Abstract
Regulatory agencies around the world have found sucralose to be a safe ingredient for use in food. A recent review by the German Federal Institute for Risk Assessment (BfR) hypothesized that sucralose use in foods heated during their manufacture might pose a health risk, by resulting in the formation of certain chlorinated compounds; specifically, polychlorinated dibenzodioxins (PCDDs), polychlorinateddibenzofurans (PCDFs) and/or free or bound 3-monochloropropanediol (3-MCPD), some of which are considered potential carcinogens. The BfR further encouraged the European Food Safety Authority (EFSA), which is in the process of conducting a staged re-evaluation of a range of food additives, including sucralose, to specifically address their hypothesis. This paper reports the results of new studies requested by EFSA to analyze for the presence of PCDDs, PCDFs and 3-MCPDs in a range of foods. As requested, foods were prepared with typical sucralose use levels and thermally processed under typical food processing conditions. The presence of the compounds of interest were analyzed using validated and accepted analytical methods (e.g. US Environmental Protection Agency (EPA); American Oil Chemists Society (AOCS)). The results of these new analytical studies show no evidence for the formation of these compounds due to sucralose presence. This paper also reports a critical analysis of the studies cited in the BfR review as the basis for its hypothesis. This analysis shows that the cited studies do not represent food manufacturing conditions and are thus not reliable for predicting the fate of sucralose in foods. This work reaffirms that sucralose is safe for use in food manufacture, including when heating is required.
Highlights
The current work refutes a recent review that hypothesized that sucralose use in foods exposed to heat during manufacturing may pose a risk to human health, by possibly resulting in the formation of certain chlorinated compounds; specifically, polychlorinated dibenzodioxins (PCDDs), polychlorinated dibenzofurans (PCDFs), and/or free or bound monochloropropanediols (3-MCPDs).
Direct analyses reported herein show that sucralose is not a cause for the formation of any of these compounds (PCDDs, PCDFs or free or bound 3-MCPDs) in a range of sucralose-containing baked/cooked foods made under typical food processing conditions.
A critical analysis of the studies cited as the basis for concern over the use of sucralose in foods heated during their manufacture also reveals no empirical evidence for the concern.
The collective research adds further evidence that sucralose is safe for use in food manufacture, including cooking/baking.
Introduction
Regulatory agencies around the world have found sucralose to be a safe ingredient for use in food manufacture. These agencies recognize that food manufacturing conditions, whether in the home or in mass production facilities, expose ingredients in food to different types of conditions—conditions that may alter the nature of the ingredients present and thereby affect the safety of the final food product. As a consequence, with the proposal of any new food ingredient, studies are required to demonstrate stability of the ingredient under conditions representative of normal food manufacture. 1,2 Such studies were a part of the sucralose food additive petitions submitted to regulatory agencies. 3,4 Several books and papers discuss the key findings from this research, 5 –7 which shows that sucralose has remarkable stability. This is reflected in regulatory rulings permitting the use of sucralose in food, which includes use in a wide range of categories. 3,8,9
As an example, the US Food and Drug administration (FDA) permits sucralose as a general purpose sweetener, meaning it can be used to sweeten any food product. 9 This ruling permits use under all conditions of normal food manufacture, including those that require heating for cooking/baking or sterilization purposes.
More recently, certain studies have questioned whether sucralose is stable under food manufacturing conditions that expose it to heat. A review of these studies, by the German Federal Institute for Risk Assessment (Bundesanstalt für Risikobewertung or BfR), concluded that use of sucralose, a chlorinated disaccharide, in foods that are heated might lead to the formation of potentially toxic chlorinated compounds, such as congeners of polychlorinated dibenzodioxins (PCDDs), polychlorinateddibenzofurans (PCDFs) and 3-monochloropropanediol (3-MCPD), although noting that “there are currently insufficient data to draw final conclusions.” 10 A recent paper by Eisenreich et al. 11 similarly reports/discusses the BfR review process and conclusions.
The BfR further encouraged the European Food Safety Authority (EFSA) to specifically address their hypothesis. EFSA is currently in the process of re-evaluating sucralose as part of staged plan to re-evaluate all food additives approved for use in the EU prior to January 20, 2009 by year end 2021, as required by a 2008 European Union (EU) Directive. 12,13 To support its re-evaluations, EFSA launched a public call for technical and toxicological data regarding the food ingredients in scope, which includes sucralose. Related to this, EFSA recently requested (unpublished) specific food preparation studies to investigate the potential for sucralose to result in the formation of specific PCDDs and PCDFs and 3-MCPD. It should be noted that these substances are found commonly in a wide range of foods, 14 –18 however, some animal and cellular research has indicated potential carcinogenicity and, for this reason, some countries have set upper limits for the presence of certain of these substances in certain foods. 19,20 That said, the public health risk is considered to be low, 14 and human studies do not provide evidence of a safety risk from exposure to these substances with usual food intake. 21
The current paper reports the conduct and results of the new studies requested by EFSA. It also provides a critical analysis of the studies that the BfR review cited as a rationale for needing such new research on sucralose. These undertakings add to the collective understanding of sucralose safety. This can have importance to both food manufacturers and consumers who wish to create products with lower sugar. Not all low/no calorie sweeteners are as stable as sucralose in foods, 22 meaning sucralose can be used to replace sugar in some applications where other sweeteners cannot. In general, low-calorie sweeteners approved for use have been recognized as a way to help reduce carbohydrate intake, important, for example, in diabetes meal planning 23,24 and to help reduce caloric intake, which can aid in weight management strategies. 25 –27 In addition, sucralose is used already in many food applications. 22 The new work reported herein should help to clarify the safety of sucralose when used normally for the manufacture of food products.
Research
Materials and methods
Experimental design
EFSA requested that realistic uses of sucralose in a range of foods subjected to heating under realistic manufacturing conditions be analyzed for the presence of specific non-sucralose chlorinated substances. The foods specified were:
Wafers using sucralose as a sweetener
Pizza made with sucralose-sweetened pizza sauce
Cake and biscuits (cookies) made with a sucralose-containing tabletop sweetener following typical household directions for use.
Each product was to be evaluated for the presence of:
17 PCDF + PCDD congeners with 2,3,7,8-chlorine substitution with a LOQ of at least 2 pg WHO2005-TEQg fresh weight;
3-MCPD 3-Monochloropropandi-2-ol (free and bound) with a LOD of at least 5 µg/kg.
In order to determine the effects of typical baking heat processing treatments on the formation of compounds of interest, four model food systems were formulated and processed under controlled conditions. Model food systems comprised a wafer, cake, biscuit (cookie) and pizza sauce (placed on a typical pizza crust to recreate typical heat treatment conditions and then collected after baking) (Tables 1 -4).
Each model food system incorporated the use of either sugar (control), sucralose or a retail sucralose-based sweetener with maltodextrin (SPLENDA® Granulated), which provides sugar-like bulk with relatively minimal calories per serving, or maltodextrin alone. The use levels chosen for sucralose, or retail sucralose-based sweetener with maltodextrin, represented amounts typically found in commercial applications and levels commonly recommended for home product preparation or greater. In order to avoid complicating experimental variables, when sucralose ingredients were applied in the experimental recipes, no additional ingredients were added to rebalance the recipes for the removal of sugar.
The presence of the toxic compounds of interest were analyzed using validated and accepted analytical methods - high resolution gas chromatography (HRGC)/high resolution mass spectrometry (HRMS) for PCDDs and PCDFs 28 and GC-MS/MS for free and bound 3-MCPD. 29
Ingredients
Sucralose (SPLENDA® Sucralose, micronized powder) and maltodextrin (STARDRI® 100) were obtained from Tate & Lyle Ingredients Americas LLC (Hoffman Estates, IL, USA). SPLENDA® Granulated (1% sucralose and 99% maltodextrin by weight), a dry, granulated, low-calorie sweetener designed to replace sugar spoon-for-spoon, on a sweetness basis, and all other recipe ingredients were purchased from a local retail grocery store. Ingredients were representative of those typically used in home and/or industrial baking applications.
Formulations and preparation
Food products were prepared as described below (Tables 1 -4). Stainless steel cookware and utensils were used for preparation of pizza sauce samples. The pizza sauce samples were cooked using a Vollrath Induction burner. Dough or batter mixing was accomplished using a Hobart® planetary mixer (Model N50A) with paddle attachment. All traditional baking was accomplished using a Picard Revolution reel gas heated oven and stainless steel baking sheets or muffin tins. A stainless steel non-stick cast electric waffle cone baker (Cone Baker Model XC-XG-1) was used for preparation of the wafer samples.
Each food type was produced in triplicate. Samples (minimum 250 g) were collected from each production run, placed in a stainless steel bowl, homogenized with an immersion blender (Braun MQ505) and ground until visually homogenous. Samples of this ground material were placed in 100 mL glass sample containers sealed with plastic lids, placed in frozen storage and held at -23°C until samples were shipped to the contract laboratory for analysis.
Wafer preparation
Wafer model formula.
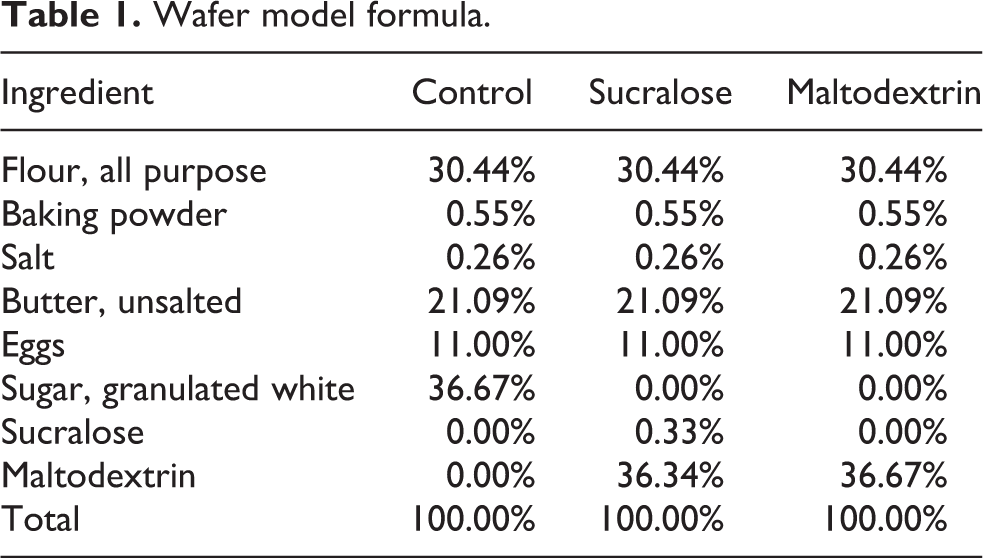
Wafers (Table 1) were prepared by combining, blending and sifting all dry ingredients. Eggs and butter were then added and mixed for 1 min on low speed to form a homogeneous batter. The batter was then transferred to and electric heating plate at 160°C and baked for 1.5 min prior to removal and cooling.
Biscuit (Cookie) preparation
Biscuit (Cookie) formula.
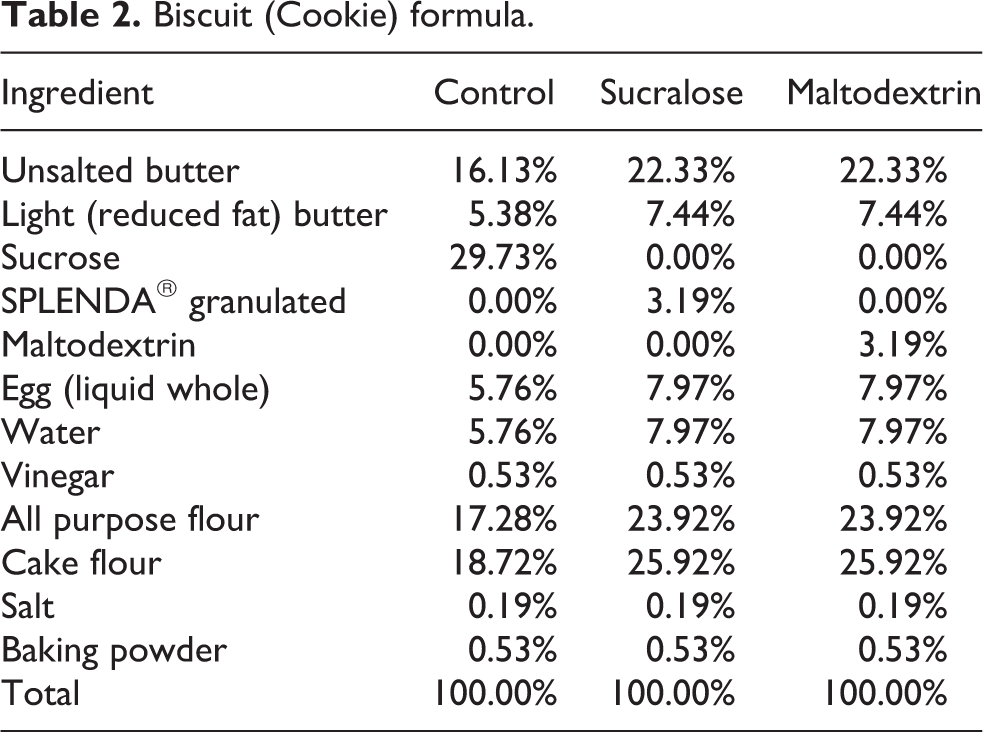
Biscuits (Table 2) were prepared by combining softened butter with sucralose, sugar or maltodextrin ingredients (according to the formulas shown). Egg, water and vinegar were then added and mixed for 30 s on low speed. Flour salt and baking powder were then added and mixed for an additional 15–30 s until a dough was formed. The dough was divided and refrigerated for 1 h at 4°C. The biscuits were portioned using a cookie scoop and placed on a baking sheet. The sheets were then transferred to an oven at 180°C and baked for 18 min prior to removal and cooling on wire racks.
Pizza sauce preparation
Pizza sauce formula.
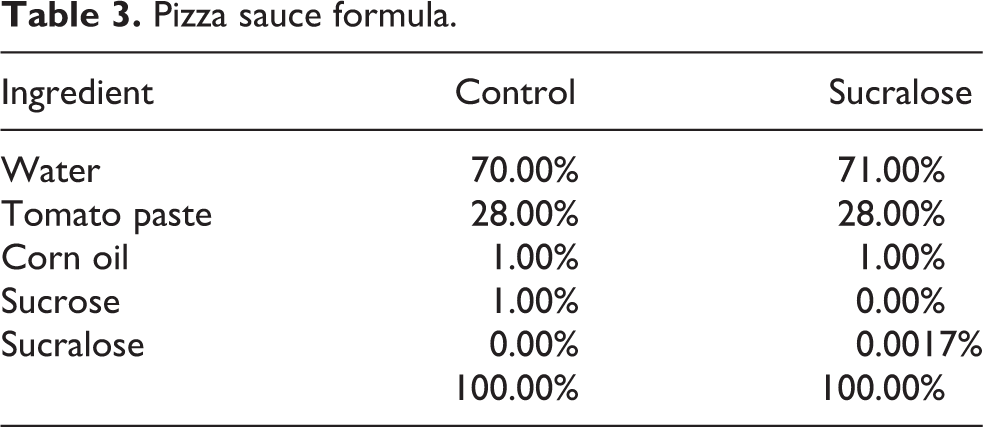
Pizza sauce (Table 3) was prepared by combining ingredients and mixing until homogeneous. The mixture was then heated to 90°C and held for 5 min. Following this hold time, the sauce was placed on a commercial par-baked pizza crust and baked in an oven at 180°C for 16 min. The pizza was then removed from the oven and allowed to cool before the sauce was collected by scraping it from the pizza surface.
Cake preparation
Cake formula.
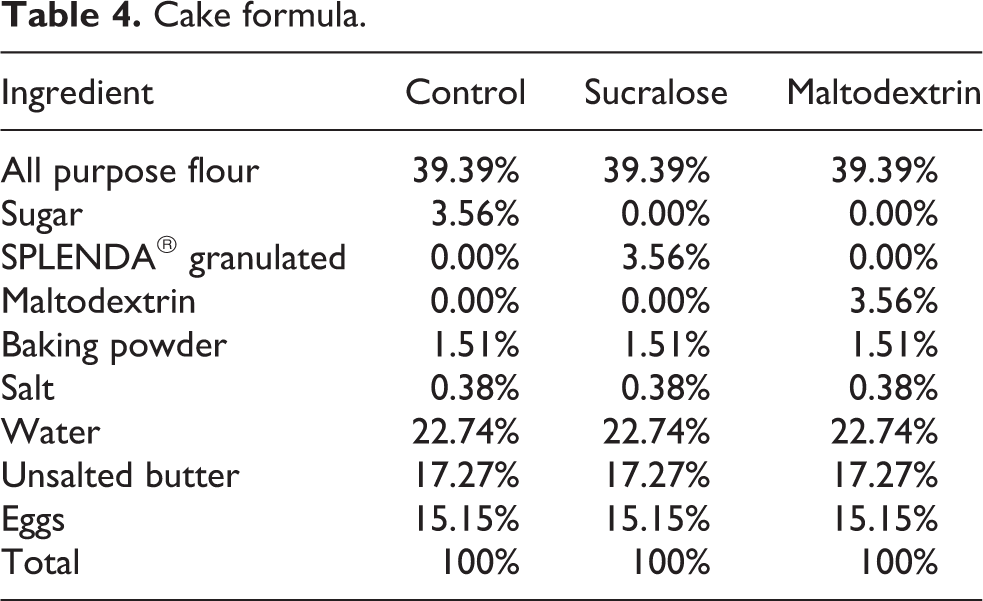
Cakes (Table 4) were prepared by creaming the SPLENDA® Granulated, sugar or maltodextrin with the unsalted butter on low speed for 15 s, then medium speed for 1 min. The flour, salt and baking powder were sifted together. The eggs were beaten and then added into the creamed mixture at low speed for 15–30 s. The sifted ingredients were added, and mixing was continued at low speed for an additional 30 s. The batter was portioned by placing approximately 40 g into muffin tin wells, and the cakes were baked at 180°C for 20 min. After baking, the products were allowed to cool on a wire rack.
Sample preparation and analysis
For each of the three individual baking run replicates (for each of the experimental variables), representative samples were shipped in 100 ml glass sample containers, sealed with plastic lids in insulated shipping containers held at approximately 0-5°C. Samples were shipped to Eurofins Lancaster Laboratories Environmental, Lancaster, PA, USA, for the analysis of PCDD and PCDF, and to Eurofins Food Integrity & Innovation, Madison, WI, USA, for the MCPD analyses. Upon receipt of samples, they were kept at 0-6°C at both locations until sample preparation and analysis.
PCDD/PCDF
A 10 g sample was blended with sodium sulfate. Isolute® HM-N (Fisher Scientific), a cross-linked styrene-divinylbenzene copolymer that is used to help bind water and other unwanted polar materials and also helps with keeping solid materials dispersed and flowable, was added if the sample was especially moist. As a precaution, Isolute® was blended in with these samples. The spikes were added directly into the sample, which had already been transferred to the microwave tubes. Thirty ml of 1:1 methylene chloride and acetone were added to the sample which was then microwaved for about 1 h. The sample extract, from the microwave vessel, was then transferred to a mini-Kuderna/Danish concentrator through a sodium sulfate column to further dry the extract. The extract was then concentrated on a steam bath, followed by a solvent exchange to hexane. The hexane extract was pretreated by slurrying with a 1:1 mixture of PSA, (Primary and Secondary Amine exchange sorbent; Agilent Technologies, Inc.), and acid-treated silica gel. The hexane extracts were then passed through a multi-bed clean-up column consisting of acid-treated silica gel, base treated silica gel and neutral silica gel. The hexane extract was concentrated to nominal 1 ml for transfer to instrumental analysis. Prior to instrumental analysis, 10 µl of isotopically labeled internal standards in nonane were added to the hexane extract and the extract concentrated to approximately 10 µl. Another 10 µl of nonane was added to make a final volume of 20 µl. The extract was then analyzed by high resolution gas chromatography (HRGC)/high resolution mass spectrometry (HRMS). 28
3-MCPD
2-MCPD, 3-MCPD and their fatty acid esters and glycidyl fatty acid esters were extracted from samples based on three stages of Heat-Ultrasonic-Pressure-supported Solvent Extraction (HUPsSE) using methanol (HPLC grade; Fisher) and tert-butyl methyl ether (t-BME; HPLC grade; Sigma-Aldrich). After removing the solvents, the polar fraction was separated from the non-polar fraction using saturated sodium sulphate (anhydrous, ACS grade, >99%; Fisher). The free 2-MCPD and free 3-MCPD in aqueous phase were extracted into diethyl ether (anhydrous, analytical reagent grade; Fisher), whereas the 2-MCPD esters, 3-MCPD esters and glycidyl esters were extracted into a mixture of hexane (HPLC grade; Fisher) and t-BME. The free 2-MCPD and free 3-MCPD in diethyl ether fraction were derivatized with phenylboronic acid (>97%; Acros Organics) followed by evaporation of the extract and concentration in isooctane (HPLC grade; Sigma-Aldrich). The phenylboronic acid derivatives of free 2-MCPD and free 3-MCPD was determined by GC-MS/MS (GC - Agilent 7890B; MS - Agilent 7010 GC/MS Triple Quad; column - Restek RTX5-MS. 20 m length, 0.25 mm diameter, 0.25 micron film thickness). The 2-MCPD esters, 3-MCPD esters and glycidyl esters in hexane/t-BME fraction were converted into their free forms using slow alkaline-catalyzed ester cleavage at low temperature. The reaction was stopped using acidified sodium bromide (Acros Organics) solution. Under these conditions, released glycidol was reacted rapidly with inorganic bromide and converted to a stable monobromopropanediol (3-MBPD). The fatty acid methyl esters were removed from the extract with hexane. The released free 2-MCPD, free 3-MCPD and 3-MBPD were extracted with diethyl ether/ethyl acetate (HPLC grade; Sigma-Aldrich) and derivatized with phenylboronic acid prior to analysis by GC-MS/MS. 29
Spike recovery
PCDD and PCDF
An additional 8 samples (2 samples for each of the 4 products) were subjected to spike recovery analysis of PCDD/PCDF. Spike recovery was conducted as follows:
A 10 g portion of the homogenized sample was blended with Isolute® and sodium sulfate to make the mixture more granular/flowable.
The blended mixture was spiked with 100 µl of a solution containing the native PCDD/PCDF components. The target spike amount was 10 ng/kg for the tetra congeners, 100 ng/kg for penta-, hexa-, and hepta congeners, and 200 ng/kg for octa congeners.
The blended/spiked mixture was transferred to a microwave vessel, solvent was added and the vessel was microwaved for the extraction of the PCDD/PCDF constituents.
The resulting extract was further purified and analyzed by high resolution gas chromatography (HRGC)/high resolution mass spectrometry (HRMS) (EPA 1613B, October 1994).
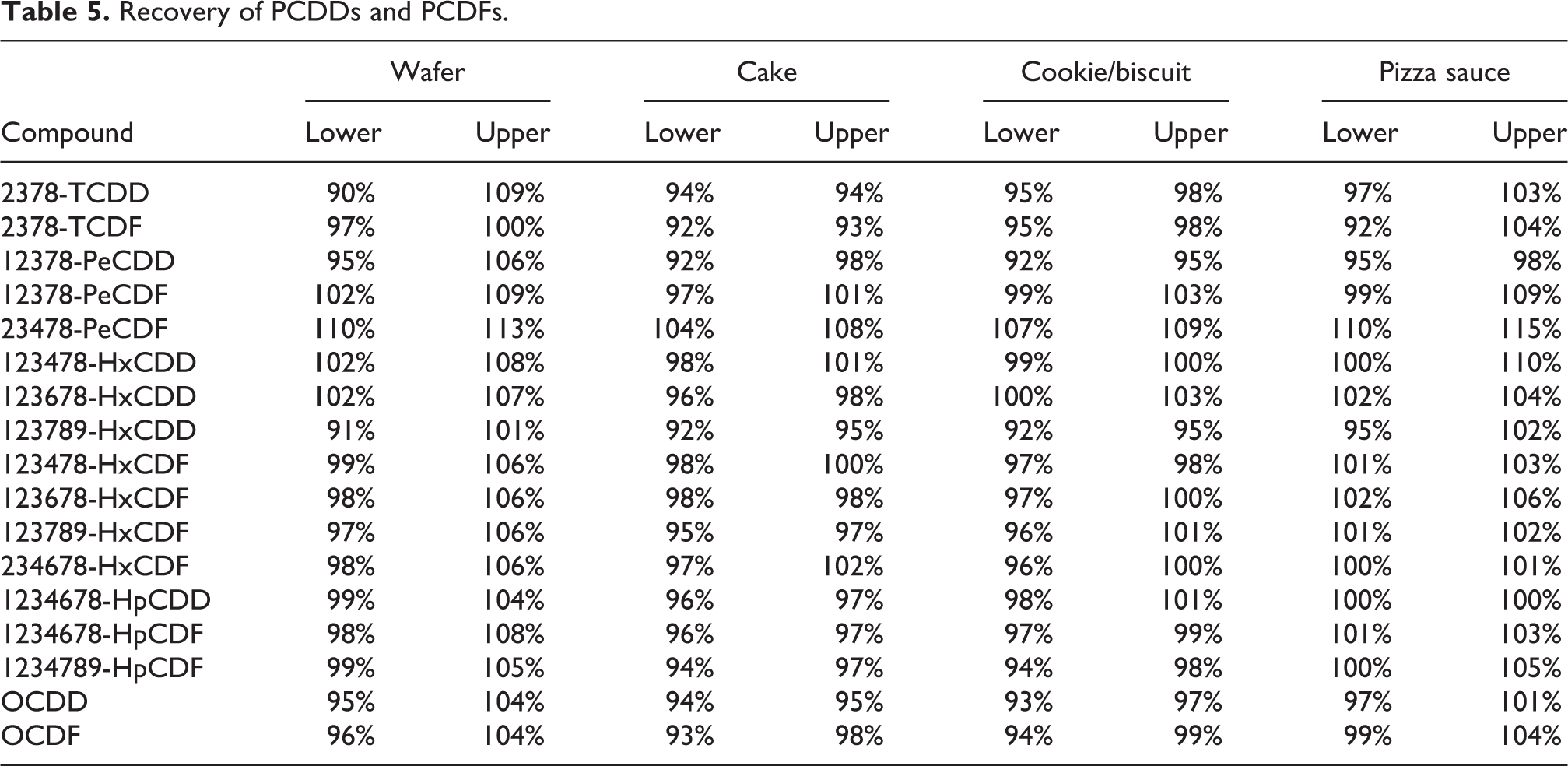
Spike recovery results for PCDD and PCDF are presented in Table 5.
Recovery of PCDDs and PCDFs.
3-MCPD
Four samples (two solid samples and two liquid samples) were used for spike recovery analysis of free and bound 3-MCPD. An appropriate amount of spiking reagent was added to the sample tube as follows: for solid samples, 100 µL of a mixed solution of 3-MCPD ester, 2-MCPD ester, and glycidol to obtain sample spiked at 0.05 µg/g of ester forms, and 200 µL of a mixed solution of free 3-MCPD and free 2-MCPD to obtain sample spiked at 0.01 µg/g of free forms; for liquid samples, 100 µL of a mixed solution of 3-MCPD ester, 2-MCPD ester, and glycidol to obtain sample spiked at 0.01 µg/g of ester forms, and 50 µL of a mixed solution of free 3-MCPD and free 2-MCPD to obtain sample spiked at 0.005 µg/g of free forms. The following standards were used: 1,2-bis-palmitoyl-3-chloropropanediol (PP-3-MCPD; Toronto Research Chemicals, B515200), 1,2-bis-palmitoyl-3-chloropropanediol-d5 (PP-3-MCPD-d5; Toronto Research Chemicals, B515202), 3-chloropropane-1,2-diol (3-MCPD; Sigma-Aldrich, 32406), and 3-chloropropane-1,2-diol-d5 (3-MCPD-d5; C/D/N Isotopes, D-1842). Samples were vortexed to mix and then analyzed using GC-MS/MS [AOCS Official Method Cd 29b-13 (2013), modified]. Spike recovery results for free and bound 3-MCPD are presented in Table 6.
Recovery of free and bound 3-MCPD.

Overall, the spike recovery data for PCDDs and PCDFs demonstrated recoveries of 90–115% for the 17 congeners of PCDD and PCDF in the four product types (Table 5). The spike recovery data for free and bound 3-MCPD demonstrated recoveries of 100–134% for free and bound 3-MCPD in solid and liquid samples (Table 6). This demonstrates that the analytical methods used were working well.
Statistics
Mean and standard deviation were calculated for each group (control, sucralose, and maltodextrin) of three samples each. For results reported as less than the method limit, the value was assumed to be the reporting limit for calculation of means. Groups within a product category were compared to each other using one-way ANOVA. P<0.05 was considered statistically significant.
Results
PCDD and PCDF
All 17 congeners of PCDD and PCDF were below the method reporting limit (Limit of Quantitation; LOQ) for all the samples from the baking study (Table 7). The LOQ for the tetra congeners was 0.2 pg/g, for penta, hexa, and hepta congeners was 1 pg/g, and for octa congeners was 2 pg/g. The total TCDD Toxic Equivalence (TEQ) LOQ was 2.28 pg/g.
Analytical results for PCDD and PCDF TEQ.
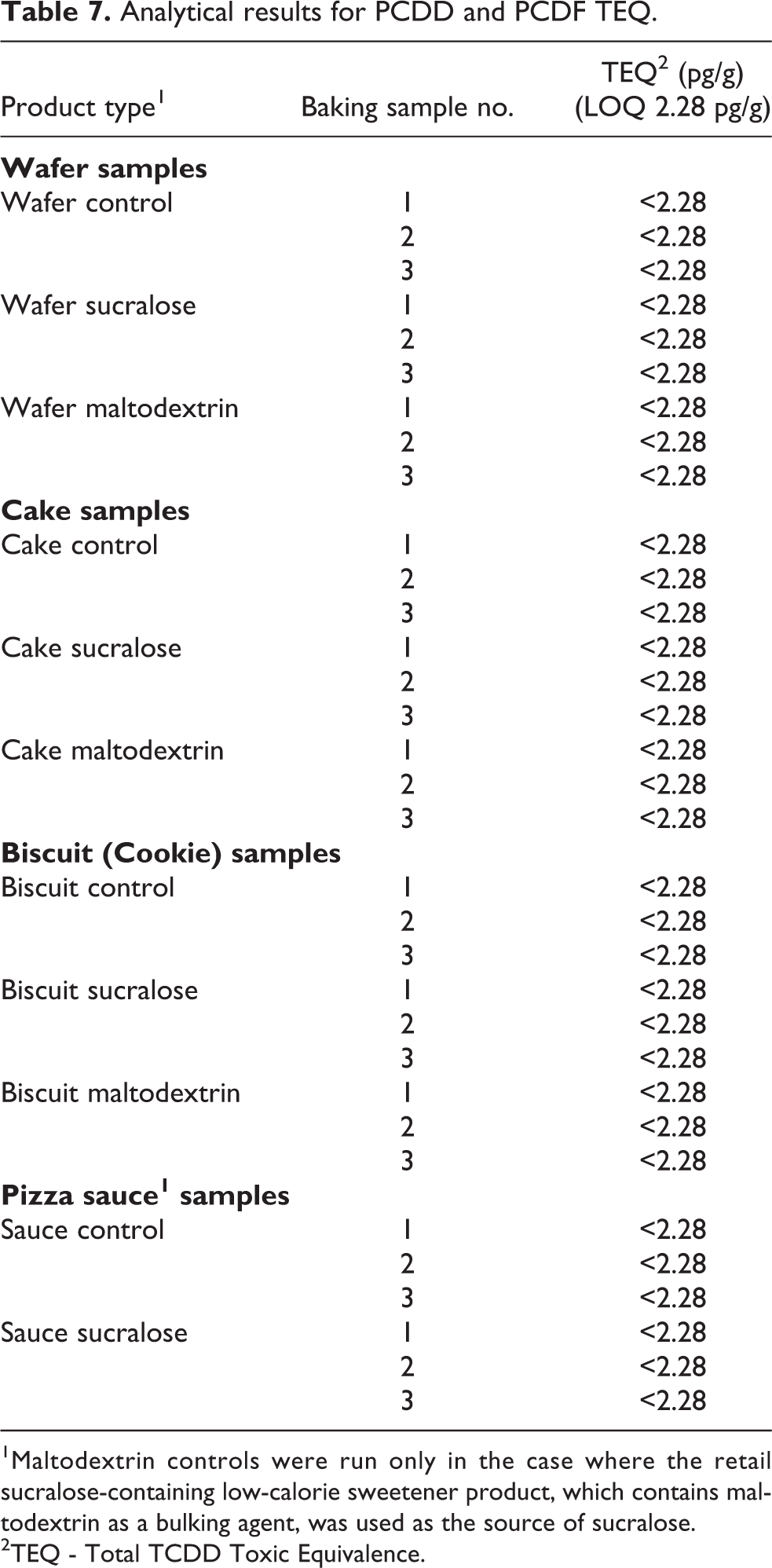
1 Maltodextrin controls were run only in the case where the retail sucralose-containing low-calorie sweetener product, which contains maltodextrin as a bulking agent, was used as the source of sucralose.
2TEQ - Total TCDD Toxic Equivalence.
3-MCPD
Wafer results
Bound 3-MCPD levels for controls, sucralose, and maltodextrin-containing wafer samples were below the LOD of 0.005 µg/g and LOQ of 0.01 µg/g (Table 8). Free 3-MCPD levels in most of the wafer samples were below the LOQ of 0.005 µg/g, excepting one control sample and one of the sucralose-containing samples. There were no statistically significant differences observed for bound or free MCPD among the wafer samples.
Analytical results for free and bound 3-MCPD from the baking study.
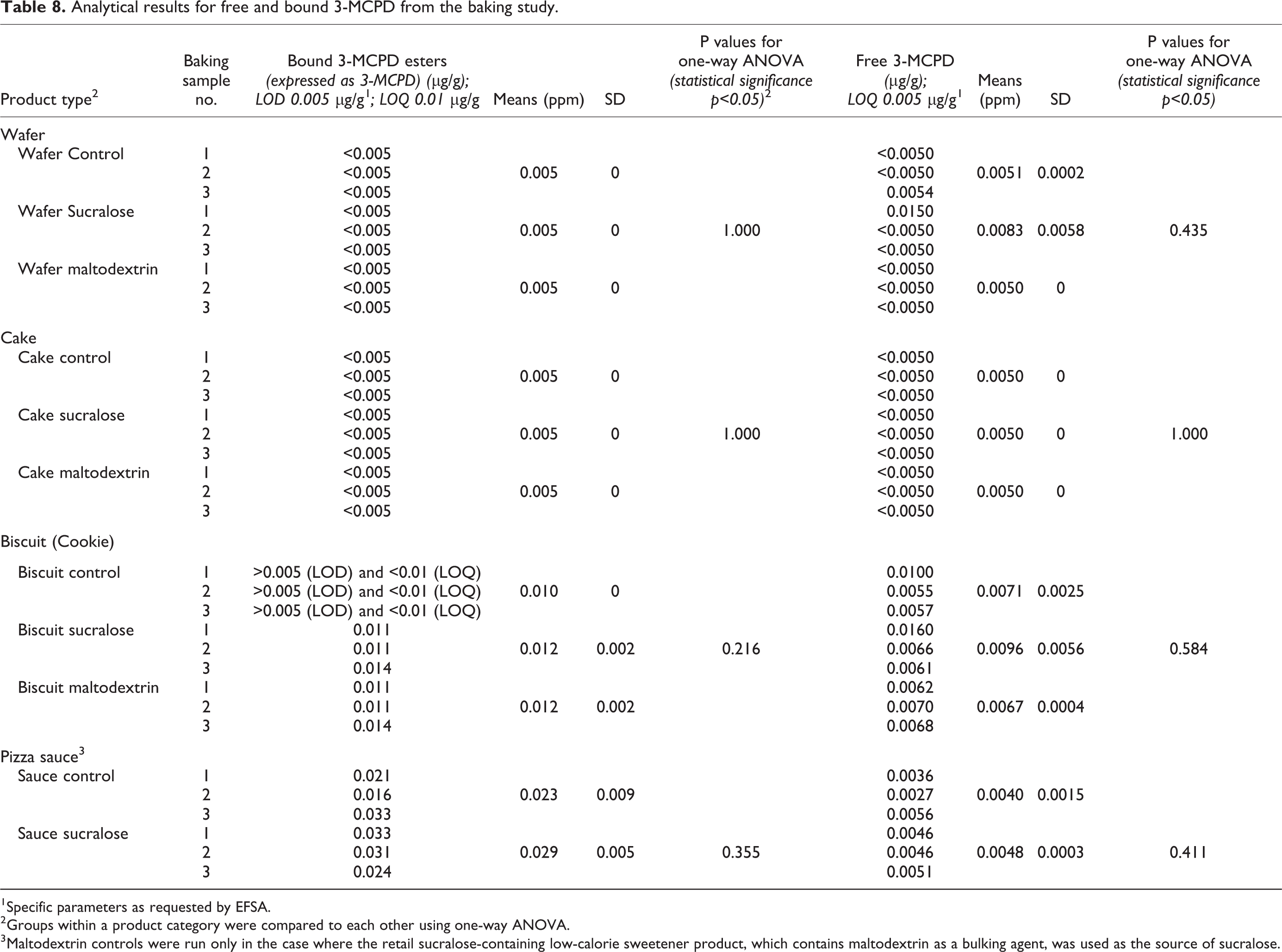
1Specific parameters as requested by EFSA.
2Groups within a product category were compared to each other using one-way ANOVA.
3Maltodextrin controls were run only in the case where the retail sucralose-containing low-calorie sweetener product, which contains maltodextrin as a bulking agent, was used as the source of sucralose.
Cake results
Bound 3-MCPD levels for controls, sucralose, and maltodextrin-containing cake samples were below the LOD of 0.005 µg/g and LOQ of 0.01 µg/g. Free 3-MCPD levels in all of the cake samples were below the LOQ of 0.005 µg/g. Hence, there were no statistically significant differences observed among the cake samples.
Biscuit (Cookie) results
Bound 3-MCPD levels for sugar-containing control samples were above the LOD of 0.005 µg/g but below the LOQ of 0.01 µg/g. For the sucralose and maltodextrin-containing samples bound MCPD levels ranged between 0.011 and 0.014 µg/g. Free 3-MCPD levels in all of the cookie samples were above the LOQ of 0.005 µg/g. For sugar-containing controls free MCPD levels ranged between 0.0055 and 0.01 µg/g, between 0.0061 and 0.016 µg/g for sucralose samples, and between 0.0062 and 0.007 µg/g for maltodextrin controls. There were no statistically significant differences observed for bound or free MCPD among the cookie samples.
Pizza sauce results
Bound and free 3-MCPD were detected in all control and sucralose-containing pizza sauce samples. There were no statistically significant differences observed for bound or free 3-MCPD among these samples.
Background
In addition to conducting the analyses requested by EFSA to investigate the potential for sucralose to lead to the formation of non-sucralose chlorinated compounds that could be harmful to human health, it is important to assess critically the basis for the BfR conclusion that sucralose use in foods subjected to heat during their manufacture may result in the formation of non-sucralose chlorinated compounds that could be harmful to health.
This BfR conclusion was first reported in a review 10 of publications that BfR found through a literature search and assigned a “Klimisch” Category rating. This review was followed by a publication in Food Chemistry, 11 providing similar information.
The Klimisch Category rating system is a published scoring system designed to help with assessing data reliability. 30 The highest Klimisch category rating is 1, to be used when data are considered “reliable with no restrictions.” Next is 2 denoting data considered as “reliable with restrictions.” A 3 or 4 rating is for, respectively, data considered as “not reliable” and “not assignable.”
In forming its conclusions, the BfR determined to consider only publications to which it had assigned a Klimisch Category level of 1 or 2, i.e. publications BfR considered to be “reliable,” with or without “restriction.” The BfR rated no publications found as Category 1 and nine as Category 2, so these nine publications were evaluated in the BfR review. Six publications were found that that BfR assigned a Klimisch Category rating of either 3 or 4, and the BfR did not rely upon these in forming its opinions on the safety of sucralose when used in foods exposed to heat. 7 The 3 and 4 Klimisch Category ratings were assigned generally on the basis that BfR found the publication to present insufficient details on methodology and/or findings and/or the publication represented “secondary literature” (i.e., a book chapter).
The below critically analyzes both the studies that were the basis for the BfR opinion on sucralose (BfR assigned Klimisch Category level of 2), and the merits of reaching conclusions regarding sucralose safety without consideration of other information, including publications dismissed by BfR in evaluating available information.
Critical analysis of studies that were the basis for the concern raised by BfR
A critical analysis of these studies shows that none are reliable for assessing the safety of sucralose use in foods that are subjected to heat during manufacturing. They are studies that imposed conditions that would not be found in expected food manufacturing processes and, conversely, could be expected to affect reaction kinetics. It is well known that solute concentrations and environmental conditions can affect reaction kinetics. In many of the studies relied upon by BfR in its assessment of the potential fate of sucralose in food, the concentration of sucralose employed was extremely high, either in the absence of any other ingredient, or at high molar concentration and massively in excess of concentrations that could be expected to be found in food matrices. In addition, the testing environment commonly excluded the presence of water. Previous studies have reported already that neat (dry) sucralose can, with sufficient exposure to heat, eventually lead to chloride ion release by reaction with ambient hydrogen, to form hydrogen chloride, 31,32 which can further catalyze chloride release. In general, most of the studies cited by the BfR as evidence of concern for sucralose stability in food found this type of chloride release. What is not taken into account by the BfR review, however, is that sucralose is a highly water soluble molecule 33 that will preferentially partition to the water phase in food matrices, 6 which changes the dynamics for chemical reactions. Importantly, an aqueous matrix limits the heat level to which sucralose could be exposed during food manufacturing conditions. Indeed, loss of chloride from sucralose is not seen when sucralose is present at low levels in food. 5 The majority of primary reaction sites on the sucralose molecule are also occupied, 5 reducing its ability to interact with other substances. This helps to explain its general resistance to chemical interactions and/or breakdown in foods. Scientific publications are available that support this conclusion: Sucralose has been shown to be remarkably stable to chemical interactions with a wide range of types of substances that could be expected to be found in foods, (e.g., bases, oxidizing and reducing agents, aldehydes, ketones and metallic salt) and to pH levels normally found in food, including high acidity foods, such as carbonated soft drinks. 5 In a baking study, that investigated the stability of sucralose in a range of baked goods (cookies, cake, graham crackers), no loss of sucralose was detected. 7 The latter study used radiolabeled sucralose and extracts of radiolabeled material were analyzed by thin-layer chromatography, using two solvent systems independently as eluents. This showed only material eluting at a time consistent with sucralose standard. Sucralose has also been found to be 100% stable in a range of foods subjected to heat-pasteurization, e.g. ketchup, canned fruits, dairy products and still beverages. 3
What the studies ranked by BfR as Category 2 provide evidence of, is that, consistent with other research, release of free chloride in the presence of fats or fatty acids will result in the formation of such compounds as PCDD/Fs and/or 3-MCP and its congeners. Indeed, it is the availability of chloride ion that is key to the formation of chloropropanols. In a review of 3-MCPD formation from soy sauce production, for example, it was noted that removal of chloride ions in acid hydrolysis would eliminate the formation of 3-MCPD. 34 Also important to note is that the formation of chloropropanols in foods is not unique to foods containing sucralose. They are found in a wide range of foods, including, for example, baked goods (e.g., bread, cookies, crackers), cooked meats (e.g., hamburgers, sausages, bacon), battered fish filets, cheeses, malted beverages (beer and others), roasted coffee, soups, cereals, oils and infant formula, and more 14 –18 Their formation occurs naturally in “fat- and salt-containing foods processed at high temperatures,” 16 via the interaction of free chlorine (chloride), which is readily available from salt, and glycerol (glycerine), which is readily available from fatty acids.
Further, even if sucralose could be a source of free chloride under normal food manufacturing conditions, the thermodynamics of which are exceedingly unsupportive, to protest the use of sucralose based on a potential for it to add chloride to food, is akin to protesting that salt cannot be used in foods. This is particularly true when sucralose use levels in foods are exceedingly low due to its high sweetening potency. On average, sucralose is about 600× sweeter than sugar. 35
Finally, it must be taken into consideration that the extreme and rapid heating applied to sucralose in many of the studies discussed below, if applied in the manufacture of a typical food containing sucralose, would also be likely to induce pyrolysis (burning) of other ingredients, such as the carbohydrate in flour, which thus would seriously impact the likelihood of human exposure due to unacceptable product quality.
The below summarizes the conduct and findings of research studies that were the basis for the BfR questions regarding sucralose use in food, which illustrate the non-applicability of these studies to food safety assessment.
Hutchinson 36 investigated the thermal stability of aqueous sucralose, and other sweeteners, at varying pH and temperatures, which was reported in a doctoral thesis. 36 In this non-peer-reviewed series of studies, the stability of a 0.1 M (3.97%) solution of sucralose (sucralose purity unspecified; 3.97 g in 100 mL water), or of a 0.1 M solution of sucralose and 0.1 M glycine, was evaluated after 1 h at temperatures of 100°C, 140°C, and 180°C at different pH levels (3, 7, and 11). The solutions were placed in stainless steel Whitey reaction cylinders, sealed, and were heated in an oil bath. Heating in such a sealed system would lead to higher temperatures that would not be expected with usual heating of sucralose in an aqueous environment. Such investigative methodology, while useful for thermal stability research, does not represent standard cooking procedures, from either a household or industrial scale perspective. The concentrations of sucralose used by Hutchinson represents about a 2% sucralose concentration, whereas sucralose concentrations in food matrices is far (at least 100×) less. Hutchinson also noted that the inclusion of certain ions in the buffered solutions used in the testing may have interacted with sucralose hydrolysis, thus complicating the interpretation of the effect of temperature and pH on sucralose stability, including findings of chloride loss.
Bannach et al. 37 investigated the effect of rapid heating of sucralose by simultaneous thermogravimetry and differential thermal analysis (TG-DTA). Neat, dry sucralose (two suppliers; one with 98% purity, the other with purity undefined) was applied to the metal sample plate of the thermal analysis system and introduced to a nitrogen-purged furnace sample tube, that was enveloped by a micro-furnace, which increased the heat of the sample tube at a rate of ∼10°C/min up to 500°C. Thermal decomposition of sucralose was noted to occur in three separate phases, the first decomposition change being detected at about 120°C and consistent with loss of chlorine, which was empirically substantiated by detection of hydrogen chloride.
This study is then, effectively, a study of the isolated burning of neat sucralose.
de Oliveira et al., 38 using thermogravimetric apparatus, similarly assessed rapid heating (10°C/min) of neat, dry technical-grade sucralose (two suppliers; purity not specified). The authors reported the fate of sucralose pyrolysis based on differential scanning calorimetry and thermogravimetric analysis coupled with infrared spectroscopy (DSC/TGA/IR), hot-stage microscopy (HSM), and high resolution mass spectrometry (HRMS).
It is worth noting that, in these studies, as with the Bannach et al. 37 studies, mass loss, consistent with chloride loss and HCl formation, was begun to be detected at about 125°C, and images of sucralose evaluated by HSM showed that the dry sucralose crystals underwent a fusion process followed by decomposition. If sucralose in food matrices were to so decompose with food manufacturing processes, including heat, it would be unlikely that it would be suitable for achieving sweetness.
Finally, HRMS analyses of the gaseous phase following heating of sucralose (in a closed vial) to 98°C in a water bath, was reported to reveal the presence of specific polychlorinated aromatic hydrocarbons. The finding of polychlorinated aromatics in the heated headspace at low ppm concentrations seems inconsistent with the relatively mild conditions involved (98°C for 15 min). Formation of these substances is not known to occur, generally, at such a low temperature, 39 and this temperature is lower than that reported to be associated with the thermal decomposition of sucralose. 31 This could be the result of contamination or some other source of chlorine other than sucralose. The use of a closed vial when heating the samples may also have caused pressure changes that may have affected internal temperature and/or chemical reaction potential, and as such are not clear as having relevance to sucralose stability in food preparation.
Rahn and Yaylayan 31 investigated sucralose thermal decomposition and the potential of this to lead to formation of other chlorinated substances, in particular, chloropropanols. In these investigations, sucralose alone or in a 1:2 M ratio with glycerine was inserted into a pyrolysis (250°C-heated) chamber for 20 s. Rahn and Yaylayan also report pyrolysis of sucrose (common table sugar) under similar conditions. With pyrolysis, there can be oxidation of other elements, like sulfur and chlorine. 40 –42 The investigators showed that the pyrolysis of pure sucralose resulted in a degradation pathway with levoglucosenone as a major product, which is also a product found with the pyrolysis of sucrose and other saccharide molecules such as β-d-glucopyranose, cellobiose and cellulose. 43,44 The formation of levoglucosenone is not surprising as sucralose is made from sucrose with selective chlorination at three of the primary hydroxyl groups, and release of chloride through oxidation would enable this type of degradation product. Levoglucosenone formation is preceded by, in the case of both sucrose and sucralose, cleavage at the glycosidic linkage that connects the inherent monosaccharides or, in the case of sucralose, chlorinated monosaccharide moieties. Indeed, Rahn and Yaylayan noted that “being [a] sucrose analog, one anticipates [sucralose] degrading in a similar fashion.” The investigators also showed that subsequent degradation of the two monosaccharide derivatives from sucralose is related to chlorine loss. The finding of chlorine loss under these particular experimental conditions is again consistent with the previously discussed thermal degradation studies.
The pyrolysis studies of a 1:2 molar mixture of sucralose and glycerine, whether in the presence of air or moisture, also showed some formation (in addition to formation of the main degradation product, levoglucosenone) of 3-monochloropropanediol (3-MPCD) and 1,2- and 1,3-dichloropropanols (1,2- and 1,3-DCP, respectively), based on chromatographic analyses. The findings are consistent with those of Rahn and Yaylayan, 31 which exposed a 1:2 molar mixture of sucralose and glycerol to extreme and rapidly-applied heat, would yield chloropropanols. While chloropropanol formation resulted, as discussed above, this can be expected when conditions force free chloride release in the presence of glycerol, but such molar concentrations and experimental conditions are not present in the production of food products.
Wu et al. 45 investigated the potential for formation of various polychlorinated dibenzo-p-dioxin and dibenzofuran congeners (PCDD/Fs) by heating sucralose or 1,3-dichloropropan-2-ol (1,3-DCP) combined with oil (soybean oil) or oil + minced beef (∼1/3 ratio, w/w). The sucralose and 1,3-DCP concentration in the heated oil was 22% and 19%, respectively. The sucralose and 1,3-DCP w/w concentrations in the beef/oil mixtures ranged from ∼0.005 to 5% and ∼0.00005 to 5%, respectively. The oil mixtures were heated to ∼250°C and maintained at this temperature for another 10 min. The beef/oil +/- sucralose or 1,3-DCP mixtures were heated using the same apparatus used for heating the oil, and after 15 min the mixture temperatures were ∼145°C. All control and text mixtures were then allowed to cool for 10 min. The temperature of all the beef/oil mixtures rose slightly, to about 165°C, by the end of this period. The air above the heating pans (head space) was also trapped during the entire heating and cooling phases for subsequent analysis.
Analysis of the fluid phases (oil, oil mixtures or residual fluids after heating beef/oil +/- sucralose or 1,3-DCP) after heating showed that the concentrations of PCDD/Fs of the oils with or without sucralose or 1,3-DCP, as well as the beef/oil mixtures, were all “rather low, and most of the target PCDD/F congeners were not detected.” Analysis of the head space samples and solid phases from the beef-containing mixtures showed the presence of various PCDD/F congeners, however, the contribution was predominantly from the gas, vs. the solid phase, i.e. predominantly not in the food matrix. Thus, this study does not represent conditions of food manufacture, where sucralose would be dissolved in the water phase of a food matrix. Further, as noted by the investigators, heating the oil used in their study could produce hydrocarbons. The PCDD/F levels found with inclusion of either sucralose or 1,3-DCP, both of which have been shown to be able to be dechlorinated with high heat and in low/zero-moisture conditions, 31,46 likely stems from free chloride interacting with the generated hydrocarbons. As noted previously, however, any possible chloride release would not be likely to meaningfully increase exposure to PCDD/Fs beyond what would already be possible with high heat application to foods containing salt and fat.
Dong et al. 47 conducted studies that were very similar to those conducted by Wu 45 and, indeed, were done in the same lab. These studies investigated the potential for formation of particular polychlorinated biphenyls (PCBs) when sucralose or 1,3-DCP was added to either minced beef or minced beef in soybean oil (3:1 w/w ratio). The w/w concentration of sucralose or DCP in the beef/oil mixtures was ∼0.5 – 5% and 0.005 – 5%, respectively. The mixtures were added to a stainless steel bowl kept at 400°C and heated for 15 min, and then allowed to cool for 10 min. The temperatures of the raw minced beef or minced beef mixtures was ∼160°C at the end of the heating time.
The research found that the concentrations of PCBs in control beef declined by more than 50% after cooking with or without the presence of oil. This also occurred when either sucralose or 1,3-DCP was added to the beef/oil mixtures, except at the highest 1,3-DCP concentration, which was still a PCB concentration not greater than the level found in uncooked beef. As in the Wu 45 study, PCB congener levels were increased in the oil fumes produced after cooking. The authors said that results of their study suggest that sucralose or 1,3-DCP could act as chlorine sources, and so promote formation of PCBs in oil fumes. While it is unknown whether the formation of PCBs by reaction of chlorine with hydrocarbons occurs within the beef, the decline in PCBs in the cooked beef is evidence that any such putatively formed PCBs are subsequently volatilized and, thus, do not add to the innate meat PCB content. Nonetheless, it is more probable that the heating of the oil, while either sucralose or 1,3-DCP was present, led to free chloride, by dechlorination, and free chloride then combined with hydrocarbons from the oil, to result in PCB congeners that were then volatilized to the head space. It is also noteworthy that, compared with the total PCB content of raw beef (∼75 pg/g), the content of PCBs in oil fumes captured after cooking the beef/oil/sucralose mixtures was comparatively low (<3 pg/g). In addition, while the investigators report an increasing concentration of PCBs with increasing sucralose concentration, the maximum increase was within one order of magnitude, while the range of sucralose concentrations represented a 1000-fold increase.
Dong et al. 48 later investigated the effect of heating oil (peanut or olive) with or without either sucralose or sodium chloride, using the same type of cooking apparatus and general methodology, and analyzing for the presence of polychlorinated napthalenes (PCNs). The authors noted that due to the fact that sucralose is hydrophilic, the sucralose/oil mixtures were mixed with a glass rod to affect a sucralose suspension as homogeneous as possible. The w/w concentration of sucralose or sodium chloride in the oils was 10%. As noted by the investigators, this is an unrealistic use of sucralose in food preparation. The mixtures were heated to ∼ 200 or 245°C. The investigators also reported heating neat sucralose under the same conditions as the control or test oil mixtures and similarly analyzed for the presence of PCNs.
No PCNs were detected, in heated neat sucralose, or any oil mixture or collected head space (off-gassed fumes following heating) when heated to ∼200°C. At 245°C, no PCNs were found in neat sucralose or the oil mixtures containing either salt or sucralose. PCNs were detected in the off-gassed oil fumes when the sucralose/oil mixture was heated to ∼245°C, but not with heating either neat sucralose or oil mixtures with sodium chloride. The investigators note that this suggests that the PCNs detected are a consequence of free chloride, released from sucralose as a result of the cooking in oil, interacting with carbon sources from the cooked oil, at this temperature. They further note that the PCNs detected could be the result of [higher] temperatures achieved in the oil mixtures in the field close to the thermal heating plate, leading to PCNs being evaporated into the oil fumes. As with the previous studies from this lab, the experimental conditions utilized do not represent conditions that would be encountered in normal food production.
Dong et al. 39 reported a further investigation generally using the methodology described by Wu, 45 where neat sucralose (5 g) was heated to 350°C or 400°C in utensils made of different materials and in the presence or absence of particular “rusts,” including Al2O3, Fe2O3, and CuO, and analyzed for the presence of PCDD/Fs. Heating sucralose up to 400°C in “cooking utensils” made of quartz, aluminium, or copper, and polished to be free of rust, failed to generate detectable amounts of PCDD/Fs. Whereas, when sucralose was heated to 350 or 400°C in a stainless steel vessel, PCDD/Fs were detected primarily in the resulting smoke. It should be noted that other research has shown that certain metal ions, and particularly Fe3+ and Fe2+, enhance the formation of 3-MCPD when vegetable oil is heated. 49 With heating to 400°C and addition of 1 g of either Al2O3, Fe2O3, or CuO, there were increases in the levels of PCDD/Fs detected in the smoke. No significant amounts of PCDD/Fs were detected when sucralose was heated in the presence of the metal oxides at 300°C. In general, these studies are effectively pyrolysis studies, which do not represent conditions during normal food manufacture. The addition of pure rusts at high w/w concentration, which could act as catalysts for other reactions in the thermal degradation of sucralose, is also unrealistic for food manufacturing conditions. Further, the amount of dry sucralose that was heated has a sweetness equivalent to ∼3000 g sugar – hence, a concentration that has no relevance to normal use of sucralose in foods.
Dong et al. 50 conducted one additional study that had a design and methodology very similar to the previously described study, 39 this time looking for the presence of PCNs. Dry, neat sucralose (5 g) was again heated by itself to high temperatures (350 and 400°C) and PCNs were found primarily in the resulting smoke. The patterns of PCNs detected were found to be “consistent with the patterns of PCNs unintentionally produced from waste incineration and metal smelting.” The addition of rusts in the heating process demonstrated, effectively, that heated metal oxides can catalyze PCN formation processes when dry sucralose is heated at such high temperatures. The authors also noted that CuO, the added oxide resulting in the largest PCN increase, can react with HCl to produce Cl2, and “Cu (II) would be expected to promote formation of biphenyl structures, while Cl2 can, of course, promote chlorination of aromatic compounds.” The investigators also detected no polycyclic aromatic hydrocarbons (PAHs) in sucralose prior to heating, although they were found in the smoke after heating sucralose at the high temperatures utilized. This indicates that the heating induced effective burning of sucralose, such that aromatic hydrocarbons were made available from the breaking of the disaccharide basis of sucralose, and, along with release of free chloride. The released free chloride and available hydrocarbons can then lead to generation of PCNs.
Consideration of the scope of research, and application of Klimisch Category ratings, in the BfR reviews 10,11
The Klimisch category ratings were originally created for evaluating the quality of experimental toxicological and ecotoxicological data and in comparison to already established standards for the conduct of such studies 30 relating to preferably standardized methodology. 51 While its use has recently been extended to other types of studies, the application of this rating system to exploratory chemistry studies on ingredient stability under variable conditions is questionable. Importantly, use of the ratings infers that all studies conducted should be available, in full detail, in the published literature, and those that don’t have full details should not have weight in scientific consideration of stability evidence. There is, however, no regulatory requirement for publication of stability studies of food ingredients and there is also no historical precedent set by food ingredient research publications that should lead to an expectation for such detailed reports. In general, very detailed stability studies of any food ingredient are commonly not found in the published literature. In addition, Klimisch Category ratings of 3 and 4 do not have to be excluded in a weight of evidence approach, as they may still convey important information. 52 Despite the BfR ratings applied to publications found, appropriate stability studies are required by regulatory agencies in the assessment of any proposed new food ingredient 2,53 and, in the case of sucralose, key studies have been described in various publications, 7,22,54 –57 including, but not limited to, ones found by BfR with their literature search. 5,58 A summary of these studies is shown in Table 9.
* This is not intended to be a comprehensive list of studies that were reviewed by regulatory agencies, which can include studies beyond those reported in the published literature.
While the publications that provide this information were largely publications that summarize sucralose stability research (the primary reason BfR assigned a Klimisch Category rating of 3 or 4 to studies it found in its literature search), they provide a good picture of the types of research that was submitted to regulatory agencies for their full and complete assessment.
These publications also provide information on other research, some of which would have been outside the scope of the BfR literature search, that helps food scientists to better comprehend how sucralose might behave in food systems. This includes, for example, effects of concentration, stability in dry or low-moisture conditions, food-chemical interaction studies, and physicochemical characteristics of sucralose. 5,6,54,58
Notably, this research makes clear that sucralose is freely soluble in water (up to 28.2 g/100 ml at 20°C) and has poor lipid solubility (insoluble in corn oil <0.1g/100 g at 20°C), 33 and will always partition with the aqueous portion of a food system comprising both lipid and aqueous phases. 6 Because the presence of surrounding water limits the level of heat in the food matrix, this information must be taken into account when considering the meaningfulness of certain types of research studies, e.g. the studies cited by BfR in raising their concern about the safety of foods made with sucralose.
Discussion and conclusion
The current paper extends the evidence that sucralose is suitable and safe for its intended uses as permitted by regulatory authorities. The new analytical studies covering a range of foods heated during their manufacture, and analyzing each food type for 17 congeners of PCDDs and PCDFs and for free and bound 3-MCPD, shows no evidence for the formation of these substances as a result of sucralose presence in the food.
The types of foods studied were those recommended for evaluation by EFSA. They include low- and higher-moisture baked goods, including wafers, biscuits (cookies) and cakes, and pizza made with a sucralose-containing pizza sauce. EFSA also requested that a table-top sucralose-based sweetener be used in the preparation of foods like cakes and cookies to represent common household uses of sucralose. Production of each of these types of foods involves exposure of the food to relatively high temperatures. Pizza sauce also represents a relatively low pH food vs. baked goods, a variable that could influence stability of some ingredients, and also represents the category, generally, of sauces that are applied topically to foods.
The strength of the conclusion that sucralose does not lead to the formation of PCDD/Fs or 3-MCP is supported by evaluation of a variety of food products with typical to high sucralose concentrations and prepared under typical manufacturing conditions, which include high heat for different periods of time, and the analytical methodology which allows for very low detection and quantification limits of multiple congeners of dioxins, furans and chloropropanols. Further, the analytical methods used for the determination of PCDDs, PCDFs, and MCPD are validated and accepted by scientific and regulatory bodies. 28,29 While a weakness could be the possibility of PCDD/F formation below the detection limit for the assays conducted, the physicochemical characteristics of sucralose and early stability studies do not support that sucralose would be able to lead to formation of chloropropanols. This is made clear by the critical analysis of the BfR review, 10,11 which shows that the studies cited by BfR as cause for concern are not reliable for predicting the fate of sucralose in food manufacture. Sucralose is a hydrophilic substance and will be dissolve into the aqueous phase of food matrices. Because of its high sweetening potency, only minute quantities would also be present in these matrices. In general, the studies that were the basis for the questions raised in the BfR review, were studies that created environments resulting in scenarios that differed radically from those in which sucralose is used in normal food manufacture, including during cooking and baking. In many of the studies, sucralose was mixed with oil. Heating sucralose in oil, in which it would very likely not be dissolved, would mean it would most likely be present as a moisture-free dispersion. Such conditions with high heat application could be expected to actually promote pyrolysis, vs. use of sucralose in food matrices where it would be dissolved in available water. Other studies were extremely rapid, high-heating pyrolysis studies of neat, dry sucralose, and/or studies of very high concentrations of sucralose with only one or two other ingredients present and with little to no water present, or studies with other conditions (addition of rust at high w/w concentrations vs. food matrices) that could seriously affect reaction dynamics and kinetics. Moreover, the BfR review did not consider, in arriving at their concern about sucralose use in foods that are heated, other publications that support that sucralose is stable under various types of thermal processing. This includes studies that investigated the fate of sucralose when used in baking (cookies, cakes [higher-moisture baked goods] and graham crackers [low-moisture baked goods]), 7 and in foods subjected to pasteurization, sterilization or UHT processing. 5 In some of these studies, radiolabeled sucralose was also used, either 14C or 36Cl. These uses indicate no formation of any kind of PCDD/F or 3-MCPD nor loss of chlorine from sucralose when subjected to high heat in food production. In contrast, the finding of PCDD/Fs and/or 3-MCPDs in the thermal degradation, non-food-prototype studies cited by BfR, 10 almost certainly reflects a loss of chlorine from sucralose. A more recent study reported by Moldoveanu 32 also confirms the loss of chloride from sucralose under conditions of pyrolysis and notes that, “The elimination of HCl from the molecule is, in many respects, similar to the elimination of H2O from simple sugar molecules [undergoing pyrolysis].” It also notes that sucralose pyrolysis leads to “compounds similar to those generated from other sugars.” Rahn and Yaylayan 31 also show that sucralose degrades in a manner similar to sucrose (common table sugar) under conditions of pyrolysis, except with the loss of chloride (Figure 1). However, the conditions used in these studies, with little to no moisture present and extraordinarily high sucralose concentrations relative to what would ever be found in foods, cannot be expected to predict the fate of sucralose in normal thermal processing of food. Indeed, were sucralose to undergo pyrolysis (effectively, rapid burning), one could also expect significant degradation of other ingredients present, including starches commonly found in baked goods, and thus affect product acceptability.
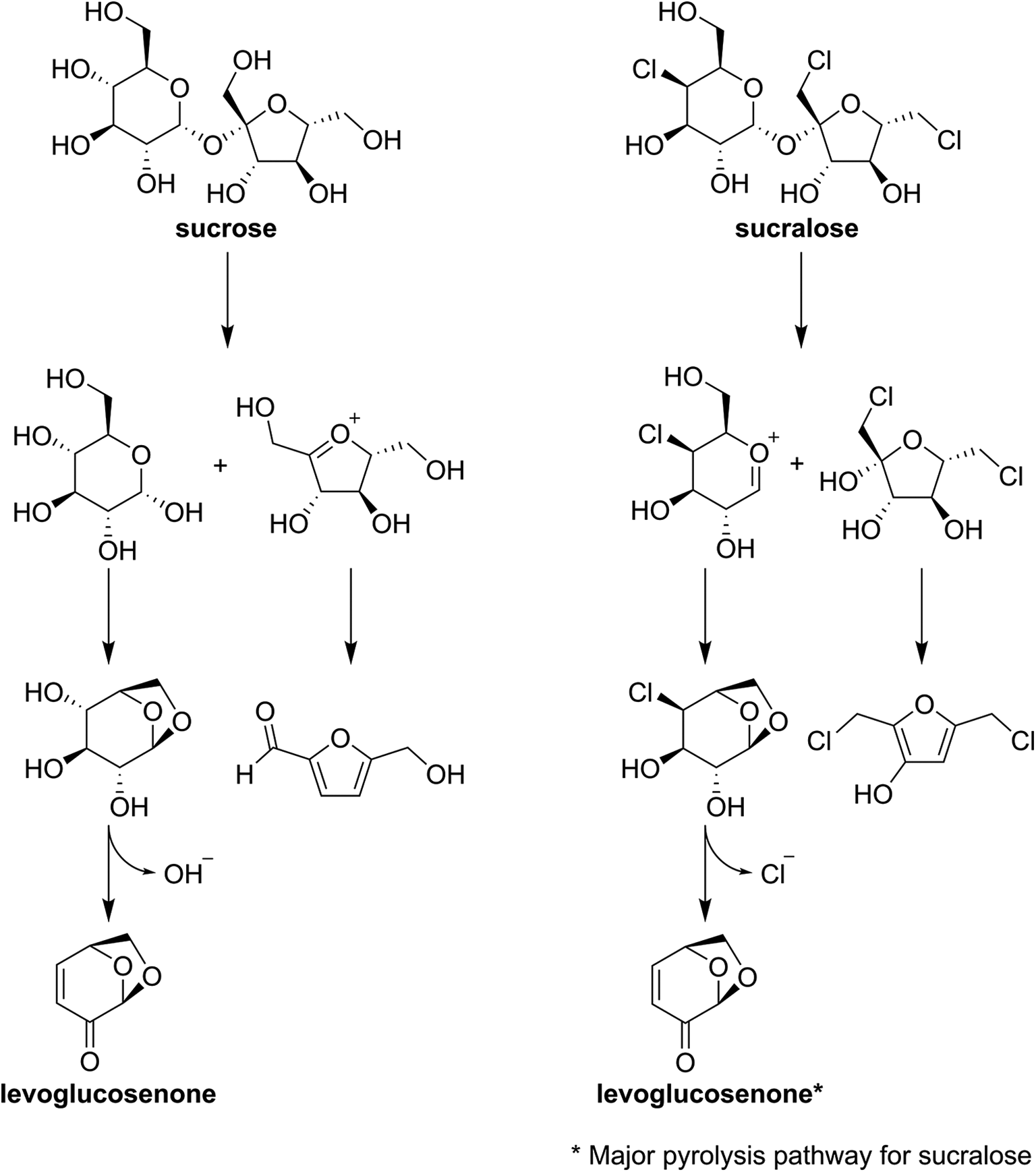
Comparison of the degradation pathways of sucrose and sucralose under pyrolytic conditions.
Even if sucralose were to be degraded to the point that it would give up free chloride that could be free to participate in the formation of PCDD/Fs or 3-MCPDs, it is highly improbable that this could result in any meaningful contribution to existing intake of chloropropanols. As discussed earlier, they are commonly found in a wide range of foods resulting from the interaction of free chloride in food and glycerol, which is released from fats when heated. Glycerol is also sometimes used as a food ingredient, which can, itself, contain chlorinated impurities such as 3-MCPD. 59 Again, this is likely a result of glycerol interaction with free chloride. Indeed, EFSA has set a limit of 0.03% free chloride in glycerol to help limit the possible formation of chlorinated compounds that could potentially pose a carcinogenic risk. 59 Free chloride also be present without the addition of table salt, as it is commonly found in the water supply and can be present in food ingredients like eggs. Indeed, the use of water and/or salt in the food products developed for the new analyses reported herein may explain why a very low level of 3-MCPD was detected in all of these products, including control products with no sucralose. The amounts detected were in the range reported elsewhere for baked goods 14,15,60 and were 40-50 times less than the maximum level permitted in oils typically used for either commercial or home food production, per a recent (2020) EU regulation. 19
Numerous studies show also that the temperature for sucralose thermal degradation increases by diluting it with a carrier, which can be anything from the application of a bulking agent used in the manufacture of retail dry sucralose-based low-calorie sweetener products, to ingredients typically found in the preparation of sweet foods, importantly including water. 58,61 Such information was not considered in the BfR deliberations on sucralose safety.
In sum, none of the studies reviewed by BfR provide any empirical evidence that sucralose could cause the formation of harmful chemical compounds when used under conditions of normal food processing, including thermal food processing. Further, the new analytical studies reported herein extend the evidence that sucralose is suitable and safe for its intended uses, including uses in cooked and baked foods, as permitted by regulatory authorities.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The analytical work reported in this publication was funded solely by Tate & Lyle Ingredients Americas LLC. All PCDDs/PCDFs and 3-MCPD analyses were conducted under contract by Eurofins Lancaster Laboratories Environmental (Lancaster, PA, USA) and Eurofins Food Integrity & Innovation (Madison, WI, USA), respectively. The manuscript preparation was funded by Tate & Lyle Ingredients Americas LLC (authors J. Gujral, J. Carr, C. Darwen, and D. Tonucci), and Heartland Food Products Group (author V. Lee Grotz, consultant to Heartland Food Products Group).

