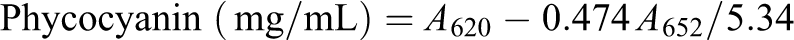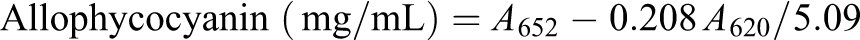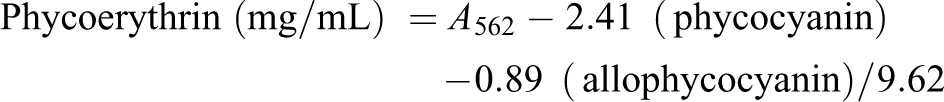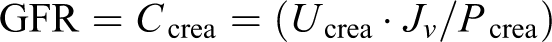Abstract
Arthrospira maxima (Spirulina) is considered a nutraceutical or functional food because it provides health benefits and it is used as nephroprotector because it contains nucleophilic compounds as phycobiliproteins and phycocyanin that prevent oxidative stress and cellular damage process. Also, it is known that inorganic mercury is bioaccumulated and exerted kidney toxicity. Despite the nephroprotective effect of Spirulina and its components, there is not enough information about the effect of them on renal function as well as the apoptosis process inhibition. This work aimed to investigate whether phycobiliproteins and phycocyanin of Spirulina can improve HgCl2-related glomerular and tubular renal dysfunction as well as the Bax, Bcl2, and effectors caspases alterations. Male mice were administrated with Spirulina, phycobiliproteins or phycocyanin 30 min before 5 mg/Kg HgCl2 administration. The nutraceuticals were administrated for the next 5 days. Then, the mice were euthanized. The renal function, caspases 3 and 9 activities, as well as Bax and Bcl2 expression were evaluated. Spirulina and its components prevent HgCl2-related apoptosis induction and glomerular dysfunction. We concluded that phycobiliproteins and phycocyanin of Spirulina reduce glomerular damage but not the tubular dysfunction in a mercury-related acute kidney injury.
Introduction
Mercury (Hg2+) has been used for the chemical industry, but the inappropriate sewage treatment propitiates water bodies contamination that affects all living beings. The kidney is the target organ where Hg2+ is taken up. It has been observed that Hg2+ is bioaccumulated in pars recta and it interferes with organic anion transport. 1 Also, Hg2+ interacts with thiol-containing groups of proteins and enzymes of intracellular organelles, including mitochondria, cytoskeleton, endoplasmic reticulum, and acidic compartments, leading to cell death through necrosis, apoptotic, and autophagic pathways. 2,3 In this way, the reabsorption process, hydroelectrolytic balance, and redox state are modified, and it is associated with acute kidney injury (AKI). 4 The Kidney Disease Improving Global Outcome, Clinical Practice Guidelines, defines AKI respect to the changes in serum creatinine levels or urine volume, according to that, AKI is defined as any of the following: increase in serum creatinine by ≥0.3 mg/dL within 48 h, increase in serum creatinine to ≥1.5 times baseline (the value of prior 7 days), or urine volume <0.5 mL/kg/h for 6 h. 5 AKI is one of the most concerning health problems in both developing and developed countries, where it is encountered in 45% of the patients admitted to the intensive care unit. 6 AKI is associated with 1.7 million deaths per year. 7 On the other hand, Arthrospira maxima (Spirulina) is considered nutraceuticals or functional food because it provides physiological benefits in the prevention and treatment of several diseases. Spirulina has been used as an antioxidant protector in different toxicological models that involved the kidney. 8 –12 The nephroprotective activity of Spirulina and its components is associated with the antioxidant capacity of them. Spirulina contains vitamins (A, B, C, and E), essential amino acids, and fatty acids; as well as another pigment as carotenoids, chlorophylls, and phycobiliproteins as phycocyanin that acts as nucleophilic compounds and they can neutralize free radical and reactive species. 13 Moreover, the Food and Drug Administration considers Spirulina as “generally recognized as safe” food as well as Human and the Dietary Supplements Information Expert Committee concluded that there is not a risk to human health Spirulina consumption. 14
It has been observed that Spirulina has a nephroprotective action against drug or toxicant that causes oxidative stress and renal damage. 9,15,16 Despite the benefits to renal health by Spirulina, there is not enough information about the effect of Spirulina on HgCl2-related glomerular and tubular renal dysfunction in AKI, although it has been reported that Spirulina and its components as phycobiliproteins and phycocyanin can reduce renal dysfunction in chronic kidney disease. 8 This work aimed to investigate whether phycobiliproteins and phycocyanin of Spirulina can improve HgCl2-related glomerular and tubular renal dysfunction as well as apoptosis process in kidney of rats.
Material and methods
Animals and housing
Forty-eight-male albino mouse weighed between 25 and 30 g were used. They were housed in groups of six in a Plexiglas cage, with food and water ad libitum, in a room with constant temperature (21 ± 2°C), 60% of relative humidity, and a 12 h light: 12 h dark cycle (lights on at 0800). All experimental procedures described in this study follow the Mexican Official Standard NOM-082-ZOO-1999 as well as the Guide for the Care and Use of Laboratory Animals from National Research Council (US) Committee (National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals, 2011). Also, the protocol was approved by the Internal Bioethics Committee (CEI-ENCB) with number approbation CEI-ENCB 019/2014.
The animals were randomly assigned into eight groups with six animals each one. The groups received 100 mM phosphate buffer (oral gavage route) + 0.9% saline solution (intraperitoneal route), 100 mM phosphate buffer (oral gavage route) + 5 mg/kg HgCl2 (intraperitoneal route), 1000 mg/kg of A. maxima (Spirulina) (oral gavage route) + 0.9% saline (intraperitoneal route), 1000 mg/kg of A. maxima (Spirulina) (oral gavage route) + 5 mg/kg HgCl2 (intraperitoneal route), 100 mg/kg of phycobiliproteins (oral gavage route) + 0.9% saline (intraperitoneal route), 100 mg/kg of phycobiliproteins (oral gavage route) + 5 mg/kg HgCl2 (intraperitoneal route), 100 mg/kg of phycocyanin (oral gavage route) + 0.9% saline solution (intraperitoneal route), and 100 mg/kg of phycocyanin (oral gavage route) + 5 mg/kg HgCl2 (intraperitoneal route).
The doses used were established according to the antioxidant effect compared with previous studies. 9,17
The administration of A. maxima (Spirulina), phycobiliproteins, phycocyanin, or 0.9% saline solution was done 30 min before HgCl2 administration and repeated for the next 5 days at the same hour (1100). Every day, the animals were weighed. Five days after mercury administration, renal function was evaluated using the metabolic cage for 6 h without food and fluids. After this period, urine samples were collected, also by abdominal compression to ensure the complete emptying of the bladder. Mice were euthanized by decapitation, blood was collected, the serum separated by centrifugation at 3500 r/min for 10 min, and kidneys were quickly removed and weighed. The right kidney was cut longitudinally, and it was used for p-aminohippuric (PAH) uptake test. Meanwhile, the left kidney was used to measure caspases 3 and 9 activities.
A. maxima (Spirulina) culture and phycobiliproteins and phycocyanin purification
A. maxima (Spirulina) was grown in repeated batches in Zarrouk medium (150 mM NaHCO3, 2.87 mM K2HPO4, 29.4 mM NaNO3, 5.7 mM K2SO4, 17.1 mM NaCl, 1.4 mM MgSO4, 0.4 mM CaCl2, 36.9 mM FeSO4, 0.2 mM EDTA, 46.2 mM H3BO3, 9.3 µM MnCl2, 0.95 µM ZnSO4, 2.03 µM Na2MoO4, 0.49 µM Ca(NO3)2, and 0.77 µM CuSO4) at 35 ± 2°C. The culture was supplied with the constant illumination of 180 µE/m2s provided by a halogen lamp over the curse of 24 h. 8 The aeration was constantly using an air pump (Rolf C. Hagen Inc., Montreal, Canada). We used 5 g of the washed cell mass in 20 mL of distilled water to purify phycobiliproteins. It was freezing at −20°C over the course of 24 h twice. The resultant slurry was centrifuged at 10,000 r/min for 10 min to remove the cell debris for 10 times. The phycobiliproteins in the supernatant were spectrophotometrically characterized as described by Bennett and Bogorad, using the equations 18
The phycocyanin was extracted from the phycobiliprotein fraction obtained of A. maxima (Spirulina) as previously, we described. Twenty-five milliliter of the phycobiliproteins-rich extract was put on a column (33-cm length × 4.7-cm diameter) of Sephadex G-250 (Sigma, Co., St. Louis, USA) that had been equilibrated with 100 mM phosphate buffered (PB), pH 7.4. The Sephadex column was eluted with a PB, pH 7.4, with a linear gradient from 100 to 6.5 mM. Five fractions were obtained and were precipitated with a saturated solution of (NH4)2SO4 at 4°C with agitation. After that, each fraction was kept in the dark for 48 h at 4°C. Subsequently, the fractions were dialyzed in distilled water at 4°C for 48 h in the dark, were then dehydrated, and were then collected in tubes and stored at −70°C.
The dialyzed fractions four and five were put on a Diethylaminoethyl-cellulose (DEAE–cellulose) (Sigma, Co.) column (3.3-cm length × 4.7-cm diameter) that had been equilibrated with 50 mM acetate buffer, pH 5.5. The column was eluted with a 50 mM acetate buffer, pH 5.5. The bluish fractions were collected, and they were precipitated with added finely powdered (NH4)2SO4, to achieve a saturated solution at 4°C for 48 h in the dark. They were then dialyzed in distilled water at 4°C for 48 h in the dark and were dehydrated, and stored at −70°C until characterization. 8,9
Renal function evaluation
Renal function was evaluated by determination of glomerular filtration, tubular secretion, and electrolytic balance as previously reported by Estévez-Carmona et al. 19
Glomerular filtration was calculated from the concentration of creatinine in serum and urine (a modified method of Jaffe alkaline picrate) using the conventional equation
where glomerular filtration rate (GFR) or C crea is the creatinine clearance rate (mL/min), U crea and P crea are the creatinine concentrations (mg/dL) in urine and serum, respectively, and Jv is the urinary flow rate (mL/min).
Sodium and potassium concentrations were measured in both urine and serum samples using a Flamephotometer Coleman 51 Perkin-Elmer, Norwalk, Conn. Urinary excretion and clearance of sodium and potassium were calculated from urinary flow and serum and urinary concentration of these cations.
Tubular secretion was measured using PAH technique. PAH was used as an indicator of the renal secretory pathway of organic anions as previously described. 19 Renal cortex slices were obtained, and they were incubated in Ringer solution containing PAH (1 mM) for 1 h. The slices were then removed from the incubation media, the wet weight of the tissue pieces was recorded, the PAH concentration was measured and expressed as the tissue/medium ratio. Also, the glucosuria and proteinuria was evaluated using RANDOX commercial kits.
Effect of Spirulina, phycobiliproteins, and phycocyanins on caspase 3 and caspase 9 activities
Left kidneys were homogenized in 3 mL of 10 mM phosphate buffer (pH 7.4). Caspases 3 and 9 were evaluated spectrophotometrically using the Chemicon International kits (Catalog No. APT131 and APT173, respectively). The caspases activities were detected by cleavage of p-nitroaniline (pNA) from the N-Acetyl-Asp-Glu-Val-Asp p-nitroanilide(Ac-DEVD-pNA) (caspase 3) and Leu-Glu-His-Asp-p-nitroanilide (LEHD)-pNA (caspase 9). Finally, was measured spectrophotometrically at 405 nm.
Bax and Bcl2 expression
We evaluated the proapoptotic (Bax) and antiapoptic (Bcl2) mitochondrial proteins by Western blot assays. Briefly, 50 μg of protein was charged in 10% polyacrylamide gels and separated by electrophoresis and then electrotransferred to polyvinylidene-flouride (PVDF) membranes in a semi-dry chamber. Membranes were blocked for 1 h in blocking buffer (Phosphate buffered saline with tween 20 [PBST]; 0.05% tween 20 in saline phosphate buffer containing 5% low-fat milk Svelty®) under constant agitation. After blocking, membranes were incubated overnight with the primary antibodies diluted 1:1000 (Bax and BCl2 obtained from Santa Cruz Biotechnology, Dallas, Texas, USA; sc-493 and sc-783), at 4°C. After incubation, membranes were washed three times with fresh PBST (20 min per wash) and then incubated in 1:1500 diluted secondary antibody (HPR-conjugated goat anti-rabbit; Life technologies, Rockford, Illinois, USA; 65-6120) for 1 h at room temperature under constant stirring. Membranes were washed three times with fresh PBST, and finally, protein bands were revealed in photographic plates (JUAMA, México) by chemiluminescence, using Luminata TM Forte®, USA (Millipore). Protein β-actin expression was used as charge control and constitutive protein (Santa Cruz Biotechnology; sc-1615, dilution: 1:4000). Optical density (OD) from all bands obtained was analyzed by Image J program (NIH, Bethesda, Maryland, USA; version 1.51p), according to program specifications. 20 Proteins expression was expressed as OD Bax or Bcl2/OD β-actin ratio.
Statistical analysis
For all variables, the results are given as the mean ± SE, and they were evaluated by one-way analysis of variance and Student–Newmann–Keuls post hoc. Values of p < 0.05 were considered statistically significant.
Results
Figure 1 shows that the HgCl2 intoxication causes glomerular dysfunction because it reduces urinary flux, and creatinine clearance with a serum creatine increase. Meanwhile, the A. maxima (Spirulina) and its phycobiliproteins partially prevent the mercury effect on glomerular function. Only the phycocyanin avoids the mercury intoxication that causes glomerular dysfunction.
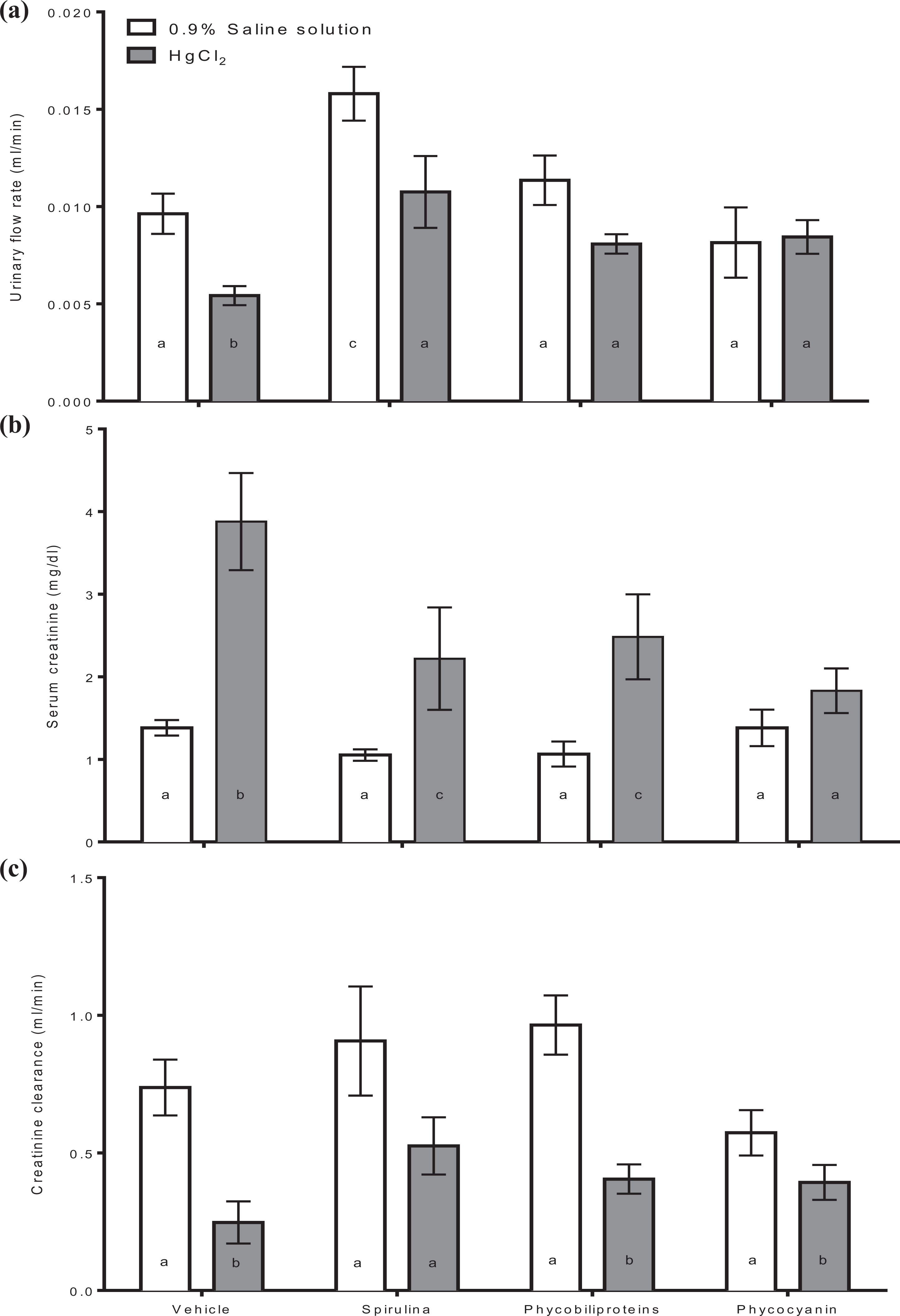
Determination of glomerular function as urinary flow rate (a), serum creatinine (b), and creatinine clearance (c) in intoxicated mice with HgCl2 treated with phycobiliproteins and phycocyanin of Arthrospira maxima (Spirulina). The values represent the mean of 6 independent data ± standard error. p < 0.01 a ≠ b ≠ c. One-way ANOVA and Student–Newman–Keuls post hoc test. ANOVA: analysis of variance.
Table 1 shows that the HgCl2 causes electrolytic misbalance because mercury intoxication reduces the urinary excretion of sodium and potassium and its respective clearances. Respect to electrolytic balance, all treatments partially prevents the electrolytic disturbance of potassium, but the sodium alterations were normalized.
Protective effect of Arthrospira maxima (Spirulina) and its components (phycobiliproteins or phycocyanin) on urinary excretion and clearance of sodium and potassium in intoxicated mice with HgCl2. a
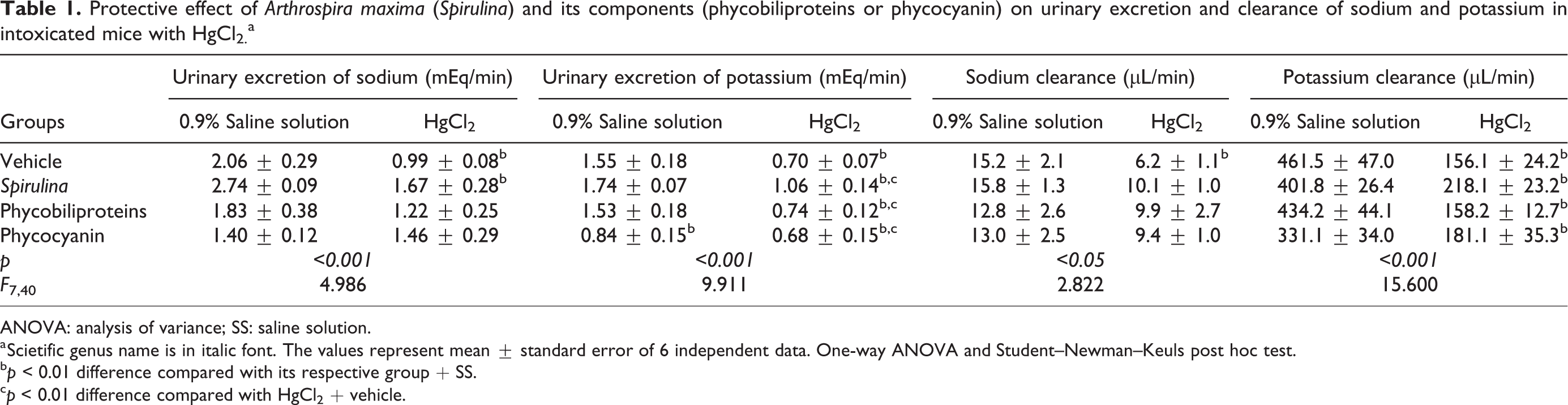
ANOVA: analysis of variance; SS: saline solution.
a Scietific genus name is in italic font. The values represent mean ± standard error of 6 independent data. One-way ANOVA and Student–Newman–Keuls post hoc test.
b p < 0.01 difference compared with its respective group + SS.
c p < 0.01 difference compared with HgCl2 + vehicle.
Respect to renal tubular function, the Figure 2 demonstrates that mercury intoxication causes the reduction of relation t/m in the PAH test (panel a), glucosuria (panel b), and proteinuria (panel c). Meanwhile, all treatment partially avoids t/m PAH decrement, but Spirulina and phycobiliproteins treatments showed a better effect on this variable. Respect to glycosuria and proteinuria, none treatment decreased the mercury effect on tubular dysfunction.
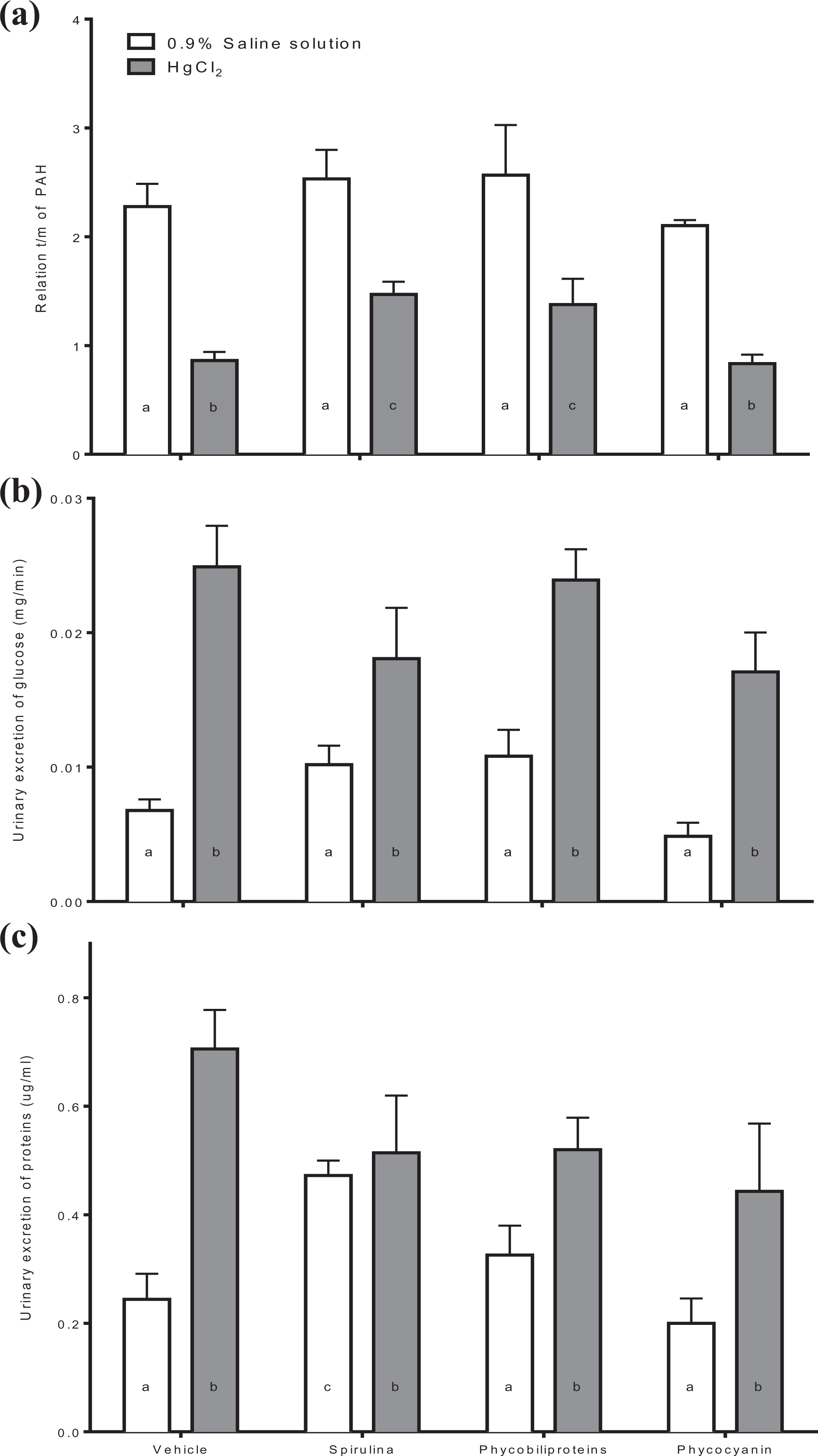
Determination of tubular function as relation t/m of PAH (a), urinary excretion of glucose (b), and urinary excretion of proteins (c) in intoxicated mice with HgCl2 treated with phycobiliproteins and phycocyanin of Arthrospira maxima (Spirulina). The values represent the mean of 6 independent data ± standard error. p < 0.01 a ≠ b. One-way ANOVA and Student–Newman–Keuls post hoc test. PAH: p-aminohippuric; ANOVA: analysis of variance.
Figure 3 shows the effect of the Spirulina and its components on caspases 3 (panel a) and 9 (panel b) activities. It was shown that all treatments avoid mercury that causes overactivation of caspases 3 and 9 in kidney.
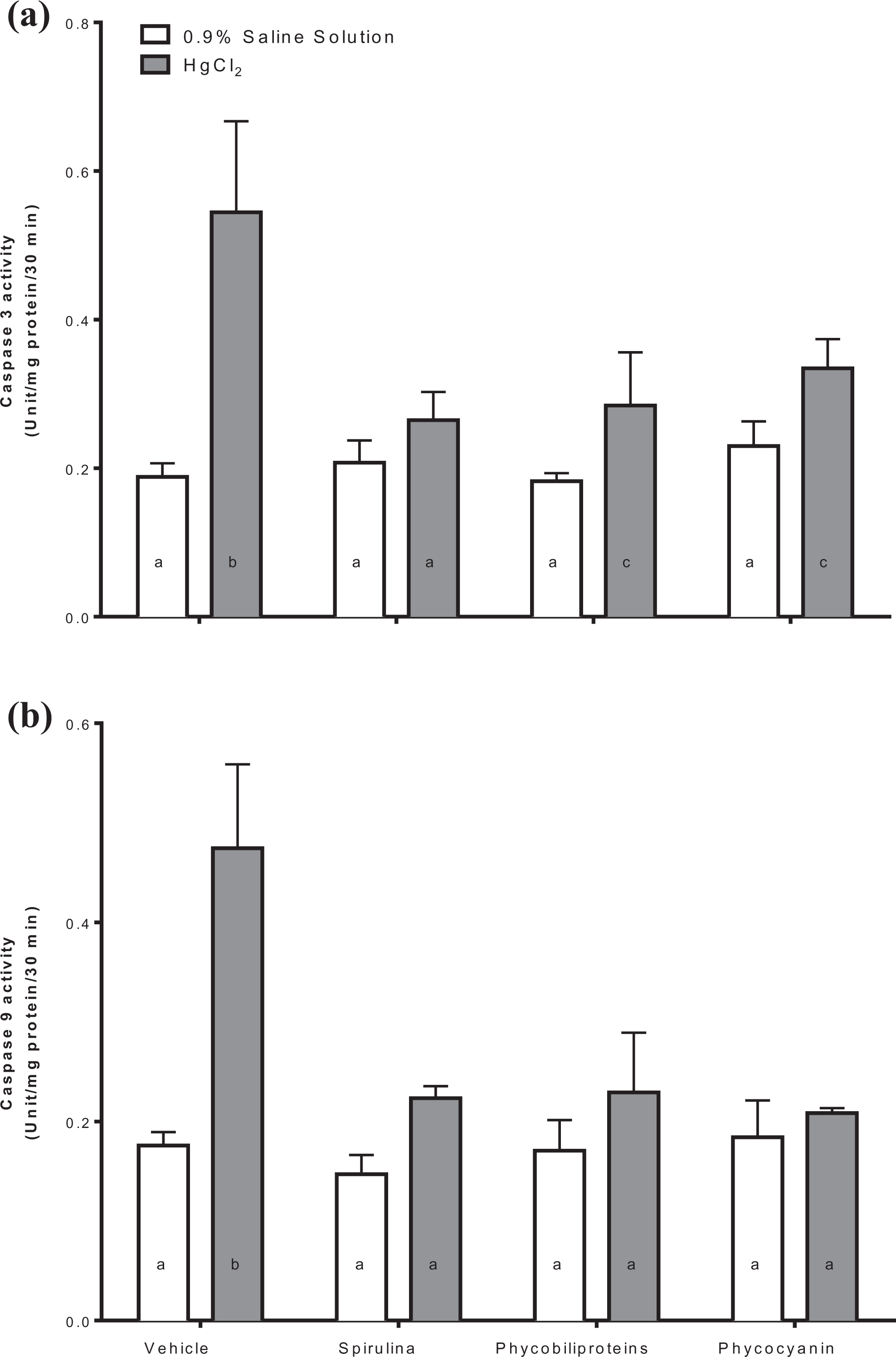
Determination of caspase 3 (a) and caspase 9 (b) activities in intoxicated mice with HgCl2 treated with phycobiliproteins and phycocyanin of Arthrospira maxima (Spirulina). The values represent the mean of 6 independent data ± standard error. p < 0.01 a ≠ b. One-way ANOVA and Student–Newman–Keuls post hoc test. ANOVA: analysis of variance.
We evaluated the proapoptotic protein Bax (Figure 4, panel a) and antiapoptotic Bcl2 (Figure 4, panel b) expression as well as the Bax/Bcl2 ratio (Figure 4, panel c). We can show that HgCl2 enhances Bax expression and it reduces the Bcl2 expression. Thus, HgCl2 causes an enhance of Bax/Bcl2 ratio. The treatments with Spirulina, phycobiliproteins, or phycocyanin prevent HgCl2 causes alteration on Bax and Bcl2 expression in the kidney.
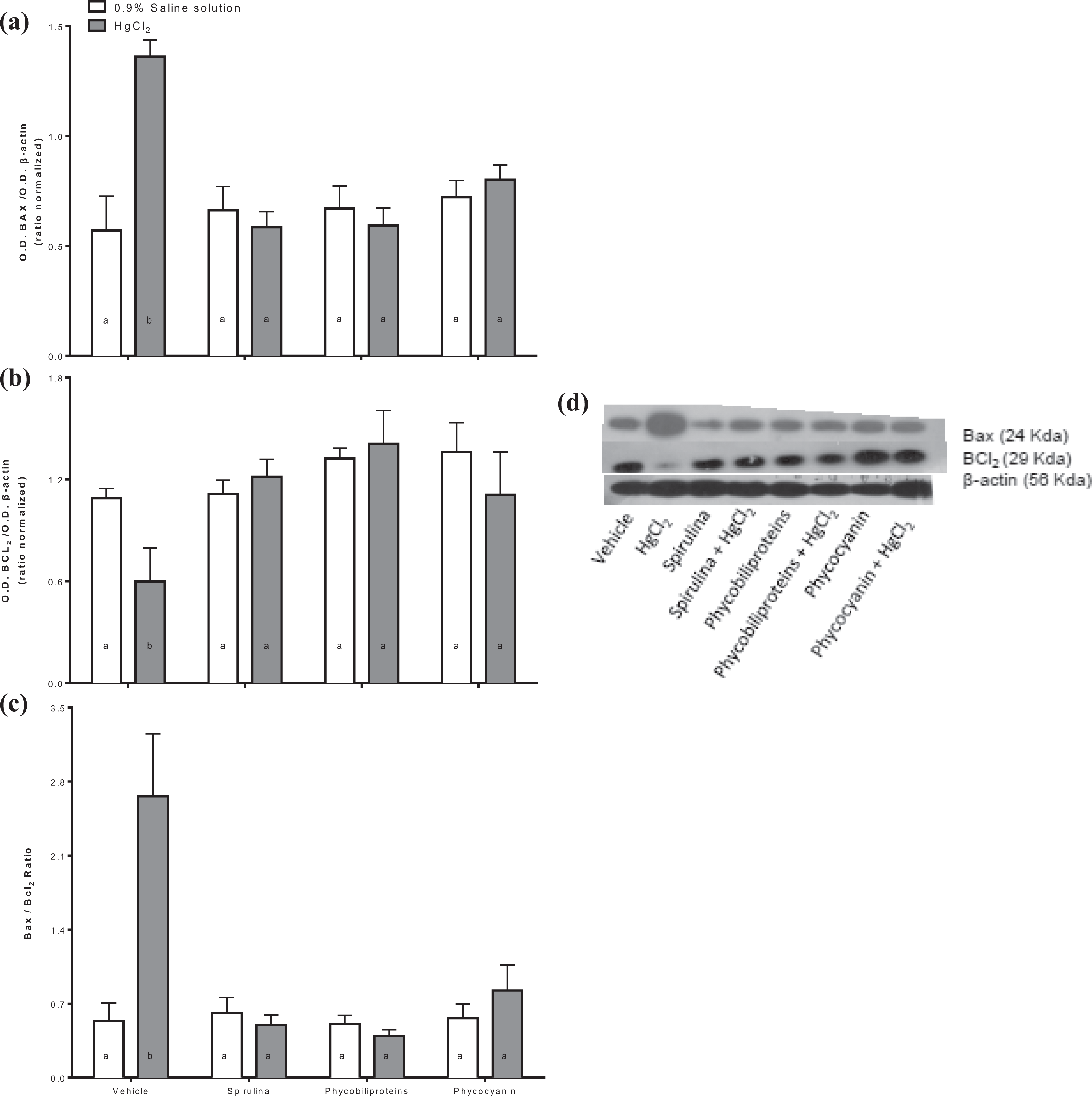
Determination of Bax (a), Bcl2 (b), and Bax/Bcl2 ratio (c) in intoxicated mice with HgCl2 treated with phycobiliproteins and phycocyanin of Arthrospira maxima (Spirulina). (d) Representative blots of the Western blot technique. All values represent the mean of 6 independent data ± standard error. p < 0.01 a ≠ b. One-way ANOVA and Student–Newman–Keuls post hoc test. ANOVA: analysis of variance.
Discussion
In the last years, some scientific research has been reported a nephroprotective effect of Spirulina against the heavy metals intoxication, it prevents the oxidative stress, and renal damage when the animals were intoxicated with cadmium or mercury. 10,21 In search of the bioactive molecules, it has been observed that phycobiliproteins from cyanobacteria have nephroprotective activity against heavy metals-related AKI. 9,22 Even more, it has been reported that phycocyanin has a protective action against heavy metal that causes cellular and renal damage. 23 It is essential mentioned that phycocyanin is the most abundant phycobiliprotein in some cyanobacterium as A. maxima (Spirulina). The nephroprotective property of phycobiliproteins is associated with the antioxidant action of the molecules. Phycobiliproteins contain a tetrapyrrole compound that acts as a nucleophilic compound with the ability to neutralize reactive species and free radicals. 24 However, the effect of phycobiliproteins and phycocyanin on renal function is still unknown in mercury-related AKI. There are some studies which proposed that phycocyanin decrease renal dysfunction, but only using elemental tests such as serum creatinine and blood urea nitrogen (BUN) determination 25 or in another kidney disease as chronic kidney disease. 8 However, our results demonstrated that the administration of Spirulina and its components, phycobiliproteins, and phycocyanin, reduce glomerular but not the tubular dysfunction of mercury-related AKI and this protection could be associated with lower caspases 3 and 9 activations.
The alterations of glomerular and tubular dysfunction caused by mercury intoxication were well documented previously. The Hg2+ is bioaccumulated in kidney, and it causes oxidative stress because of Hg2+ binding with intracellular sulfhydryl-containing proteins and compounds of low molecular weight. 26 Where redox environment is affected, and oxidative stress process begins to damage the most important biomolecules which affect renal function. 27 Also, the Hg2+ reduced the mitochondrial activity affecting the energy production in renal cells which participate in the apoptosis process in resident renal cells. 28 –30 Other toxic effects of inorganic mercury are associated with a modification in plasma membrane permeability. Notably, the Na+-K+-ATPase expressed in basolateral membrane participates in the transepithelial transport process affecting the hydroelectrolytic balances. 4 Although the cellular mechanisms above described were evaluated in vitro and in vivo, the hidroelectrolytic balance was not considered a critical point in the evaluation of new nephroprotective drugs against mercury poisoning. We think that this point is essential because sometimes the reduction in the oxidative process and morphological renal damage do not reflect the renal function. 26
In this research, we evaluated the Spirulina nephroprotective effect on glomerular function. It is remarkable that Spirulina and its components showed an improvement in glomerular function in mice intoxicated with mercury. The intoxicated mice reduced the concentration of serum creatinine, in agreement with some authors, who only considered this variable as a marker of glomerular damage. 31 The mechanism of glomerular toxicity by mercury was described previously by Diamond and Zalups. 1 They proposed that Hg2+ affects the GFR because of the oncotic pressure is equal to hydraulic pressure along the glomerular capillary due to a tubular blockage and damage in renal endothelium followed by spreading and flattening in epithelial cells as podocytes. 1 Thus, the reduction of GFR results in renal dysfunction together with the oxidative stress process and cellular damage; there is a loss of functionality. 9 We observed that Spirulina-treated animals prevented Hg2+ causes a creatinine clearance reduction. Creatinine clearance is a variable related with GFRbecause it gives information during the time. 19 Also, the groups treated with Spirulina, both the control and mercury-intoxicated groups, had an enhance of urinary flow respect their vehicle groups. We can suggest a possible diuretic effect of Spirulina because it can modify the physicochemical forces involved in the GFR as the oncotic or hydrostatic pressures. However, this information is worth because the treated mice with Spirulina or its components could avoid the glomerular dysfunction. The antioxidant capacity of the phycobiliproteins and phycocyanin were associated with the reduction in glomerular dysfunction. 9,22,24 Another possible mechanism of our treatments, which could reduce the cellular damage and renal dysfunction, is the chelating capacity of the phycobiliproteins. 32 Also, it is important mentioning that Spirulina and its metabolites partially prevent the glomerular dysfunction because the glycosuria and proteinuria, variables related to tubular damage, are presented.
The effect of Spirulina and its components on renal tubular function is notorious in this research. Spirulina was capable of attenuating the damage produced by mercury on tubular section of the nephron. We observed that t/m relation is lower in the intoxicated mercury groups, but only the Spirulina and phycobiliproteins partially prevent this reduction at tested doses. The t/m relation is an indicator of active transport in the proximal tubule. Values major to unit indicate the free flux across cells with the help of organic anion transporters dependent on energy, oxygenation, and protein saturation. Thus, the partial prevention of tubular dysfunction could be associated with the antioxidant, and chelating activities as above mentioned because it has been observed that antioxidants maintenance the active transport in proximal tubule as the ATP-dependent transport PAH. 19 Also, the urinary excretion of glucose and proteins indicates the tubular capacity to reabsorb substances and are indicators of renal damage at the proximal tubular area. 4 However, the electrolytic balance improved only in the sodium but not in the potassium systems except the Spirulina treatment, which presented the maximum improvement. The electrolytic balances in Spirulina-treated groups are in agreement with the hypothesis of diuretic activity of cyanobacteria. However, sodium clearance was not modified. Another relevant result was the low excretion of potassium presented by phycocyanin control group per se; this biological property is relevant on therapeutics because the potassium lost is the secondary adverse effect of many diuretic drugs. Thus, our treatments showed a slight improvement in tubular dysfunction.
Finally, the phycobiliproteins and phycocyanin of Spirulina avoid mercury-increased effector caspases activities (3 and 9) and subsequent probably reduced apoptotic events in renal cells. We hypothesized that because in previous studies the morphometrical analysis in kidney reveal less damage when Spirulina or its metabolites were administrated. 9,10 It is known that Hg2+ modifies calcium mitochondrial current promoting the formation of regulated proteaceous pores which cooperate to form the mitochondrial megachannel an expression of proapoptotic Bax protein. This proposal was probed because we show an overexpressión of Bax and a low expression of antiapoptotic Bcl2 in kidney of intoxicated animals with mercury. The consequences of this biological process are the collapse of the mitochondrial transmembrane potential, uncoupling respiratory chain, hyperproduction of free radical superoxide anions, disruption of mitochondrial biogenesis, the outflow of matrix Ca2+ and glutathione, as well as the release of soluble intermembrane proteins. 2,3 Also, Hg2+ promotes the apoptosome formation with the consequent activation of the caspases 3 and 9, which in the end will promote the renal cell death. 1,4,33,34 Recent evidence suggests that Spirulina reduces the expression of caspase 3 in lead causes neurotoxicity 35 and cyclosporine- or gamma radiation causes nephrotoxicity. 36 Moreover, the phycocyanin reduces the mitochondrial dysfunction which prevents apoptosis events in cell culture, 37 as well as phycocyanin, reduced the caspases 3 and 9 expressions on cisplatin-induced nephrotoxicity. 38 Thus, it is probably that Spirulina and its metabolites limit mercury-related cell death process in kidney because they prevent the disruption of the mitochondrial transmembrane potential and the apoptosome formation. These findings have significant implications for the development of new therapies for the treatment of AKI. Considering Spirulina and its components (phycobiliproteins and phycocyanin) as a treatment in the mercury-related AKI, because they have less adverse effects than chelants.
Conclusion
Spirulina and its components (phycobiliproteins and phycocyanin) reduce mercury chloride-related glomerular and effector caspase activation. However, we demonstrated in this work that phycobiliproteins and phycocyanin of Spirulina reduced glomerular dysfunction in AKI caused by mercury. However, our treatments did not avoid tubular dysfunction in a mercury-related AKI.
Footnotes
Authors’ note
The researchers are fellows of EDI, COFAA, and SNI.
Acknowledgments
The authors thank INSTITUTO POLITÉCNICO NACIONAL, SECRETARÍA DE INVESTIGACIÓN Y POSGRADO-IPN, and CONACyT for financial support.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: This study was partially supported by CONACyT (221057) and SIP-IPN (20171416, 20171284, 20170594, 20171233, 20171152, 20181115, and 20180911).