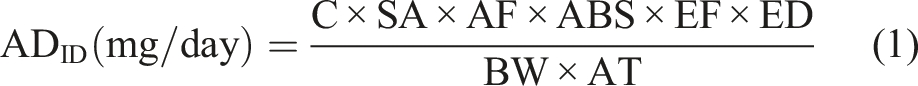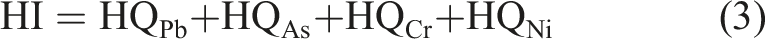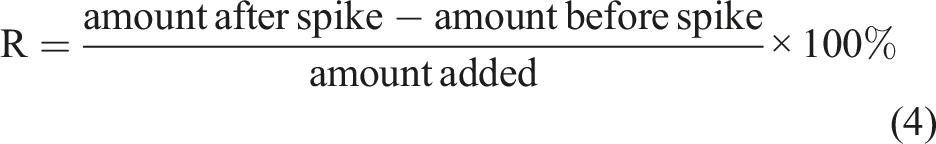Abstract
Introduction
Heavy metals in the environment such as lead (Pb), chromium (Cr), nickel (Ni) and arsenic (As) arise from both natural and anthropogenic sources.1–3 The anthropogenic sources of these heavy metals have resulted in elevated levels of metal pollutants in the environment.4,5 The pursuit of physical beauty by men and women has increased the use of cosmetics around the world.6,7 In particular, the use of cosmetics products, especially skin lightening cosmetics has been found to be a common practice in Africa, Asia, Middle East and Latin America countries. 8 In the early years of cosmetics use, it was assumed that these products worked only on the surface of the skin and would not be absorbed through the skin. 6 However, studies later established that some of the additives in these products can have systemic absorption through the skin which raised concerns about their safety. 6 The detrimental health effects of heavy metals Pb, Cr, As and Ni are well documented.9,10 The adverse health effects from exposure to Pb are more prominent in children than adults with levels of Pb as low as 10 μg/dl in blood leading to damage of the brain, impaired vision and slowed growth. 11 In adults, the detrimental health effects of Pb include reproductive problems, decreased fertility and kidney dysfunction. 1 Studies such as the one carried out in Italy were able to establish a link between children’s makeup containing heavy metals such as Ni and Cr and skin irritation. 12 Several studies have linked dermatitis and allergic reactions to the use of cosmetic products such as children’s bath products, facial care products and body care products.13,14 In addition, the disposal of these products is not regulated and in most cases products are washed down the drain which has the potential to contaminate ground water. 10 This has culminated into more studies to ascertain for the safety of these products, stringent regulations governing product quality and user protection. For example, in 1976, the European Union (EU) laws for cosmetics banned the use of Pb and its compounds in cosmetic products since 1976. 15 Furthermore, the United States Food and Drug Administration (US FDA) through the Federal Food, Drug and Cosmetic Act (FD&C Act) frequently carries out product sampling for examination and analysis which assesses the plant and import compliances to safety standards. 16 It is important to note that cosmetic products are regulated by the US FDA before their release into the mainstream market and this task has been relegated to manufacturers who are required to assess the safety of their products before reaching consumers. 17
However, several developing countries such as Botswana, Zimbabwe, Lesotho and Swaziland lack legislations that regulate the import, distribution and use of cosmetics products in line with standards that are commensurate with those set up by the US FDA and EU.18–20 Studies have recommended that in the absence of laws regulating levels of toxic metals in cosmetic products, a concept called ‘Threshold of Toxicological Concern’ (TTC) be used for the evaluation of the safety of cosmetic ingredients.21,22 TTC considers the chemical structure and toxicity information of compounds that are related in structure. 21 It compares the approximated oral intake of a particular compound with the TTC value obtained from toxicity data of similar compounds.
Currently, there is no regulation in Botswana governing quality checks and controls for determination of metals in cosmetic products imported into the country. This is the first study to be carried out in Botswana to ascertain the presence of toxic heavy metals in cosmetic products imported and sold in Botswana. The objective of this study, therefore, is to assess the presence of various trace metals such as as Pb, Cr, Ni and As in cosmetic products sold in Botswana market. Due to their detrimental health effects even at low concentrations (for example; 10 μg/dl of Pb in blood), these trace metals have been listed in the EU Cosmetics Directive in the class of compounds that have been banned for use in cosmetics. 23 This study intends to raise awareness on the prevalence of toxic metals in cosmetic products and the risks associated with the use of such products. Lastly, the study will also provide baseline data for further studies on epidemiological patterns and determinants of skin related problems in Botswana.
Materials and methods
Sampling location and sample collection
The cosmetic products were facial and body creams and lotions purchased from different beauty shops in Gaborone, Botswana. A total of 14 skin lightening cosmetic samples were used in the study (Figure 1). Unfortunately, the labelling on the products did not indicate the active ingredients responsible for bleaching of the skin. The cosmetic products were chosen based on their frequency of purchase by users from the stores as per the store assistants and therefore their resultant popularity among users. The shop assistants were asked verbally about cosmetic products that consumers frequently purchased and based on that information, products that are popular with the consumers were bought and used in the study. During sampling, information about the manufacturers, product name, brand, colour and the product’s ingredients on the label were all recorded. The products used in this study originated from Hong Kong (3), China (4), Italy (2), South Africa (2), Switzerland (1), Spain (1) and Democratic Republic of Congo (DRC) (1). The samples were analysed for heavy metals Pb, Cr, Ni and As. Skin lightening products used in the study.
Reagents and chemicals
All reagents, solvents and chemicals used in this study were of analytical grade. Hydrochloric acid (35%, v/v), nitric acid (65%, v/v) and hydrofluoric acid (40%, v/v) were all supplied by LabChem (Johannesburg, South Africa). Stock standard solutions of Pb, Cr, Ni and As (1000 mg/l) were also purchased from Sigma Aldrich (St Louis, USA). Double deionized water was produced from the chemistry lab and was used for dilution of samples prior to analysis using inductively coupled plasma optical emission spectrometry (ICP-OES; Perkin Elmer, Optima 7300DV).
Sample preparation and chemical analysis
Exactly 1.0 g of each sample was weighed accurately into a Teflon microwave digestion vessel. About 10 mL of a mixture of nitric acid (65%, v/v) and hydrofluoric acid in the ratio of 4:1 was added to the samples. The Teflon vessel was capped and swirled to thoroughly mix the sample with the acid. This was followed by wet digestion of the sample at 200°C for 15 min in a microwave digestion system (Microwave Reaction system MARS 6, CEM corporation, USA). The solution was then allowed to cool to room temperature, filtered using Whatman no. 42 into a 100 mL volumetric flask and diluted to the mark with deionized water. The precision and accuracy of the method were determined by analysis of procedural blanks, calibration standards and triplicate of samples. The sample solution was analysed for Pb, Cr, Ni and As using ICP-OES.
Health risk assessment
Heavy metals contained in cosmetic products may enter the body through absorption through the skin. The dermal absorption of these heavy metals such as Pb is promoted by the presence of fat-soluble components in the products resulting in absorption of Pb through the skin as organo-Pb.
24
However, some of the heavy metals such as Cr are water-soluble which makes it easier for them to be absorbed through the skin.25,26 The health risk assessment was evaluated using the average daily intake for exposure dose through dermal contact (ADID) and hazard quotient (HQ) based on the concentrations of heavy metals detected in cosmetic products.27,28 The ADID was estimated for all samples using equation (1)
Where C (mg/kg) is the measured concentration of the analyte of interest, SA (cm2) is the exposed skin area given as 565 cm2 for skin lightening creams and lotions, AF (mg/cm2/day) is the adherence factor which is given as 0.07 for leave on cosmetics, ABS is the dermal absorption factor (ABS ∼0.001 for Pb, Cr, Ni and As), EF (day/year) is the frequency of the exposure which is taken as 350 days/year, ED (year) is the exposure duration which is averaged to 30 years, BW (kg) is the body weight of a human being (taken as 70 kg) and AT (day) is the averaging time (10,950 days).28,29
On the other hand, the hazard quotient assesses whether there are potential adverse health effects emanating from exposure to cosmetic products.
28
The hazard quotient of the different metals in samples was evaluated as the ratio between the average daily intake for exposure dose through dermal contact (ADID) and the reference dermal dose (Rfddermal) using equation (2).
30
A hazard quotient of less than one is considered to be associated with no adverse effects
The RfD (mg/kg day) values for Pb, Cr, Ni and As were 1.4 × 10−4, 1.5, 2.0 × 10−2 and 3.0 × 10−4 respectively.28,31 The oral reference dose is the estimated daily human exposure to metals that has no hazardous effect during lifetime. The hazard index (HI) from all metals studied was estimated from the summation of the HQ of individual metals using equation (3)
Quality control
Glassware was washed with soap and tap water and then rinsed many times with tap water before being soaked overnight in 5% HNO3 solution. They were subsequently rinsed with double distilled water and dried in oven at 70°C. Before each sample was analysed, three blank runs were carried out and the average blank measurement was subtracted from the sample measurement. Samples measurements were taken in triplicates. Due to lack of a cosmetic certified reference material, the accuracy and precision of the analytical methods employed were validated through the spike recovery procedure. This was done by introducing into fresh portions of analysed samples a known concentration of the analysed element and percent recovery for each element addressed the accuracy of the analytical procedure. The percent recoveries (R) were calculated using equation (4);
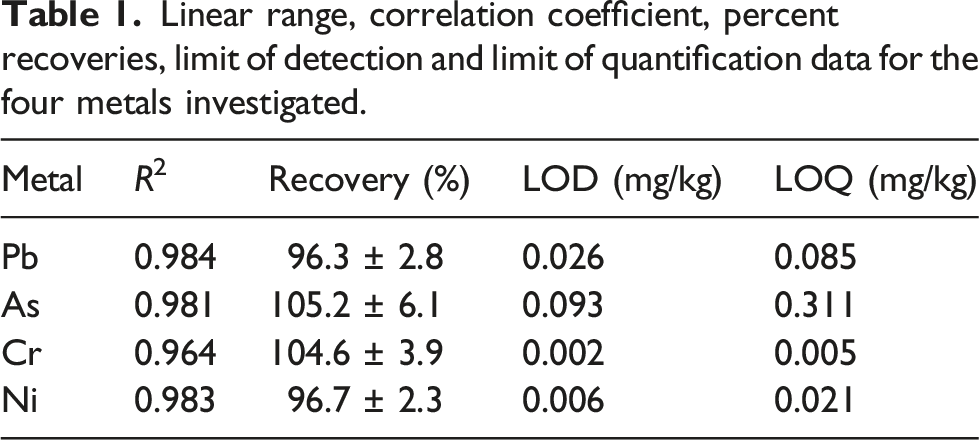
The limit of detection (LOD) and limit of quantification (LOQ) were determined experimentally from 3s/m and 10s/m respectively where s is the standard deviation of the signal measure of the blank solutions and m is the slope of the calibration curve for the specific test metal. 32 Intra and inter-day precisions were also carried out using a 5 mg/kg multi-element standard solution following the optimized analytical procedure used for analysis of the cosmetic samples. For intra-day precision, 5 mg/kg multi-elemental standard solution was analysed under optimized conditions for five times twice in a day and response concentrations were recorded. For inter-day precision, 5 mg/kg multi-elemental standard solution was analysed five times for three consecutive days under optimized conditions and the response concentrations were recorded. The calculated %RSD values for both inter-day and intra-day were less than 2% indicating that the data set are closer to each other.
Linear range, correlation coefficient, percent recoveries, limit of detection and limit of quantification data for the four metals investigated.
Results and discussion
Total metal concentration in cosmetic products
Mineral elements detected in cosmetic samples.
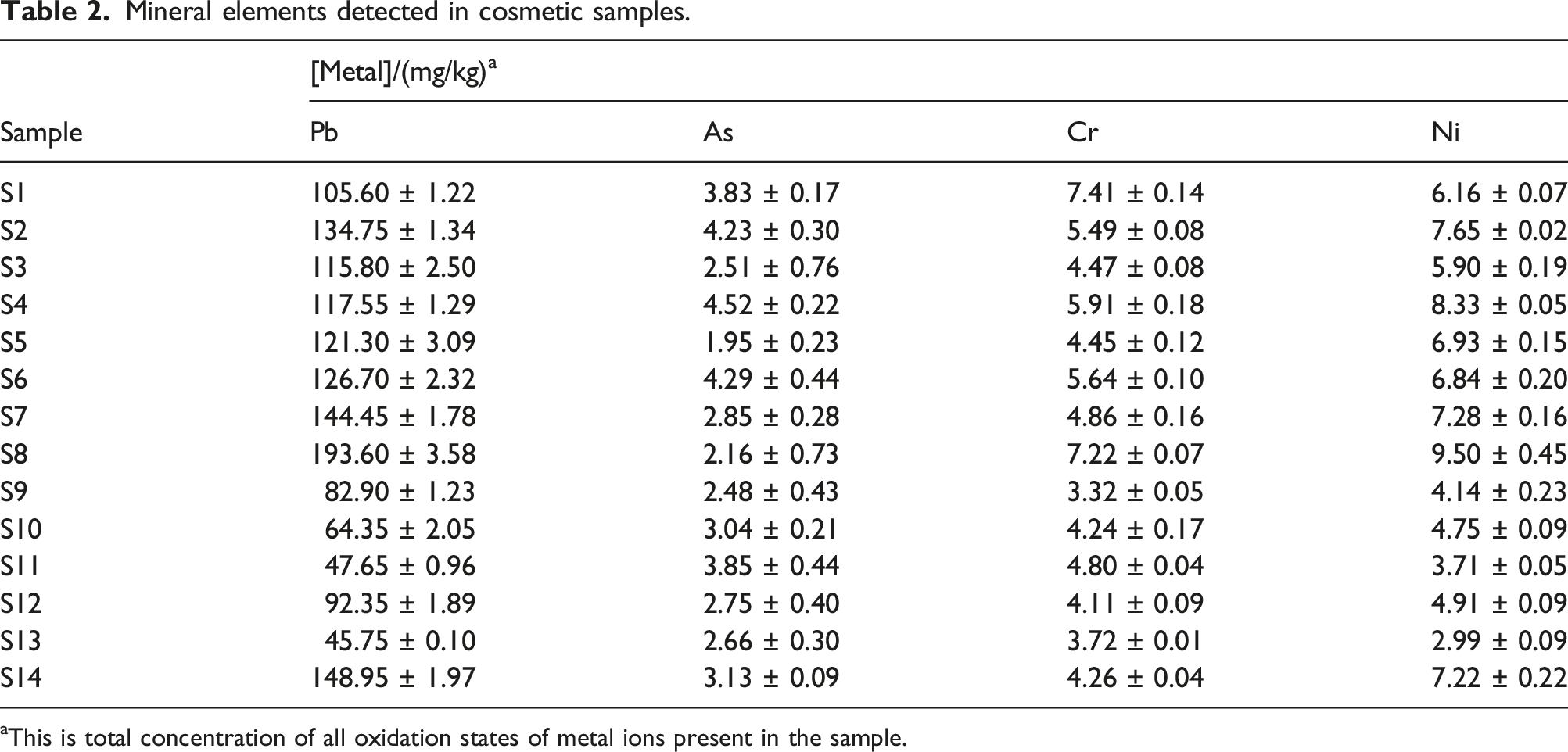
aThis is total concentration of all oxidation states of metal ions present in the sample.
There are currently no regulations in Botswana governing maximum contaminants limits for contaminants such as metal ions in cosmetic products. Regulatory agencies such as the FDA have set maximum contaminant limits for contaminants and impurities in some color additives used in cosmetics. For example, contaminant limits have been set for Pb and As to 10 and 3 mg/kg respectively and for Ni and Cr to 5 mg/kg in colour additives that are subject to certification and are allowed for use in foods, drugs, cosmetics and medical devices. In the absence of Botswana’s regulations for maximum contaminant limits of metals in cosmetic products, the results obtained in this study were interpreted based on the FDA regulations stated above. Applying these set limits, this means that the total concentration of Pb in all 14 sampled analysed significantly exceeded the maximum contaminant limit of 10 mg/kg for Pb in cosmetics. 34 In some samples the concentration of Pb was more than 15 to 20 times above the set limit. Similarly, S8 was found to contain the highest concentration of Ni (9.50 ± 0.45 mg/kg). Only five out of the 14 cosmetic samples studied contained concentration of Ni above the set maximum contaminant limit of 5 mg/kg. 35 The largest concentration of Cr was determined in the S1 (7.41 ± 0.14 mg/kg) sample that also originated from Guangzhou. Out of the 14 samples analysed for Cr, nine were found to contain Cr above the set maximum contaminant limit of 5 mg/kg. 35 On the other hand, the highest concentration of As (4.52 ± 0.22 mg/kg) was found in the S4 sample that has been imported from Italy. Only six out of the 14 products analysed contained As above the set limit of 3 mg/kg. 34 The high concentrations of metals in the skin lightening products should sound an alarm for the users because of the adverse health effects of these metals. It is also noteworthy that the metals found in cosmetic products are classified as unintentional contaminants and seen as impurities in the products and therefore it is not a requirement that should be listed on the product labels. 9 These metal impurities come about either as a byproduct of the manufacturing process or contamination arising from raw materials used in the production of cosmetics. 9 Heavy metals such as Pb, As, Cr and Ni are naturally occurring in the environment and their sources include soils, rocks, water and organic matter and are bound to be present in raw materials used in the manufacture of various products including cosmetics. As mentioned earlier, the quality and authenticity of cosmetic products has been relegated to the manufacturers to remove the impurities and toxic ingredients from the products before they reach consumers. However, only few manufacturers such as those in Canada heed this advice from governments and remove impurities from the final product. 36 In some instances, some manufacturers may not even be aware that their products are contaminated with such metals because of lack of manufacturer testing and regulatory oversight.
Health risk assessment of tested cosmetic products
The health risks presented by exposure to heavy metals such as Pb, As, Cr and Ni were evaluated using ADID, HQ and HI. It is critical that the adverse effects of these heavy metals on human health should be taken seriously because of the daily application of these products on the face and their contact with vital body organs such as eyes, mouth, nose and ears that may serve as passageways through which such toxic metals are absorbed into the body. 37 Some of these heavy metals such as Pb have been found to be detrimental to human health even at low concentrations (10 μg/dl) in the blood. 11 For example, low concentrations of Pb can act as a selective blocker of voltage-dependent calcium channels and can also decrease re-absorption of small organic molecules on renal mitochondria. 27
Health risk assessment of cosmetic product.
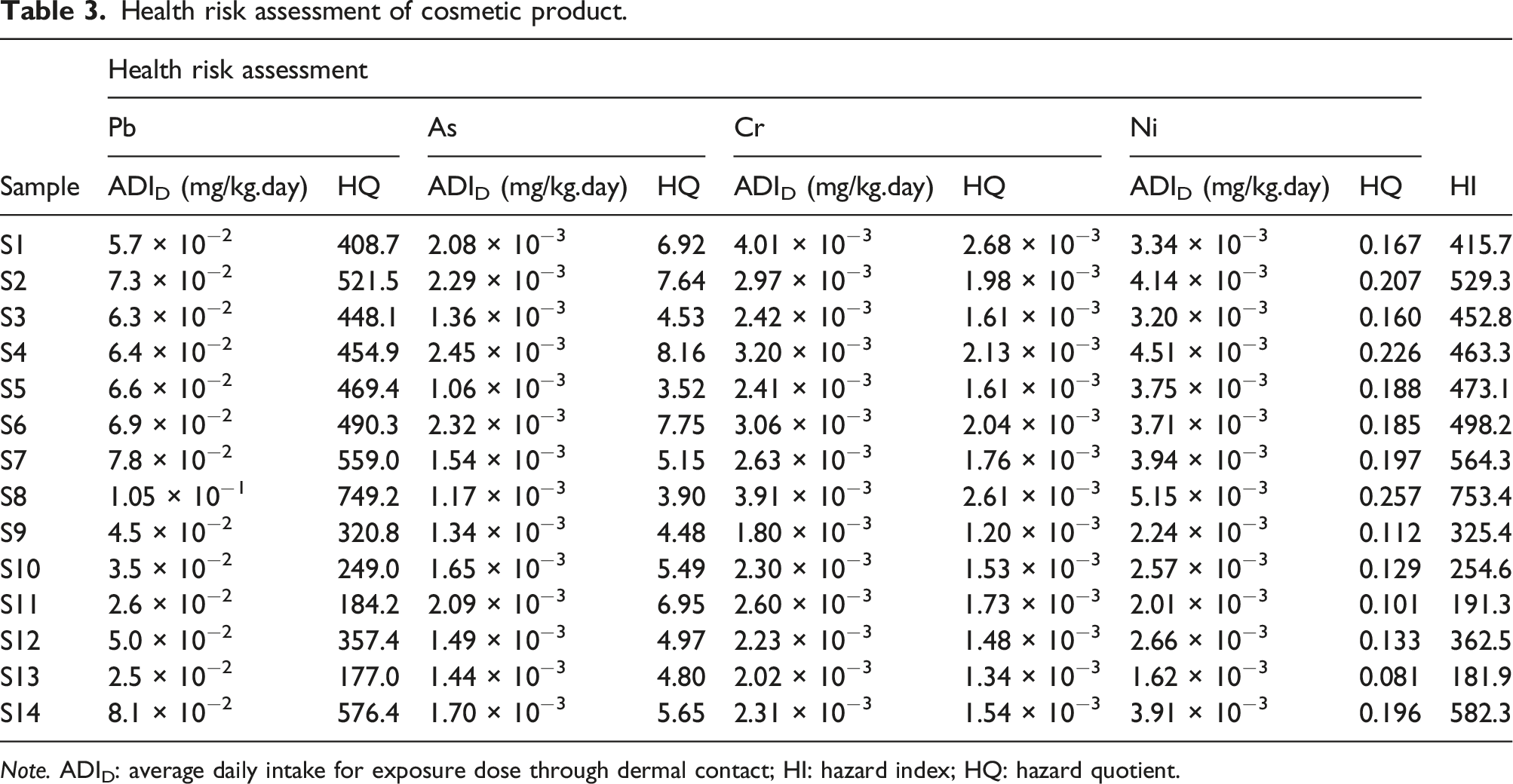
Note. ADID: average daily intake for exposure dose through dermal contact; HI: hazard index; HQ: hazard quotient.
Conclusion
This study demonstrates ICP-OES analysis of Pb, Cr, As and Ni in skin lightening cosmetics with excellent recoveries ranging from 96.3–105.2% which indicates reproducibility and reliability of the analytical procedure. Total metal concentration for Pb in all 14 skin lightening cosmetics examined far exceeded the set maximum contaminant level as per the USFDA. Total concentrations of Cr, As and Ni were higher than the set maximum contaminant limits in 9, six and five of the 14 samples examined respectively. The hazard quotients for Pb in all 14 cosmetic samples were greater than 1, an indication of potential adverse health effects from Pb in all products analysed. Furthermore, the daily dermal exposure and average daily intake for Pb in all 14 samples both exceeded the reference daily doses. Thus, health risk assessment results emanating from hazard quotient, daily dermal exposure, average daily intake and health risk index for Cr, As, and Ni indicate potential risk to human health. It is important that the health risk assessment of the metals under study was estimated based on total concentration of all metal ion species present in the samples and specification of individual metal ions was not taken into consideration. This might have inflated the health risks of the metals investigated. This presents a weakness for this study and therefore, future studies should focus on the various species of these metal ions present in cosmetic products to better understand their topical and potential transdermal absorption. These findings indicate that facial skin lightening cosmetics imported into the country are likely to have harmful health effects to consumers. This, therefore, calls for strict regulatory guidelines on heavy metals in skin lightening cosmetics imported into Botswana. Furthermore, there are currently no regulations in Botswana governing maximum contaminants limits for contaminants such as metal ions in cosmetic products. One of the strengths of this study include public awareness of toxic heavy metals in skin lightening cosmetics needs to be raised and consumers need to be educated on the potential adverse human health effects from topical exposure to heavy metals. Lastly, strategic partnership with neighbouring countries such as South Africa that have well laid out regulations on the importation and distribution of cosmetic products could go a long way in assisting Botswana combat the importation of these products in the country.
Footnotes
Acknowledgements
The authors would like to thank Botswana International University of Science and Technology (BIUST) for wholly sponsoring this study.
Author contributions
PD: Conceptualisation; methodology; student supervision; manuscript writing; data analysis. OM: Data collection; data analysis; writing – initial draft. TTK: Data analysis; manuscript reviewing. RK: Methodology; validation; manuscript revisions. All authors have read and agreed to the published version of the manuscript.
Declaration of conflicting interests
The authors report no potential conflict of interest regarding research, authorship and publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research work was wholly financially supported by the Botswana International University of Science and Technology (BIUST), Research Initiation Grant Number BIUST/ds/R&I/26/2016.
Ethical approval
Not applicable
Informed consent
Not applicable
Availability of data
Not applicable