Abstract
Nicotinamide riboside (NR) is an analog of vitamin B3, which is naturally present in trace amounts in selected foods. Several B3 analogs are known precursors in the synthesis of nicotinamide adenine dinucleotide, including nicotinic acid (niacin) and nicotinamide (niacinamide). The safety of nicotinamide riboside hydrogen malate (NRHM), a salt of NR, was assessed in a bacterial reverse mutagenesis assay (Ames), in vitro micronucleus assay in human peripheral lymphocytes, a 14-day acute toxicology test (Sprague-Dawley rats), and a 90-day sub-chronic toxicology study in Sprague-Dawley rats. Based on the results of the 14-day study, the 90-day sub-chronic study was conducted comparing oral NRHM at 1000, 1500, and 3000 mg/kg/day with a vehicle control. No genotoxicity was observed. There were no mortalities or clinical observations attributed to the NRHM test substance administration over the course of the study. The no-observed-adverse effect level for NRHM was determined to be 3000 mg/kg bw/day in the female rats but 2000 mg/kg bw/day in the male rats due to a significantly lower body weight in males at the high dose. The results of this study imply that NRHM can be manufactured at a high level of purity, is not genotoxic, and is well-tolerated in in Sprague-Dawley rats.
Keywords
Introduction
Background Information
Nicotinamide riboside (NR) is an important precursor in the biosynthesis of nicotinamide adenine dinucleotide (NAD+) and the related NAD family of molecules. 1 Several B3 analogs are known precursors in the synthesis of nicotinamide adenine dinucleotide (NAD), including nicotinic acid (niacin) and nicotinamide (niacinamide) and due to the nicotinamide moiety, NR is considered a form of vitamin B3. 2 The nicotinamide moiety of NR is covalently linked to a riboside moiety and carries a positive charge at the nitrogen atom of the pyridine ring. NAD plays a variety of roles within the cell. However, NAD most notably acts as a hydride ion acceptor/donor and an energy modulator in several oxidation-reduction reactions of cellular energy metabolism, including glycolysis, the tricarboxylic acid cycle, and oxidative phosphorylation. 3 Primarily, NAD functions in the degradation of carbohydrates, fats, proteins, and alcohols, which are energy-producing, catabolic reactions. In contrast, NAD-phosphate (NADP) plays an anabolic role and serves as a redox agent in the synthesis of macromolecules such as fatty acids and cholesterol. 1 NAD+ also acts as a substrate for NAD+-dependent enzymes such as poly-adenosine diphosphate-ribose polymerases, sirtuins (SIRTs), and CD38 and CD157. 4
Several biosynthetic pathways exist for NAD, including the de novo biosynthesis pathway, which employs the amino acid, tryptophan, as its precursor; the Preiss-Handler pathway, in which NAD is derived from nicotinic acid; and the Salvage Pathway, which utilizes NR or nicotinamide. 5 NR is naturally present in cow’s milk and is also available as a nutraceutical that can be supplemented to increase NAD. 6 In humans, many clinical studies have been conducted to assess the safety and efficacy of NR chloride (NRCl) as a supplement.7-11 In addition, the labs of Marinescu and Conze have demonstrated the safety of NRCl through cytotoxicity and preclinical safety studies.
Previous toxicological studies of synthetic NRCl have demonstrated the relative safety of the NR compound and found that it showed no genotoxicity. In 2016, Conze et al. performed several toxicity studies including a 90-day repeat dose toxicity study, in which male and female Sprague-Dawley rats were given oral doses of NRCl at 300 mg/kg/day, 1000 mg/kg/day, and 3000 mg/kg/day. Assuming a human body weight of 60 kg, the upper intake level (UL) for NR was determined to be 180 mg/day or 3.0 mg/kg/day. These values were based off a no-observed-adverse effect level (NOAEL) of 300 mg/kg/day and a 100-fold safety factor. The lowest-observed adverse-effect-level was found to be 1000 mg/kg/day. At the 3000 mg/kg/day dosage, treatment-related effects were seen in the liver, kidneys, testes, epididymides, and ovaries, as demonstrated by clinical chemistry markers. 2
Recently, Marinescu et al. conducted a series of similar genotoxic studies of NR-E, a synthetic, nature-identical NR-Cl compound. The researchers performed a 90-day oral toxicity study, in which male and female Sprague Dawley rats were given doses of 300, 500, or 1200 mg/kg/day of NR-E. The researchers found that there were no mortality or clinical observations deemed attributable to NR-E at any of the dosages. The authors did note that a small statistically significant decrease in body weight was observed in the 1200 mg/kg/day NR-E-treated males when measured on day 92. This finding was consistent with previous reports of NR by Conze et al., who found an 8-9% overall decrease in body weight in the 300 and 1000 mg/kg/day males and a 17% reduction in body weight at the 3000 mg/kg/day dose in males. 2 No treatment related effects were seen at the NOAEL of 300 mg/kg/day, previously established by Conze et al. As such, Marinescu and their team adjusted the NOAEL of systemic toxicity for NR-E to be 500 mg/kg/day for males and 1200 mg/kg/day for females. The authors concluded, based on previous results and their own, that a UL of 300 mg/day for males and 720 mg/day in females was acceptable. 4
In the present study, a newly developed NR salt was assessed, which contains NR ionically bonded with malate. Based on the previous safety profiles of NRCl and the relative safety of malate, NR hydrogen malate (NRHM) was expected to be safe, non-toxic, and well-tolerated at 1500, 2000, and 3000 mg/kg/day in Sprague-Dawley rats. To analyze the safety profile of NRHM the present GLP compliant studies were conducted: a bacterial reverse mutation (Ames) assay, an in vitro micronucleus assay and, and a 90-day repeat dose oral sub-chronic toxicity study in male and female Sprague-Dawley rats. A non-GLP compliant 14-day oral range finding study in male and female Sprague Dawley rats was also conducted.
Materials and methods
Nicotinamide riboside hydrogen malate test material description
Nicotinamide riboside hydrogen malate is an ionic compound with the molecular formula C15H20N2O10 and molecular weight of 388.33 g/mol (see Figure 1 for molecular structure). It consists of 66% NR and 34% malate (by weight). Malate was chosen as the anionic moiety for NR because of superior manufacturing and solubility parameters when compared to other salt forms. In addition, malate was theorized to be of low toxicity and very-well tolerated because of its status as a metabolic intermediate and its significant prevalence in the food supply (e.g. apples). At 20°C, the solubility of the salt is 1.05 g/mL and, thus, is predicted to dissociated full in aqueous solution. pH does not exhibit undue behavior modification, in stability terms, on the disassociation of NRHM. During the preparation of the solutions of the test material, there were no issues with solubility, even at higher concentrations. Nicotinamide-beta-D-riboside hydrogen-L-malate of purity 98.00% (CAS No. 2415659-01-5) was supplied by Thorne Research, Inc (Summerville, SC, USA). Chemical structure of nicotinamide riboside hydrogen malate.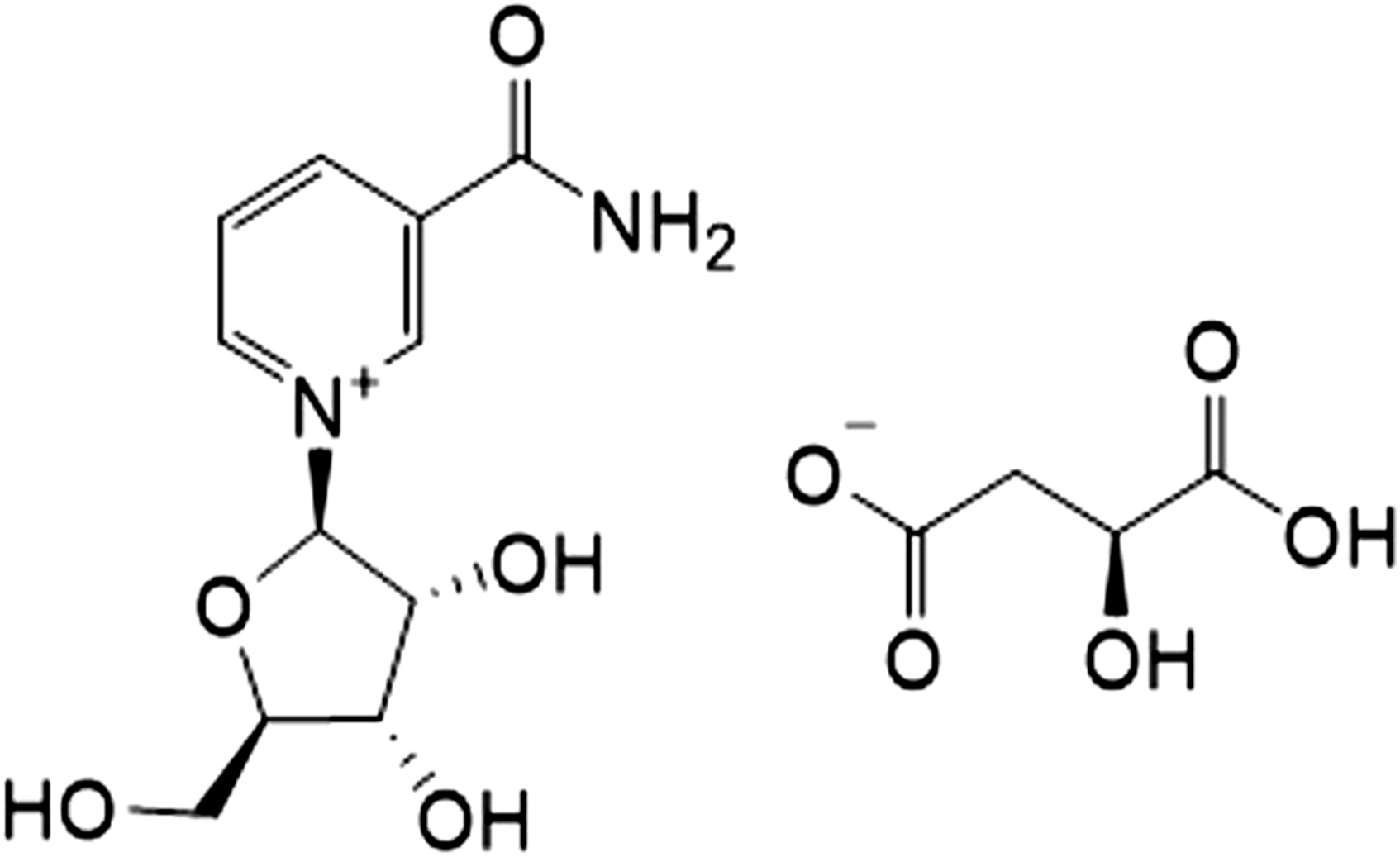
Bacterial reverse mutagenicity (ames assay)
The bacterial reverse mutation (Ames) assay was performed in compliance with the U.S. FDA Principles of Good Laboratory Practices (GLPs) and OECD Guideline No. 471. 12 All positive control articles, including sodium azide, ICR 191 acridine, daunomycin, methyl methanesulfonate, 2-aminoanthracene, S. typhimurium (TA1535, TA1537, TA98, TA100), and E. coli (WP2 uvrA) were purchased from Molecular Toxicology, Inc. (Boone, NC).
The mutagenicity of NRHM was assessed utilizing the plate incorporation method, in which 0, 0.0158, 0.05, 0.158, 0.5, 1.58, 5.0, 15.8, 50, 158, 500, 1580, and 5000 μg doses of NRHM were mixed with standard volumes of vehicle control, test article solution, or positive control. Samples were also mixed with 500 μL S9 mix (+S9) or substitution buffer (-S9), 100 μL bacterial preparation (S. typhimurium or E. coli) and 2.0 mL overlay agar maintained at approximately 45°C. The mixtures were poured over the surface of a minimal plate agar, inverted, and then incubated at approximately 37°C until growth was adequate for enumeration. Each test run was performed in triplicate.
Confirmatory assay
If there was no indication of mutagenicity in the initial test, verification via a second examination was not deemed necessary. However, negative, or equivocal results were confirmed with a repeat of the assay using modified conditions. The pre-incubation method of treatment was used for the confirmatory tests and employed the same study design as the main assay.
In the presence of metabolic activation (+S9), 2-aminoanthracene was used as the positive control for all strains. In the absence of metabolic activation (-S9), the positive controls for strains TA100 and TA1535, TA1537, TA98, and WP2 uvrA were sodium azide, ICR 191 acridine, daunomycin, and methyl methanesulfonate, respectively.
Toxic effects of NRHM are indicated by the partial or complete absence of background lawn of the non-relevant bacteria or a substantial dose-related reduction in revertant colony counts compared with lower dose levels and concurrent vehicle control taking into account the laboratory historical control range. Results were considered to be indicative of mutagenic potential if the results showed a substantial increase in revertant colony counts, i.e. a response Mutation Factor ≥2 for strains TA98, TA100, and WP2 uvrA or MF ≥3 for strains TA1535 and TA1537, with mean values outside the laboratory historical control range. Otherwise, results were considered negative. This increase must be dose related and/or reproducible.
In vitro micronucleus assay
The in vitro micronucleus assay was performed in compliance with U.S. FDA GLPs and OECD Guideline No. 487. 13 Human peripheral blood lymphocytes were obtained from healthy, non-smoking donors with no known recent exposure to genotoxic chemicals. Blood was collected from a single donor through venous puncture and collected in heparinized tubes. Mammalian S9 liver microsomal fraction homogenate was obtained from Trinova Biochem Gmb, Giessen, Germany.
A pre-experiment was conducted under identical conditions to the short exposure main experiment in order to determine the toxicity of the test material. The following concentrations were tested: 3.9, 7.8, 15.6, 31.3, 62.5, 125, 250, 500, 1000 and 2000 μg/mL (Experiment I - without S9 mix) and 4.1, 8.2 16.4, 32.8, 65.7, 131.3, 262.5, 525, 1050, 2100 μg/mL (Experiment II - with S9 mix). Following the recommendations of the corresponding OECD testing guidelines 487, the concentration of 2000 μg/mL was considered the highest test concentration to be used. Experiment I of the main experiment was a short-term (4h) exposure with and without metabolic activation at concentrations of 1000, 1500, and 2000 μg/mL. The lymphocytes were incubated with the test item for 4 h in presence or absence of metabolic activation. At the end of the incubation, the treatment medium was removed, and the cells were washed twice with phosphate buffered saline (PBS) + 10% fetal bovine serum (FBS). Subsequently the cells were incubated in complete culture medium + 6 μg/mL cytochalasin B for 40 h to 42 h at 37°C and 5% CO2. 14
Experiment II was a long-term exposure (44 h) without metabolic activation at concentrations of 1000, 1500, and 2000 μg/mL. The whole blood cultures were pre-cultured in the presence of phytohemagglutinin (PHA) for 44 to 48 h prior to exposure to the test item. Then the test item was added in complete culture medium. 1 h later 6 μg/mL cytochalasin B were added and the cells were incubated for further 43 h at 37°C. At the end of the treatment, the cell culture medium was removed, and the cells were prepared for microscopic analysis.
Subsequently, the cells were treated with cold hypotonic solution (0.075 M KCl) for some minutes at room temperature and immediately centrifuged. The pellet was resuspended with a solution consisted of fixation solution + NaCl 0.9% (1+1) and centrifuged. After that the cells were fixed with methanol + glacial acetic acid (3+1). The cells were gently resuspended, and the suspension was dropped onto clean glass slides. Consecutively, the cells were dried on a heating plate. The cells were stained with acridine orange solution.
All slides, including those of positive and negative controls were independently coded before microscopic analysis. For each dose group at least 2000 binucleated cells (if possible) per concentration (1000 binucleated cells per slide) were analyzed for micronuclei according to the criteria of Fenech, i.e. clearly surrounded by a nuclear membrane, having an area of less than one-third of that of the main nucleus, being located within the cytoplasm of the cell and not linked to the main nucleus via nucleoplasmic bridges. 15 Mononucleated and multinucleated cells and cells with more than six micronuclei were not considered. 16
14-Day range finding study
A 14-day repeat dose study was conducted in 40 male and female Sprague-Dawley rats in accordance with the OCED Guidelines for testing of Chemicals and Food Ingredients, Section 4 (Part 407). However, the Guidelines were applied to a 14-day study instead of a 28-day study. The rats were supplied by Charles Rivers Laboratories, Inc. with the 2016 certified Envigo Teklad Global Rodent Diet® (from Envigo Teklad, Inc.) that was used during the study. Nicotinamide-beta-D-riboside hydrogen-L-malate 98.00% (CAS No. 2415659-01-5) was supplied by Thorne Research, Inc. (Summerville, SC, USA).
Animals were housed in a controlled environment according to the latest Guide for the Care and Use of Laboratory Animals. 17 The animals were acclimated to the housing conditions for at least 5 days prior to testing. Body weights and clinical observations were recorded at least two times prior to study start. Both feed and water were provided ad libitum throughout the duration of the study.
For dose selection, a human dose was chosen based on our consumption target. This dose was then converted to a rat dose using conversion tables and a standard 100 times safety factor was applied. This was the minimum doses in rats. Given this minimum dosing number, a dosing schedule was chosen that was several times higher than the minimum dose and was graduated so that our lowest dose in the dose ranging study would cover our objective dose, at minimum, in the 90-day toxicology study. Using this process, we expected to get our desired NOAEL in humans. NRHM was mixed in distilled water and administered daily by oral gavage. Fresh formulations containing 100 (low dose), 250 (intermediate dose), and 500 (high dose) mg/mL of NRHM were prepared daily for a dosing volume of 10 mL/kg bw/day. These formulations correspond to 1000, 2500, and 5000 mg/kg bw/day of NRHM, respectively. Formulations were sonicated and stirred at ambient temperature until the mixtures were visually homogeneous. Stability analysis was performed by the PSL analytical lab using a validated HPLC method specific to NRHM and a NRHM standard.
All animals were observed at least twice daily for viability. Cage-side observations of all animals were performed daily during the study with all findings being recorded. Detailed clinical examinations were conducted on Day 1, prior to treatment with NRHM, and approximately weekly thereafter (∼days 1, 8, 15). Examinations include assessment of skin, fur, eyes, and mucous membranes, occurrence of secretions and excretions and autonomic activity (e.g. lacrimation, piloerection, pupil size, unusual respiratory pattern). Likewise, changes in gait, posture and response to handling as well as the presence of clonic or tonic movements, stereotypies (e.g. excessive grooming, repetitive circling), or bizarre behavior (e.g. self-mutilation, walking backwards) were also recorded when necessary. Body weight and food consumption measurements were also taken on these days. Body weight gain was calculated for selected individuals and for the study overall. Individual food consumption will be measured and recorded to coincide with body weight measurements. All animals were euthanized and examined for gross pathological changes including examination of the external surface of the body, all orifices, musculoskeletal system, and the cranial, thoracic, abdominal, and pelvic cavities with their associated organs and tissues. The results of this study were utilized to refine the doses for the 90-day study.
Statistical analysis
Product Safety Labs performed statistical analysis of all data collected during the in-life phase of the study as well as clinical pathology and organ weight data. Significance was judged at a probability value of p < 0.05. Male and female rats were evaluated separately. For all in-life endpoints that are identified as multiple measurements of continuous data over time (e.g. body weight parameters, food consumption, and food efficacy), treatment and control groups were compared using a two-way analysis of variance (ANOVA), testing the effects of both time and treatment, with methods accounting for repeated measures in one independent variable (time; Motulsky, 2014). Significant interactions observed between treatment and time as well as main effects were further analyzed by a post hoc multiple comparisons test (e.g. Dunnett’s test; Dunnett, 1964 and 1980) of the individual treated groups to control.
90-day subchronic toxicity study
A 90-day oral subchronic toxicity study was conducted in 80 male and female Sprague-Dawley rats (n = 10/sex/group) in accordance with the US FDA GLP: 21 CFR Part 58, 198 and the OECD Guidelines for Testing of Chemicals, Section 4 (No. 408): Health Effects, Repeated Dose 90-Day Oral Toxicity Study in Rodents (2018). The study was performed at Product Safety Labs (Dayton, NJ, USA), which is AAALAC accredited and certified in the appropriate care of all live experimental animals. The rats were supplied by Charles Rivers Laboratories, Inc. with the 2016 certified Envigo Teklad Global Rodent Diet® (from Envigo Teklad, Inc.) that was used during the study. Nicotinamide-beta-D-riboside hydrogen-L-malate 98.00% (CAS No. 2415659-01-5) was supplied by Thorne Research, Inc. (Summerville, SC, USA).
Animals were group housed, with no more than two animals per cage, in a controlled environment according to the latest Guide for the Care and Use of Laboratory Animals. 17 The animals were acclimated to the housing conditions for 6 days prior to testing. Body weights and clinical observations were recorded at least two times prior to study start.
Water was provided ad libitum, and with the exception of an overnight fast prior to being euthanized, feed was provided ad libitum throughout acclimation and the duration of the study. NRHM was mixed in distilled water and administered by oral gavage. Fresh formulations containing 150 (low dose), 200 (intermediate dose), and 300 (high dose) mg/mL of NRHM were prepared daily. These formulations correspond to 1500, 2000, and 3000 mg/kg bw/day of NRHM, respectively. The formulations were stirred at ambient temperature until a visually homogenous mixture was achieved.
Based on the results of a previous method validation study, homogeneity, and stability results indicated that the NRHM test substance was considered to have been uniformly distributed and stable at all concentrations as mixed in the vehicle.
All animals were observed at least twice daily for viability along with cage side observations of all animals daily. Detailed observations were conducted on Day 1 and approximately weekly thereafter for the duration of the study. Examinations include assessment of skin, fur, eyes, and mucous membranes, occurrence of secretions and excretions and autonomic activity (e.g. lacrimation, piloerection, pupil size, unusual respiratory pattern). Likewise, changes in gait, posture and response to handling as well as the presence of clonic or tonic movements, stereotypies (e.g. excessive grooming, repetitive circling), or bizarre behavior (e.g. self-mutilation, walking backwards) were also be recorded when necessary. Body weight and food consumption measurements were also taken on these days. The animals were also weighed prior to sacrifice in order to calculate organ to body weight ratios. Body weight gain was calculated for selected intervals and for the study overall. A final fasted body weight was collected prior to sacrifice. Ophthalmologic evaluations were performed during the acclimation period. A functional observational battery (FOB) was conducted once on all animals in the study during week 13. Motor activity was also evaluated on all animals during week 13 using an automated system for a single one-hour session. Urine and blood samples were collected following placement in metabolism cages and an overnight fast. Animals were then euthanized by exsanguination followed by a full necropsy. Urine was analyzed for bilirubin, blood, color, clarity, glucose, ketone, microscopic urine sediment, pH, total protein, quality, specific gravity, volume, and urobilinogen.
Clinical pathology was assessed on blood samples collected from the sublingual vein and included hematology, coagulation, and clinical chemistry parameters. Hematological parameters included hematocrit, hemoglobin concentration, mean corpuscular hemoglobin, mean corpuscular volume, platelet count, red blood cell count, red cell distribution width, reticulocyte count, white blood cell count, and differential leukocyte count. Mean corpuscular hemoglobin concentration was calculated.
Blood samples were analyzed for clinical chemistry measures including albumin, alkaline phosphatase (ALP), bilirubin, blood creatinine, total cholesterol (TC), fasting glucose, globulin, high-density lipoprotein (HDL), low-density lipoprotein (LDL), serum alanine aminotransferase (ALT), serum aspartate aminotransferase (AST), total serum protein, sorbitol dehydrogenase, triglycerides, blood urea nitrogen (BUN), and electrolytes (i.e. calcium, chloride, inorganic phosphorus, potassium, and sodium).
Organs that were collected and weighed wet, immediately following dissection, including adrenals, brain, epididymides, heart, kidneys, liver, ovaries with oviducts, spleen, testes, thymus, and uterus. Additional tissues weighed in the control and high dose animals included the inguinal fat pad, retroperitoneal fat pad, left gastrocnemius muscle and the left soleus muscle. The following organs and tissues were collected and preserved in 10% neutral buffered formalin: accessory genital organs (prostate and seminal vesicles), adrenals, all gross lesions, aorta, bone (femur), bone marrow (from femur and sternum), brain (medulla/pons, cerebellar, and cerebral cortex), cecum, cervix, colon, duodenum, esophagus, Harderian gland, heart, ileum with Peyer’s patches, jejunum, kidneys, larynx, liver, lungs, lymph node mandibular, lymph node mesenteric, mammary gland, nasal turbinates, nose, ovaries oviducts, pancreas (with islets), parathyroid, peripheral nerve (sciatic), pharynx, pituitary gland, rectum, salivary glands (sublingual, submandibular, and parotid), skeletal muscle, skin, spinal cord (3 levels: cervical, mid-thoracic, and lumbar), spleen sternum, stomach, thymus, thyroid, trachea, urinary bladder, uterus, and vagina. The left tibia was collected from all animals in the control and high dose groups for measurement of length and width and were then discarded.
Organs and tissues from the control and the high dose groups, were subject to histopathological examination. Based on findings in the high dose animals, the stomach, testes, kidney, spleen and thymus were also evaluated from the low and mid dose groups. The fixed tissues were trimmed, processed, and embedded in paraffin, sectioned with a microtome, placed on glass microscope slides, stained with hematoxylin and eosin, and examined by light microscopy. Slide preparation and histological assessment, by a board-certified veterinary pathologist, were performed at StageBio (Mount Jackson, VA).
Statistical analysis
Significance was judged at a probability value of p < 0.05. Mean and standard deviations were calculated for all quantitative data. Statistical analyses included a two-way analysis of variance (ANOVA) a post hoc multiple comparisons test (e.g. Dunnett’s test),18,19 homogeneity of variances 20 and normality, 21 a non-parametric analysis of variance, 22 and Dunn’s test to compare treated versus control groups when non-parametric analysis of variance was significant. 23
Results
Bacterial reverse mutagenicity (ames assay)
Nicotinamide riboside hydrogen malate was not genotoxic at any doses demonstrated by the mean revertant colony counts for each strain treated with the vehicle being close to or within the expected range, considering published values.24,25 The positive control substances (sodium azide, acridine, daunomycin, methyl methanesulfonate, and 2-aminoanthracene) caused the expected substantial increases in revertant colony counts in both the absence and presence of S9 mix. Thus, confirming the sensitivity of the test and the activity of the S9 mix.
For all strains, there was no concentration related or substantial test substance related increases in the number of revertant colonies using either the plate incorporation or the pre-incubation method (Supplementary Table 1). Therefore, NRHM did not elicit evidence of bacterial mutagenicity in the Ames assay.
In vitro micronucleus assay
Nicotinamide riboside hydrogen malate was evaluated for cytotoxicity using human peripheral blood lymphocytes via the in vitro micronucleus assay. In experiment I without and with metabolic activation, no increase of the cytostasis above the established threshold of 30%. In addition, no biologically relevant increase of the micronucleus frequency was noted in experiment I when treated with NRHM. These results were verified using the nonparametric χ2 Test, confirming that no statistically significant enhancement (p < 0.05) of the cells with micronuclei occurred in the dose groups of NRHM in experiment I.
In experiment II, without metabolic activation, an increase of the cytostasis above 30% was noted at a concentration of 2000 μg/mL. Without metabolic activation, in experiment II, an increase of micronucleus frequency was noted in the lowest concentration (1000 μg/mL, p = 0.0406). However, this increase was not outside the historical control limits of the negative control and was not dose dependent. Higher concentrations tested showed no increase compared to the negative control. In addition, the effect observed with the lowest concentration was deemed as not biologically relevant.
The χ2 (Chi Squared) Test for trend was performed to test whether there was a concentration-related increase in the micronucleated cells frequency in the experimental conditions. No statistically significant increase in the frequency of micronucleated cells under the experimental conditions of the study was observed in experiment I and II (Supplementary table 2).
Methylmethanesulfonate (MMS, 25 μg/mL and 65 μg/mL) and cyclophosphamide (CPA, 15 μg/mL) were used as clastogenic controls. Colchicine (Colc, 0.02 μg/mL and 0.4 μg/mL) was used as aneugenic control. All induced distinct and statistically significant increases of micronucleus frequency. This demonstrates the validity of the assay.
14-day toxicology study
There were no mortalities or clinical observations attributed to the oral administration of NRHM. Statistically significant body weight parameters were observed in Group 3 (2500 mg/kg/day) and Group 4 (5000 mg/kg/day) male rats, and to a lesser extent in Group 3 and 4 female rats. The observed decreases in body weight were a 9 and 16% decrease from control values for Group 3 and 4 males, as well as 4 and 7% decreases from control in female rats, respectively (Supplementary Table 3). Body weight decreases were not accompanied by decreases in food consumption or any other in-life endpoint.
Mean weekly and daily body weight gains for male rats in Groups 2-4 were generally comparable to the control Group 1 throughout the study. However, statistically significant weekly decreases (p < 0.001–0.01) were observed on Days 8-15 in Group 4 and statistically significant daily lower wight gain (p < 0.001–0.05) was observed at both intervals between Days 1-15 in Groups 3 and 4 male rats. Mean weekly body and daily body weight gains for female rats in Groups 2-4 were comparable to control Group 1 animals throughout the study (Supplementary Table 4). There were no significant changes in food consumption in male or female rats administered NRHM (Supplementary Table 5).
Increases in hematocrit (11%) and hemoglobin (13%) were served in Group 3 and 4 males and were observed outside the historical control data for this age and strain of rat. Red blood cell count was significant in Group 4 male rats only but within historical control ranges. Mean platelet count for male and female rats were decreased in all groups, significantly in Group 2 and 3 males, and 3 and 4 females, as compared to Group 1 controls. These changes were between 14-20% of control values for the same groups. Mean platelet counts for Group 2 and 3 males as well as Group 3 and 4 females were within historical control values. Hematology data is presented in Supplementary Table 6. Statistically significant increases in Group 4 male alanine aminotransferase (72% from control) and aspartate aminotransferase (32% from control) were noted and fell within historical control levels. Bilirubin levels in Group 3 and 4 females was significantly increased from control values (92% and 104%, respectively. These bilirubin values were within historical control values. All other noted changes in clinical chemistry values were within historical control values, did not occur in a dose-dependent manner, and were considered to be of no toxicological significance (See Supplementary Table 7).
90-day subchronic toxicity study
Mortality, body weight, and food consumption
There were no mortalities or clinical observations attributed to NRHM test substance administration over the course of the study. All observations throughout the study were considered to be incidental and/or present across all groups, including control, and not considered to be toxicologically significant.
Statistically significant differences in mean absolute body weight were observed in Groups 3 and 4, and to a lesser extent Group 2 male rats, compared to the Control Group (Figure 2). Statistically significant decreases in the rate and degree of body weight gain were also reported in all NRHM treated females, as compared to the concurrent control group (Figure 2). Some statistically significant changes in mean daily body weight gain were reported in the NRHM treated animals (Supplementary Table 8). Trajectory of male and female bodyweight between dosing groups.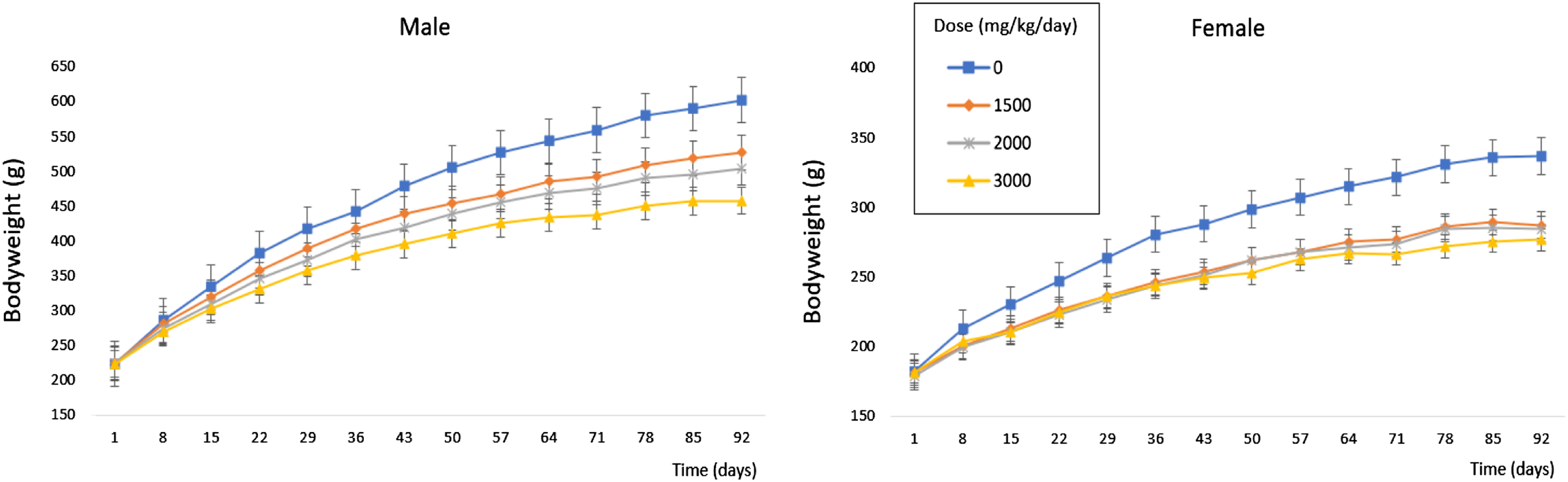
Decreases in the mean daily food consumption were reported in the males and females, as compared to the concurrent controls (Supplementary Table 9). Some decreases reached statistical significance and were considered to be test material related and a contributing factor to the decreased body weights, however the decreases were not considered to be adverse.
Hematological, clinical chemistry, and urinalysis tests
Change in hematological parameters.
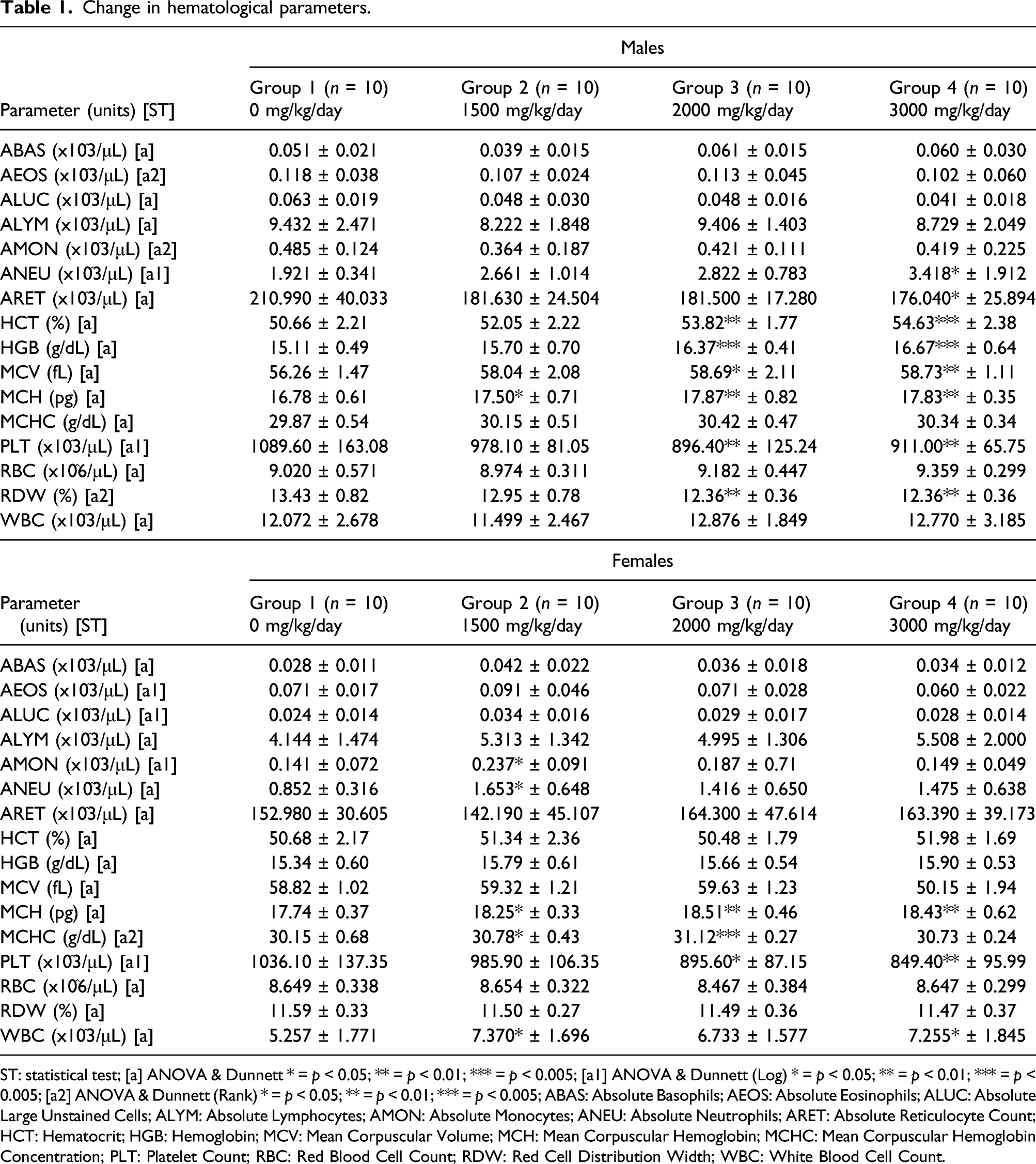
ST: statistical test; [a] ANOVA & Dunnett * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a1] ANOVA & Dunnett (Log) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a2] ANOVA & Dunnett (Rank) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; ABAS: Absolute Basophils; AEOS: Absolute Eosinophils; ALUC: Absolute Large Unstained Cells; ALYM: Absolute Lymphocytes; AMON: Absolute Monocytes; ANEU: Absolute Neutrophils; ARET: Absolute Reticulocyte Count; HCT: Hematocrit; HGB: Hemoglobin; MCV: Mean Corpuscular Volume; MCH: Mean Corpuscular Hemoglobin; MCHC: Mean Corpuscular Hemoglobin Concentration; PLT: Platelet Count; RBC: Red Blood Cell Count; RDW: Red Cell Distribution Width; WBC: White Blood Cell Count.
Changes in clinical chemistry parameters.
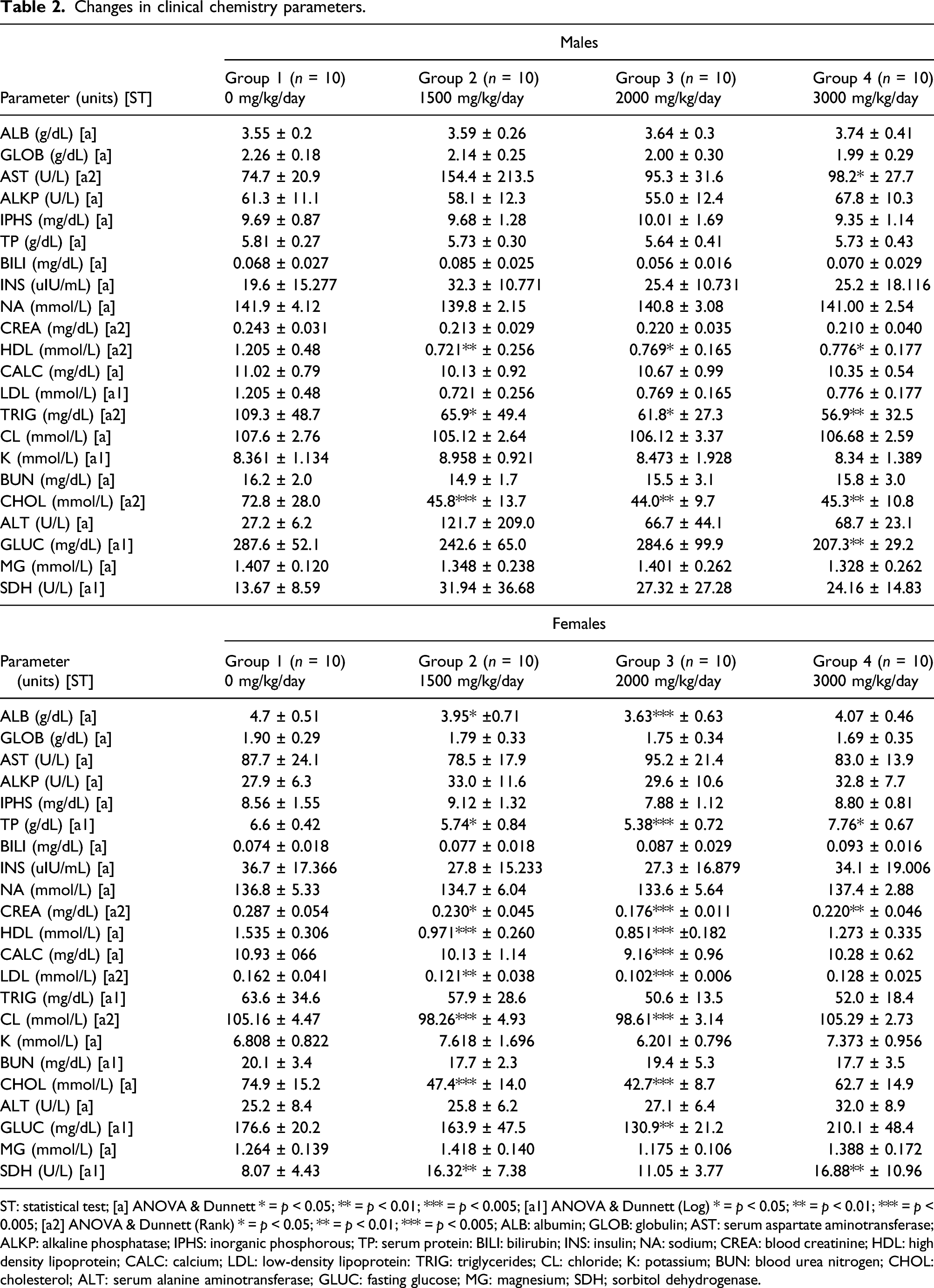
ST: statistical test; [a] ANOVA & Dunnett * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a1] ANOVA & Dunnett (Log) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a2] ANOVA & Dunnett (Rank) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; ALB: albumin; GLOB: globulin; AST: serum aspartate aminotransferase; ALKP: alkaline phosphatase; IPHS: inorganic phosphorous; TP: serum protein: BILI: bilirubin; INS: insulin; NA: sodium; CREA: blood creatinine; HDL: high density lipoprotein; CALC: calcium; LDL: low-density lipoprotein: TRIG: triglycerides; CL: chloride; K: potassium; BUN: blood urea nitrogen; CHOL: cholesterol; ALT: serum alanine aminotransferase; GLUC: fasting glucose; MG: magnesium; SDH; sorbitol dehydrogenase.
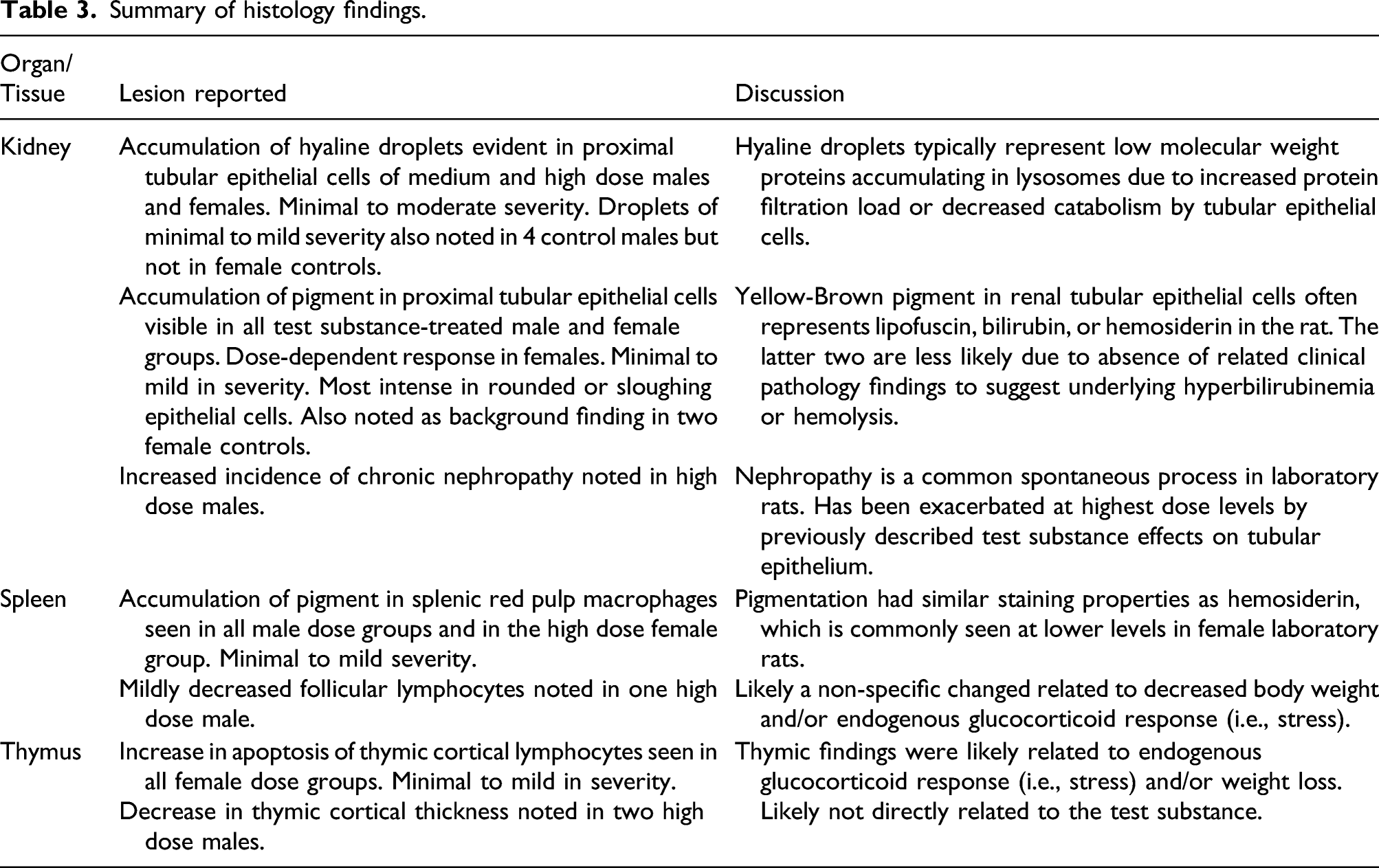
Summary of histology findings.
Decreased platelet count at 2000 and 3000 mg/kg/day were seen in both males and females, as well as decreased reticulocyte in the male 3000 mg/kg/day group. However, these were considered to be incidental changes, as the values were within historical control ranges and were not associated with any microscopic changes in hematopoietic organs. All other changes in hematological parameters were considered unrelated to the test substance and due to biological variance.
Increased ALT activity in the 3000 mg/kg/day males was considered a test substance-related non-adverse change. Increased ALT in the 1500 and 2000 mg/kg/day male groups was considered to be incidental. The presence of outliers, as indicated by the high standard deviations, led to higher mean values. Decreased triglycerides, total cholesterol, and HDL cholesterol in all male group resulted from reduced food consumption and body weight reduction and was not considered to be adverse. All other clinical chemistry parameters were considered to be unrelated to the test substance and are likely due to biological variability.
All changes in urinalysis parameters were considered to be unrelated to the test substance due to biological variance among rats as the magnitude of variation was minimal.
Ophthalmologic, FOB, and motor activity
All animals in the study were normal upon ophthalmic examination. Therefore, NRHM in not considered an ocular toxicant. There were no adverse FOB findings attributed to NRHM. Mean quantitative measurements of grip strength and foot splay for the treated male and female rates were comparable to the control group values.
There were no statistically significant changes in overall motor activity in any of the treatment groups, either male or female, that were considered to be adverse or attributable to NRHM administration. Significant decreases in total movement in Group 2 and 3 females were noted in the sixth and final activity interval only, and were not observed in Group 4. No RMANOVA treatment by time interaction, as well as no dose-dependent trend activity counts indicate that these changes were not treatment related.
Pathology
No substance-related gross pathological observations were noted at necroscopy by the testing facility. Spontaneous, incidental observations included a right epididymal mass in one animal at the 1500 mg/kg/day dose. Microscopically, this finding corresponded to a sperm granuloma). Additionally, another female animal in the 2000 mg/kg/day was observed with a fluid-filled uterus, which microscopically corresponded to estrous cycle-related dilation of the lumen.
Notable findings related to the test substance were evident in the kidneys, spleen, and thymus (Table 3). An accumulation of hyaline droplets was observed in the proximal tubular epithelial cells of the 2000 and 3000 mg/kg/day groups for both males and females, with males being more affected. This finding was of minimal to moderate severity and characterized by distorted cellular outlines, compressed tubular lumens, and was occasionally accompanied by cytoplasmic invaginations into the nucleus of affected cells or sloughing of individual epithelial cells into the tubular lumens. Droplets were not associated with tubular casts. Droplets of minimal severity were observed in 4 of the control group males and were absent in control females. Hyaline droplets in rats typically represent low molecular weight proteins accumulating in lysosomes due to increased protein filtration load or decreased catabolism by tubular epithelial cells. 26
Golden-brown pigment was visible in the proximal tubular epithelial cells of male and female rats in all test substance groups. A clear dose response was noted in female rats was noted. The accumulation of pigment was of minimal to mild severity. Pigment was found in two male control mice and was absent in female controls. Yellow-brown pigment in renal tubular cells in the rat often represents lipofuscin, bilirubin, or hemosiderin. In the present study, it is unlikely that the pigmentation was bilirubin or hemosiderin due to the absence of correlating clinical pathology findings to suggest underlying hyperbilirubinemia or hemolysis.
Compared to controls, the incidence of chronic nephropathy, characterized by chronic tubular degeneration and regeneration with interstitial fibrosis and mononuclear cell infiltrates, was increased in the 3000 mg/kg/day male rats. The incidence in lower dose groups was no different than that in the control group. No test substance-related effects were seen in females. Nephropathy is a common spontaneous process in laboratory rats and has been exacerbated at the highest dose levels by the previously described test substance effects on tubular epithelium.
An increase in golden-brown pigment was also present in the splenic red pulp macrophages in males in all dose groups with a clear dose-dependent effect. Pigmentation was also observed in the 3000 mg/kg/day female dose group. Pigmentation was of minimal to mild severity and had similar staining properties as hemosiderin commonly seen at lower levels as a background chance in female laboratory rats. Follicular lymphocytes were mildly decreased in the 3000 mg/kg/day a single male rat and was attributed to a non-specific change related to decreased body weight and/or stress.
An increase in apoptosis of thymic cortical lymphocytes was noted in all female dose groups and was considered minimal to mild severity. No difference in apoptosis was seen across dose groups in male rats. However, a decrease in thymic cortical thickness was noted in two males from the 3000 mg/kg/day dose group. Thymic findings were likely non-specific changes related to an endogenous glucocorticoid response (i.e. stress) and/or weight loss rather than directly related to the test substance.
Bilateral degeneration of seminiferous tubules was evident in 2 rats in the 3000 mg/kg/day male dose group and was characterized as minimal to mild. In one of the rats, the degeneration of the seminiferous tubules was associated with cellular debris in the epididymis. A focal unilateral degenerative tubular change was seen in one of the male rats in the 1500 mg/kg/day dose group. Bilateral degeneration could represent non-specific changes to decreased feed intake or a sporadic incidental finding. It is unlikely that bilateral degeneration is directly related to the test substance, given how few males in the study were affected. The unilateral lesion was undoubtedly spontaneous and incidental.
Organ weights
Males
Statistically significant decreases in absolute organ weights in males occurred in the epididymides of the 2000 and 3000 mg/kg/day dose groups (p < 0.05), the hearts of each dosage group (p < 0.05-0.001), 3000 mg/kg/day liver, (p < 0.01), 3000 mg/kg/day pituitary glands, all dose groups spleens (p < 0.05–0.001), and all dose groups thymus weights (p < 0.05). In the 3000 mg/kg/day dose group’s gastrocnemius muscle and retroperitoneal fat pads (p < 0.001). In addition, a decrease in tibial width was observed in the 3000 mg/kg/day male dose group when compared to the control (Table S12).
Statistically significant increases in organ to body weight ratios were observed in brain-to-body (p = 0.05–0.001) in all male dose groups, epididymides-to-body (p < 0.01) in the 3000 mg/kg/day dose group, kidneys-to-body (p < 0.001) in both the 2000 and 3000 mg/kg/day, and prostate/seminal vesicle-to-body (p < 0.01) and testes-to-body (p < 0.001) in the 3000 mg/kg/day dose group (Table 5).
Statistically significant decreases in organ-to-brain weights occurred in heart-to-brain ratio in the 1500 and 3000 mg/kg/day (p < 0.05–0.01), spleen-to-brain (p < 0.05–0.01) in all dose groups, and thymus-to-brain (p < 0.05) in the 1500 and 3000 mg/kg/day dose groups (Table 6).
Females
Absolute organ weight.
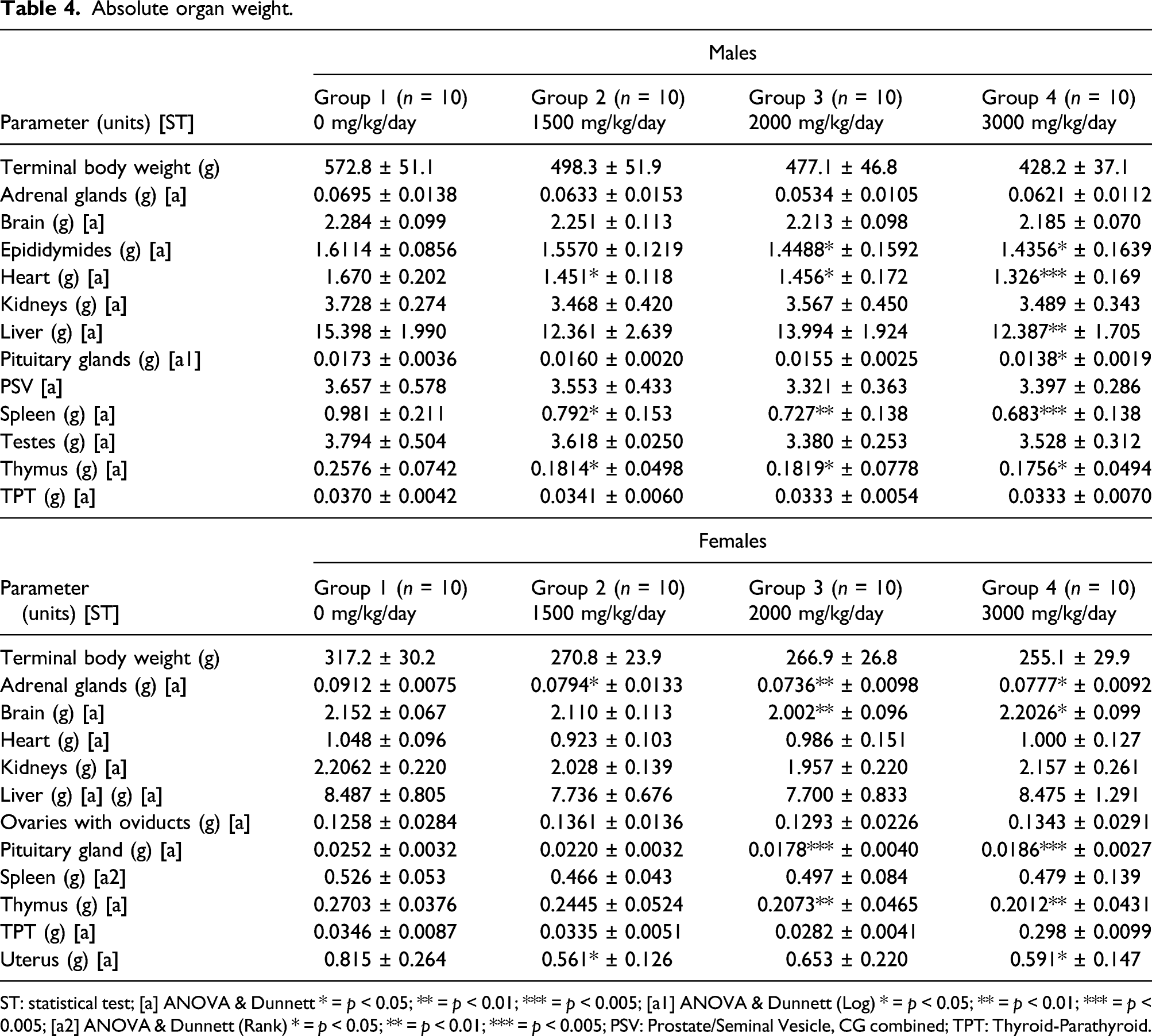
ST: statistical test; [a] ANOVA & Dunnett * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a1] ANOVA & Dunnett (Log) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a2] ANOVA & Dunnett (Rank) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; PSV: Prostate/Seminal Vesicle, CG combined; TPT: Thyroid-Parathyroid.
Changes in organ weights in relation to body weight.
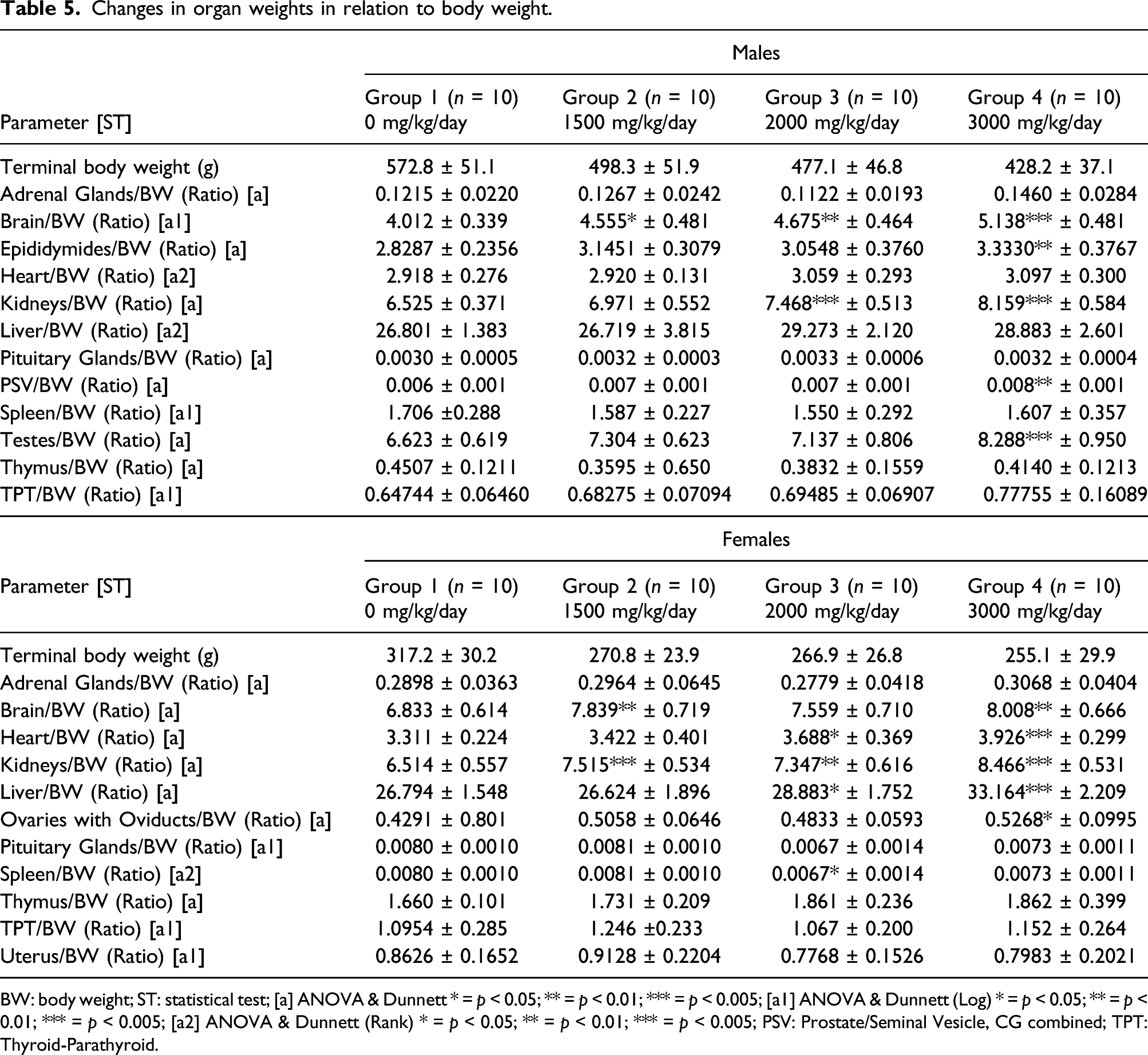
BW: body weight; ST: statistical test; [a] ANOVA & Dunnett * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a1] ANOVA & Dunnett (Log) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a2] ANOVA & Dunnett (Rank) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; PSV: Prostate/Seminal Vesicle, CG combined; TPT: Thyroid-Parathyroid.
Organ to brain weight.
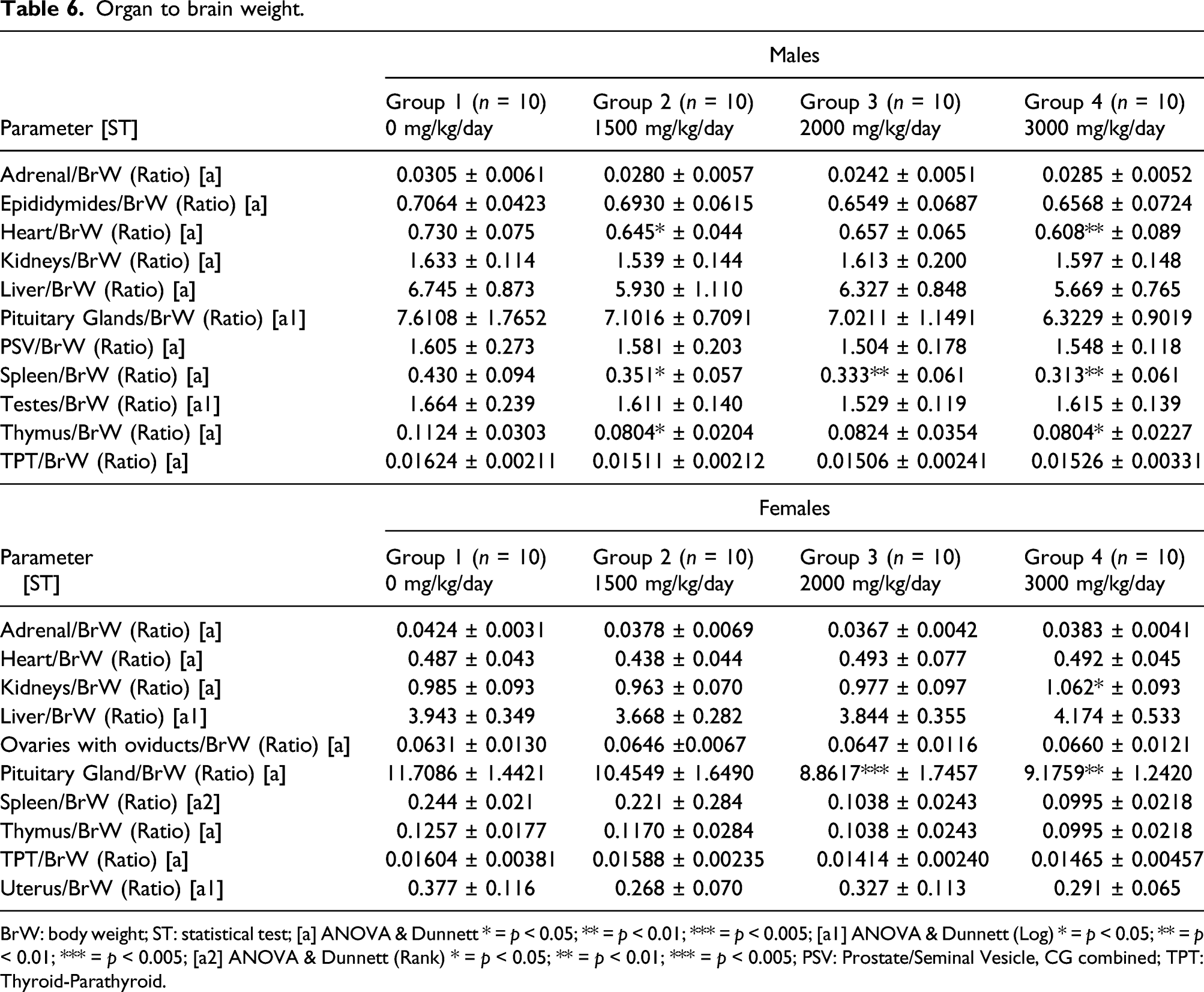
BrW: body weight; ST: statistical test; [a] ANOVA & Dunnett * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a1] ANOVA & Dunnett (Log) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; [a2] ANOVA & Dunnett (Rank) * = p < 0.05; ** = p < 0.01; *** = p < 0.005; PSV: Prostate/Seminal Vesicle, CG combined; TPT: Thyroid-Parathyroid.
Discussion
Nicotinamide riboside is considered to be a form of vitamin B3 because of the nicotinamide moiety and is an important precursor in the biosynthesis of NAD+. NAD+ can be found in all living cells and is important in enzyme-catalyzed redox reactions. 4 Tissues can produce NAD+ de novo from tryptophan and nicotinic acid, however this is much less efficient than production of NAD+ from vitamin B38. The importance of NAD+ in health and disease is well established. Declining levels of NAD+ in various tissues is being examined for a variety of reasons, including the impact of NAD+ on aging, alcohol use, and DNA damage.e4,8.
Nicotinamide riboside hydrogen malate is a newly developed NR salt which has been shown to be safe based on the results of the in vitro and in vivo studies described herein. NRHM was not genotoxic at doses up to 5000 μg/plate in a GLP compliant bacterial reverse mutation (Ames) assay and did not induce a statistically significant increase in the frequency of micronucleated cells in the GLP compliant in vitro micronucleus assay conducted with human peripheral lymphocytes. An in vivo 14-day repeat dose oral toxicity study was conducted to determine the appropriate doses to use in a GLP compliant 90-day repeat dose oral toxicity study in male and female Sprague Dawley rats. The 90-day oral repeat dose study was conducted in accordance with OECD Guideline 408, and no mortalities, NRHM related abnormal clinical observations or ophthalmological changes were reported.
No adverse functional effects in the FOB or motor activity assessment were reported. However, a significant NRHM-related reduction in degree and rate of body weight gain was reported in all treated males and females. Specifically, there was a 12, 16, and 24% decrease in the low, mid and high dose males and a 15, 16 and 18% decrease in the low, mid and high dose females, respectively, as compared to concurrent controls. There were differences in the mean daily body weight gain in all treated animals, both with and without statistical significance, that progressively contributed to the overall lower body weight. The decreases in the mean daily food consumption reported in both NRHM treated male and females were not considered to be adverse but did contribute to the decreases in body weight reported. The changes reported in the hematology, clinical chemistry, coagulation, thyroid hormone levels and urinalysis parameters were considered to be unrelated to NRHM exposure and likely due to biological variability. No gross pathological changes were reported which were related to NRHM exposure. However, histopathological findings in the kidneys, spleen and thymus were reported which were linked to NRHM exposure. The microscopic changes in the kidneys and spleen were not related to an adverse functional effect as note by the lack of corresponding adverse changes in any other parameters evaluated. The changes in the thymus were considered to be non-specific and related to stress and/or weight loss, rather than a direct effect of NRHM exposure.
The reduced degree and rate of weight gain in the NRHM treated animals was not an unexpected finding and is not considered to be adverse. Other studies conducted with NR or NR containing test materials reported that NR exposure led to reduced weight gain or impacted body weight gain in preclinical studies.2,4,11,27 In the current study, there were no adverse clinical signs reported and no change in the motor activity of the NRHM treated animals. The significantly decreased mass reported for the inguinal and retroperitoneal fat pads in the high dose males and females, the evaluation of the tibia length and width and the lack of adverse clinical signs demonstrate that the reduced body weight gain was impacted by a selective reduction in body fat with no evidence of malnutrition. While changes in specific fat depots are reported here, changes in body weight or composition have not been reported in human clinical trials in which subjects received up to 2000 mg/day of NR or a NR containing product. 4 The addition of a recovery group to the 90-Day subchronic toxicity study would have been beneficial to gauge reversibility of the effect on body weight of the test material.
The 90-day toxicology study reported here was conducted in Sprague-Dawley rats, which is the same species in which other pre-clinical toxicology studies of NR have been reported to date. This permits a degree of direct comparison between the different salt forms of NR. Based on the aggregate of the comparative data and the results of the studies reported here, NRHM has a safety profile similar to other NR containing products and is a safe source of NR at similar and even higher doses.
Conclusion
The results of the studies reported here indicate that, under the conditions reported, NRHM is not genotoxic and that, based on a GLP compliant 90-day oral repeat dose study, the NOAEL for NRHM is 2000 mg/kg bw/day and 3000 mg/kg bw/day for male and female Sprague-Dawley rats, respectively. The lower NOAEL in the male rats is based only on the reduced degree and rate of weight gain observed in the high dose group.
Supplemental Material
Supplemental Material - Preclinical safety assessments of nicotinamide riboside hydrogen malate
Supplemental Material for Preclinical safety assessments of nicotinamide riboside hydrogen malate by Margitta Dziwenka and Amy Mozingo in Toxicology Research and Application
Footnotes
Author contributions
M.D. was involved in conceptualization, protocol development, data analysis, drafting and finalizing the manuscript. A.M. was involved in conceptualization and editing the final manuscript.
Availability of data
batasets generated and/or analyzed during the current studies are available in the manuscript or as supplementary data provided with the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
(include full name of committee approving the research and if available mention reference number of that approval}: Product Safety Labs Institutional Animal Care and Use Committee.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
