Abstract
Chemicals classified as known human carcinogens by International Agency for Research on Cancer (IARC) show a low level of concordance between rodents and humans for induction of pulmonary carcinoma. Rats and mice exposed via inhalation for 2 years show a low level of concordance in both tumor development and organ site location. In 2-year inhalation studies using rats and mice, when pulmonary tumors are seen in only male or female mice or both, but not in either sex of rat, there is a high probability that the murine pulmonary tumor has been produced via Clara cell or club cell (CC) metabolism of the inhaled chemical to a cytotoxic metabolite. Cytotoxicity-induced mitogenesis increases mutagenesis via amplification of the background mutation rate. If the chemical being tested is also negative in the Ames Salmonella mutagenicity assay, and only mouse pulmonary tumors are induced, the probability that this pulmonary tumor is not relevant to human lung cancer risk goes even higher. Mice have a larger percentage of CCs in their distal airways than rats, and a much larger percentage than in humans. The CCs of mice have a much higher concentration of metabolic enzymes capable of metabolizing xenobiotics than CCs in either rats or humans. A principal threat to validity of extrapolating from the murine model lies in the unique capacity of murine CCs to metabolize a significant spectrum of xenobiotics which in turn produces toxicants not seen in rat or human pulmonary pathophysiology.
Executive summary
In the text to follow, a number of concepts will be developed and supported including the following: Clinical observations of the carcinogenicity of workplace chemicals predated conduction of the first animal cancer bioassays. Rats and mice exposed via inhalation for 2 years show a low level of concordance in both tumor development and organ site location. Chemicals classified as known human carcinogens by IARC show a low level of concordance between rodents and humans for induction of pulmonary carcinoma. Cytotoxicity-induced mitogenesis increases mutagenesis via amplification of the background mutation rate. Mitogenesis in the presence of genotoxic agents synergizes the carcinogenic effect. Increased lung cancer rates in cigarette smokers with chronic obstructive pulmonary disease (COPD) represent a clinical demonstration of this process. Pulmonary inflammation can induce a localized hypoxia that selects for preferential survival and clonal expansion of epidermal growth factor receptor (EGFR)–positive tumors by non-genotoxic mechanisms. In many scenarios, oxidative stress from pulmonary inflammation can exceed the amount of reactive oxygen species (ROS) and reactive nitrogen species (RNS) resulting from direct chemical damage to mitochondria. Mice but not humans display high spontaneous background lung cancer rates. In contrast with high spontaneous background lung cancer rates, cigarette smoking duration is a stronger predictor of lung cancer mortality than is cigarette smoking intensity. Mouse lung tumors are much less histologically diverse than human lung cancers. Human and murine lung tumors display different behavior patterns including reduced vascularization, stromatogenesis, and metastasis in murine tumors. The histogenetics of human and mouse lung cancers differ significantly. Mice have a larger percentage of Clara cells or club cells (CCs) in their distal airways than rats, and a much larger percentage than in humans. The CCs of mice have a much higher concentration of metabolic enzymes capable of metabolizing xenobiotics than CCs in either rats or humans. The increased xenobiotic metabolic capacity of mouse CCs is proportional to its increased tendency to develop chemically induced lung tumors as compared with the rat. A principal threat to validity of extrapolating from the murine model lies in the unique capacity of murine CCs to metabolize a significant spectrum of xenobiotics which in turn produces toxicants not seen in rat or human pulmonary pathophysiology.
Section one: Historical role of cancer bioassays
The original investigations of workplace-related cancers were initiated by the observations of unusual tumors in association with certain occupations 1 including chimney sweeps, 2,3 mule spinners, 2,4,5 aniline dye workers, 6 clock workers using radium-impregnated dye, 7 and chemical workers exposed to benzene. 8 Following detection in humans, the causative agents were later studied in animals. 9 –14
In general, the ongoing efforts to remove carcinogens from the workplace in developed countries have resulted in amelioration of exposure. 15 The paradigm of testing for chemical induction of cancer in rodents has shifted from confirmation of observation to intended prophylaxis. The misperceptions resulting from anhistoric presentism have inverted the chronological sequence of clinical observation and animal testing and inappropriately elevated animal testing to a preeminent role in detecting rather than describing environmental carcinogens. 16 Table 1 shows a representative subset of IARC-classified agents that were recognized as cancerous (or potentially cancerous) prior to testing in any animal. 17 –45 The average time lapsing from first reported observation or collection of formal epidemiological data to conduction of the first animal tests is 64 years (range 9–387 years). The intent of this animal testing was to provide data to substantiate that the suspect agents were indeed dangerous and that certain manufacturing, handling, shipping, and storage precautions and or regulations were needed for the safety of workers and consumers.
IARC agents where clinical observation or epidemiological cancer data preceded animal cancer testing.
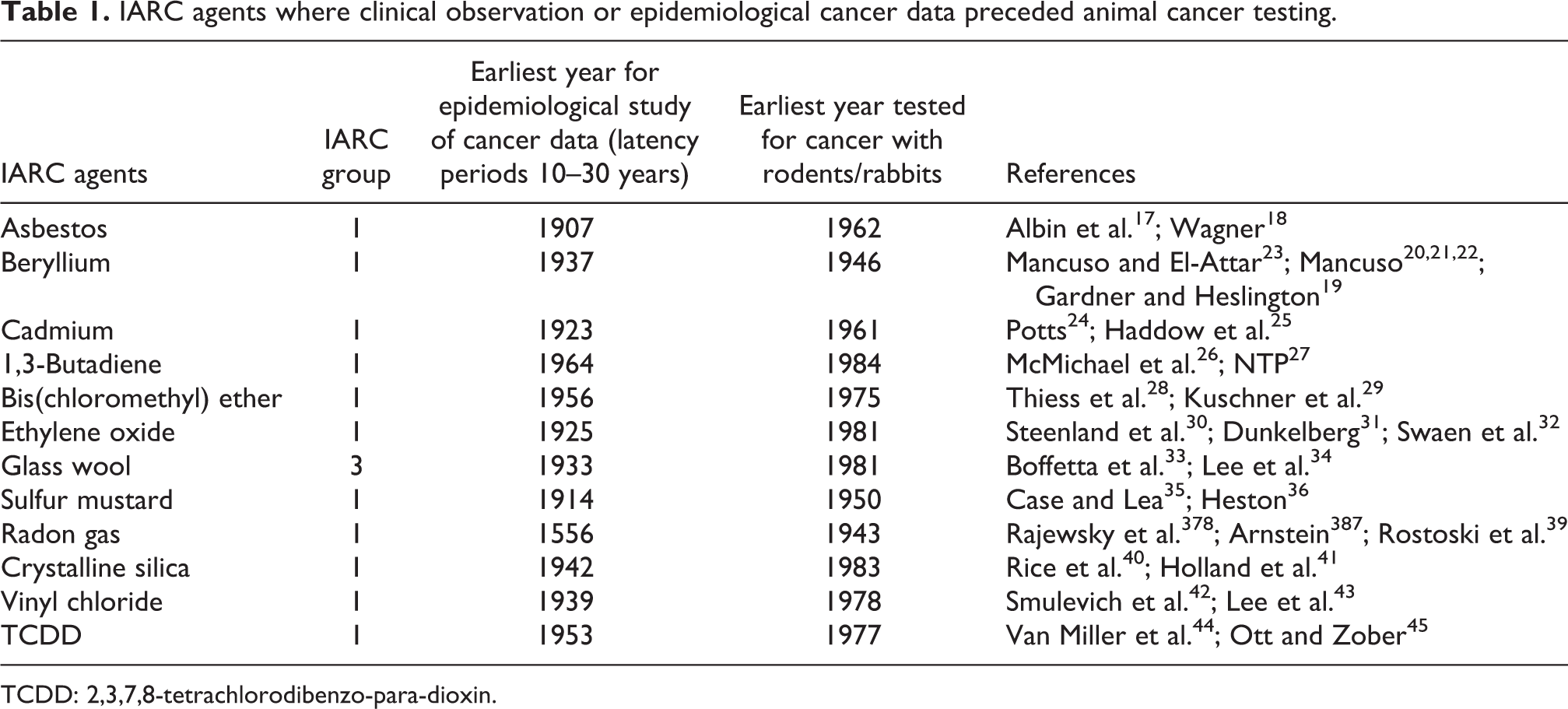
TCDD: 2,3,7,8-tetrachlorodibenzo-para-dioxin.
In addition to transposing the chronological sequence of clinical observation and animal studies, the degree of concordance between rodents and humans regarding pulmonary carcinoma has been overstated. 16 In 2-year National Toxicology Program (NTP) inhalation studies, Smith and Anderson 46 have shown that concordance of pulmonary tumor formation between phylogenetically similar rats and mice is low, thereby questioning the potential degree of concordance between much more phylogenetically distant rodents and humans. Krewski 47 has reported the concordance between humans and mice for lung tumors associated with IARC group 1 (known human) carcinogen exposure. Using a κ statistic at 90% confidence, IARC group 1 agents showed higher concordance than did group 2a, 2b, and group 3 chemicals and mixtures. The κ value for the concordance between humans and mice in group 1 chemicals was 0.17 (−0.2–0.53). A κ value of between 0.01 and 0.20 represents only a slight concordance. The concordance (human vs. mouse) for lung tumors was slight and equal to tumors of the urinary system (0.12 (−0.05–0.12)). The concordance (human vs. mouse) for all other organ systems was higher than the lung system (κ 0.51–0.64 = moderate to substantial).
Section two: “Clear” evidence of neoplasia in NTP studies is required to facilitate statistical and mechanistic analysis
Over the history of the NTP testing program up through early 2018, 594 different 2-year rodent bioassays have been conducted via different routes of exposure including inhalation, feed, gavage, drinking water, dermal, and intraperitoneal injection. These 594 bioassays resulted in successful completion of final NTP reports for 479 chemicals or chemical mixtures, with additional three chemicals described in two Report on Carcinogens (RoC) reports. Our research group has statistically and mechanistically analyzed the results from all completed final NTP reports. 46,48 –52
NTP considers results from the Ames assay test to be very important in its deliberations as illustrated by the following statement from a recent RoC.
53
DNA reactivity combined with Salmonella mutagenicity is highly correlated with induction of carcinogenicity in multiple species/sexes of rodents and at multiple tissue sites.
54
A positive response in the Salmonella test was shown to be the most predictive in vitro indicator for rodent carcinogenicity (89% of the Salmonella mutagens are rodent carcinogens).
55,56
Additionally, no battery of tests that included the Salmonella test improved the predictivity of the Salmonella test alone.
Section three: Interactive role of inflammation and proliferation
Non-genotoxic chemicals frequently induce neoplasia via increased cellular proliferation
In the NTP database, 180 chemicals have been identified whose current genotoxicity test results are negative but that induce at least one tumor in either rats or mice. 52 Our examination of the NTP database suggests that it is not possible to retrospectively determine which of these 180 chemicals are definitively non-genotoxic due to the absence of independent verification for each particular assay. While the validity of an Ames test on a particular chemical in the NTP database might not be definitive, when all routes of administration are combined, the overall dependability of the Ames test results in the NTP database is high. The overall validity of the NTP database Ames results are demonstrated by correlation of Ames “positive” status, Ames “negative” status, categorical rank (1–48), and ordinal rank (1–135). The Mann–Whitney–Wilcoxon rank sum test shows that the trend in Ames versus category ranking is highly significant (Z = −5.69; p value near 0); that is, positive Ames results are strongly associated with categorical ranks of increased tumorigenicity. The Mann–Whitney–Wilcoxon rank sum test shows that the trend in Ames versus ordinal ranking is highly significant (Z = −5.65; p value near 0); that is, positive Ames results are strongly associated with ordinal ranks of increased tumorigenicity. 50
A large and elegant body of research conducted in the 1980s explains the induction of tumors in 2-year rodent bioassays by non-genotoxic chemicals. The mechanism is amplification of the background mutation rate via cytotoxicity induced by high doses of the test chemicals, thereby leading to increases in reparative cellular proliferation rates. 52 In the 1980s, Cohen and colleagues conducted a series of studies that demonstrated that cellular proliferation could amplify the background mutation rate thereby increasing tumor formation in experimental animals. 66 –69 The studies of Moolgavkar and Knudson also played an important role during this era. 70 Throughout the 1990s, Ames and Gold incorporated these new findings into their thinking, resulting in a series of publications, one of which is highlighted by Smith and Perfetti, 52 that is, Ames and Gold. 71 This paper published in Science was actually a commentary on the Cohen and Ellwein paper published in the same issue. 67 A broader model incorporating the potential environmental influence on cell number, proliferation rates, and mutation rates (secondary to DNA reactive carcinogens) formed the basis for the Cohen and the Moolgavkar models.
In 2015 and 2017, Tomasetti and colleagues
72,73
provided evidence for the clinical relevance in humans of the Cohen and Ellwein,
66,67
Cohen et al.,
68
Moolgavkar and Knudson,
70
and Ames and Gold
71
mechanism regarding the amplification of the background mutation rate. These authors sought to determine the relative contribution to human cancers from inherited mutations, mutations induced by environmental factors, or mutations resulting from DNA replication errors (R). They compared the number of normal stem cell divisions with the risk of 17 cancer types occurring in 69 different countries. Tomasetti and Vogelstein
73
reported the following results: The data revealed a strong correlation (median = 0.80) between cancer incidence and normal stem cell divisions in all countries, regardless of their environment. The major role of R mutations in cancer etiology was supported by an independent approach, based solely on cancer genome sequencing and epidemiological data, which suggested that R mutations are responsible for two-thirds of the mutations in human cancers.
Genotoxicity and increased cellular proliferation interact to increase neoplasia
In 2015, Kiraly et al. 75 provided an elegant demonstration of inflammation-induced cell proliferation greatly potentiating exposure-induced mutations. For many years, it has been recognized that cancer risk was raised in certain chronic inflammatory diseases, for example, phagedenic ulcer of the skin, reflux esophagitis with Barrett’s esophagus, chronic atrophic gastritis, chronic ulcerative colitis, cirrhosis of the liver, cholelithiasis of the gallbladder, and Paget’s disease of the bone. 68 Ulcerative colitis may represent the most dramatic example of a strong association between chronic inflammation and cancer. In patients experiencing inflammation of the entire colon (pancolitis) for 10 or more years, the relative risk for developing colon cancer as compared with normal controls is 20- to 30-fold. 76 Thirty-five years following diagnosis, these relative risks are equivalent to an absolute risk of 30%. 77 (Fortunately, if initial pathologic examination does not reveal dysplasia, the rate of progression to dysplasia and carcinoma is much lower than the 30% figure noted in the aforementioned worst-case scenario. 78,79 ) With an understanding of the relationship between cancer risk and chronic inflammatory disease and pathogen-induced inflammation in hand, Kiraly et al. 75 designed two experiments.
In the first experiment, FYDR mice were exposed to a potent inducer of pancreatic inflammation called cerulein. (FYDR refers to “fluorescent yellow direct repeat” mice harboring a reporter that detects misalignments during homologous recombination (HR).) The cerulein-induced pancreatic inflammation causes double-stranded breaks (γH2AX foci). These double-stranded breaks are associated with an increase in cell proliferation. An acute inflammation by itself did not increase HR. In contrast, HR was increased significantly when inflammation-induced DNA damage and inflammation-induced cell proliferation overlapped.
In the second experiment, Kiraly et al.
75
exposed mice to the alkylating agent methylnitrosourea. While alkylation alone induced some measure of HR, when paired with inflammation-induced cell proliferation, HR was produced in a synergistic fashion. These authors summarized their findings as: Taken together, these results show that, during an acute bout of inflammation, there is a kinetic barrier separating DNA damage from cell proliferation that protects against mutations, and that inflammation-induced cell proliferation greatly potentiates exposure-induced mutations.
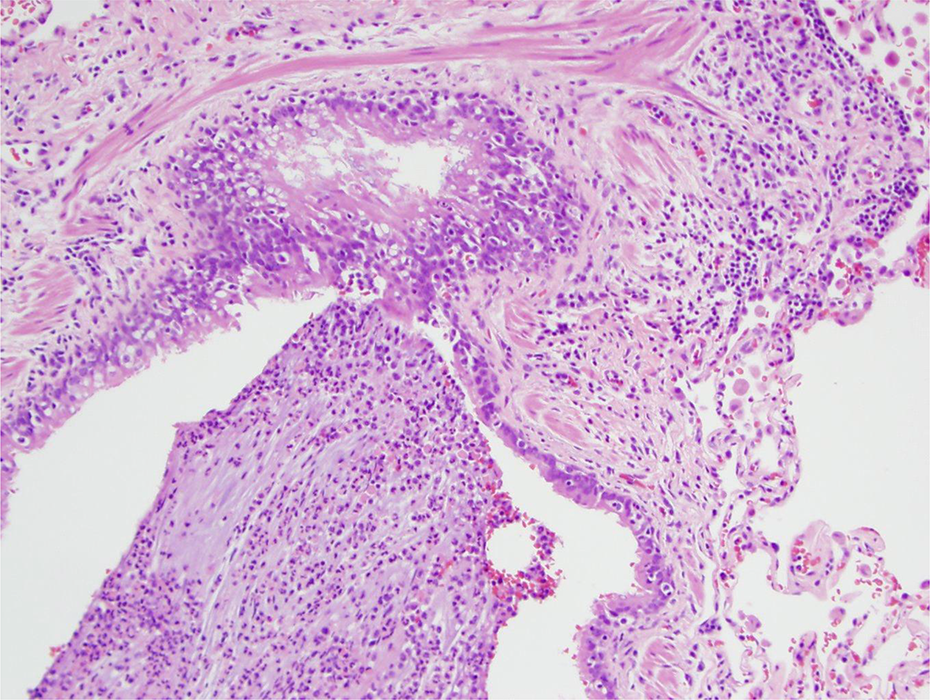
Acute inflammation (neutrophils) in bronchial branch (lumen) (lower left) and chronic inflammation in the wall (upper right). H&E stain 10× objective (100×). H&E: hematoxylin and eosin.
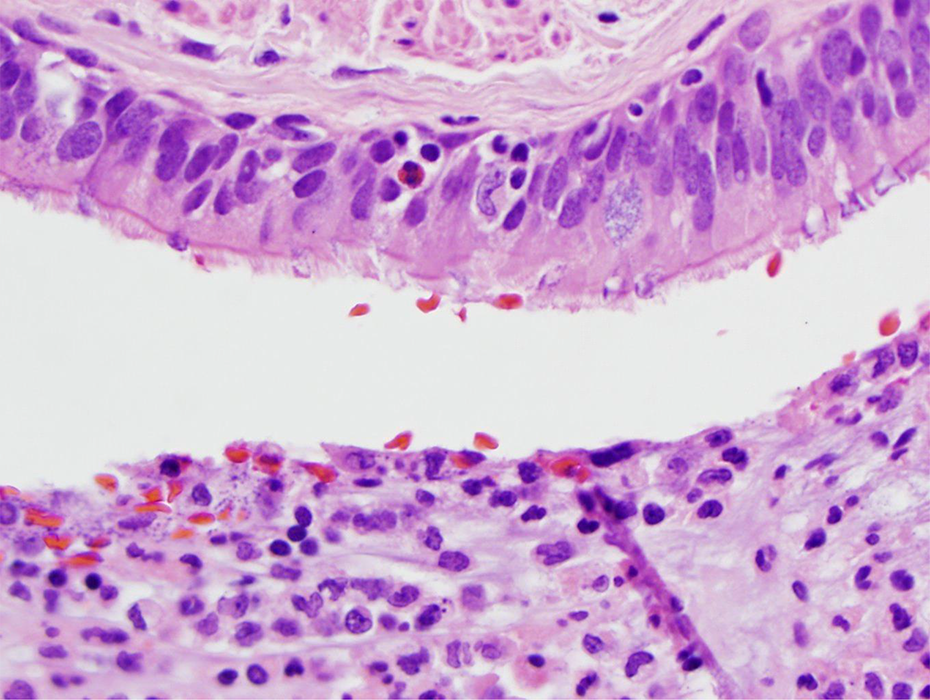
Respiratory epithelial cells with cilia (upper part of photo) with few inflammatory cells in the layer (including an eosinophil). Bottom of photo shows neutrophils in the lumen of the bronchial branch. H&E stain 40× objective (400×). H&E: hematoxylin and eosin.
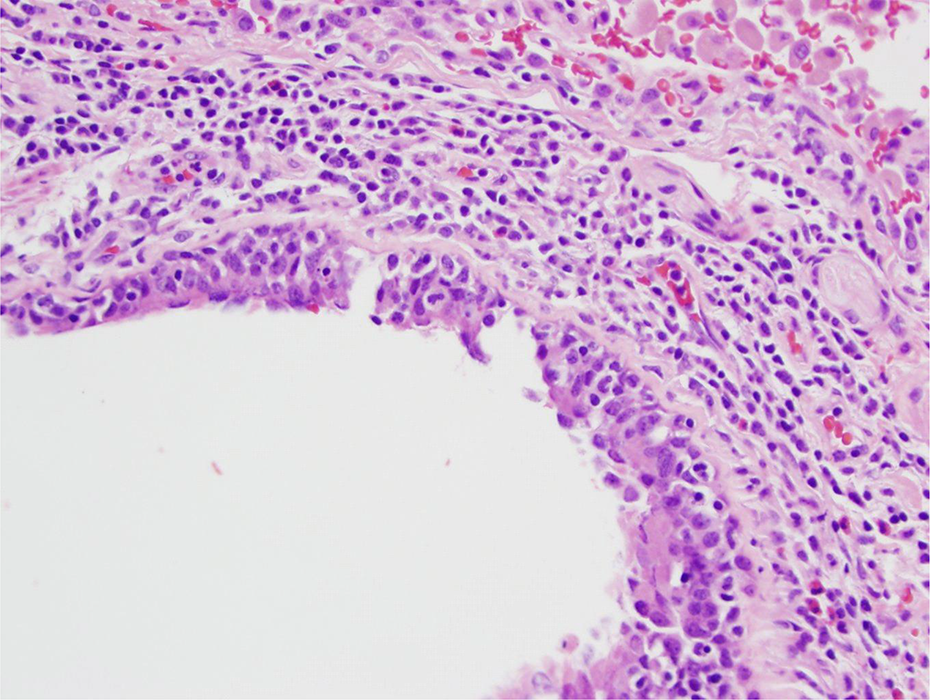
Bronchial branch with ciliated respiratory epithelial cells. Lumen to the lower left. Chronic inflammation (and few eosinophils) in the wall. Some inflammatory cells in the respiratory epithelial cell layer. H&E stain 20× objective (200×). H&E: hematoxylin and eosin.
A number of different cell types are stimulated to release activated oxygen and nitrogen species (Figure 4) in the lungs of COPD patients including cells involved in innate immunity (neutrophils, macrophages, eosinophils, mast cells, natural killer cells, γδ T cells, innate lymphoid cells, and dendritic cells), adaptive immunity (T and B lymphocytes), and structural cells (airway and alveolar epithelial cells, endothelial cells, and fibroblasts) (Table 2). 81 –112
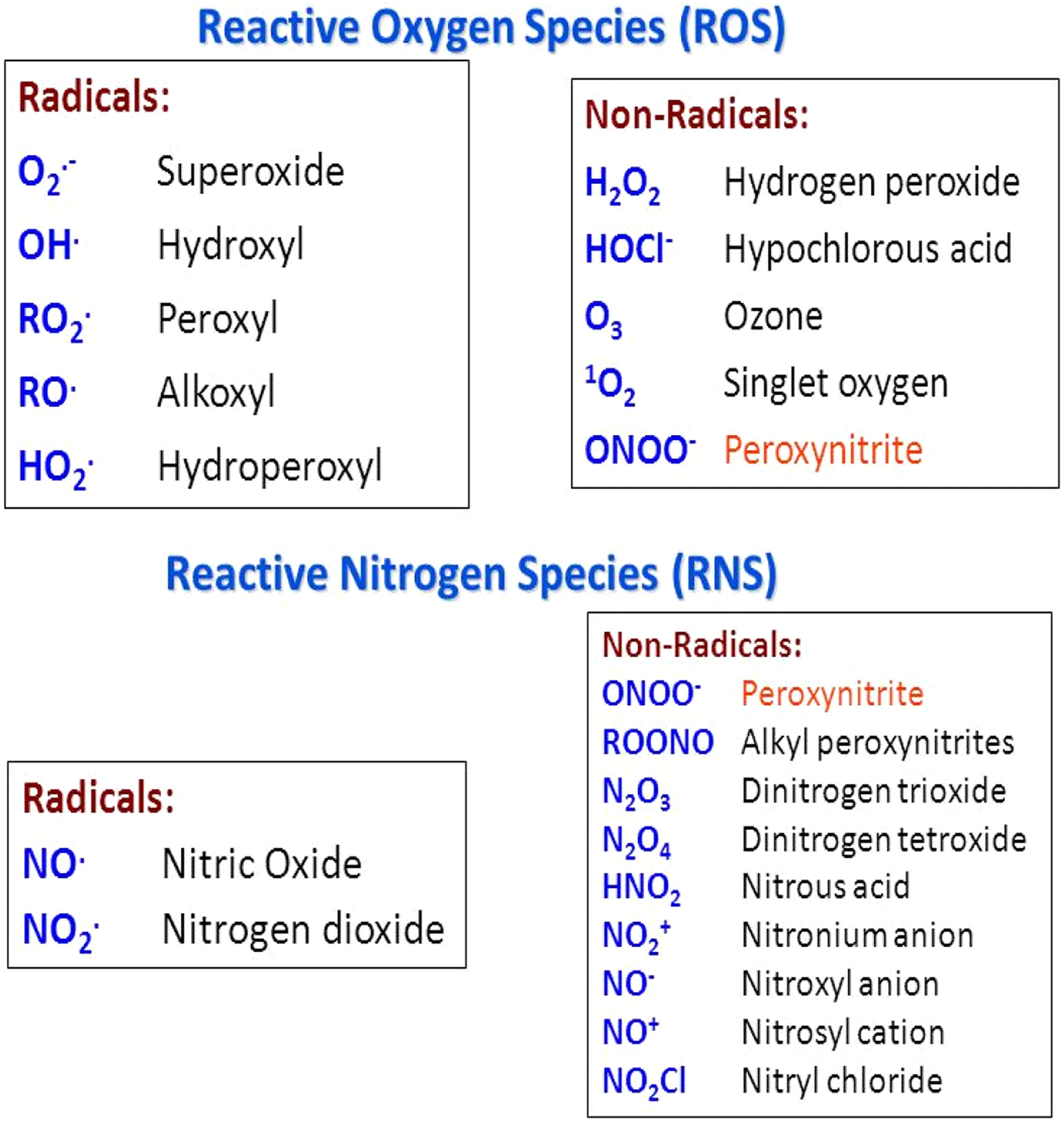
ROS and RNS. Source: https://slideplayer.com/slide/5372691/17/images/6/Reactive+Oxygen+Species+%28ROS%29+Reactive+Nitrogen+Species+%28RNS%29.jpg. RNS: reactive nitrogen species; ROS: reactive oxygen species.
Inflammatory cells and their secreted ROS and RNS.

ROS: reactive oxygen species; RNS: reactive nitrogen species.
Recent screening studies have reaffirmed the results from many previous studies 113 –120 that smokers with COPD are particularly susceptible to lung cancer. 121 The chronic inflammation associated with COPD is postulated to both cause cellular damage and promote cellular proliferation. 122 –134 In addition, the chronic inflammation in emphysema appears capable of conferring some level of lung cancer risk even in the absence of the genotoxic exposure from smoking. 135,136
Inflammatory states are associated with hypoxia
Pulmonary inflammation can result from a wide variety of agents and actions causing injury to the cells lining the airways, including inhalation of a wide variety of chemicals including irritants, cytotoxic compounds, and certain metals and metallic complexes. 46 The vast majority of neutrophils and macrophages found in pulmonary inflammation are not normally resident in the lung, but rather are recruited to inflammatory lesions. 137 One of the metabolic changes associated with active inflammation is development of hypoxia with concomitant accumulation of lactic acid and sometimes metabolic acidosis depending on the amount of lactic acid and the degree of buffering in the tissue environment. 138 –140 A number of factors can contribute to inflammation-induced tissue hypoxia including increased metabolic demands of cells and reductions in metabolic substrates caused by thrombosis (blood clots), trauma, compression (interstitial hypertension), or atelectasis (airway plugging). 141
Hypoxia due to inflammation is a stressor that can preferentially select for expansion of clonal populations of cells with particular mutations, for example, preferential survival of EGFR-positive clones. 142 In hypoxic mice with lung tumors induced by urethane, significant overexpression of EGFR, fibroblast growth factor receptor 2 (FGFR2) and platelet-derived growth factor receptor (PDGFR) were seen. 142 Similarly, using the HCC827 NSCLC cell line, Lu et al. 143 provided evidence that hypoxia/HIF2α activation mediates upregulation of EGFR protein levels. This observation provides a potential non-mutational explanation for the EGFR overexpression often seen in human adenocarcinomas. The authors hypothesized that, “The data presented in this contribution also introduce the intriguing possibility that the tumor microenvironment may act as a universal oncogenic trigger that drives the autonomous growth of tumor cells.”
Human epidermal growth factor (EGF) is a 6 kDa protein and cytokine 144,145 comprised of 53 amino acid residues and containing three intramolecular disulfide bonds. 146 Epidermal Growth Factor Receptor (EGFR) is a protein found on the surface of some cells and to which EGF binds. Cell division is stimulated following the binding of EGF to EGFR. EGFR is found at abnormally high levels on the surface of many types of cancer cells, so these cells may divide excessively in the presence of EGF. Alternate names for EGFR are Erythroblastic leukemia viral oncogene type B1 (ErbB1) and Human Epidermal growth factor Receptor 1 (HER1). 147
Non-small cell lung cancers (NSCLC) are a group of lung cancers so named for either the normal cell of origin or the microscopic appearance of the tumor cells. The three main types of NSCLC are squamous cell carcinoma, large cell carcinoma, and adenocarcinoma. NSCLC is the most common kind of lung cancer. 148 Approximately 10% of patients with NSCLC in the United States and 35% in East Asia have a mutation in the EGFR gene in the DNA of their lung tumor. 149 –151 In both the United States and East Asia, EGFR mutations are more common in tumors from female never smokers (<100 cigarettes in patient’s lifetime) with adenocarcinoma histology. 149 –151 The percentage of EGFR mutation frequency rises to 60–65% in female East Asian never-smoker adenocarcinoma patients. 152 However, EGFR mutations are sometimes seen in squamous cell carcinoma and large cell carcinoma in both former and current smokers. 153
The EGFR mutations occur within exons 18–21. (Both the DNA sequence within a gene and the corresponding sequence in RNA transcripts are termed exons. 154 ) EGFR exons 18–21 encode a portion of the EGFR kinase domain. EGFR mutations are usually heterozygous and display gene amplification, that is, increase in number of copies. 155 The overwhelming majority, that is, approximately 90% of EGFR mutations, are deletions in exon 19 or point mutations in exon 21 L858 R. 156 These mutations increase the kinase activity of EGFR. Kinase-induced phosphorylation activates signaling pathways that block apoptosis. 157 In the vast majority of cases, EGFR mutations seen in NSCLC do not overlap with KRAS mutations observed in the same tumors, 153 although KRAS mutations are found in 25–35% of newly diagnosed non-small cell, non-squamous cell patients. 152
Direct chemical induction of oxidative stress versus indirect oxidative stress from inflammation
An imbalance between the production of ROS such as superoxide anion (O2 ·−), hydroxyl radical (·OH), hydrogen peroxide (H2O2), and singlet oxygen (1O2) and their detoxification results in oxidative stress thereby leading to cellular damage. When a chemical is inhaled at a dose sufficient to induce cellular damage, oxidative stress can be induced by two different although not completely independent sources—(a) direct toxicity to cells lining the airways; and (b) toxicity to cells lining the airways from ROS, RNS, and enzymes released via inflammatory processes.
The process of direct oxygen-related toxicity to cells can be best illustrated by cell culture studies where a single parenchymal cell type is cultured in the absence of the 12 cell types that can participate in inflammatory processes listed in Table 2. The mitochondrial respiratory chain generates the majority of ROS. The electron flow rate through respiratory chain complexes is the primary modulator of mitochondrial ROS production. Approximately 1–4% of mitochondrial oxygen consumption is diverted to the formation of ROS under physiological conditions. 158 Based on the aforementioned ROS conversion rate, and an average rate of utilization of oxygen in each human cell of approximately 2.5 × 10–18 mol/s (i.e. 2.2 × 1010 molecules/day), almost 1 billion molecules of ROS are being produced by each cell every day in vivo. 158
Mechanistic studies are sometimes conducted in cultured cells in an attempt to elucidate the mode of action (MOA) of chemicals. The partial pressure (pO2) of ambient atmospheric oxygen is 150 mm/Hg, which is equivalent to 21% O2. Inhaled O2 levels progressively decrease in humans as the gas reaches various internal organs and tissues. 159 The level of O2 and its tissue distribution depends on the balance between capillary blood flow and oxygen utilization. In healthy humans, arterial pO2 averages approximately 100 mm/Hg or 14% O2. As blood flow reaches the highly vascularized parenchymal organs such as lungs, liver, and kidneys, O2 levels range from 4% to 14%. 159 In less vascularized organs and tissues including brain, eye, and bone marrow, the O2 concentration ranges between 0.5% and 7%. 159 In contrast with the relatively low O2 concentrations seen in human organs, in vitro cultures of immortalized cells use a higher O2 concentration of 21%. Under these higher O2 concentrations, ROS production can be increased several-fold. Cultured primary cells are cultured at lower O2 levels than 21% due to their inability to grow at higher O2. Although excess ROS can lead to oxidative stress, moderate to low levels of ROS function in cellular signaling pathways. 160 Oxidative stress occurs in all cell culture media. It is especially important in serum-free and protein-free media because many of the anti-oxidation properties of serum are missing.
Cells in culture may behave differently from cells in vivo in many ways. Immortalized cells cultured under high oxygen tension can demonstrate a higher inflammatory response and greater redox imbalance. 159 Halliwell 161,162 has argued that cells that survive and grow in culture might use ROS-dependent signal transduction pathways that rarely or never operate in vivo. In addition, cell culture media can catalyze the oxidation of certain test chemicals, resulting in apparent cellular effects due in fact to oxidation products such as ROS. Such artifacts may have affected many studies on the effects of ascorbate, thiols, flavonoids, and other polyphenolic compounds on cells in culture. 161
Since 1978, NTP has conducted 596 studies and issued 480 reports. The term oxidative stress was introduced in 1985. 163,164 Three hundred of the 596 studies had been completed prior to the use of the term oxidative stress. Since the introduction of the term in 1985, NTP has proffered oxidative stress as a potential contributor to the formation of rodent tumors on 58 occasions (Supplemental Table). The 58 instances result from an amalgamation of attributions. First, in some reports written after 1985, the authors retrospectively note that some chemicals tested before 1985 would have invoked oxidative stress as a possible carcinogenic mechanism. Second, the majority of NTP reports do not speculate about mechanisms underlying the observation of rodent tumors. Third, 146/480 (30.4%) NTP reports show no evidence of neoplasms. Fourth, many of the chemicals tested by NTP have also been tested by non-NTP scientists. Several of these non-NTP studies invoked oxidative stress as a possible mechanism for carcinogenicity even when the NTP report authors did not. 165,166 For example, since 2014 there have been 832 articles written on the carcinogenicity of benzene. Of these, 121 (14.5%) have indicated that oxidative stress was the primary cause of cancer by benzene. 167
When a rodent inhales a chemical, it is difficult to disentangle contributions to oxidative stress from the following scenarios: (1) Direct damage to mitochondria of cells lining the distal airways that does not induce inflammation. (Significant cellular damage occurring in vivo and not eliciting any degree of inflammation appears unlikely.); (2) Direct damage to mitochondria of cells lining the distal airways, exposing either antigenic or immunogenic intracellular contents or extracellular membrane components that then elicits additional oxidative stress from activation of one or more of the 12 different cell types described in Table 2; (3) Metabolic conversion of the inhaled chemical to a cytotoxic metabolite that then either damages the mitochondria, or exposes antigenic or immunogenic cellular components that stimulate the inflammatory process; and 4) Without damaging cellular mitochondria lining the distal airways, a chemical, particle, or dust elicits an inflammatory response that secretes large amounts of ROS and RNS. In the human lung, the inflammatory response can make a profound contribution to the levels of oxidative stress as seen in the clinical sequelae of a number of diseases or conditions including silicosis, 168,169 black lung (pneumoconiosis) in coal miners, 170,171 idiopathic pulmonary fibrosis, 172,173 adult respiratory distress syndrome, 174,175 bird fanciers lung, 176,177 and other fibrotic lung conditions.
Section four: Differences between human and mouse lung tumors
Pulmonary anatomy and physiology differ greatly between humans and mice
There are many anatomical and physiological differences between humans and mice. These differences might be relevant to development of neoplasia in 2-year inhalation exposure studies in mice because inflammatory processes that would compromise lung function in humans are tolerable in mice 178 thereby facilitating the cascade of inflammation-cell damage-reparative cell proliferation-non-genotoxic tumor development. 52 The total lung capacity (TLC) of a mouse is only approximately 1 ml. By comparison, the TLC of a rat is about 10 times larger at 10 ml, and the TLC of a human male is approximately 6000 ml. The right lung in humans has three lobes: inferior, middle, and superior. The mouse has four lobes in the right lung—superior, middle, inferior, and postcaval. The human left lung has an upper and lower lobe, while the left lung of the mouse has but a single lobe. The branching patterns of the mouse and human also differ with mice having fewer respiratory generations (13–17) and displaying monopodial (from single branch) branching. Human lungs branch dichotomously and have 17–21 generations. Airways constitute 11% of the mouse lung and 5.7% of the rat lung. The mouse trachea has comparatively poorly organized cartilage with only the upper trachea possessing complete rings seen in other mammals that become plates with distal progression. 179 The compositional percentages of total lung occupied by parenchyma differ among mice (18%), rat (24%), and human (12%).
A very significant difference between human and mouse lung physiology is the extremely high respiration rate in mice (84–350 breaths per minute), 180,181 as compared with humans (12–20 breaths per minute). 182 (The high respiration rate in mice facilitates body temperature maintenance.) As compared with humans, mice have a large airway caliber. 183,184 If mice did not have such a large airway caliber, the flow-resistive load resulting from 250–350 breaths per minute would be prohibitive. 180 Submucosal glands are either absent or sufficiently scarce as to eliminate the secretion of mucus in association with pulmonary inflammation in mice. 182 The combination of a large airway caliber and absence of mucus facilitates a high tolerance to pulmonary inflammation in mice.
Mice but not humans display high spontaneous background lung cancer rates
Different mouse-inbred strains display wide variations in the appearance of spontaneous lung tumors. Several inbred mouse strains have been examined for spontaneous lung tumor rate. By decreasing incidence of spontaneous lung tumors, A/J (82%) > SWR/J (47%) > BALB/c (33%) > CBA (17%) > C3 H (9%) > C57BL/6 (3%). 183 The O2O strain is also classified as intermediately sensitive similar to the BALB/c strain, and the DBA strain is comparable to the highly resistant C57BL/6. The primary mouse model employed by the NTP is the B6C3F1 (NTP 2013). 184 This mouse strain is produced by crossing a female C57BL/6 (3% spontaneous rate) and male C3 H mouse (9% spontaneous rate). The cross product B6C3F1 mice have a higher spontaneous tumor rate than either of the parent strain mice, that is, male B6C3F1 at 27.7% and female B6C3F1 at 9.5% (n = 950/sex; NTP 2013), 184 demonstrating that the genetic factors underlying spontaneous tumor formation do not interact in a linear fashion. Mouse strains with high background spontaneous tumor formation are also more susceptible to chemically induced tumorigenesis.
Human lung cancer cases display a pattern of development antithetical to spontaneous tumor formation. Although less than 10% of smokers develop lung cancer, 185 smoking causes between 80% and 90% of cases. 186 The average age of smoking initiation is approximately 20, while the average age at diagnosis occurs some 50 years later at about age 70. 187 Few cases are diagnosed in patients younger than age 45, with the majority of cases presenting at over age 65. 188
The relatively older ages of lung cancer presentation are consistent with a sophisticated statistical analysis of the lung cancer epidemiological literature conducted by Flanders et al. 189,190 These authors found that cigarette smoking duration is a stronger predictor of lung cancer mortality than is cigarette smoking intensity. This finding held regardless of age in both men and women. Lung cancer risk was proportional to approximately the second or third power of cigarette smoking duration among men and women 40–49 years of age.
Mice and rats are highly discordant in development of chemically induced pulmonary tumors
The NTP reports 60 2-year inhalation studies in both mice and rats on single agents or closely related agents. Fifty-eight of the 60 NTP inhalation studies were amenable to statistical analysis. For the 58 compounds tested via inhalation by NTP, there is a high degree of discordance between mice and rats in the susceptibility to develop lung tumors. The causation of tumors at anatomical sites outside the lung via the inhalation route is also discordant in mice and rats, for example, 11/58 (19%) of agents tested in the NTP inhalation studies using mice and rats were negative in the Ames assay test and showed lung tumors in mice only. Mouse and rat data are discordant regarding the ability to induce tumors at organ sites outside the lungs—0/58 as compared with 16/58, respectively. Mice and rats display distinctly different patterns of both lung tumor development and development of tumors outside the lungs. 46 Mice and rats separated along the evolutionary tree sometime between 12 and 24 million years ago. In contrast, humans separated from rodents approximately 80 million years ago. 191 Differential responses in rats and mice to chemicals can be influenced by idiosyncratic P450 metabolism, immune system surveillance, DNA repair mechanisms, macroanatomical differences (e.g. airways), and microanatomical differences (e.g. predominance of CCs lining mouse lung epithelium). 192 –196 Rats and mice in the NTP database are highly discordant in both the development of tumors and the site of tumor formation, despite their relative phylogenetic closeness. This high discordance begs the question of the potential degree of tumor discordance between humans and rodents.
The ability to form lung tumors in mice in the absence of genotoxicity demonstrates that other mechanisms, for example, cytotoxicity followed by reparative cellular proliferation, might be involved. Ten of the 11 chemicals (90.9%) are insoluble or only slightly soluble in water, soluble in organic solvents, and have moderately hydrophobic log base 10 octanol–water partition coefficients of 0.17, 1.85, 2.10, 2.13, 2.42, 2.53, 2.61, 3.15, 3.30, 3.66, and 3.80. These moderate log p (log Kow) values are near the optimum values for penetrating the lipid bilayer membranes of cells. 197 These chemicals induce hyperplasia in the airways of mice. (Hyperplasia is an increase in the number of cells resulting from cellular proliferation.) 198
Mouse lung tumors are much less histologically diverse than human lung cancer
Table 3 shows the extremely diverse histological presentation of human lung cancers. In contrast, the vast majority of rodent lung tumors are of a single histologic type 200,201 termed bronchioloalveolar adenomas or bronchioloalveolar carcinomas (BACs). 199 –202 This rodent pulmonary lesion is named geographically for its location in the bronchiolar alveolar region. 203 Murine BACs reportedly follow a progressive continuum from hyperplasia to adenoma to carcinoma. 204 Much less common rodent tumors termed cystic keratinizing epitheliomas and squamous cell carcinomas can be observed following inhalation of dioxin (2,3,7,8-tetrachlorodibenzo-para-dioxin (TCDD)), titanium dioxide, talc, and nickel oxide but these uncommon tumors are not seen in unexposed control rodents. 205 Even these uncommon rodent pulmonary tumors are located in regions similar to the location of the more common BACs, that is, distal bronchioles and alveolar acini.
College of American Pathology Histologic Typing of Human Lung Cancer.

Source: Adapted from Butnor et al. 199
+Data elements preceded by this symbol are not required for accreditation purposes. These optional elements may be clinically important but are not yet validated or regularly used in patient management.
Human lung cancers are divided into small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC). 206 SCLC was previously known as oat cell carcinoma. SCLC is a neuroendocrine carcinoma that exhibits aggressive behavior, rapid growth, and early spread to distant sites. 207 –210 NSCLCs are a group of lung cancers so named for either the normal cell of origin or the microscopic appearance of the tumor cells. The three main types of NSCLC are squamous cell carcinoma, large cell carcinoma, and adenocarcinoma. NSCLC is the most common kind of lung cancer. 148 The lower portion of Figure 5 shows a low power view of a section of human lung with adenocarcinoma. The upper portion of Figure 5 shows normal human lung. Figure 6 shows a higher magnification of adenocarcinoma of the human lung with notable nuclear pleomorphism and mitotic figures. Proponents of employing mouse models in quantitative risk assessment represent the rodent BAC tumor as comparable to human adenocarcinomas. 204
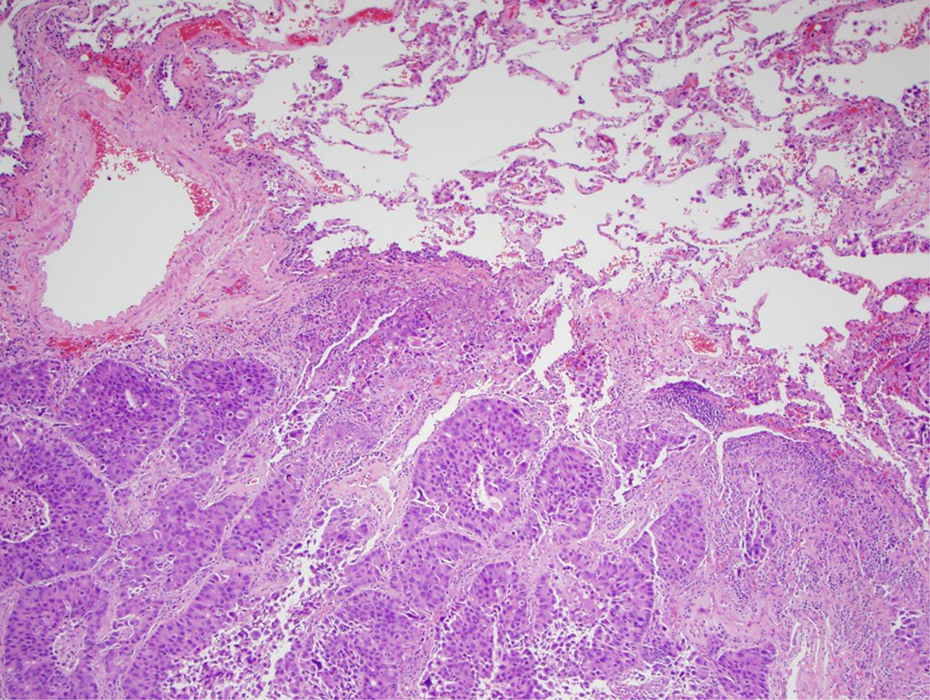
Low power view of section of human lung showing adenocarcinoma in the lower portion of the image and normal lung in the upper portion of the image (H&E stain) (4× objective [40×]). H&E: hematoxylin and eosin.
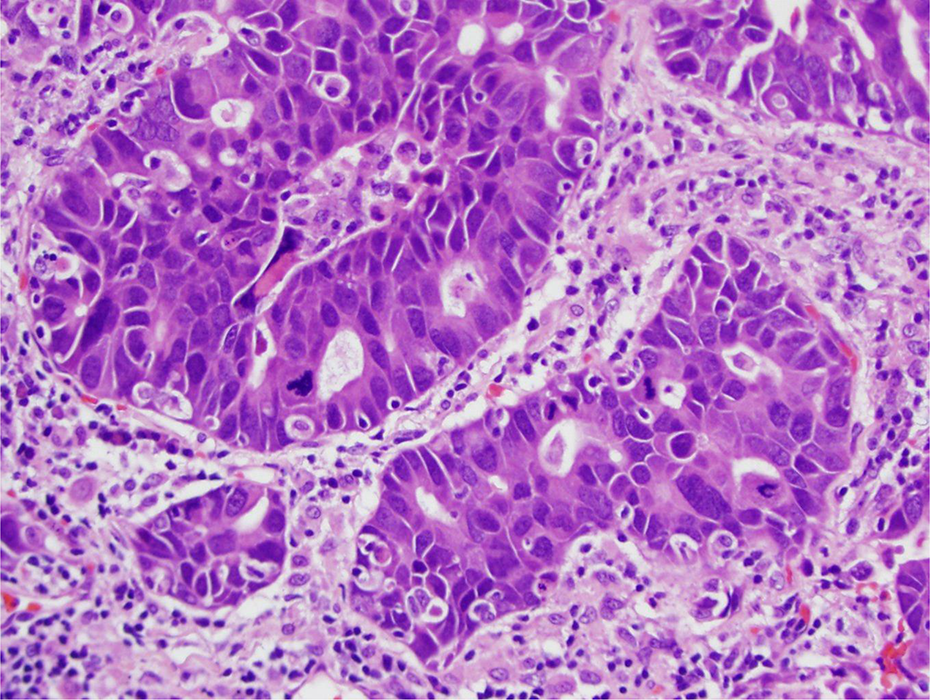
Higher magnification of adenocarcinoma of the lung. Note the nuclear pleomorphism and mitotic figures (H&E stain) (20× objective [200×]). H&E: hematoxylin and eosin.
Human and murine lung tumors display different behavior patterns
In contrast with rodent lung tumors, human lung cancers can display an intense stromal response termed desmoplasia. A desmoplastic reaction leads to the buildup of tumor-associated connective tissue, resulting in a thickening similar to scarring. 211 Many tumors invade the surrounding tissue prior to metastasizing, 212 with stromatogenesis playing an integral role in the invasive process. Stromatogenesis entails the formation of new, specific type, stroma (matrix of connective tissue and blood vessels) at sites of active tumor cell invasion. 213 Endophytic tumors grow inward into tissues in fingerlike projections. 214 In a process termed intramural stromatogenesis for endophytic tumors, newly formed stroma wedges between tissue planes of least resistance thereby cleaving paths for invading tumor cells. Exophytic tumors grow outward. 214 In a somewhat less frequent process, new stroma forms toward a void space at either an internal or external free space. Interaction between the invading tumor cells and adjacent activated fibroblasts is postulated as stimulating the formation of this new stroma. 213
Consistent with the absence of a significant stromal response, mouse lung tumors also possess a much lower metastatic potential than human lung tumors. 204 The metastatic process involves the acquisition of genetic and/or epigenetic alterations within tumor cells. 212 Metastatic potential is among the most clinically relevant tumor behavioral characteristics as primary tumors that have not yet metastasized usually do not kill the patient unless located in a particularly unfortunate area that damages essential anatomical structures. 215
Section five: Histogenetics of human and murine lung tumors
Cellular and genetic origins of human lung adenocarcinoma subtypes
Regardless of histological subtype, human lung cancers derive from normal lung tissues. The major anatomical components of normal lung tissues are the air-conducting system and the peripheral lung parenchyma. Gases are exchanged in peripheral lung parenchyma, normally carbon dioxide for oxygen. During embryonic development of the lung, two lung buds are formed, followed by the morphogenesis of repeated branching thereby producing conducting airways and finally the terminal sac and alveoli. 206
Thyroid transcription factor 1 (TTF-1) is potentially a lineage-specific survival oncogene of some lung adenocarcinomas. 216,217 TTF-1 amplification and overexpression contribute to lung cancer cell proliferation rates and survival. During the later stages of embryonic development, TTF-1 is ubiquitously expressed in peripheral lung epithelial cells such as small bronchioles and alveoli. 218 The peripheral bronchioloalveolar compartment contains the terminal bronchioles, alveolar ducts, and alveoli. These three structures are termed the terminal respiratory unit (TRU). CCs and type II pneumocytes are normal cells found within the peripheral bronchioloalveolar compartment that can be transformed to tumor cells and give rise to tumors expressing TTF-1. 219 Figure 7 shows a section of normal human lung stained for TTF-1 with nuclear stain. Figure 8 presents a section of human lung adenocarcinoma illustrating nuclear staining for TTF-1. In contrast with the TRU, there are two different potential candidate progenitor cells that give rise to tumors in the central conducting airways (bronchi): bronchial basal cells and mucous cells. 219,220 These central airway tumors are TTF-1 negative.
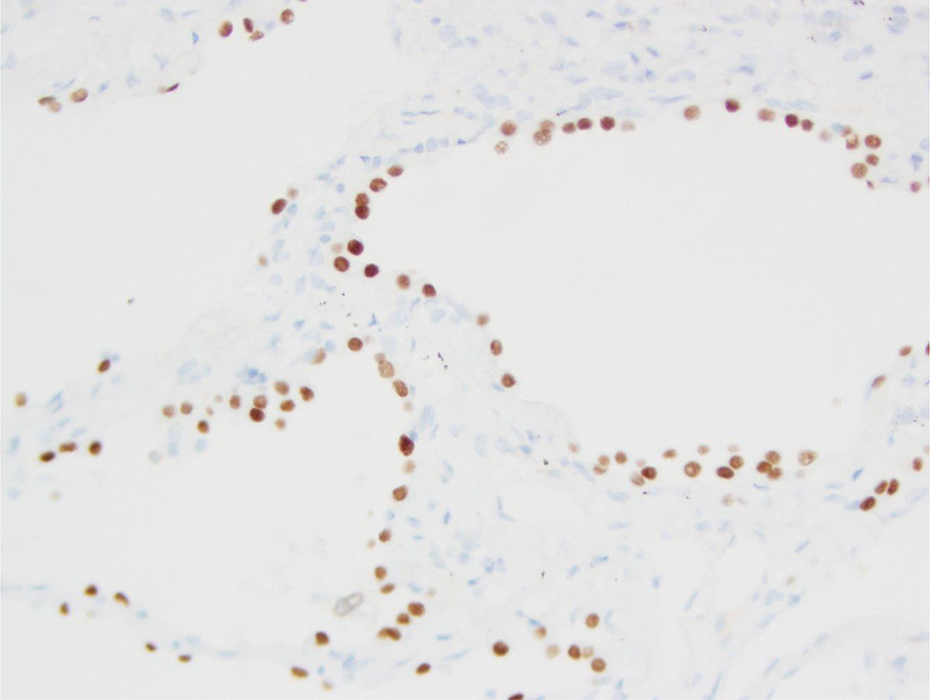
Section of normal lung stained for TTF-1 (nuclear stain) (20× objective [200×]). TTF: thyroid transcription factor.
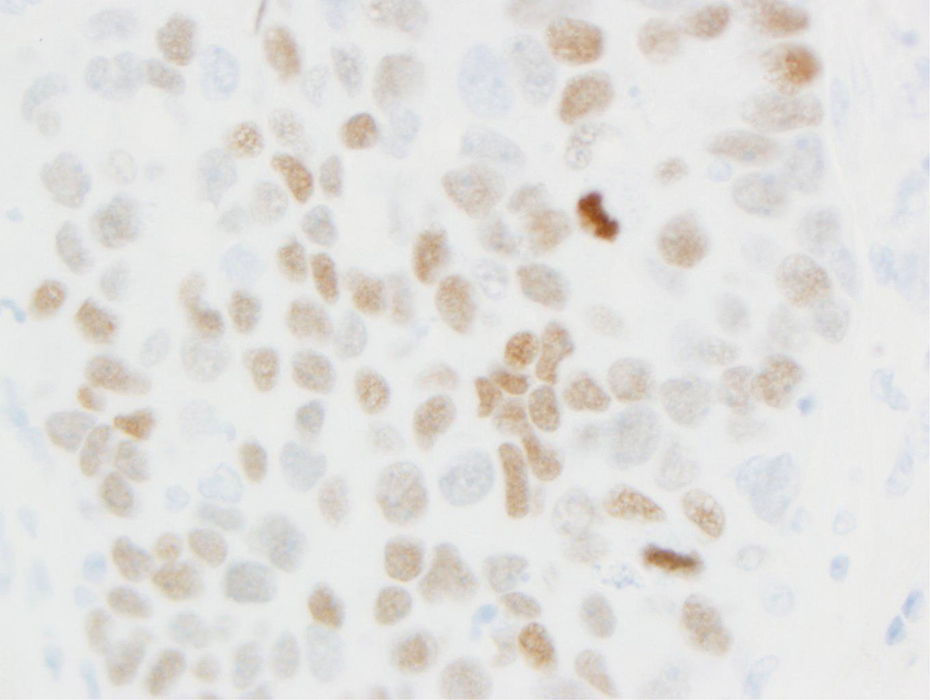
Section of lung adenocarcinoma showing nuclear staining for TTF-1 (40× objective [400×]). TTF: thyroid transcription factor.
Hierarchical cluster analysis is an algorithm that organizes similar objects into groups termed clusters. While each cluster is distinct from every other cluster, the objects within each cluster are broadly similar to each other. 221,222 Hierarchical cluster analysis has been conducted on biopsies of adenocarcinomas based on the TTF-1 expression profile and has demonstrated the presence of two major histogenetic clusters: TRU- and non-TRU-type adenocarcinomas. Therefore, there are two major subsets of human adenocarcinoma with distinct histogenetic origins. 223
The international adenocarcinoma classification study group 206 has hypothesized that a subset of human lung adenocarcinomas progress from atypical adenomatous hyperplasia (AAH) to adenocarcinoma in situ to invasive carcinoma. Following transformation (initiation) of a normal cell to a malignant phenotype, multiple genetic changes might facilitate the tumor progression process in a stepwise fashion. 224 –227 EGFR and KRAS mutations can be seen in normal epithelium 228,229 and in premalignant AAH and are also observed in invasive adenocarcinoma. However, increased numbers of EGFR gene copies only become widespread in later stages of adenocarcinoma invasion and metastases. 230,231 Amplification (multiple gene copies) of EGFR, KRAS, and TTF-1 all characterize the progression process. 217,230,231 Invasive adenocarcinomas contain more p53 tumor suppressor gene mutations than noninvasive adenocarcinomas. 232 –237 Despite the positive association with invasiveness, the p53 mutation has not been identified as a reliable prognostic marker or a therapeutic target.
The histogenetics of lung cancer in mice differs greatly from that seen in human lung cancer. The cellular origin of rodent BACs is frequently reported as type II pneumocytes or CCs but demonstrating the precise cellular origin of mouse lung tumors can be technically challenging. 238,239 In an extremely thorough analysis of mouse models for human lung cancer, Meuwissen and Berns 240 state that the cellular origin of murine pulmonary adenocarcinomas is unknown. Murine pulmonary adenocarcinomas might arise from CCs, alveolar type II pneumocytes, multipotent stem cells, or from derivative lineages descended from these cells. 241 –247
In Meuwissen and Berns, 240 these authors provide a detailed summary of many of the genetic changes associated with murine mouse lung cancer models. The wide susceptibility across inbred mouse strains to chemically induced neoplasia has led to a search for etiological clues in strain-associated differences in molecular genetics. The more chemically sensitive mouse strains are hypothesized to be sensitive to lung cancer 248 due to enhanced KRAS expression 249 caused by a polymorphism in intron 2 of KRAS. 250,251 In addition to the strain-associated susceptibility toward KRAS mutation, various strains of mice exposed to a variety of different chemical agents have been reported to be related to a voluminous array of tumor-associated genetic configurations not seen in humans including the following representative but not comprehensive list: Cdkn2a polymorphism; 252 –254 three pulmonary adenoma susceptibility (PAS) loci; 255,256 Pas-1; 257,258 linkage of K-Ras to the Pas-1 locus; 259 two non-Pas-1 lung tumor susceptibility loci on chromosome 6; 260 12 additional Pas loci; 256,261 –263 and 30 different loci conferring susceptibility to lung cancer, abbreviated as Sluc. 264 –266
Molecular analysis of murine tumors has been conducted as a function of tumor initiation and development timeline. In both spontaneous and chemically induced murine tumors, hyperplastic lesions have displayed KRAS mutations 267,268 suggesting that this genetic change can be an early event. 269 In contrast with the early event of KRAS mutation, tumor-suppressor gene inactivation is usually a late event in chemically induced mouse lung tumor development. 270
Section six: Role of CCs in human respiratory diseases and mouse lung tumor development
Unique biology of human CCs
In humans, the respiratory bronchioles form the margin between the larger conducting airways and the distal respiratory zone where gases are exchanged. 271 Five different cell types are found in the respiratory bronchioles: ciliated cells (most common); microvillar cells (few); small granule cells (few); and bronchiolar cells known as club cells or Clara cells. In the epithelium of the alveoli, there are four different cell types including type I pneumocytes, type II pneumocytes, type III pneumocytes (rare), and numerous pulmonary macrophages. 272 CCs are also known as bronchiolar cells or non-ciliated non-mucous secretory cells of the bronchiolar epithelium. 273 The presence of a large number of mitochondria facilitates a high level of metabolic activity in CCs. Each CC usually contains six, approximately 0.3 µm diameter dense granules located near the basement membrane. The composition of the granule contents includes proteins, glycoproteins, and lipids. CCs contain substantial amounts of cytochrome P450 enzymes and mixed-function oxidases working collaboratively. In both an apocrine (membrane budding) and merocrine (secretion) manner, 273,274 CCs produce and secrete a number of different substances. Innervating adrenergic fibers stimulate the secretory activity of CCs. 275
Nonhuman primates are much closer phylogenetically to humans than are rodents, for example, the rhesus macaque monkey and humans shared a common ancestor only approximately 25 million years ago, 276 as compared with 80 million years ago for the lineal divergence of rodents and humans. 191 Differences in pulmonary anatomy and physiology between even monkeys and humans are sufficiently large that Boers et al. 277 stated that “Even species with an extensive branching of respiratory bronchioles, e.g. nonhuman primates, 278 are far from an ideal model for the human lung, in contrast to the speculation of Plopper and colleagues.” 279 Airway organization, epithelial cell composition, and CC ultrastructure differ between humans and all other animals.
Based on an autopsy study on seven healthy human lungs, CCs comprise approximately 9% of the total population of airway epithelial cells. CCs are almost absent in the mucus membranes of the proximal segments of the bronchial tree including the trachea, primary bronchi (first branch), lobar bronchi (narrower secondary bronchi), and segmental bronchi (narrower tertiary and further branching bronchi). Approximately 11–22% of CCs (0.99–1.98% of total lung epithelial cells) are located in the terminal bronchioles. Despite their scarcity, under physiological conditions CCs constitute approximately 15–44% of all proliferating cells in the terminal bronchioles. 277,280 Possibly due to the difference in CC numbers, the proliferation percentage in the respiratory bronchioles (44%) is higher than in the terminal bronchioles (15%).
Direct experimental evidence on the proliferative response of the human lung to injury cannot be collected under the clinical exigencies of treating such an injury. Circumstantial evidence suggests that chronic pulmonary injury in humans might reduce rather than increase CC numbers. CC numbers are reduced in cigarette smokers. 281 –283 In addition, CC secretory proteins CC10 and P1 detected in serum and bronchoalveolar lavage fluid are decreased in smokers, 281 patients with bacterial pneumonia, 284 and patients with COPD and lung cancer. 285 In contrast, in rodents CCs clearly proliferate as a response to lung injury. Results from oxidant gas pulse-chase exposure studies suggest that the majority if not all rabbit and rat CCs proliferate in response to injury. 286 CCs participate in cell renewal in hamster bronchial epithelium. 287 CC division is the predominant contributor to the proliferative response of the bronchiolar epithelium after exposure of rats to NO2 288 or O3. 289
In an elegant series of publications, Cruzan and colleagues have demonstrated that the MOA of the mouse-specific lung carcinogen styrene is neither quantitatively nor qualitatively relevant to humans. 290 The pulmonary CCs in mice contain high levels of the metabolic enzyme CYP2F2 which hydroxylates the aromatic ring of styrene producing 4-hydroxystyrene, 3,4-dihydroxystyrene, and 4-hydroxystyrene-7,8-oxide as metabolites. 291 The hydroxylation of aromatic rings in the synthesis of Coenzyme Q is thought to be the normal function of CYP2F2. 292 In addition to styrene, the tumorigenicity of naphthalene and coumarin in mouse lung also results from a similar metabolism of these chemicals by high levels of CC CYP2F2. 293 Rats do not develop lung tumors from inhaling styrene, naphthalene, and coumarin because although rat CYP2F4 appears to be equally active to mouse CYP2F2 in metabolizing these chemicals, CYP2F4 occurs at a much lower concentration in rat CCs with cytotoxic metabolite production insufficient to cause reparative cell proliferation and tumor development. The difference between humans and mice is dramatically greater than between rats and mice. Human lungs contain very few CCs as compared with rats and especially mice. Human lung microsomes only marginally or do not metabolize styrene, naphthalene, and coumarin. In addition, morphological differences between human CCs and mouse CCs render the mouse cells as much more sensitive to damage via reactive metabolites. 293
Cytotoxic and proliferative effects of hydrophobic chemicals
Up through 2017, the NTP had tested a total of 60 single agents or mixtures in 2-year inhalation studies in both rats and mice. Fifty-eight of the 60 inhalation studies were amenable to statistical analysis. Eleven out of 58 agents tested in the NTP inhalation studies using rats and mice were negative in the Ames assay and showed lung tumors in mice only. 46 Ten of the 11 chemicals (90.9%) are insoluble or slightly soluble in water, soluble in organic solvents, and have moderately hydrophobic log base 10 octanol–water partition coefficients: Nitrobenzene, CAS No. 98-95-3, slightly soluble in water, soluble in organic solvents. Log p = 1.85; Trichloroethylene, CAS No. 79-01-6, slightly soluble in water, soluble in ethanol, acetone, diethyl ether, and chloroform, and miscible in oil. Log p = 2.61; Vinylidene chloride, CAS No. 75-35-4, clear volatile liquid, insoluble in water but miscible with most organic solvents. Log p = 2.13; 1-Bromopropane, CAS No. 106-94-5, slightly soluble in water, soluble in most organic solvents. Log p = 2.10; Cumene, CAS No. 98-82-8, alkylated benzene volatile at room temperature. Log p = 3.66; Divinylbenzene-HP, CAS No. 1321-74-0, insoluble in water and soluble in methanol and ether. Log p = 3.8; Naphthalene, CAS No. 91-20-3, not soluble in water, soluble in organic solvents. Log p = 3.3; Chloroprene, CAS No. 126-99-8, practically insoluble in water, soluble in alcohol, and miscible with acetone, benzene, and ethyl ether. Log p = 2.53; Ethylbenzene, CAS No. 100-41-4, practically insoluble in water but soluble in most organic solvents. Log p = 3.15; Nitromethane, CAS No. 75-52-5, soluble in water, alcohol, ether, acetone, and dimethylformamide. Log p = 0.17; and Isoprene, CAS No. 78-79-5, Log p = 2.42.
These moderate log p (log Kow) values of 0.17, 1.85, 2.10, 2.13, 2.42, 2.53, 2.61, 3.15, 3.30, 3.66, and 3.80 are near the optimum values for penetrating the lipid bilayer membranes of cells. 198 These chemicals induce hyperplasia in the airways of mice. Of these 11 chemicals, the non-genotoxic mechanism of mouse lung tumor induction described above by Cruzan et al. 293 has been elucidated for cumene and naphthalene. A third member of the list of 11 chemicals, trichloroethylene is both acutely toxic and carcinogenic to the mouse lung via the production of reactive metabolites by CCs following exposure via inhalation. As in the cases of styrene, cumene, and naphthalene, the absence of metabolic capacity in human lung suggests that the risk of human lung cancer from trichloroethylene is minimal. 234
The induction of pulmonary tumors via CC metabolism of a chemical to a cytotoxic metabolite that elicits cellular proliferation is not limited to mice and rats. Hukkanen et al. 294 describe the following species as capable of metabolizing the listed chemical to a pneumotoxic metabolite(s): 3-methylindole metabolized via CYP2F and CYP4B1 by the CCs of cow, goat, mouse, and rat; 4-ipomeanol metabolized via CYP4B1 by the CCs of the rabbit; coumarin metabolized via CYP2B subfamily by the CCs of the mouse; dichloroethylene metabolized via CYP2E1 by the CCs of the mouse; and ethyl carbamate and vinyl carbamate metabolized by CYP2E1 by the CCs of the rat and mouse. In summary, the discovery that a chemical is metabolized to a pneumotoxic metabolite in an animal, especially a mouse, but not in a human is a common finding.
The differential anatomic distribution between the few CCs in human airways and the many CCs in murine airways probably affects the absorption and metabolism of hydrophobic chemicals. Many inhaled, highly lipophilic compounds, for example, polycyclic aromatic hydrocarbons (PAHs), have longer retention times with resultant higher local doses in bronchial and bronchiolar epithelium than less lipophilic compounds. 277, 295,296 –298 Gerde et al. 299 have constructed a dosimetric model for inhaled PAHs in which a larger fraction of inhaled PAHs is deposited in the alveolar region, that is, respiratory bronchioles. PAHs depositing in this region absorb into circulating blood at such a rapid rate that there is little time for local metabolism. Only 5% of human lung cancers develop in this region. In humans, this region of low metabolism is the same area where the few CCs that are proliferating are found, that is, proliferation percentage in human respiratory bronchioles (44%) is higher than in the terminal bronchioles (15%). In “Genotoxicity and increased cellular proliferation interact to increase neoplasia” section, results from Kiraly et al. 75 were discussed to illustrate that the coincidence of exposure to mutagens in the presence of cellular proliferation is of special concern to elevation of cancer risk. The relative scarcity of human lung tumor development in this alveolar region is consistent with a lack of chemical metabolism coinciding with the vast majority of the CCs that are proliferating, although the absolute number of CCs in human epithelium is small. In stark contrast with the distribution of proliferating CCs in the human lung, both the proximal intrapulmonary epithelium and the terminal bronchiolar epithelium in the mouse are predominately lined with CCs, 59–61% and 60–80%, respectively. 300
Section seven: Conclusions relevant to quantitative risk assessment
In 2-year inhalation studies using rats and mice, when pulmonary tumors are seen in only the male or female mice or both, but not in either sex of rat, there is a high probability that the murine pulmonary tumor has been produced via CC metabolism of the inhaled chemical to a cytotoxic metabolite. If the chemical being tested is also negative in the Ames Salmonella mutagenicity assay, and only mouse pulmonary tumors are induced, the probability that this pulmonary tumor is not relevant to human lung cancer risk goes even higher.
In addition to increasing awareness of the idiosyncratic nature of mouse pulmonary tumors, an emphasis on recent, state-of-the-art genotoxicity test batteries rather than reliance on older non-good laboratory practice (non-GLP) genotoxicity results on chemicals lacking a certificate of analysis is needed to facilitate proper MOA analysis of chemically induced tumors. 52 In summary, bronchioloalveolar lung tumors induced in “mice only” by non-genotoxic chemicals are not useful for quantitative assessment of pulmonary adenocarcinoma risk in humans.
Supplemental material
SmithPerfettiKingSupplemental - Bronchioloalveolar lung tumors induced in “mice only” by non-genotoxic chemicals are not useful for quantitative assessment of pulmonary adenocarcinoma risk in humans
SmithPerfettiKingSupplemental for Bronchioloalveolar lung tumors induced in “mice only” by non-genotoxic chemicals are not useful for quantitative assessment of pulmonary adenocarcinoma risk in humans by Carr J Smith, Thomas A Perfetti, and Judy A King in Toxicology Research and Application
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
