Abstract
The herbicidal action of diquat dibromide (DD) on plant cells is due primarily to the initiation of reactive oxygen species (ROS) formation, lipoperoxidation, and apoptotic cell death. It has been demonstrated that oxidative stress also occurs in animal cells exposed to high concentrations of DD; however, observations of DD’s effects on animal cells at concentrations below the reported ROS-initiation threshold suggest that some of these effects may not be attributable to ROS-induced cell death. Our results suggest that DD causes disruption of the Wnt pathway, calcium dysregulation, and cytoskeletal damage during development. Using embryos of the pond snail Lymnaea palustris as our model organism, we observed increased mortality, developmental delay and abnormality, altered motility, calcium dysregulation, decreased heart rate, and arrhythmia in embryos exposed to DD. Sperm extracted from adult snails that were exposed to DD exhibit altered motility, increased abundance, and high mortality. Effects were quantified via real-time imaging, heart rate assessment, flow cytometry, and mortality scoring. We propose that there are two models for the mechanism of DD’s action in animal cells: at low concentrations (≤28 µg/L), apoptotic cell death does not occur, but cytoskeletal elements, calcium regulation, and Wnt signaling are compromised, causing irreversible damage in L. palustris embryos; such damage is partially remediated with antioxidants or lithium chloride. At high concentrations of DD (≥44.4 µg/L), calcium dysregulation may be triggered, leading to the establishment of an intracellular positive feedback loop of ROS formation in the mitochondria, calcium release from the endoplasmic reticulum, calcium efflux, and apoptotic cell death. Permanent cellular damage occurring from exposure to sublethal concentrations of this widespread herbicide underscores the importance of research that elucidates the mechanism of DD on nontarget organisms.
Introduction
Diquat dibromide (DD) is a compound in many Roundup formulations, as well as other commercial herbicides. The effects of DD on nontarget organisms have been minimally studied, though previous studies indicate that herbicidal runoff into surface waters poses a risk to the fecundity, development, and viability of organisms that come into direct contact with DD. 1,2 DD is known to initiate the formation of reactive oxygen species (ROS) in plant tissues, leading to lipoperoxidation and apoptotic cell death. 3,4 While ROS are essential during animal development and regeneration 5 and serve as second messengers in several signaling pathways, 6 overproduction of ROS is implicated in a number of diseases and comprises a stress response to toxins. 7 –9 Using the pond snail Lymnaea palustris, a rapidly developing aquatic invertebrate, this study sought to characterize specific DD-induced cellular and developmental effects on animal embryos.
At high concentrations (≥44.4 µg/L), DD has been shown to elicit oxidative stress and apoptosis in animal cells, as determined by transcription quantification of oxidative stress responsive proteins, including superoxide dismutase, glutathione reductase, catalase, and heat-shock proteins (hsp-40 and hsp-70), but not at lower concentrations. 8 In a study where rat testicular cells were exposed to very high concentrations of DD, Cavalli et al. 10 demonstrated that DD initiates a significant increase in calcium uptake, followed by ROS formation, preceding massive calcium efflux and cell death. These findings are consistent with our results and support a model for the mechanism of DD at high concentrations (≥44 µg/L) where DD causes calcium uptake leading to high intracellular calcium concentration and mitochondrial uptake, causing increased ATP production while simultaneously increasing ROS formation. 11,12
One study suggested that L. stagnalis embryos, though developmentally delayed, make a full recovery after 24-h exposure to DD at 13.7 µg/L followed by a return to control medium. 1 However, 24-h exposure of L. palustris embryos to 3.5 µg/L DD or greater causes permanent heart defects including arrythmia and decreased average heart rate. We hypothesize that there are different mechanisms for the action of DD at high (≥44 µg/L) and low (≤28 µg/L) concentrations. Our observations of DD-induced defects at a range of concentrations suggest an alternative mechanism apart from ROS formation and apoptotic cell death at lower DD concentrations (≤28 µg/L) that causes developmental arrest, phenotypic abnormality, and cytoskeletal defects. Such defects occur upon exposure of embryos to DD concentrations well within the US EPA recommended maximum contaminant level goal (MCLG) of DD for human drinking water (0.02 mg/L). 13 Here, we provide evidence for effects of DD on cytoskeletal elements and calcium dynamics. These and other cellular effects may be linked by a DD-induced disruption of both canonical and noncanonical Wnt pathways.
Wnt signaling is essential to many stages of determination and differentiation during embryogenesis, ongoing cellular proliferation, and regeneration. 14,15 Cytoskeletal organization, heart development, and calcium regulation are representative of the effects of Wnt signaling, and we demonstrated that these are specifically compromised in embryos exposed to even extremely low concentrations of DD during development. We thus questioned whether canonical and/or noncanonical Wnt signaling was being disrupted by DD. DD has also been demonstrated to negatively affect fecundity in L. palustris adults. 2 While DD may indirectly decrease fecundity by altering abundance of steroidogenic enzymes in these hermaphroditic organisms, it is possible that DD may also directly interfere with reproduction by acting at the cellular level in sperm production, maturation, and/or motility. Wnt/Ca2+ signaling, specifically mediated by Wnt 11 and Wnt 5a, 16 is required for sperm maturation and motility, 17 which are aberrant in sperm extracted from DD-treated snails. Since calcium homeostasis affects cellular adhesion and division, 18 ciliogenesis and myogenesis, 19 and later developmental events, a disruption in the Wnt/Ca2+ or other Wnt pathways during a crucial developmental window has the potential to disrupt subsequent development. 20,21
A primary goal of this study was to determine whether the phenotypic damage observed in L. palustris embryos exposed to sublethal DD concentrations could be reversed by a return to control media or by treatment with calcium-adjusted medium, antioxidants, or the Wnt agonist lithium, 22 since the defects we observed were consistent with calcium dysregulation and increased ROS production, and previous studies have demonstrated that lithium augments Wnt signaling. 23 –25 Although they may reach maturity and appear phenotypically normal, embryos exposed to DD at even trace concentrations (<14 µg/L) exhibit permanent cytoskeletal damage, such as decreased average heart rate or arrhythmia; these phenotypes are distinct from the apoptotic cell death induced by ROS overproduction typical upon exposure to high DD concentrations (≥44 µg/L). We sought to elucidate the effects of low concentrations of DD on cells and embryos and to provide initial characterization of the putative disruption to Wnt signaling.
Materials and methods
Animals and reagents
Lymnaea (Stagnicola) palustris is a Basommatophora gastropod mollusc found worldwide in freshwater lakes, streams, and rivers. Lymnaea is the preferred genus to Stagnicola 26 for the Eurasian species palustris, which is very highly related to the North American elodes, and these have been considered conspecific. 27 L. palustris are hermaphroditic and suitable for use as ecotoxicological indicators due to hardiness, year-round reproductive output, rapid developmental progression, 28 ability to colonize diverse ecosystems, 29 and high sensitivity to mutagens as documented in closely related species. 30,31 Adult laboratory-reared L. palustris snails were kept in filtered and aerated breeding tanks on a 12-h light/12-h dark cycle at 21 ± 1°C, and pH maintained at 7.4 ± 0.2 in artificial pond water (APW). Snails were fed rinsed organic romaine lettuce ad libitum.
Control concentration of calcium in APW in the form of calcium chloride dihydrate (CaCl2 · 2H2O) was 0.027 g/L. High calcium medium was defined as twice the control calcium concentration, and low calcium medium defined as half the control calcium concentration.
DD (45422; Sigma-Aldrich, St Louis, Missouri, USA) was made in APW at concentrations given in Table 1, with “1X” based on the MCLG for DD. 13 Calcium ionophore (A23187; Santa Cruz Biotechnology, Dallas, Texas, USA) was used at 0.5 µM; antioxidants Trolox (10011659; Cayman Chemicals, Ann Arbor, Michigan, USA; 50 µM), and L-ascorbic acid (Cayman Chemicals 14656; 75 µM) were applied together.
DD concentrations.a

EPA: environmental protection agency; MCLG: maximum contaminant level goal; DD: diquat dibromide.
a1X concentration (28 µg/L) is equivalent to EPA MCLG for DD.
Embryo assays
Encapsulated gastrula-stage embryos were removed from jelly masses and transferred to either fresh APW or treatment medium in 12- or 24-well plates (typically 10–20 embryos per 2.5-mL well). 32 A chronic treatment was defined as continued exposure to a specific treatment or chemical for the duration of an experiment, typically 1–2 weeks. An acute treatment was defined as an exposure up to 24 h, followed by transfer to APW.
Motility was assessed by counting the number of stops per minute during the normal rotational pattern observed in gastrula stage embryos, using an EVOS FL inverted compound microscope (Thermo Fisher Scientific, Waltham, Massachusetts, USA).
Quantification of heart rate defects and arrhythmia was assessed by counting the number of beats per minute (bpm) of embryos and juvenile snails at equivalent developmental stages with a counter and 1-min timer, using the compound microscope. 33
Atomic absorption
Calcium standards ranging from 0.54 ppm to 4.32 ppm were analyzed by atomic absorption (Perkin Elmer Instruments, Waltham, Massachusetts, USA, AAnalyst 100 Atomic Absorption Spectrometer) to assess changes in calcium concentration over time in samples conditioned for 24 h with embryos. Media were collected from embryos incubated for 24 h in APW ±1X DD (approximately 200 embryos per 50-mL solution) and compared to both APW and 1X DD media without embryos (unconditioned controls). Solutions were sterile-filtered (0.22 µM) and analyzed by atomic absorption; wavelength of analysis 422.7 nm, slit width 0.7 nm, air-acetylene oxidizing (clear, blue).
Sperm extraction for motility and viability assays
Adult snails of similar weight (within 0.1 g) were housed in one of three treatments: control (APW), 5X DD, and 10X DD. Habitats were cleaned and snails fed fresh lettuce three times a week for 1- and 2-week experimental periods. At the end of the exposure period, snails were dissected and ovotestes and seminal vesicles were removed and placed into 300 µL sterile saline solution. Samples were rocked at approximately 24 r/min for 30–60 min with occasional gentle agitation to encourage free sperm release. Slides were prepared using 1/10 (30 µL) of the sample and coverslipped for viewing on the compound microscope.
Flow cytometry
For sperm density and viability assessment, sperm were extracted as mentioned above for use with Millipore Sigma (Burlington, Massachusetts, USA) Guava EasyCyte Flow Cytometer and Software system. For abundance, sperm cells were stained with 0.3-mg/mL 4′,6-diamidino-2-phenylindole, dihydrochloride (DAPI; Thermo Fisher Scientific D1306) 34 and assessed using the Express Plus program. For viability, sperm cells were stained with 0.01-mg/mL propidium iodide (PI; Thermo Fisher Scientific P3566) 35,36 and assessed using the ViaCount program.
Statistical analysis
Statistical analysis for quantification of results was performed in Microsoft Excel, t-test: Two-Sample Assuming Equal Variances; hypothesized mean difference = 0; p (T ≤ t) one-tail. Confidence intervals: *p < 0.05, **p < 0.01, and ***p < 0.001.
Results
DD-induced developmental abnormalities
Previous work in our lab demonstrated deleterious developmental effects of DD on L. palustris. 37 Typically, the toxic effects of DD were observed following prolonged exposure to high concentrations (≥140 µg/L). Here, we used 24-h exposures at low concentrations of DD and observed that phenotypic abnormality and death occur in high percentages; embryos exposed to 0.5X–1X DD exhibit significantly higher rates of mortality compared to controls 9 days after acute exposure (Figure 1(a)). To test whether DD inhibited calcium uptake, 5X DD-treated embryos were incubated in calcium-altered media. However, significantly higher percentages of abnormality were observed with the addition of the 5X DD in all media except calcium-free, suggesting instead that DD may augment calcium uptake (Figure 1(b)); calcium-free medium alone causes high percentages of mortality during L. palustris embryonic development but 5X DD addition does not cause mortality percentage to increase. DD interferes with calcium dynamics in gastrula-stage embryos, causing significantly higher calcium efflux compared to control (Figure 1(c)), and the observed DD-induced calcium dysregulation occurred in less than 24-h exposure to 1X DD concentration (28 µg/L).
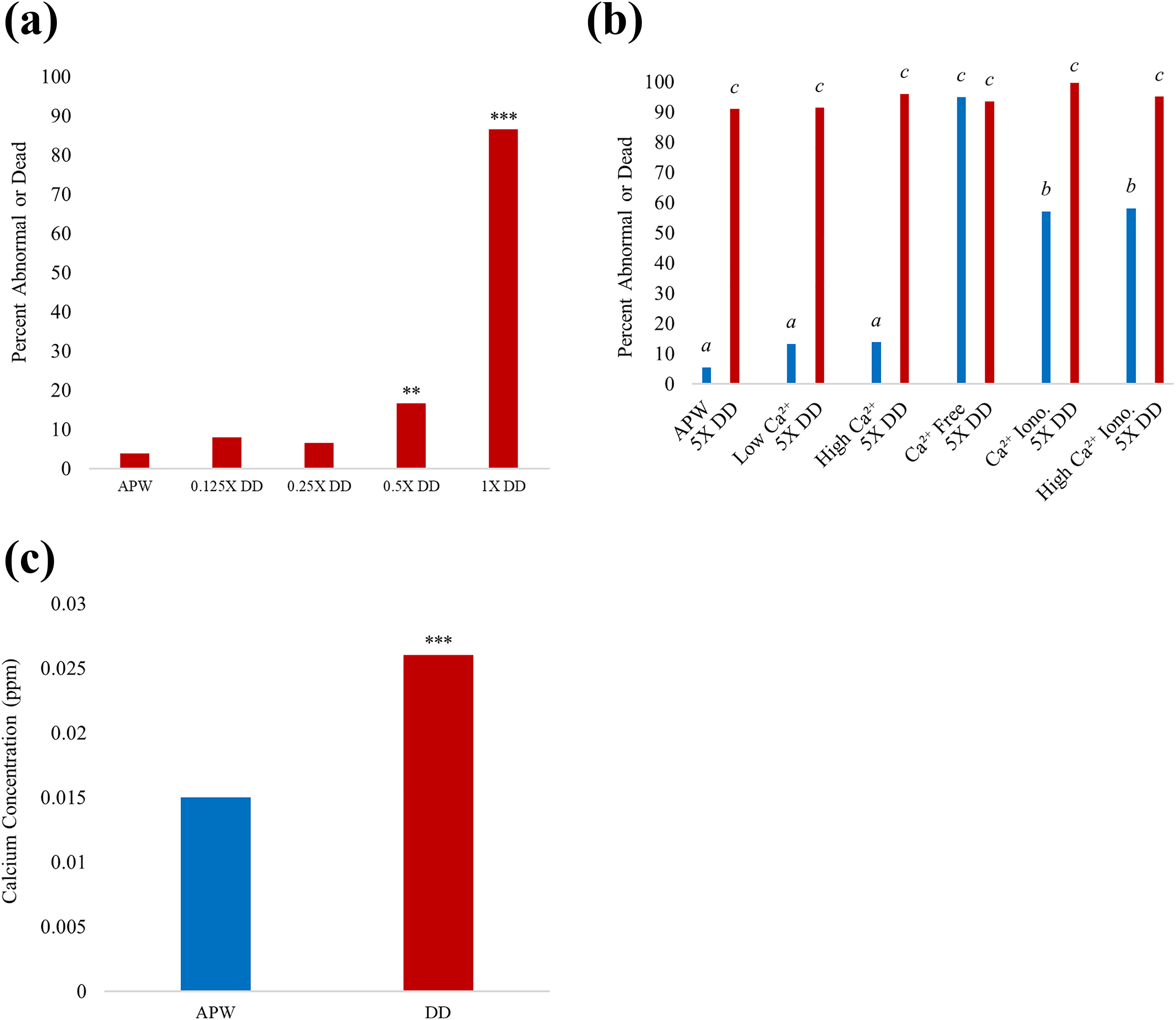
(a) Percent of inviable embryos (abnormal or dead) assessed 9 days after acute exposure at gastrula stage to DD concentrations (indicated on the x-axis; see “Materials and methods” section for concentrations), demonstrating dose-dependent effect (**p ≤ 0.01; ***p ≤ 0.001). (b) Percent of inviable embryos (abnormal or dead) assessed after 12 days of chronic exposure to 5X DD (140 µg/L) beginning at gastrula stage, in calcium-adjusted media. APW: normal calcium; low Ca2+: half normal calcium APW; high Ca2+: twice normal calcium APW; Ca2+ free: no-calcium APW; Ca2+ iono: calcium ionophore in APW (or in high Ca2+ APW); control embryos: blue; DD-treated embryos: red. (c) Calcium efflux in media conditioned by gastrula-stage embryos for 24 h in APW and 1X DD in APW at 21°C. Baseline calcium concentration removed to measure net calcium efflux. Calcium standard R 2 = 0.9985 (***p ≤ 0.001). APW: artificial pond water; DD: diquat dibromide.
Effects of DD on heart rate
DD was found to significantly decrease heart rate in developing snails at even the lowest concentration (0.125X DD) in a dose-dependent manner with a robust linear correlation (Figure 2(a)). DD’s effects upon heart rate were observed in both chronically exposed embryos and in those returned to APW following acute exposure. Heart rate defects worsened from 3 days postexposure to 7 days postexposure, with a decrease of up to 60% (chronic 5X DD; Figure 2(b)). Interestingly, embryos acutely exposed to 0.125X DD continue to display decreasing heart rate compared to embryos acutely exposed to 0.5X DD, from 3 days to 6 days postexposure (Figure 2(b)).
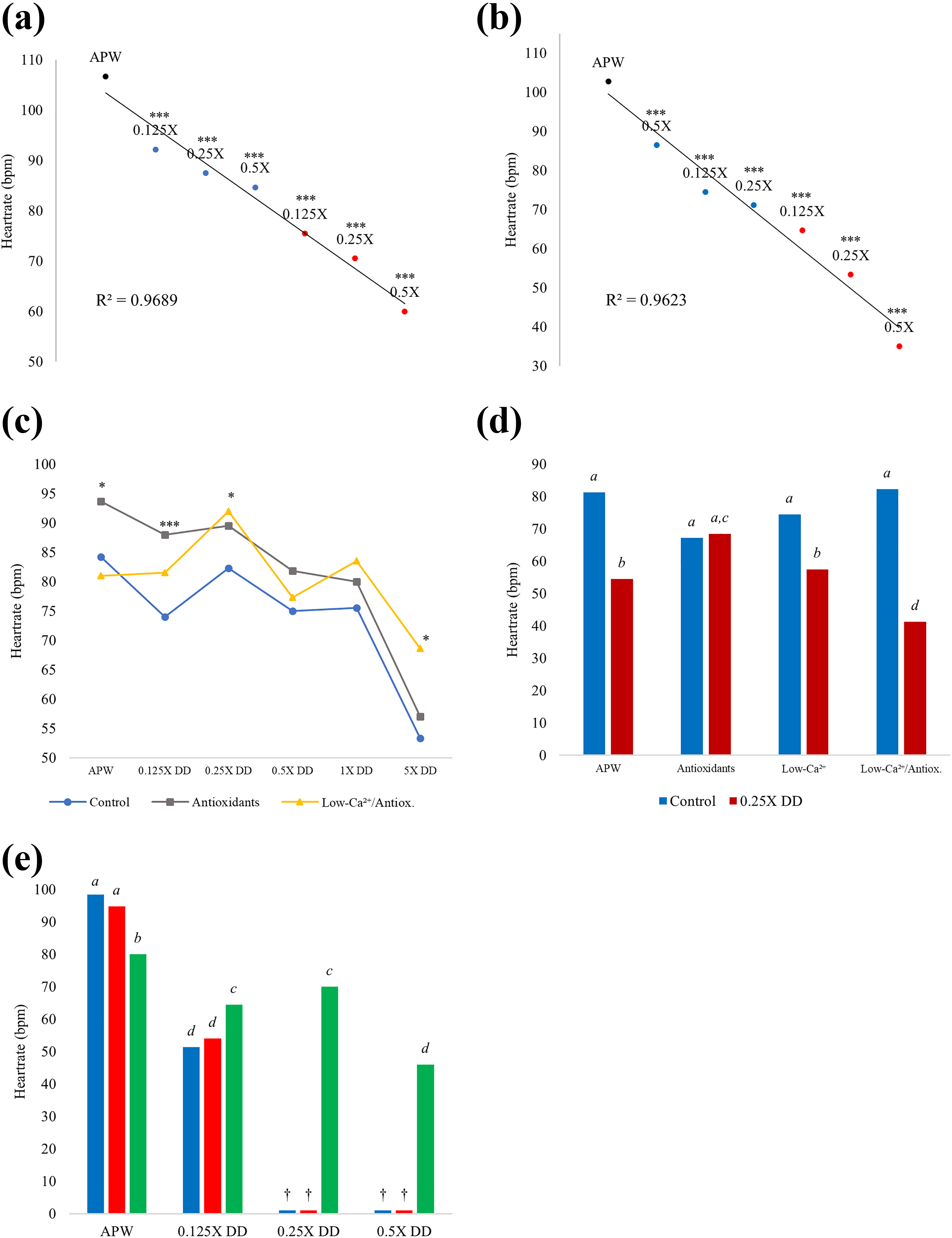
(a) Average heart rates of embryos 3 days following chronic or acute DD exposure at gastrula stage. Acute treatment (blue points): embryos exposed to DD for 24 h; chronic treatment (red points): embryos continuously exposed to DD. t-Test compared each to control (APW, black point) heart rate (N = 5 per group; ***p ≤ 0.001). (b) Average heart rates of embryos 7 days following chronic or acute DD exposure at gastrula stage. Acute treatment (blue points): embryos exposed to DD for 24 h; chronic treatment (red points): embryos continuously exposed to DD. t-Test compared each to control (APW, black point) heart rate (N = 5 per group; ***p ≤ 0.001). (c) Average heart rates of embryos treated for 24 h at gastrula stage, assessed 6 days after acute exposure to DD at concentrations indicated on the x-axis. Media: APW control = blue/circles, low-calcium and antioxidants = yellow/triangles, and antioxidants = grey/squares (75 µM ascorbic acid and 50 µM Trolox). t-Test compared heart rates in each DD concentration between control and other media (N = 5 per group; *p ≤ 0.05; ***p ≤ 0.001). (d) Average heart rates of embryos chronically exposed to 0.25X DD (7 µg/L) for 12 days, beginning at gastrula stage. Four media, with or without DD: APW control, antioxidants, low Ca2+ (half normal calcium), and low Ca2+ with antioxidants. In each pair, blue bar is medium without DD and red bar is medium with 0.25X DD. Bars marked a are not significantly different from control heart rate; b are significantly different from a or c; c is significantly different from b but not from a; d is significantly different from a, b, and c (differences at p ≤ 0.05; N = 5 per group). (e) Average heart rates of embryos chronically exposed to LiCl and varying DD concentrations (indicated on the x-axis) for 6 days, beginning at gastrula stage. Blue bars: no LiCl; red bars: 10 mM LiCl; and green bars: 30 mM LiCl. † = 0 bpm; death. Each letter indicates significant difference at p ≤ 0.05 (N = 5 per group). APW: artificial pond water; DD: diquat dibromide; LiCl: lithium chloride; bpm: beats per minute.
Embryos exposed to 0.25X and 5X DD for 24 h and co-treated with antioxidants/low-Ca2+ exhibited significantly higher average heart rates compared to those exposed to DD alone when assessed 6 days following the acute exposure (Figure 2(c), yellow). Similarly, embryos cocultured with antioxidants in DD (Figure 2(c), grey) had significantly higher heart rates than those in DD alone at the lowest concentration (0.125X DD) and in control medium (APW). It is important to note that antioxidants may increase heart rate in animals under control conditions, as previously demonstrated in zebrafish. 38 Following 12 days of chronic exposure to 0.25X DD, antioxidants were shown to be the most effective in improving DD-induced heart rate defects compared to other treatment variations (Figure 2(d)). The average heart rates of embryos co-treated with antioxidants and DD were shown to be significantly higher than DD-treated embryos in low-Ca2+ and antioxidant/low-Ca2+ media. Additionally, embryos co-treated with antioxidants and DD had an average heart rate that was not significantly different compared to control embryos (Figure 2(d)). Though low calcium availability was observed to be slightly more effective than high calcium medium or medium with calcium ionophore at mitigating DD-induced mortality, calcium-adjusted medium is not enough to rescue or significantly improve DD-induced phenotypes (Figure 1(b)). Prolonged treatment with low-calcium medium is known to have adverse effects independent of herbicidal exposure and is a likely explanation for decreased heart rate observed in embryos chronically exposed to 0.25X DD and treated with antioxidants/low-Ca2+ (Figure 2(d)).
Embryos typically do not survive chronic exposure to DD at concentrations > 0.125X; however, embryos cocultured with 30 mM lithium chloride (LiCl) and DD not only survived, but also maintained moderate heart rates up to 0.5X DD 6 days postexposure (Figure 2(e)). For interpretation of results, it should be noted that the average normal heart rate of L. palustris embryos changes during development, from an average heart rate of approximately 99–110 bpm at the onset of heartbeat 4–5 days post-oviposition (Figure 2(a), (b), and (e)), to average heart rates of approximately 80–88 bpm in young juveniles approaching hatching (7–8 days post-oviposition).
Decreased motility with DD exposure
The normal spinning of gastrula-to-veliger stage embryos typically is smooth, rhythmic, and generally uninterrupted by cessation in motion and is essential to development and ciliogenesis. 39 Embryos exposed to DD exhibit a significantly higher number of stops per minute than control embryos (Figure 3(a)); the number of stops increases linearly with increasing DD concentration as well as with prolonged exposure time (Figure 3(b)). Acute exposure to DD with the addition of 30 mM LiCl (Figure 3(c)) was found to significantly decrease the number of stops per minute in gastrula embryos, though LiCl slightly increases the number of stops per minute in medium without DD. Embryos cocultured with 10 mM LiCl did not exhibit significantly different results compared to controls after 24 h. Embryos acutely exposed to DD and reassessed 72-h postexposure exhibited a significantly reduced number of stops per minute if cocultured in 10 mM or 30 mM LiCl (Figure 3(d)), although 30 mM LiCl was more effective in reducing motility stops at all DD concentrations. These results suggest that LiCl mitigates the DD-induced motility phenotype, although it is developmentally deleterious when not in co-treatment with DD. Embryos chronically exposed to DD with 10 mM and 30 mM LiCl exhibit significantly reduced number of stops per minute compared to those without LiCl treatment (Figure 3(e)); 30 mM LiCl treatment completely restores normal motility.
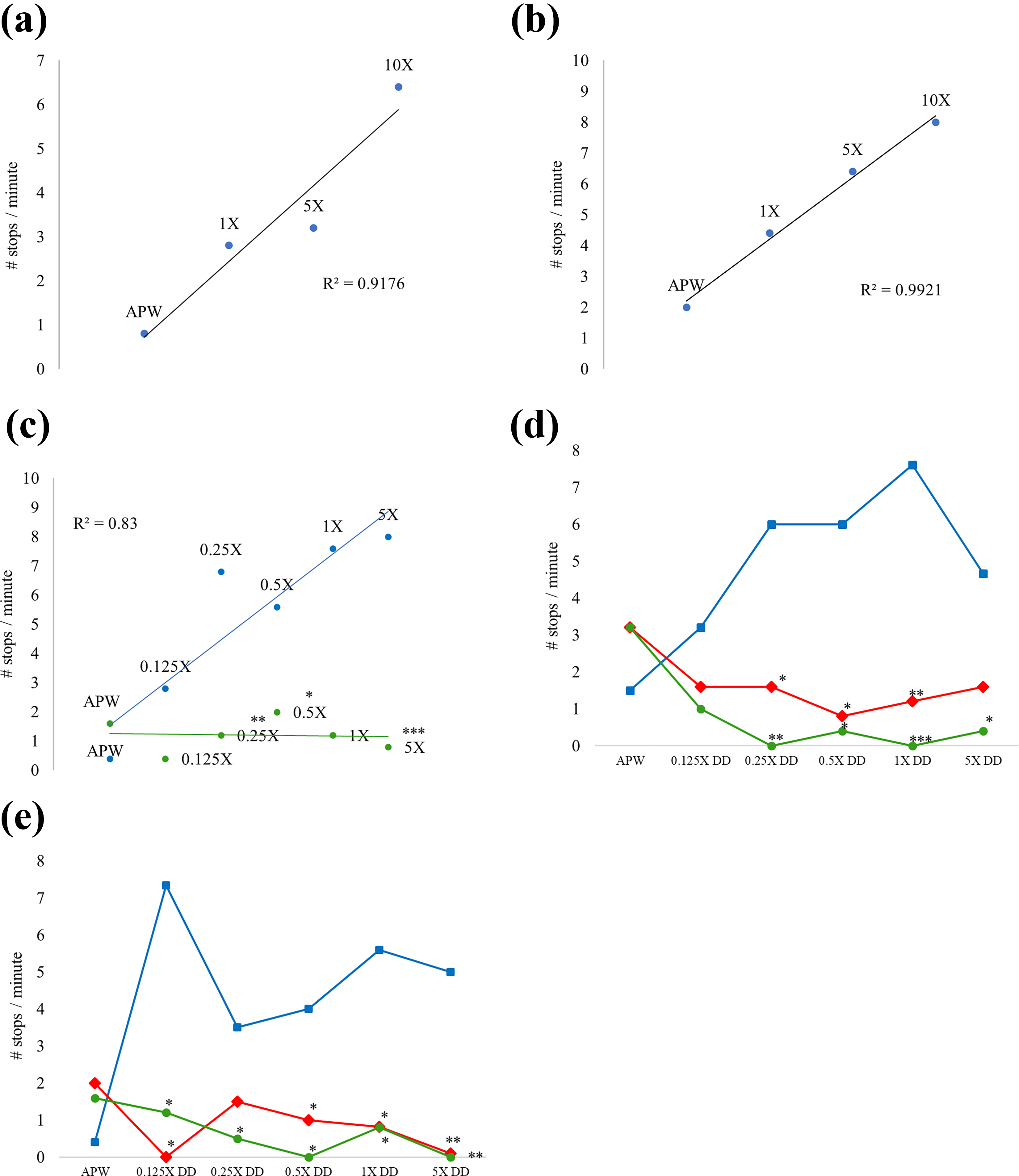
(a) Average number of stops in normal embryonic ciliary motility per minute following 1 day chronic DD exposure at 1X, 5X, and 10X concentrations (N = 5 per group). (b) Average number of stops in normal embryonic ciliary motility per minute following 3 days chronic DD exposure at 1X, 5X, and 10X concentrations (N = 5 per group). (c) Average number of stops in normal embryonic ciliary motility per minute following 24-h exposure to 0.125X, 0.25X, 0.5X, 1X, and 5X DD in control media or media supplemented with LiCl. Blue points/trendline: control APW; and green points/trendline: 30 mM LiCl with DD exposure. R 2 value shown for control (blue) trendline (*p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001; N = 5 per group). (d) Average number of stops in normal embryonic ciliary motility per minute following exposure to DD for 24 h then culture in APW (control), 10 mM LiCl-APW, or 30 mM LiCl-APW for 48 h before assessment. Blue squares/line: control untreated; red diamonds/line: embryos 10 mM LiCl; green circles/line: 30 mM LiCl (N = 5 per group). (e) Average number of stops in normal embryonic ciliary motility per minute following 72 h of chronic exposure to DD in APW (blue squares/line), 10 mM LiCl (red diamonds/line) or 30 mM LiCl (green circles/line). N = 5 per group; *p ≤ 0.05; **p ≤ 0.01. APW: artificial pond water; DD: diquat dibromide; LiCl: lithium chloride.
In addition to DD increasing the number of stops per minute in developing embryos, erratic motility is also observed (Online Supplemental Data 1). LiCl treatment with DD reduced the number of stops per minute and restored normal smooth, rhythmic motility in DD-treated embryos.
Sperm density, motility, and mortality
Chronic exposure consistently increased the size of seminal vesicles in adult L. palustris snails exposed to 5X (140 µg/L) and 10X (280 µg/L) DD. Normal seminal vesicles of control snails were dissected and compared to those of snails chronically exposed to DD for 3 weeks (Figure 4(a)). It has been reported that DD reduces fecundity and steroid sex hormone levels, in part through a significant reduction in steroid acute regulatory protein (StAR) expression 2 ; however, the increased seminal vesicle size and sperm count (Figure 4(b) and (d)) suggest that sperm release and/or maturation are compromised as well. Sperm collected from snails chronically exposed to 5X or 10X DD exhibit greater density, erratic motility, and tendency to form twisted cable-like structures (Figure 4(b)) in contrast to the smoother sinusoidal movements of control sperm (Figure 4(c); Online Supplemental Data 2). Differences in sperm density were quantified using nuclear (DAPI) staining and flow cytometry, demonstrating higher sperm count in 5X and 10X DD-treated snails in a dose-dependent manner (Figure 4(d)); extracted sperm were stained with DAPI to assess total sperm count (Figure 4(f)). However, live/dead analysis of the sperm samples reveals that sperm viability was not enhanced in DD-treated snails; staining of inviable cells (PI) followed by flow cytometry reveals greater numbers of dead cells in both 5X and 10X DD (Figure 4(e)); and extracted sperm were stained with PI to assess inviable sperm count (Figure 4(g)).
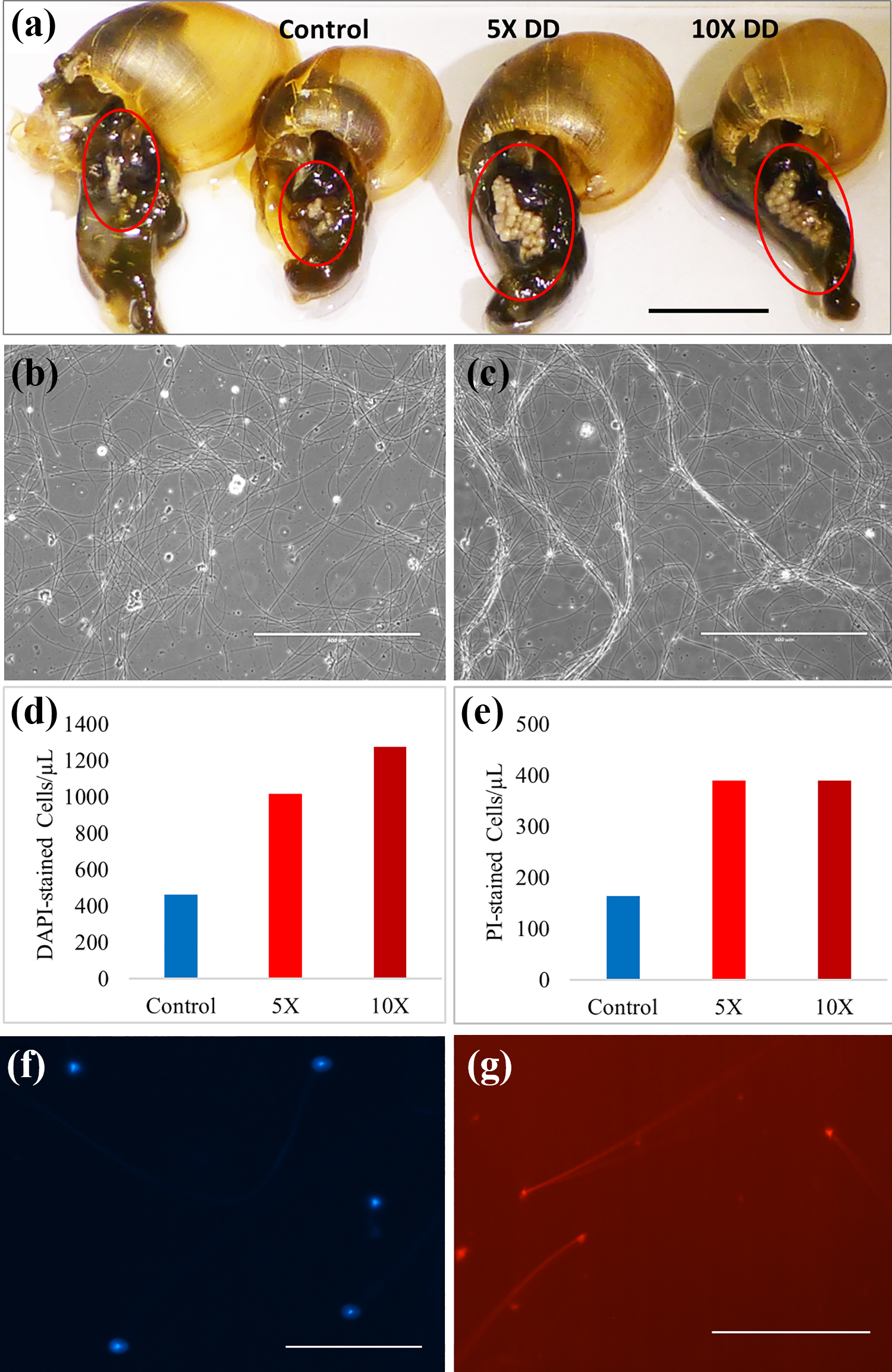
(a) Comparison of seminal vesicle size in dissected adult Lymnaea palustris housed in either APW (two control snails shown), 5X DD (140 µg/L), or 10X DD (280 µg/L) for 2 weeks (bar = 1 cm). (b) Sperm extracted from adult snails in control APW medium (bar = 400 µm). (c) Sperm extracted from adult snails exposed to 10X DD for 2 weeks showing bundles of sperm (bar = 400 µm). (d) Sperm density determined by flow cytometry of DAPI stained sperm from adult snails exposed to either control (APW), 5X DD (140 µg/L), and 10X DD (280 µg/L) for 2 weeks prior to collection. (e) Number of dead sperm (cells/µL) as determined by flow cytometry and PI staining. Sperm were from adult snails exposed to either control APW, 5X, or 10X DD for 2 weeks prior to collection. (f) Sperm stained with DAPI for flow cytometry (bar = 100 µm). (g) Sperm stained with PI for flow cytometry (bar = 100 µm). APW: artificial pond water; DAPI: 4′,6-diamidino-2-phenylindole, dihydrochloride; PI: propidium iodide; DD: diquat dibromide.
Discussion
DD and Wnt signaling
The results of this study provide compelling evidence in support of DD’s interference with Wnt signaling. Our results suggest that the Wnt/Ca2+ pathway is directly inhibited by DD due to evidence for disruption of Ca2+ regulation, impaired Ca2+-dependent adhesion between cells during development, and abnormal development of structures such as the shell. The Wnt/Ca2+ pathway is also an essential regulator of heart development, which we confirmed to be compromised in the presence of DD. 21,40 Interestingly, the noncanonical planar cell polarity (Wnt/PCP) pathway is intricately involved in cytoskeletal rearrangement which is critical during early development for two specific events, both compromised by DD: the first during gastrulation in which an actin meshwork surrounding L. palustris embryos plays a pivotal role in normal developmental patterning; the second is the onset of motility following gastrulation by patches of cilia, beating in a rhythmic motile motion believed to be essential to agitate the capsulary fluid. 28,41 Our results show that DD interferes with the motility of gastrula-to-veliger stage embryos which was completely reversed by co-treatment with 30 mM LiCl (Figure 3(d) and (e)) and causes decreases in average heart rate, which was remediated, though not fully restored by LiCl treatment (Figure 2(e)). It is important to note that after 12 days of chronic exposure, only embryos kept in very low DD concentrations (≤0.25X; 7 µg/L) can be assessed for heart rate, as chronic exposure to higher concentrations yields embryos too developmentally delayed, abnormal, or inviable for heart rate assessment. The fact that control and 10 mM LiCl-treated embryos were unable to survive past 6 days of chronic DD exposure while embryos cocultured with 30 mM LiCl not only survived, but also exhibited normal developmental morphology and heart rates ≥ 40 bpm, is both surprising and provides compelling evidence that DD either directly or indirectly interferes with Wnt signaling.
LiCl treatment alone has been shown to cause both decreased heart rate and inhibition of motility in zebrafish embryos, 24,42 which coupled with transcriptional analysis demonstrate an important role of Wnt signaling in these physiological processes. 38 We provide preliminary evidence for DD’s interference with both canonical and noncanonical Wnt signaling pathways which may be partially or completely remediated by treatment with LiCl. The observed effects on development, motility, and heart rate may be caused by DD directly inhibiting the protein Disheveled (dsh) which activates downstream G-proteins following the binding of Wnt ligands to the Frizzled receptor (Fz), which is known to coordinate all three known Wnt pathways: canonical or Wnt/β-catenin, Wnt/Ca2+, and Wnt/PCP pathways. 43,44 However, more research is needed to validate this hypothesis and explore other potential targets of DD.
DD-induced calcium dysregulation
Our results establish that DD interferes with calcium regulation which may explain in part the significant percentages of developmental delay, abnormality, and death observed in DD-treated embryos (Figure 1(a)). Adjusting calcium levels alone is not sufficient to fully counteract the effects of DD (Figure 1(b)); however, comparable or lower percentages of abnormality were observed in calcium-free medium upon addition of DD. This suggested that DD may initially cause increased calcium uptake, possibly via voltage-dependent calcium channels as demonstrated by Cavalli et al. 10
A more likely explanation may be that DD alters the Wnt/Ca2+ signaling pathway that regulates intracellular calcium release from the endoplasmic reticulum via the inositol phosphate pathway. Wnt signaling and Li+ treatment negatively regulate the inositol phosphate pathway by inhibiting the inositol monophosphatase enzyme that converts inositol monophosphate (IMP) to myoinositol, and inositol polyphosphate 1-phosphatase enzyme which converts membrane-bound phosphatidylinositol 4,5-bisphosphate (PIP2) to IMP, and forms the basis of the myoinositol depletion hypothesis. 22 If Wnt/Ca2+ signaling and subsequently the negative feedback mechanism for the inositol phosphate pathway is inhibited by DD, the conversion of PIP2 to secondary messengers diacylglycerol and inositol triphosphate causes increased Ca2+ release from the endoplasmic reticulum. 20
Through either mechanism, increased intracellular calcium concentration is known to initiate oxidative stress, release of calcium from the endoplasmic reticulum, upregulation of ATP and ROS production in the mitochondria, and initiation of cell death. DD’s inhibition of the Wnt/Ca2+ pathway may provide an explanation for the significant calcium efflux observed following 24-h exposure to DD (Figure 1(c)) at very low concentrations not known to cause oxidative stress. Therefore, calcium dysregulation is a likely mechanism through which oxidative stress is achieved in animal cells exposed to DD. Importantly, at sublethal DD concentrations, brief exposure and an immediate calcium efflux event alone could have serious implications on development following gastrulation when key patterning and signaling events take place.
Decreases in average heart rate with DD exposure
The effect of DD upon heart rate was determined to be both concentration and exposure-time dependent. As hypothesized, absent of any remediation, DD alone at even the lowest concentrations and short exposure times causes significant decreases in heart rate, and also cardiac arrythmia. Additionally, embryos exposed to DD at the gastrula stage developed abnormally small and weak hearts (Online Supplemental Data 3) compared to controls. Both canonical and noncanonical Wnt pathways are integral to heart development and may act antagonistically during the process of cardiac cell patterning, 45 with canonical Wnt signaling keeping cardiomyocyte cells in a proliferative state, and noncanonical Wnt signaling initiating differentiation of cardiac cells. 21,40,42 Wnt 3a and Wnt 8c associated with the canonical Wnt/β-catenin pathway inhibit cardiomyocyte formation, whereas cardiomyocyte formation is stimulated by the noncanonical Wnt 11 via the PCP pathway, which directly inhibits Wnt/β-catenin signaling. 46 These findings further validate the high percentages of abnormality and mortality observed in embryos treated with sublethal concentrations of DD for brief periods of time, as well as the proposed mechanism of DD’s disruption of the Wnt/Ca2+ pathway.
As hypothesized, the combined treatment of low-Ca2+ medium and antioxidants remediated heart rates at most concentrations, even at five times the MCLG of DD as recommended by the EPA. 13 Interestingly, antioxidants alone were effective in improving heart rate compared to controls at DD concentrations ≤ 1X (28 µg/L), where DD-induced oxidative stress is not reported to occur. 1,8 Although DD has not been demonstrated in the literature to elicit significant oxidative stress at concentrations ≤ 44 µg/L, 0.5X (14 µg/L), DD may elicit slight oxidative stress, increasing biological defenses to counter the effects of DD following acute exposure (Figure 2(b)). However, it is important to note that embryos chronically exposed to all concentrations of DD exhibit no such improvements over time in DD-induced heart rate defects (Figure 2(a) and (b)). Antioxidant/prooxidant treatment alone is known to increase average heart rate; 38 therefore, additional investigation is needed to determine the mechanism by which antioxidants increase heart rate in DD-treated embryos at concentrations below the suspected ROS initiation threshold (44 µg/L).
The effects of DD on sperm
We have observed an increase in sperm abundance and decrease in sperm viability in DD-treated snails, which may indicate that sperm release and/or maturation is inhibited by DD. A recent study using mice found that sperm maturation and release is regulated by Wnt/Ca2+ signaling in the epididymis. 17 This research confirms the importance of Wnt signaling in transcriptionally silent sperm for continued development, maturation, and motility. Our results suggest that DD, by inhibiting Wnt signaling, may attenuate sperm maturation and release, causing enlargement of the seminal vesicles observed in adult snails exposed to DD (Figure 4(a)). Additionally, higher mortality in sperm extracted from DD-treated snails may be due to morphological abnormalities at the cellular level, oxidative stress, and initiation of programmed cell death. 6,47
Sperm extracted from snails exposed to 5X and 10X DD (140 and 280 µg/L) for 1- and 2-week periods exhibited erratic and prolonged motility compared to controls, remaining motile past cessation of motility in control sperm (data not shown). Aberrant motility appeared to increase with increased density and therefore increased DD concentration. Impaired StAR expression and hormonal regulation, 2 hypertrophy of the seminal vesicles as a possible consequence of DD-induced disruption in Wnt signaling, and increased ROS formation are all factors that likely play a role in producing the observed irregularities in sperm abundance, viability, and motility, though the exact mechanism is unclear; these may all contribute to the severely impaired fecundity of snails exposed to DD.
Proposed models for the mechanism of DD
The results from this study and related literature support dual mechanisms for the action of DD upon animal cells. Calcium dysregulation and Wnt signaling likely play a role in both mechanisms. At high concentrations (≥44 µg/L), DD initiates calcium influx through voltage-dependent calcium channels or increases calcium release from the endoplasmic reticulum (ER) via aberrant Wnt/Ca2+ signaling, such that these mechanisms work either separately or agonistically to increase intracellular calcium concentration. As intracellular calcium concentration increases, ATP production and subsequently ROS formation are upregulated in the mitochondria. Though a negligible amount of ROS formation accompanies ATP production under normal conditions, an abnormal ATP production increase is known to lead to potentially lethal levels of intracellular ROS. 6,10,47,48 As ROS accumulates in the cell, more calcium is released from the ER, establishing a positive feedback loop of intracellular calcium release from the ER and ROS formation in the mitochondria. 11,49 Following the establishment of the positive feedback loop, large amounts of calcium are effluxed from the cell, triggering apoptotic cell death (Figure 5(a)).
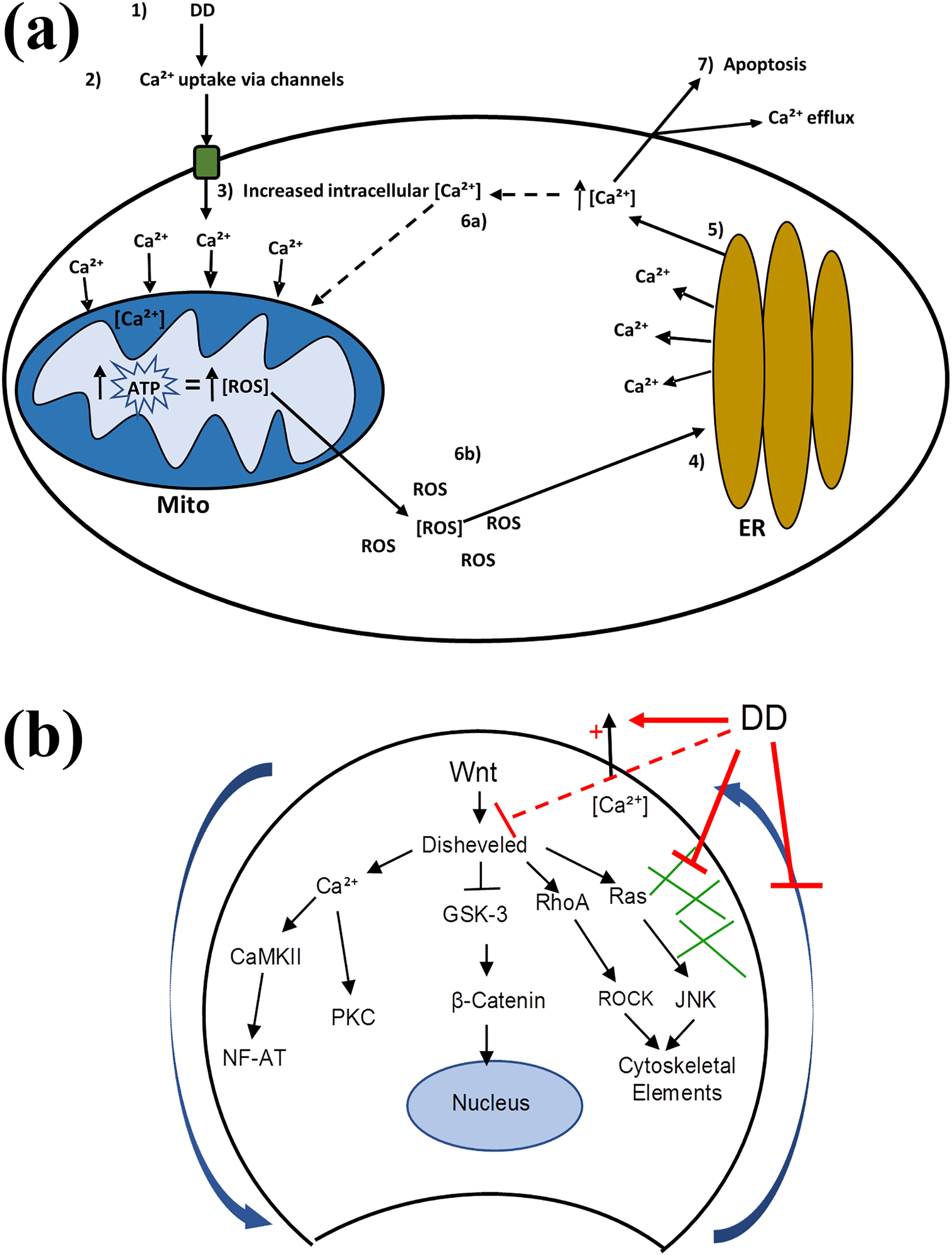
(a) Proposed mechanism of DD at concentrations ≥ 44.4 µg/L. (1) Cells exposed to high concentrations of DD; (2) DD causes calcium uptake from the medium through calcium channels; (3) intracellular concentration of calcium rises and is taken up by the mitochondria, increasing ATP production and consequentially, ROS formation; (4) and (5) the increased intracellular concentration of ROS causes calcium release from the endoplasmic reticulum as a stress-response mechanism; (6a) and (6b) the increase of ROS and release of calcium from the ER establishes a positive feedback loop; and (7) high intracellular levels of calcium lead to massive calcium efflux, triggering apoptotic cell death. (b) Proposed mechanism of DD in animal cells at all concentrations: DD inhibits both canonical and noncanonical Wnt signaling pathways (potentially through inhibition of disheveled), disrupts motility (blue arrows) mediated by cytoskeletal elements (green lines), and causes significant calcium efflux (potentially via the Wnt/Ca2+ pathway). ER: endoplasmic reticulum; Mito: mitochondrion; DD: diquat dibromide; ROS: reactive oxygen species.
At low DD concentrations (≤28 µg/L), evidence suggests that DD directly disrupts both canonical and noncanonical Wnt signaling, possibly via interaction with dsh proteins. 44,50 This may lead to altered Wnt/Ca2+ signaling, which regulates intracellular calcium, cellular adhesion, motility, and heart development; the PCP pathway which through the RhoA-ROCK and Ras-JNK pathways is required for cytoskeletal elements and arrangement and antagonistic development of other Wnt pathways; and canonical Wnt signaling that is critical for embryonic growth and developmental patterning (Figure 5(b)). It is probable that the low-dosage effects of DD that we have described (Figure 5(b)) are occurring at all concentrations, while the high-dosage effects of DD leading to ROS-induced cell death (Figure 5(a)) occur only at DD concentrations ≥ 44 µg/L.
Conclusions
The results of this study establish a strong connection between the observed DD-induced effects of developmental abnormality, calcium dysregulation, cytoskeletal damage, and heart rate impairment, which are all intricately coordinated through canonical and noncanonical Wnt pathways. The findings of this study call for future investigation into the dynamics of DD’s interaction with elements of Wnt signaling, canonical and noncanonical pathway crosstalk, and the complex differences of Wnt signaling in vertebrates compared to invertebrates. These and future experiments may highlight the underrepresented danger that DD poses to nontarget organisms both ecologically, and particularly to human health and the hazards of frequent exposure to DD-containing pesticides.
Supplemental material
Supplemental_Data_Legends_FINAL - Cellular effects of diquat dibromide exposure: Interference with Wnt signaling and cytoskeletal development
Supplemental_Data_Legends_FINAL for Cellular effects of diquat dibromide exposure: Interference with Wnt signaling and cytoskeletal development by Amaris S Jalil, Sneha B Reddy and Carol Zygar Plautz in Toxicology Research and Application
Footnotes
Acknowledgements
Samantha Mines Schildt and Colleen J Nolan made important contributions to this work. Thanks to Robert Warburton for assistance with flow cytometry (WV-INBRE P20GM103434) and to Dan DiLella for assistance with atomic absorption.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported through student research funding provided by the NASA-WV Space Grant Consortium and SOARS at Shepherd University (SURE Program of WV-EPSCoR/West Virginia Science & Research).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
