Abstract
Naturally sweet proteins have no glycemic effect and offer a fundamentally new approach to sweetness and health for individuals seeking to reduce their added sugar intake. However, unlike many commercial sweeteners, little research has been performed on the potential safety implications of adding these uniquely sweet proteins to food and beverages. In this study, a naturally sweet protein found in the West African Oubli plant (Pentadiplandra brazzeana), referred to as Oubli fruit sweet protein or brazzein, was expressed in Komagataella phaffii (formerly Pichia pastoris) and produced via precision fermentation, and a safety and risk assessment was undertaken for its use as a sweetener in food and beverages. Potential consumption levels of brazzein were estimated to be 3 mg/kg body weight/day based on the National Health and Nutrition Examination Survey. The safety of brazzein derived from K. phaffii was evaluated through in silico allergenicity, in vitro genotoxicity (reverse mutation and mammalian micronucleus assays), and a 90-day dietary oral toxicity study in rats. There was no indication of allergenicity in the in silico analyses. Brazzein was non-genotoxic in the in vitro assays and showed no adverse effects in the 90-day oral toxicity study up to the highest dose tested, where the no-observed-adverse-effect level (NOAEL) was 978 and 985 mg/kg body weight/day in males and females, respectively. The totality of evidence in the in silico allergenicity, in vitro genotoxicity, and 90-day dietary toxicity studies demonstrates that brazzein derived from K. phaffii is considered safe for use as a sweetener in food and beverages.
Introduction
As long as humans have existed, so has our inherent taste for sugar. 1 A ready source of energy, natural sugars such as fructose, sucrose, and lactose have traditionally shaped our relationship with sweetness and many of the foods we eat from infancy through adulthood. 2 For early human diets, these readily accessible calories were likely derived from seasonal fruits, vegetables, and other wild sources. As humankind has now transitioned to an age of mass sugar production and consumption from sugarcane, corn, and other domesticated sugar crops, both the cost and impact of added sugar on global diets has drastically changed. 3 Despite this rise in abundance of inexpensive sugar, our innate affinity and taste for sweetness remains the same, leading many to consume food and beverage products high in added sugar. 4
Over the past several decades, the role of sugar intake in human health and disease has been well-studied. 5 Excess sugar intake has been correlated with increased incidence of obesity, diabetes, cardiovascular disease, and cancer.6,7,8,9 As part of efforts to reduce sugar intake in the 1960s through the 1990s, a number of synthetically produced high-intensity sweeteners were developed for use in food.10,11 This trend has continued with the commercialization of high-intensity, low-calorie sweeteners of natural origin, even if not directly extracted from plants. 12
In addition to high-intensity sweeteners, interest in the properties of naturally sweet macromolecules such as proteins has intensified. 13 Due in large part to their amino acid-based composition and structure, sweet proteins offer a fundamentally different path to both sweetness and health than the incumbent small molecule sweeteners.14,15 By their nature, sweet proteins do not contribute to the glycemic index of food and beverage products, which is often a key marker for fluctuations in blood sugar. Their sugar-sweet taste in combination with no glycemic effect not only helps satisfy the ever-increasing demand for low-calorie sweeteners, but also addresses the needs of millions of individuals seeking to decrease sugar intake in their diets for critical health reasons. 13
The discovery of sweet proteins found predominantly in plants suggests that their uniquely sugar-like taste profile is no accident, but instead due to their evolution alongside humankind and its inherent taste for sugar. It was once thought that large macromolecules such as proteins were unable to elicit sweet taste response and would generally be considered tasteless relative to small molecules like sugars.16,17 More recently, however, plant-based proteins have been discovered throughout nature that possess sweetening properties only for humans and higher primates. 18 Sweet plant proteins likely evolved out of competition for efficient seed dispersal amongst plant species. 19 These “trickster” plants were able to improve their overall energy efficiency with lower sugar fruits by producing small amounts of “sweet proteins” that mimic the taste of sugar. 20 Examples of these naturally sweet plant proteins include thaumatin, monellin, mabinlin, brazzein, and curculin.17,21 At present, this has led to the commercialization of one approved plant protein, thaumatin, as a sweetener and flavor enhancer.22,23,24
Oubli fruit sweet protein, also referred to interchangeably as brazzein in this report, has been identified as another such “sweet protein.” Derived from the West African Oubli plant (Pentadiplandra brazzeana), brazzein has a sweetness 500 to 2000 times that of sucrose on a weight-weight basis, with the 53-amino acid sequence (referred to as brazzein-53) showing the highest sweetness potency.25,26,27,28,29 The full amino acid sequence of brazzein-53 is as follows:
DKCKKVYENYPVSKCQLANQCNYDCKLDKHARSGECFYDEKRNLQCICDYCEY
The physio-chemical properties of brazzein allow for its potential use in a wide range of food and beverage categories and food preparation/cooking modalities. 30 As part of the effort to commercialize brazzein for use in the food and beverage industry, brazzein-53 was produced by Oobli, Inc. via precision fermentation using Komagataella phaffii (formerly Pichia pastoris).
While traditionally consumed in West Africa from the fruit of the plant, very little data have been published on the safety of brazzein and its potential for allergenicity and toxicity when used as an ingredient in food and beverages. In this study, a series of in silico, in vitro and in vivo investigations were conducted to develop a full safety profile for brazzein produced as described above. The significance of allergenicity was assessed in silico due to the ability of certain proteins to induce an allergic response in susceptible individuals. The novel genotoxic potential of brazzein was investigated in two in vitro genotoxicity tests: the bacterial reverse mutation assay (OECD 471) and the in vitro micronucleus test in mammalian cells (OECD 487). Finally, the safety of brazzein was evaluated in a 90-day oral dietary toxicity study (OECD 408) in rats designed to assess potential systemic toxicity. The in vitro genotoxicity studies and the 90-day oral dietary rat study were all conducted in accordance with their respective relevant Organisation for Economic Co-operation and Development (OECD) Test Guidelines and Good Laboratory Practice (GLP) requirements.31,32,33,34
Materials and methods
Good laboratory practice and regulatory guidelines
The toxicology studies, namely the in vitro bacterial reverse mutation (Ames) assay (OECD 471) and 90-day subchronic dietary toxicity study (OECD 408) in rats, were conducted at Product Safety Labs (PSL) of Dayton, New Jersey (USA). Each study was conducted according to GLP31,35 and respective OECD Test Guidelines.31,33,34 PSL is AAALAC (Association for Assessment and Accreditation of Laboratory Animal Care) accredited and certified in the appropriate care of all live experimental animals, and they maintained current staff training, ensured animals were handled humanely during the experimental phase of this study, and met all guideline standards, including NRC. 36
The in vitro micronucleus study (OECD 487) was conducted at Eurofins BioPharma Product Testing Munich GmbH (Munich, Germany). This was also conducted under GLP conditions 31 and in accordance with the respective OECD Test Guideline (487) and relevant European Union (EU) regulations32, 37 (Commission regulation (EU) 2017/735 B.49).
Test material, production, and estimated daily intake
Composition of Brazzein powder produced from K. phaffii.
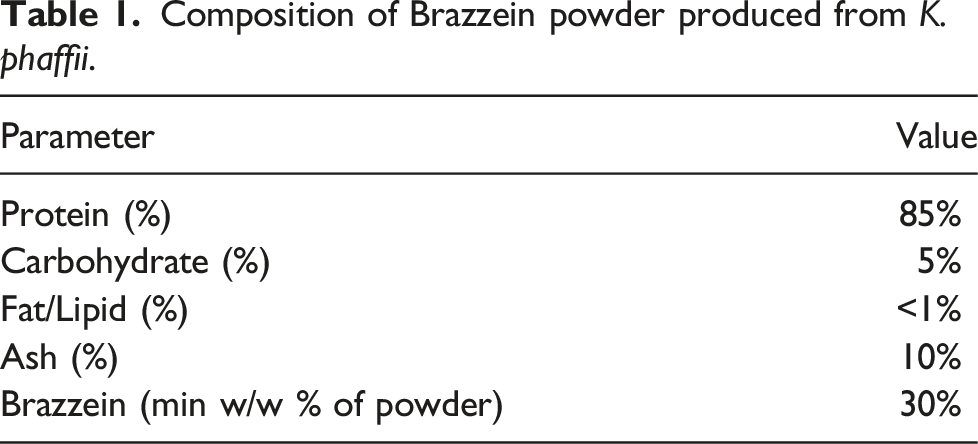
The K. phaffii strain BG10 (ATUM, PPS-9010) was derived from the parent strain K. phaffii NRRL Y-11430 which is a non-toxigenic, non-pathogenic, well-characterized strain lineage that has a history of safe use in manufacturing proteins for use in food and pharmaceuticals and follows current Good Manufacturing Practices.38,39,40,41,42 The K. phaffii NRRL Y-11430 is a well-characterized, commonly used strain, which has been deposited in multiple public culture collections (ATCC 76273, NRRL Y-11430). The production strain for Oobli’s brazzein was derived from K. phaffii BG10 through a series of transformations with different expression constructs to enable the biosynthesis of the brazzein. The promoter and terminator within the expression cassette are native sequences of K. phaffii. The gene encoding for brazzein was synthesized de novo and codon-optimized from Pentadiplandra brazzeana (UniProtKB P56552). The master cell bank was prepared from a single colony and grown in defined medium, and its DNA was sequenced to confirm its identity. A further evaluation of the BG10 K. phaffii production organism was performed using the principles of the safety assessment procedure for enzyme preparations, as described by Pariza and Johnson. 43
Based on the physio-chemical and organoleptic properties of brazzein, potential human exposures from its inclusion in food have been estimated using the National Health and Nutrition Survey (NHANES) 2017-2018 database.44,45,46 This database, in conjunction with anticipated food use categories and inclusion levels, can be used to produce an estimated daily intake (EDI) value. On a consumer-only basis, the resulting mean and 90th percentile EDI for brazzein for the total population was estimated to be 89 mg/person/day (1.30 mg/kg body weight/day) and 199 mg/person/day (2.88 mg/kg body weight/day), respectively. Among the individual population groups, and on an absolute basis, male adults had the highest mean and 90th percentile intakes of brazzein, while on a body weight basis, the highest mean and 90th percentile intakes were estimated in young children (2 to 5 years). The EDI was used in part to estimate dose levels that would be used in the 14-day dose range-finding study. The doses for the dose-range study were set based on approximately 50-, 100-, and 200-fold of the EDI, which were 250, 500, and 1000 mg/kg body weight/day, respectively.
In silico assessment of allergenicity
According to the United States Food and Drug Administration (FDA), food allergies and other types of food hypersensitivities affect millions of Americans and their families. As such, assessing the potential for allergenicity of a new food ingredient is critical to its overall safety evaluation. Two peer-reviewed, curated allergen databases have been developed and validated to assess the potential allergenicity of proteins and peptides according to the recommendations of the Codex Alimentarius Commission and the Food and Agricultural Organization (FAO)/World Health Organization (WHO): AllergenOnline 47 and Allermatch. 48 The potential allergenicity of brazzein (UniProtKB: P56552) was evaluated through a series of sequence homology searches against putative allergenic proteins from the AllergenOnline Database (Version 21; http://www.allergenonline.org/) and the Allermatch Database (https://allermatch.org/). Sequence homology searches were conducted in a stepwise manner as recommended by the Codex Alimentarius 49 and the FAO/WHO 50 using a sliding 80-amino acid window derived from the full-length amino acid sequence of brazzein as well as an 8-amino acid exact match. A search was also conducted with the full-length amino acid sequence of brazzein. All searches were performed using the default settings of each allergen database using FASTA36. For the 80-amino acid sliding window, a percent identity cut-off of 35% was used to determine whether significant sequence homology exists between brazzein and the allergenic protein. In addition, the E-value and bit-score were also considered as indicators of whether the matches reflect a biologically relevant similarity.51,52
In vitro genotoxicity tests
Bacterial reverse mutation test (OECD 471)
The bacterial reverse mutation assay (Ames test) was conducted using Salmonella typhimurium (S. typhimurium) strains TA98, TA100, TA1535, and TA1537, and Escherichia coli (E. coli) WP2 uvrA, both in the presence and absence of metabolic activation by S9 mix. S. typhimurium and E. coli strains were obtained from Molecular Toxicology, Inc. (Boone, North Carolina). Overlay agar (supplemented with biotin and limited amounts of histidine and tryptophan) and minimal glucose agar plates were purchased from Molecular Toxicology, Inc. The methods followed those reported by Ames et al. 53 and Maron and Ames. 54
The main test was conducted with and without metabolic activation by the plate incorporation method. Eight dose levels (1.58, 5.0, 15.8, 50, 158, 500, 1580, and 5000 μg/plate) were utilized with testing conducted in triplicate. Sterile water was used as the negative (vehicle) control. Positive controls sodium azide (Chemical Abstracts Service [CAS] No. 26628-22-8), 9-aminoacridine hydrochloride monohydrate (CAS No. 52417-22-8), 2-nitrofluroene (CAS No. 607-57-8), 4-nitroquinoline N-oxide (CAS No. 210222-18H), 2-aminozanthracene (CAS No. 210222-21H), and benzo(a)pyrene (CAS No.210222–22H) were obtained from Sigma-Aldrich and formulated using dimethyl sulfoxide.
A mix of test article, positive or negative control article (0.1 mL of each) at the indicated concentration, 0.5 mL of S9 or substitution (sodium phosphate) buffer, 0.1 mL bacteria suspension (1 × 109 colony forming units/mL), and 2 mL overlay agar were poured over the surface of a prepared minimal agar plate. Plates were allowed to set and incubated at 37°C until adequate growth for colony enumeration was observed (∼65 hours). Sterility control plates were included to ensure reagent sterility.
A confirmatory test using the pre-incubation method was also performed. For this method, test article, positive or negative control, S9 or substitution buffer, and bacteria suspension were mixed in the same ratios as for the main test, and incubated with agitation for 30 minutes at 37°C prior to mixing with the overlay agar. After mixing with the overlay, confirmatory testing was conducted as described for the plate incorporation method.
For both the main and confirmatory tests, revertant colonies were counted manually and/or with an automatic colony counter (Colony-Doc-It™). Toxicity of the test material was evaluated based on precipitation of test article and evaluation of the background bacterial lawn for abnormalities. A test was considered positive if the mutation factor (i.e., number of revertants in treated group/number revertants in controls) was ≥2-fold for strains TA98, TA100, and WP2 uvrA or ≥3-fold for strains TA1535 and TA1537. For a result to be considered positive, the mean value was also required to be outside of the historical control range, be dose related, and occur at more than one experimental point. Otherwise, tests were considered negative for mutagenic potential.
In vitro mammalian cell micronucleus test (OECD 487)
An in vitro micronucleus assay was undertaken following previously described methods.32,55,56,57 Human peripheral blood lymphocytes from healthy, non-smoking donors were collected via venous puncture and collected in heparinized tubes. Each donor received training as per Standard Operating Procedures (SOPs), signed a consent form, and received confidentiality through randomization, where all data was protected by German data protection laws. Blood was stored at 4°C for a maximum of 4 hours prior to use. Whole blood samples were incubated with a mitogen (phytohaematogglutinin) for 44–48 hours prior to exposure to the test article. Test article and positive controls were dissolved in vehicle (negative) control medium (RPMI 1640). Clastogenic controls consisted of 50 μg/mL methylmethanesulfonate (Sigma) without metabolic activation and 15 μg/mL cyclophosphamide (Sigma) with metabolic activation. Aneugenic control was 0.4 μg/mL colchicine (Sigma) without metabolic activation. The S9 liver microsomal fraction was obtained from Trinova Biochem GmbH, Giessen, Germany. Male Sprague-Dawley rats were induced with phenobarbital/β-naphthoflavone.
An initial analysis for test article cytotoxicity was conducted with and without S9 metabolic activation. Tests were conducted using 15.6, 31.3, 62.5, 125, 250, 500, 750, 1000, 2500, and 5000 μg/mL test article. Cytotoxicity was assessed via cytokinesis-block proliferation index (CBPI) using the formula: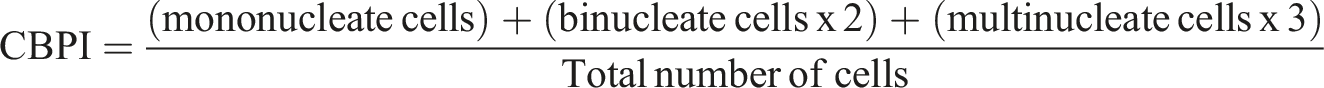
The CBPI was used to calculate the % cytostasis, which indicated the inhibition of cell growth of treated cultures in comparison to control cultures: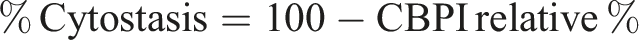
For the short-term exposure experiment, duplicate cultures were tested at 62.5, 125, 250, 500, 1000, and 2000 μg/mL of test article with and without S9 activation. Cultures were exposed to the test article for 4 hours, after which the treatment medium was washed twice with 10% phosphate-buffered saline. Cells were then incubated in complete culture medium containing 6 mg/mL cytochalasin B for 40 to 42 hours at 37°C and 5% CO2.
For the long-term exposure experiment, duplicate cultures were tested at the same concentrations as for the short-term experiment without S9 activation only. Cultures were exposed to test article for 1 hour in complete culture medium, followed by addition of 6 μg/mL cytochalasin B and incubation for 43 hours at 37°C and 5% CO2.
All experimental cells were prepared for microscopic analysis at the end of the culture period by removal of the culture medium and treatment with 4°C hypotonic solution (0.075 M KCl) at room temperature, and centrifuged. The cell pellet was then resuspended in a solution consisting of fixation solution and 0.9% NaCl and centrifuged again. Next, cells were fixed with methanol and glacial acetic acid (3:1), placed on clean glass slides, dried, and stained with acridine orange. At least 2000 binucleated cells (1000 for each replicate) for each dose group were analyzed for micronuclei as per previously published methods. 55 Mononucleated and multinucleated cells and cells with more than 6 micronuclei were not considered. 58
The test was considered to be clearly positive if: 1) at least one of the test concentrations exhibited a statistically significant increase compared with concurrent negative control as determined by the χ2 test; 2) the increase was concentration-dependent; and, 3) the results were outside the distribution of the historical negative control data (Poisson-based 95% confidence intervals). The test was considered clearly negative if none of these criteria were met.
In vivo 14-day dietary palatability and range-finding study in rats
Four groups of adult CRL: Sprague-Dawley® CD® IGS rats (5/sex/group) were maintained on diets prepared to contain 3000, 6000, and 12000 ppm of brazzein, for target daily intakes of 250, 500, and 1000 mg/kg/day. As this was a range-finding study, it was not conducted according to GLP. The target daily doses for this study were based on multiples of the upper 90th percentile human EDI derived using the NHANES exposure database.
The animals were individually housed in suspended stainless steel cages which conform to the size recommendations in the latest Guide for the Care and Use of Laboratory Animals. 36 Litter paper placed beneath the cage was changed at least three times/week. The animal room had a 12-hour light/dark cycle and was kept clean and vermin-free.
The animals were observed at least once daily for viability, signs of gross toxicity, and behavioral changes, and weekly for a battery of detailed observations. Body weights were recorded two times during the acclimation period (including prior to initial dietary administration on Day 0), on Days 3, 7, 10, and 14, and immediately prior to sacrifice. Individual food consumption was also recorded to coincide with body weights. Food efficiency and dietary intake were calculated. A gross necropsy was performed on all animals at study termination (Day 15).
In vivo (subchronic) repeated dose 90-day oral (dietary) toxicity study in rats
For the 90-day study, Sprague-Dawley rats [Crl:CD(SD)] were obtained from Charles River Laboratories, Inc. at 7 to 8 weeks of age. Male and female rats were acclimated for a minimum of 5 days prior to commencement of testing. Forty rats of each sex were assigned by randomization to ensure no statistically significant difference among group body weights by sex. Animals were allowed free access to a commercial diet (2016 Certified Envigo Teklad Global Rodent Diet) containing the test article and tap water provided ad libitum. Animals were individually housed at 21 ± 2°C, with relative humidity of 50 ± 20%, and a 12-hour day/night cycle.
Brazzein was administered in the diet at varying concentrations to achieve dosages consisting of zero (negative control; control diet), 250, 500, and 1000 mg/kg body weight/day. To achieve the target doses, the dietary concentrations of brazzein were adjusted weekly on the basis of the previous weeks’ food consumption and body weight data. First week dietary concentrations were prepared based on initial body weights and historical control data for feed consumption. The doses for the 90-day subchronic study were established based on the results of the 14-day dose range-finding study.
Brazzein was added to 2016 Certified Envigo Teklad Global Rodent Diet® and thoroughly mixed in a high-speed mixer. Control diet was mixed under the same conditions as the diets prepared with the test substance. All diets were refrigerated following preparation, unless presented to the test animals on the same day as diet preparation. All diets were prepared approximately weekly or more frequently, as needed. The test diets were presented to their respective groups on Day 0 of the study. Additional diet was provided as needed throughout the study to ensure ad libitum feeding. Animals were exposed to the test diets for at least 90 days. The brazzein-containing diets were analyzed using a validated HPLC method for concentration verification (Weeks 1, 6 or 7, and 13 for representative dietary concentrations) and homogeneity (Weeks 1 and 13), as well as stability.
Animals were observed for clinical signs such as abnormalities in appearance, nutritional condition, posture, behavior, and excretions three times a day. Body weights were recorded twice in the first week of the study, once a week thereafter, and at necropsy. Food consumption was recorded twice during the initial week of the study, and once a week thereafter. Animals were administered ophthalmologic examinations using an indirect ophthalmoscope before and at the beginning of Week 13 of the study.
Functional observational battery (FOB) tests were conducted once prior to test substance administration, again during Week 1, and monthly thereafter. Motor activity (MA) was also be evaluated (PSL SOP #735). The MA assessments coincided with the FOB assessments for the same animals. Activity was monitored using an automated Photobeam Activity System® (San Diego Instruments, Inc.). The system monitored up to 20 animals during one session.
In the FOB tests, each rat was evaluated during handling and while in an open field for excitability, autonomic function, gait, and sensorimotor coordination (open field and manipulative evaluations), reactivity and sensitivity (elicited behavior), and other abnormal clinical signs, including but not limited to convulsions, tremors unusual or bizarre behavior emaciation, dehydration, and general appearance. In addition to the above observations, forelimb and hindlimb grip strength and foot splay measurements were made and recorded.
At necropsy, Study Day 92 for males and 93 for females (differing days for logistical reasons), approximately 500 µL of blood was collected in a pre-calibrated tube containing dipotassium ethylenediaminetetraacetic acid for hematology assessments. Approximately 1.8 mL of blood was collected in a pre-calibrated tube containing 3.2% sodium citrate for analysis of blood coagulation parameters. Approximately 1000 µL of blood was collected into tubes containing no preservative for clinical chemistry assessments. These samples were centrifuged in a refrigerated centrifuge and the serum transferred to a labeled tube. Blood was also examined for triiodothyronine (T3), thyroxine (T4), and thyroid-stimulating hormone (TSH) using an enzyme-linked immunosorbent assay method.
Urinalysis was conducted on the day of necropsy. Urine samples were collected every 4 hours under deprivation of food but free access to water, and 20-hour urine samples were collected with free access to both food and water.
At terminal sacrifice, vaginal smears were collected from all female rats on the day of terminal sacrifice to determine the stage of estrus (PSL SOP #740).
At the end of the study, all animals were sacrificed and necropsied. The following organs were weighed, and absolute and relative organ weights determined: brain, pituitary, thyroid, adrenal, thymus, spleen, heart, lung, salivary gland, liver, kidney, testis, prostate, seminal vesicle, ovary and uterus. Additional histopathological studies were conducted on organs after fixation and staining. Organs were examined microscopically from each of the control and high-dose groups. Organs examined included: cerebrum, cerebellum, spinal cord (thoracic), sciatic nerve, eyeball, optic nerve, harderian gland, pituitary, thyroid, parathyroid, adrenal, thymus, spleen, submandibular lymph node, mesenteric lymph node, heart, thoracic aorta, trachea, lung, tongue, esophagus, stomach, duodenum, jejunum, ileum, cecum, colon, rectum, submandibular gland, sublingual gland, liver, pancreas, kidney, urinary bladder, testis, epididymis, prostate, seminal vesicle, ovary, uterus, oviduct, vagina, mammary gland, sternum, femur, femoral skeletal muscle, skin, nasal cavity, and Zymbal gland.
Statistical parameters of body and organ weights, food and water consumption, urinalysis, and hematology were calculated as the mean ± standard deviation. An analysis of variance was conducted by the Bartlett test (p > 0.01). Homogeneous data were then compared (control and test groups) by the Dunnett’s test (p > 0.05 and p > 0.01, two-tailed) while heterogeneous data were compared by the Steel’s test (p > 0.05 and p > 0.01, two-tailed). All analyses were performed by SAS Release 9.1.3 (SAS Institute, Inc.).
Results
In silico allergenicity analyses
Brazzein did not share any significant sequence homology matches with any known allergens from the AllergenOnline Database or Allermatch Database. All matches shared less than 35% identity in all 80-amino acid sliding window searches, and no 8-amino acid exact matches were identified with any known allergens from the AllergenOnline Database or Allermatch Database. The full-length amino acid sequence search identified a match sharing >50% identity with a pathogenic-related protein from peach (Prunus persica). The corresponding E-value and bit-score were 0.76 and 24.6, respectively. The full-length amino acid sequence search against the Allermatch Database identified matches with 20 putative allergens sharing >50% identity.
In vitro genotoxicity tests
Bacterial reverse mutation test
Bacterial reverse mutation test conducted with Brazzein.
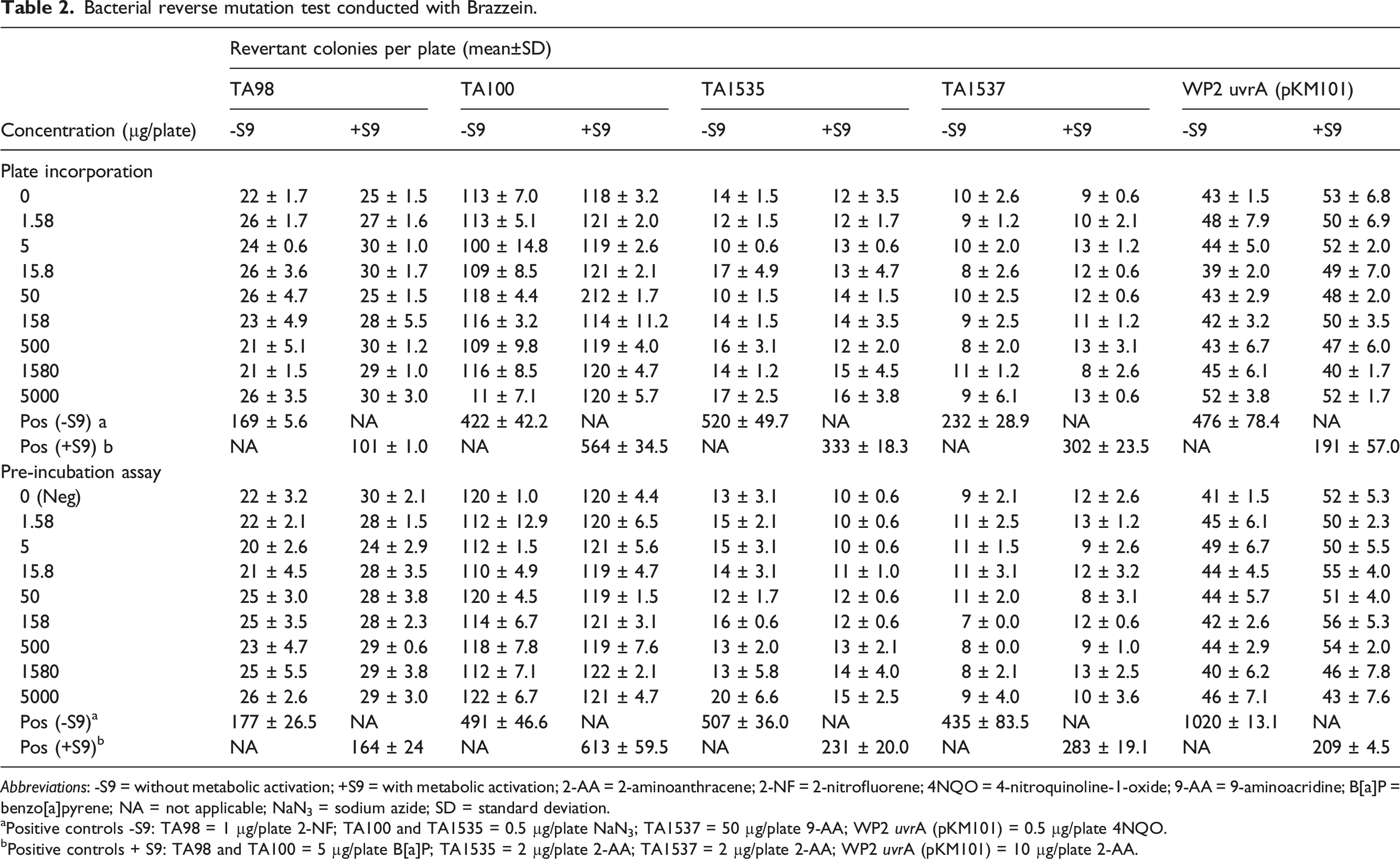
Abbreviations: -S9 = without metabolic activation; +S9 = with metabolic activation; 2-AA = 2-aminoanthracene; 2-NF = 2-nitrofluorene; 4NQO = 4-nitroquinoline-1-oxide; 9-AA = 9-aminoacridine; B[a]P = benzo[a]pyrene; NA = not applicable; NaN3 = sodium azide; SD = standard deviation.
aPositive controls -S9: TA98 = 1 µg/plate 2-NF; TA100 and TA1535 = 0.5 µg/plate NaN3; TA1537 = 50 µg/plate 9-AA; WP2 uvrA (pKM101) = 0.5 µg/plate 4NQO.
bPositive controls + S9: TA98 and TA100 = 5 µg/plate B[a]P; TA1535 = 2 µg/plate 2-AA; TA1537 = 2 µg/plate 2-AA; WP2 uvrA (pKM101) = 10 µg/plate 2-AA.
In vitro mammalian cell micronucleus test
In vitro mammalian cell micronucleus test in human peripheral blood lymphocytes treated with Brazzein.
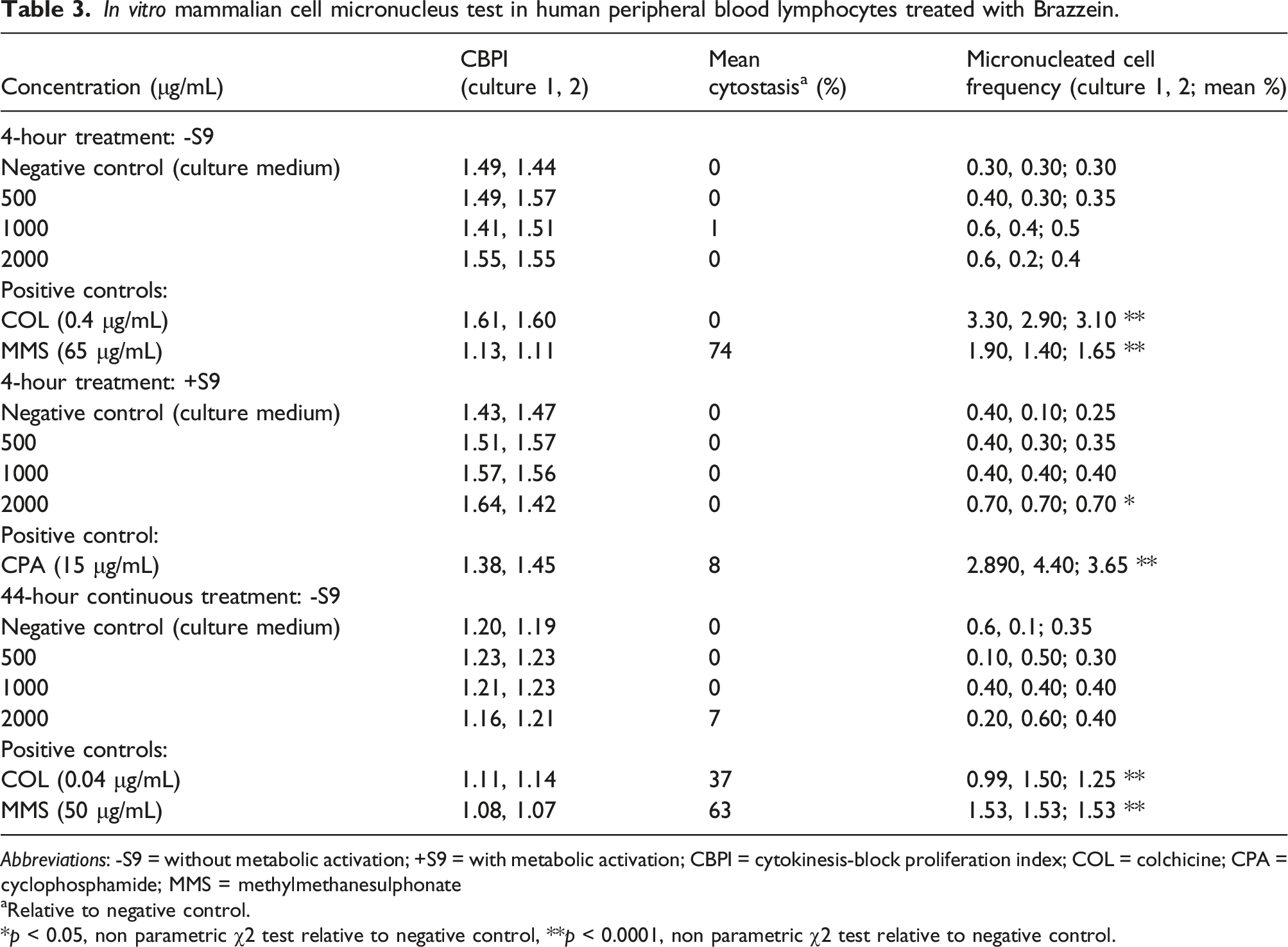
Abbreviations: -S9 = without metabolic activation; +S9 = with metabolic activation; CBPI = cytokinesis-block proliferation index; COL = colchicine; CPA = cyclophosphamide; MMS = methylmethanesulphonate
aRelative to negative control.
*p < 0.05, non parametric χ2 test relative to negative control, **p < 0.0001, non parametric χ2 test relative to negative control.
The test as a whole was considered to be valid as all conditions of validity were met: concurrent negative/vehicle controls were consistent with laboratory historical controls; concurrent positive controls induced a statistically significant increase in micronuclei; cell proliferation as measured by CBPI was ≥70% (≤30% cytostasis); and an adequate number of cells (≥500 cells per treatment) and concentrations (≥3) were analyzed. The positive controls induced biologically relevant and statistically significant increases in the percentage of micronucleated cells compared with vehicle controls, with mean values within historical positive control ranges. In summary, brazzein showed neither mutagenic nor cytotoxic potential in the in vitro micronucleus test.
In vivo 14-day dietary palatability and range-finding study in rats
In the 14-day dose range-finding study, there were no effects of brazzein treatment at up to nominal doses of 1000 mg/kg body weight/day on mortality, clinical signs, body weight, body weight gain, or food consumption. No treatment-related abnormalities were noted upon macroscopic examination at necropsy. The results of the study supported the use of 1000 mg/kg body weight/day as a high dose in the 90-day dietary toxicity study.
In vivo repeated dose 90-day oral (dietary) toxicity study in rats
Brazzein was found to be incorporated into the diet in a homogenous and accurate manner and was demonstrated to be stable as prepared for use in the 90-day study.
Body weights for male and female rats administered Brazzein via the diet for at least 90 days.
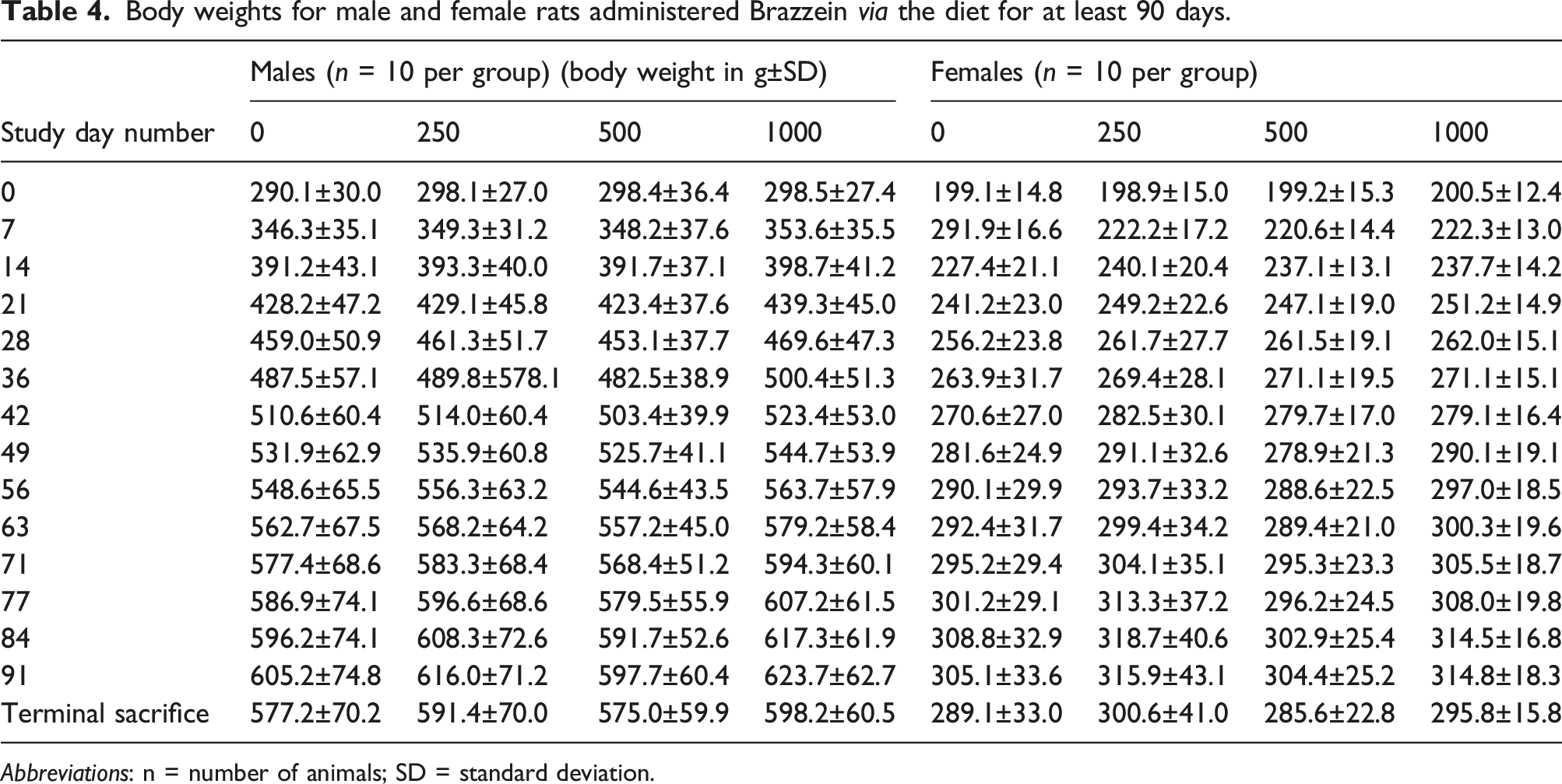
Abbreviations: n = number of animals; SD = standard deviation.
Feed consumption and achieved intake for representative periods for rats fed Brazzein via the diet for at least 90 days.
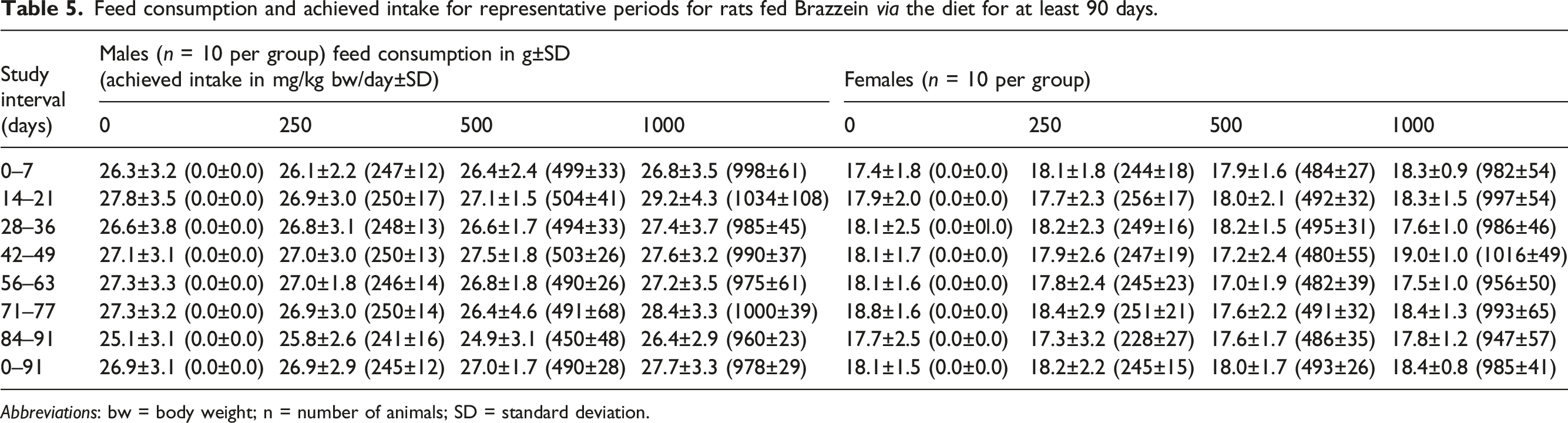
Abbreviations: bw = body weight; n = number of animals; SD = standard deviation.
Treatment with brazzein was not associated with any increased incidence of clinical symptomology, change in general condition, or change in the results of the ophthalmological examinations (data not shown).
Results of the functional observational battery tests in male and female rats administered Brazzein via the diet for at least 90 days.
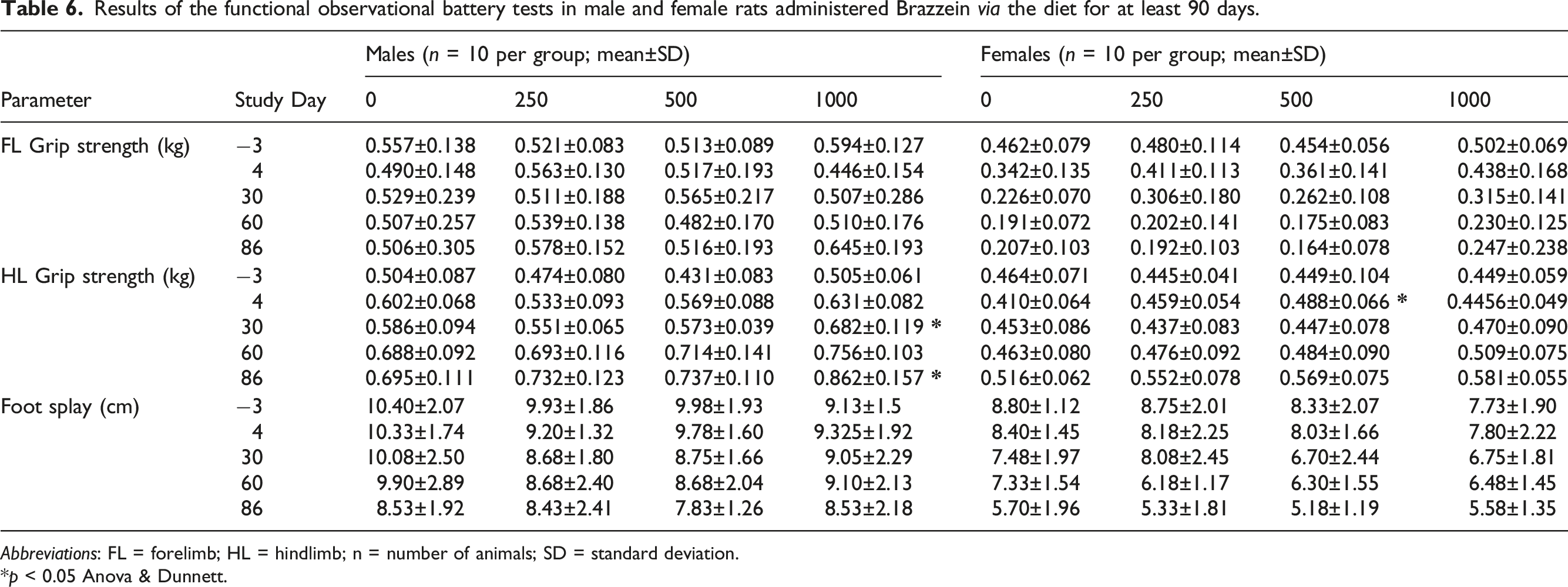
Abbreviations: FL = forelimb; HL = hindlimb; n = number of animals; SD = standard deviation.
*p < 0.05 Anova & Dunnett.
Hematology and coagulation values for male and female rats administered Brazzein via the diet for at least 90 days.
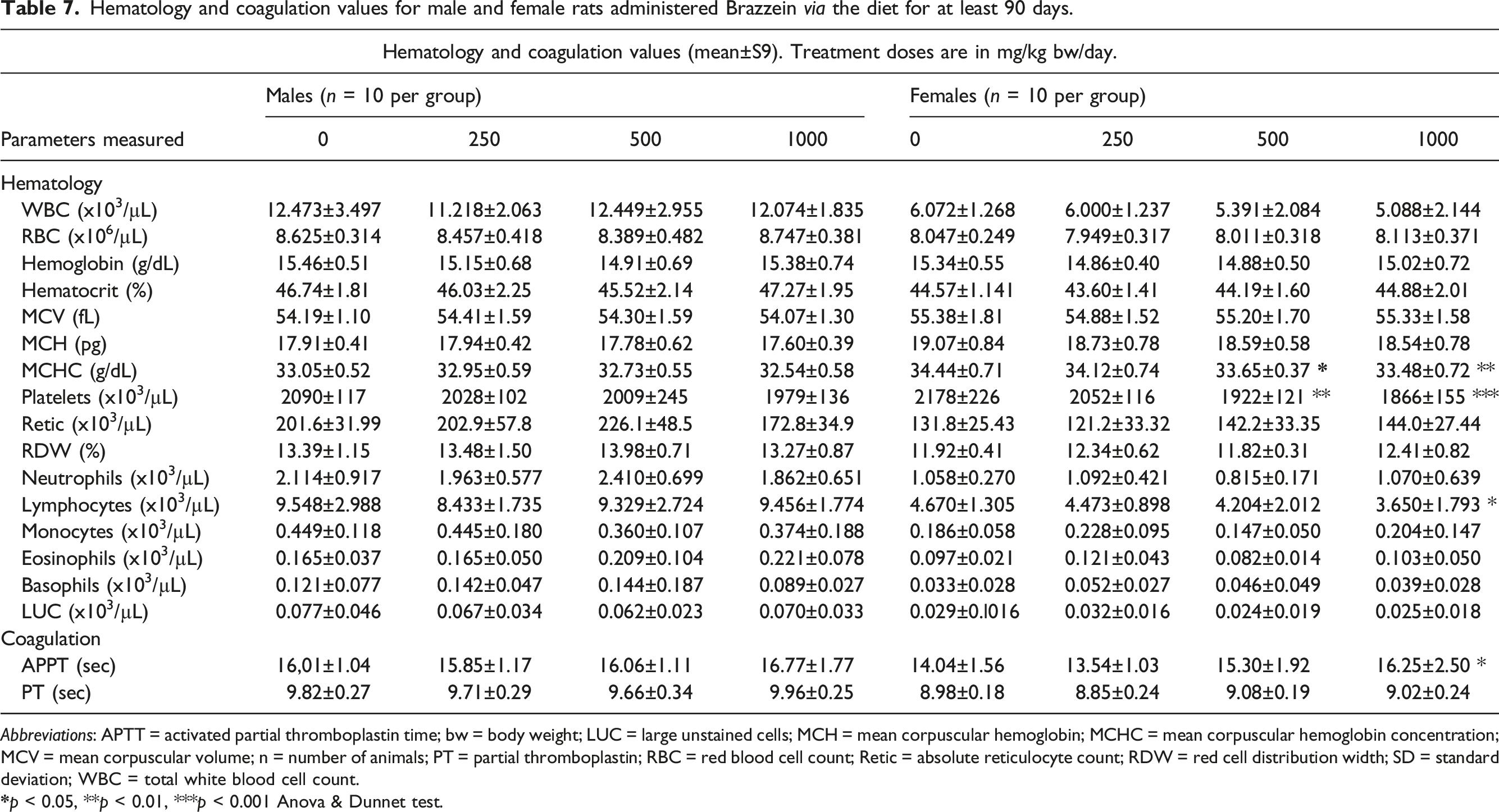
Abbreviations: APTT = activated partial thromboplastin time; bw = body weight; LUC = large unstained cells; MCH = mean corpuscular hemoglobin; MCHC = mean corpuscular hemoglobin concentration; MCV = mean corpuscular volume; n = number of animals; PT = partial thromboplastin; RBC = red blood cell count; Retic = absolute reticulocyte count; RDW = red cell distribution width; SD = standard deviation; WBC = total white blood cell count.
Clinical chemistry values for male and female rats administered Brazzein via the diet for at least 90 days.
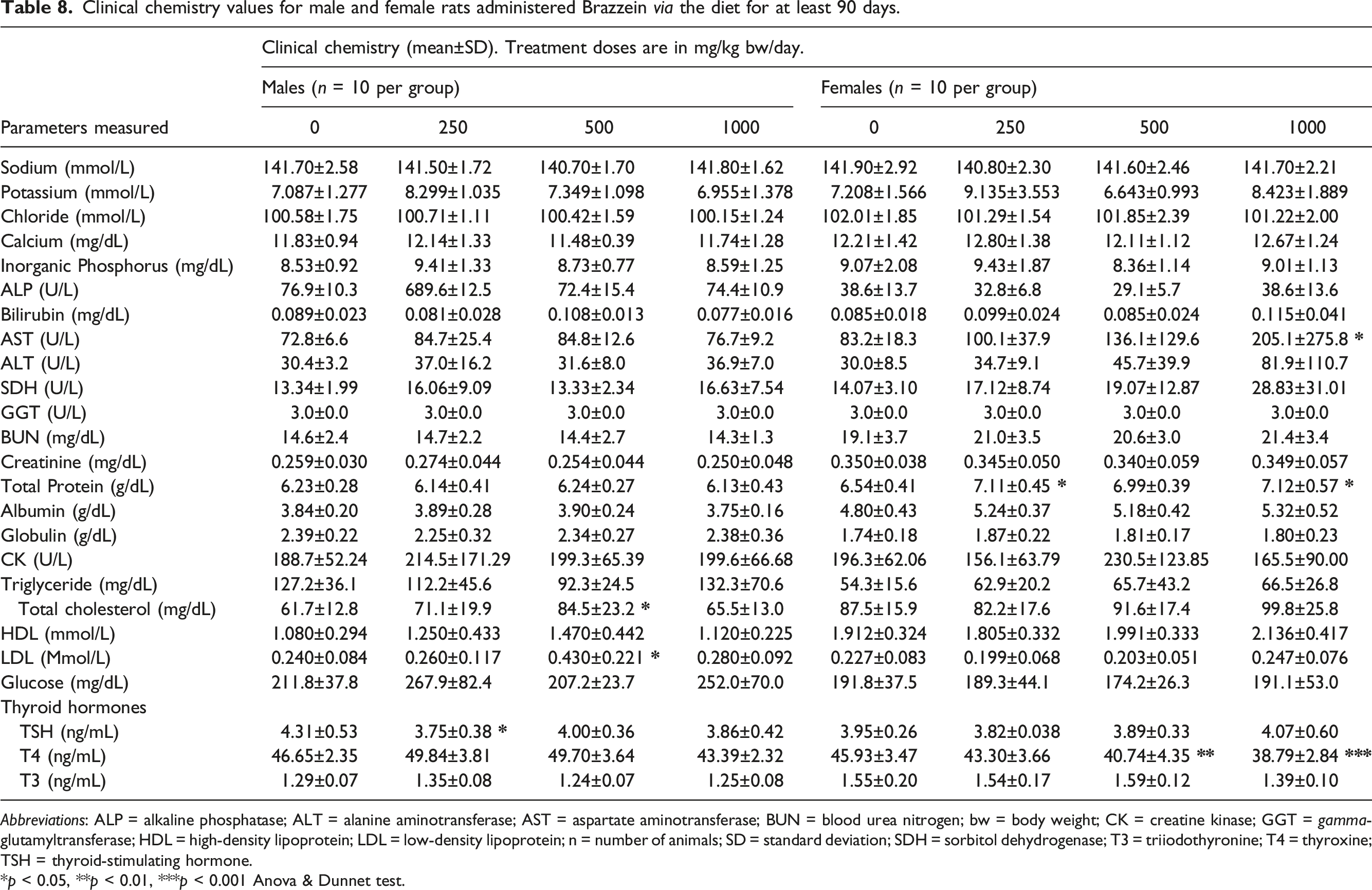
Abbreviations: ALP = alkaline phosphatase; ALT = alanine aminotransferase; AST = aspartate aminotransferase; BUN = blood urea nitrogen; bw = body weight; CK = creatine kinase; GGT = gamma-glutamyltransferase; HDL = high-density lipoprotein; LDL = low-density lipoprotein; n = number of animals; SD = standard deviation; SDH = sorbitol dehydrogenase; T3 = triiodothyronine; T4 = thyroxine; TSH = thyroid-stimulating hormone.
*p < 0.05, **p < 0.01, ***p < 0.001 Anova & Dunnet test.
Urinalysis values for male and female rats administered Brazzein via the diet for at least 90 days.
Abbreviations: bw = body weight; SD = standard deviation.
A few statistically significant changes were observed in the hematology and clinical chemistry parameters evaluated (see Tables 7 and 8, respectively). With respect to hematological parameters, statistically significant findings included: decreased mean cell hemoglobin and platelet counts in the mid- and high-dose females, a slight decrease in lymphocyte counts in high-dose females, and a slight increase in activated partial thromboplastin time (APTT) in high-dose females only. There were no statistically significant changes in any of the male dose groups.
Statistically significant changes in clinical chemistry parameters included: increased aspartate aminotransferase (AST) in high-dose females, slightly increased total protein in low- and high-dose females, and increased total cholesterol and low-density lipoprotein in mid-dose males. The AST value in the high-dose females remained within the historical control range (mean 86, range 42 to 341 U/L) and was not accompanied by any statistically significant increases in other enzymes associated with liver function (i.e., alanine aminotransferase [ALT], alkaline phosphatase [ALP], succinate dehydrogenase [SDH], and gamma-glutamyl transferase [GGT]). A few statistically significant changes in thyroid hormones were recorded, including a slight decrease in TSH in low-dose males and a slight decrease in T4 in the mid- and high-dose group females.
Organ weights for male and female rats administered Brazzein via the diet for at least 90 days.
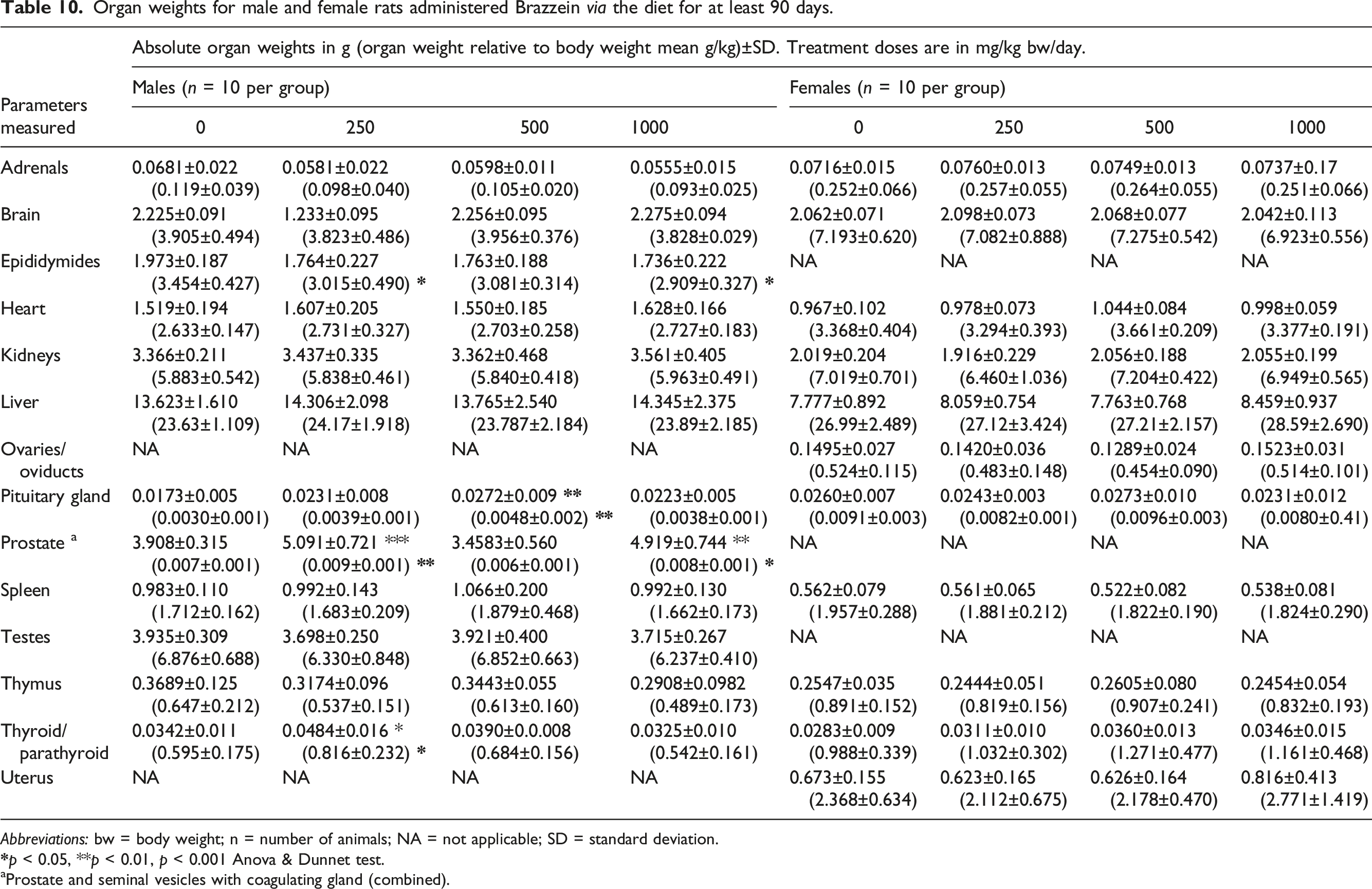
Abbreviations: bw = body weight; n = number of animals; NA = not applicable; SD = standard deviation.
aProstate and seminal vesicles with coagulating gland (combined).
The results of the macroscopic observations at necropsy were unremarkable between treatment groups and the incidence of histopathological lesions in all tissues examined was similar between the high-dose rats of both sexes and their respective controls. The few statistically significant changes reported from the FOB, hematology, clinical chemistry, and organ weight analyses did not correlate with any adverse histopathological findings.
Overall, there were no indications that brazzein treatment at up to 985 mg/kg body weight/day had any adverse effect on any of the parameters measured in the 90-day study. The no-observed-adverse-effect level (NOAEL) was considered to be 978 mg/kg body weight/day in males and 985 mg/kg body weight/day in females.
Discussion
In the in silico evaluation, the full-length amino acid sequence search identified one match sharing >50% identity with an E-value of 0.76 and bit-score of 24.6 with a putative allergen from peach (P. persica) from the AllergenOnline Database, and 20 matches sharing >50% identity with putative allergens from the Allermatch Database. The corresponding E-values and bit-scores of these 20 matches were greater than 1.1 and less than 24.1, respectively. The biological relevance of sequence homology matches is inferred by the identity match, E-value, and bit-score.51,52 Previous reports have suggested that cross-reactivity between proteins requires at least 70% identity, and an identity match of less than 50% is suggestive that cross-reactivity is rare. 59 The E-value and bit-score are used to investigate whether significant sequence homology exists, where an E-value of less than 0.001 and bit-scores greater than 40 can be reliably used to infer homology. 51 Recently, Abdelmoteleb et al. 52 demonstrated that an E-value less than 10−7 reflects a functional similarity between two proteins, and likely suggests a biologically relevant similarity for allergenic cross-reactivity potential. Therefore, considering the match with the pathogenic-related protein from peach had an E-value of 0.76 and bit-score of 24.6, while the E-values and bit-scores of the 20 matches from the Allermatch Database were greater than 1.1 and less than 24.1, respectively, these matches are not considered to be biologically or functionally significant, and are not suggestive of allergenic potential of brazzein. Using the in silico search guidelines recommended by the FAO/WHO and Codex Alimentarius, no matches were identified in the 80-amino acid sliding window search and 8-amino acid exact match. The overall findings suggest brazzein lacks the potential for allergenicity, and that the risk of allergenicity of brazzein when used under the conditions of intended use in food and beverage products is low.
The lack of evidence for genotoxicity in the two in vitro assays is consistent with the proteinaceous nature of the brazzein preparation tested. In the in vitro micronucleus test, a slight, but statistically significant increase was noted in the 4-hour treatment high-concentration (2000 µg/mL) group. However, this result was marginal and well within the historical control range; hence, it was not considered as a “positive” response. Proteins would not typically be expected to show genotoxicity activity. 60
The 90-day study showed no evidence of adverse effects of brazzein up to the highest dose tested of 985 mg/kg body weight/day.
The few statistically significant findings in the hematology and clinical chemistry data were not of toxicological significance. Specifically, for mean cell hemoglobin concentration, the decrease was modest in nature and not seen in males, and all values remained within the normal historical control range (mean 32.7, range 30.6 to 36.1 g/dL). The decrease in platelet count was considered of no toxicological significance, as in each group, including the controls, the values were actually above the historical control range (mean 1033.0, range 698.0 to 1641.0 × 103/μL) and no decreases were noted in males. Likewise, the slight increase in APTT time in high-dose females was slight, not seen in males, and not accompanied by any changes in prothrombin time.
An apparent dose-related increase in AST in the high-dose females was observed. However, the mean AST value in the high-dose females still remained within the historical control range (mean 86, range 42 to 341 U/L) and was not accompanied by any statistically significant increases in other enzymes associated with liver function (i.e., ALT, ALP, SDH, and GGT). In addition, there was no evidence of any increase in AST in males or changes in liver histopathology indicative of an adverse effect of treatment in either sex. Examination of the individual animal data showed that one female in the high-dose group had an abnormally high AST value at 980 U/L, and all others remained within historical control range, which resulted in the finding of the statistically significant increase in AST. There was no histological correlate in the liver for the animal with elevated AST. The changes in thyroid hormones were all modest in nature, occurred in only one sex, were not correlated with any other changes in thyroid parameters or histopathological findings, and remained close to historical control mean values. As a result, these changes were considered not treatment-related.
The slight decreases in epididymides weights (relative to body and brain weight), not always achieving statistical significance, were considered unrelated to treatment given that the decreases were minimal in nature and did not show clear dose responses, and given that the absolute organ weights were unaffected, the weights remained within historical control values, the testes weights were unaffected, and there were no correlates in any reproductive tissues found at either macroscopic or histopathological examination. All other statistically significant findings in organ weights were minimal in nature, showed no dose-response, did not correlate with any histopathological changes, and/or occurred only in one sex.
The results of the histopathological examinations found no treatment-related effects. Several changes were identified in various organs but were considered not test article-related due to sporadic occurrences, the lack of supporting changes to suggest a pattern or biological mechanism, and the fact that the microscopic findings consisted of lesions commonly seen in this age and strain of rat. 61 There was no effect of treatment on the distribution of the identified lesions with similar incidences in treated and control groups in both sexes.
There are no other standard toxicology studies on brazzein available in the scientific literature. One study has been conducted with mice to assess the potential anti-obesity effects of brazzein. 62 In this study, groups of seven male C57BL/6 J mice (7 weeks old) were exposed to brazzein containing mutations 31 R/E36D/E41 A (i.e., “3M-brazzein”) produced from Kluyveromyces lactis or aqueous solutions of 10% sucrose equivalents in their drinking water for a period of 15 weeks. A group of six control mice received water. The authors reported that relative to sucrose, body weight gain and fat accumulation were reduced. Moreover, Kim et al. 62 reported that administration of brazzein did not affect glucose homeostasis or indices of insulin resistance and inflammation. Nothing from this study suggests toxic potential of brazzein. The results of Kim et al. 62 are consistent with the lack of effect of brazzein on body weight gain, and overall lack of adverse effects, noted in our 90-day study of brazzein.
Beyond the series of toxicological studies, the safety of brazzein produced via fermentation could be influenced by the safety of the production strain. The safety of the production organism traditionally follows the principles of the safety assessment procedure for enzyme preparations, as described by Pariza and Johnson, 43 based on the primary consideration that there is availability of data and information substantiating that the production organism is from a safe lineage that has been the subject of previous toxicological evaluations. The safe strain lineage of strain NRRL Y-11430 has previously been reviewed by the FDA as part of Generally Recognized as Safe (GRAS) Notice (GRN) 737, GRN 967, and GRN 1001, demonstrating its non-pathogenic and non-toxigenic properties, and its safe and suitable use as a source organism for the production of food ingredients.39,40,63 All changes introduced into the production strain are stably integrated in the genome, and the production strain does not contain any antibiotic resistance genes or plasmid sequence. Therefore, it is unlikely to pose any risk of transferring antibiotic resistance to non-related organisms.
Proteomics assessment of the brazzein demonstrated the presence of only six residual host cell proteins from the production strains at levels of greater than 1% up to 5.6% based on the total protein spectrum count (data not shown). The proteins include a fusion protein (UniProt No. C4R0U2), transaldolase (UniProt No. C4R245), vacuolar aspartyl protease (UniProt No. C4R6G8), peptide hydrolase (UniProt No. C4QYZ6), chitin deacetylase (UniProt No. C4QW42), and S-adenosylmethionine synthase (UniProt No. C4R5U7). In silico assessment of these residual K. phaffii proteins demonstrated that residual host cell proteins from the production strain are non-toxigenic and of low allergenic potential for cross reactivity to major food allergens. Therefore, any native residual proteins from the K. phaffii production organism would not pose a safety concern with respect to allergenicity or toxigenicity; these findings are consistent with those reported by Jin et al. 64 and Reyes et al. 65 for the same host organism.
Conclusion
Oubli fruit sweet protein, also referred to as brazzein, was evaluated in a series of in silico allergenicity and in vitro and in vivo toxicology studies. The results of these investigations provide data to indicate that brazzein is not allergenic. In addition, these data show brazzein to be non-genotoxic, and to have no adverse effects following the 90-day oral dietary treatment of Sprague-Dawley rats at doses of up to 985 mg/kg body weight/day (i.e., the NOAEL). Under the conditions of intended use, the NOAEL for the 90-day study is more than 340-fold higher than the total population in the 90th percentile (consumer only) for estimated daily intake (EDI) of 2.88 mg/kg body weight/day derived from exposure estimates using the NHANES database. The NOAEL is also more than 200-fold higher than the EDI (4.25 mg/kg body weight/day) for the highest-exposure group in the 90th percentile (young children, ages 2–5). The strain used to produce brazzein via fermentation was found to be safe for its intended use and does not pose any safety concerns. On the basis of the preceding, Oubli fruit sweet protein (brazzein) is considered safe for use as a sweetener in food and beverages, offering a new approach to sweetness for millions of people actively working to reduce sugar in their diets to improve their health.
Footnotes
Acknowledgements
The authors would like to acknowledge YH. Chiang, D. Ellison, S. Rodriguez, T. Salvucci, and C. Gabotero for test material production and L. Huskins for analytical chemistry support; N. Sachs and A. Wing for critical evaluation of the manuscript; N. Rainer and M. Bauter for assistance in study design.
Author Contributions
Barry Lynch contributed to methodology, validation, formal analysis, drafting and critically revising the manuscript; Tina Wang contributed to conception and design, methodology, investigation, resources, drafting and critically revising the manuscript, supervision, and project administration; Trung Vo contributed to formal analysis, data curation and investigation; Shahrzad Tafazoli contributed to conceptualization, resources, drafting and critically revising the manuscript; Jason Ryder contributed to conceptualization, resources, and critically revising the manuscript.
Declaration of conflicting interests
The author(s) declared potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tina Wang and Jason Ryder are employees of Oobli, Inc. Intertek Health Sciences, Inc. received financial support from Oobli, Inc. to assist in the preparation of the manuscript.
Funding
Oobli, Inc. is the only source of funding for this study. Oobli, Inc. is responsible for the study design, the data collection, analysis, and interpretation, and the decision to submit the article for publication.
Data availability
Data generated and/or analyzed during the current study are available from the authors upon request.
