Abstract
Bacopa monnieri is an important medicinal plant widely used in various food systems and gaining interest these days for its health benefits such as boosting brain function and improving quality of life. The objective of the present study was to examine the safety of Bacognize®, a standardized botanical extract obtained from the whole herb Bacopa monnieri (L.) Wettst., in subchronic toxicity and genotoxicity studies conducted in compliance with Good Laboratory Practice (GLP) and test guidelines established by the Organization for Economic Cooperation and Development (OECD). In the subchronic toxicity study, treatment with Bacognize® did not result in any toxicologically significant treatment-related changes in clinical observations, and in the clinical pathology as studied by hematology, serum chemistry, urinalysis, and terminal necropsy. Treatment-related adverse effects were not observed in ophthalmic examinations, body weights, body weight gains, feed consumption, and organ weights. The results of genotoxicity studies as assessed by gene mutations in Salmonella typhimurium, in vitro chromosome aberrations and in vivo micronucleus test did not reveal any genotoxicity of Bacognize ® . The no-observed-adverse-effect level (NOAEL) for Bacognize® was established based on the subchronic study and was determined as at least 1000 mg/kg bw/day. These results indicate that Bacognize® does not cause significant adverse effects and suggest its tolerability up to 1000 mg/kg (highest dose tested) for the daily administration of 90 days in rats.
Introduction
Bacopa monnieri (B. monnieri), is a creeping, prostrate, somewhat succulent perennial herbaceous plant comprising of stems, runner stems and leaves. In India, Ayurvedic practitioners have used B. monnieri as an herb for almost 3000 years in traditional preparations for boosting brain function and improving the quality of life. 1 In various ancient Ayurvedic treatises, the use of B. monnieri has been mentioned for the management of a range of mind disorders such as anxiety, poor cognition and lack of concentration. Bacopa monnieri is also known for its efficacies in maintaining the acuity of intellect and memory. 2
In addition to its traditional use in alternative systems for its health benefits, B. monnieri was reported and identified as a wild food plant. 3 In another report, it was mentioned as a wild edible plant, as it was used as a cooked vegetable. 4 In a review article, Hussain et al., 5 described the use of herbs and their active ingredients, including B. monnieri, for incorporation into foods. The use of B. monnieri has been reported as an ingredient in food supplements, teas and cosmetic products. 6 There are numerous food products such as drinks, biscuits, syrups, jellies, and breakfast cereals available in the Indian market containing B. monnieri as a functional food ingredient. 7
Many phytochemical studies have shown the chemical constituents of B. monnieri to be bacosides, and bacogenins, steroids triterpene, alkaloids brahmine, herpestine, nicotine, saponin, monierin, hersaponin, and bacosine. 8 The major chemical entities shown to be responsible for the memory-enabling action of B. monnieri are dammarane types of triterpenoid saponins with jujubogenin or pseudojujubogenin moieties as the aglycone units known as bacoside A and B. 9 Bacoside A is a mixture of four triglycosidic saponins, namely bacoside A3, bacopaside II, bacopasaponin C and the jujubogenin isomer of bacosaponin C (bacopaside X). 10
In recent years, multiple human clinical studies have been conducted to evaluate the effects of B. monnieri. A randomized, double-blind, placebo-controlled study conducted to assess the effect of a standardized dry extract of a whole plant of B. monnieri on cognition function, safety and tolerability in healthy elderly participants revealed the potential for safely enhancing cognitive function. 11 In another randomized, open-label study in healthy adult volunteers, BacoMind™ (standardized to bioactive compounds of B. monnieri) was evaluated for short-term safety and tolerability. Participants orally given one single capsule of BacoMind™ daily for 30 days, (i.e., 300 mg for first 15 days and 450 mg for next 15 days) showed no adverse events at the dose administered for the given duration of the trial period. 12 Also, a subchronic oral toxicity study for 90 days in rats with BacoMind™ at the dose levels of 85, 210 and 500 mg/kg did not reveal any evidence of toxicity. 13
As alluded to above, although human clinical studies and toxicology studies have been conducted on extracts of B. monnieri, to assess their efficacy and safety, a comprehensive safety profile on bacopa glycosides (bacosides) at 1000 mg/kg bw/day has not yet been established. Hence, a standardized extract Bacognize® from B. monnieri was used to establish the safety through a series of toxicological studies. Bacognize® is a standardized hydroalcoholic extract of the whole herb Bacopa monnieri (L.) Wettst. It is standardized by the United States Pharmacopeial Convention method (using High-performance liquid chromatography) to 12% total bacopa glycosides (Bacopaside I, Bacoside A3, Bacopaside II, Jujubogenin isomer of Bacopasaponin C, Bacopasaponin C) as depicted in Figure 1.
14
The objective of the present study was to investigate adverse effects, if any, of a standardized extract Bacognize® from B. monnieri in genotoxicity studies as assessed by the Ames test, in vitro chromosomal aberration assay, in vivo micronucleus test in rats, and a repeat-dose subchronic toxicity study in rats. The effects of Bacognize® was investigated in a dose-response manner. Chromatogram of 100 mg/mL dose formulation of Bacognize®. Bacognize® standardized to NLT 12% total bacopa glycosides (Bacopaside I, Bacoside A3, Bacopaside II, Jujubogenin isomer of Bacopasaponin C, Bacopasaponin C) using HPLC by the USP method.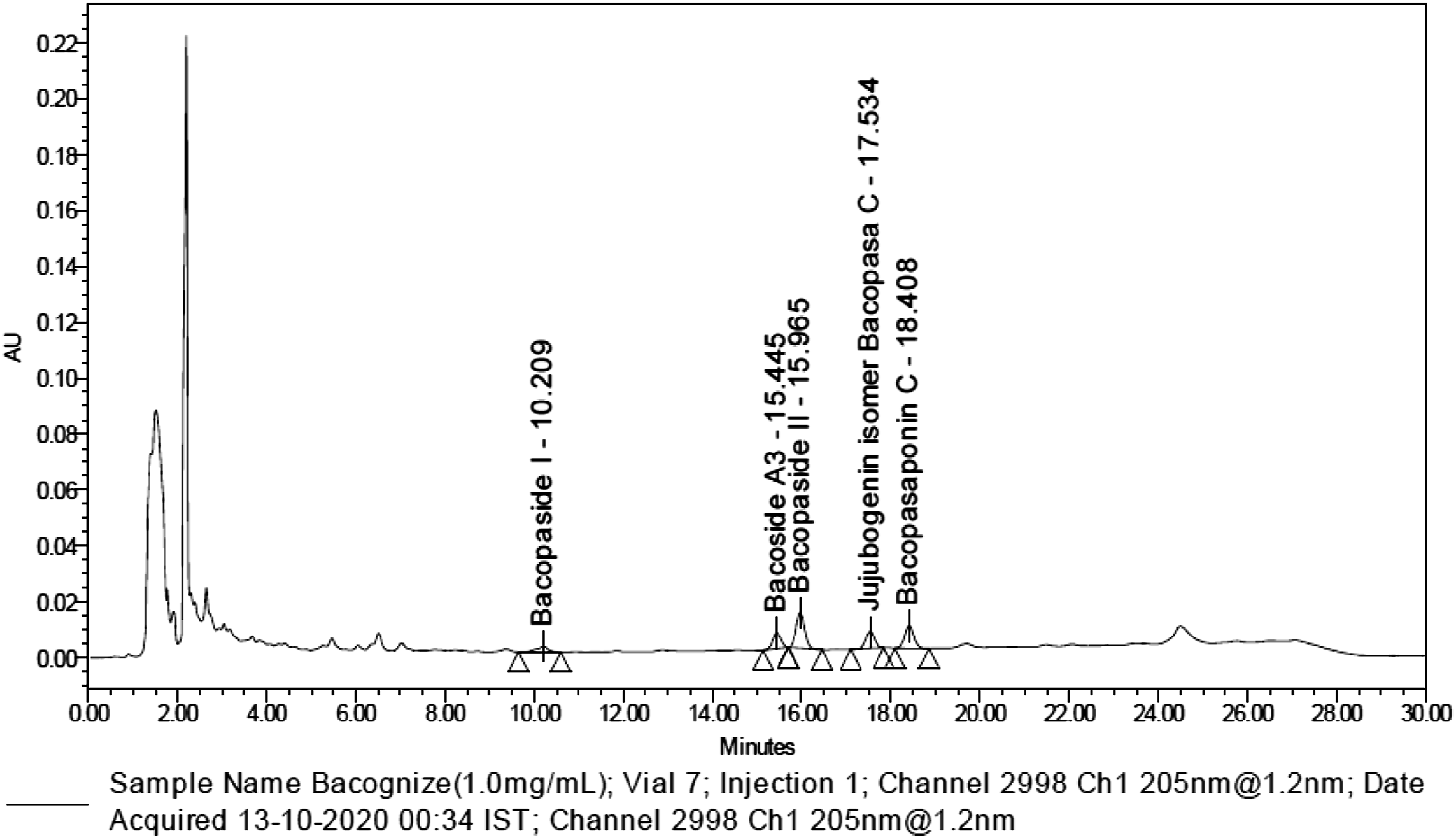
Materials and methods
Test substance
Specifications of Bacognize®.

Genotoxicity studies
Ames assay
To examine the potential of Bacognize® to induce gene mutations, an Ames test was performed at RCC, Hyderabad, India, in compliance with The Organisation for Economic Co-operation and Development (OECD) 471 and adhering to the principles of Good Laboratory Practice (GLP). Salmonella typhimurium strains TA98, TA100, TA102, TA1535, TA1537 were used, and the plate incorporation method in the presence or absence of a S9 metabolic activation system was applied in Trial 1. Based on the solubility and precipitation test results, eight different concentrations namely, 0.002, 0.005, 0.016, 0.050, 0.158, 0.501, 1.582 and 5 mg/plate, were selected for pre-experiment. Based on the pre-experiment results, Bacognize® was tested with the following concentrations 0.050, 0.158, 0.501, and 5 mg/plate for the main study, both in the presence of metabolic activation (+S9) and in the absence of metabolic activation (-S9) by the pre-incubation method (Trial 2). Chemicals used as positive controls for assays without or with metabolic activation included, sodium azide, 4-nitro-o-phenylenediamine, methyl methane sulfonate, and 2-aminoanthracene and were purchased from Sigma‐Aldrich. S9 fraction (Aroclor 1254-induced; Analab, USA) with co-factor was used to mimic the metabolic activation system. Each batch of S9 mix was tested with 2-Aminoanthracene as well as benzo (a) pyrene for its efficiency. For each strain and dose level, including the controls, three plates (triplicate) were used. The following materials were mixed in a test tube and poured onto the selective agar plates: 100 μL test solution at each dose level, negative control, vehicle control and reference mutagen solution (positive control), 500 μL S9 mix (for the test with metabolic activation) or S9 mix substitution buffer (for the test without metabolic activation), 100 μL bacterial suspension, and 2000 μL overlay agar.
In the pre-incubation assay, 100 μL test solution, 500 μL S9 mix/S9 mix substitution buffer and 100 μL bacterial suspensions were mixed in a test tube and incubated at 37 ± 2°C for 60 min. After pre-incubation, 2.0 mL overlay agar (47 ± 2°C) was added to each tube. The mixture was poured on minimal agar plates. After solidification, the plates were incubated in the inverted position for 48 h at ± 2°C. The colonies were counted manually. The mean values of the plates for each concentration, together with the standard deviation, were compared to the spontaneous reversion rates. Microsoft Office Excel-based calculations were used for descriptive statistical analysis. Essentially, means and standard deviations were analyzed for all quantitative data collected.
The bacterial reverse mutation assay was considered acceptable if it met the following criteria: (a) regular background growth in the negative control and solvent (vehicle) control (b) the positive control substances produced a significant increase in mutant colony frequencies (c) the spontaneous reversion rates in the negative control were in the range of in-house historical data. A test item was considered a mutagen if a biologically relevant increase in the number of revertant exceeding the threshold was of twice (strains TA 98, TA 100 and TA102) or thrice (strains TA 1535 and TA 1537) the colony count of the corresponding solvent control was observed. A dose-dependent increase was considered biologically relevant if the threshold exceeded at more than one concentration. An increase exceeding the threshold at only one concentration is judged as biologically relevant if reproduced in an independent second experiment. A dose-dependent increase in the number of revertant colonies below the threshold was regarded as an indication of a mutagenic potential if reproduced in an independent second experiment. However, whenever the colony counts remained within the historical range of negative control and vehicle control such an increase was not considered biologically relevant.
In vitro chromosomal aberration assay
The potential of Bacognize® to induce structural and numerical chromosome aberrations was evaluated in human lymphocytes. At Accuprec, Gujrat, India, the GLP and OECD guidelines for testing of chemicals #473 was followed for the In vitro Mammalian Chromosome Aberration Test (1997), in the presence and absence of a metabolic activation system. At least three different doses based on the solubility of the test item were tested. The induction of cytogenetic damage in human lymphocytes was assessed with, and without, metabolic activation. Before the selection of doses, the mitotic index of the cells with Bacognize® at different doses were chosen for the study. The maximum concentration used was 5 mg/mL, unless limited by solubility, precipitation, and effect on the media pH of the test item. The highest treatment concentration chosen for the experiment was such that it would inhibit the mitotic index by more than 50%.
The treatment of cultures with the test substance was conducted in a minimum of two independent phases. For Phase – I 1% and for Phase – II 2% of the S9 mixture was used with the metabolic activation system. Proliferating cells were used and the cell culture was prepared in RPMI-1640 medium 48–50 h prior to exposure. Negative and vehicle control (solvent), and positive control were maintained, both in the presence and absence of a metabolic activation system. The medium of the proliferating blood culture was removed by centrifugation at 1000–1500 r/min for 10 min. Cells were suspended in the plain medium mixed with S9 mix and in the complete media mixed with phosphate buffer for treatment in the presence and absence of a metabolic activation system. For Phase II, cultures were harvested at the end of incubation of 22–25 h after treatment.
Harvesting and processing cells
Before 3–5 h of harvesting, 240 μL of colcemid (10 μg/mL) was added to each of the culture tube/flasks and incubated at 37 ± 2°C. The cultures were harvested by centrifugation at 1000–1500 r/min for 10 min, 22–25 h after the beginning of treatment. The supernatant was discarded and the cells were re-suspended in approximately 7–8 mL of freshly prepared, warm (37 ± 2°C) hypotonic solution of potassium chloride (0.075 M KCl). The cell suspension was allowed to stand for 30 min, following which it was centrifuged and the supernatant was removed. Freshly prepared chilled (5–6 mL) Carnoy’s fixative (3:1 methanol: acetic acid solution) was added and allowed to stand for 5 min. This process was repeated twice and after final centrifugation, the supernatant was removed completely, and the cell pellet was re-suspended in 0.3–5 mL of Carnoy’s fixative. Slides were prepared by dropping the cell suspension onto a clean microscope slide and stained with 5% fresh Giemsa stain. All slides, including positive and negative controls, were independently coded before microscopic analysis.
In vivo micronucleus test
The mammalian erythrocyte micronucleus assay was performed at Cadila Pharmaceuticals, Gujrat, India, in accordance with the OECD guideline for the testing of chemicals #474 (2016). The animal usage and experiment procedures for the present study were approved by Institutional Animal Ethics Committee. This study was performed as per the recommendations of the CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals) guidelines, following all the ethical practices as laid down in the CPCSEA guidelines for animal care. Swiss albino mice (about 6–10 weeks old) were quarantined and acclimated for a minimum of 5 days before dosing for this dose-response study. Bacognize® was administered by oral gavage to animals that were randomly assigned to a treatment group (5/sex/group). Bacognize® was suspended in 0.5% carboxy methyl cellulose (CMC) and administered to mice at dose levels of 0, 500, 1000 and 2000 mg/kg bw. The preparation of different doses varied in concentration to allow a constant dose volume of 10 mL/kg for each group. Animals in the control group were treated with 0.5% CMC and served as the vehicle control. Colchicine (2 mg/kg bw) was chosen as the positive control, prepared using water and administered intraperitoneally. Between 18–24 h following the last treatment, animals from all dose groups were sacrificed using carbon dioxide gas. A femur from the sacrificed animals was excised and the epicondyle tips were removed. The bone marrow content was expelled in centrifuge tubes by flushing the bone marrow with 3 mL of fetal bovine serum. After collection, the aspirated bone marrow was mixed thoroughly using a syringe and needle to dissociate the cells and also avoid the formation of a cell clump. The centrifuge tubes with bone marrow content were centrifuged at ∼1500 r/min for 10 minutes. The supernatant was discarded, leaving about 0.2–0.3 mL of the medium with a cell pellet. The cell pellet was dissociated thoroughly by repeated pipetting. A drop of the thoroughly mixed suspension was placed on the clean pre-identified slide; a thin smear was prepared and allowed to air dry.
The slides were identified with a study number, animal number and slide number. Three slides were prepared for each animal to meet the required number of cells. The smear was fixed with absolute methanol and air-dried. Slides were stained with Giemsa, allowed to air dry and mounted with dibutyl phthalate in xylene (DPX). In order to prevent bias in the scoring procedure, the slides were blind-coded and decoded after scoring.
The required number of slides of each animal was used for screening of percent immature erythrocytes and micronucleated immature erythrocytes, whereas the remaining slides were kept in reserve for scoring if required. The slides were examined for the proportion of immature among total (immature and mature) erythrocytes in each animal by counting a minimum of 500 erythrocytes. At least 4000 immature erythrocytes per animal were scored for incidence of micronucleated immature erythrocytes.
The data on the percent micronucleated immature erythrocytes and percent immature erythrocytes for both sexes were statistically analyzed. For the experimental groups, data was analyzed by ANOVA and Bartlett’s test, along with Dunnett’s test. Where Bartlett’s test was non-significant (homogeneous data), the statistical decision was considered according to ANOVA. For non-significant ANOVA, results of ANOVA were considered and for significant ANOVA, results of Dunnett’s test were considered for interpretation. Where Bartlett’s test was significant (data not homogeneous), non-parametric ANOVA (Kruskal–Wallis test) along with Dunn’s test was performed. For non-significant Kruskal-Wallis, results of the Kruskal–Wallis test were considered. For the significant Kruskal–Wallis test, the results of Dunn’s test were considered for interpretation.
Sub-chronic study
Study design
The OECD Guidelines for Testing Chemicals #408 was followed for 90-Day Oral Toxicity Study in Rodents. The study was conducted at Cadila Pharmaceuticals, Gujrat, India, in compliance with the technical requirements of the protocol, and all applicable guidance and regulations that included Good Laboratory Practices for Non-Clinical Laboratory Studies (FDA, 21 CFR, Part 58 and OECD, ENV/MC/CHEM(98)17, revised 1997).
Animals
The animal usage and experiment procedures for the present study were approved by Institutional Animal Ethics Committee. This study was performed as per the recommendations of the CPCSEA (Committee for the Purpose of Control and Supervision of Experiments on Animals) guidelines, following all the ethical practices as laid down in the CPCSEA guidelines for animal care. The number of animals that were selected for use in this study was considered the minimum requirement to meet rational scientific endpoints and regulatory guidelines followed for this type of study. The Wistar rats were housed in autoclaved standard rat cages with stainless steel top grill. Autoclaved corn cob was used as bedding. The frequency of change for bedding material was at least once in 4 days. A maximum of three rats was housed per cage. Cages were rotated at least once a week. The animals were kept in a clean environment with 12-h light and 12-h dark cycles. The air was conditioned between 19.0 to 23.8°C and the relative humidity was maintained between 43.0 to 68.9%. The pelleted rat feed manufactured by Altromin, Germany was provided ad libitum throughout the study period, except during overnight fasting (∼17–19 h) before blood collection. Autoclaved drinking water was provided ad libitum in polypropylene bottles with a stainless steel sipper tube throughout the study to all the animals.
Treatment
A repeated dose oral (90 days) toxicity study of Bacognize® was evaluated on 50 male and 50 female Wistar rats. The animals selected for the study were 7–8 weeks old with a weight range within ±20% of the mean body weight of each sex at the time of randomization. The doses were selected based on the literature no-observed-adverse-effect level (NOAEL) of Bacopa extract, which was 500 mg/kg in 90 days oral toxicity study in rats. 13
The animals were divided into six different groups during randomization and were numbered as groups I to VI. There were 10 animals per sex in Group I to Group IV (treatment group) and five animals per sex in Group V and Group VI (reversal groups). Group I and Group V were dosed with vehicle 0.5% Carboxymethylcellulose (CMC) as vehicle control (0 mg/kg). Groups II, III, IV and VI were dosed with Bacognize® at different doses (Group II-500 mg/kg body weight as low dose; Group III- 750 mg/kg body weight as mid-dose; Groups IV and VI- 1000 mg/kg body weight as high-dose). The test article or vehicle was administered to the animals by oral gavage daily for 90 days.
Formulation analysis was performed on the first week (Day 3) and last week (Day 86). Animals of treatment groups were observed for clinical signs and mortality up to Day 90. The animals of reversal groups were further observed for 28 days (up to Day 118) without any treatment. Percent mean accuracy of dose formulation analysis (Day 3) for 50, 75 and 100 mg/mL samples were 106.4, 108.9 and 109.0 of their nominal value, respectively. The mean of % relative standard deviation of dose formulation analysis (Day 3) for 50, 75, and 100 mg/mL samples were 0.41, 0.11, and 0.40, respectively. Percent mean accuracy of dose formulation analysis (Day 86) for all layers of 50, 75 and 100 mg/mL samples were 100.1, 101.6 and 98.1 of their nominal value, respectively. % relative standard deviation of dose formulation analysis (Day 86) for 50, 75, and 100 mg/mL samples were 1.72, 0.58, and 0.48, respectively. Both accuracy and precision for concentration and homogeneity analysis of test article in dose formulation were found within acceptance criteria which suggests that all dose formulation preparation was accurate and homogenous.
Parameters evaluated
Clinical signs, body weights and feed consumption
Detailed clinical observations were made on all animals once before the first exposure and weekly thereafter. All animals were observed once daily (additionally shortly after dosing from Day 47 onwards) for abnormal clinical signs and behavioral changes. The appearance, change, and disappearance of these clinical signs were recorded for 90 days for the treated dose group and 118 days for the reversal dose group. Detailed clinical observations were made on all animals once before the first exposure and weekly thereafter. Animals were observed for changes in the skin, fur, eyes and mucous membranes, the occurrence of secretions and excretions. Animals were also observed for an autonomic activity like lacrimation, piloerection, pupil size and unusual respiratory pattern, changes in gait, posture, response to handling, presence of clonic or tonic movements, stereotypes like excessive grooming and repetitive circling or bizarre behavior like self-mutilation, walking backward, etc. Ophthalmologic examination of all animals was carried out 1 day before initiation of dosing and 1 day before necropsy in control and high-dose animals. Body weights along with feed consumption were measured at weekly intervals.
Clinical pathology
At the end of the observation period, all the male and female animals in treatment groups were subjected to terminal sacrifice after blood collection on Day 91 and for the reversal groups on Day 119. Hematological and biochemical parameters were studied. During the last week of observation, urine was collected and urine parameters were studied for all groups. Before necropsy, the oestrus cycle of female animals was determined by taking a vaginal smear. During necropsy, organs were collected and preserved in 10% neutral buffered formalin (NBF) for further histopathological examination. The absolute weights of organs were recorded.
After completion of the treatment period of 90 days, blood was collected from all overnight fasted animals under carbon dioxide anesthesia (Day 91 for groups I to IV and Day 119 for groups V and VI). Approximately 4 mL blood was collected by retro-orbital method and transferred in three different containers. One container each, coated with K2EDTA (for routine hematological investigation), sodium citrate (for coagulation parameters) and another plain container, which was allowed to coagulate and serum was separated after centrifugation for biochemical analysis.
Hematology parameters analyzed included: Red blood cell counts (RBC), White blood cell counts (WBC), Platelet counts (PLT), Hemoglobin (HB), Hematocrit (HCT), Mean Corpuscular Volume (MCV), Mean Corpuscular Hemoglobin (MCH), Mean Corpuscular Hemoglobin Concentration (MCHC), and WBC differential (neutrophils-NEUT; eosinophils-EOSIN; basophils-BASO; monocytes-MONO; lymphocytes-LYMPH). Clinical biochemistry parameters analyzed included: Albumin (ALB), Alkaline phosphatase (ALP), Total bilirubin (T-BIL), Alanine aminotransferase (ALT), Aspartate aminotransferase (AST), Gamma-glutamyl transferase (γ-GT), Total protein (TP), Creatinine (CRE), Blood Urea nitrogen (BUN), Cholesterol (CHO), Triglycerides, Sodium (Na), Potassium (K), Glucose (GLU), Calcium (Ca) and Phosphorus (P).
During the last week of observation, urine was collected using metabolic cages and urine parameters such as appearance, color, specific gravity, pH, total protein, glucose, ketone, urobilinogen and bilirubin were studied for all groups.
Necropsy, organ weight and histopathology
Organs from all terminally sacrificed animals belonging to all dose groups were preserved in 10% neutral buffered formalin except for eyes, testes, and epididymis, which were fixed in modified Davidson’s fixative for 24–72 h, then transferred to neutral buffered formalin (NBF). The absolute weights of organs were recorded immediately after collection and before preserving them in NBF. The histopathological examinations were performed for the animals belonging to vehicle control (group I), treated high-dose (group IV) and both reversal dose groups (group V and VI) and all gross lesions. High-dose group animals did not reveal any treatment-related microscopic findings; hence, microscopic examination was not extended to mid and low-dose groups.
At necropsy, the oestrus cycle of all female animals was determined by taking a vaginal smear.
Statistical analysis
Group means and standard deviation were calculated for body weight, body weight change, feed consumption, hematology, biochemical parameters, absolute and relative organ weights and urinalysis (numerical) for each sex. For treated dose groups (groups I to IV), data were analyzed by Bartlett’s test and ANOVA. Where Bartlett’s test was non-significant (homogeneous data), the statistical decision was considered according to ANOVA. For non-significant ANOVA, results of ANOVA were considered and for significant ANOVA, results of Dunnett’s test were considered for interpretation. Where Bartlett’s test was significant (data not homogeneous), non-parametric ANOVA (Kruskal–Wallis test) along with Dunn’s test was performed. For the non-significant Kruskal–Wallis test, results of the Kruskal–Wallis test were considered and for the significant Kruskal–Wallis test, results of Dunn’s test were considered for interpretation. For the reversal dose groups (groups V and VI), data were analyzed by Paired “t”test. Where p-value <0.05 was considered as statistically significant.
Results
Genotoxicity studies
Ames assay
Results from the plate incorporation and pre-incubation methods of the Ames assay.
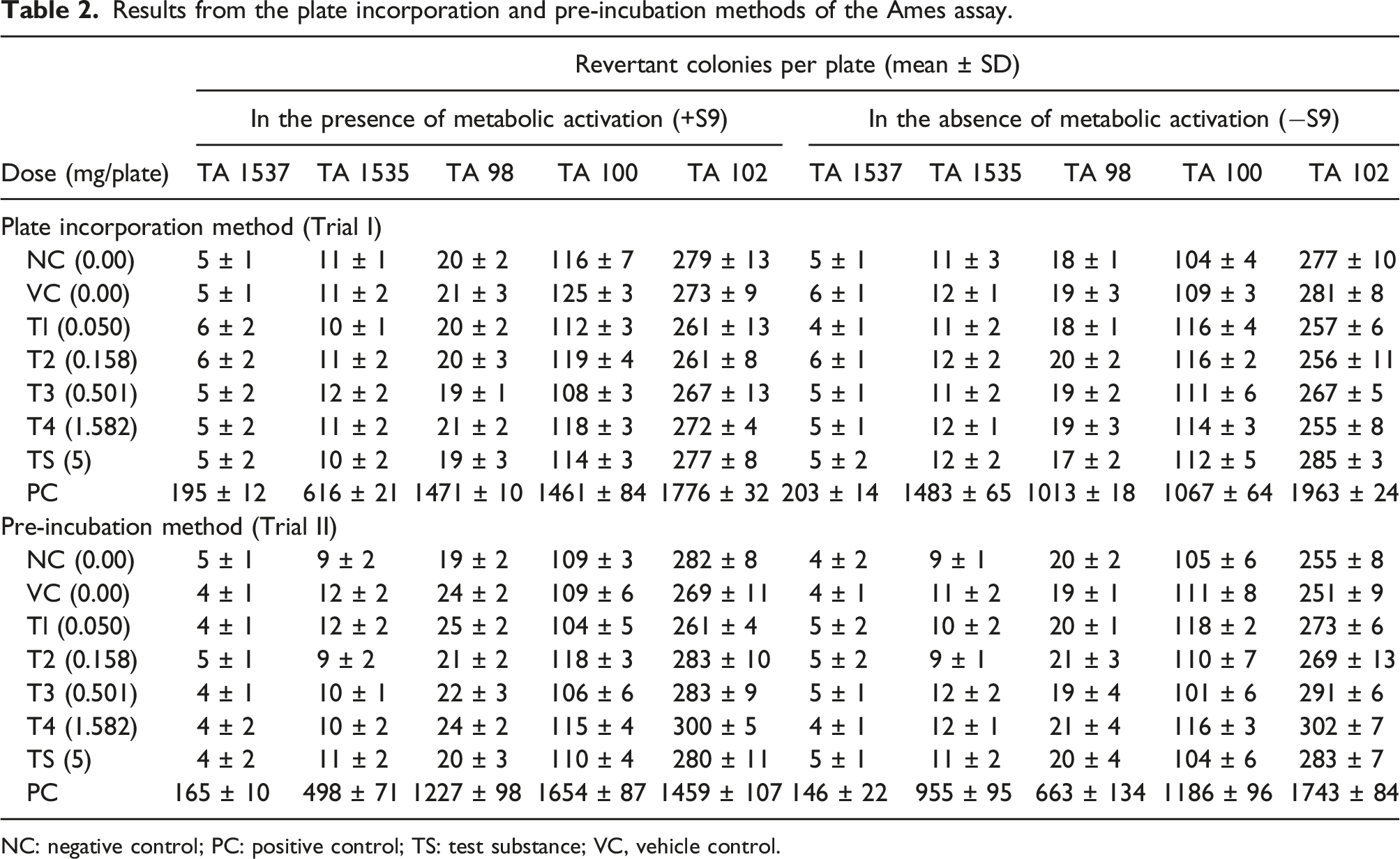
NC: negative control; PC: positive control; TS: test substance; VC, vehicle control.
In vitro chromosomal aberration assay
Results of in vitro chromosomal aberration assay.
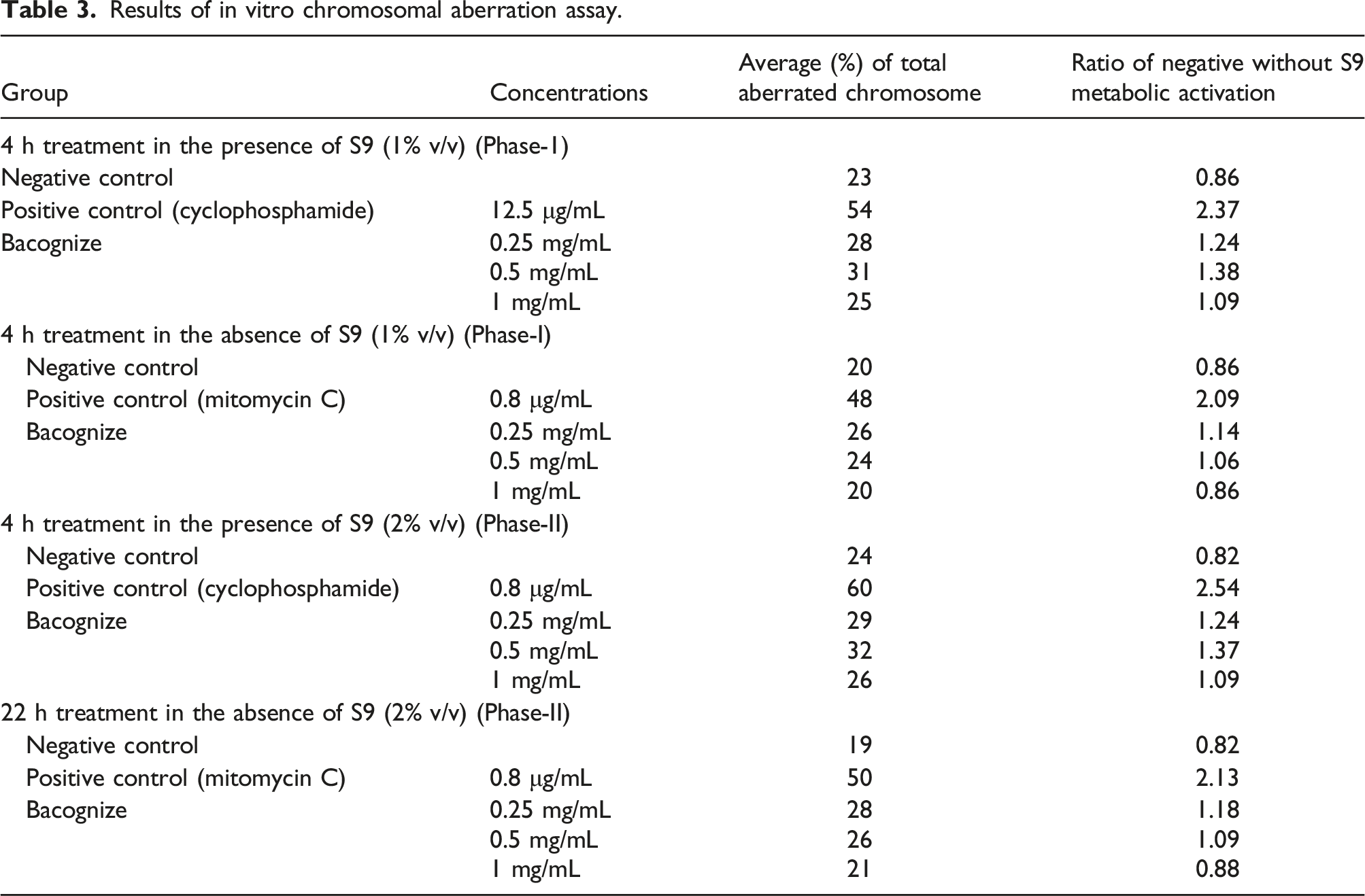
In phase I, at the concentration of 1, 0.5 and 0.25 mg/mL Bacognize® the corresponding percent chromosomal aberrations were 20% and 25%; 24% and 31%; and 26 and 28% without and with metabolic activation, respectively. In phase II, at the concentration of 1, 0.5 and 0.25 mg/mL Bacognize® the corresponding percent chromosomal aberration were 21% and 26%; 26% and 32%; and 28 and 29% without and with metabolic activation, respectively. Since the ratio of percent chromosomal aberration between the positive and negative controls were more than 2; while that of the Bacognize® treated sample and negative control were less than 2, it was concluded that Bacognize® does not have the potential to induce structural/numerical chromosomal aberrations in both the phases under the experimental conditions of the study.
In vivo mammalian erythrocyte micronucleus test
Bacognize® was suspended in 0.5% carboxy methyl cellulose (CMC) and administered to mice at dose levels of 0, 500, 1000 and 2000 mg/kg bw. Throughout this study, there were no statistically significant differences in mean body weights, abnormal clinical symptoms or deaths of animals observed in any group of animals. No statistically significant (p < 0.05) changes in % immature erythrocytes and % micronucleated immature erythrocytes in male and female animals belonging to the treatment groups were observed when compared to the vehicle control group. Within 19–22 h following the last treatment, animals from all dose groups were sacrificed using carbon dioxide gas. The PCE% (Polychromatic erythrocytes) of the vehicle control versus positive control, when compared in females and males, revealed a decrease in the PCE% in the positive control group that indicated inhibition of erythropoiesis by colchicine. Nevertheless, the PCE% in all the treatment groups were comparable to the vehicle control group indicating that Bacognize® did not inhibit erythropoiesis (data not shown).
Dose-response and micronucleus frequency on treatment of mice with Bacognize
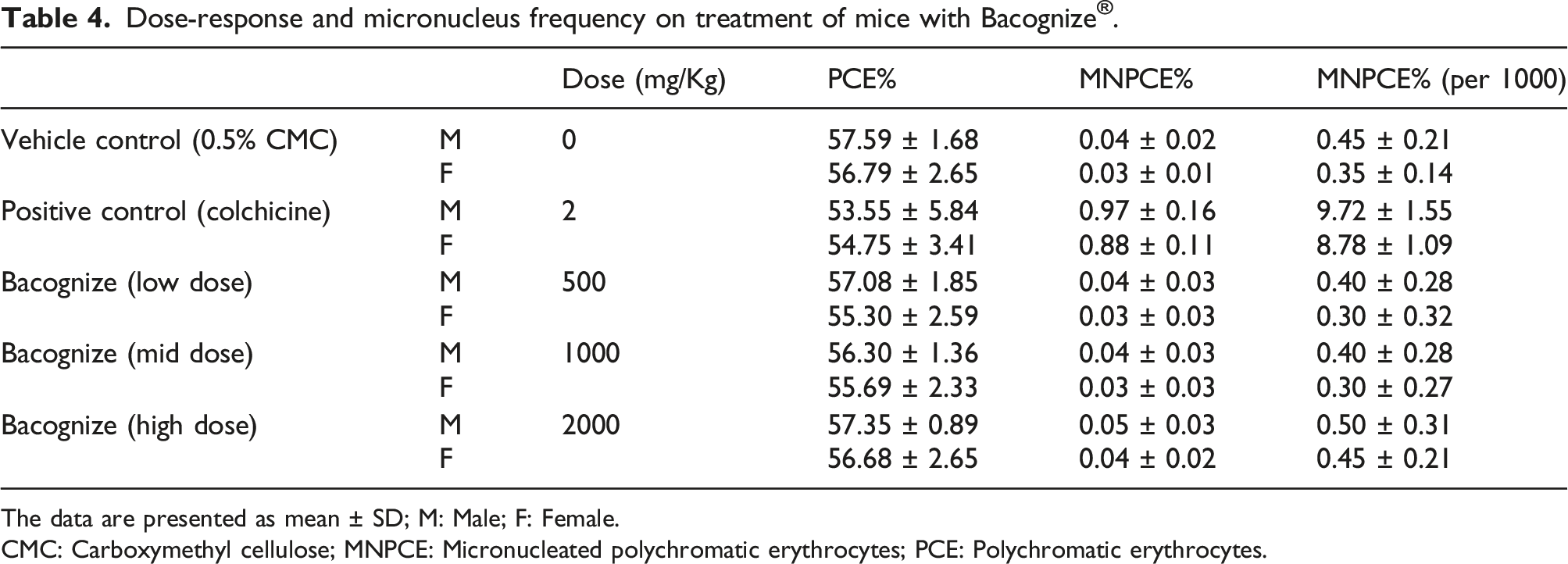
The data are presented as mean ± SD; M: Male; F: Female.
CMC: Carboxymethyl cellulose; MNPCE: Micronucleated polychromatic erythrocytes; PCE: Polychromatic erythrocytes.
Since Bacognize® did not cause any substantial increases in the incidence of micronucleated immature erythrocytes or any substantial decreases in percent immature erythrocytes, it is concluded that Bacognize® did not show evidence of causing chromosome damage or bone marrow cell toxicity when administered orally in this in vivo test conditions up to 2000 mg/kg body weight.
Subchronic study
Survival, clinical observations, body weights and feed consumption
During the study period, there were no mortality or treatment-related abnormal clinical signs in any of the groups. Although minimal to moderate salivation shortly after dosing (reversible by same day) was observed in male and female Bacognize® treated animals, salivation did not have an adverse impact on the overall health of animals and was not considered an adverse effect of the test article. The increased salivation in the study is attributed to the bitter taste of the test substance. In the literature, saliva is known to have modulating effects on sour, salt, and or umami/bitter taste. It has a diminishing effect on the sour taste as a result of the buffering by salivary bicarbonate. 15 There were no abnormal ophthalmic changes throughout the observation period.
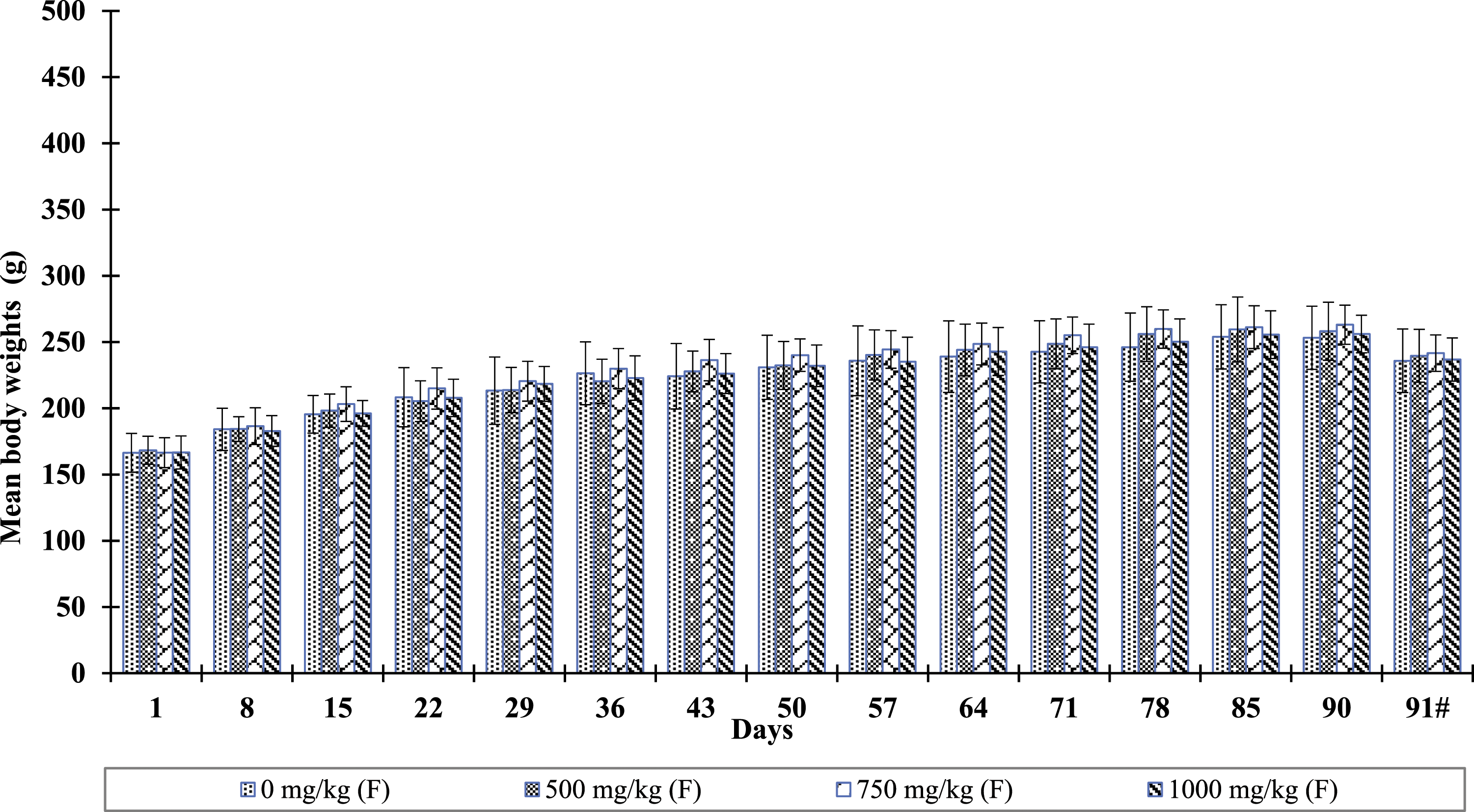
Male and female animals in the Bacognize® treated dose groups did not reveal statistically significant (p < 0.05) changes in body weights and body weight change when compared to the weights of male and female animals of the respective control group. The percent body weight change on Day 15 was significantly lower in female animals of group VI (1000 mg/kg) as compared to female animals of group V (vehicle control). This was a single incidence without any definitive pattern and it was considered biological variation/incidental instead of an adverse effect of the test article. The summary of body weight and body weight change data are depicted in Figures 2 and 3. Also, male and female animals in the Bacognize® treated dose groups did not reveal statistically significant (p < 0.05) changes in feed consumption when compared to male and female animals of the respective control group (data not shown). Bacognize® treatment effect on mean body weights (mean ± SD) in male rats. Note: Fasted body weight. Bacognize® treatment effect on mean body weights (mean ± SD) in female rats. Note: Fasted body weight.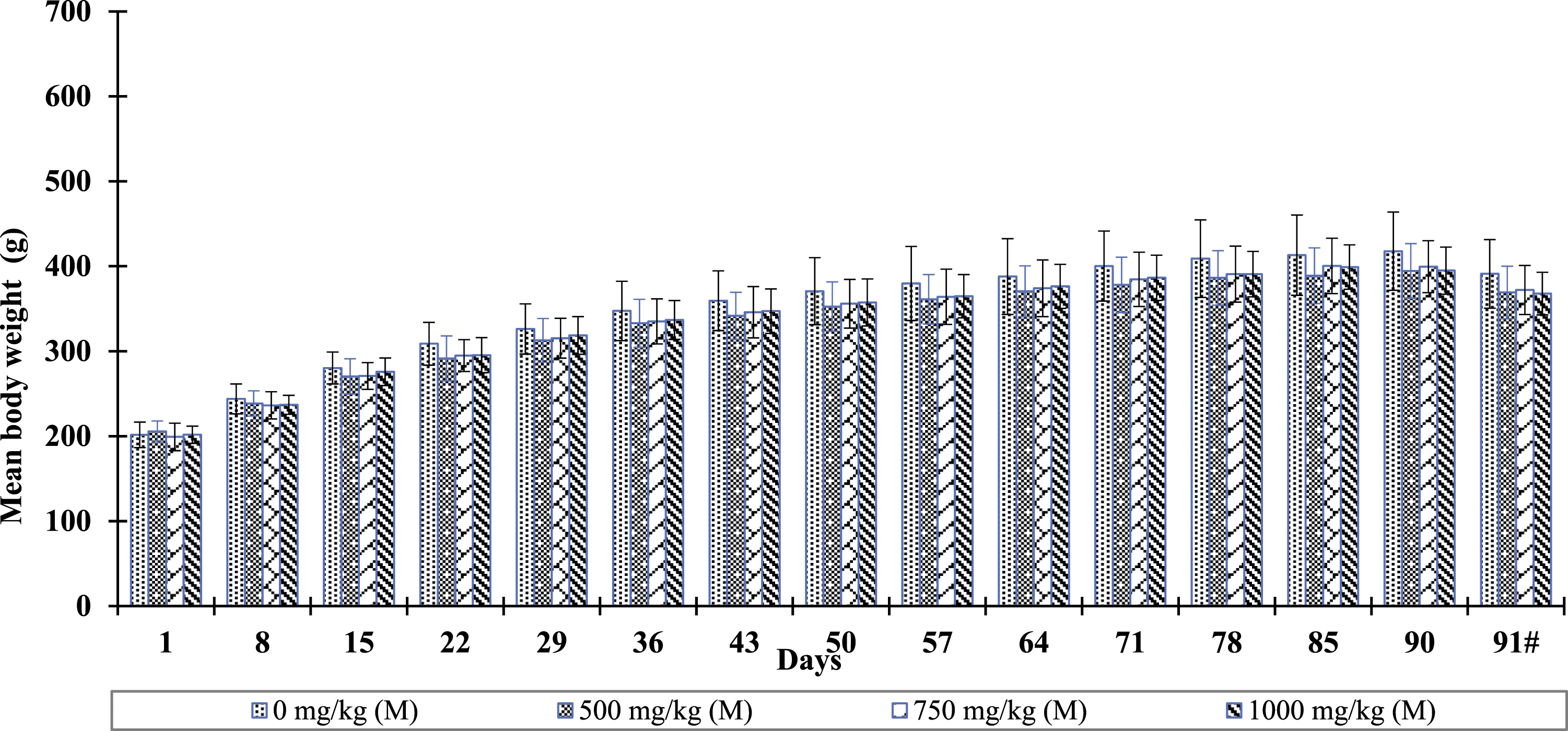
Clinical pathology
Urinalysis data
Bacognize
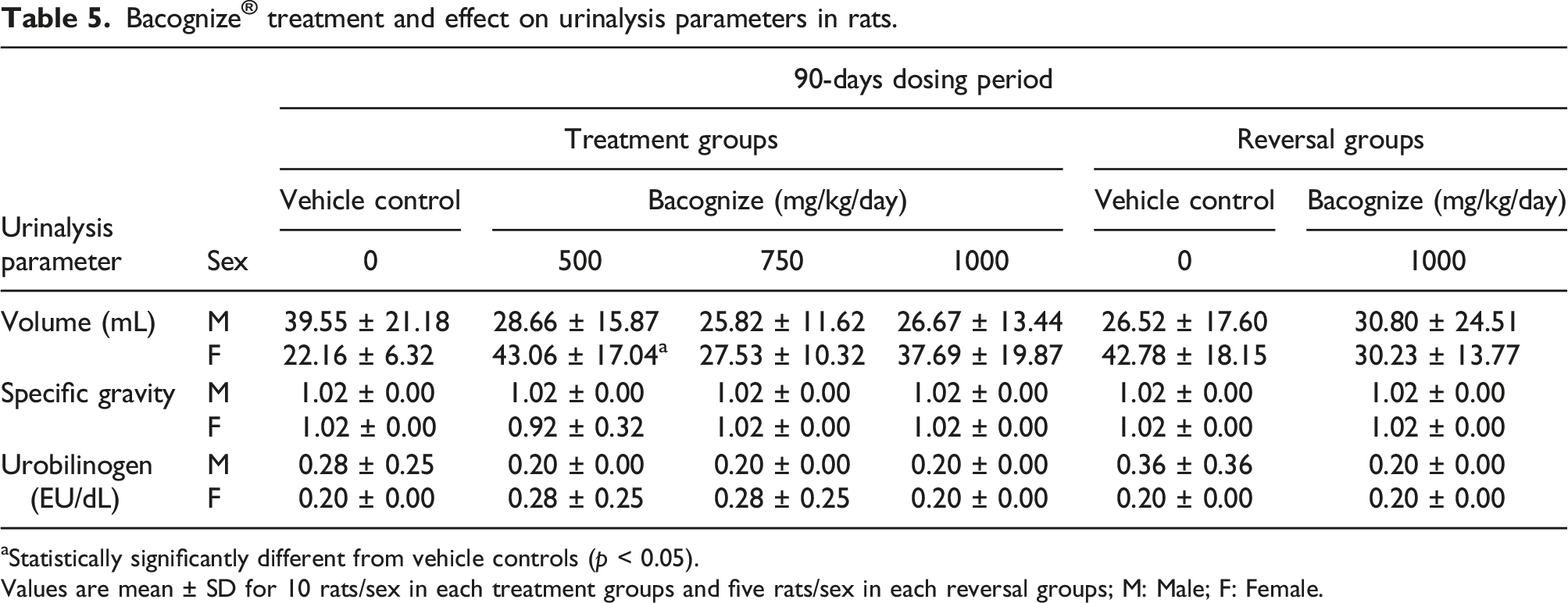
aStatistically significantly different from vehicle controls (p < 0.05).
Values are mean ± SD for 10 rats/sex in each treatment groups and five rats/sex in each reversal groups; M: Male; F: Female.
Hematology
Outcome on hematological parameters on treatment of rats with Bacognize
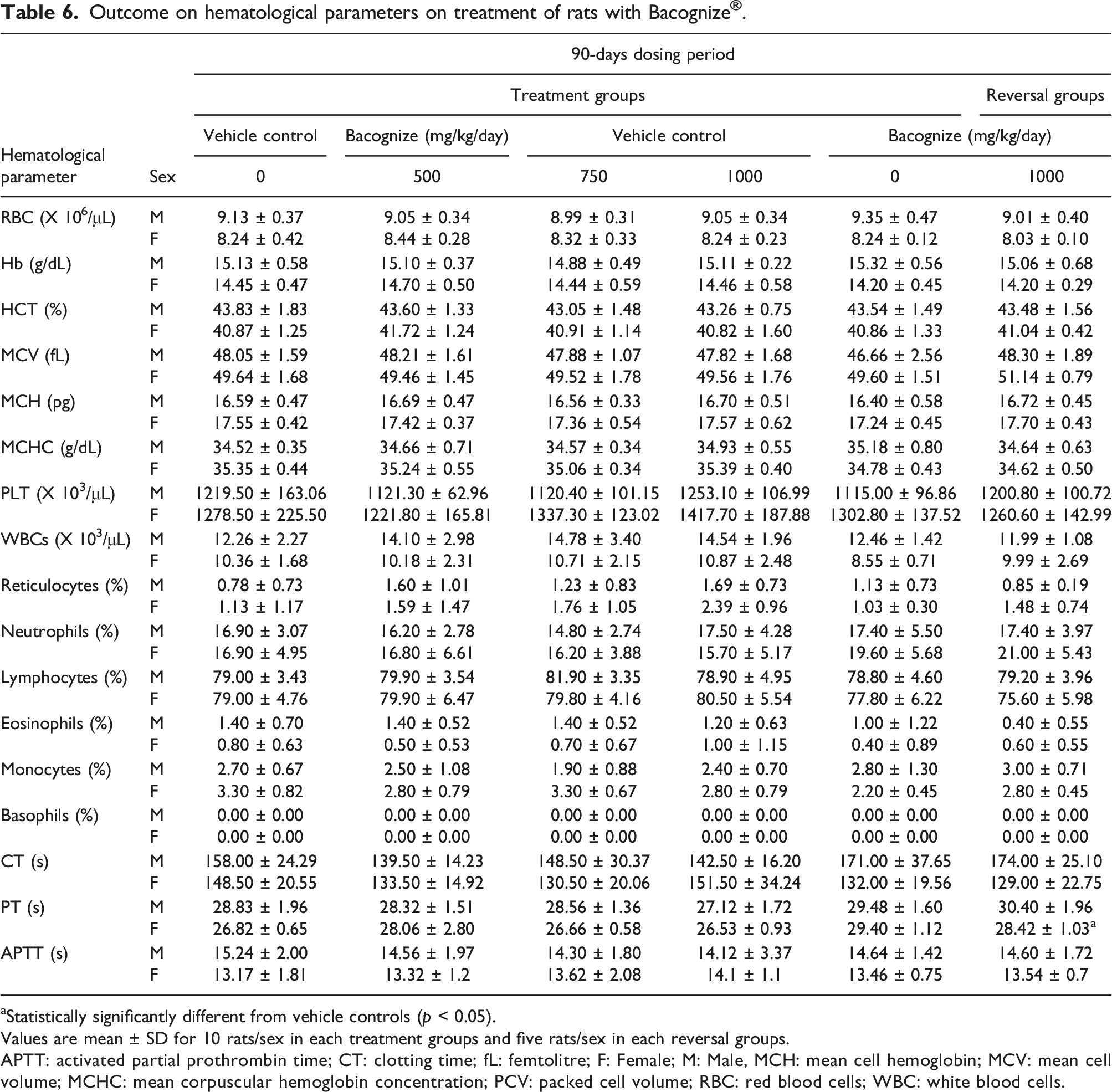
aStatistically significantly different from vehicle controls (p < 0.05).
Values are mean ± SD for 10 rats/sex in each treatment groups and five rats/sex in each reversal groups.
APTT: activated partial prothrombin time; CT: clotting time; fL: femtolitre; F: Female; M: Male, MCH: mean cell hemoglobin; MCV: mean cell volume; MCHC: mean corpuscular hemoglobin concentration; PCV: packed cell volume; RBC: red blood cells; WBC: white blood cells.
Biochemical investigations
Effect of Bacognize
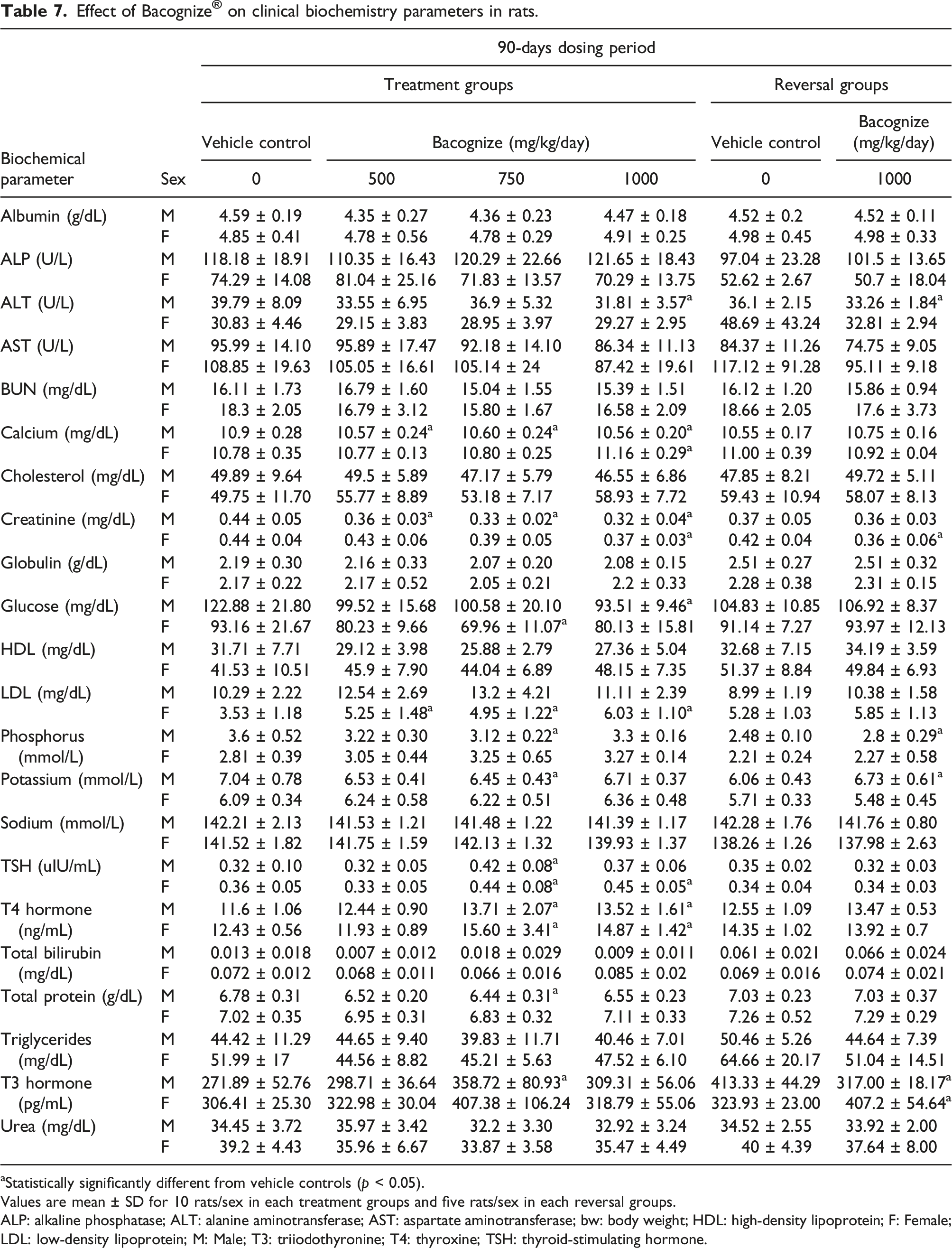
aStatistically significantly different from vehicle controls (p < 0.05).
Values are mean ± SD for 10 rats/sex in each treatment groups and five rats/sex in each reversal groups.
ALP: alkaline phosphatase; ALT: alanine aminotransferase; AST: aspartate aminotransferase; bw: body weight; HDL: high‐density lipoprotein; F: Female; LDL: low‐density lipoprotein; M: Male; T3: triiodothyronine; T4: thyroxine; TSH: thyroid‐stimulating hormone.
In male rats, as compared to the control group, significant (p < 0.05) decreases in serum calcium levels and creatinine in low, mid and high dose groups (500, 750 and 1000 mg/kg bw/day, respectively), alkaline transferase (ALT) in the high-dose and high-dose reversal group (1000 mg/kg bw/day) and glucose levels in the high-dose (1000 mg/kg bw/day) group was observed. Statistically significant increases in T4 in mid-high dose group (750 and 1000 mg/kg bw/day, respectively) and in T3 in mid dose group (750 mg/kg bw/day) were observed. Concurrently, a statistically significant decrease in T3 was observed in the high-dose reversal group compared to the control reversal group. Statistically significant decreases were observed in serum phosphorus, total protein and potassium in the mid-dose group (750 mg/kg bw/day), while an increase was observed in serum phosphorus and potassium in the high-dose reversal group (1000 mg/kg bw/day) compared to the control reversal group. An increase in TSH in mid dose (750 mg/kg bw/day) was also noted.
Similarly, in female rats, compared to the control groups, statistically significant decreases were observed in serum levels of creatinine in the high-dose and high-dose reversal group (1000 mg/kg bw/day), and glucose in the mid-dose (750 mg/kg bw/day). A statistically significant increase was observed in the LDL in the low to high-dose groups (500, 750 and 1000 mg/kg bw/day), T3 in the high-dose reversal (1000 mg/kg bw/day) group, T4 in the mid and high-dose groups (750 and 1000 mg/kg bw/day), serum calcium and TSH level in the high-dose (1000 mg/kg bw/day) and TSH in the mid dose group (750 mg/kg bw/day). There were no other statistically significant differences when the respective control and/or treatment groups were compared.
All significant data mentioned above did not reveal dose-dependency except for the values of creatinine in both male and female animals in treated groups. Decreased values of creatinine did not have a clinical correlation with necropsy or histopathology. Moreover, the animals in reversal groups did not reveal such changes in the values of creatinine and hence the changes were considered incidental instead of a treatment effect. The observations for T3, T4 and TSH were limited to treatment phase groups only and did not demonstrate dose dependency. Changes observed in T3 values in the reversal dose groups exhibited a reverse trend among male and female animals with either no significance (female) or no dose-dependency (male) in treatment phase dose groups. Accordingly, the variation in T3 level was considered a biological variation and not an adverse effect of the test article administration.
The data analysis revealed that all described changes in serum glucose, phosphorous, alkaline phosphatase and proteins were well within the normal laboratory control range and hence considered incidental changes or biological variations and not Bacognize® treatment-related effects. The observed statistically significant changes did not exhibit dose dependency. Other qualitative parameters like total protein, ketone, pH, appearance, glucose, etc. in the Bacognize® treated groups were comparable to the respective control groups.
Organ weights
Effect of Bacognize® on organ weights relative to body weight in rats.
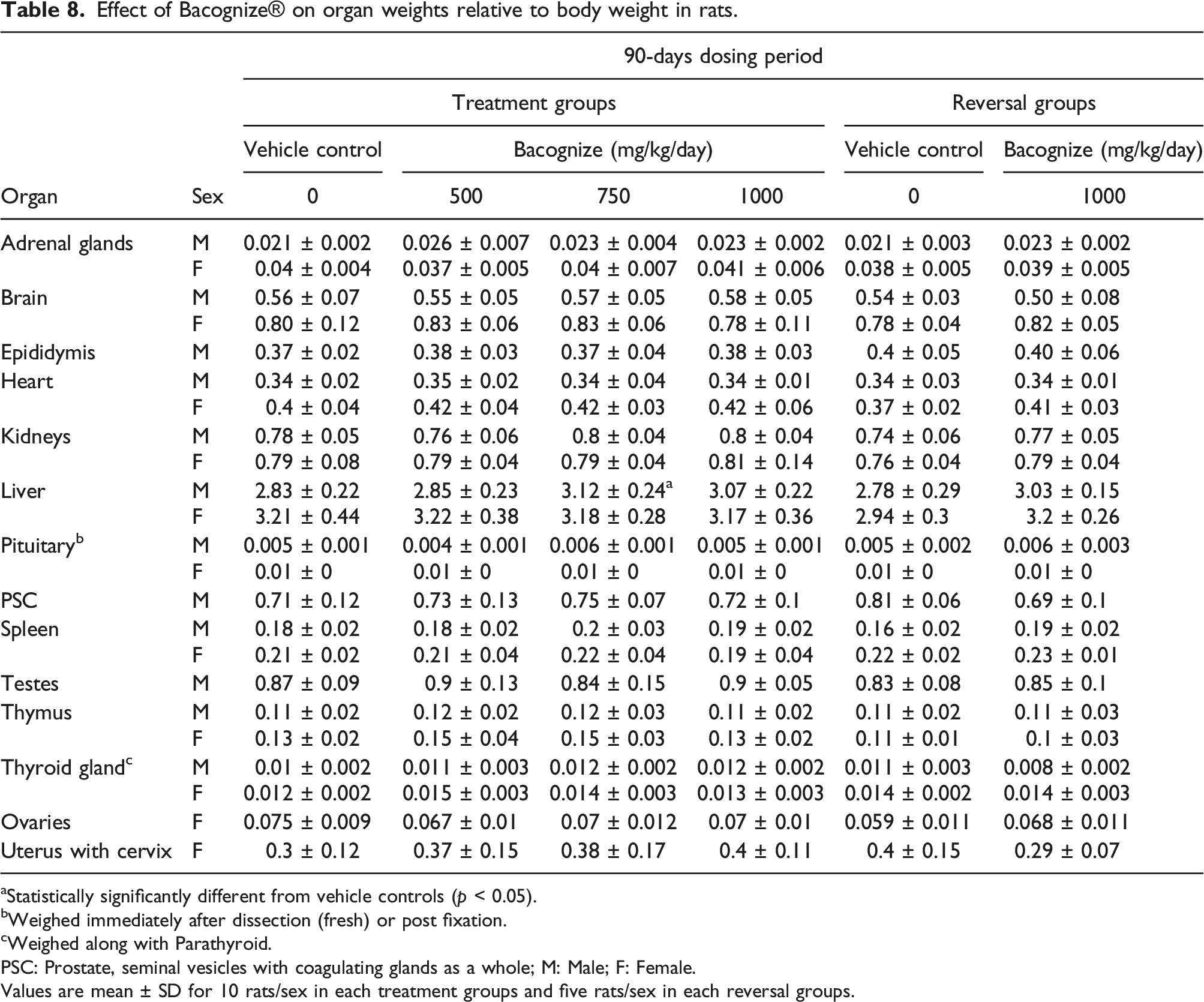
aStatistically significantly different from vehicle controls (p < 0.05).
bWeighed immediately after dissection (fresh) or post fixation.
cWeighed along with Parathyroid.
PSC: Prostate, seminal vesicles with coagulating glands as a whole; M: Male; F: Female.
Values are mean ± SD for 10 rats/sex in each treatment groups and five rats/sex in each reversal groups.
Observation of the oestrus cycle in the female animals belonging to groups I to VI did not reveal abnormal changes and all females exhibited normal cyclic changes. The values of all the above mentioned significant changes were without an absolute dose-dependent pattern or there was no correlation with necropsy and histopathology; hence, these changes were considered as biological variations instead of treatment effects.
Histopathological examinations
No treatment-related macroscopic findings were noted in any of the groups at the scheduled necropsy following the administration of Bacognize® to rats. However, gross pathological findings such as testes and epididymis atrophy and whitish material on the uterus were observed and correlated with microscopic changes. The observed gross lesions were sporadic in nature and observed randomly in animals of different groups, including the control group. Hence, the observed changes could not be considered as treatment-related changes.
Histopathological findings (incidence and severity) in rats post Bacognize® treatment.
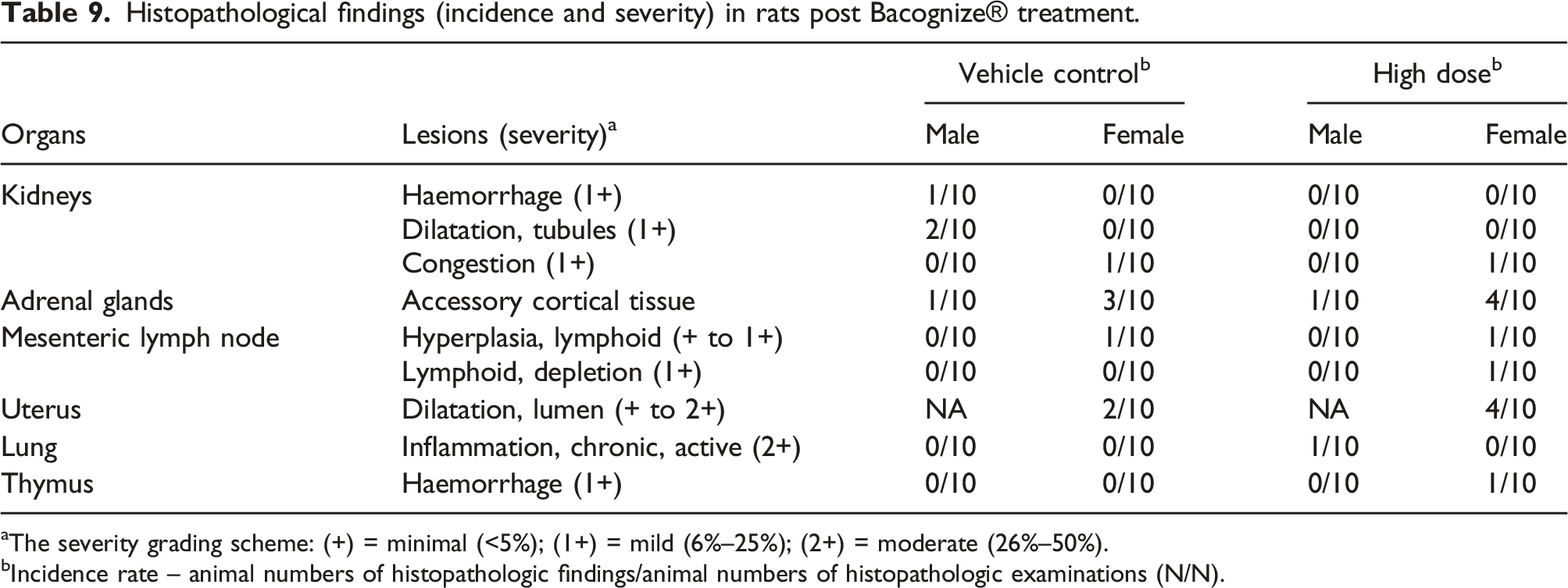
aThe severity grading scheme: (+) = minimal (<5%); (1+) = mild (6%–25%); (2+) = moderate (26%–50%).
bIncidence rate – animal numbers of histopathologic findings/animal numbers of histopathologic examinations (N/N).
Microscopic lesions observed in terminally sacrificed animals were comparable in animals of treatment groups with animals of the vehicle control group and were considered as non-treatment related findings. Also, microscopic lesions observed in reversal control and reversal high-dose group animals were observed in only a single animal of the various groups including the control group animals and were not be considered as treatment-related changes. The other microscopic lesions were comparable in animals of the reversal high-dose treatment group with animals of the reversal control group and were considered as non-treatment related findings.
Discussion
Data from the present subchronic dose-response study in rats did not reveal adverse effects of Bacognize® treatment, at dose levels up to 1000 mg/kg bw/day, as assessed by several parameters which included the appearance of the animals, growth, feed consumption, clinical pathology, organ weights and histopathology. Although statistically significant differences were observed in the Bacognize® treated rats and solvent control with regard to certain parameters, they were not considered as adverse effects of the test article. This was because the changes were either incidental or not related to the treatment. It was also evident that the significant differences were limited to one sex and/or returned to the levels of the solvent control in the reversal groups.
For example, treatment with Bacognize® in male rats revealed a significant decrease in serum calcium levels and a decrease in creatinine in rats receiving 500, 750 and 1000 mg/kg bw/day; glucose levels in rats receiving 1000 mg/kg bw/day; and an increase in T4 and T3 in male rats receiving 750, and 1000 mg/kg bw/day. In female rats, a statistically significant decrease in serum levels of creatinine in rats receiving 1000 mg/kg bw/day; and glucose in rats receiving 750 mg/kg bw/day, and an increase in the LDL in rats receiving 500, 750 and 1000 mg/kg bw/day and an increase in T4 in rats receiving 1000 mg/kg bw/day (Table 7).
Many times the significant changes were not supported by any other changes in clinical parameters or histopathological observations. The microscopic lesions observed in various organs were spontaneous in nature and either comparable with the control group animals or were observed singly in the group and therefore not considered to be a result of test item administration. Therefore, it was concluded that oral administration of Bacognize® did not reveal any adverse change in histopathology of male and female animals up to 1000 mg/kg body weight.
In several Pharmacopoeia from around the world, B. monnieri is listed as a known and accepted herb in many countries. B. monnieri is included in U.S. Pharmacopoeia, American Herbal Pharmacopoeia, British Pharmacopoeia, Indian Pharmacopoeia (IP), the Ayurvedic Pharmacopoeia of India (API), Indian Herbal Pharmacopoeia, the Siddha Pharmacopoeia of India and the Unani Pharmacopoeia of India. Bacopa plant has been used as an ingredient in products of commerce. 16 It appears to be used as a dietary supplement component under provisions of the Dietary Supplement Health and Education Act of 1994. 17 In addition to its traditional use in alternative systems of therapy, Garcia (2006) reported that over 75% of the non-tribal mothers as well as non-tribal children and teens in Wayanad, Western Ghats, India, have identified the plant B. monnieri as a wild food plant. 3 In another report, the inhabitants of Koch Bihar district of West Bengal, India, have identified B. monnieri as a wild edible plant, as it was used as a cooked vegetable. 4 In a review article, Hussain et al. 5 described the use of herbs and their active ingredients, including B. monnieri, for incorporation into foods. Information from other countries also suggests the use of Bacopa in food. In a recent study, Baig et al. 18 optimized calcium gluconate and B. monnieri extract levels in calcium-enriched herbal ice cream by employing response surface methodology and suggested the use of Bacopa in food.
The published literature does not reveal any significant adverse effects of B. monnieri or its preparations at commonly used levels. In an open-label, prospective, uncontrolled, non-randomized trial, investigations were conducted on the effect of Bacopa extract on cognitive functions in Alzheimer’s disease patients. 19 All subjects took 300 mg of Bacopa extract (Bacognize®) orally twice a day (600 mg/day) for 6 months. At the end of the trial, no adverse effects of the treatment were reported. 19 The safety of a daily intake of 600 mg Bacognize®/day is supported by the clinical study and by the extrapolation of an acceptable daily intake (10 mg/kg/day) for a 60 kg individual, from the NOAEL (1000 mg/kg bw/day) established from this subchronic toxicity study in rats after applying a 100-fold safety factor (10 for intraspecies and 10 for interspecies variations).
In some preclinical studies, besides efficacy, relevant safety end-points were also investigated and evaluated from a safety and tolerability point of view. The most relevant is the 90-days repeated dose oral toxicity studies to characterize the safety of BacoMindTM, an enriched phytochemical composition standardized to nine constituents of B. monnieri and indicated for memory enhancement. The authors demonstrated that BacoMindTM was given orally for 90 days to Sprague–Dawley rats up to a dose of 500 mg/kg bw and did not cause any mortality or clinical abnormalities. 13 The present study with Bacognize® compares well with that of BacoMindTM, where the study revealed a very small incidence of clinical signs that were considered to be incidental in the absence of dose dependence and the only difference in the test substances being in the constituents (five vs nine) of the standardized composition. Also, the maximum dose tested with Bacognize® in this study was 1000 mg/kg bw/day. In another pre-clinical chronic repeat-dose toxicity study, oral gavage administration of B. monnieri extracts at doses of 30, 60, 300, and 1500 mg/kg bw/day to rats for 270 days did not produce any evidence of toxicity. 20 In addition, as with Bacognize® in a series of in vitro studies, potential mutagenicity and genotoxicity of Bacopa extract has been investigated and concluded as non-genotoxic. 21 Taken together, the findings from the present study suggest that the intake of Bacognize® by humans for health benefits are unlikely to cause adverse effects.
In summary, the results of the present subchronic toxicity study suggest that oral administration of Bacognize® at levels up to 1000 mg/kg bw/day does not cause adverse effects in male and female rats. Based on the results of this study, the no-observed effect level (NOAEL) of Bacognize® was found to be 1000 mg/kg bw/day, the highest dose tested. The findings from present investigations also suggest that Bacognize® is unlikely to cause any genotoxic effects.
Footnotes
Acknowledgements
We thank Pharmanza Herbal Pvt Ltd for supplying standardized Bacognize® for use in these studies. Editorial assistance (formatting of the manuscript) was provided by Ms Nikeeta Kheradia, Verdure Sciences, and the listed authors have authorized her for the submission of this manuscript.
Author contributions
All the authors contributed to the conceptualization, methodology, investigation and review of the manuscript, while the majority of the writing, review and editing was performed by JM and BM.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JM has worked as a consultant with Verdure Sciences under a cost-reimbursable contract for this project.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
