Abstract
Exposure of organisms to environmental contaminants is a growing concern. We have investigated the effects of the individual active ingredients of the herbicide Roundup (glyphosate and diquat dibromide [DD]) since Roundup causes alterations in reproduction, mortality, and development in the aquatic snail
Introduction
An abundance of herbicides and pesticides in the environment poses an increasing threat to nontarget species. These chemicals may reach freshwater habitats by surface runoff, groundwater contamination, or aerial drift. Roundup is a widely-used and highly water-soluble herbicide. Its main active ingredient, glyphosate, has been widely studied
DD is a broad-spectrum herbicide, often used in combination with other herbicides. Due to the rapid dissipation of diquat in water
14
and subsequent binding to sediment and soil, DD has been assumed safe for direct application to water systems to control aquatic weeds, and US EPA Tolerance Reassessment Progress and Risk Management Decision (TRED) reports indicate no harm will result from exposure to DD within established and proposed tolerances, up to 2 mg/L for fish and 20 mg/L for shellfish. This has led to increased exposure of freshwater animals to DD, organisms for which the effects of this compound are largely uncharacterized.
15
Depending upon application time, proximity to agricultural areas, and rainfall, runoff to surface water causes Roundup concentration to regularly equal or exceed the US EPA recommended maximum contaminant level (MCL) of glyphosate for human drinking water (0.7 mg/L) as well as the maximum contaminant level goal (MCLG) of DD (0.02 mg/L) in many regions of the United States and worldwide.
7,16,17
Studies have documented altered fecundity, developmental delays,
18
and altered transcriptional and enzymatic activity of markers of oxidative stress following acute DD treatment in
Since steroid acute regulatory (StAR) protein is the rate-limiting factor in steroidogenesis
23,24
and has been shown to be disrupted in mammalian cells exposed to a variety of compounds, we sought to determine whether StAR abundance in molluscs is altered
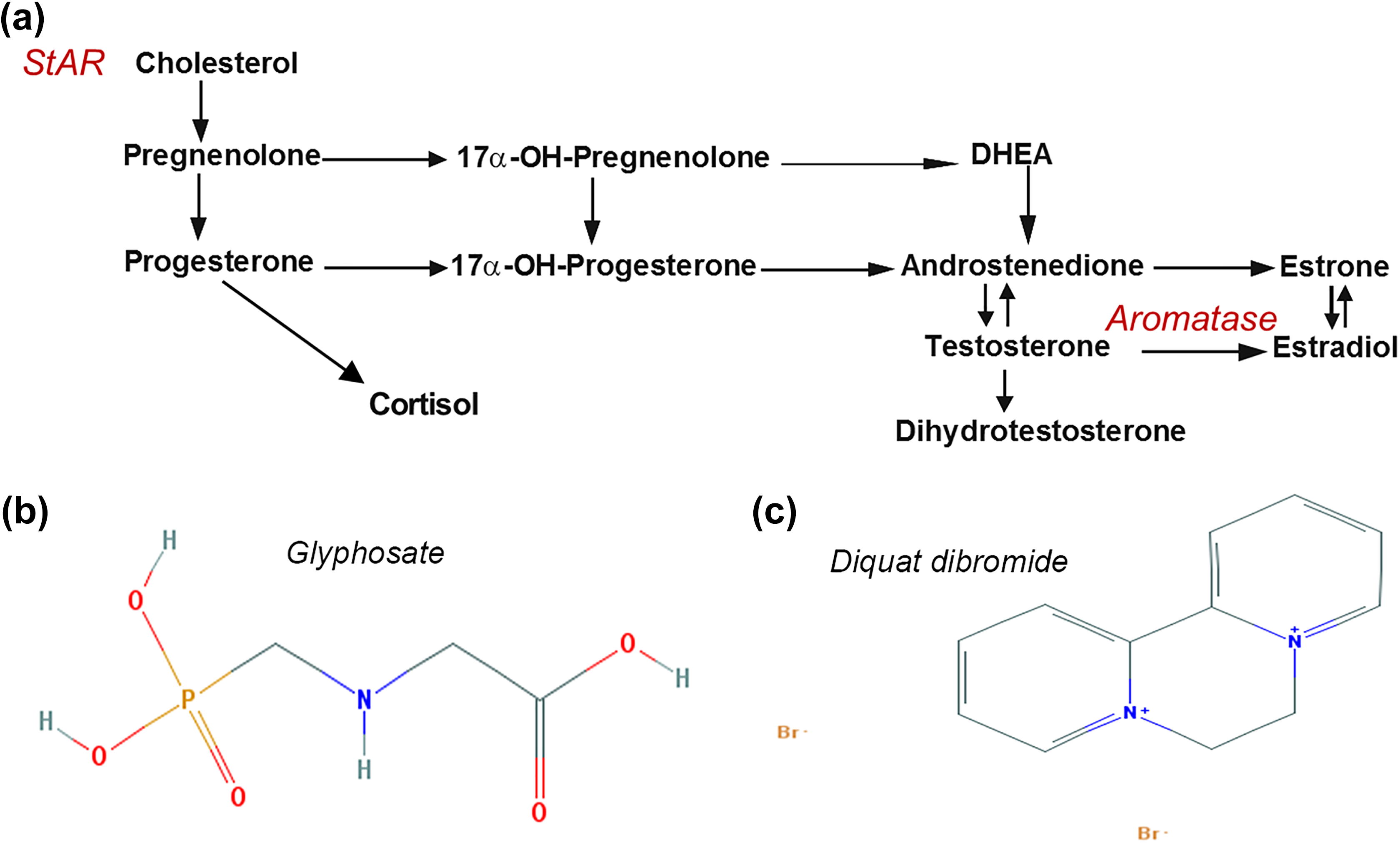
(a) Steroidogenic pathway in molluscs. StAR is the rate-limiting factor in steroidogenesis and ushers cholesterol to the inner mitochondrial membrane. P450-aromatase (CYP19 or aromatase) converts testosterone to estradiol. Panel adapted from Janer and Porte. 28 (b and c) Chemical structures of herbicides used in this study, generated by National Center for Biotechnology Information PubChem Compound Database: (b) structure of glyphosate and (c) structure of diquat dibromide. StAR: steroid acute regulatory protein.
Methods
Animal culture and treatment
Laboratory-reared
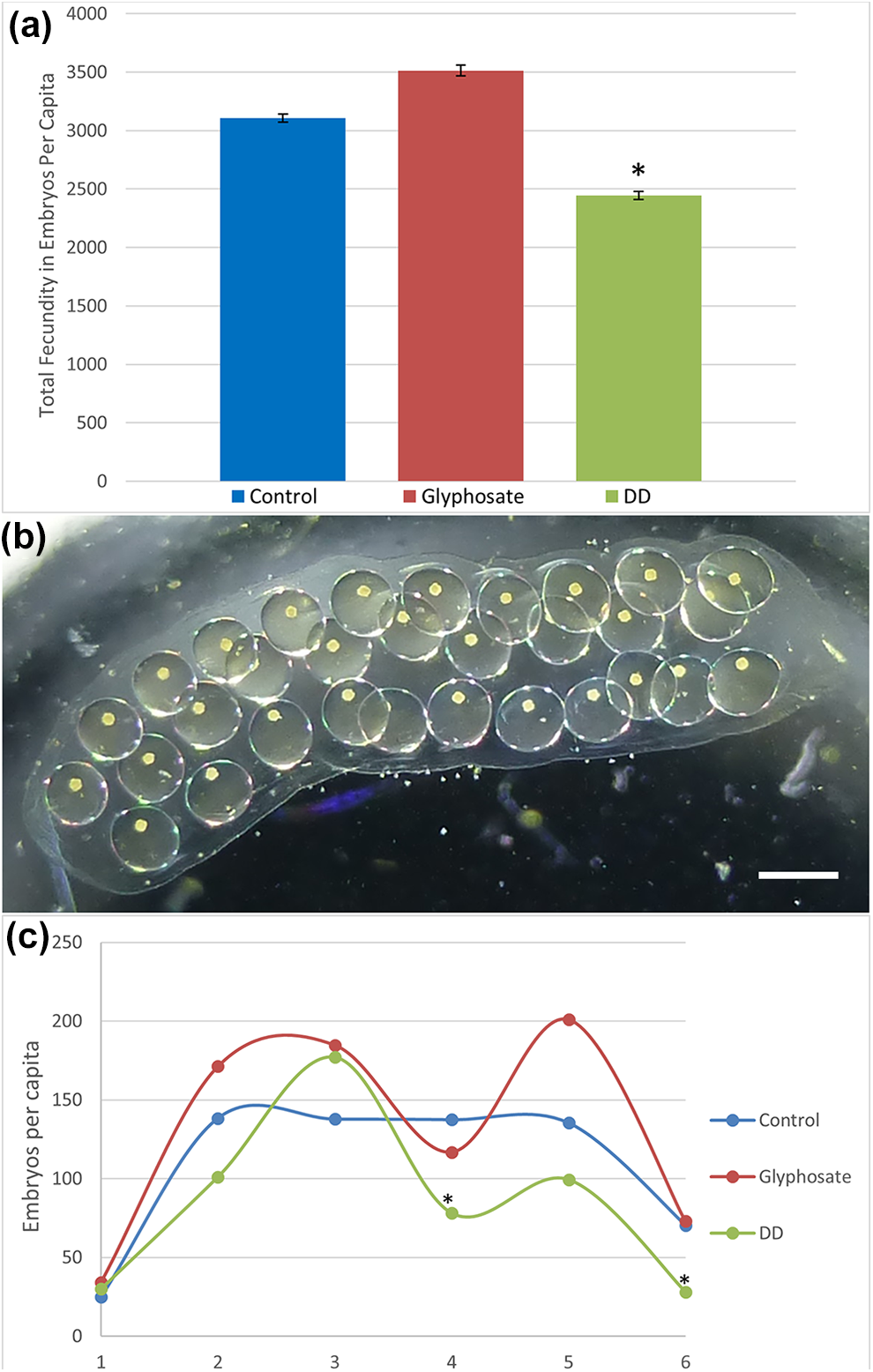
Fecundity of
Three mesocosms were established for each of three treatment types (
Steroid analysis
Hemolymph was drawn weekly (once at outset of study, and following each of 3 weeks of treatment) from snails using a noninvasive, nonlethal method whereby the animal is induced to extrude hemolymph by a poke on the foot with a pipette tip.
35
Hemolymph was flash frozen in liquid nitrogen and stored at −80°C until analysis by enzyme-linked immunosorbent assay (ELISA). Hemolymph, approximately 50 µL per animal per draw, was pooled from all individuals in one mesocosm at a given time point. ELISA analyses for estradiol and testosterone were conducted and data analyzed according to manufacturer’s instructions (Cayman Chemicals, Ann Arbor, MI 582701, 582251). Absorbance of samples and standards was read in BioTek, Winooski, VT Synergy HT Multi-Detection Microplate Reader and calculations made in Microsoft Excel (
Protein analysis
To analyze differential aromatase abundance, ovotestes and associated structures (prostate and albumen glands) were harvested at the end of the study period. Tissues were mechanically disrupted in RIPA buffer (ThermoFisher, Waltham, MA 89900) containing protease inhibitor cocktail (ThermoFisher 78425) according to manufacturer’s directions, and snap frozen in liquid nitrogen followed by storage at −80°C prior to analysis by Western blot. Samples were
To analyze StAR abundance in whole snail, ovotestis, kidney, or brain, tissues were isolated and disrupted similarly using ReadyPrep Protein Extraction Kit (Bio-Rad 163-2086). Samples were quantified using EZQ Protein Quantitation Kit (Invitrogen Carlsbad, CA/ThermoFisher R33200), and 30 µg of total protein or 10 µg of individual organ protein was loaded in each lane for 12% PAGE and transfer as above. Primary anti-StAR at 1:1000 and subsequent secondary, treatment, and analysis as described above to quantify relative intensity of bands in control and treated samples.
Statistical analysis
Paired
Results
Fecundity
Embryo counts collected from each mesocosm were normalized by the number of snails alive at that time point to ensure accurate representation of the fecundity per individual for each treatment type over the 3-week study (Figure 2(a)), since a low level of spontaneous mortality occurred. As the study progressed, DD-treated snails exhibited a marked decrease in embryo production (*
Steroid hormone levels
Hemolymph extracted from animals prior to the outset of the study and weekly for 3 weeks during treatment was analyzed by ELISA for estradiol and testosterone concentrations. In glyphosate-treated animals, we observed a consistent trend of estradiol concentration comparable to or exceeding that of control animals (Figure 3(a) and (b)). Testosterone levels in glyphosate-treated animals are statistically comparable to control animals (Figure 3(c);
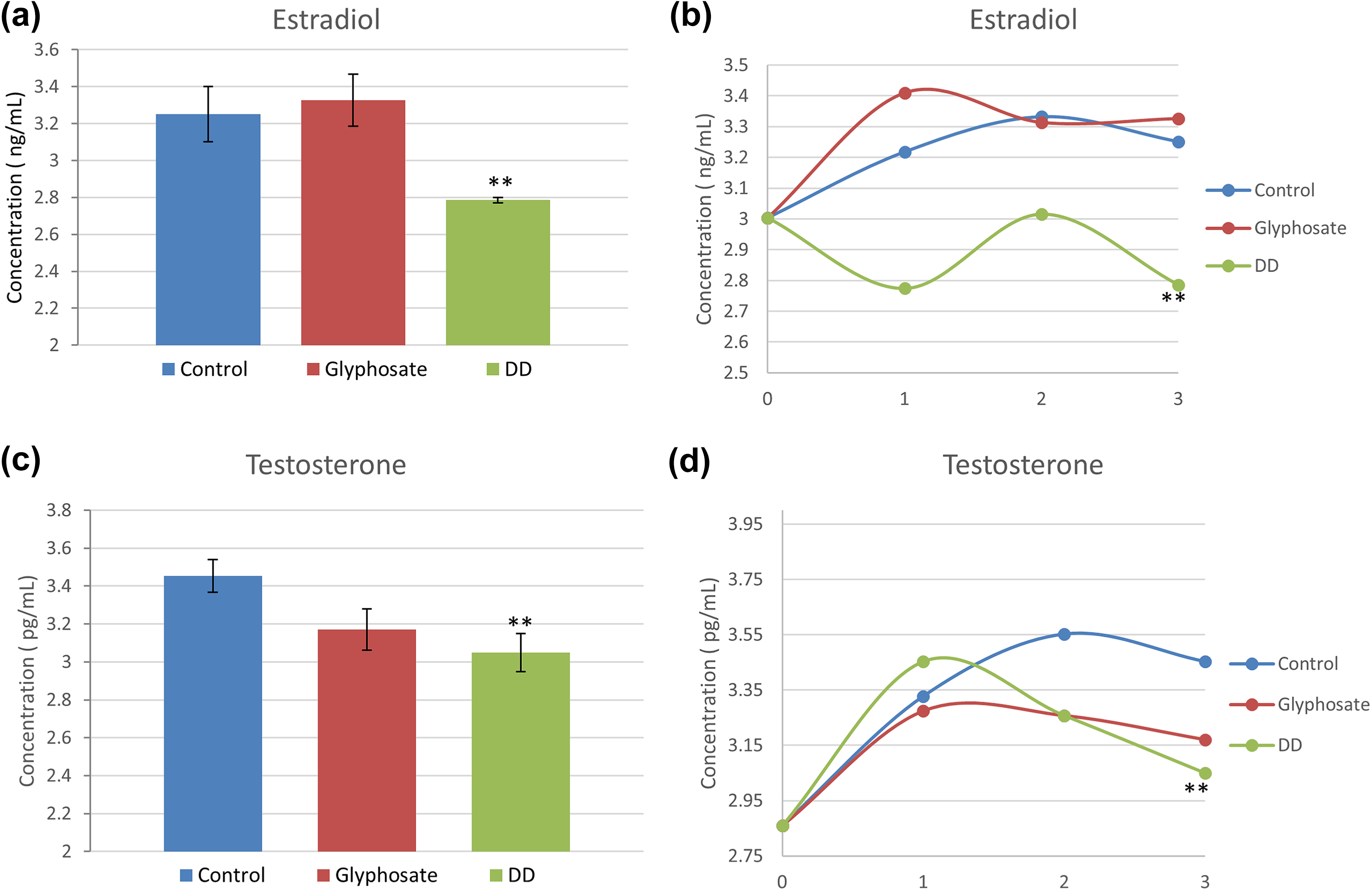
Testosterone and estradiol levels measured by ELISA in
Hemolymph of animals treated with DD contains significantly lower estradiol (Figure 3(a) and (b)) and testosterone (Figure 3(c) and (d)) concentrations relative to control animals. The differences were most pronounced at the 3-week mark, when estradiol and testosterone concentrations in DD-treated animals are lowest relative to controls (**
Western blot analyses
Western blot analysis on isolated ovotestes from the various treatment groups at the conclusion of the 3-week study revealed that glyphosate-treated samples on average had a larger quantity of aromatase protein compared to control samples, and DD-treated samples had a lower quantity of aromatase protein compared to control samples. Although some DD-treated ovotestis samples had comparable aromatase or approximately two- to three-fold less than control samples, most had little to no detectable aromatase, although GAPDH detection reveals there was equivalent protein present in the sample (Figure 4(a)). Ovotestes from glyphosate-treated animals contained the highest levels of aromatase protein across all samples analyzed (
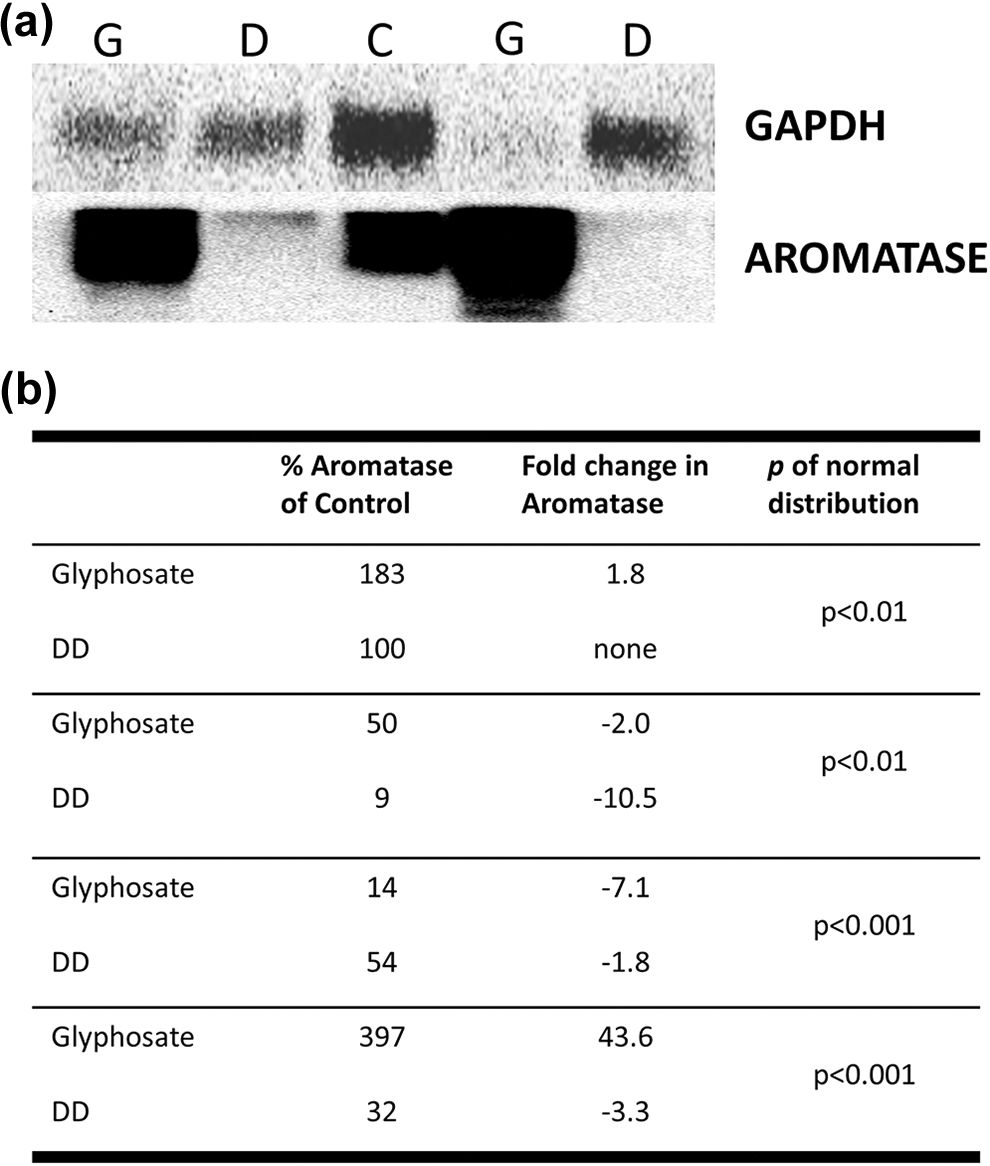
(a) Representative Western blot of ovotestes harvested from animals exposed to 3-week treatment in control APW, glyphosate (3.5 mg/L), or DD (140 µg/L). Top: Western blot of GAPDH for internal standardization of samples. Bottom: Western blot of aromatase (G: glyphosate; D: DD; C: control APW). (b) Percent aromatase quantity in treated ovotestes relative to aromatase quantity in control ovotestes for four Western blots, demonstrating high variability of aromatase quantity in glyphosate-treated samples and consistent though variable decrease in aromatase quantity in DD-treated samples. Aromatase quantity in treated ovotestes relative to aromatase quantity in control ovotestes is also represented as fold change in aromatase.
StAR protein is readily detected in snail brain, kidney, and ovotestes, but is not detected in other snail organs tested (lung, heart). Following 6-week chronic treatment in Roundup, DD, or glyphosate, whole snails as well as isolated kidney, brain, and ovotestes were analyzed to quantify decrease in StAR expression (Figure 5). Glyphosate-treated whole animal samples exhibited a 42% reduction in StAR, Roundup-treated samples on average exhibited a 65% reduction in StAR, and DD-treated whole animal samples exhibited a 70% reduction in StAR (Figure 5(a) and (b)), suggesting that both active ingredients of Roundup may contribute to the downregulation of StAR in the whole animal when chronically exposed. When individual organs from chronically treated animals were analyzed, the organs were affected differentially by the same treatments (Figure 5(c)). The kidney exhibited a less than two-fold reduction in StAR following 6-week treatment with Roundup (62% of control) or DD (55% of control), while the brain exhibited greater than four-fold reduction in StAR following 6-week treatment with Roundup (22% of control) or DD (23% of control), and the ovotestis was the steroidogenic organ most affected by Roundup (6% of control) or DD (9% of control) with greater than 13-fold reduction in StAR.

(a) Representative Western blots for StAR of whole snails exposed for 6 weeks to control (APW) conditions or supplemented with Roundup (19.5 mg/L), glyphosate (3.5 mg/L), or DD (140 µg/L). Representative samples from 12 control snails, 3 glyphosate-, 3 Roundup-, and 3 DD-treated snails are shown (30 µg protein total loaded per lane). C: control; G: glyphosate; R: Roundup; D: diquat dibromide. (b) Percent StAR quantity in treated whole snail extracts relative to StAR quantity in control whole snail extracts; all treated individuals except one in glyphosate exhibited a reduction in StAR, for an average reduction in StAR quantity of 41.7% for glyphosate, 65.0% for Roundup, and 70.3% for DD; average fold change in StAR in each treatment group is also shown. Each treatment resulted in significant reduction in StAR protein compared to control whole snail StAR content by
A summary of the individual effects of glyphosate and DD in our system is found in Table 1. While glyphosate is associated with a transient rise in fecundity, a trend toward decreased testosterone and increased estradiol and fecundity leading to a modest increase in estradiol to testosterone (E:T) ratio, and potential elevation of aromatase level, the main impact of 3-week glyphosate treatment is a reduction in StAR level. In contrast, the effects of DD treatment include significant decreases in StAR, fecundity, testosterone, and estradiol leading to a modest reduction in E:T ratio, and a trend toward lower aromatase levels.
Trends in reproductive indicators exhibited overall in snails exposed to glyphosate or DD relative to control snails.

StAR: steroid acute regulatory protein; DD: diquat dibromide; E:T: estradiol to testosterone ratio.
a
b The control E:T value is 0.95.
Discussion
Fecundity of
Given the differences in concentrations of circulating gonadal steroids, we additionally sought to analyze whether the abundance of aromatase was a potential target of DD and/or glyphosate, since aromatase is directly responsible for the conversion of testosterone to estradiol. Studies by Walsh et al.
1
demonstrated that Roundup decreases the expression of StAR and the ability of a cyclic adenosine monophosphate analog (dibutyryl cAMP) to induce steroidogenesis in cultured MA10 cells, while Richard et al.
37
observed that low doses of Roundup and glyphosate both decreased aromatase activity and Roundup decreased the amount of aromatase mRNA present in cultured placental cells. A recent study by Uren Webster et al.
36
reported that a low dose of Roundup decreased expression of testicular steroidogenic enzymes in zebrafish and increased expression of these enzymes in the ovary. Given that hermaphrodites possess both ovaries and testes, the findings of these research groups suggest a similar effect could contribute to suppressed steroidogenesis that subsequently decreases gametogenesis and reduces fertility and fecundity seen in
It is accepted that Roundup and its components are not alone in altering reproduction as there are a plethora of herbicides and pesticides that have been identified as endocrine disruptors
38
and many of these endocrine disruptors are known to impact gonadal steroidogenesis in aquatic and terrestrial species. For example, another dipyridyl compound, paraquat, has been demonstrated to exert reproductive disturbances
39
in freshwater snails. In addition, exposure to atrazine, one of the most commonly used pesticides, has been shown to produce feminization in
Many elements of the steroidogenic pathway have been found in common between humans and molluscs, and numerous studies demonstrate that sex steroids are synthesized from cholesterol precursors in molluscs. 28,43 There is evidence that elevated estradiol levels increase and extend gastropod mollusc oviposition in seasonal reproducers 44 ; this is consistent with our observations that higher fecundity, increased aromatase abundance, and elevated estradiol levels are present in glyphosate-treated snails. Lazzara’s study 45 clearly links testosterone and estradiol levels to reproductive output in zebra mussels; endocrine disruption by tributyltin and other compounds in these bivalve molluscs led to reduced testosterone levels, an altered androgen/estrogen ratio, and reduction in fecundity.
While there are commonalities in steroidogenesis, there may be differences in enzymes and/or enzymatic activity between species. Although cytochrome P450 enzymatic activity has been widely demonstrated in invertebrates including molluscs, aromatase
27
or aromatase-like
46
activity specifically (CYP19/p450-AROM) has been less well-characterized. This may be due to changes in this enzyme over evolutionary time, and the resulting difficulty in identifying an invertebrate homologue to this and other steroidogenic pathway elements.
47
In 1974, De Longcamp et al.
48
identified a putative gonadal steroidogenic pathway in molluscs that was similar to that in vertebrates and confirmed the presence of testosterone in gonads. A more recent review by Matthiessen and Gibbs
49
discussed the production of intersex and imposex individuals in neogastropod molluscs by addition of tributylin which increased testosterone and that this effect could be blocked by use of an androgen agonist. Bearing these studies in mind, it will be important to further characterize steroidogenesis in molluscs and identify the enzymes responsible for this pathway in
The level of conservation between higher vertebrates and molluscs of molecules and activities in the steroidogenic pathway is becoming ever more apparent, and points to the increasing usefulness of molluscs as indicator organisms in which we are able to not only determine that toxicological harm exists, but to elucidate the specific mechanisms involved. Further, it is becoming established that hormones such as testosterone and estradiol likely play physiological roles in mollusc reproduction, and that such steroid sex hormones act as endogenous modulators of gametogenesis in molluscs.
50,51
It is clear that using an aquatic invertebrate, such as
Footnotes
Acknowledgments
Jessica Cain Seal, Jessica Chaney Hansroth, Samantha Mines Schildt, Christopher Seal, Allison Brooks, Preetha Phillips, Priya Arumuganathan, and Shruthi Sreekumar made important contributions to this work. StAR antibody was kindly supplied from the laboratory of Dr Douglas Stocco, Texas Tech University.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Student research funding was provided by the NASA-WV Space Grant Consortium and SOARS at Shepherd University (SURE Program of WV-EPSCoR/West Virginia Science & Research).
