Abstract
The oil palm, an ancient tropical tree species that originated in West Africa, has a history of centuries-long use both as a food and a medicine. Based on its higher saturated fatty acid composition, primarily palmitic acid, concerns have been flagged about its nutritional attributes. Elevation of low-density lipoprotein cholesterol effects of the associated palmitic acid is far less profound than animal sources of this fatty acid and is linked with multiple health benefits. Recently, the European Food Safety Authority raised issues about potential health risks of the chloropropanols, heat- or acid-induced food contaminants created during the refining of all edible oils, and some hydrolyzed proteins. Despite the fact that the levels of 3-monochloropropane1,2 diol and its glycidyl esters are generally <800 ppb and without demonstrated toxicological effects in humans, the chloropropanols in palm oil appear to be a lightning rod for global criticism. The toxicological data are reviewed and evaluated, and an approach for mitigation of the emerging challenge is suggested.
Introduction
In March 2016, the European Food Safety Authority (EFSA) issued an extensive report warning about the possible health consequences of contaminants created during the processing of edible oils. 1 EFSA specifically focused on 3-monochloropropane1,2 diol (3-MCPD), which has been classified as a group 2B (possible human carcinogen) since 2013. 2
Importantly, EFSA did not recommend dietary changes based on its findings, likely in light of the methodologic limitations of in vitro and rodent studies, the clustering of associated MCPD esters with a differential spectrum of absorption and metabolism, and the apparent capacity of certain cell lines either to potentiate or to reduce distribution and downstream effects of the putative toxicants. A paucity of uniform and long-term toxicity or carcinogenicity data likely also obviated dietary change recommendations. While selected European infant and follow-on formulas containing palm oil exceeded the established tolerable daily intake (TDI) of 3-MCPD, these levels were based upon projections from current consumption patterns and projected exposure scenarios which may not accurately reflect actual intake. Thus, a number of potential concerns seemed to have contributed to the EFSA report being provocative but equivocating and somewhat confusing.
While the chloropropanol family of compounds appears to present a well-defined hazard, the actual risk to human health has yet to be clearly defined and appears small in light of the accumulated data of potential exposures through foods.
As suggested previously, the overarching issue of palm oil is complicated by growing evidence-based claims for its cardiovascular health promotion effects in replacing trans-fatty acids, as well as the concerns about the sustainability practices of the palm oil industry in approximately 43 countries with the principal five production areas in Malaysia, Indonesia, Thailand, Columbia, and Nigeria.
Suffice it to say, both the potential public health and economic consequences of colliding claims and priorities require rigorous and clear-eyed assessment. The inevitable media storm may further obfuscate and emotionally charge the issues and make it even more important to systematically, and in an evidence-based fashion, place the key issues in perspective. The consulting firm, Europe Economics Research, Ltd, estimated that the palm oil industry contributes US $39 billion of Gross Domestic Product(GDP) growth to the global economy annually, mainly in developing regions. 3 It is safe to say that the multifaceted public health and economic factors at issue make it imperative that we understand the key variables as well as possible. The following review attempts to accomplish this end.
Palm oil and saturated fatty acids
The oil palm (tree) is an ancient tropical plant that originated from West Africa. Palm oil has a history of centuries-long use as both food and medicine. Crude palm oil (CPO) is obtained from the flesh or mesocarp of the fruit from the oil palm tree (Elaeis guineensis). The oil is rich in saturated palmitic acid (approximately 50% w/w), monounsaturated oleic acid (40% w/w), and polyunsaturated linoleic acid (10% w/w). CPO also contains an array of carotenoids (500–700 ppm), including βcarotene (300–420 ppm) and various vitamin E tocopherol and tocotrienol isomers (600–1000 ppm). CPO is normally refined and may be further fractionated into liquid palm olein and solid palm stearin in order to diversify its food and nonfood applications. Palm oil is highly stable during frying especially due to its unique fatty acid composition, very low levels of linolenic acid (subject to oxidation), and its relatively high content of vitamin E tocols (natural antioxidant properties). 4 It is important to note that the carotenoid content in refined palm oil is near zero, and if it were present, it is quite unstable at typical deep frying temperatures (177–191°C).
Elevation of low-density lipoprotein cholesterol effects of the constituent palmitic acid is far less profound than animal sources of this fatty acid because it is present predominantly in the sn1 and sn3 position as opposed to sn2 position as in animal fats such as lard. As such, palm oil, like other vegetable oils, including olive oil, has the favorable oleic acid in this position. Therefore, palm oil has been properly embraced as equally health promoting as olive oil. 5,6
Palmitic acid, the main saturated fat in palm oil, has a similar effect on the lipid profile as the monounsaturated fat oleic acid (18:1), typically associated with olive oil. In addition, palm oil contains oleic (18:1) and linoleic acids (18:2) and vitamin E tocotrienols which may be considered as antioxidants and may reduce or inhibit cholesterol synthesis. 7 –10 Reduction of cholesterol synthesis by fatty acids via the downregulation of acetyl-coenzyme A carboxylase and 3-hydroxy-3-methylglutaryl coenzyme A reductase has been documented in a variety of human cell lines. 11 –13
Oleic acid (C18:1n-9) in particular has positive impacts on various tissues in general and rarely is associated with negative health effects. Olive oil composition includes high amount of oleic acid (55–85%). Considering the effects of oleic acid on the cardiovascular system, it has been suggested that oleic acid intake is associated with decrease in the myocardial infarction rate, platelet aggregation, and secretion of TXA2, as well as reduction of systolic blood pressure. 14
Palm oil appears to mitigate oxidative reperfusion injury in isolated rat heart tissue and in hyperlipemic rats. 15 –19 Dietary red palm oil (RPO) supplementation has been shown to enhance functional recovery in hearts subjected to ischemia–reperfusion. Kruger et al. examined the effects of 2 mL RPO administered with a high-cholesterol diet on mitogen-activated protein kinase (MAPK) phosphorylation and apoptosis. 16 A subsequent significant reduction in p38-MAPK was associated with reduced apoptosis as indicated by significant reductions in caspase-3 and poly (Adenosine diphosphate-ribose [ADP-ribose]) polymerase cleavage. In a more recent study, Bester et al. suggested several other mechanisms involving increase in glutathione peroxidase (GPX) activity in the myocardium, but the pathway behind this increase in GPX activity remains unknown. 20 Antioxidant activity is known to play a role in prosurvival kinase signaling. Bester’s findings also suggest that RPO may increase GPX activity through upregulation of the mRNA levels of GPX4.
The palm tocotrienols may further enhance or potentiate the impact of the component fatty acids by a variety of mechanisms.
21
In addition to desirable medium-chain fatty acids, those with 6–10 carbons as found in palm kernel oil and absent in palm oil, palm oil contains an array of plant-derived compounds often associated with health benefits. Those innate substances include carotenoids (α, β, and γcarotenes), plant sterols (sitosterol, stigmasterol, and campesterol), vitamin E (tocopherols and tocotrienols), and potential antioxidants, phenolic acids, and flavonoids.
Interestingly, dietary intervention studies comparing a palm oil–rich diet with diets rich in industrial trans-fats demonstrated significantly higher levels of High Density Lipoprotein (HDL) cholesterol and apolipoprotein AI and significantly lower apolipoprotein B, triacylglycerols (TAGs), and Total Cholesterol (TC)/HDL cholesterol. 7 These results suggest palm oil may not contribute to cardiovascular disease risk factors but rather just the opposite.
Trumping all of the beneficial and probable beneficial effects of globally consumed palm oil is its nutritional contributions in infant formula products. This application highlights the need to continue palm oil production, for standardized and precise analytical detection methods for known and unknown hazardous contaminant, and for validated and cost-effective production-scale mitigation of putative contaminants. 22
As noted in the Introduction, in March 2016, the EFSA issued a report warning about the health consequences of chloropropanols, the thermally induced food contaminants created during the processing and refining of edible oils. EFSA specifically identified 3MCPD, which was classified as a possible human carcinogen by International Agency for Research on Cancer (IARC). 2 The regulatory arm of the European Union, namely the European Commission (EC) Scientific Committee on Food (SCF), adopted the TDI of 2 μg/kg body weight (bw), which includes an uncertainty factor of 500, based on a 40% dry matter content for 3-MCPD in soy sauce and hydrolyzed vegetable protein (HVP), which came into force in April 2002. 23 This large uncertainty factor is considered conservative because instead of the typical default uncertainty factor of 100 for intra and interspecies differences, the SCF based its conclusion on the fact that the TDI was derived from an Lowest Observed Adverse Effect Level (LOAEL) and not an No Observed Adverse Effect Level (NOAEL) for renal tubular hyperplasia. Furthermore, other limitations in the database included minimal reproduction and developmental toxicity studies. The Joint WHO/FAO Expert Committee on Food Additives (JECFA) also recommended a provisional maximum TDI of 2 μg/kg bw for 3-MCPD and also concluded that 1,3-dichloropropan-2-ol (1,3-DCP) is genotoxic in vitro. 24
The UK Committee on Carcinogenicity of Chemicals in Food, Consumer Products and the Environment and the Committee on Mutagenicity of Chemicals in Food, Consumer Products and the Environment have recently considered the fatty acid esters (of 3-MCPD): 1,3-dichloropropan-2-ol (DCP and 2,3-dichloro-1-propanol (2,3-DCP). These expert committees concluded that it would be prudent to regard the esters as potentially genotoxic in vivo. 25,26 The toxicity of 2,3-DCP has been shown to be lower than that of 1,3-DCP because of differences in the reactivity of their respective metabolites. 27
It is important to note that there are no adequate human toxicological data available yet on 3-MCPD and its glycidol esters. The primary toxicological concern appears to be based on the potential release of 3-MCPD or glycidol from the parent esters by lipase-catalyzed hydrolysis in the gastrointestinal (GI) tract. Although 3-MCPD is assessed as a nongenotoxic carcinogen with a TDI of 2 μg/kg bw, glycidol is considered to be a genotoxic carcinogen, which induces tumors in numerous organs of rodents. The initial exposure estimates, conducted by the German Federal Institute for Risk Assessment (BfR) under the assumption that 100% of the 3-MPCD and glycidol are released from their esters, revealed especially that infants being fed commercial infant formula could ingest harmful amounts of 3-MCPD and glycidol. However, the actual oral bioavailability of 3-MCPD is unknown and may be lower than the projected TDI.
It is noteworthy that the cautionary data from in vitro investigations suggest that 3-MCPD esters are accepted as substrates by gut lipases and thus potentially could be hydrolyzed in the mammalian gut. In studies by Robert et al., formation of 3-MCPD from vegetable oils and fats was observed in the presence of mammalian, vegetable, and fungal lipases. 28 Hydrolysis of 3-MCPD esters was demonstrated by incubating monoesters (3-MCPD mono-stearate, 3-MCPD mono-oleate, 3-MCPD mono-palmitate, and 3-MCPD mono-myristate) and diesters (3-MCPD palmitate–oleate) in an intestinal model containing pancreatic lipase and porcine bile extract.
Moreover, in recent in vitro studies with differentiated Caco-2 cells, a model system of the human small intestine, it was found that 3-MCPD monoesters could be efficiently hydrolyzed in the presence of Caco-2 cells, whereas 3-MCPD diesters were metabolized by the cells. This metabolic process importantly resulted in the loss of the 3-MPCD moiety. It was also observed that free 3-MCPD could cross a Caco-2 monolayer by a paracellular diffusion mechanism. While these are in vitro data that do not model the in vivo effects of the food matrix, they do suggest that 3-MCPD fatty acid esters may be hydrolyzed and release 3-MCPD that could be absorbed in the human intestine. Also, it is noted that 3-MCPD esters were detected in human breast milk, suggesting that dietary 3-MCPD esters might be bioavailable in humans. 29,30 Importantly, these data do not necessarily reflect exposure or clinical significance. Bioavailability of these esters in a food matrix introduced via normal enteral feeding, and perhaps subsequently found in breast milk, is simply not known.
Braeuning et al. present an analysis of proteomic alterations in rat liver upon oral repeated-dose exposure to 3-MCPD or its ester 3-MCPD dipalmitate. 31 Previous studies have suggested that 3-MCPD esters are not systemically bioavailable but are efficiently cleaved to free 3-MCPD and fatty acids in the GI tract, thus potentially leading to comparable intestinal absorption of 3-MCPD following exposure either to the free or an esterified version of 3-MCPD. 32,33 Therefore, it has been suggested that toxic responses to 3-MCPD and 3-MCPD esters are similarly caused by free 3-MCPD or its metabolites and further support for this view comes from recent observations at the proteomic level, indicating similarities in the response of rat testis to 3-MCPD or 3-MCPD dipalmitate exposure. 34 Again, this issue is considered of critical relevance since dietary consumption of 3-MCPD and 3-MCPD esters simultaneously present in foodstuff (i.e. free 3-MCPD and the 3-MCPD released from esters in the intestine) might lead to exceeding the TDI for 3-MCPD of 2 μg/kg bw. 25,35,36 Adding a measure of alarm to this theoretical possibility, the BfR calculated that infants might be exposed to up to 25 μg/kg bw/day 3-MCPD, corresponding to a 12.5-fold exceedance of the TDI, while the worst case assumption for adults resulted in an approximately 5-fold exceedance of that threshold value. 25 However, if the benchmark dose (BMD) approach and a BMDL10 value as point of departure instead of the LOAEL are used, part of this uncertainty factor of 500 would no longer be needed, and one could derive a TDI that is no longer below the current worst case intake estimates, indicating there is no need for concern. Furthermore, glycidol, a genotoxic carcinogen, risk assessment can be based on estimating a margin of exposure using the BMDL10 value derived from the 2-year National Toxicology Program (NTP) carcinogenicity study on glycidol administered via gavage. 37 Based on this approach, it emerges that for most groups within the population, except for babies fed infant formula, there would be only a low level of concern. Under the very conservative estimates used for California’s Safe Drinking Water and Toxic Enforcement Act of 1986, the no significant risk level for glycidol was calculated to be 0.54 µg/day.
Human exposure is well below the animal LOAEL and the doses of 3-MCPD or 3-MCPD dipalmitate administered in the BfR study which were equivalent to 2.5 or 10 mg/kg bw 3-MCPD per day, respectively. 29 However, as infants might exhibit an increased sensitivity toward the toxicity of 3-MCPD, the BfR risk assessment came to the conclusion that infants who are fed commercial infant formulas might be at risk from 3-MCPD at the doses taken up via the diet.
Toxicological animal studies using mice and rats have shown that the most sensitive target organs for 3-MCPD toxicity are the kidney and the male reproductive system. 38 –40 The metabolic studies have showed that 3-MCPD is distributed in the kidney at high concentrations, while subchronic studies showed that nephrotoxicity is an early effect of 3-MCPD, progressing throughout the lifetime of exposed rats. 30,33 In some cases, especially in rats fed with high doses of 3-MCPD for a prolonged period, hyperplasia and tumors in kidneys and reproductive organs have been reported. 31,41
Lynch et al. and Bakhiya et al. have shown that 3-MCPD can be oxidized via β-chlorolactaldehyde to β-chlorolactic acid, finally yielding oxalic acid. 29,38 Such metabolites are assumed to be involved in the nephrotoxic mechanisms. 32 β-chlorolactic acid has been reported to suppress the glycolytic enzymes glyceraldehyde-3-phosphate dehydrogenase and triosephosphate isomerase. 29,30,33 Hence, the renal injury caused by 3-MCPD might be associated with glycolysis and energy production. Using a proteomic approach, it was shown that 3-MCPD caused the perturbation of carbohydrate, amino acid, and fatty acid metabolism, and the urea cycle and the TCA cycle as well as the respiratory chain in the rat kidney. Furthermore, glutathione S-transferase pi1 has been shown to be a sensitive marker for early 3-MCPD-induced toxicity. 29
Thus, the putative human risk relates to potential renal and testicular effects, not to liver toxicity, as kidney and testis appear to be the most sensitive target organs of 3-MCPD toxicity in animal studies. Subtle hepatic effects of 3-MCPD dipalmitate, however, have been reported in a 90-day feeding study with Wistar rats which received a dose of 267 mg/kg bw/day via the diet. 42 Not surprisingly, suspected mechanisms of 3-MCPD toxicity have long been generally discussed in the same terms as many other putative toxicants and include the inhibition of glycolysis and oxidative stress.
Stevenson and Jones found that the nature of 3-MCPD antifertility in boars was related to the inhibition of the glycolysis pathway. 43 Glycolysis is closely related to sperm motility, as it is a supplement for the lack of oxidative phosphorylation. 31,44,45 The blockage of glycolysis by 3-MCPD in tyrosine protein phosphorylation impairs the cyclic Adenosine Monophosphate / Protein Kinase A (cAMP/PKA) pathway in isolated rat epididymal spermatozoa, and 3-MCPD treatment induces the decrease of intracellular Adenosine Triphosphate (ATP) contents, cAMP levels, and protein kinase substrates dramatically in rat sperm. 46 In male rats, it was demonstrated that the spermatotoxic effect of 3-MCPD was mediated by reduced H+-ATPase expression and altered pH level. 47 In rat testicular cells, 3-MCPD has been shown to induce apoptosis at moderate levels with consistent increases in the activity of caspase-3 in a 90-day toxicology study that administered three doses, 29.5, 7.37, and 1.84 mg/kg bw/day. 48
Sun et al. exposed 3-MCPD to R2C Leydig cells and found that this substance significantly induced early apoptosis using a single cell gel/comet assay and morphological analysis. This report showed the involvement of the expressions of steroidogenic enzymes (Steroidogenic Acute Regulatory protein [StAR], P450scc, and 3 β-HSD) in immature Leydig cells caused by 3-MCPD. The findings suggest that the 3-MCPD may be mediated by Matrix Metalloproteinase protein (MMP) and cAMP levels subsequent to a reduction in R2C cells progesterone production. In addition, 3-MCPD induced morphological changes and DNA damage of Leydig cells, demonstrating that 3-MCPD might have caused early apoptotic cell death. 49
The toxicological potential of 2,3-DCP regarding hepatotoxic effects seems to be lower than that of 1,3-DCP. Limited data show that 2,3-DCP occurs only in trace amounts in food, indicating that exposure to 2,3-DCP also seems to be of lower concern for human health. The dietary 2-monochloro-1,3-propanediol (2-MCPD) burden also appears to be lower than that of 3-MCPD. An adequate risk assessment for 2-MCPD cannot be performed due to limited data on the toxicology and occurrence in food. 32
Despite numerous studies to elucidate the toxicity of 3-MCPD, the molecular mechanisms accounting for its cytotoxicity have not been fully clarified in human-derived cells. One study was exceptional in that it was undertaken to examine the cytotoxic effects of 3-MCPD in embryonic kidney cells derived from a human. 50 The results suggested that 3-MCPD induced apoptosis through mitochondrial-mediated caspase cascade-dependent pathway. Especially, the dysfunction of mitochondrial oxidative phosphorylation and the blockage of energy production played pivotal roles in 3-MCPD-triggered apoptosis. Induction of apoptosis in vitro is always intriguing, but the clinical significance of this observation serves to raise questions that require further investigation.
Summary of implicit challenges and confounds of the EFSA reportEFSA did not put the data into context
It is apparent that there may be potential problems, but relevant toxicokinetic data among humans do not exist. There is not any direct evidence that current 3MCPD levels in foods, and at their typical consumption amounts, trigger any adverse health effects, even following years of potential exposure to the human population including infants globally. Thus, public health advisements are based on limited animal studies and projected exposure among humans of various population or age groups. The data indicate that fewer and lower amounts of 3MCPD compounds are present in some sources of Malaysian palm oil than oils processed outside of Malaysia. This observation may be ascribed to a variety of reasons, including better crop management in the field, variations in harvesting of fruit bunches, and their subsequent oil extraction in the mills coupled with refining techniques that may possibly alter opportunities for formation of the chloropropanols despite the high temperatures and pressure involved.
Nonetheless, this observation indicates the need for a better grasp of the palm oil process variances among countries and indeed even among different refineries. There is a need to carefully evaluate each step and identify the conditions under which contaminants, such as 3MCPD and related chloropropanols, are generated. There are some data, for example, that indicate the timing of the harvest and fruit milling, as well as processing temperature, can significantly affect the generation and subsequent levels of these contaminants.
The toxicology is not fully vetted.
As of 2011, there were not any toxicological data available either for 3MCPD esters or glycidol esters, except for a bacterial mutagenicity assay performed with glycidyl stearate. Several investigators noted toxicological data gaps and call for further research. More recent data have allowed several regulatory and authority bodies to develop a TDI of 2 μg/kg bw for 3-MCPD. There remains a need for additional research that assesses potential causes and effects of 3MCPD on human health when consumed at typical levels over a life span. 51
Balancing concerns with health potential
RPO has been recognized as a rich source of pro-vitamin A (PVA) carotenoids for decades. Indeed, it was used to ameliorate vitamin A deficiency (VAD) in India in the 1930s and West Africa in the 1940s. Because of its exceptionally high PVA carotenoid content, RPO has been proposed as a food-based intervention to prevent VAD and tested for this purpose in various small-scale human feeding studies. 52–54
While palm oil may be at the center of the 3MCPD controversy, care should be taken not to neglect the economical, sustainable source of healthful nutrients innate to palm oil. The best quality RPO is processed using molecular distillation processes, and the levels of 3MCPD and glycidyl esters are almost nonexistent in such products.
In the realm of food processing, the removal of 3MCPD from refined oil has been viewed as dependent upon the efficiency and efficacy of the deodorization process. This is the last step of refining in which unwanted constituents are removed. There is also some evidence that repeated heating of oils, such as in a quickservice environment, may accelerate or enhance the amount of cocontaminants in these food components. The end users would need to be educated on safer use guidelines, including monitoring and changing their oil based on the potential adverse threshold levels of these compounds.
It is important to note that Razak et al. studied 105 CPOs and found that 80% contained no detectable 3MCPD esters. 55 Even more significantly, for palm oils from different locations, Matthäus et al. found impressive variation in the amounts from 3 to 14 mg/kg of 3MCPDs formed in refined oil from varying regions within Malaysia. 56 This was hypothetically attributed to differences in climate, soil, growth conditions, all of which impact the distribution and concentration of chloride salts in the water used for irrigation, soil and in fertilizer, cultivar genotype, harvest technique, and approach to processing.
Discussion: The path forward
Recent in vivo Absorption, Digestion, Metabolism, Excretion (ADME) data suggest that 3MCPD esters are hydrolyzed in the mammalian GI tract. Exposure estimates further suggest that infants being fed commercial formula could well ingest potentially harmful amounts of 3MCPD. While all of these toxicological data serve to posit a hazard, the current data simply do not support a definite increased risk, especially in light of the importance of palm oil in a variety of foods, foods which are consumed across population groups and throughout the life cycle. However, the recent EFSA report suggests that it is incumbent upon the edible oil industry to explore technologies that would mitigate the potential 3MCPD hazard in the food supply.
The free form of 3MCPD (3monochloropropanediol fatty esters) was discovered in 1978, and its esterified form was found in 1980. On the basis of some carcinogenicity and genotoxicity studies in animals, the JECFA and the EC SCFs have set a TDI of 2 μg/kg bw/day and a regulatory limit of 20 μg/kg bw/day. 57 Despite conflicting data, most regulatory agencies, including the US FDA in 2006, have recommended that 3MCPD not exceed 1 ppm in foods, such as acid HVP and Asian-style sauces that humans consume. One of the points of controversy is that palm oils may contain as high as 2.9 ppm of 3MCPD, whereas sampled vegetable oils, including palm kernel, rapeseed, coconut, soybean, corn, peanut, and sunflower seed oils typically contain 0.5–0.8 ppm. 1
Several investigators have delineated the formation mechanisms of monochloropropandiol-fatty esters (MCPD-FE) during palm oil refining. 58 –60 Because the highest levels of MCPD appear to be in palm oils, our focus is on palm oil production and refining (see flow diagram (Figure 1) adapted from Craft et al. 61
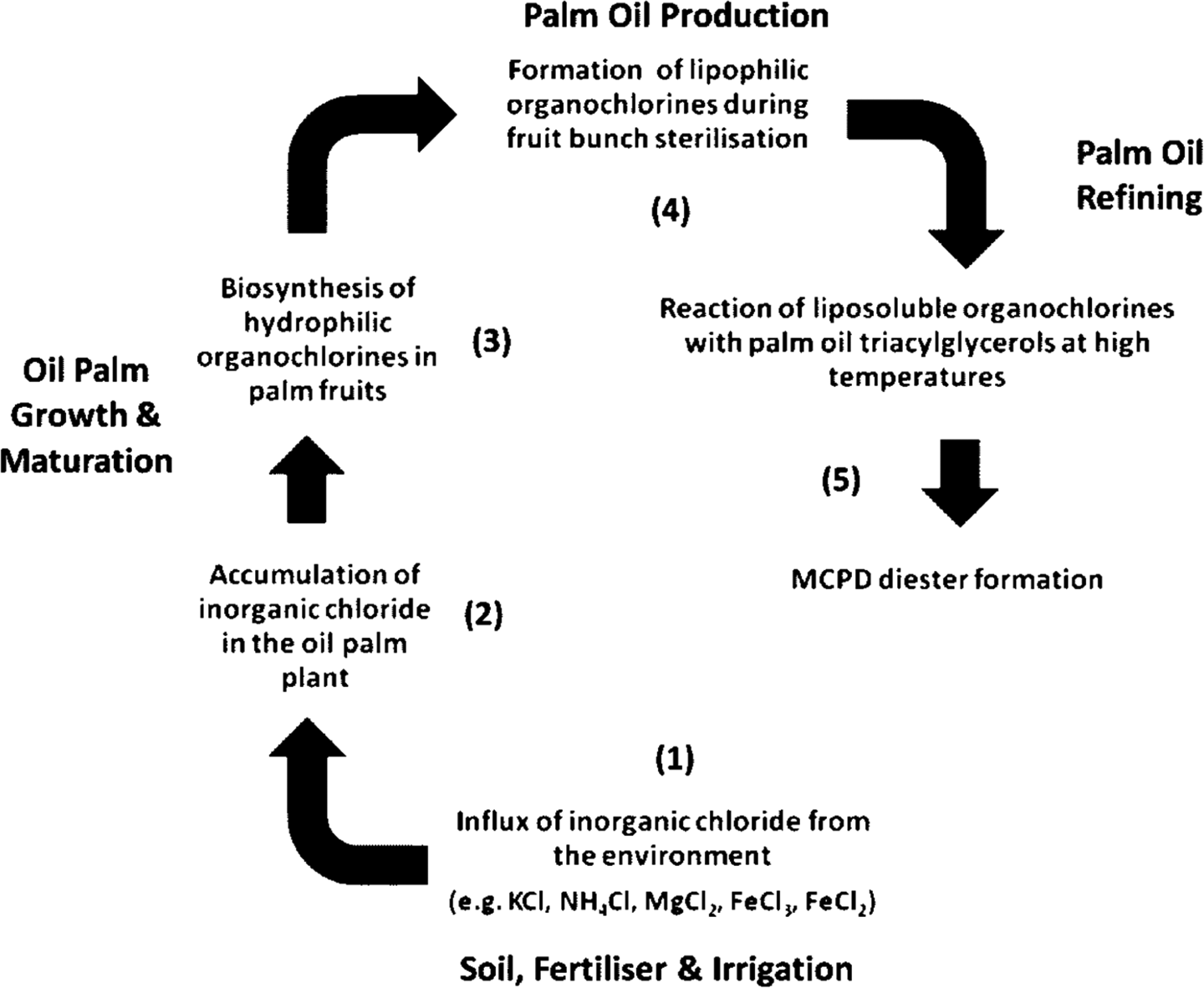
Root-cause analysis of the factors involved in the formation of MCPD diesters within the palm oil process chain. (1) Chlorine input into oil palm production, (2) accumulation of inorganic chloride in the plant, (3) bioconversion of inorganic chlorides to organochlorines in the fruits, (4) formation of liposoluble organochlorines during fruit bunch sterilization, and (5) reaction of liposoluble organochlorines with palm oil TAG during oil refining and resulting in MCPD diesters. MCPD: monochloropropanediol; TAG: triacylglycerol.
Regardless of the uncertainty of hazard identification and risk assessments, the industry must focus on mitigation both in the laboratory, on the pilot plant scale and finally at the production level. It is important to understand that the formation of 3MCPD may result from one of the three pathways: The first is from acid hydrolysis of vegetable protein. Note, like the majority of commercial edible vegetable oils in the United States, neither palm nor palm kernel oil contains protein; the second involves thermal processing and domestic cooking; and the third occurs with the release of free 3MCPD from the bound esterified form (which can occur in the human GI tract).
A key emphasis is the precursor compounds that contribute to the formation of chloropropanols, the most important of which may be chlorine in both organic and inorganic forms. A spectrum of constituent lipids may interact with chlorine donated from iron, magnesium, and calcium chlorides, as well as from endogenous plant metabolites. 15 The most critical lipid precursor or substrate appears to be the TAGs, which seem to preferentially react with chlorine donors, that are apparently pervasively present, at a certain temperature to form MCPDFE. 62
Despite inadequate data and ongoing controversy, the recent EFSA report calls attention yet again to levels of a myriad of putative toxicants known to be present in all refined vegetable oils, infant foods, as well as human breast milk. Edible oil producers know that their success depends on consumers’ trust that the oils they buy are healthy and safe and without potential hazards. Thus, updated awareness of this issue and an accurate appreciation of its clinical implications are imperative. Like many well-founded efforts to evaluate possible toxicants in food, this goal is complicated by the absence of clinical studies on humans, and the complexities and variation in food harvesting, processing, refining, preparation, and patterns of consumption. 15
Conclusion and recommendations
While increased scrutiny and enhanced analytical methods facilitate ever lower detection of compounds such as 3MCPD, one question before us is whether the quantities and the possible metabolic downstream health impacts are clinically significant hazards. 63 The edible oil industry has already begun to move toward exploration of approaches to mitigation of 3MCPDs, but overreaction based upon low level or insufficient evidence may demonize a nutritionally important group of foods, namely edible vegetable oils. Impulsive elimination of edible oils from the diet could have more serious health consequences than continued inclusion even with the presence of chloropropanols, which are destined to be minimized or removed by additional processing technologies such as short path distillation. 64
From a global perspective, all seed oil processing, including palm, soybean, canola, rapeseed, and sunflower seed, produces 3-MCPD and related contaminants. It may be important also to note that data indicate that fewer 3-MCPD compounds are present in Malaysian palm oil than that which is processed outside of Malaysia. This observation indicates the need for a better grasp of the palm oil process variances among countries. There is a need to carefully evaluate each step and identify the conditions under which contaminants, such as 3-MCPD and related chloropropanols, are generated. There are some data, for example, that indicate the timing of the harvest and pressing, as well as processing temperature, that can significantly affect the generation and subsequent levels of these contaminants. It is incumbent upon the seed oil processing industry to move forward to eliminate such contaminants from their products to maintain the confidence and trust of their consumers.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
