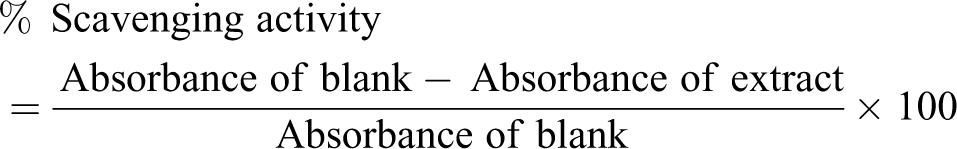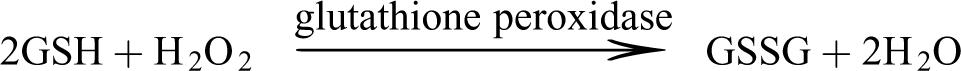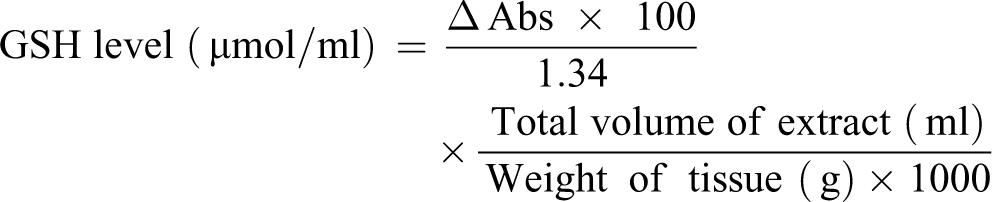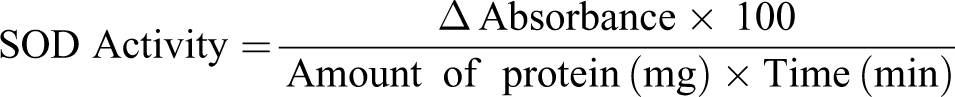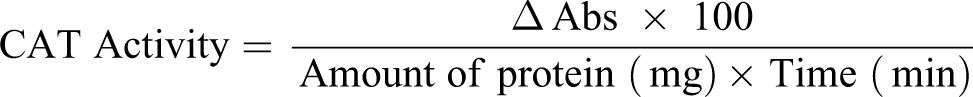Abstract
The pharmacological effects of medicinal plants are due to the presence of certain chemical compounds present in them. Previous studies have shown that crude extracts of Tapinanthus bangwensis (T. bangwensis) possess hepatocurative potential. However, the present study aims on evaluating the antioxidant activity as well as identifying the chemical compounds in ethylacetate fraction of T. bangwensis that ameliorate carbon tetrachloride-induced hepatotoxicity in Wistar rats. Six subfractions of ethylacetate fraction were obtained by column chromatography. The antioxidant activity was determined by 2,2-diphenyl-1-picrylhydrazine (DPPH), biochemical assay determined by spectrophotometry, and compound elucidation by liquid chromatography–mass spectroscopy (LC-MS) analysis. The DPPH result shows that subfraction F0 exhibited the strongest antioxidant activity followed by F4 while F1 exhibited the lowest. Oral administration of 100 mg/kg body weight of the subfractions of ethylacetate fraction of T. bangwensis increases superoxide dismutase and catalase activities and also glutathione level and decreases malondialdehyde level compared to the positive control group. Subsequently, aspartate aminotransferase, alanine aminotransferase, alkaline phosphatase, total bilirubin, and conjugated bilirubin were reduced while albumin and total protein levels increased compared to the positive control group. However, there were no significant differences between the positive control group and the group induced and treated with the subfractions at p < 0.05. The histopathology study shows normal hepatocyte distribution with no fat deposit in the induced and treated groups while fatty liver was observed in the positive group. The anti-hepatotoxic effect was higher in F4 than in other subfraction treated groups. Hence, the LC-MS analysis of F4 reveals the presence of 8-hydroxyluteolin-8-glucoside, Avicularin, Fisetin-7-glucoside, Isoscutellarein-7-xyloside, and Isovitexin, respectively, and has been reported to exhibit antioxidant and hepatocurative activities. It can be concluded that the hepatocurative effect could be due to the chemical compounds identified above.
Keywords
Introduction
The liver performs over 500 biological and metabolic functions such as detoxification, deamination, synthesis, storage, homeostatic regulation of the body, and so forth. 1 –3 The susceptibility of the liver to metabolism-dependent drug-induced damage is due to its defective detoxifying function. 4,5 This is because active metabolites (free radicals) when produced during xenobiotic biotransformation could periodically cause changes in liver’s microscopic structures, bile acid transporters, and biomolecules consequently causing liver apoptosis/necrosis and metabolic disorders. 6,7 Carbon tetrachloride (CCl4) is one of the widely known hepatotoxic agents that elucidate liver damage by enzymatic activation of CCl4 by cytochrome P450 to produce carbon trichloromethyl free radicals (*CCl3) within the membrane of the endoplasmic reticulum. These free radicals bind covalently to polyunsaturated membrane lipid resulting in increased formation of lipid peroxyl free radicals through lipid peroxidation and influence pathological changes in the liver’s structure and function. 8 Due to the harmful effects of these free radicals and the adverse side effects associated with synthetic drugs, researchers considered phytotherapy as an alternative remedy. 9 Antioxidants are well-known free radical scavengers and plants are considered to be rich in phytochemical compounds with potent antioxidant properties such as flavonoid, saponin, tannin, and phenol, and the mechanism of action could be by either (1) stimulating the liver to release certain chemicals or enzymes to detoxify agents that potentially cause liver damage (2) improving liver damage repair mechanism or (3) biotransforming active oxygen molecules (species) into nontoxic compounds. 10,11 Tapinanthus bangwensis (T. bangwensis) (Engl. & K. Krause) Danser is a citrus-dependent plant of African origin. In Africa, it is popularly called African mistletoe. The English acronyms are “all heal tree,” “bird lime,” or “tree of life.” Traditionally, in Nigeria, T. bangwensis in Hausa is called Kauci (Kanchi), Yoruba—Afomo onisana, and Igbo—Awurusie or Apari. 12 Ethno-medicinally, it has been reported to possess anti-inflammatory, antimicrobial, anticancer, hypotensive, hypoglycemic effects, and so on. 13,14 Previous studies provide limited information on the chemical compounds responsible for the hepatocurative potential of T. bangwensis. Therefore, the present study was undertaken in order to evaluate the antioxidant activity and identify the chemical compounds present in ethylacetate fraction of T. bangwensis that ameliorate CCl4-induced hepatotoxicity in Wistar rats.
Materials and methods
Chemical reagents
All the chemical reagents used in this work such as methanol, ethylacetate, hexane, n-butanol, glacial acetic acid, CCl4, potassium acetate, Folin–Ciocalteu reagent, hydrogen peroxide (H2O2), 2,2-diphenyl-1-picrylhydrazine (DPPH), 5,5-dithio-bis(2-nitrobenzoic acid) (DTNB) (Ellman’s reagent), sodium pyrophosphate, methosulfate phenazine, nitroblue tetrazolium chloride, nicotinamide dinucleotide (NADH), 1,1,3,3-tetraethoxypropane, trichloroacetic acid (TCA), thiobarbituric acid (TBA), ascorbic acid, and potassium chloride (KCl) were of analytical grade from Zigma in the United Kingdom.
Preparation of plant material
Fresh leaves of T. bangwensis were obtained from Lagos, botanically identified by Mr Adeleke, Department of Pharmagnosy, University of Lagos, and deposited at the University’s herbarium with the voucher number LUH 3856. The leaves were washed, dried for 5 days under dry climatic condition at room temperature, and pulverized into a fluffy mass using electric blender.
Extraction of plant material
About 2.15 kg of T. bangwensis ground leaves was extracted with 10 L of 100% methanol solvent using cold maceration method. The mixture was left for 48 h with intermittent shaking at intervals to increase the surface area for readily solvent penetration. The mixture was filtered using Whatman paper and the filtrate concentrated by evaporation at room temperature until a constant weight of 187.5 g (8.71%) was obtained. About 60 g of the concentrated methanol extract was further extracted by solvent partition method using separating funnel. The solution mixture was ethylacetate–water in 3:2. Two layers were observed. The upper portion was ethylacetate fraction which was collected and air-dried at room temperature until a constant weight of 20.45 g was obtained.
Column chromatographic extraction
The method of Vivex et al. 15 was adopted. A cylindrical glass column (55 × 22 cm2) was packed with 100 g of silica gel (mesh size of 60–120) (stationary phase) dissolved in hexane solution. A 15 g of the dried ethylacetate fraction was mixed with 30 g of the silica gel to form a slurry particle. The slurry particle was then placed on top of the prepacked silica gel column and the sample covered with a layer of cotton wool. Four solvents of different polarities were used, namely hexane, ethylacetate, methanol, and water. The solvent mixtures were hexane (100%), hexane–ethylacetate (80:20, 60:40, 40:60, 20:80, and 100%), ethylacetate–methanol (80:20, 60:40, 40:60, 20:80, and 100%), methanol–water (80:20, 60:40, 40:60, 20:80, and 100%), and finally water only (100%). A total of 105 fractions were collected in 100 mL bottles and numbered consecutively. The fractions were covered with perforated aluminum foil and concentrated by air-drying. The various concentrated fractions were then pooled together based on their retention factors via an activated thin layer chromatography (TLC) and a total of six subfractions were obtained labeled as F0, F1, F2, F3, F4, and F5, respectively.
Animal model and management
Forty apparently healthy male Wistar rats weighing 100 g were used. They were maintained under standard laboratory conditions according to the animal welfare regulation. 16 The rats were acclimatized for 2 weeks under standard environmental conditions (temperature, 25°C; humidity, 55%; and photoperiod, 12:12 h light:dark cycle) in airy plastic cages and were fed with rat feed and distilled water.
Experimental design
The rats were divided into eight groups with five rats in each group: Group A: Rats received feed and water only (normal control group). Group B: Rats induced with 120 mg/kg body weight (bwt) of CCl4 intraperitoneally (positive control group). Group C: Rats induced with 120 mg/kg bwt of CCl4 and then administered orally 100 mg/kg bwt of subfraction (F0) of ethylacetate fraction of T. bangwensis daily for 14 days. Group D: Rats induced with 120 mg/kg bwt of CCl4 and then administered orally 100 mg/kg bwt of subfraction (F1) of ethylacetate fraction of T. bangwensis daily for 14 days. Group E: Rats induced with 120 mg/kg bwt of CCl4 and then administered orally 100 mg/kg bwt of subfraction (F2) of ethylacetate fraction of T. bangwensis daily for 14 days. Group F: Rats induced with 120 mg/kg bwt of CCl4 and then administered orally 100 mg/kg bwt of subfraction (F3) of ethylacetate fraction of T. bangwensis daily for 14 days. Group G: Rats induced with 120 mg/kg bwt of CCl4 and then administered orally 100 mg/kg bwt of subfraction (F4) of ethylacetate fraction of T. bangwensis daily for 14 days. Group H: Rats induced with 120 mg/kg bwt of CCl4 and then administered orally 100 mg/kg bwt of subfraction (F5) of ethylacetate fraction of T. bangwensis daily for 14 days.
Blood collection and storage
After the 14th day of treatment with the subfractions of ethylacetate fraction of T. bangwensis, rats were fasted overnight for 12 h. They were anesthetized and blood collected through the jugular vein in the neck region. The blood samples were centrifuged at 3000 r/min for 5 min, supernatants (sera) collected using a Pasteur pipette and then stored at 4°C before use for the determination of liver function indices such as alanine aminotransferase (ALT), alkaline phosphatase (ALP), and aspartate aminotransferase (AST) activities. Total protein (TP), albumin (ALB), total bilirubin (T.Bil), and conjugated bilirubin (C.Bil) levels were also analyzed.
DPPH assay
The method of Burtis and Bucar 17 was adopted. A 0.5 mL of the extract (or subfraction) in methanol at different concentrations (100, 300, 400, and 600 µg/mL) was added to 2.0 mL of DPPH solution (4 mg DPPH/100 mL methanol). The mixture was incubated at 25°C for 30 min and centrifuged at 3000 r/min for 15 min. The supernatant was collected and absorbance read at 517 nm. The blank contained 2.0 mL of DPPH only
Liver tissue homogenate
The excised rat livers were washed in 1.15% KCl solution blotted and weighed. Then, 0.5 g of the liver was homogenized gently with 5.0 mL of 0.1 M phosphate buffer at pH 7.2 using ceramic mortar and pestle. The resulting homogenate was centrifuged at 2500 r/min for 15 min and the supernatant collected by Pasteur pipette and stored at −20°C for analyses of liver biomarkers of oxidative stress such as glutathione (GSH) and malondialdehyde (MDA) levels as well as superoxide dismutase (SOD) and catalase (CAT) activities, respectively.
Determination of GSH level
The GSH level was estimated using the method of Zerargul et al. 18 This method is based on the reduction of DTNB (Ellman’s reagent) by sulfhydryl group to form 2-nitro-5-mercaptobenzoic acid. A 50 µL of liver homogenate was diluted in 1.0 mL of 0.1 M phosphate buffer at pH 8.0; 20 µL of 0.01 M DTNB was mixed with 3.0 mL of the mixture of dilution. A yellow color developed and absorbance was read at 412 nm after 5 min. The GSH level was then determined by the formula as shown below
Determination of SOD activity
SOD activity was measured according to the method of Kakkar et al. 19 A 0.05 mL of the liver homogenate was mixed with 1.2 mL of 0.05 M sodium pyrophosphate buffer at pH 8.3, 0.1 mL of 0.186 mM methosulfate phenazine, 0.3 mL of 0.3 mM nitroblue tetrazolium chloride, and 0.2 mL of 0.78 mM reduced NADH. The reaction was stopped after 90 s by the addition of glacial acetic acid. The color intensity of the chromogen was extracted in 20 mL butanol with vigorous shaking. The mixture was then centrifuged at 3000 r/min for 10 min. The supernatant was collected and the absorbance read at 500 nm
Determination of CAT activity
CAT activity was measured by the method of Claiborne. 20 This method is based on the breakdown of H2O2 to water and oxygen by CAT. Briefly, the assay mixture contained 1.95 mL of 0.05 M phosphate buffer at pH 7.4, 1.0 mL of 19 mM H2O2, and 0.05 mL liver homogenate in a total volume of 3.0 mL. The absorbance was read at 240 nm and CAT activity expressed as
Determination of MDA level
The level of MDA was determined by the method of Okhawa et al. 21 This method is based on the interaction of MDA with two molecules of TBA in an acidic medium at 40°C to form a pink color. A 0.5 mL of the liver homogenate was mixed with 0.5 mL of 20% TCA and 1.0 mL of 0.67% TBA and the mixture boiled for 15 min. Then, 2.0 mL of n-butanol was added, centrifuged at 3000 r/min for 15 min and the supernatant collected and read at 532 nm. A standard curve of 1,1,3,3-tetraethoxypropane in different concentrations was plotted from where the MDA concentration was determined.
Biochemical assay of liver function indices
Serum AST and ALT activities were determined by the method of Schmidt and Schmidt, 22 while serum ALP activity was determined by the method of Babson et al. 23 Serum ALB level was determined using Bromocresol Green according to Doumas et al., 24 while serum T.Bil and C.Bil levels were determined by the method of Tietz. 25 Serum TP was estimated using the protocol of Tietz. 26
Liver histology
The method of Million et al. 27 was followed. After the rats were sacrificed, livers were excised and fixed in 10% formalin solution for 72 h, dehydrated through ascending alcohol gradient (50, 70, 90, and 100%), and cleared in two changes of xylene I and II and embedded in paraffin wax. Serial transverse section of 4–5 µm thickness was prepared using the Leica microtome (Leica Biosystems, USA) and then stained with hematoxylin and eosin. The samples were then viewed under light microscope with high resolution and observations recorded.
Elucidation of chemical compounds
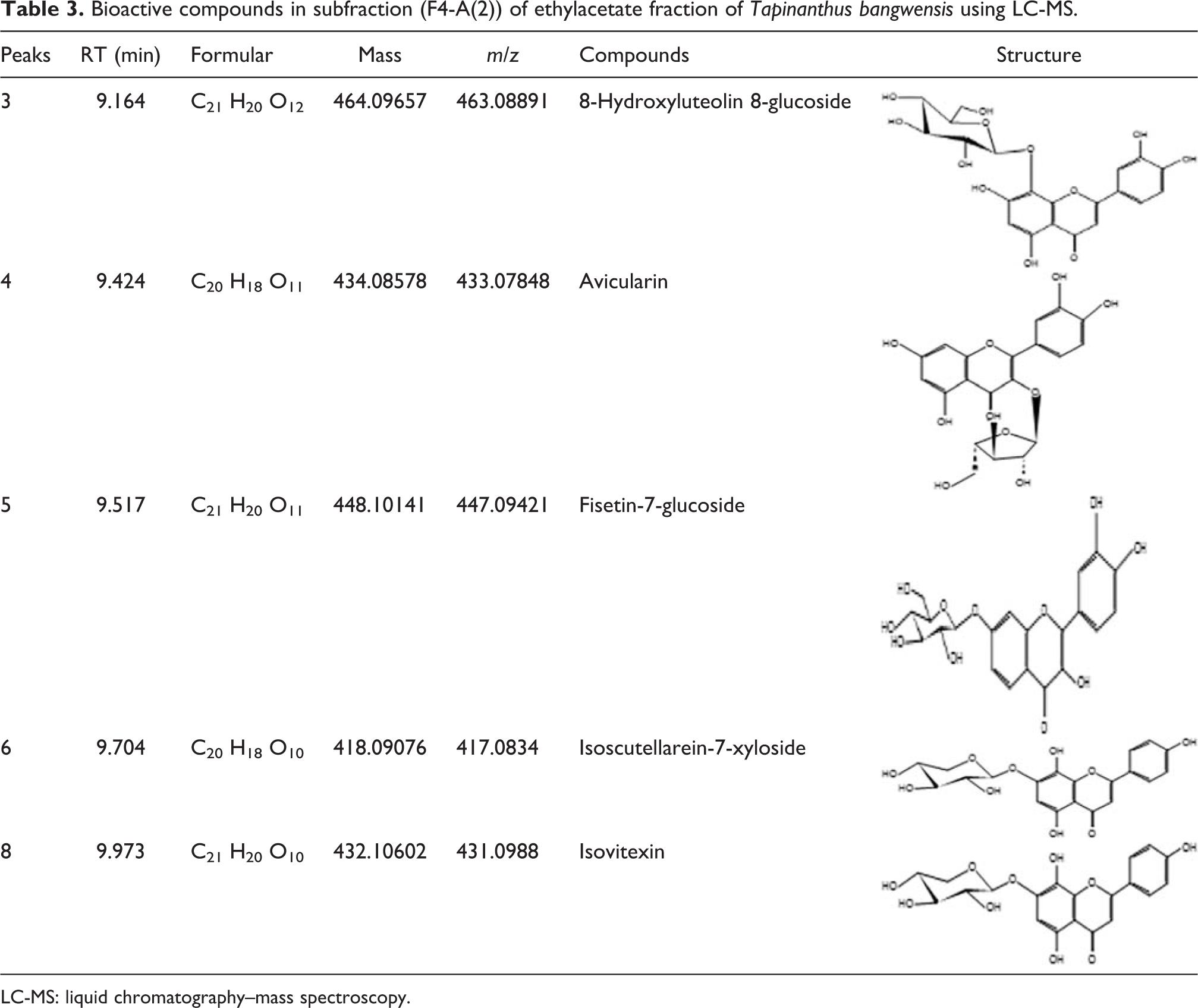
The chemical compounds present in subfraction (F4-A(2)) of ethylacetate fraction of T. bangwensis were elucidated by liquid chromatography–mass spectroscopy (LC-MS) technique. The instrument used was Agilent 1290 infinity liquid chromatography system coupled to Agilent 6520 (Agilent Technologies Inc., USA) Accurate mass Q-TOF mass spectrometer with dual ESI source. A 3.0 mL liquid sample of subfraction (F4-A(2)) was diluted with methanol in 1:1. Subsequently, 1.0 µL of the resulting solution was injected into the column and read against a methanol as blank.
Statistical analysis
The data obtained were statistically analyzed as mean ± standard deviation. One-way analysis of variance and Tukey multiple comparison test were used, respectively. The confidence limit was at p < 0.05.
Results
Table 1 shows the result of the recovery profile of the subfractions of ethylacetate fraction of T. bangwensis. A total of 105 column chromatographic fractions were obtained in 100 mL bottles. They were pooled together into six subfractions and labeled as F0, F1, F2, F3, F4, and F5, respectively. The subfraction F1 indicated the highest yield of 3.63 g (24.76%) with a retention factor of 0.3, while subfraction F5 showed the lowest yield of 1.46 g (9.96%) with a retention factor of 0.7. Figure 1 shows the result of the inhibition of oxidized DPPH free radical (DPP*). The inhibition concentration (IC50) result revealed that subfraction F0 exhibited the strongest antioxidant activity followed by subfraction F4, while subfraction F1 exhibited the lowest antioxidant activity. However, the antioxidant activity of ascorbic acid was higher compared to other subfractions. The effects of the subfractions of ethylacetate fraction of T. bangwensis on liver function indices of oxidative stress such as GSH and MDA levels as well as SOD and CAT activities were evaluated as shown in Figure 2. The GSH level increased in the induced and treated groups except in the group treated with subfractions F0 and F1 compared to the positive control group. However, subfraction F4 effect was higher compared to other induced and treated groups. SOD activity reduced in the positive control group but increased in the induced and treated groups except in the groups treated with subfractions F2 and F3. Conversely, subfraction F4 treated group improved SOD activity than in other induced and treated groups. There was no significant difference between the positive control group and the group induced and treated with subfractions at p < 0.05. CAT activity was found to be reduced in the positive control group. On treatment with 100 mg/kg bwt of the subfractions, CAT activity increased. However, the group treated with subfractions F1 and F3 improved CAT activity compared to other treated groups (Figure 2). There was no significant difference between the positive control group and the groups treated with subfractions F0, F3, F4, and F5 at p < 0.05. The MDA level increased in the subfraction F2, F3, and F5 treated groups compared to the positive control group, however, the group induced and treated with subfractions F0, F1, and F4 showed reduction in MDA level compared to the positive control group. However, there was no significant difference between the positive control group and the induced and treated groups at p < 0.05 (Figure 2). The liver function indices assayed were AST, ALT, and ALP activities as well as ALB, TP, T.Bil, and C.Bil levels (Table 2). The AST activity after treatment was found to be lower in the groups induced and treated with the subfractions compared to the positive control group. The ALT activity reduced in the groups induced and treated with the subfractions compared to the positive control group, nevertheless the effect was higher in the group induced and treated with subfraction F3. Furthermore, ALP activity was reduced in the groups induced and treated with subfractions F0 and F4, however, the ALP activity was higher in the other induced and treated groups compared to the positive control group. There were increase in the levels of serum ALB and TP in the induced and treated groups and normal control group except in the subfraction F1 and F2 treated groups compared to the positive control group, while the levels of T.Bil and C.Bil decreased in the induced and treated groups compared to the positive control group; however, there were no significant differences between the positive control group and the induced and treated groups in the liver function indices at p < 0.05. The histopathology study shows liver steatosis in the positive control group (plate B), but after 14 days of oral administration of subfractions F0, F1, F2, F3, F4, and F5, normal hepatocyte distributions and no fatty changes were observed as shown in plates C, D, E, F, G, and H, respectively, nonetheless, sinusoidal congestion was observed in plate G. The normal control group shows normal hepatocyte distribution, no fatty change, and no sinusoidal congestion, respectively (plate A) (Figure 3). The result of the DPPH bioguided assay used for screening the preparative-TLC (Prep-TLC) bands of subfraction (F4) of ethylacetate fraction of T. bangwensis for structural elucidation, as shown in Figure 4, shows that band A(2) exhibited stronger antioxidant activity compared to band B(1) though ascorbic acid exhibited the strongest antioxidant activity compared to the bands (Figure 4). Therefore, LC-MS analysis of subfraction (F4-A(2)) as shown in Table 3 reveals the presence of five chemical compounds such as 8-hydroxyluteolin-8-glucoside, Avicularin, Fisetin-7-glucoside, Isoscutellarein-7-xyloside, and Isovitexin, respectively.
Recovery profile of the subfractions of ethylacetate fraction of Tapinanthus bangwensis.
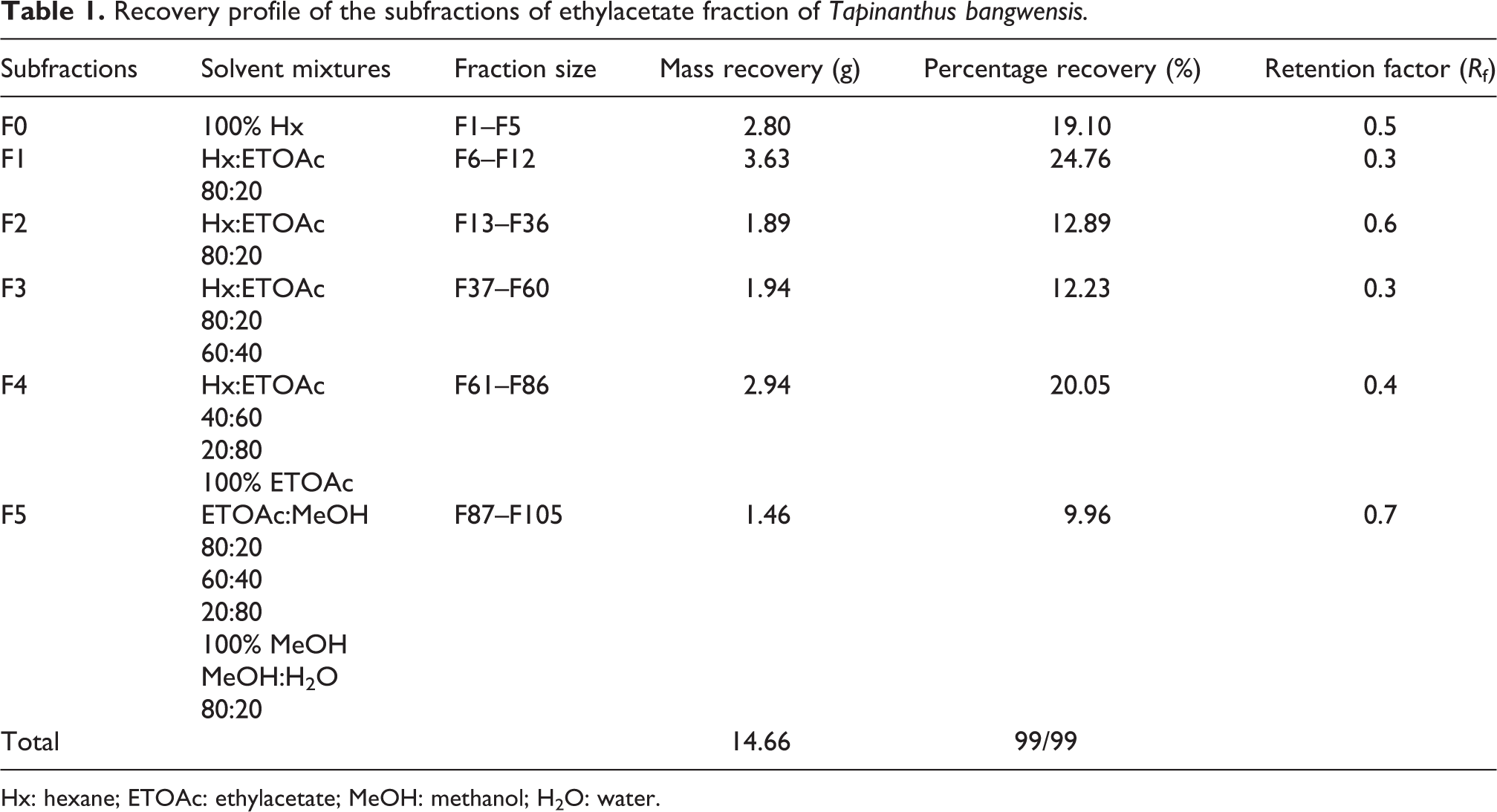
Hx: hexane; ETOAc: ethylacetate; MeOH: methanol; H2O: water.
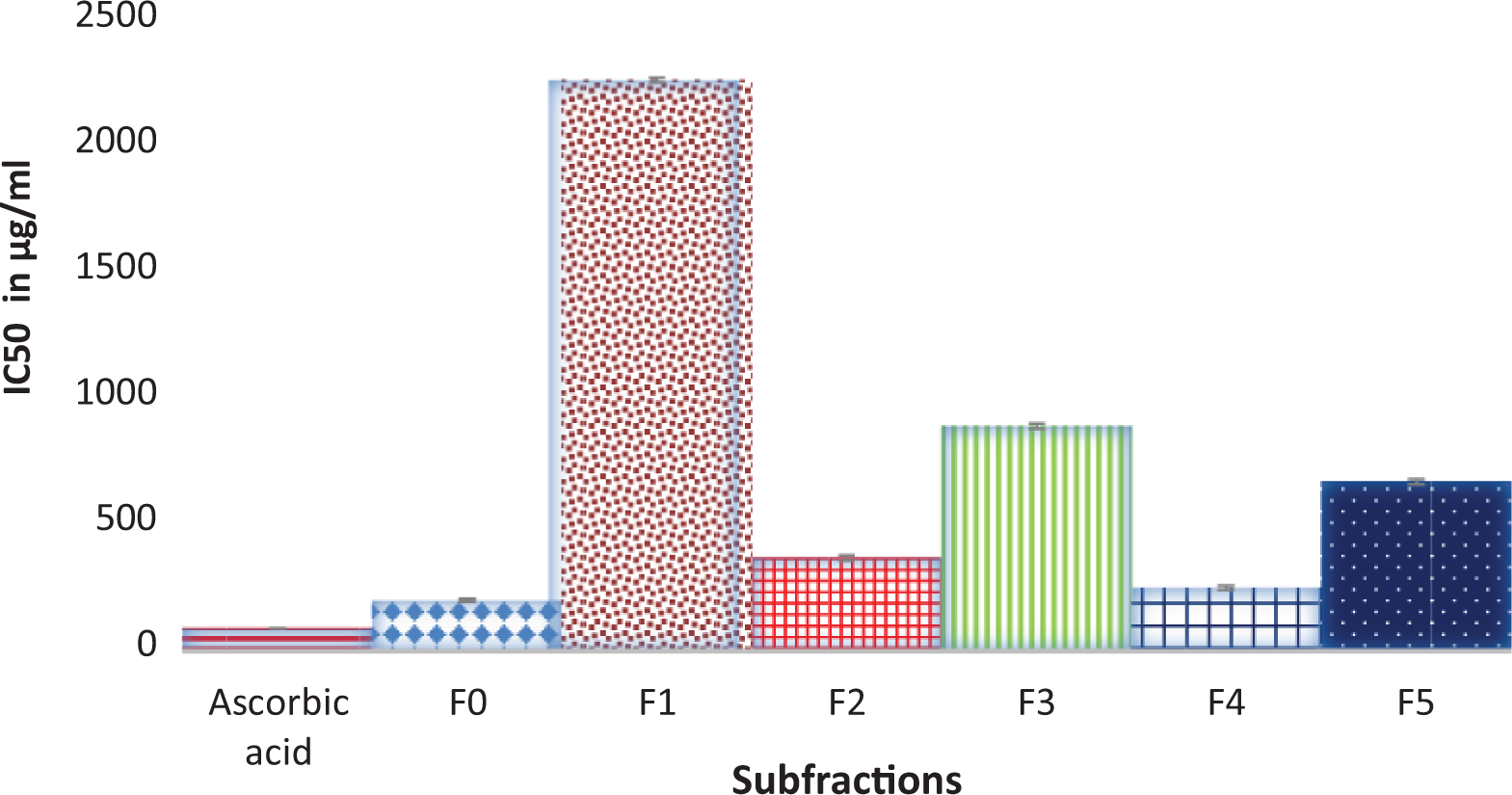
DPPH antioxidant activity of subfractions of ethylacetate fraction of Tapinanthus bangwensis. DPPH: 2,2-diphenyl-1-picrylhydrazine.
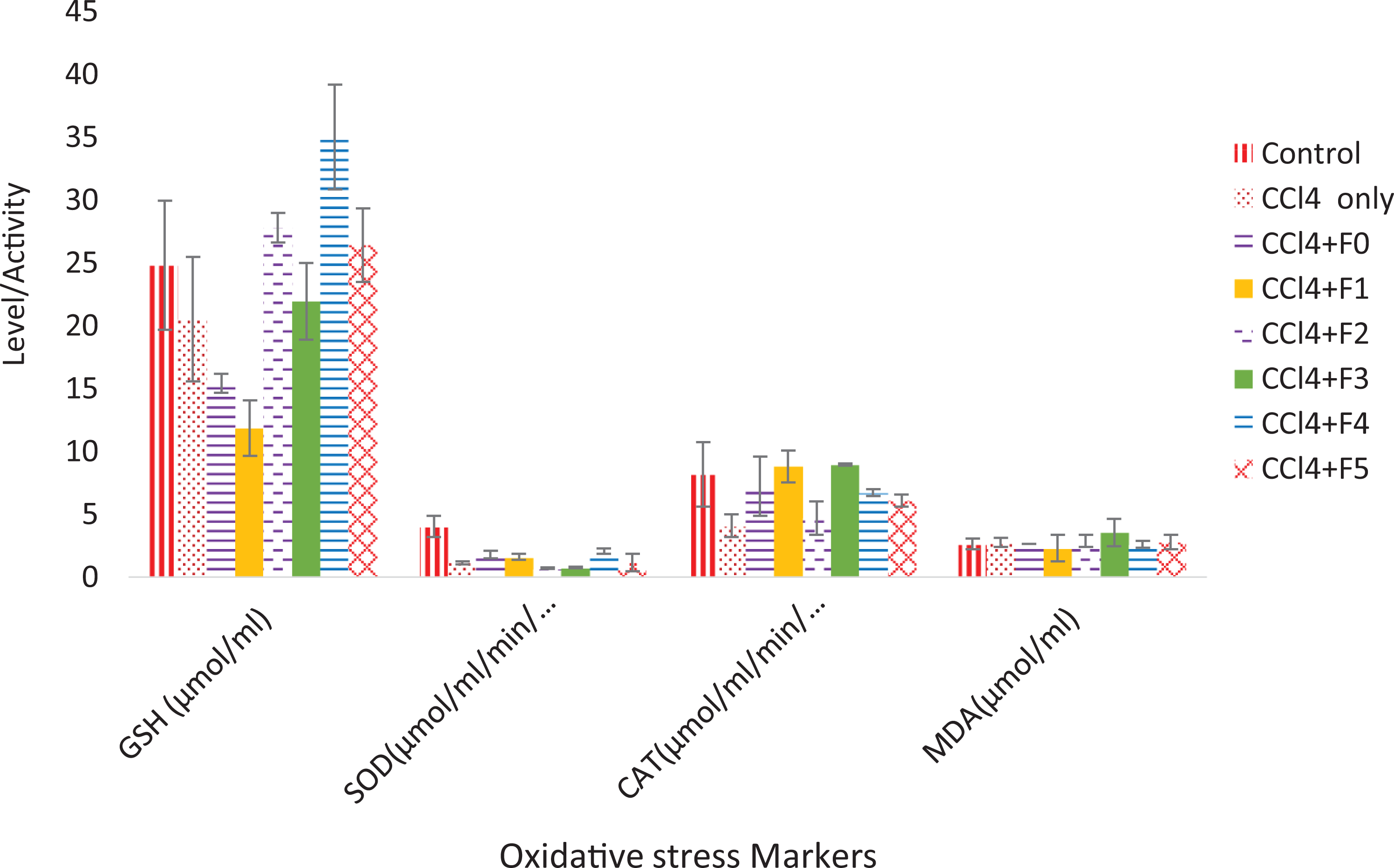
Effect of 100 mg/kg bwt of subfractions of ethylacetate fraction of Tapinanthus bangwensis on liver biomarkers of oxidative stress in CCl4-induced Wistar rats. CCl4: carbon tetrachloride; bwt: body weight.
Effect of 100 mg/kg bwt of subfractions of ethylacetate fraction of Tapinanthus bangwensis on serum liver function indices in CCl4-induced Wistar rats.
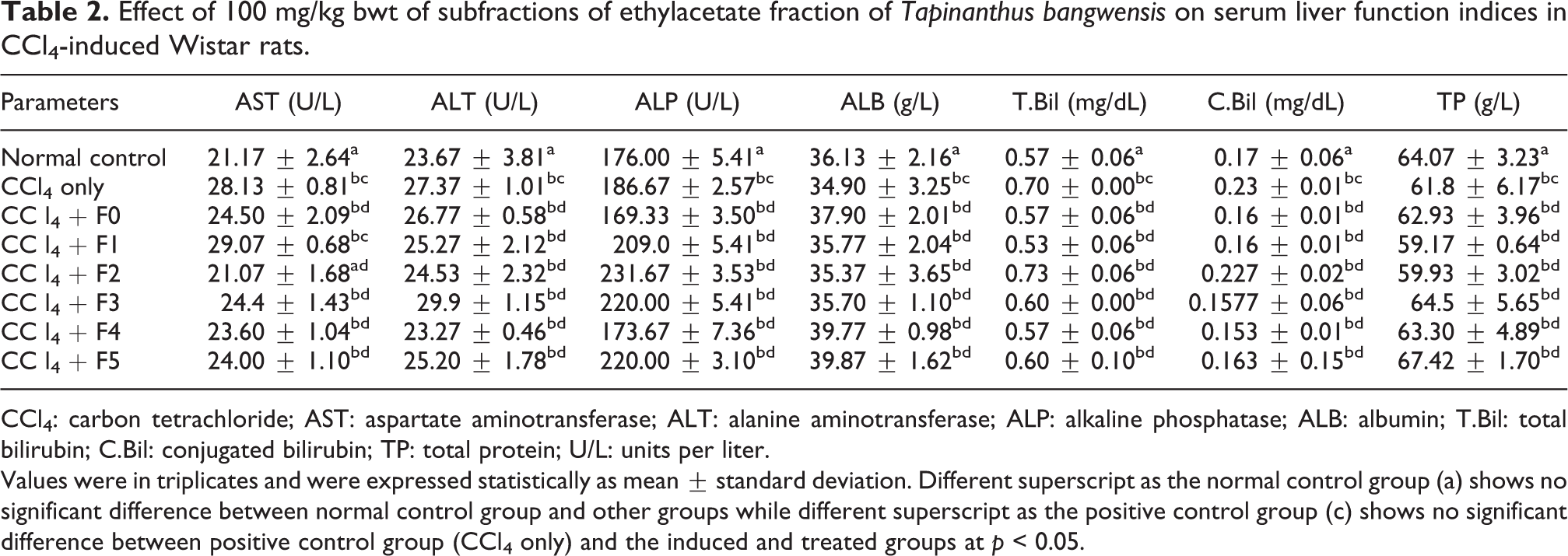
CCl4: carbon tetrachloride; AST: aspartate aminotransferase; ALT: alanine aminotransferase; ALP: alkaline phosphatase; ALB: albumin; T.Bil: total bilirubin; C.Bil: conjugated bilirubin; TP: total protein; U/L: units per liter.
Values were in triplicates and were expressed statistically as mean ± standard deviation. Different superscript as the normal control group (a) shows no significant difference between normal control group and other groups while different superscript as the positive control group (c) shows no significant difference between positive control group (CCl4 only) and the induced and treated groups at p < 0.05.
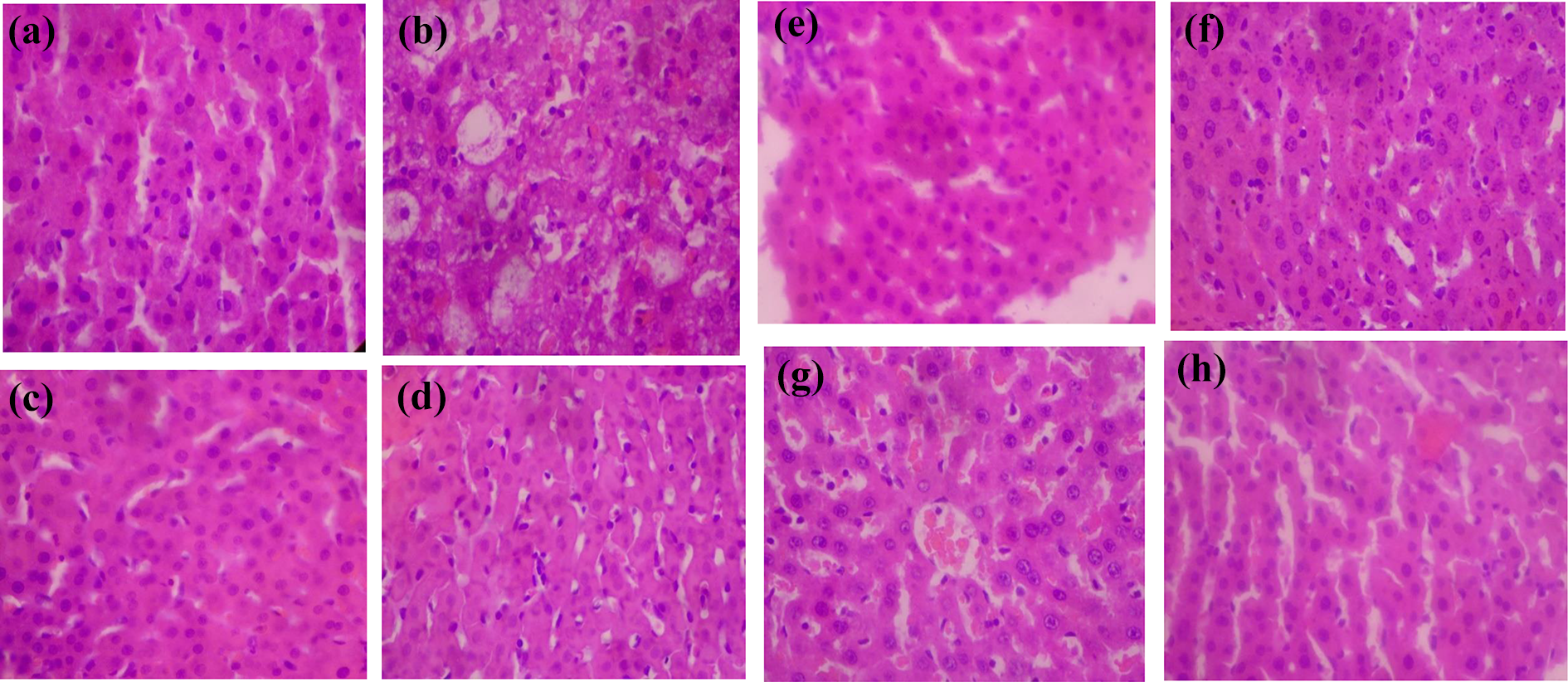
Histopathology study of the effect of oral administration of subfractions of ethylacetate fraction of Tapinanthus bangwensis in CCl4-induced Wistar rats (HE ×100). (a) Normal hepatocyte distribution and no fatty changes observed (normal control group), (b) fatty changes (or liver steatosis) observed (positive control group), and (c to h) normal hepatocyte distribution and no fatty changes observed in the induced and treated group with subfractions F0, F1, F2, F3, F4, and F5, respectively, of ethylacetate fraction of T. bangwensis. CCl4: carbon tetrachloride; HE: hematoxylin and eosin.
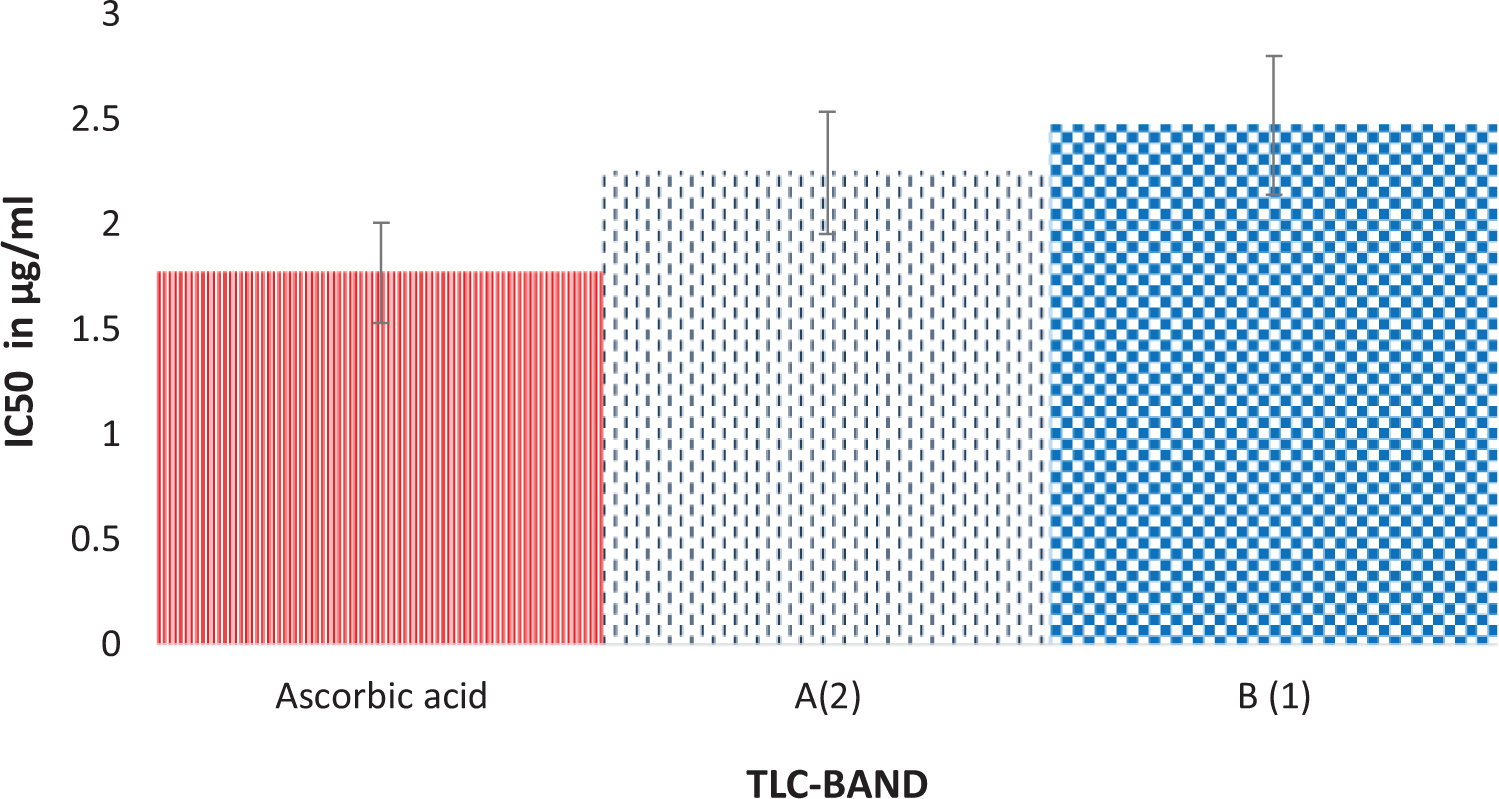
DPPH antioxidant activity of Prep-TLC bands of subfraction (F4) of ethylacetate fraction of Tapinanthus bangwensis. DPPH: 2,2-diphenyl-1-picrylhydrazine; Prep-TLC: preparative-thin layer chromatography.
Bioactive compounds in subfraction (F4-A(2)) of ethylacetate fraction of Tapinanthus bangwensis using LC-MS.
LC-MS: liquid chromatography–mass spectroscopy.
Discussion
CCl4 hepatotoxic effect is caused due to the release of carbon trichloromethyl free radicals (*CCl3) resulting from the metabolic activation of CCl4 by cytochrome P450 dependent mixed oxidase present in the liver endoplasmic reticulum. These free radicals attack lipoprotein-deoxyribonucleic acid membrane in the presence of oxygen forming trichloromethylperoxyl free radicals (*CCl3OO) which induce lipid peroxidation. 28,29 This metabolic action leads to liver damage, loss of metabolic enzyme activation, protein inhibition, and lipoprotein secretion. 30 The decrease observed in SOD and CAT activities in the positive control group may be due to accumulation of superoxide anion radicals and H2O2, respectively, in the system. 31 Furthermore, the decrease in GSH level could be due to the extrusion of GSH from the liver or increased utilization of GSH for free radical detoxification, while the increase in MDA level could be due to the attack by lipid peroxyl free radicals on biological membrane produced via lipid peroxidation. 32,33 These abnormalities in liver function indices of oxidative stress were ameliorated after oral administration of 100 mg/kg bwt of the different subfractions of ethylacetate fraction of T. bangwensis. The improvement in these liver parameters suggests that the plant possess chemical compounds that exhibit strong antioxidant activity against the effect of lipid peroxyl free radical activity. 34,35 Another attributable evidence of hepatocytic damage is the increase in the activities/levels of hepatic biomarkers (or liver function indices) found in the circulatory system. 36 The elevated serum enzyme activity indicates tissue perforation, hepatocellular leakage, and loss of functional integrity of liver’s plasmalemma and could be due to the stimulation of lipid peroxidation and depletion of antioxidant reservoirs. 37 –39 Furthermore, elevated levels of serum T.Bil and C.Bil indicate either decrease uptake and conjugation of bilirubin, decrease secretion by the liver or obstruction of the bile acids. The reduction in these serum liver biomarkers observed in this study indicates the antioxidative effect of T. bangwensis against lipid peroxyl free radical attack. 40 This result correlates with reported findings. 14,41 Some studies suggest that decrease in ALB and TP levels could be attributed to reduction in protein biosynthesis caused by disruption and dissociation of polyribosomes on rough endoplasmic reticulum. 10,42 This phenomenon was observed in the positive control group as shown in Table 2. However, oral treatment with the subfractions of ethylacetate fraction of T. bangwensis elevated ALB and TP levels and subsequently decreased AST, ALT, and ALP activities and T.Bil and C.Bil levels, respectively, compared to the positive control group. Oxidant substances (free radicals) generated from chemicals, drugs to mention a few cause oxidative stress. Free radicals are unstable substances as a result of the presence of unpaired electron(s), thus are called electron-deficient molecules. 43 This instability makes them to attack membrane macromolecules resulting in membrane disruption/damage. The DPPH assay which is a stable and widely used method for evaluating antioxidant power of medicinal plants 44 shows that T. bangwensis exhibits strong antioxidant activity and may be attributed to the antioxidant activities of Isoscutellarein-7-xyloside and Fisetin-7-glucoside as reported by research findings. 45,46 The liver fatty deposit observed in the positive control group indicates defective lipid metabolism and excretion thereby leading to impaired blood flow through the portal vein resulting in the onset of hepatic hypertension. 47 After oral treatment with the subfractions of ethylacetate fraction of T. bangwensis, ameliorative effects were observed. The LC-MS analysis of subfraction (F4-A(2)) of ethylacetate fraction of T. bangwensis reveals the presence of 8-hydroxyluteolin-8-glucoside, Avicularin, Fisetin-7-glucoside, Isoscutellarein-7-xyloside, and Isovitexin, respectively. These chemical compounds identified are phytochemical compounds of flavonoid derivatives in which a sugar moiety is attached at different positions of the flavonoid ring at either position 3, 7, or 8, respectively. The 8-hydroxyluteolin-8-glucoside (also called Hypolaetin-8-O-β-D-glucoside) is a flavonoid glycoside and has been reported to exhibit both antioxidant and hepatocurative activities. 48 Avicularin (also called Avicularine, Avicularoside, fenicularin, or Quercetin-3-α-L-arabinofuranoside) is also a flavonoid glycoside in which an α-L-arabinofuranosyl residue is attached at position 3 of quercetin via a glycosidic linkage and has been reported to possess both antioxidant and hepatocurative properties. 49 Furthermore, Fisetin-7-glucoside (also called 2-[3′,4′-dihydroxyphenyl]-3,7-dihydroxy-4H-chromen-4-one-7-α-L-glucoside) is a flavonol glycoside with a reported antioxidant activity 46 while Isoscutellarein-7-xyloside, a novel flavone glycoside compound in which xylose sugar residue is attached at position 7 of the flavonoid ring. It exhibits antioxidant property. 45 Finally, Isovitexin (also called homovitexin or saponaretin) which is a flavone glycoside phytochemical compound, rich in polyphenols has been reported to exhibit antioxidant and hepatocurative properties. 50
Conclusion
In lieu of the life-threatening effects of hepatotoxicity, huge financial cost associated with liver transplant, and adverse side effects of synthetic drugs, the present study validates that the antioxidant activity and hepatocurative potential of ethylacetate fraction of T. bangwensis is due to the presence of the chemical compounds mentioned above. Hence, it can be concluded that T. bangwensis has a promising medicinal potential against CCl4-induced hepatotoxicity in Wistar rats.
Footnotes
Acknowledgment
We acknowledge the technical support of Mr Maicah Chijoke, Department of Pharmacology, University of Lagos, and Gladys Ihegboro, Sandra Ihegboro, Moses Olua, Creda Esebamen, and Israel Ikuemelo, Department of Biochemistry and Forensic Science, Faculty of Sciences, Nigeria Police Academy, Wudil, Kano, Nigeria, for their inputs.
Author contributions
GOI, AJA, and MSS designed and supervised the work while GOI and CJO prepared the manuscript and analyzed the data obtained. All authors proofread the manuscript and made corrections where appropriate.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
The ethical approval for this research work was obtained from the ethics committee for the care and use of laboratory animals of Nigeria Police Academy, Wudil, Kano, Nigeria.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.