Abstract
Background:
Metformin-induced vitamin B12 deficiency state or metformin-induced hypocobalaminemia is gradually becoming an epidemic among diabetic patients on moderate-to-high doses of metformin or those diabetic patients on metformin for a long period of time. The potential effect of chronic metformin pharmacotherapy to cause vitamin B12 deficiency with abnormalities in haematologic indices and central/peripheral neuropathy has been widely reported. Long-term usage of metformin has been reported to be associated with intestinal malabsorption of vitamin B12 culminating in vitamin B12 deficiency with likely associated haematologic abnormalities (including macro-ovalocytic anaemia and immune dysfunctioning due to hypersegmentation of polymorphonuclear leukocytes), central/peripheral neuropathy and manifestation of biochemical derangements such as elevated homocysteine and methyl malonate levels.
Aim:
This study aimed to determine the correlation between serum vitamin B12 levels and various haematologic indices among metformin-treated type 2 diabetic patients in a clinical practice setting with the rational purpose of alleviating/preventing the associated derangements.
Materials and Methods:
This was a case-control, prospective, analytical, observational study of 200 adult participants (100 per group) attending the Endocrinology Out-patients Clinic of Irrua Specialist Teaching Hospital, Irrua, Edo State, Nigeria. For each participant, serum vitamin B12 level was determined using a vitamin B12 immunoassay technique, while the corresponding complete blood count was done using PCE-210N autohaematology analyser. Data were presented using tables and figures. Chi-square test was used to compare categorical variables, Student t-test was used in comparing means of continuous variables, while Pearson’s correlation study was done to determine the existence of any statistically significant correlation(s) between the serum vitamin B12 levels and various haematologic indices among the participants.
Results:
Approximately 41% versus 20% of the metformin-treated and metformin-naive diabetic patients, respectively, had frank vitamin B12 deficiency. There was a statistical difference between the total serum vitamin B12 levels in male and female diabetic patients with p = 0.048. Also, statistically significant differences existed with respect to mean corpuscular volume (MCV), mean corpuscular haemoglobin and total white blood cells count among the metformin-treated and metformin-naive diabetic patients. Furthermore, a statistically significant weak positive correlation existed between pack cell volume (PCV) and serum vitamin B12 level (r = +0.148, p = 0.037), but a statistically significant weak negative correlation existed between MCV and serum vitamin B12 level (r = −0.245, p = 0.0001). In addition, the test for associations between the serum vitamin B12 categorization status or metformin exposure status and the peripheral neuropathy components assessment revealed that there were statistically significant associations between the serum vitamin B12 categorization status or metformin exposure status versus pain sense (p < 0.0001 or <0.001), vibration sense (p < 0.0001 or <0.001) and light touch sense (p < 0.0001 or <0.001) among the participants.
Conclusion:
In this study, statistically significant weak positive and weak negative correlations existed between serum vitamin B12 level versus PCV, and serum vitamin B12 level versus MCV, respectively. The peripheral neuropathy components assessment revealed that there were statistically significant associations between the serum vitamin B12 categorization status or metformin exposure status versus pain sense, vibration sense and light touch sense among the participants.
Keywords
Introduction
Metformin-induced vitamin B12 deficiency state or metformin-induced hypocobalaminemia is gradually becoming an epidemic among diabetic patients on moderate-to-high doses of metformin or those diabetic patients on metformin for a long period of time. The potential effect of chronic metformin pharmacotherapy to cause vitamin B12 deficiency with abnormalities in haematologic indices and central/peripheral neuropathy has been widely reported. Long-term usage of metformin has been reported to be associated with intestinal malabsorption of vitamin B12 culminating in vitamin B12 deficiency with likely associated haematologic abnormalities (including macro-ovalocytic anaemia and immune dysfunctioning due to hypersegmentation of polymorphonuclear leukocytes), central/peripheral neuropathy and manifestation of biochemical derangements such as elevated homocysteine and methyl malonate (MMA) levels. 1,2
The metformin-induced vitamin B12 deficiency state, also known and referred to as metformin-induced hypocobalaminemia (due to metformin-vitamin B12 interaction) belongs to the category form of mild/minor (type B) drug–nutriceutical interaction whose medico-clinical significance is acutely unknown, until when taken chronically for more than average of 5 years before gradual decrease in plasma vitamin B12 level will begin to ensue as a result of hepatic and muscular storage sites depletion. It is also a form of mild adverse drug reaction (ADR) whose occurrence is duration of use-dependent (type C/chronic use ADR) and onset prolongation-dependent (type D/delay-onset ADR).
According to the year 2014 annual report of the International Diabetes Federation, the estimated prevalence of diabetes mellitus (DM) was 4.6% in Nigeria. 1,2 In 2016, an estimated 422 million adults globally are said to be living with DM according to World Health Organization. 3
The management of type 2 DM involves the use of several oral hypoglycaemic agents; however, metformin is widely used as a first-line pharmacotherapeutic agent in conjunction with dietary modifications. 4 Despite having these benefits, it has been implicated to cause vitamin B12 deficiency as a result of chronic administration, thereby contributing to the problems of DM patients by leading to clinical manifestations of vitamin B12 deficiency such as macro-ovalocytic (megaloblastic) anaemia, immune dysfunctioning, distal sensory polyneuropathy and cognitive impairment. 5
Metformin is currently one of the first-line pharmacotherapeutic agents used for the treatment of type 2 diabetes. It belongs to the biguanide class of antidiabetic drugs. 6,7 Metformin has been found to significantly improve peripheral insulin sensitivity, blood glucose control via decrease hepatic gluconeogenesis, and has been associated with decreased cardiovascular mortality, the major cause of death among patients with diabetes. 8 Metformin has a good safety profile and limited side effects although early discontinuations due to gastrointestinal intolerance occur in up to 20% of patients. Metformin has been reported to lower vitamin B12 levels in a dose-dependent manner and based on duration of use. 9 But, the precise mechanism by which metformin causes vitamin B12 deficiency has not been clearly elucidated; however, proposed mechanisms include alterations in small bowel motility due to calcium deficiency which stimulates bacterial overgrowth and consequential vitamin B12 malabsorption. Metformin has also been shown to inhibit the calcium-dependent endocytosis active transportation absorption process of vitamin B12-intrinsic factor of castle (vitamin B12-IF) complex at the terminal ileum. 10 –13 This inhibitory effect is reversed by oral co-administration of calcium supplement. 14
A study done in Ibadan, Nigeria, found a prevalence of vitamin B12 deficiency in metformin-treated type 2 DM patients to be 8.6%. 5 The above study at Ibadan, Nigeria, however, did not evaluate for the presence of haematologic indices derangements associated with vitamin B12 deficiency which this study will thoroughly investigate and correlate. In sub-Saharan Africa, including Nigeria, there has been an increase in the prevalence rates of DM and vitamin B12 deficiency in metformin-treated type 2 DM patients. 4 –6
Vitamin B12 or cobalamin is a water-soluble vitamin that plays a very essential role in DNA synthesis, optimal haematopoiesis and neurological function. 4 Several studies have obviously demonstrated that vitamin B12 deficiency is highly prevalent in chronic metformin-treated adult diabetic patients. 5,12,13 The most notable risk factor associated with vitamin B12 deficiency in adult individuals with diabetes in clinical practice is long-term and high dose metformin therapy. 6,7,13 Clinically overt and untreated vitamin B12 deficiency has been associated with neurological impairments such as disabling sensory polyneuropathy and subacute degeneration of the spinal cord which may clinically mimics diabetic neuropathy, anaemia, depression and/or neurodegeneration disorders-induced cognitive impairment. 7,8 Vitamin B12 deficiency is associated with a significant increase in serum homocysteine and MMA concentrations. 7 Hyperhomocysteinemia may negatively impact diabetic patients health, as elevated homocysteine levels are associated with an increased risk of cardiovascular disease (atherosclerosis), 8 independently of classic cardiovascular risk factors. 9 In type 2 diabetes, homocysteine concentrations higher than 15 μmol/L have been correlated to an increased cardiovascular risk 10 and seem to be a higher risk factor for mortality than in subjects without diabetes. 11 Consequently, the fact that chronic metformin pharmacotherapy may raise homocysteine levels through vitamin B12 deficiency, 12,13 particularly in the absence of exogenous supplementation of folic acid or B-group vitamins, 14,15 could worsen its associated cardiovascular, haemopoietic and neurological morbidity. Despite these above-mentioned clinical conditions associated with vitamin B12 deficiency and its high degree of occurrence in adult individuals with diabetes, there are neither global nor national guidelines recommending the routine assessment of serum vitamin B12 levels, especially in Nigeria, a nation with a high prevalence of nutritional deficiencies, as there are poor availability of data on the burden and predictors of vitamin B12 deficiency in adult individuals with DM. 15 –17
In fact, frank serum vitamin B12 deficiency has been found to manifest with laboratory picture of haematologic abnormalities such as anaemia, macro-ovalocytosis and hypersegmented neutrophils. 17 –20 Briefly, pack cell volume (PCV) is a measure of the proportion of red blood cells and haemoglobin (Hb). When PCV value is low, a patient is said to be anaemic. Considering the expensive cost of regular screening using vitamin B12 assay as a limiting factor, the full blood count with peripheral microscopic blood film smears can be done frequently in type 2 DM patients on long-term metformin therapy to detect macro-ovalocytic anaemia and hypersegmented polymorphs followed by occasional biochemical vitamin B12 assay analysis periodically in rural areas. 21 –24
Metformin causing vitamin B12 deficiency among type 2 diabetic patients has become a traditional myth in medical literatures. 10 –14,25,26 We have learnt that not all low plasma vitamin B12 level actually means ‘an absolute deficiency state’ and not all high plasma vitamin B12 level actually means ‘an absolute sufficiency state’. Furthermore, what this aforementioned statement implies is that: ‘borderline serum vitamin B12 level alone without disturbances in the metabolic markers has no significant diagnostic value. In contrast, higher serum vitamin B12 level in metformin-naïve type 2 diabetic patients may give the wrong impression that vitamin B12 deficiency is less common among type 2 diabetic patients’. 25,26
When assimilated vitamin B12 molecule is internalized and is able to regulate and maintain the vitamin B12-dependent cellular enzymes’ activities (i.e. methionine synthase and methyl malonyl-CoA mutase enzymatic functions), the plasma total homocysteine (tHcy) and MMA levels will be within the normal reference range. This revolution in diagnosing vitamin B12 deficiency state has provided convincing evidence that metformin is not as bad as it is being reputed. 25,26
All available evidences suggest that with chronic metformin pharmacotherapy, there is lowering of plasma vitamin B12 levels (and if not associated with increased metabolite markers) would mean increased cellular uptake of vitamin B12. In line with this view, it was recently shown that animals treated with metformin accumulate more vitamin B12 molecules in their liver tissues, which may help explain the observed lowering effect on plasma vitamin B12 levels. 25,26
This study was designed to correlate the serum vitamin B12 levels with the various haematologic indices among metformin-treated type 2 diabetic patients in a clinical practice setting with the rational purpose of alleviating/preventing the associated derangements.
Materials and methods
Study location and population
This study was conducted at the Irrua Specialist Teaching Hospital (ISTH), Irrua. It is a Tertiary Teaching Hospital located at Irrua town, Esan Central Local Government Area in Edo State, Nigeria. The hospital is at a distance of about 87 km north of Benin City, the Edo state capital. The ISTH is a 343-bed capacity hospital involved in the management of patients and training of medical students, resident doctors and nursing students. The Endocrinology clinic holds once a week with about 46 diabetic patients seen per clinic day. The study population comprised of adult medical patients with type 2 DM attending the Endocrinology Out-patients Clinic at ISTH, Irrua, Edo State. Their ages were ≥30 years and were fulfilling the inclusion criteria for recruitment, evaluation and followed up.
Inclusion and exclusion criteria
The inclusion criteria for evaluation in this study were known adult medical patients with type 2 DM attending the Endocrinology Out-patients Clinic at ISTH, Irrua, Edo State after commencing the study provided they granted their informed consents to participate in the study and were ≥30 years of age. They also fulfilled the diagnostic inclusion criteria used to define type 2 DM according to the American Diabetes Association (ADA).
The exclusion criteria for this study were as follow: Known type 1 DM patients. Diabetic patients <30 years of age whose diagnosis of whether type 1 DM or type 2 DM were highly ambiguous and uncertain. Patients who do not fulfilled the diagnostic inclusion criteria used to define type 2 DM according to the ADA. Adult type 2 DM patients who did not grant their informed consents to participate in the study. Adult type 2 DM patients who at the time of recruitment for the study were pregnant women, purely vegetarians, receiving vitamin B12 containing supplement or chronic alcoholics (>21 units and >14 units per week for male and female for >10 years, respectively), patients taking cytochrome P450 microsomal enzyme inducer/inhibitor substances such as omeprazole and other proton pump inhibitors, rifampicin, barbiturates, cimetidine, grapefruit juice, ritonavir, ketoconazole, carbamazepine or phenytoin, patients taking neomycin or having comorbidities such as undernutrition (BMI < 18.5 kg/m2), tuberculosis, benign/malignant neoplasms on chemotherapy, human immunodeficiency virus infection/acquired immunodeficiency syndrome, chronic liver disease, chronic kidney disease/acute kidney injury with estimate glomerular filtration rate < 60 mL/min/1.73 m2, history of irritable bowel syndrome, pernicious anaemia or inflammatory bowel disorders such as Crohns disease or ulcerative colitis. Adult type 2 DM patients who had done previous surgeries such as bariatric surgery, partial/total gastrectomy or partial/total ileal resection. Patients without DM taking metformin for other medical reasons/conditions such as polycystic ovarian syndrome or insulin resistance syndrome.
Study design
This was a case-control, prospective, analytical, observational study.
Sample size determination
The formula for estimating sample size is:
N = [Z 2 × P × (1 − p)/e 2]; where N = required sample size, and Z is the critical value at 95% confidence level = 1.96.
P = prevalence of vitamin B12 deficiency in patients with type 2 DM on metformin obtained from a previous study 5.8%, 5 e = error margin that the investigator was willing to accept and in this instance = 0.05. Note: (1 − p) = q, this was the proportion of the sample population not covered by the study.
n = 1.96 × 1.96 × 0.058 (1 − 0.058)/0.05 × 0.05; n = 84; n = 97 (with 15% attrition). This was rounded up to 100 patients for each group.
Sampling method
Consecutively consented adults with type 2 DM who met the inclusion criteria for cases and controls were selected from the outpatient clinic at ISTH until the desired sample size was achieved. Both cases and controls were matched for age and sex.
Clinical examination
A structured questionnaire was administered to the selected participants by the researchers. The structured questionnaire contained information about socio-demographic characteristics, possible co-morbidity, surgery, alcohol ingestion, diet (vegetarian or not vegetarian), and symptoms of anaemia and gastrointestinal symptoms associated with vitamin B12 deficiency, medication information including the dose and duration of therapy.
All participants were made to undergo thorough clinical examination in order to document the presence of relevant haematological signs. Evidence of Pallor was examined for in areas like the conjunctiva, oral mucosa and palms.
Subjects’ preparation and sample collection
The laboratory assessments comprised of baseline haematologic parameters (complete blood count (CBC)) and serum vitamin B12 level. Venepuncture site was cleaned with 70% methylated spirit and allowed to dry. Ten millilitres of whole blood was collected from the antecubital vein of each participant. Of this, 6 mL of venous blood was collected into a labelled vacutainer sample bottle coated inside with potassium ethylene diamine tetraacetate for CBC and peripheral blood film (PBF). The remaining 4 mL of whole blood was dispensed into a labelled plain vacutainer sample bottle, allowed to clot, centrifuged for 15 min at 3000 r/min and separated into another plain bottle for vitamin B12 assay. Freshly prepared sera were stored in aliquots of at least 2 mL at a temperature of −20°C and pooled until the desired sample size was met for analysis. Repeated freezing/thawing cycles were avoided due to constant power supply at the storage unit.
Baseline haematologic parameters
CBC, including red cell indices were obtained using an autohaematology analyser which requires blood sample drawn by the instrument (PCE-210N ERMA Inc. Tokyo, Japan). Blood sample collection and preparation of PBFs were done. Viewing of PBFs was done by haematologists. The standard reference intervals for CBC parameters are as follow: Hb = 13–18 g/dL male, 11.5–16.5 g/dL female, PCV = 40–52% male, 36–48% female, mean corpuscular volume (MCV) = 77–95 fL, mean corpuscular haemoglobin (MCH) = 27–32 pg, mean corpuscular haemoglobin concentration (MCHC) = 32–36 g/dL, total white blood cells (WBCs) count = 4.0–11.0 × 103 cells/μL, neutrophil = 2.0–7.5 × 103 cells/μL, lymphocytes = 1.5–4.5 × 103 cells/μL, eosinophils = 0.04–0.4 × 103 cells/μL, basophils = 0.0–0.1 × 103 cells/μL and platelets = 150–400 × 103 cells/μL.
Preparation and staining of PBFs
First, a spreader was made from a glass slide that had a smooth end by breaking the corner of the slide leaving a width of 18 mm as the spreader. 16 A drop of blood was placed using a capillary tube at the centre of another glass slide that was dried and degreased, about 1 cm from one end. And without delay, the spreader was placed in front of the drop of blood at an angle of about 30° to the slide, it was then moved back to make contact with the drop of blood. The drop spreads out quickly along the line of contact. With a steady movement of the hand, a spread of the drop of blood along the slide was made. Then the slide was left to dry. The blood film was immediately labelled after spreading with a grease pencil in the 1 cm space at the end of the slide. The air dried blood smear was fixed for 15 min in 100% methanol. It was then transferred without blotting to a staining jar containing May–Grünwald stain freshly diluted with an equal volume of buffer solution (phosphate buffer pH 7.2) for 15 min. The slide was allowed to dry on a filter paper without blotting, and then transferred into a jar container followed by the placement of Giemsa staining solution freshly diluted with 9:1 volume ratio of phosphate buffer at pH 7.2 for 30 min. After, it was transferred into a phosphate buffer solution (pH 7.2) and agitated for 10–20 s, then removed. The slide was allowed to dry in a vertical position. The film was viewed by haematologist under a light microscope to assess the red and white cells morphology using 40× microscope objective lens, while the proportion of platelets was viewed with 100× microscope objective lens.
Measurement of serum vitamin B12 levels
Vitamin B12 ELISA assay kit, Cat. No. EE-L-0010, Lot AK0017APRO1014 Elabscience Biotechnology Company Ltd, China, was used for the measurement of serum vitamin B12 levels. 20 The vitamin B12 quantitative test is based on the principle of the enzyme linked immunosorbent assay. The lower reference limit for healthy state serum vitamin B12 levels depends on the technique used for the bioassay and it was set at 200 pg/mL (148 pmol/L) during this study in similarity and comparison to the other previous studies done. 20
Serum vitamin B12 levels less than 200 pg/mL is diagnostic of vitamin B12 deficiency, values greater than or equal to 400 pg/mL confirm absence of vitamin B12 deficiency, while values from 200 to 399 pg/mL are considered as borderline vitamin B12 status levels. 20
Ethical consideration
Ethical approval for this study was obtained from the Health Research Ethical Committee of ISTH and the assigned protocol identification number was ISTH/HREC/2016/March/026. Duly written informed consents were obtained from all cases and controls. These written informed consents were documented in the respective medical case file for each of these patients and then counter signed by each of them for appropriate record purpose. All the study participants were counselled before and after clinical assessment and serum vitamin B12 screening. In addition, they were assured of the confidentiality of all their records. This study also served to notify and treat those participants with vitamin B12 deficiency and borderline vitamin B12 status.
Data analysis
The statistical package software for social sciences (SPSSR) version 21.0 was used for data entry, validation and analysis. Frequency distribution tables and figures were generated for the categorical variables. Tests of significance were generated for different variables. Logistic regression was used to assess the relationship between vitamin B12 deficiency and independent variables in metformin participants. Data were examined for normality. Chi-squared test was used to test categorical variables, independent t-tests was used to test the significance of means between two groups, while Pearson’s correlation study was done to determine the relationship between continuous variables. The level of statistical significance was defined by p < 0.05.
Results
A total number of 200 participants, comprising 100 metformin-treated and 100 metformin-naive type 2 DM patients, were recruited into this study from November 2016 to September 2017. The socio-demographic parameters include age with its categorization and gender distribution. The age of the participants ranged from 31 to 80 years with a mean of 55.80 ± 9.3 years. Majority of the participants; 39 (39%) for metformin-treated type 2 DM patients and 37 (37%) for metformin-naive type 2 DM patients were between the age group of 51–60 years. Both groups were matched for age and gender. This was represented in Table 1 alongside with other socio-demographic profiles of the participants. Furthermore, Table 1 showed that 49% versus 51% among the metformin-treated group were males and females, respectively; the opposite was recorded for the metformin-naive group. Majority of the participants were married (96%) and income less than 100 dollars a month. The marital status is meant to assess the social or financial support/well-being of the respondents; as low income may be a contributing factor and reason for nutritional deficiency/malnutrition in this study.
Socio-demographic profiles of the metformin-treated and metformin-naive type 2 DM study groups.
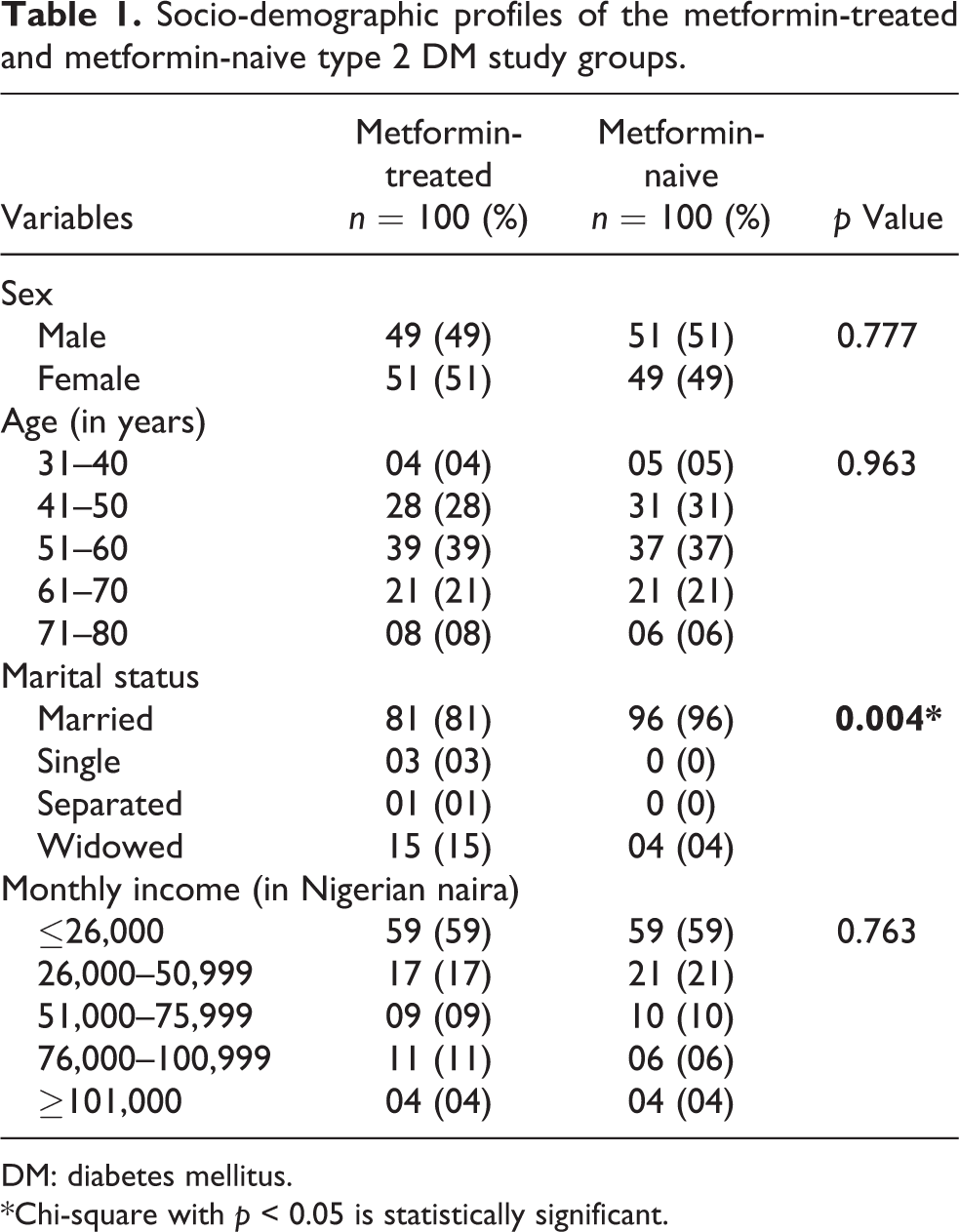
DM: diabetes mellitus.
*Chi-square with p < 0.05 is statistically significant.
Table 2 below shows the anthropometric and socio-demographic parameters of study participants. The mean for age, height and weight were higher for male participants when compared with their female counterparts (58.8 ± 8.9 years versus 56.1 ± 9.4 years; p = 0.316), (1.62 ± 0.08 m versus 1.59 ± 0.07 m; p = 0.512), (67.5 ± 8.9 kg versus 66.7 ± 8.8 kg; p = 0.408) for males and females, respectively. However, the reverse was observed concerning the mean for BMI (25.7 ± 2.8 kg/m2 versus 26.4 ± 3.6 kg/m2; p = 0.491), duration of DM diagnosis (4.92 ± 4.5 years versus 5.14 ± 4.9 years; p = 0.705) for males and females, respectively, although all their p values were not statistically significant.
Comparison of the anthropometric and socio-demographic parameters of the study participants based on gender difference.
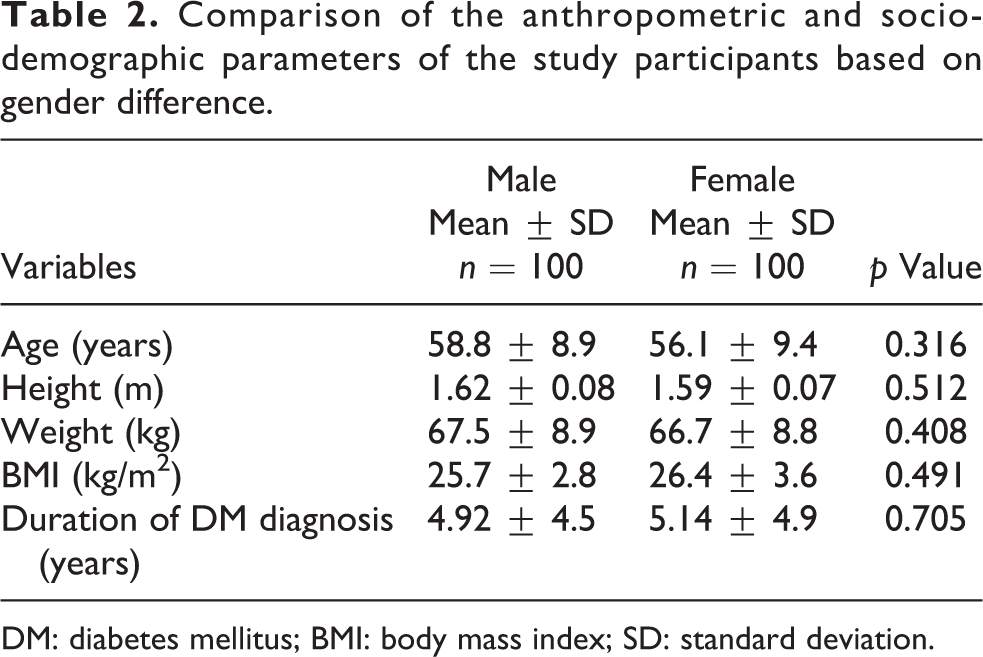
DM: diabetes mellitus; BMI: body mass index; SD: standard deviation.
Table 3 summarizes the haematologic parameters, total serum vitamin B12 levels and fasting blood glucose values for male and female participants at recruitment contact during this study. The mean MCH for males is 28.7 ± 1.42 pg versus 28.0 ± 1.3 pg for females with p = 0.025, mean serum vitamin B12 level was slightly lower for male participants 234.9 ± 79.2 pg/mL versus 247.0 ± 78.8 pg/mL for females with p = 0.048. This revealed statistically significant differences for the mean MCH and the mean serum vitamin B12 levels with respect to gender (sex) for the participants. Furthermore, the mean fasting plasma glucose (FPG) levels appear to be better controlled for males compared to females 132.9 ± 43.2 mg/dL versus 135.6 ± 58.1 mg/dL with p = 0.470, but this was not statistically significant. Other parameters such as PCV (34.8 ± 6.4% versus 33.8 ± 5.9%; p = 0.492), Hb (11.74 ± 2.2 g/dL versus 11.4 ± 2.2 g/dL; p = 0.506), WBC (5210 ± 2386 cells/µL versus 5800 ± 3700 cells/µL; p = 0.781), neutrophil (59 ± 16% versus 56 ± 17.5%; p = 0.160), lymphocytes (43 ± 17.38% versus 41 ± 17.88%; p = 0.382), platelets (214,000 ± 98,919 cells/µL versus 246,000 ± 106,000 cells/µL; p = 0.078), MCV (83.6 ± 7.7 fL versus 83.1 ± 8.1 fL; p = 0.455), MCHC (33.4 ± 2.4 g/dL versus 34.6 ± 2.0 g/dL; p = 0.673) for males and females, respectively, did not show any statistically significant differences as none had their p values <0.05.
Various haematologic parameters and FPG levels at recruitment contact among the study participants based on gender difference.
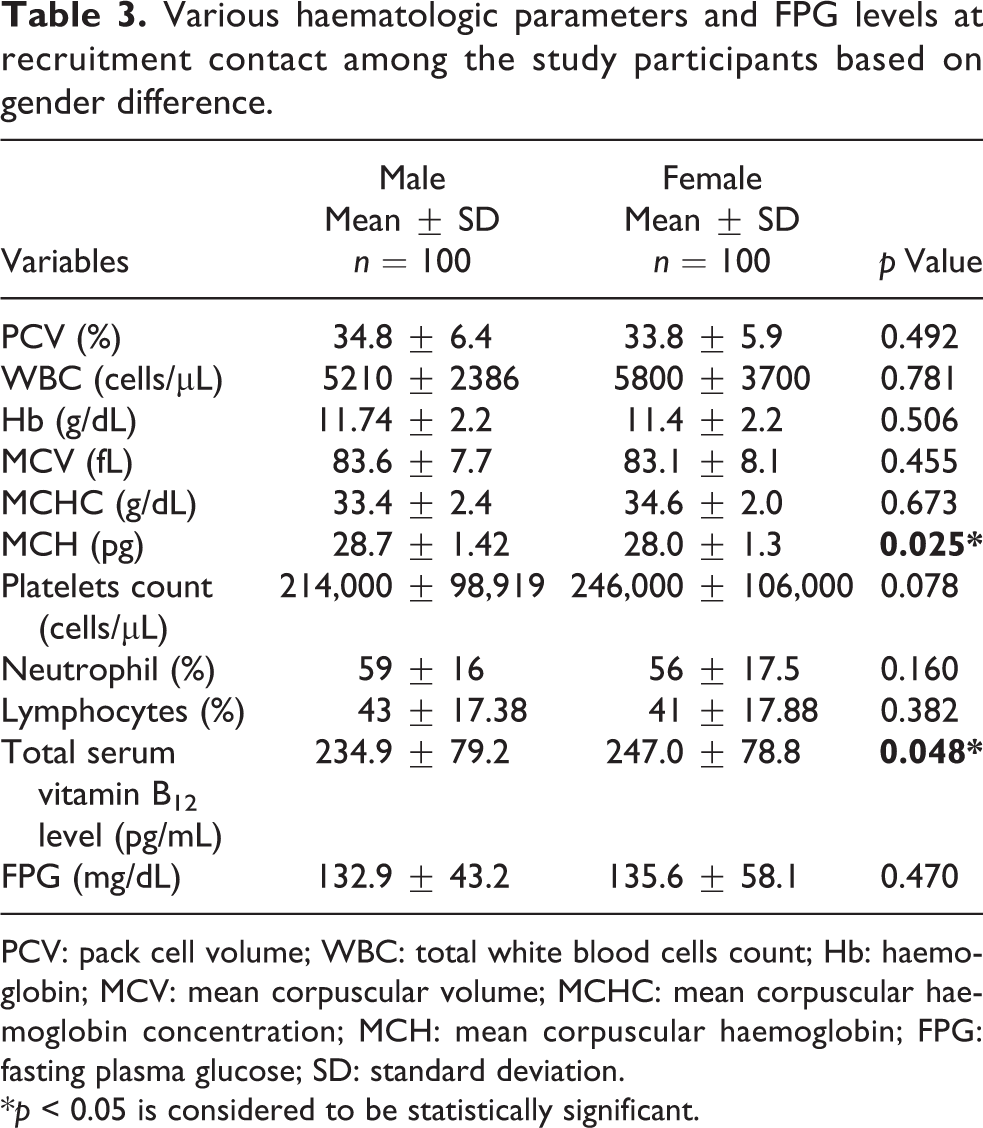
PCV: pack cell volume; WBC: total white blood cells count; Hb: haemoglobin; MCV: mean corpuscular volume; MCHC: mean corpuscular haemoglobin concentration; MCH: mean corpuscular haemoglobin; FPG: fasting plasma glucose; SD: standard deviation.
*p < 0.05 is considered to be statistically significant.
The prevalence rates for frank vitamin B12 deficiency state among the metformin-treated and metformin-naive type 2 diabetic patients were 41% and 20%, respectively, which revealed a statistically significant difference between both groups (p < 0.001). Furthermore, the prevalence rates for frank anaemia (PCV value < 30% with Hb concentration < 10 g/dL) among the metformin-treated and metformin-naive type 2 diabetes patients were 29% and 14%, respectively, which revealed a statistically significant difference between both groups (p < 0.05).
Table 4 shows frank vitamin B12 deficiency levels and various haematologic parameters among the metformin-treated and metformin-naive participants at recruitment contact during this study. The mean serum vitamin B12 levels among the 41 (41%) patients in metformin-treated group and 20 (20%) patients in metformin-naive group with frank vitamin B12 deficiency state (i.e. mean serum vitamin B12 level ≤ 199 pg/mL) were 158.29 ± 29.27 pg/mL and 173.95 ± 14.21 pg/mL, respectively (p = 0.028). This was significantly lower for the metformin-treated group compared to metformin-naive group with respect to the participants with frank vitamin B12 deficiency state. There were statistically significant differences in the mean for MCV (85.04 ± 7.83 fL versus 81.63 ± 7.70 fL; p = 0.01), MCH (28.50 ± 1.37 pg versus 28.10 ± 1.28 pg; p = 0.02) and WBC (5000 ± 2775 cells/µL versus 4150 ± 2450 cells/µL; p = 0.01) between both participants. But, there were no significant statistically differences between both participants with respect to their mean Hb concentration, PCV, MCHC, Neutrophils, lymphocytes and platelets count; as none had their respective p values <0.05.
Frank vitamin B12 deficiency levels and various haematologic parameters among the metformin-treated and metformin-naive study groups.
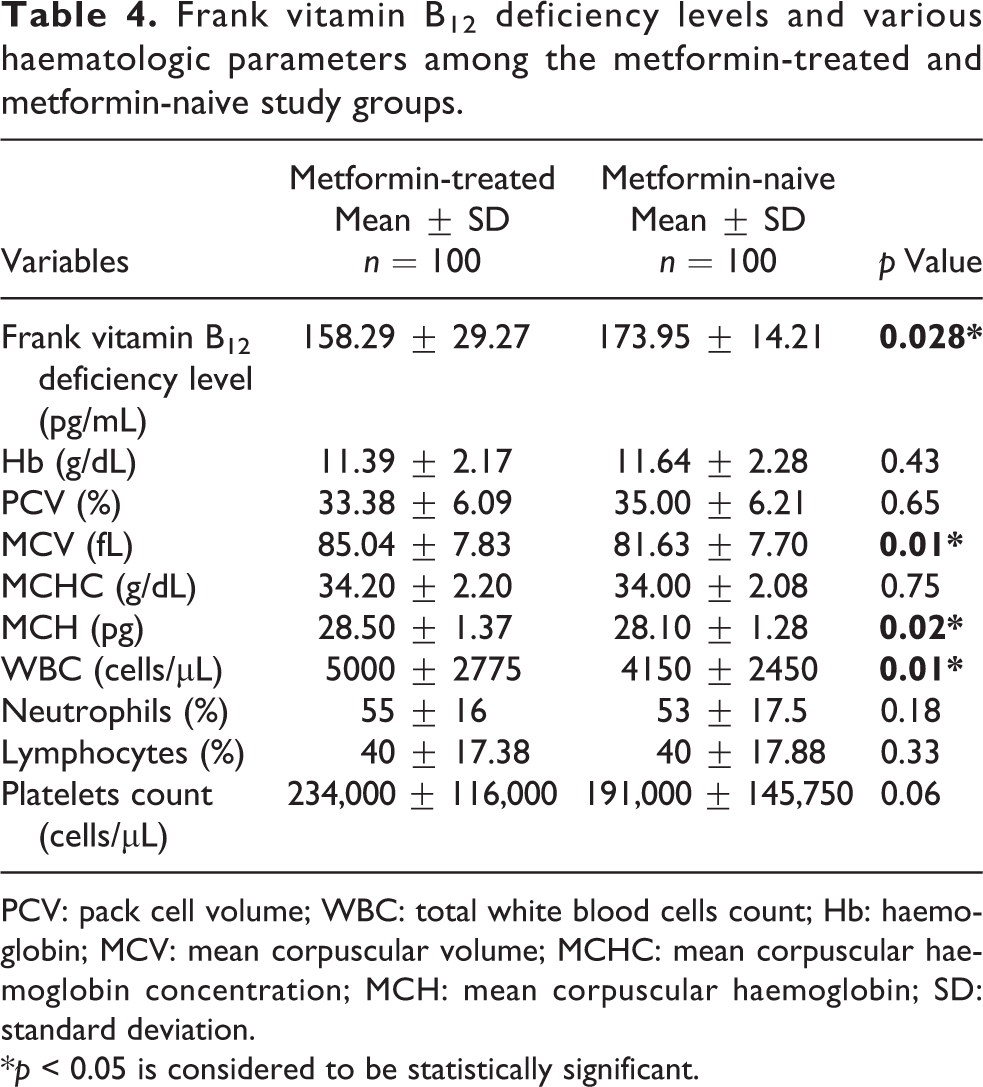
PCV: pack cell volume; WBC: total white blood cells count; Hb: haemoglobin; MCV: mean corpuscular volume; MCHC: mean corpuscular haemoglobin concentration; MCH: mean corpuscular haemoglobin; SD: standard deviation.
*p < 0.05 is considered to be statistically significant.
Table 5 categorically depicts the correlation between serum vitamin B12 levels and various haematologic parameters among the study participants. There was a statistically significant weak positive correlation between serum vitamin B12 levels and PCV (r = +0.148, p = 0.037). But, there was a statistically significant weak negative correlation between serum vitamin B12 levels and MCV (r = −0.245, p = 0.0001). There were no statistically significant correlations between serum vitamin B12 levels and other various haematologic parameters such as WBC (r = −0.045; p = 0.525), platelets count (r = −0.047; p = 0.524) or Hb concentrations (r = +0.087; p = 0.222); as their respective p values were more than 0.05.
Correlation of serum vitamin B12 levels with various haematologic parameters among the study participants.
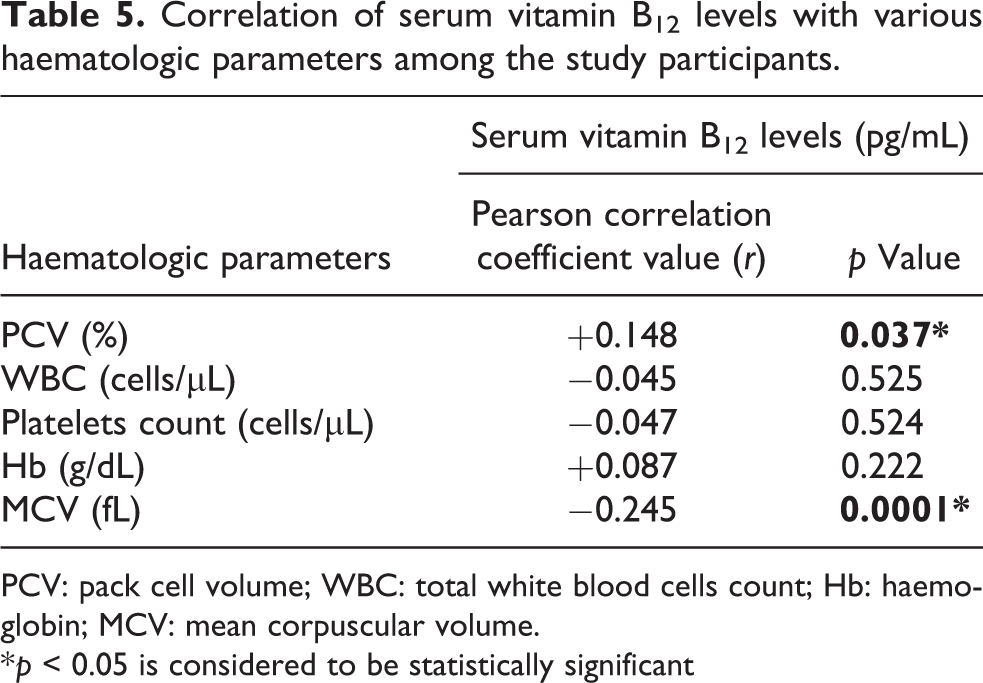
PCV: pack cell volume; WBC: total white blood cells count; Hb: haemoglobin; MCV: mean corpuscular volume.
* p < 0.05 is considered to be statistically significant
Table 6 shows frequency distribution for the occurrence of oval macrocytic red blood cells in the PBFs of metformin-treated and metformin-naive groups. There was a statistically significant difference between the metformin-treated and metformin-naive participants with respect to ovalocytosis in 18% versus 6%, respectively (p = 0.009). But, there was no statistically significant difference between the metformin-treated and metformin-naive participants with respect to macrocytosis in 5% versus 0%, respectively (p = 0.06). A high MCV was recorded for very few participants (i.e. 5%) in the metformin-treated group, but it was normal for all the participants in the metformin-naive group.
Frequency distribution for oval macrocytic red blood cells among metformin-treated and metformin-naive groups.a
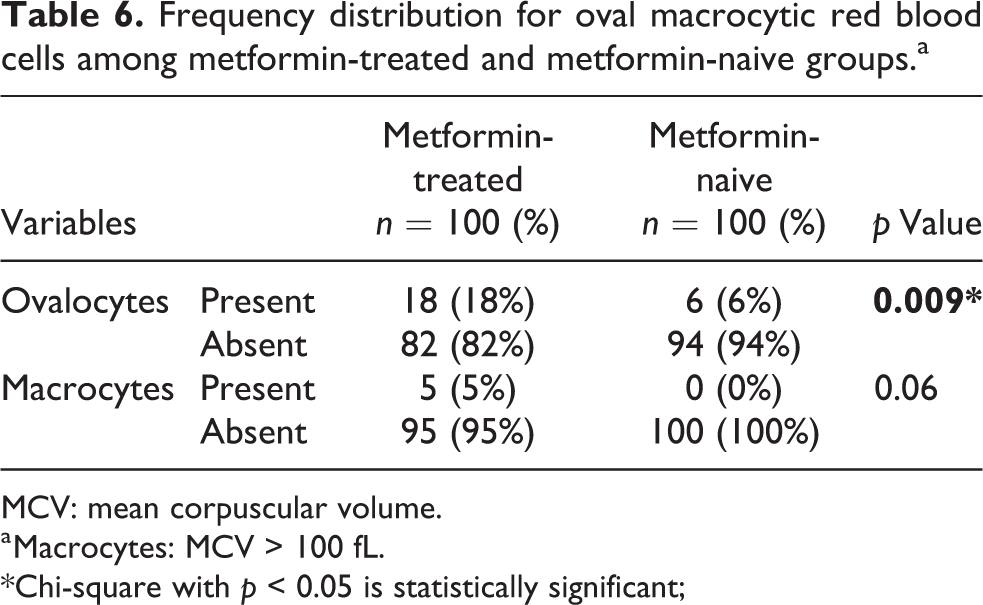
MCV: mean corpuscular volume.
a Macrocytes: MCV > 100 fL.
*Chi-square with p < 0.05 is statistically significant;
The peripheral neuropathy components assessed during this study comprised of pain sense, light touch sense, vibration sense, joint position sense, knee and ankle jerks, and planter reflex response. So peripheral neuropathy assessment is not a single entity. The statistically significant components of peripheral neuropathy evaluated during this study were impaired pain sense (paraesthesias), impaired vibration sense and impaired light touch sense among the metformin-treated group versus metformin-naive group with their respective prevalence rates of 86% versus 39% (p < 0.001), 83% versus 43% (p < 0.001) and 83% versus 39% (p < 0.001). Table 7 depicts this illustration.
The respective prevalence rates for statistically significant components of peripheral neuropathy evaluated during this study.

*p < 0.05 is statistically significant.
Table 8 shows the test for associations between the serum vitamin B12 categorization status and the peripheral neuropathy components assessed during this study. There were statistically significant associations between the serum vitamin B12 categorization status versus pain sense (p < 0.0001), vibration sense (p < 0.0001) and light touch sense (p < 0.0001) among the participants. But, there were no statistically significant associations between the serum vitamin B12 categorization status versus joint position sense (p = 0.0509), knee jerk reflex (p = 0.547), ankle jerk reflex (p = 0.547), planter reflex response (p = 0.915) and Romberg’s sign occurrence among the participants.
Test for associations between the serum vitamin B12 categorization status and the peripheral neuropathy components assessed during this study.
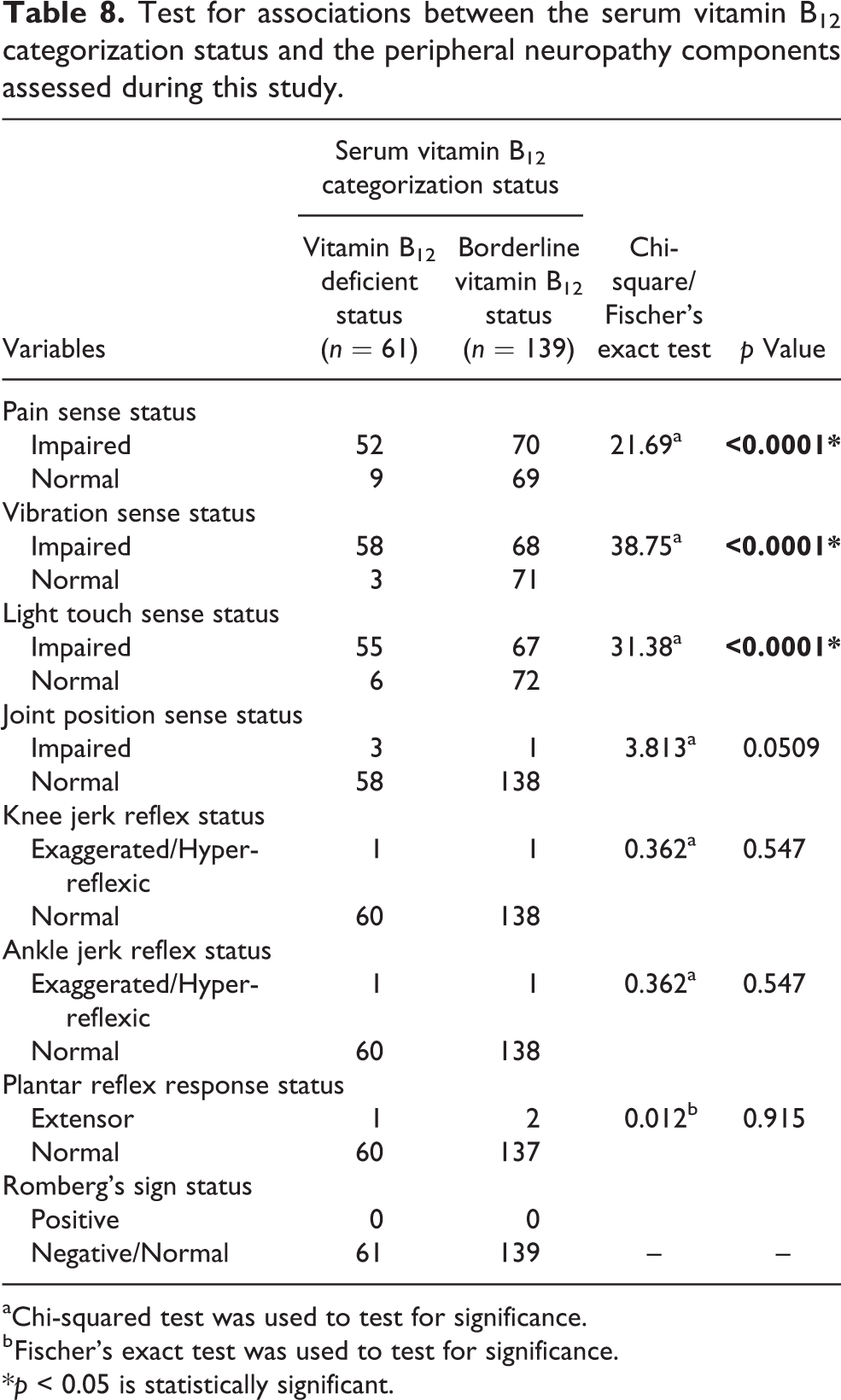
aChi-squared test was used to test for significance.
b Fischer’s exact test was used to test for significance.
*p < 0.05 is statistically significant.
Table 9 shows the test for associations between the metformin exposure status and the peripheral neuropathy components assessed during this study. There were statistically significant associations between the metformin exposure status versus pain sense (p < 0.001), vibration sense (p < 0.001) and light touch sense (p < 0.001) among the participants. But, there were no statistically significant associations between the metformin exposure status versus joint position sense (p = 0.430), knee jerk reflex (p = 0.095), ankle jerk reflex (p = 0.095), planter reflex response (p = 0.246) and Romberg’s sign occurrence among the participants.
Test for associations between the metformin exposure status and the peripheral neuropathy components assessed during this study.
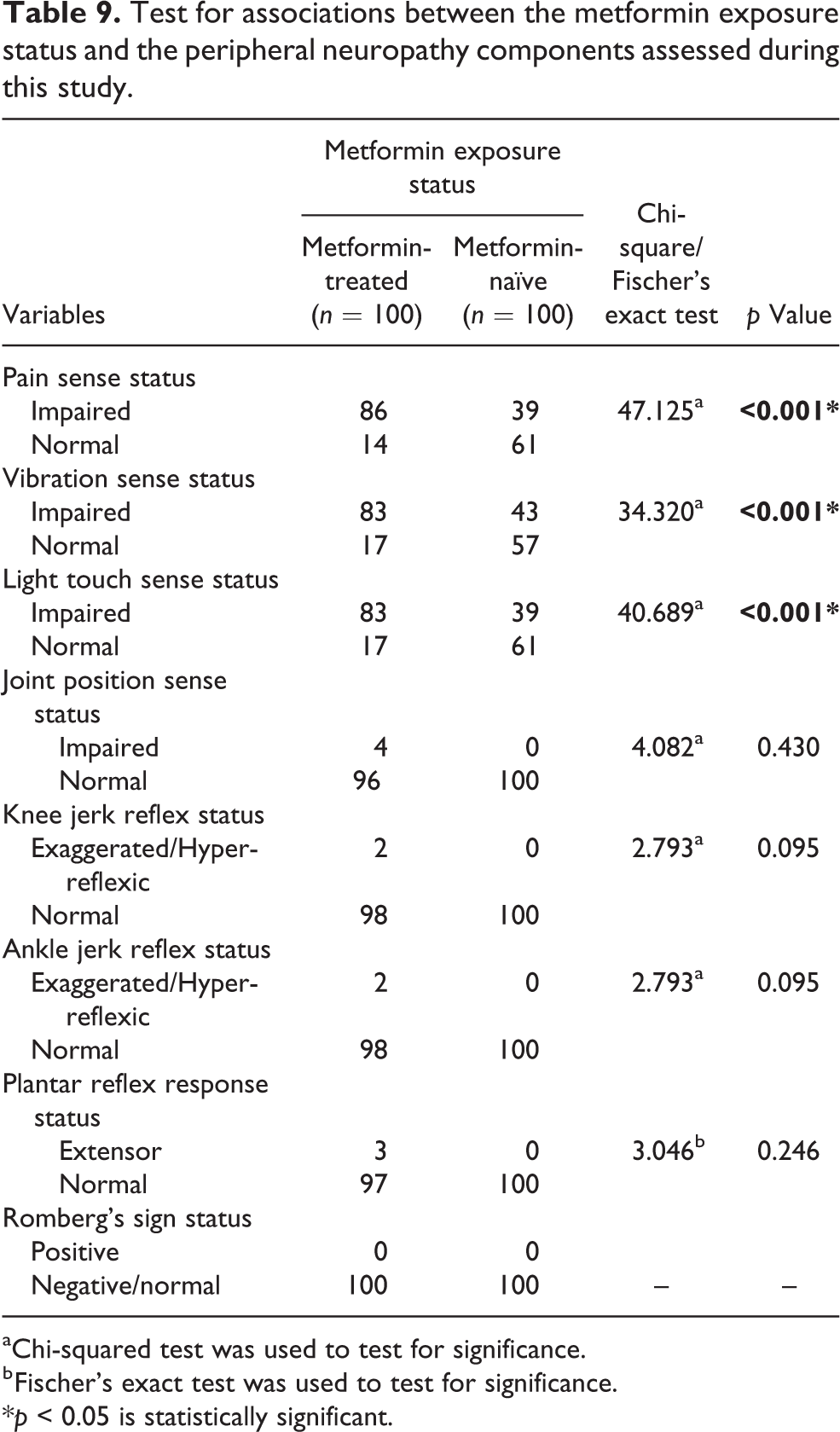
aChi-squared test was used to test for significance.
b Fischer’s exact test was used to test for significance.
*p < 0.05 is statistically significant.
In addition, the statistically significant weak negative correlation between serum vitamin B12 levels and MCV (r = −0.245, p = 0.0001) was depicted in Figure 1. While, the statistically significant weak positive correlation between serum vitamin B12 levels and PCV (r = +0.148, p = 0.037) was illustrated in Figure 2.
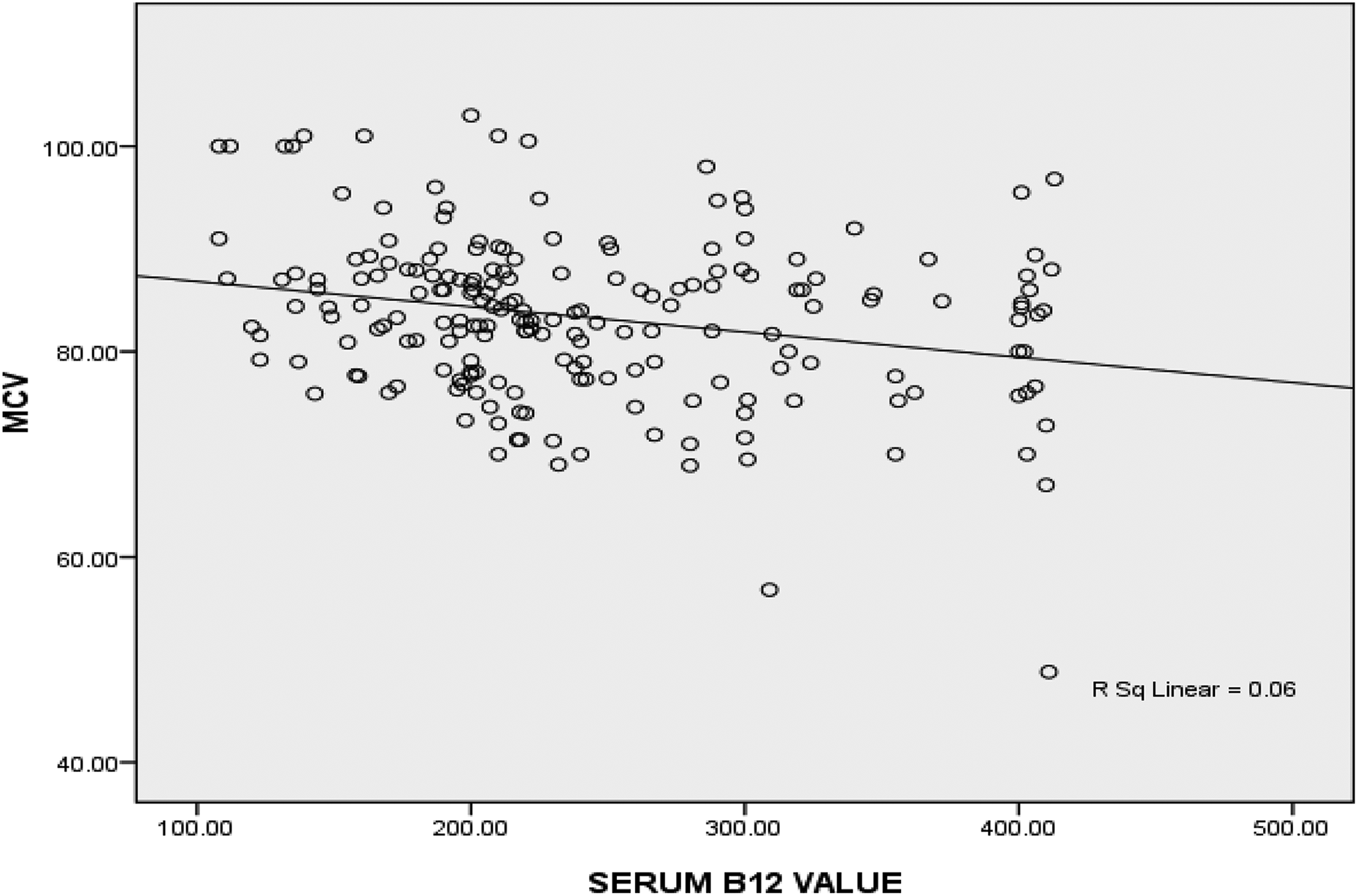
Correlation plot of serum vitamin B12 levels (measuring unit in pg/mL) and MCVs (measuring unit in fL) among the study participants (r = −0.245; p = 0.0001). MCV: mean corpuscular volume.
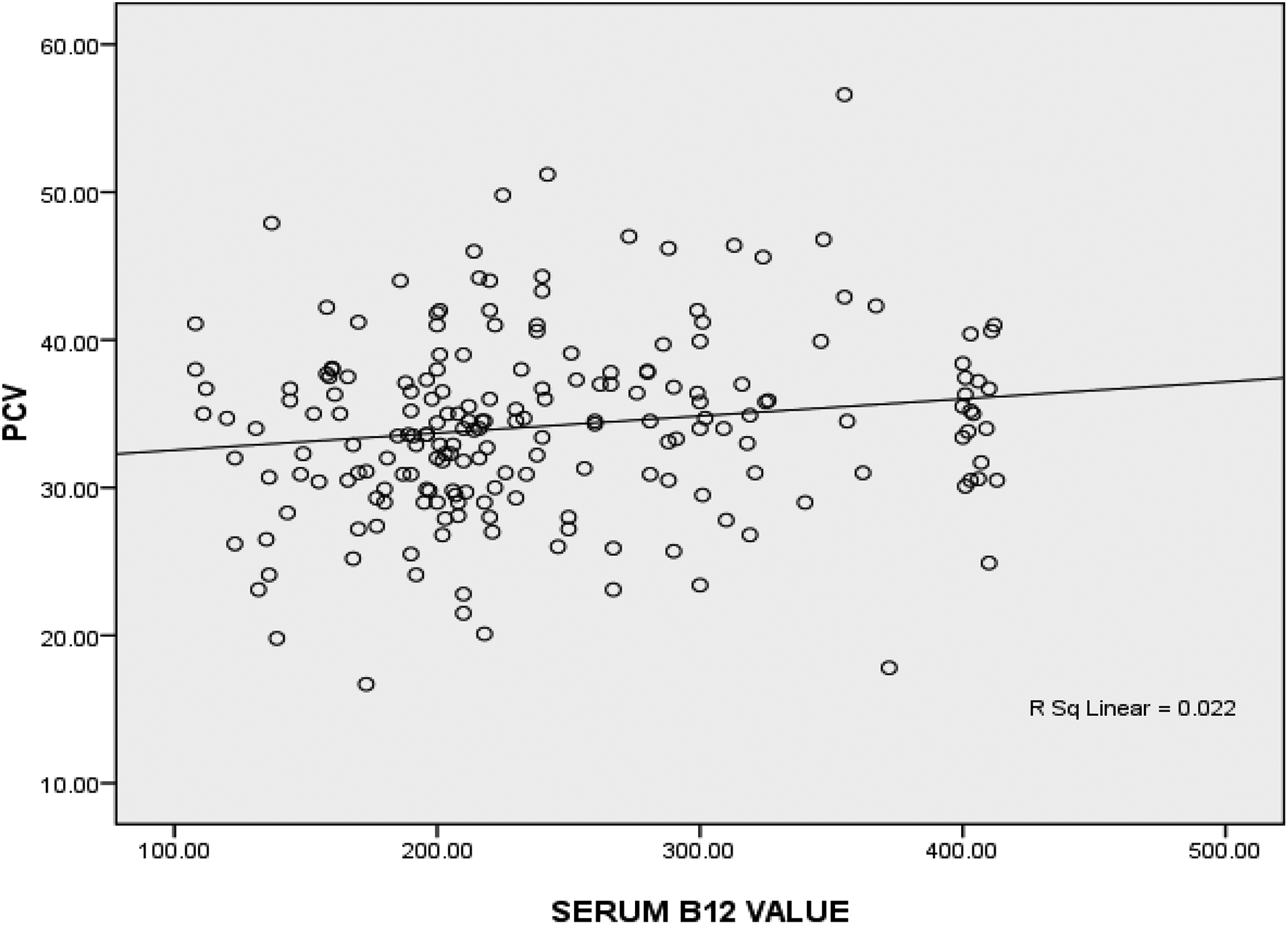
Correlation plot of serum vitamin B12 levels (measuring unit in pg/mL) and PCVs (measuring unit in %) among the study participants (r = +0.148; p = 0.037). PCV: pack cell volume.
In this study, regarding the metformin-naive participants, 70% were on insulin alone, 10% were on pioglitazone and vidagliptin, 10% were on pioglitazone and glibenclamide, 5% were on pioglitazone and glimepiride, while the remaining 5% were on glibenclamide alone. Concerning the metformin-treated participants, 70% were on metformin and glibenclamide, 10% were on metformin and glimepiride, 5% were on metformin and vidagliptin, 5% were on metformin, vidagliptin and pioglitazone, 5% were on metformin, glibenclamide and pioglitazone, while the remaining 5% were on metformin, glimepiride and pioglitazone. All the participants were also on dietary measures too.
Both case and control groups had fair glucose control using their mean FPG at presentation during diagnosis versus recruitment contact for sample collections in this study. The mean FPG at presentation during diagnosis versus recruitment contact for sample collections in this study among the metformin-treated group was 167.8 ± 29.1 mg/dL versus 140.8 ± 60.2 mg/dL (p < 0.001), respectively. While the FPG at presentation during diagnosis versus recruitment contact for sample collections in this study among the metformin-naive group was 186.8 ± 33.4 mg/dL versus 128.1 ± 42.1 mg/dL (p < 0.001), respectively. Therefore, the glycaemic index for the participants at the recruitment contact for sample collections in this study may not likely have impacted the outcome of our findings since their mean FPG levels revealed that they were better controlled.
Moreover, there was no statistically significant correlation between the increasing doses of metformin and serum vitamin B12 levels (r = −0.77, p = 0.45). Finally, there was also no statistically significant correlation between the duration of metformin use and serum vitamin B12 levels (r = −0.002, p = 0.98).
Discussions
As earlier mentioned, this study was designed to evaluate for the correlation between serum vitamin B12 levels and various haematologic indices among metformin-treated type 2 diabetic patients in a clinical practice setting with the rational purpose of alleviating/preventing the associated derangements. Metformin-induced vitamin B12 deficiency state or metformin-induced hypocobalaminemia is gradually becoming an epidemic among diabetic patients on moderate-to-high doses of metformin or those diabetic patients on metformin for a long period of time. According to previous studies done, long-term metformin pharmacotherapy has been reported to be associated with intestinal malabsorption of vitamin B12 culminating in vitamin B12 deficiency with consequential haematologic abnormalities, neurological impairments and biochemical derangements such as elevated homocysteine and MMA levels. 1,3,4,6,8,18,19,22 Our study revealed that 41% of metformin-treated diabetic patients have vitamin B12 deficiency. However, this only occurs in 20% of metformin-naive diabetic controls and there was a statistically significant difference between both groups (p < 0.001). This finding is in agreement with the prevalence of vitamin B12 deficiency among the Indians where close to one-third of metformin-treated diabetic patients have vitamin B12 deficiency. 1 Other studies supporting this showed 10–30% reduction in vitamin B12 level in metformin-treated diabetic patients. 3 A similar study by Akinlade et al. 5 conducted in Ibadan, South-Western, Nigeria, revealed a lower prevalence of vitamin B12 deficiency. This difference could be attributed to the different locations in which both studies were conducted. We conducted this study in a rural town area, while Akinlade and colleagues 5 conducted their study in Ibadan, an urbanized city with higher population mass and better socioeconomic condition in Nigeria. This study revealed the occurrence of oval macrocytes in about one-third of the metformin-treated diabetic patients. This was similar to the study conducted in Greece by Kalitsa et al. 6 where evidence of megaloblastic anaemia in type 2 diabetic patients was reported with MCV being significantly higher among metformin-exposed case group than the metformin-naive control group. Moreover, this study showed no statistically significant difference between male and female diabetics in terms of age, anthropometry, duration of diagnosis and haematologic indices except for MCH which was statistically significant p < 0.05. However, there was a statistically significant difference between the mean serum vitamin B12 level in male and female diabetic patients with p < 0.05. Also, there were statistically significant differences in PCV, Hb concentration, MCV, MCH and total WBC count between the metformin-treated and metformin-naive diabetic patients with p < 0.05. This finding showed the effect of metformin on haematologic parameters of diabetic patients. Furthermore, the prevalence rates for frank anaemia (PCV value < 30% with Hb concentration < 10 g/dL) among the metformin-treated and metformin-naive type 2 diabetes patients were 29% and 14%, respectively, which depicted a statistically significant difference between both groups (p < 0.05). The absorption of vitamin B12 has been reported to be affected by decreased intestinal motility, increased bacterial overgrowth, hypocalcaemia, hypokalaemia, hypomagnesemia and alteration in vitamin B12-IF complex. 3 A statistically significant weak positive correlation exists between PCV and serum vitamin B12 concentration in this study (r = +0.148; p = 0.037). This finding also showed the effect of serum vitamin B12 levels on haemopoiesis. A similar result was reported among the Palestinians where a statistically significant strong positive correlation was seen between PCV and serum vitamin B12 level (p = 0.001). 7 Akabwai et al. 2 also reported an association between Hb concentrations and serum vitamin B12 levels among Ugandan population. The low level of PCV seen among the participants in this study could be as result of their poor socio-economic status, as most of them are rural area dwellers and low income earners. This study reports a statistically significant weak negative correlation between MCV and serum vitamin B12 concentration (r = −0.245, p = 0.0001). This finding was in agreement with a negative correlation reported by Yajnik et al. 8 between MCV and serum vitamin B12 level among rural and urban Indians. However, there was no statistically significant correlation between total WBCs count and serum vitamin B12 levels. Also, there was no statistically significant correlation between platelets count and serum vitamin B12 levels. This study did not show any statistically significant correlation between the increasing doses of metformin and serum vitamin B12 levels (r = −0.77, p = 0.45). In addition, there was also no statistically significant correlation between the duration of metformin use and serum vitamin B12 levels (r = −0.002, p = 0.98). This finding, was however in contrast and discrepancy to the cohort study in Pakistan where the duration and dose of metformin was found to have a statistically significant influence on the serum vitamin B12 levels among the participants (p < 0.05). 4 The reason behind this observational finding may be because the patients in this study were not followed-up for a longer period of time to ensure drug (Metformin) compliance, as this may explain the fact that clinically overt features of vitamin B12 deficiency takes a long period of time, about 5–10 years before manifesting due to large amount of hepatic storage following cessation/impairment of gastrointestinal (GIT) absorption process. In addition, the serum vitamin B12 measurement was done at one particular point in time during this study as it was a short time prospective observational study of about 11-month duration.
In this study, the test for associations between the metformin exposure status and the peripheral neuropathy components assessment revealed that there were statistically significant associations between the metformin exposure status versus pain sense (p < 0.001), vibration sense (p < 0.001), and light touch sense (p < 0.001) among the participants. But, there were no statistically significant associations between the metformin exposure status versus joint position sense (p = 0.430), knee jerk reflex (p = 0.095), ankle jerk reflex (p = 0.095), planter reflex response (p = 0.246), and Romberg’s sign occurrence among the participants. In addition, the test for associations between the serum vitamin B12 categorization and the peripheral neuropathy components assessment revealed that there were statistically significant associations between the serum vitamin B12 categorization status versus pain sense (p<0.0001), vibration sense (p < 0.0001), and light touch sense (p < 0.0001) among the participants. But, there were no statistically significant associations between the serum vitamin B12 categorization status versus joint position sense (p = 0.0509), knee jerk reflex (p = 0.547), ankle jerk reflex (p = 0.547), planter reflex response (p = 0.915), and Romberg’s sign occurrence among the participants. Importantly, these findings regarding the peripheral neuropathy components assessment in this present study were in concordance (agreement) with the cross sectional study carried out by Singh et al. 22 involving 136 participants with type 2 DM (84 metformin-exposed and 52 metformin-naive) in India. Also, the presence of worsened clinical neurologic signs of overt vitamin B12 deficiency among the metformin-treated type 2 DM patients in this present study was similar to findings in the research conducted by Wile and Toth, 19 who compared 59 type 2 DM patients on metformin for at least 6 months and 63 others not using metformin for evidence of neuropathy, but they found more severe peripheral neuropathy in the former group using metformin.
According to the study conducted by Bauman et al., 14 the easiest way to lower the risk of Metformin-induced vitamin B12 deficiency among type 2 diabetic patients is through proper and adequate supplementation with oral calcium; as increase oral intake of calcium reverses vitamin B12 malabsorption induced by metformin. The precise mechanism by which metformin causes vitamin B12 deficiency has not been clearly elucidated, however proposed mechanisms include; alterations in small bowel motility due to calcium deficiency which stimulate bacterial overgrowth and consequential vitamin B12 malabsorption. Metformin has also been shown to inhibit the calcium-dependent endocytosis active transportation absorption process of vitamin B12-IF of castle complex at the terminal ileum. This inhibitory effect is reversed by oral co-administration of calcium supplement. We therefore recommend that vitamin B12 supplement should be routinely prescribe for metformin-treated type 2 diabetic patients via the parenteral route of administration, most preferably the intramuscular site injection; in order to prevent the occurrence of frank vitamin B12 deficiency with its associated complications that include central/peripheral neuropathy, haematologic abnormalities such as macro-ovalocytic anaemia and immune dysfunctioning due to hypersegmentation of polymorphonuclear leukocytes, and manifestation of biochemical derangements such as elevated homocysteine and MMA levels.
Finally, the limitation of this present study was that the patients were not followed up for a longer period of time to ensure metformin drug compliance. In addition, the serum vitamin B12 measurement was done at one particular point in time as it was a short time prospective observational study of about 11 months duration. It also aimed to look at the effect of chronic metformin therapy on serum vitamin B12 levels, but not folate. Chronic metformin therapy does not contribute to folate deficiency; however, folate deficiency may have contributed to megaloblastic (macro-ovalocytic) anaemia among some of the participants but this was not within the scope of our study design. This study did not assess for the level of HbA1C among the participants since it was not part of the scope of this research work and also the glycaemic index for the participants at recruitment contact for sample collections in this study may not likely have impacted the outcome of our findings because their mean FPG levels revealed that they were better controlled. It is also important to note that it takes quite a long duration of time, which is about 5–10 years for vitamin B12 deficiency to manifest during laboratory measurement or clinically due to hepatic storage compared to HbA1C which measures plasma glucose control over a period of 3 months duration. In addition, these other antidiabetic medications that was co-administered with metformin in this study has not been shown by any previous research to affect serum vitamin B12 levels in anyway. A long-term assessment with evaluation of the associations between various different serum vitamin B12 levels and metformin use using a case-control analytical prospective observational longitudinal cohort study design to monitor vitamin B12 deficiency, progression and associated laboratory (haematologic) abnormalities and clinical features would have been desirable. Also, intrinsic factor assay, as well as transcobalamin profile assay, serum transferrin receptors’ status, red cell folate, serum transferrin, total iron binding capacity (TIBC), serum copper, serum MMA and serum homocysteine levels would have been desirable but the financial implication/cost was a limiting factor.
Conclusion
In this study, a statistically significant weak negative correlation existed between serum vitamin B12 level and MCV among the metformin-treated type 2 diabetic patients. However, there a statistically significant weak positive correlation existed between serum vitamin B12 level and PCV among the metformin-treated type 2 diabetic patients. Also, the peripheral neuropathy components assessment revealed that there were statistically significant associations between the serum vitamin B12 categorization status or metformin exposure status versus pain sense, vibration sense, and light touch sense among the participants. Finally, the MCV is an erythrocyte index that represents the size/volume of a red blood cell. A high value of MCV greater than 100 fL suggests macrocytosis; and in the presence of a low PCV which signifies anaemia; with or without the presence of oval erythrocytes (ovalocytosis), may suggest megaloblastic anaemia but not pathognomonic. Serum vitamin B12 assay, MMA level, homocysteine level, CBC with PBFs examination showing macro-ovalocytes with or without hypersegmentation of neutrophils and bone marrow biopsy are useful for the definitive diagnosis of frank vitamin B12 deficiency state.
Data availability statement
Research data are not shared.
What is already known about this subject
Vitamin B12 deficiency is gradually becoming an epidemic among diabetic patients on moderate-to-high doses of metformin or those diabetic patients on metformin for a long period of time. The potential effect of chronic metformin pharmacotherapy to cause vitamin B12 deficiency with abnormalities in haematologic indices and central/peripheral neuropathy have been widely reported. Long-term usage of metformin has been reported to be associated with intestinal malabsorption of vitamin B12 culminating in vitamin B12 deficiency with likely associated haematologic abnormalities (including macro-ovalocytic anaemia and immune dysfunctioning due to hypersegmentation of polymorphonuclear leukocytes), central/peripheral neuropathy, and biochemical derangements such as elevated homocysteine and MMA levels.
What this study adds to the body of knowledge
In this study, statistically significant weak positive and weak negative correlations existed between serum vitamin B12 level versus PCV, and serum vitamin B12 level versus MCV, respectively. The peripheral neuropathy components assessment revealed that there were statistically significant associations between the serum vitamin B12 categorization status or metformin exposure status versus pain sense, vibration sense, and light touch sense among the participants. We therefore recommend that vitamin B12 supplement should be routinely prescribe for metformin-treated type 2 diabetic patients via the parenteral route of administration, most preferably the intramuscular site injection; in order to prevent the occurrence of central/peripheral neuropathy, haematologic abnormalities such as macro-ovalocytic anaemia and immune dysfunctioning due to hypersegmentation of polymorphonuclear leukocytes, and biochemical derangements such as elevated homocysteine and MMA levels. Serum vitamin B12 assay, MMA level, homocysteine level, CBC with PBFs examination showing macro-ovalocytes with or without hypersegmentation of neutrophils and bone marrow biopsy are useful for the definitive diagnosis of frank vitamin B12 deficiency state.
Footnotes
Acknowledgements
The authors would like to thank and appreciate all the staffs at outpatient Endocrinology Clinic of Irrua Specialist Teaching Hospital, Irrua, Edo State, Nigeria, for their immense assistance during the course of this study. Their support was of paramount importance to the completion of this study.
Author contributions
All the listed authors contribute equally to the study conception and design, carrying out of literature search, writing of methodology and protocols, conduction of research, co-working on data collection, data analysis, results presentation and interpretation, co-writing of discussion, conclusion, recommendation and joint approval of the final version of this research manuscript article. The authors alone are responsible for the contents and writing of this research manuscript article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from the Health Research Ethical Committee of Irrua Specialist Teaching Hospital Irrua (ISTH) and the assigned protocol identification number was ISTH/HREC/2016/March/026.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Duly written informed consents were obtained from all cases and controls. These written informed consents were documented in the respective medical case file for each of these patients and then counter signed by each of them for appropriate record purpose. All the study participants were counselled before and after clinical assessment and serum vitamin B12 screening. In addition, they were assured of the confidentiality of all their records. This study also served to notify and treat those participants with vitamin B12 deficiency and borderline vitamin B12 status.
Principal research investigators’ statement
All the authors jointly and unanimously confirm that the Principal Research Investigators for this original research work are Dr Olumuyiwa John Fasipe and Dr Sampson Omagbemi Owhin. In addition, they both had direct clinical responsibility for the study participants that took part in this research work.
