Abstract
Clostridium butyricum MIYAIRI 588 (CBM 588) is a nonpathogenic, anaerobic, gram-positive bacillus characterized by the production of short-chain fatty acids, including butyrate. The safety and tolerance of CBM 588 was investigated as a feed additive for broiler chickens, weaned piglets, and turkeys. CBM 588 administered to broilers at doses up to 5 × 107 CFU/g feed for 42 days produced no detrimental effects on zootechnical performance, natural mortality, hematology, or biochemical parameters. Piglets receiving CBM 588 at doses up to 5 × 107 CFU/g feed for 42 days showed no significant differences from controls in zootechnical performance, mortality, or morbidity. Finally, CBM 588 administered to turkeys at doses up to 2.5 × 107 CFU/g feed for 84 days produced no detrimental effects on zootechnical performance, hematology, or biochemical parameters. Some improvements in zootechnical performance were seen with CBM 588, including improved average daily gain (ADG) and feed conversion for broilers from days 1 to 21 as well as final body weight and overall ADG for turkeys. Overall, CBM 588 administered in feed at dose up to 5 × 107 CFU/g (broilers and piglets) or 2.5 × 107 CFU/g (turkeys) was shown to be safe and well-tolerated in all tested animals and may provide some nutritional benefit when added to standard commercial feed.
Introduction
Clostridium butyricum is a gram-positive bacillus found frequently in the environment as well as in the gastrointestinal tract of animals. C. butyricum MIYAIRI 588 (CBM 588) is a strain of C. butyricum that is differentiated from other strains using KM1 bacteriophage susceptibility. This nongenetically modified, obligate anaerobe lacks pathogenicity and toxigenicity, as confirmed by the following: (1) lack of transmissible antibiotic resistance, (2) absence of plasmids with mobile genetic elements, (3) absence of clostridial toxin genes and products (botulinum neurotoxins A, B, E, and F, genes encoding for Clostridium perfringens toxins α, β, ε), (4) absence of pathogenic markers via genomic analyses, and (5) lack of hemolytic potential. 1 The safety of CBM 588 has been demonstrated in a series of studies, including genotoxicity, acute toxicity in rats, 5-week repeat oral dose toxicity in dogs, subchronic toxicity in rats, and two-generation reproduction/teratology study in mice. 1 –4
Microbial fermentation of carbohydrates in the gastrointestinal tract results in the production of short-chain fatty acids (SCFAs), including acetate, propionate, and butyrate. SCFAs are an important energy source for the animal and are readily used for tissue maintenance, growth, and lipogenesis. 5 –7 Both C. butyricum in general 8 –10 and CBM 588 specifically 11,12 have been shown to produce high levels of butyrate. CBM 588 has also been shown to increase the relative abundance of other SCFAs-producing gut bacteria, including Bifidobacterium, Coprococcus, and Bacteroides species. 13 The administration of CBM 588 to rats was shown to significantly increase butyrate, propionate, and acetate concentrations in the cecum. 14
Butyrate can be metabolized by nearly all tissues in the body and serves as the preferred energy source for colonocytes to stimulate their multiplication and differentiation. 5,7,15 –18 As such, butyrate production plays an important role in the maintenance of gut health. 16,19 SCFAs promote the absorption of water and sodium from the large bowel, with butyrate having the strongest effect. 18 This is achieved by the formation of SCFA gradients across the colonic epithelium, resulting in transient intracellular acidification and increased active transport of sodium and chloride into colonocytes. 20 –22 These physiological and nutritional effects have been well documented in livestock animals, including pigs and poultry. 23,24
As a producer of butyrate, CBM 588 is intended for use as an additive to enhance the nutritional value of feed for chickens, piglets, and turkeys. This manuscript reports the safety and tolerance of CBM 588 as a feed additive in these animals.
Materials and methods
Organic acid production
The production of organic acids by CBM 588, C. butyricum, and other bacteria was analyzed as previously reported. 12 All bacteria were cultured using Gifu Anaerobic Medium broth at 37°C for 48 h under anaerobic conditions prior to analysis by high-performance liquid chromatography.
Study design
Studies were performed to evaluate the tolerance of CBM 588 in broilers, piglets, and turkeys. Studies followed the principles of the Guidelines on Good Clinical Practice for Clinical Trials for Registration of Veterinary Medicinal Products, 25 European Union (EU) Principles for Animal Care and Experimentation, and appropriate quality standards as indicated by the European Food Safety Authority. 26 Tolerance doses were based on the intended commercial doses in the EU, which are 5 × 105 CFU/g feed for broilers and piglets and 1.25 × 105 CFU/g feed for turkeys. The maximum tested dose was set to 100× the intended dose for all studies. However, the dose intended for use in turkeys was halved before execution of the study, resulting in a maximum tested dose of 200× the final commercial dose.
Broiler tolerance study, IRTA B-307
A total of 384 Ross 308 broiler chickens (1-day old) were used and sexed at the hatchery. Breeder flock history and vaccination history of the broilers were recorded. Procedures were in agreement with the Ethical Committee for Animal Care and Experimentation of IRTA.
The study was carried out in the broiler cage unit of the IRTA experimental facilities, Valls, Catalunya, Spain. Birds were kept in a clean room and housed in 96 cages, each having an area of 0.305 m2 and a height of 37 cm. Stocking density was approximately 13 chicks/m2, similar to that practiced commercially. The room was supplied with artificial, programmable lights, automated aerotherm heating, and forced ventilation. Temperature inside the room was set at 33–35°C at study start and decreased by 3°C each week. From day 28 until study end, the temperature was set at 22°C. The lighting program was 23-h light and 1-h dark during each 24-h period. The study terminated when birds were 43 days of age.
The study design was blocked with four dietary treatments (described subsequently). Each treatment was administered to 24 cages, with each cage containing four broilers of the same sex. Thus, 12 cages of males and 12 cages of females were used per treatment (n = 48/sex/group). Replicates (cages) were spatially allocated within the house to minimize potential cross-contamination between treatments. Empty cages were used to separate blocks of treatment groups. To further safeguard against cross-contamination, personnel handled control animals first, followed by treated animals from low to high dose.
All animals received starter diet from days 1 to 21 and finisher diet from days 22 to 43. Diets are described in detail in Supplementary Table 1. The control group received the basal diet containing 10 g/kg starch. This starch content was replaced with CBM 588 powder as necessary (up to 10 g/kg) to reach the desired experimental concentration for the treatment groups. The test concentrations of CBM 588 were 5 × 105, 5 × 106, and 5 × 107 CFU/g feed. Feed analysis to ensure correct nutrient and CBM 588 content was conducted using AOAC methods on the day of manufacture. Diets were formulated without antimicrobials other than coccidiostats. Mash feed and potable water (from a local public source) were available ad libitum.
Observations were made, and data were collected as described in Table 1.
Detailed observations for broiler tolerance study.
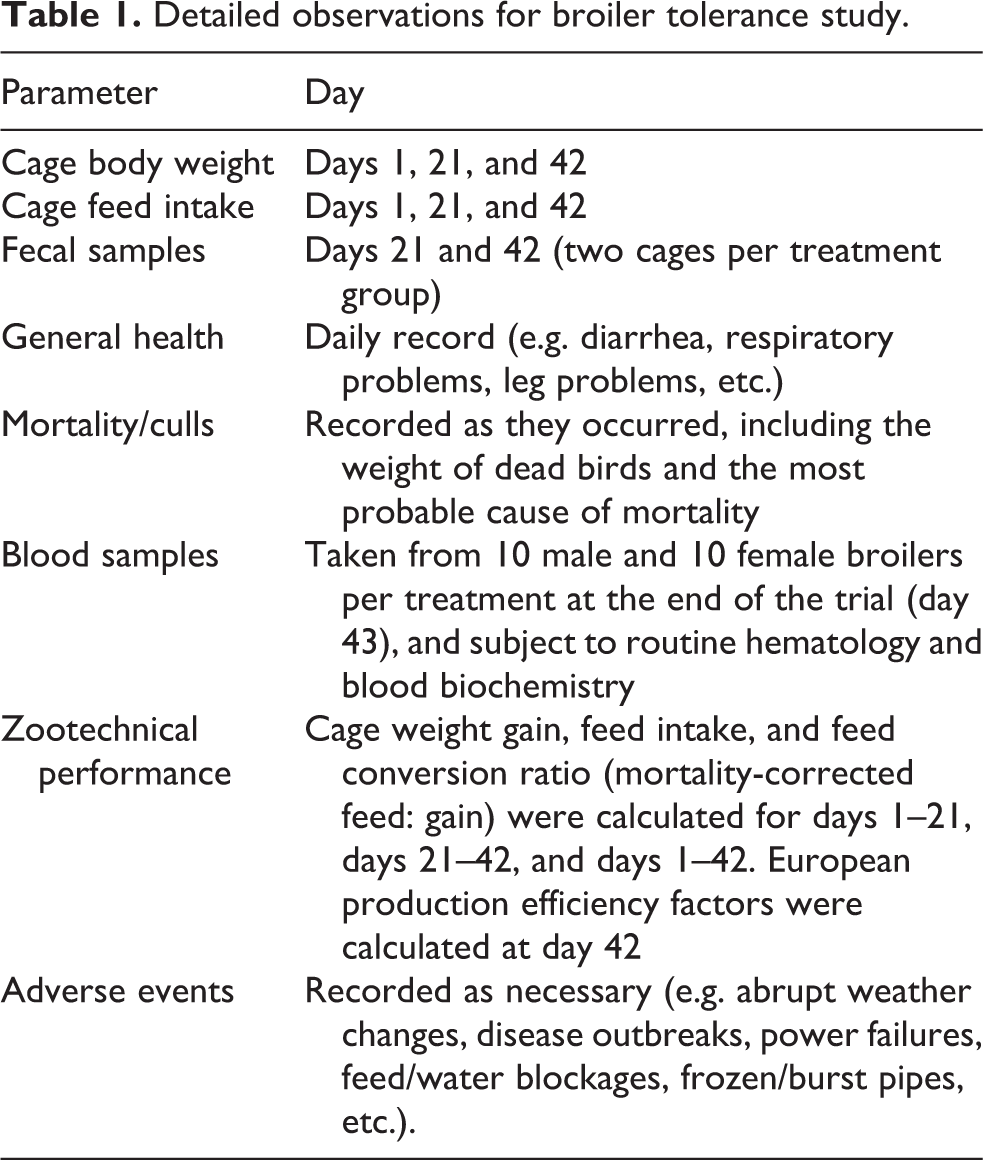
Results were analyzed using analysis of variance (ANOVA) as a completely randomized design by the generalized linear model (GLM) of SAS v.8 (SAS, 1999-2001). Statistical significance was declared at p ≤ 0.05, and 0.05 ≤ p ≤ 0.10 was considered a near-significant trend. Performance variables were analyzed as a block design with the four treatments and two sexes in a factorial arrangement and three blocks (tier location). The individual cage with four male or four female chickens (at the beginning of the trial) was the replicate unit for statistical purposes. Results from hematological and blood biochemical variables (1 bird per cage from 10 male cages and 10 female cages per treatment) were analyzed as a factorial design with four treatments and two sexes as the main factors and one bird as the replicate unit. Data were analyzed to detect possible outliers; a value was considered an outlier if it was outside the range defined by (treatment mean) ± (3 × pooled Standard Error of the Mean [SEM]). Results from microbiological counts below the limit of detection (absence) were assigned the half value of this limit and analyzed by a nonparametric test.
Piglet tolerance study, CCL-329-03-V-OO
Seventy-two healthy, mixed-sex, weaned piglets aged 28 days (Topigs Tempo boar × Topigs 20 sow) were selected from a single batch of piglets (eight litters born within a single week). Piglets were randomly assigned to the experimental treatments on the basis of body weight (BW), gender, and ancestry such that each treatment group was balanced at the start of the trial.
This study was conducted at the Laverdonk Research Farm (Heeswijk-Dinther, The Netherlands). Three identical nursery units with six pens each (four piglets per pen) were used simultaneously for this study. The housing conditions used were typical for pig husbandry in the trial location and followed Dutch IKB farm standards. Piglets were housed in pens measuring 2.60 × 1.20 m2 (area 3.12 m2). The floor in the pens was a fully slatted, plastic floor. Computer-controlled heating and mechanical ventilation systems were used to ensure similar climatic conditions between nursery units. Temperature at the onset of the study was set to 28°C, and temperature was decreased gradually to 22°C by week 5. Any effects of nursery unit on performance and health traits were considered absent or negligible, due to the use of similar housing conditions, climate control systems, and simultaneous testing.
To avoid cross-contamination with the microbial additive between pens of different treatments, each experimental treatment was assigned to pens within a single unit. Control piglets were fed, handled, and weighed first, and attendants changed boots and overalls before handling animals receiving CBM 588. Each type of feed (control, low dose, high dose) was identified by the addition of an inert color marker (red, blue, yellow) to prevent incorrect test article administration.
All diets were formulated without antibiotics and met or exceeded nutrient requirements recommended by the Dutch Centraal Veevoeder Bureau for piglet nutrition and the nutrient requirements of the National Research Council (NRC). 27 Piglets had access to a commercial creep feed during nursing. From weaning (day 0) until day 13, prestarter feed was provided. On day 14, a mixture of 50% prestarter and 50% starter feed was provided, followed by starter feed only from day 15 onward. All diets were presented as meal (not pelleted) and provided ad libitum. Potable water (from a local public source) was also provided ad libitum. Composition of the diets is detailed in Supplementary Table 2.
There were three treatment groups: control (basal diet, no CBM 588), basal diet + CBM 588 5 × 105 CFU/g feed, and basal diet + CBM 588 5 × 107 CFU/g feed (n = 24/group). CBM 588 was added to the diets in place of starch. Feed samples were tested at manufacture and after 14 and 42 days to verify CBM 588 content.
Observations were made, and data were collected as described in Table 2.
Detailed observations for piglet tolerance study.

Zootechnical performance data were analyzed by ANOVA (using Genstat software, VSNi, UK) corrected with Tukey’s method. The pen within a unit was incorporated as block factor (i.e. pen 1 within units 1, 2, and 3 is block 1; pen 2 within units 1, 2, and 3 is block 2; etc.). In addition, the following linear contrasts were analyzed: T1 versus T2, T1 versus T3, and T2 versus T3. The following model was used
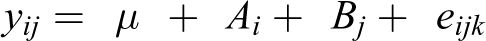
where y represents the response variable, µ represents overall mean, A represents pen within a unit, B represents treatment, and e represents error. The χ 2 procedure was used for the number of culled or lost animals and number of treatment animals. An ordinal regression analysis, following the McGullagh threshold model, was used to analyze the fecal scores. Significance was defined as probability p ≤ 0.05, with 0.05 < p ≤ 0.10 considered a near-significant trend.
Turkey tolerance study, MGT01/2011
Eight-hundred female day-old turkeys (Big 7) were purchased from a local commercial source. Birds were not routinely vaccinated. The study was conducted according to the principles set out in the relevant parts of Commission Regulation (EC) no. 429/2008 of April 25, 2008 on detailed rules for the implementation of Regulation (EC) no 1831/2003 of the European Parliament and of the Council as regards the preparation and the presentation of applications and the assessment and the authorization of feed additives.
The trial was conducted by the Department of Poultry Science, Olsztyn University of Warmia and Mazury, Olsztyn, Warmian-Masurian Voivodeship, Poland. Turkeys were housed in a clean, disinfected room on fresh wood shavings. Birds were randomly distributed into 32 floor pens, each containing 25 turkeys. Pens of different treatment groups were divided by physical barriers to avoid cross-contamination. Control animals were handled first, and attendants changed gloves and boot covers before handling animals receiving CBM 588.
Feeds met or exceeded nutritional requirements for turkeys according to NRC guidance. 28 Diets were formulated without probiotics (other than CBM 588 as indicated), enzymes, coccidiostats, veterinary antibiotics, or antibiotic alternatives (e.g. organic acids/salts, high copper/zinc, etc.). All nutrients were supplied in normal concentrations (i.e. to meet nutritional needs, not exceeding EU maximum permitted concentrations). Starter feed was provided during weeks 1–4, grower feed during weeks 5–8, and finisher feed during weeks 9–12. Diets were fed ad libitum in meal form. Potable water (from a local public source) was also available ad libitum. The composition of all diets is detailed in Supplementary Table 3.
There were four dietary treatment groups: control (basal diet, no CBM 588), basal diet + CBM 588 1.25 × 105 CFU/g, basal diet + CBM 588 2.5 × 105 CFU/g, and basal diet + CBM 588 2.5 × 107 CFU/g. Feeds containing CBM 588 were prepared after the control feed to prevent cross-contamination. All diets were calculated to be isonutritive. Feed samples were tested at manufacture and after 21, 42, and 84 days to verify CBM 588 content and homogeneity of mixing in feeds.
Observations were made, and data were collected as described in Table 3.
Detailed observations for turkey tolerance study.
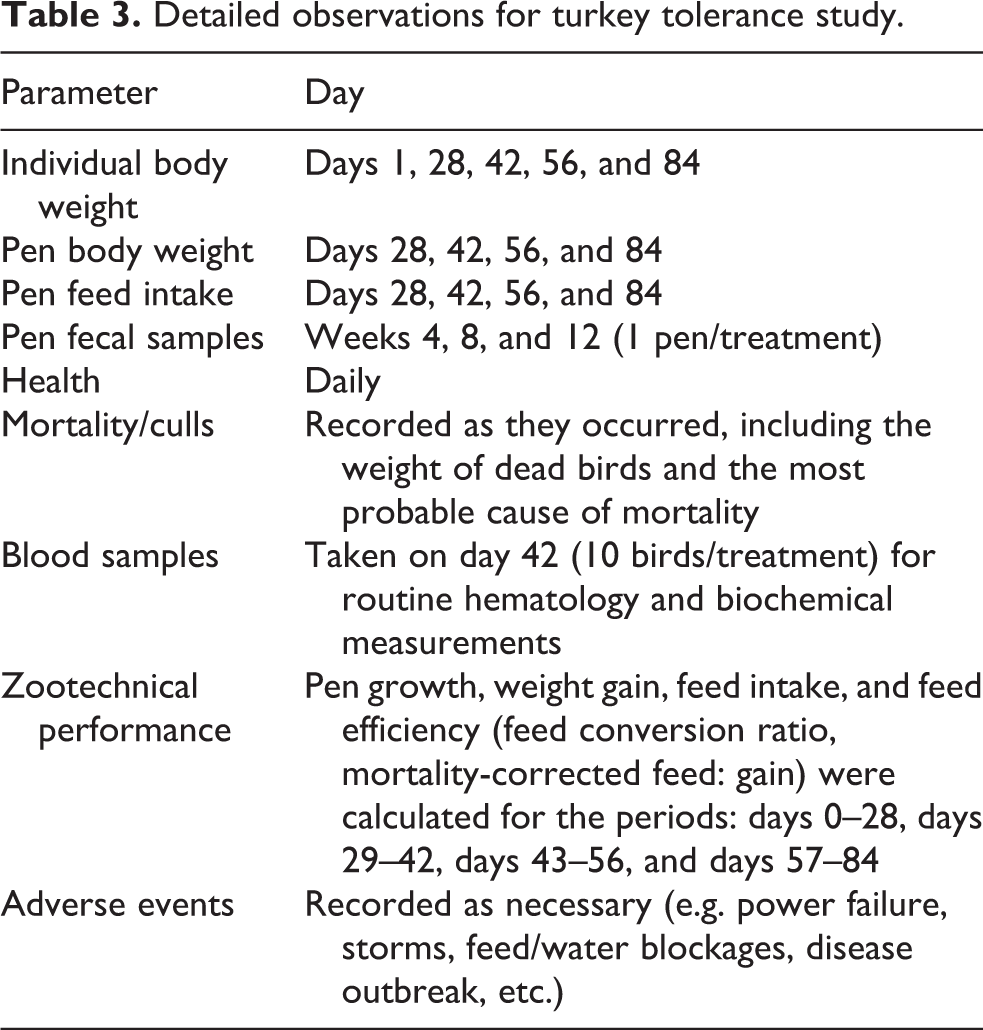
Zootechnical performance data were analyzed by ANOVA, with the pen as the experimental unit. The applied experimental model was a randomized complete block design of four treatments allocated into eight blocks. Probability was obtained from the F value in the ANOVA table. Significance was declared at p ≤ 0.05, with 0.05 < p ≤0.10 considered a near-significant trend. The GLM procedure of Statistica for Windows, version 9.0, was used. ANOVA was also used to analyze mortality/culls and hematology/biochemical parameters, with treatment as the basic experimental unit.
Results
Organic acid production
To confirm that CMB 588 generates high levels of butyrate, the production of organic acids by a variety of bacteria known to produce SCFA was compared. C. butyricum CBM 588, C. butyricum ATCC19398T, Bifidobacterium adolescentis JCM1275T, Bifidobacterium bifidum JCM1209, Bifidobacterium breve JCM1192T, Bifidobacterium infantis JCM1222T, Bifidobacterium longum JCM1217T, Enterococcus faecium JCM5804T, Enterococcus faecalis JCM5803T, Lactobacillus casei JCM1134T, Lactobacillus plantarum JCM1149T, and Lactobacillus acidophilus JCM1132T were analyzed for their ability to produce acetate, propionate, butyrate, SCFA (total), succinate, and lactate. The results of this analysis are presented in Table 4. While all tested species produced notable amounts of SCFA, only CBM 588 and C. butyricum ATCC19398T produced detectable amounts of butyrate. For both strains, butyrate represented approximately 40% of the total SCFA generated.
Production of short-chain fatty acids, succinate and lactate concentrations by Clostridium butyricum, lactic acid bacteria, and bifidobacterial.a
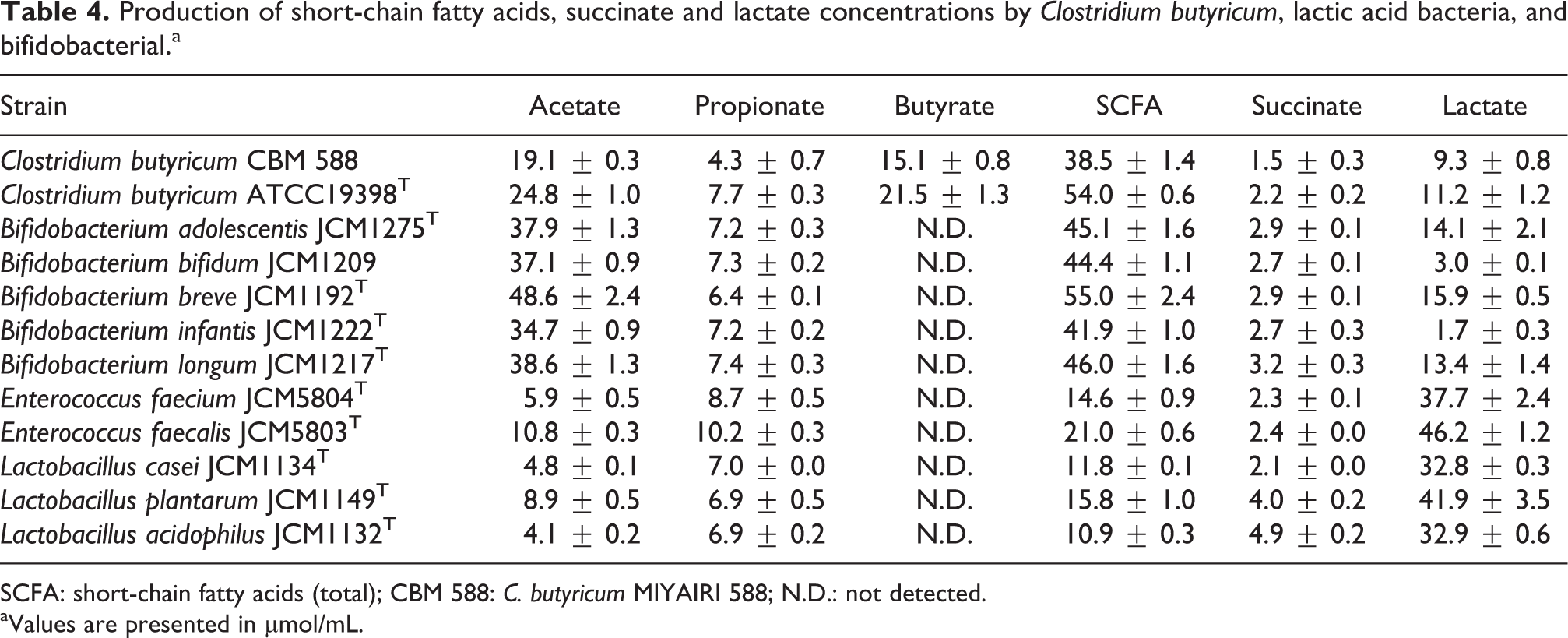
SCFA: short-chain fatty acids (total); CBM 588: C. butyricum MIYAIRI 588; N.D.: not detected.
aValues are presented in µmol/mL.
Broiler tolerance study
Feed analysis confirmed proper nutrient content of all study diets, and the nutrient content was similar for all treatments. The analyzed counts of CBM 588 in feeds were in line with target values for all treatments (see Supplementary Table 4). Fecal samples were analyzed for the presence of CBM 588 and showed a dose-dependent increase in CBM 588 counts at both day 21 (1.70 (below the limit of detection), 2.23, 3.79, and 5.29 log10 CFU/g) and day 42 (2.00, 3.17, 3.39, and 5.46 log10 CFU/g). These data confirmed exposure of birds to CBM 588 as intended by the study design.
Zootechnical performance data over the entire study period (42 days) are summarized in Table 5. Significant differences were detected between treatments within the first period of the trial (days 1–21). Birds from T4 had a lower average daily feed intake (ADFI) than other groups. As BW and average daily gain (ADG) were not significantly different, the lower ADFI was a consequence of improved (near-significant, p = 0.056) feed conversion ratio (FCR, mortality-corrected feed: gain). Similarly, in the second period of trial (days 21–42), birds from T2 had a significantly lower ADFI than other groups; this again led to significantly improved FCR. Significant differences were also detected for ADG during this second period, with T3 significantly lower than T1 and T2. Over the whole trial period (days 1–42), no significant differences were detected between treatments for final BW, ADG, and ADFI. FCR was significantly improved for T2 birds compared to T1 and T3.
Zootechnical performance of broilers.

BW: body weight; ADG: average daily gain; ADFI: average daily food intake; FCR: feed conversion ratio; EPEF: European production efficiency factor.
aAt day 28, a water blockage affected some cages of the lower tier of females; as a consequence, 1 female of T2, 3 females of T3, and 4 females of T4 died; these mortality data were not included in the analysis and the results presented in this table. Different superscripts denote differences at bc p ≤ 0.05 and AB p ≤ 0.01. n = 12/sex/group.
No significant differences were detected in mortality between groups, and the overall incidence of mortality was low at less than 2%. No deaths were attributed to the presence of CBM 588 in the feed.
Results from hematological and biochemical analyses of blood samples taken on day 43 are presented in Table 6. No significant differences were detected between groups for any of the variables studied. Moreover, values were similar between treatments.
Hematological and biochemical analyses of blood samples taken from broilers on day 43.a
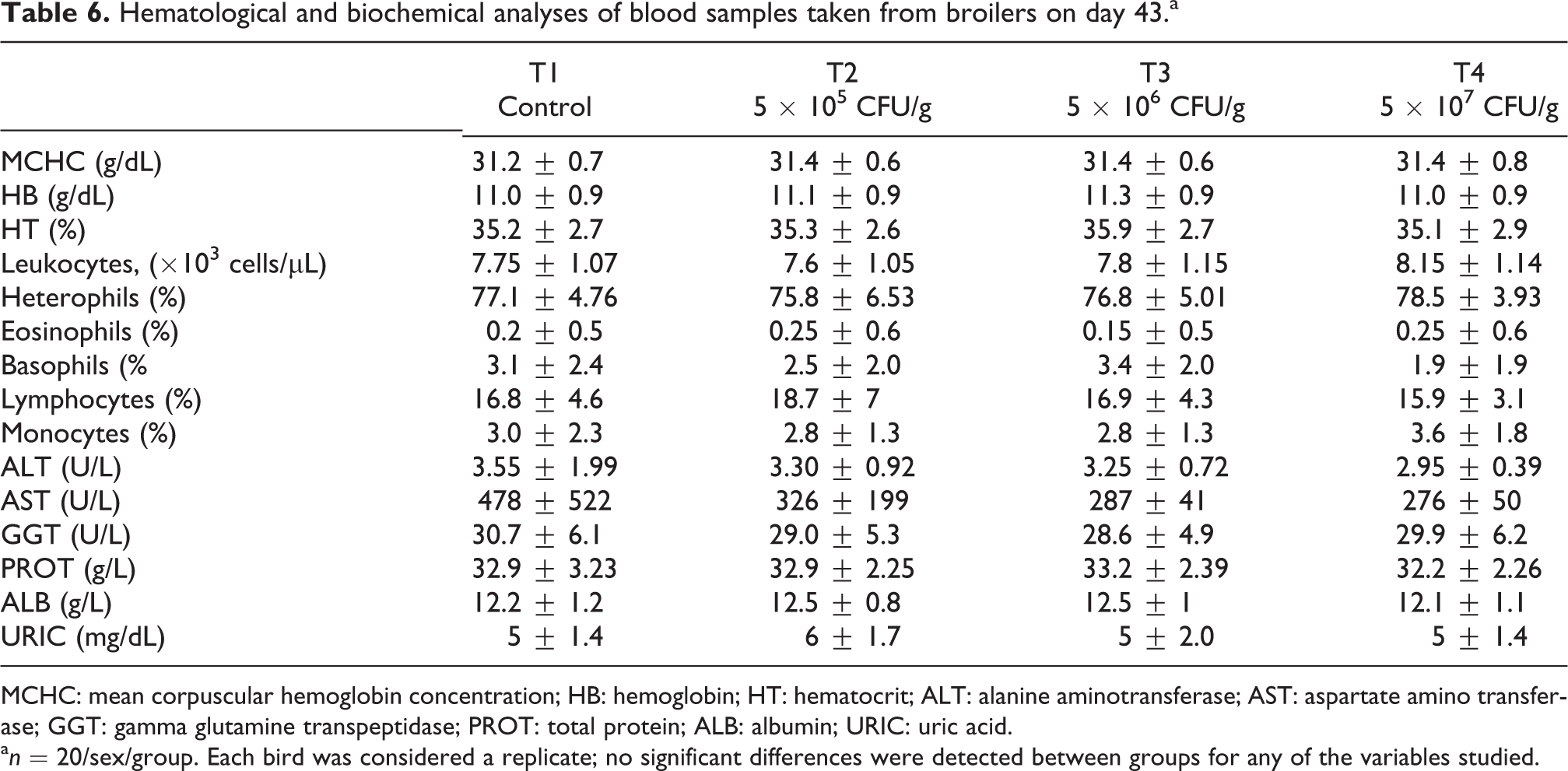
MCHC: mean corpuscular hemoglobin concentration; HB: hemoglobin; HT: hematocrit; ALT: alanine aminotransferase; AST: aspartate amino transferase; GGT: gamma glutamine transpeptidase; PROT: total protein; ALB: albumin; URIC: uric acid.
a n = 20/sex/group. Each bird was considered a replicate; no significant differences were detected between groups for any of the variables studied.
Piglet tolerance study
Feed analysis confirmed proper nutrient content of all piglet study diets, and the nutrient content was similar for all groups. The detected counts of CBM 588 in feeds were in line with target values for all treatments (see Supplementary Table 5). CBM 588 counts in fecal samples from control pens at both 21 and 42 days were below the limit of detection (<4.0 log10 CFU/g). Treated groups displayed dose-dependent increases in CBM 588 counts at both 21 days (4.4–4.5 and 5.4 log10 CFU/g for T2 and T3, respectively) and 42 days (4.3–4.4 and 5.0–5.1 log10 CFU/g, respectively). These data confirmed the exposure of piglets to CBM 588 as intended by the study design. Fecal consistency was good (average score at or near 1.00) throughout the study, and no significant differences were observed between groups (data are not shown).
The addition of CBM 588 at both a low (5 × 105 CFU/g) and high (5 × 107 CFU/g) dose did not affect zootechnical performance in weaned piglets (Table 7). ADG and FCR were considered good by current Dutch standards.
Zootechnical performance of piglets.a
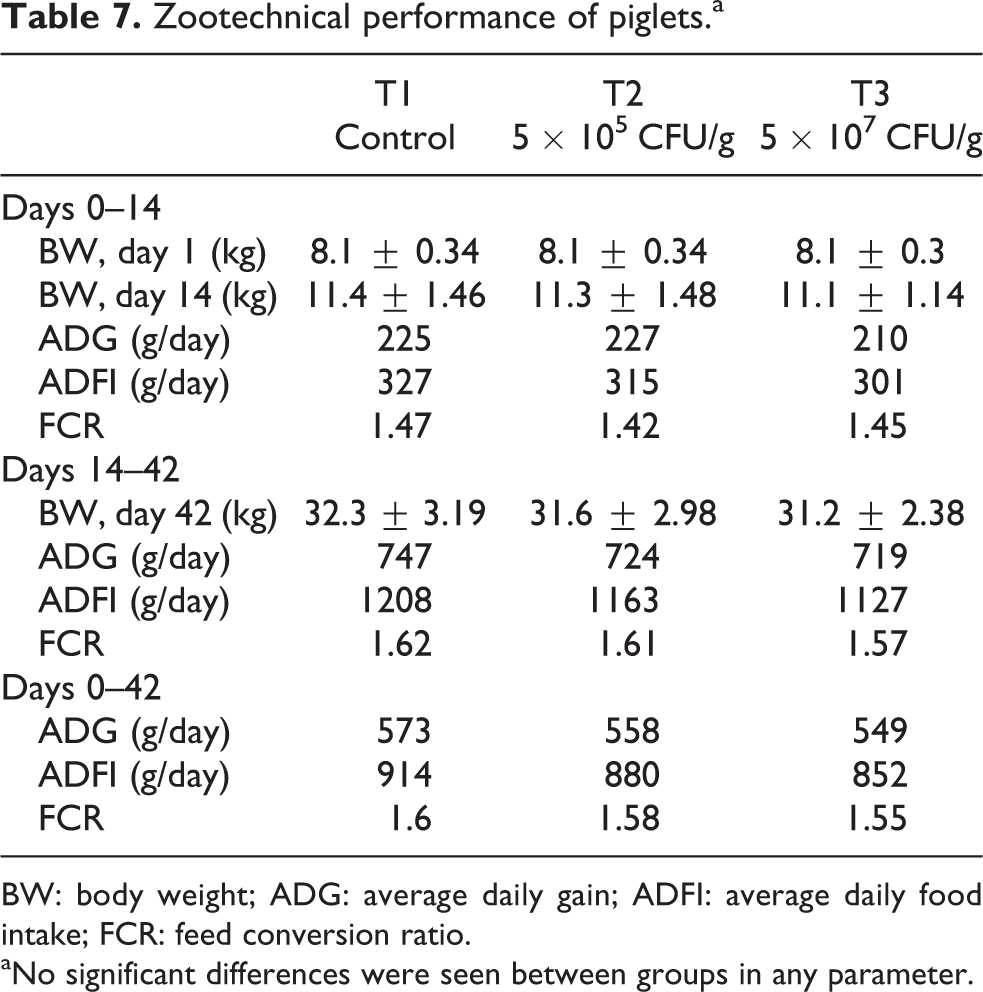
BW: body weight; ADG: average daily gain; ADFI: average daily food intake; FCR: feed conversion ratio.
aNo significant differences were seen between groups in any parameter.
One piglet in the high-dose group was treated twice with antibiotics due to poor zootechnical performance, most likely associated with porcine ileitis (Lawsonia intracellularis) and unrelated to treatment with CBM 588. No mortalities occurred in any group during the study.
Turkey tolerance study
Feed analysis confirmed proper nutrient content of all turkey experimental diets, and the nutrient content was similar for all treatments. CBM 588 was below the limit of detection in the control feed and in line with target values for all treatment-containing feeds (see Supplementary Table 6). Fecal samples displayed dose-dependent increases in CBM 588 counts at 28 days (2.43, 4.15, 5.99, and 4.79 log10 CFU/g for T1, T2, T3, and T4, respectively), 56 days (2.41, 4.26, 4.04, and 4.86 log10 CFU/g), and 84 days (3.67, 5.34, 6.04, and 6.92 log10 CFU/g). These results confirmed exposure of the birds to CBM 588 as intended by the study design.
Zootechnical performance data for turkeys receiving CBM 588 are presented in Table 8. Although there were no statistically significant differences between treatments in BW gain during the first study period (days 0–28), T4 birds reached the highest BW in comparison with the other treatments. The same effect was observed during the second and third experimental periods (days 29–42 and 43–56). During the fourth study period (days 57–84), T4 birds had significantly higher weight gain compared to T1. At day 84, T4 birds weighed significantly more than T1 birds. Additionally, the weight of T3 birds trended higher than the T1 control group (p = 0.063).
Zootechnical performance of turkeys.a
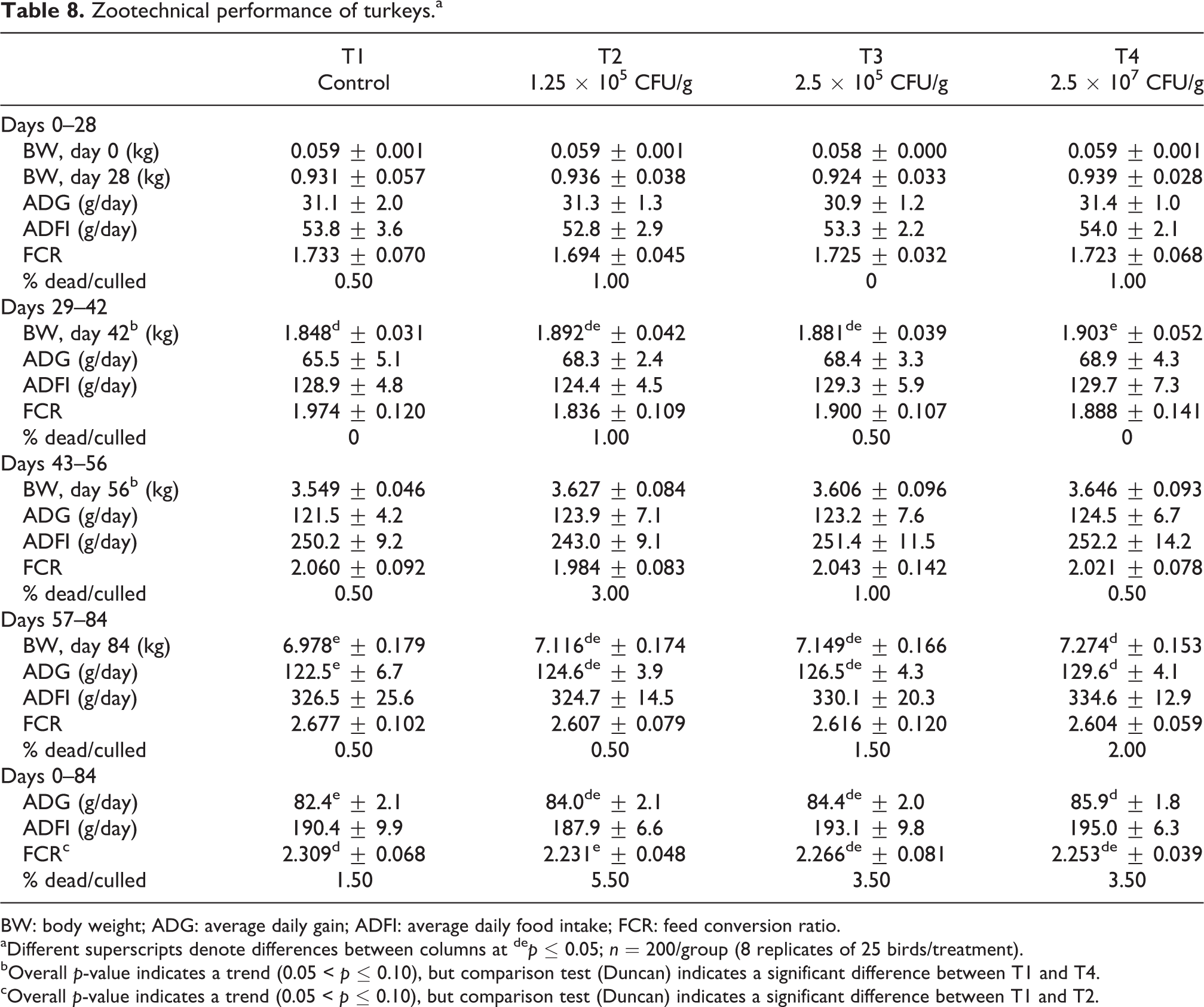
BW: body weight; ADG: average daily gain; ADFI: average daily food intake; FCR: feed conversion ratio.
aDifferent superscripts denote differences between columns at de p ≤ 0.05; n = 200/group (8 replicates of 25 birds/treatment).
bOverall p-value indicates a trend (0.05 < p ≤ 0.10), but comparison test (Duncan) indicates a significant difference between T1 and T4.
cOverall p-value indicates a trend (0.05 < p ≤ 0.10), but comparison test (Duncan) indicates a significant difference between T1 and T2.
Over the entire study period (days 0–84), birds from all treated groups (T2–T4) had improved weight gain compared to the T1 control group, but differences were only significant for T4. T3 birds showed a trend toward improved weight gain compared to T1 control birds (p = 0.062).
No significant differences were observed in feed intake between treatments. Overall, birds in groups T3 and T4 consumed more feed than birds fed the T1 control diet.
During the first (days 0–28), second (days 29–42), and third (days 43–56) study periods, no significant differences in FCR were observed between treatments. Over the entire study period (days 0–84), T2 birds had significantly better FCR compared to the T1 control group. Additionally, FCR for T4 birds showed a trend toward improvement (p = 0.090) versus T1 control birds.
Global mortality for this study was relatively low at 1.5, 5.5, 3.5, and 3.5% for groups T1–T4, respectively. Mortalities and culls were due to cachexia, weak legs, enlarged crop, and sudden death syndrome. No significant differences between groups were observed.
Results from hematological and biochemical analyses of blood samples taken on day 42 are presented in Table 9. No significant differences were detected between groups for any of the variables studied. Moreover, values were numerically similar between groups.
Hematological and biochemical analyses of blood samples taken from turkeys on day 42.a

HB: hemoglobin; HT: hematocrit; AST: aspartate amino transferase; GGT: gamma glutamine transpeptidase; PROT: total protein; TC: total cholesterol.
aDifferent superscripts denote differences between columns at de p ≤ 0.05; n = 10 birds/group, except for T3 hematology (n = 9) due to a blood clot in one sample.
bOverall p-value indicates a trend (0.05 < p ≤ 0.10), but comparison test (Duncan) indicates a significant difference between T2 and T4.
cOverall p-value indicates a trend (0.05 < p ≤ 0.10), but comparison test (Duncan) indicates a significant difference between T2 versus T3 and T4.
Discussion
CBM 588 has been approved in the EU as a feed additive for turkeys and chickens for fattening, chickens reared for laying, weaned piglets, minor avian and weaned porcine species, and as a novel food ingredient for humans. The studies presented in this article investigated the safety and tolerance of CBM 588 in broilers and weaned piglets at doses up to 100× the maximum EU recommended dose of 5 × 105 CFU/g feed, and in turkeys at doses up to 200× the maximum EU recommended dose of 1.25 × 105 CFU/g feed.
Over a trial period of 42 days, broilers receiving control diet or diet containing 5 × 105, 5 × 106, or 5 × 107 CFU CBM 588/g feed showed no significant differences in zootechnical performance parameters, including final BW, ADG, and ADFI. FCR was significantly improved for T2 compared to T1 control birds. No differences were observed in natural mortality, which was less than 2%, and no deaths were attributed to the presence of CBM 588 in the feed. Hematological and biochemical data were not affected by exposure to CBM 588. Therefore, CBM 588 was well-tolerated by broiler chickens at up to 100× the maximum recommended dose, and the inclusion of CBM 588 in the diet did not present any safety concerns. Moreover, some performance variables (ADG and FCR from days 1 to 21) were significantly improved at the highest dose (5 × 107 CFU/g).
In the piglet tolerance study, the inclusion of CBM 588 in the diet at 5 × 105 or 5 × 107 CFU/g feed had no effects on zootechnical performance. No piglets died during the study, and only one animal was treated with antibiotics due to poor condition, most likely associated with porcine ileitis (Lawsonia intracellularis) and unrelated to CBM588. Hence, CBM 588 was shown to be safe and well-tolerated in piglets at up to 100× the EU recommended dose.
The inclusion of CBM 588 in the diet of turkeys at 1.25 × 105, 2.5 × 105, or 2.5 × 107 CFU/g feed did not negatively affect zootechnical performance, blood hematology, or biochemical parameters. Some performance variables were improved by CBM 588, which produced dose-dependent increases in final BW (day 84) and overall ADG (days 0–84). These results confirm that CBM 588 is safe and well-tolerated in turkeys at up to 200-fold of EU recommended dose (2.5 × 107 CFU/g).
SCFAs, including butyrate, have been estimated to contribute 20–30% of the caloric requirements for omnivorous or herbivorous, nonruminant animals. 5 Across commercial animal species, the supplementation of butyrate has been shown to positively impact the development of the gastrointestinal tract, often resulting in notable increases in growth performance. 15 As a producer of butyrate, CBM 588 may provide additional nutrition beyond that contained in standard feed formulations. This possibility is supported by the current studies, in which CBM 588 administration had positive effects on certain zootechnical performance parameters, particularly for broilers and turkeys.
Overall, CBM 588 administered in feed at doses up to 5 × 107 CFU/g (chickens and piglets) or 2.5 × 107 CFU/g (turkeys) was shown to be safe and well-tolerated in all animals. As a source of butyrate, CBM 588 enhances the nutritional value of commercial feeds without safety concerns.
Supplemental Material
Supplementary_Material - In vivo safety evaluation of the Clostridium butyricum MIYAIRI 588 strain in broilers, piglets, and turkeys
Supplementary_Material for In vivo safety evaluation of the Clostridium butyricum MIYAIRI 588 strain in broilers, piglets, and turkeys by Kentaro Oka, Elinor McCartney, Tadashi Ariyoshi, Hayami Kudo, Borja Vilá, Lineke de Jong, Krzysztof Kozłowski, Jan Jankowski, Sarah Morgan, Claire Kruger, and Motomichi Takahashi in Toxicology Research and Application
Footnotes
Acknowledgements
The authors thank Mr A. Fukuda for technical assistance in measuring organic acid production, and Miyarisan Pharmaceutical Co. Ltd, Tokyo, Japan, for sponsoring all study experiments.
Author contributions
KO, TA, HK, BV, LJ, KK, and JJ performed the experiments; KO, EM, TA, HK, BV, LJ, KK, JJ, SM, CK, and MT conceived and designed the experiments and wrote the article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sarah Morgan and Claire Kruger are employees of ChromaDex Spherix Consulting, a business unit of ChromaDex, Inc. ChromaDex is a public company that was mandated and financed by Miyarisan Pharmaceutical Co. Ltd., to write this manuscript. Kentaro Oka, Tadashi Ariyoshi, Hayami Kudo, and Motomichi Takahashi are employees of Miyarisan Pharmaceutical Co. Ltd., the company that mandated and funded the studies presented in this manuscript and preparation of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
