cysts and a degeneration/atrophy of seminiferous tubules and giant spermatid in testes were observed. These changes were observed to the same frequency both in vehicle control and Gs-PC-treated groups and were not considered to be of any toxicological concern and related to the treatment as they are noted as common background changes in the rats housed in laboratory conditions.
In one rat from the high-dose group, a global but unilateral retinal atrophy showed a classical picture of decreased number of cells in all layers of retina and collapse of the plexiform layers. It is known that this lesion could be hereditary or could occur during the process of aging. In the same animal, a decrease in lymphocyte cellularity in cortex of thymus and a decrease in lymphocyte cellularity in white pulp of spleen were noticed, and those changes were considered to be related to some stress caused by retinal atrophy and not related to the Gs-PC treatment.
Discussion
This study presented results about the safety evaluation of a soluble fraction enriched with 30% of phycocyanin from
For the oral toxicity study, the highest dose was chosen according to literature data and for practical reason. According to literature data, a dose of 5000 mg/kg/day of
The genotoxicity of Gs-PC was assessed using two kind of in vitro tests. The first test was performed in bacteria (bacterial reverse mutation test, i.e. Ames test) and measured the mutagenic potential of chemicals, whereas the second test was performed in mammalian lymphoma cells (micronucleus assay in L5178Y TK+/− mouse lymphoma cells) and measured their clastogenic and aneugenic potential.
Results from the bacterial reverse mutation test showed that Gs-PC did not increase the number of bacterial revertants. The same results were observed in the presence of a metabolic activation system. As indicated in the method section, Gs-PC contains 54% of proteins. It should be noted that testing of protein in the Ames test may give rise to some artifactual results and should be interpreted with caution. Indeed, the potential for release of histidine from the protein extract in the treatment medium could induce an increase in the number of bacterial revertants colonies independently of a truly mutagenic activity.
30
Nevertheless, in the present study, this artifactual event seems to be unlikely since no increase in bacterial count was ever observed at any concentrations tested. It can, therefore, be concluded that Gs-PC did not show any mutagenic activity in the bacterial reverse mutation test with
The results obtained from the repeated doses 90-day oral toxicity study performed in female and male rats globally showed that Gs-PC did not exert any remarkable clinical, physiological, biochemical or histopathological alterations. Although some minor findings were observed, they were not considered to be related to the treatment. For examples, some changes were recorded for the pituitary glands, the kidneys or the ovary weights between treated and control groups. Nevertheless, those modifications were not considered to be treatment-related changes as they were comparable to the historical control values at the facility. Haematological parameters at the end of the study showed a significant increase in the packed cells volume associated to a slight but not significant increase in total red blood cell in addition to a significant decrease in the reticulocytes count and in the mean corpuscular haemoglobin concentration for the mild dose. Nevertheless, those values fell in between the values of historical control values at the facility. In addition, the decrease observed for the reticulocytes count was not dose dependent as it was not observed for treatment up to 4000 mg/kg bw/day. For these reasons, haematological changes were not considered to be related to Gs-PC treatment. Finally, Gs-PC neither affected the serum levels of thyroid hormone nor the weight or the histology of thymus or thyroid gland in animals from both sexes. Thus, Gs-PC did not present any endocrine disruptor activity at least on the thyroid axis.
A previous study with a high-protein biomass of
The tests, carried out above, significantly exceed the doses that would be seen in real usage; the World Health Organization recommended daily amount of protein ranges from 0.66 g/kg bw/day both for adult men and for women 34 and at the maximum dose level used in the present 90-day oral toxicity study (4000 mg/kg bw/day at over 50% protein), the recommended daily protein amount was exceeded by a factor around 3-fold. Nonetheless, even at this extreme dose, all clinical, hematological, biochemical and histopathological findings were generally negative.
Conclusion
In the present study, several assays were conducted to assess the risks associated to an extract of
Supplemental Material
Supplementary_Data - The safety evaluation of phycocyanin-enriched Galdieria sulphuraria extract using 90-day toxicity study in rats and in vitro genotoxicity studies
Supplementary_Data for The safety evaluation of phycocyanin-enriched
Footnotes
Acknowledgements
We would like to thank all Fermentalg employees who contributed to the production of phycocyanin from
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AAA, JD, SB-V, CA, HG and OC are employees of Fermentalg; SK and AC are members of the University of Bordeaux and received financial support to write the manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
