Abstract
Plants grown under contaminated conditions exhibit differences in metal absorption, accumulation, and transportation, and these differences are seen in different plant parts. Metal content in the soil and bark samples collected next to the Sitnica river, which passes through the industrial area of thermal power plants in Kosovo, was measured by inductively coupled plasma optical emission spectrometry. The total metal concentration in willow bark collected from the polluted area of Obilic, Kosovo, ranged 5260–22,280 mg/kg for calcium (Ca), 840–1680 mg/kg for magnesium (Mg), 66.79–910.75 mg/kg for iron (Fe), 5.09–28.66 mg/kg for copper (Cu), 56.39–140.94 mg/kg for zinc (Zn), 19.68–392.75 mg/kg for manganese (Mn), 6.49–10.09 mg/kg for nickel (Ni), 0.10–4.49 mg/kg for cadmium (Cd), 0.85–1.89 mg/kg for chromium (Cr), and 67.79–94.77 mg/kg for aluminum (Al).
Data analysis indicated that correlation between trace elements in the soil and willow bark samples varied with the highest observed in Ni(s)/Zn(p) and Fe(s)/Ca(p) followed by Fe(s)/Mg(p), Al(s)/Ca(p), Cr(s)/Mg(p), Cr(s)/Cu(p), Ni(s)/Ni(p), Cu(s)/Ca(p), and Cu(s)/Zn(p). Correlations among trace elements within willow bark samples varied. The correlation between Cr and Al concentration was the highest, followed by that between Ni and Al. A significantly strong correlation was observed between Al and Fe, Ni and Cr, Cr and Fe, Ni and Fe, and Ca and Mg. The highest transfer factor was established in Zn, followed by Cu > Ni > Cr > Al > Fe.
Introduction
The presence of metals in soil usually varies depending on the type of material and anthropogenic input of metals from industrial processes. Previous studies showed that trace elements can be present in the soil in various forms and with various chemical and physical properties, such as uncomplicated or complex ions in soil liquid; exchangeable ions related to organic material; occluded or coprecipitated with oxides, carbonates, and phosphates; or different secondary minerals and ions in the crystalline structure of principal minerals (residual portion). 1,2 Ecological, environmental, and health outcomes of metal exposure from soil are dependent upon the movement and presence of the metals. 3
Willows (Salix) are used in medicine, and thus, are constantly subject to intensive research on their capability for soil phytoremediation, phytoextraction, and phytodegradation. Willows fulfill nearly all of the requirements for plants used for these purposes, such as fast development with large root systems and the ability to store heavy metals.
Due to high element accumulation, willows are suitable as phytoextractors with high transportation of heavy metals to the shoots and biomass production. 4 The ability of Salix to accumulate high amounts of toxic elements, especially cadmium (Cd), has been established. 5 Nevertheless, of the analyzed elements (chromium (Cr), nickel (Ni), copper (Cu), zinc (Zn), Cd, and lead (Pb)), elevated content in the soil was reported only for Cd and Zn. 6 Between willow types and clones, substantial variation was reported in the uptake of metals. 7
In many climatic zones and adverse microsite states, Salix are characterized by specific physiological characteristics and rapid ecological recovery and are thus useful for conservation and environmental studies. 8 The resistance of willows to elements (Cd, Cu, Zn, Ni, Pb, and iron (Fe)) and their ability to accumulate substantial amounts of metals in their tissues has been established, indicating its potential use for metal extraction. 9
Due to the major ecological problems associated with the pollution of water and soil with trace elements, the use of plants for transferring toxins from the environment, a process known as phytoremediation, is one of the main methods for maintaining ecological balance in the natural environment. Thus, it is important to determine and analyze the elements involved. Regions near industrial areas, smelters, and mining sites are exposed to the atmospheric deposition of heavy metals, which may significantly contribute to the accumulation of trace elements, especially heavy metals in soil. 10
Elements such as Fe, Cu, Zn, manganese (Mn), and Ni are fundamental nutrients, but they become hazardous when their accumulation is greater than the recommended levels, whereas metals such as Pb, arsenic (As), and Cd are in the group of nonessential elements and are highly toxic even in small quantities. 11 The influence of toxic heavy metals on the growth and actions of soil microorganisms do not directly affect the growth of plants because their enzyme reactions are beneficial to plant metabolism. This kind of hazard reduces plant growth, which finally results in the death of the plant. 12
As only part of the elements present in the soil is potentially accessible to plants, the total content of trace elements in the soil is not a good indicator of exposure or risk to plants. 13,14 Plants receive nutrients from soil, water, and air, but they are not completely selective only to essential elements, and they might absorb trace elements, such as heavy metals, which are hazardous even at low concentrations. 15
The main aim of the present study was to determine the pollution level of a soil–willow ecosystem in terms of trace elements by evaluating the possible effect of soil on the availability of elements in polluted areas.
Materials and methods
Thermal power plant “Kosovo A and Kosovo B” specifically designates the Municipality of Obiliq and is about 6.5 km northwest of Pristina, Kosovo. The annual mean rainfall is 638.3 mm, the mean temperature in the study area is 10.9°C, and the mean insulation is 2140 h/year. The wind blows predominantly from the north to northeast with an average velocity of 3 m/s (Kosovo Meteorological Institute, Republic of Serbia). Prior to the determination of heavy metal and other element concentration, 30 soil and plant samples were collected along the 30 km stream near Sitnica river, which passes near Kosovo Thermal Power Plants (KTPP). The sample points are shown in Figure 1 (starting point (sample 1): λ longitude 21°6′41.864″ and ω latitude 42°30′51.67″ and ending point (sample 30): λ longitude 20°57′14.357″ and ω latitude 42°49′29.0712″), and the mean length between the samples was 1 km.
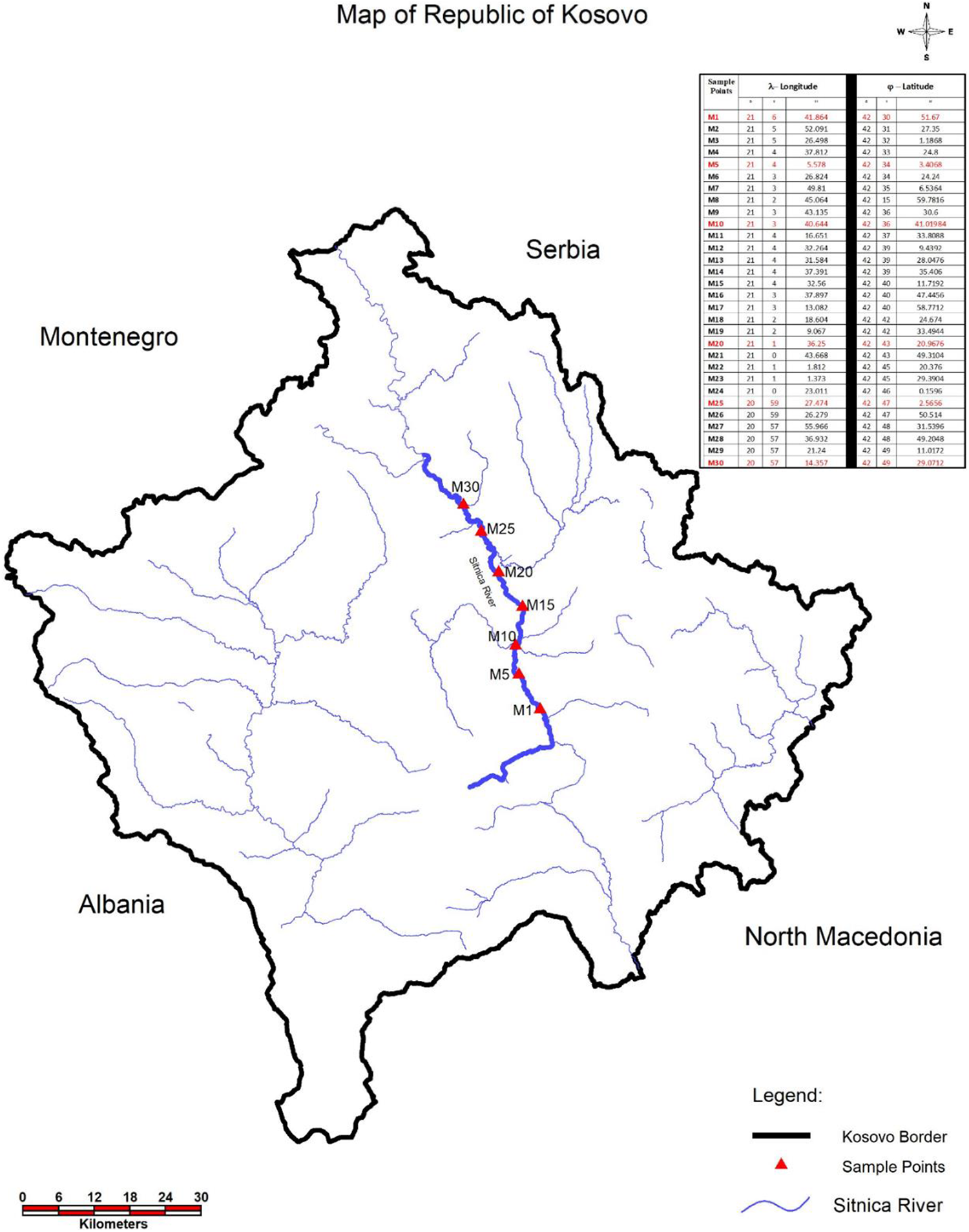
Map of sample locations and geographical locations of the Sitnica river in Kosovo.
From each plot, 2000 g of soil samples were collected from four in an area 1–2 m around the willow tree up to 30–40 cm soil depth and packed in plastic bags. From air-dried (20–25°C for 1 week) and milled out samples, 2.5 g (±1 mg) was sampled and digested with 10 mL aqua regia (mixture of concentrated nitric acid and hydrochloric acid; molar ratio of 1:3) and carefully shaken. After 20 min, the vessels were closed, and the solution was heated in a microwave as follows: 170°C and 30 bar pressure (1 min), 170°C and 30 bar pressure (10 min), 170°C and 30 bar pressure (10 min), and for 30 min (3 × 10 min) at 100°C. After the vessels were cooled to room temperature (20°C), the solution was filtered and diluted with deionized water to 100 mL in a volumetric flask.
White willow (S. alba L., family: Salicaceae) bark samples (200–250 g) were collected 1.5–2 m from the ground from trees that were an average of 3 m from the river and packed in paper bags. To remove any surface particle or impurities, bark samples were initially washed with deionized water and then dried in an oven for 24 h at 65°C. In a microwave digestion vessel containing 500 mg of minced bark samples digested in acid solution, we added 6 mL of concentrated nitric acid and 2 mL of concentrated hydrogen peroxide and carefully shaken. After 20 min, the vessels were closed and heated in a microwave as follows: 145°C and 30 bar pressure (5 min), 170°C and 30 bar pressure (10 min), 190°C and 30 bar pressure (15 min), and then for 20 min (2 × 10 min) at 100°C. After the vessels were cooled to room temperature (20°C), the solution was filtered and diluted with deionized water to 50 mL in a volumetric flask.
Metal content was measured by inductively coupled plasma optical emission spectrometry using PerkinElmer Optima 2100DV (Optical Emission Spectrometer in Markham, Ontario, Canada), and it was set up and optimized according to the manufacturer’s recommended procedures; blank samples were prepared under the same conditions.
Results and discussion
Plants, during their life circle, require basic elements for their development and growth. Some of them, such as Cu, Fe, Ni, Zn, and Mn, are essential nutrients, but others, such as Al and Cd, do not appear to have any essential role in metabolism. Plants are accumulators of air, soil, and water pollutants, and their chemical structure is widely used as an indicator of contaminated areas. 16 Thus, it is needed to determine the amount of elements in plants because at higher concentration these elements become toxic. By using modern methods and applying appropriate standards, the World Health Organization (WHO) also highlighted the necessity for establishing the quality of plants and their products. 17
Thanks to the wide possibilities of adaptation to environmental conditions, willows have found wide applications, for example, in strengthening the banks of water reservoirs, creating buffer zones by sewage treatment plants and landfills, and for the regeneration of sewage from sewage treatment plants and reclamation of degraded lands. 18
The source of the Sitnica river is in central part of Kosovo, and along its length, several rivers with industrial or residential pollution (Graqanka, Drenica) join it, which contribute to the contamination of the river. The Sitnica river passes through the industrial zone of KTPP, which is the major concern of environmental contamination in Kosovo. 19 Salix, mostly Salix alba, grows close to water, and we can find it nearby the Sitnica river. Willows have antipyretic, analgesic, anti-inflammatory, antirheumatic, astringent, and antiseptic properties. 20
All 10 chemical elements (calcium (Ca), magnesium (Mg), Fe, Cu, Zn, Mn, aluminum (Al), Ni, Cr, and Cd) were determined in the bark samples of willow, and six elements (Ni, Cr, Al, Cu, Fe, and Zn) were determined in soil samples. The concentration of the selected elements in willow bark samples collected in the polluted areas of KTPP are presented in Table 1. The plant takes basic elements, such as Zn, Fe, Mn, and Cu, from the soil owing to concentration gradients and selective uptake of these elements. 21
Mean, minimum, and maximum concentration of selected elements in willow bark samples.
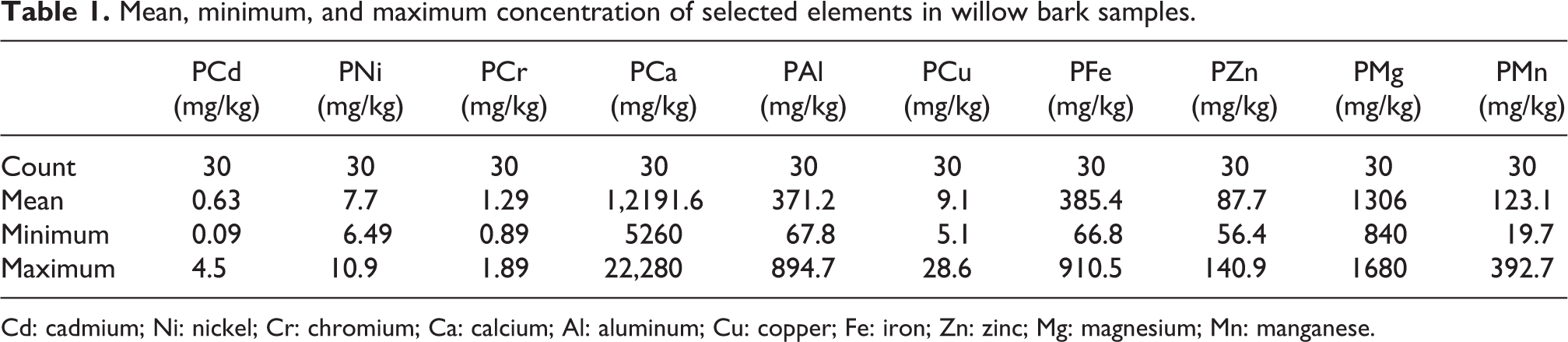
Cd: cadmium; Ni: nickel; Cr: chromium; Ca: calcium; Al: aluminum; Cu: copper; Fe: iron; Zn: zinc; Mg: magnesium; Mn: manganese.
Ca was detected in all plant samples tested ranging from 5260 mg/kg to 22,280 mg/kg. Ca is limited in nature, but too much Ca restricts plant communities on calcareous soils. 22 The highest Ca content was found in the willow bark taken from the south side of the KTPP, whereas the lowest Ca concentration was determined in samples from near the end points in the northern part of the KTPP. Plants growing with appropriate Ca in their environment have shoot concentrations of 0.1% and 5% d. wt. 23 Ca is an essential part of cell walls and is required for the formation of new cells. It helps plants to retain their foliage and in carbohydrate translocation.
The concentration of Mg in plant samples ranged from 840 mg/kg to 1680 mg/kg. The lowest Mg concentration was observed in the starting point, which was approximately 17 km from the KTPP, and the highest concentration was observed in sample 8. Mg activates the many function of enzymes and helps in the formation of fats and oils and carbon assimilation. The values of Mg in willow bark were higher than the permissible limit of Mg in plants, which is reported to be 200 µg/g. 23
The levels of Fe in willow bark samples ranged from 66.79 mg/kg, which was detected in sample 29 approximately 15 km from the KTPP in the northern part, to 910.75 mg/kg, and these results are above the recommended WHO concentration of Fe in medicinal plants (20 mg/kg). Fe is an essential element, is necessary for chlorophyll synthesis, and is a constituent of enzyme systems, helping different enzymatic reactions in plants. Fe is especially involved in oxidation and reduction processes, reduction of nitrite and sulfate, and is predominantly included in photosynthesis. 24 Shortage of Fe is a common nutritional disorder in many plants, resulting in poor yields and decreased nutritional quality. 25
Cu concentration occurred in a range from 5.09 mg/kg to 28.66 mg/kg, the higher end of which was higher than the permissible level of Cu in medicinal plants suggested by WHO (10 mg/kg). The lowest concentration was detected in willow sample 12 collected near the KTPP, whereas the highest value was detected in sample 8. Cu has several important actions in the development and growth of medicinal plants. Cu takes part in hydrogen peroxide production in cell plant walls throughout cell growth (contains amine oxidase) and acts as a catalyst in photosynthesis and respiration. Cu is an essential micronutrient necessary for enzyme function, metabolism, protein synthesis, and detoxification, but an excess could cause toxicity. 26 The anthropogenic discharge of heavy elements into the environment by industrial manufacturing, mining, smelting, agriculture, and waste disposal is a primary source for the higher concentration of Cu in nature, including plants. 27
Zn was detected in all plant samples ranging from 56.39 mg/kg to 140.94 mg/kg, all of which were higher than the suggested level of Zn in medicinal plants (50 mg/kg). The highest level of Zn was detected in samples near the KTPP, whereas the lowest values were detected at the farthest points on both sides of the KTPP. Zn has a long half-life and influences different enzyme activities and the formation of some growth hormones in plants. It stabilizes ribosomal fractions and increases the disease resistance of plants. As stated by Herrero et al., plants contain a specific system of Zn carriers to absorb this element. 28 Elevated Zn in soil suppresses metabolic activities of many plants, resulting in decreased growth and senility. The toxicity of Zn restricts the growth of root and shoot in plants. 29
The Mn content in willow samples varied between 19.68 mg/kg and 392.75 mg/kg, the higher of which was higher than the acceptable level of Mn recommended by the WHO in medicinal plants (200 mg/kg). Contrary to the other elements, Mn deficiency is a common issue, usually occurring in sandy and organic soils with a pH > 6 and in tropical soils. Mn is an essential trace heavy metal for plants that plays a key role in physiological mechanisms. It is involved in chlorophyll formation, photosynthesis, and metabolic functions in plants. It should be highlighted, however, that the amount of Mn in plants is not only a result of plant properties but also a result of the pool of available Mn, which is considerably dependent upon soil properties. 30
Al was detected in all plant samples ranging from 67.79 mg/kg to 894.77 mg/kg, whereas the typical values varies from 10,000 to 300,000 mg/kg. 30 Al decreases root respiration, interferes with enzymes, reduces the synthesis and transport of cytokines, and modifies the composition and activity of plasma membranes, which affects absorption, transport, and usage of different elements. The most simply identified toxicity with Al is the inhibition of root growth, and this symptom is an established measure of Al stress in plants. 31
The level of Cr ranged from 0.85 mg/kg to 1.89 mg/kg and was predominantly within the acceptable level of Cr suggested by WHO for plants (1.30 mg/kg). 23 The toxicity of Cr in plants is based on its valence state, that is, Cr (III) has lower toxicity compared with Cr (VI). Cr almost gets accumulated by ion transporters, such as sulfate or Fe, and not absorbed immediately by plants. Cr toxicity affects not only the germination process of plants but also total growth by influencing photosynthesis and other metabolic processes until the completion of the dry matter production. 32,33 Cr is an essential metal for plant development and is easily absorbed by the epidermis and roots of plants, which then enter the woody tissue from the internal or external membranes. 34
Cd effects biochemical, physical, and genetic levels in plants. The concentration of Cd varied from 0.10 mg/kg to 4.49 mg/kg. These values are extremely high compared with the WHO recommended level (<0.02 mg/kg). Cd in plants has no vital biological activity and is extremely toxic even at low concentration, but it has a tendency to concentrate in plants and aquatic organisms. 35
Ni level ranged from 6.49 mg/kg to 10.09 mg/kg, which is within the higher allowed level recommended by WHO (10 mg/kg). The accepted Ni limit in plant tissues is in the range of 0.5–5 mg/kg. 36 Ni is a basic nutrient for plant growth and development, but the level of Ni necessary for normal growth in plants is very low. Ni is an essential heavy metal, whereas excessive Ni levels in the soil could result in toxicity in plants. 37 Limited growth of plants and damage induced by high levels of Ni were observed over a long period.
The results in Table 1 lists that the concentration of selected elements (mean results in mg/kg) in willow bark was in the following order: Ca > Mg > Fe > Al > Mn > Zn > Cu > Ni > Cr > Cd. The experimental data presented in Table 2 revealed that Cr concentration in soil samples occurred from 18.06 mg/kg to 35.85 mg/kg and Ni concentration was from 66.74 mg/kg to129.59 mg/kg. The concentration of Al ranged from 12,021.88 mg/kg to 19,279.32 mg/kg, Cu ranged from 29.88 mg/kg to 100.02 mg/kg, Fe ranged from 13,216.97 mg/kg to 20,399.00 mg/kg, and Zn values ranged from 99.23 mg/kg to 948.71 mg/kg.
Heavy metal concentration (mg/kg) in soil samples and transfer factor values.
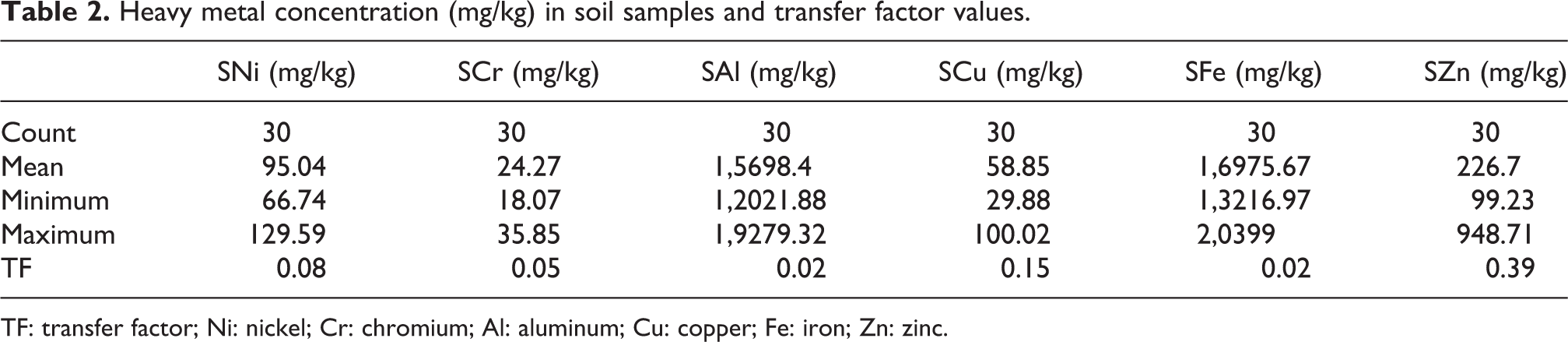
TF: transfer factor; Ni: nickel; Cr: chromium; Al: aluminum; Cu: copper; Fe: iron; Zn: zinc.
From the results of the analysis of soil and plant samples, the average accumulation of selected metals was higher than that reported in the previous studies 38 and the acceptable levels recommended by Food and Agriculture Organization (FAO)/WHO. 39 As many plant varieties are sensitive to micromolar levels of Al, the possibility of soil being Al toxic is significant. 31 Linear correlations were conducted to analyze the accumulation of selected elements in S. alba samples and their corresponding soil samples and are presented in Table 3.
Spearman’s rank correlation coefficient and the level of significance (r, p) for relationships between selected elements in soil and plant.
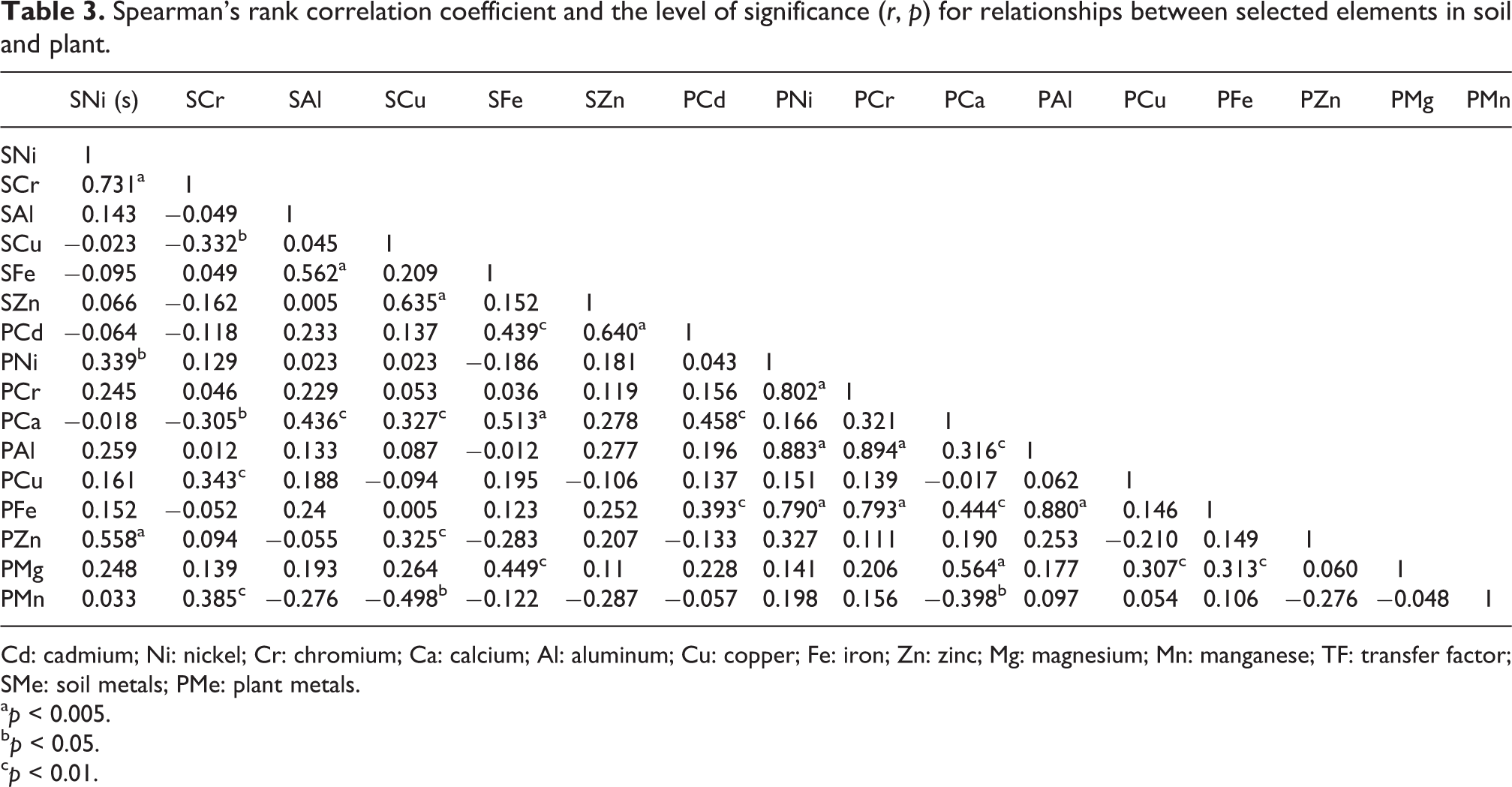
Cd: cadmium; Ni: nickel; Cr: chromium; Ca: calcium; Al: aluminum; Cu: copper; Fe: iron; Zn: zinc; Mg: magnesium; Mn: manganese; TF: transfer factor; SMe: soil metals; PMe: plant metals.
a p < 0.005.
b p < 0.05.
c p < 0.01.
The metal or metalloid toxicity can be evidently modified by interactions with other toxic and essential metals. 11,40 The correlation between the concentration of selected elements was high in Ni in soil and Zn in willow (r = 0.558), and Fe in soil and Ca in willow (r = 0.513) (Figure 2). Significant correlations between the concentration of Fe in soil and Mg in willow (r = 0.449), Fe in soil and Cd in willow (r = 0.439), Al in soil and Ca in willow (r = 0.436), Cr in soil and Mn in plant (r = 0.385), Cr in soil and Cu in plant (r = 0.343), Ni in soil and Ni in plant (r = 0.339), Cu in soil and Ca in plant (r = 0.327), and Cu in soil and Zn in plant (r = 0.325) were observed.
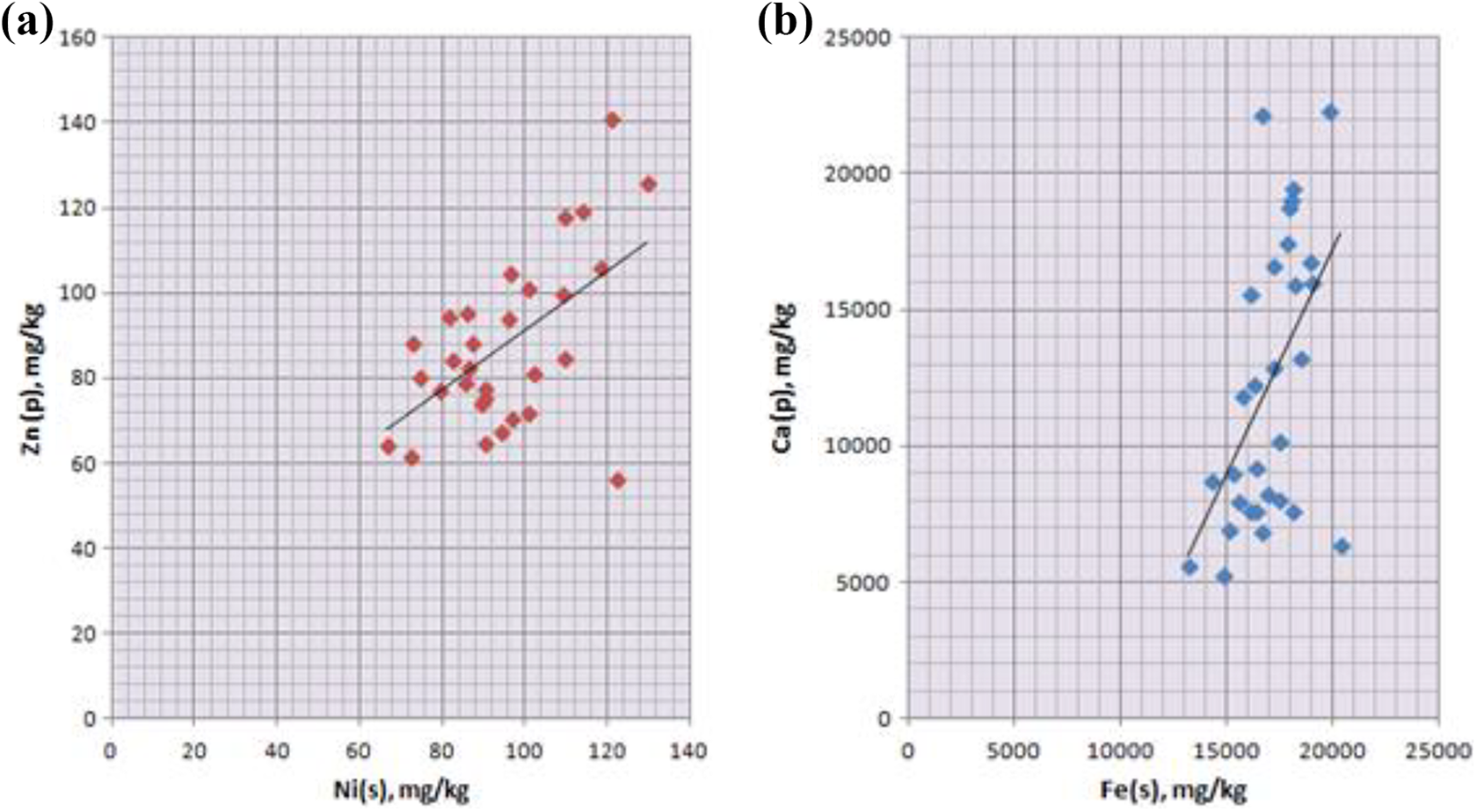
(a and b) Relationships between trace element concentrations in willow bark and local soil of Salix alba L. at its natural site of growth.
Correlations between trace elements in plant samples varied. The correlation between the concentration of Cr and Al was the highest (r = 0.894), followed by correlation of Ni with Al (r = 0.883). Significantly strong correlation was observed between Al with Fe (r = 0.880), Ni with Cr (r = 0.802), Cr with Fe (r = 0.793), Ni and Fe (r = 0.790), and Ca and Mg (r = 0.594). Moderate correlation also was detected between concentration of Cd with Ca and Fe in willow, Ca with Fe and Al, Fe with Mg, and Cu with Mg.
The transportation of elements from the soil to plant parts was analyzed by applying an index called the transfer factor (TF). The TF of the selected elements from soil in plants was determined as follows:
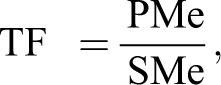
where TF is the transfer factor, PMe is the metal concentration in plant (mg/kg), and SMe is the metal concentration in soil (mg/kg).
High TF values (≥1) suggest elevated uptake of metals by the plant from the soil and higher adaptability for phytoextraction and phytoremediation of plants. Conversely, lower values suggest weak feedback of plants against metal uptake, indicating that the plant can be used for human food. 41 If the transfer index is above 0.5, then the plant will have a higher probability of pollution with elements from anthropogenic activities. 42 The TF obtained values and results with analysis of correlation between the selected elements are presented in Table 2. The highest TF was observed in Zn followed by Cu. There was low level of TFs for Ni, Cr, Al, and Fe.
Conclusion
It should be pointed that pollution through the coal flying ash, in the industrial area of Thermal Power Plants (Kosovo) cause important bioaccumulation of trace elements in the following order: Zn > Cu > Ni > Cr > Al and Fe. Cd concentration in willow bark was higher than the limits recommended by the WHO, whereas the concentration of Ni and Cr was within the upper or slightly higher than the allowed limits. Research on heavy metals in soil shows that the concentration of Ni and Cr was higher than the recommended values by FAO/WHO.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
