Abstract
Clozapine (CLZ) is considered the most effective drug in treatment of resistant schizophrenia. However, its cardiotoxic effect has raised concerns about its safety. Captopril is a well-known angiotensin-converting enzyme inhibitor with unique antioxidant properties. The aim of this study was to investigate the protective effect of captopril against CLZ-induced myocarditis, and since both drugs have hematotoxic effects, this study aimed to clarify the effect of their combined use on the bone marrow. The study was conducted for 4 weeks on 50 adult male albino rats divided into five groups: group I (negative control), group II (positive control), group III treated with captopril 5 mg/kg/day, group IV treated with CLZ 25 mg/kg/day, and group V treated with captopril (5 mg/kg) 1 hour before CLZ (25 mg/kg/day). CLZ group showed a significant increase in serum troponin I, marked histopathological changes, and immunohistochemical staining of DNA degradation product 8-hydroxy-2-deoxy guanosine (8-OHdG). It significantly increased malondialdehyde level and decreased glutathione peroxidase. Captopril coadministration decreased the histopathological hallmarks and biochemical marker of myocarditis and attenuated CLZ effects on the oxidative stress parameters and 8-OHdG, suggesting its protective action against CLZ-induced myocarditis. Complete blood count and bone marrow evaluation was normal indicating that captopril, in the protective dose given, didn’t increase the risk of CLZ-induced hematotoxicity
Keywords
Introduction
Schizophrenia is one of the most severe, debilitating, and persistent of all psychiatric disorders. 1 It starts mostly at a young age (15–35 years), lasts long, leads to permanent disability, and has very high mortality rates. 2 As such, schizophrenia is a major public health problem with an urgent need to develop strategies to improve treatment effectiveness. 3
Clozapine (CLZ), a tricyclic dibenzodiazepine derivative, is an atypical antipsychotic and a broad-spectrum antagonist drug whose profile is quite different from other antipsychotics. It is distinguished from typical antipsychotics by its greater efficacy and reduced tendency to cause extrapyramidal movement disorders. 4
CLZ is considered the gold standard antipsychotic due to its demonstrated superior efficacy in the treatment of refractory schizophrenia. 5 It is estimated that about 30%–60% of schizophrenia patients do not respond adequately to antipsychotic treatment and are known as refractory schizophrenic patients. 6,7
To date, in terms of evidences, CLZ is the acknowledged treatment of choice for patients with severe treatment refractory schizophrenia. 8 Moreover, CLZ remains the only agent Food and Drug Administration approved for treatment-resistant schizophrenia and for reduction in recurrent suicidal behavior in affected patients. 9
However, since its introduction to the market in 1961, CLZ has been plagued by controversy because of its related side-effect profile. Initially, attention was mainly related to agranulocytosis, but recently, the focus has shifted to potentially lethal cardiotoxicity. 10 Agranulocytosis, an absolute neutrophil count less than 500 per cubic millimeter, can occur in patients treated with CLZ. 11 CLZ is associated with a 7% risk of blood dyscrasias and 1%–2% risk of agranulocytosis. 12
The pathogenesis of CLZ-induced agranulocytosis is controversial whether immunological, genetic, or toxic mechanisms (bioactivation to reactive nitrenium ion) or a multistep phenomenon are responsible for this life-threatening side effect. 13 Pereira and Dean 14 reported that CLZ bioactivation induces dose-dependent toxicity of human bone marrow stromal cells.
The most probable cardiotoxic adverse effect of CLZ is myocarditis, but dilated cardiomyopathy and pericarditis have also been reported. In addition, there is evidence that CLZ and other antipsychotics may predispose to development of the metabolic syndrome, which is a risk factor for cardiovascular disease and diabetes. 15 The mechanisms by which CLZ causes cardiotoxicity remain unclear; however, it can be explained by immunoglobulin E (IgE)-mediated hypersensitivity reaction, 16 increased catecholamine levels, 17 increased release of pro-inflammatory cytokines, 18 and oxidative stress. 19
Some clinical studies have indicated that prompt diagnosis in addition to angiotensin-converting enzyme inhibitors or β-blockers may help to limit the cardiac effects of CLZ. 20
Abdel-Wahab and Metwally 19 suggested that captopril, an angiotensin-converting enzyme inhibitor, may be effective in reducing the incidence and severity of CLZ-induced myocarditis through decreased cardiac oxidative stress and pro-inflammatory cytokines production, modulation of antioxidant stress, and protection from oxidative DNA damage.
However, there is controversy regarding the use of captopril with CLZ since some authors do not suggest the use of CLZ with other agents known to suppress bone marrow as captopril, 21 while Young et al. 22 allow its use with caution. So, this study was done to detect the cardioprotective effect of captopril against CLZ-induced myocarditis and to clarify the effect of this drug combination on bone marrow.
Materials and methods
Chemicals
CLZ (8-chloro-11-(4-methyl-1-piperazinyl)-5H-dibenzo[b, e][1,4]-diazepine) was obtained from Sigma-Aldrich, Cairo, Egypt (CAS number 5786-21-0) in the form of yellow powder.
Captopril (N-[(S)-3-mercapto-2-methylpropionyl]-
Animals
Fifty adult male Wistar albino rats with an average age of 50–60 days each weighed about 150–200 g were obtained from animal’s house, Faculty of Medicine, Zagazig University. Before commencing the experimentation, experimental animals were subjected to a 14-day period of passive preliminaries to adapt themselves to the new environment, to ascertain their physical well-being, and to exclude any diseased animals. The rats received balanced food rich in all stuff necessary to maintain their health before and during drug administration. It consisted of bread, barley, and milk. Water was offered in separate clean containers.
The current study was conducted in accordance with the international guidelines for animal research and after approval from the institutional review board on animal research at Faculty of Medicine, Egypt (study number 1973/10-3-2014).
Study protocol
The rats were divided randomly into five experimental groups as follows: Group I (negative control group): Rats of this group received only a regular diet and tap water to measure the basic parameters. Group II (positive control group): Rats received 1 ml distilled water once daily orally by gavage for 4 weeks. Group III (captopril-treated group): Each rat was treated with captopril 5 mg/kg/d (therapeutic dose)
19
dissolved in 1 ml distilled water orally by gavage for 4 weeks. Group IV (CLZ-treated group): Each rat was treated with CLZ 25 mg/kg/d (1/10 of LD50)
23
dissolved in 1 ml distilled water orally by gavage for 4 weeks. Group V (CLZ + captopril-treated group): Each rat was treated once daily with captopril (5 mg/kg) dissolved in 1 ml distilled water 1 h
24
before CLZ (25 mg/kg) dissolved in 1 ml distilled water orally by gavage for 4 weeks.
Methods
Biochemical and hematology studies
At the end of the fourth week, 24 h after the last dose, rats were anesthetized with ether and then blood samples were collected from the retro-orbital plexuses.
About 1 ml of blood was collected into a glass tube containing ethylenediaminetetraacetic acid (EDTA) as anticoagulant for assessment of complete blood count (CBC) using automated hematology analyzer, Sysmex KX-21N (Sysmex America. Inc.).
Another 1 ml of blood was collected in clean test tubes without anticoagulant, and then the sera was separated and stored frozen at −20 °C for estimation of Serum troponin I level: using Life Diagnostics rat cardiac troponin I enzyme-linked immunosorbent assay kit (cat. no. CTNI-2-HS). Serum malondialdehyde (MDA) level: using MDA biodiagnostic kit according to the colorimetric method described by Yoshioka et al.
25
Serum glutathione peroxidase (GPx) level: using GPx biodiagnostic kit according to the method of Paglia and Valentine.
26
Histopathology and immunohistochemistry studies
After sacrificing rats, the hearts were excised, washed with ice-cold saline, blotted on a piece of filter paper, and then part of the heart sample was fixed in 10% formalin saline solution for histopathological examination after staining with hematoxylin and eosin (H&E) according to Bancroft and Stevens.
27
Immunohistochemical examination of the heart sample for detection of 8-hydroxy-2-deoxy guanosine (8-OHdG), one of the most widely used biomarkers of oxidative stress,
28
was performed according to the method described by Toyokuni et al.
29
Part of the ventricle of each heart was fixed with neutral 4% formaldehyde solution. Formalin-fixed, paraffin-embedded tissues were cut into 4-μm thick sections. Then, sections were subjected to dewaxing, rehydration, and blocking with hydrogen peroxide. The antigen retrieval was performed with microwave in a 10 mm citrate buffer (pH 6.0) for 10 min and cooled to room temperature. After being blocked with 1% goat serum albumin, sections were incubated with the antibody; 8-OHdG antibody (mouse anti-8-OHdG antibody, monoclonal 15A3; Santa Cruz Biotechnology (Dallas, Texas, United States) (sc-66036)) overnight at 4 °C, followed with horseradish peroxidase–labeled secondary antibodies for 30 min at room temperature. The sections were incubated with diaminobenzidine tetrahydrochloride and counterstained with hematoxylin. The intensity of the 8-OHdG immunostaining for the nuclei of myocytes was evaluated by dividing the staining reaction into four groups: group 1: negative immunostaining (>5% of cells showing nuclear positivity); group 2: weak (+) immunostaining (5%–20% of cells showing nuclear positivity); group 3: moderate (++) immunostaining (21%–80% of cells showing nuclear positivity); and group 4: strong (+++) immunostaining (<80% of cells showing nuclear positivity).
Bone marrow examination
Femurs of each animal were exposed, the muscles and soft tissues on the bone were scraped, cleaned, and the extreme ends of the femur were cut to examine the marrow. The bone marrow was flush out using a 3-ml syringe preloaded with phosphate buffer solution. The flushing and washing were repeated 3–4 times to ensure complete removal of the bone marrow (BM) contents (about 5 ml per rat) from the femur bone. The aspirate was placed in glass tubes containing EDTA as anticoagulant.
30
Bone marrow smears were prepared as quick as possible, within 2–3 min of the animal’s death, as hematopoietic elements are fragile, and cell death occurs quickly.
31
A small drop of marrow was placed about 1 cm from the end of one glass slide and then spread toward the other end using another glass slide to ensure that it will be possible to examine the thinnest part of the film, where cytological details are optimal. Slides were left to air dry, and then films were thoroughly fixed in alcohol and stained with H&E stains.
32
Bone marrow biopsy: Femurs were fixed in 10% neutral formalin, decalcified, embedded in paraffin, and sectioned at 3-μm thickness; these sections were subjected to the H&E stains and examined by light microscope.
33
The collected data were expressed as mean ± standard deviation. The statistical analysis was done by Epi-Info statistical package program version 6.04d, January 2001, using analysis of variance (ANOVA) and least significant difference test.
Results
Results of biochemical and hematology studies
Biochemical results
Using ANOVA test to compare the mean values of serum troponin I, MDA and GPX revealed that there was no significant difference among negative control, positive control, and captopril-treated group, and so the negative control group was used to be compared with other groups of the study.
There was a highly significant increase in the mean values of serum troponin I in CLZ-treated group when compared to the negative control group (p < 0.001). Its level showed a highly significant difference in CLZ and captopril when compared to the CLZ-treated group and a non-significant difference when compared to the control group (Table 1).
Comparisons between mean values of serum troponin I, MDA, and GPX in control group, CLZ group, and CLZ + captopril group using ANOVA test.
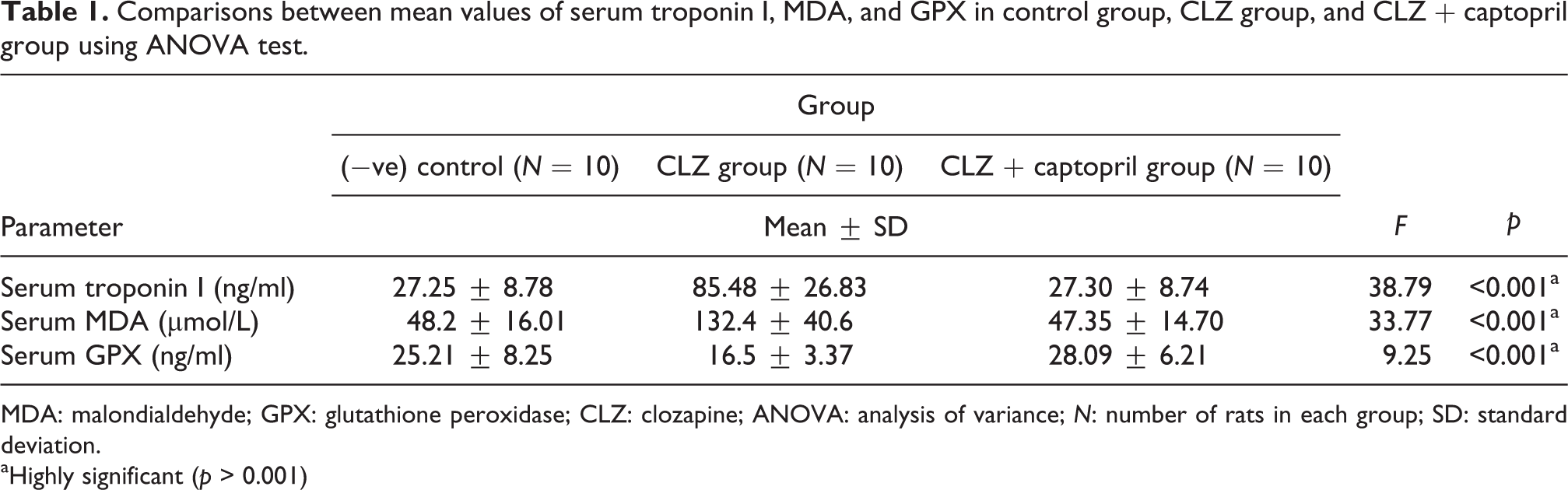
MDA: malondialdehyde; GPX: glutathione peroxidase; CLZ: clozapine; ANOVA: analysis of variance; N: number of rats in each group; SD: standard deviation.
aHighly significant (p > 0.001)
As regard to the oxidative stress parameters, there was a highly significant increase in serum MDA and a highly significant decrease in serum GPX in CLZ-treated group when compared to the negative control group. These levels showed a highly significant difference in the CLZ- and captopril-treated group when compared to the CLZ-treated group and a non-significant difference when compared to the control group (Table 1).
Hematological results
Using ANOVA test to compare the mean values of CBC among negative control, positive control, captopril-treated group, CLZ-treated group, and CLZ + captopril-treated group revealed a non-significant difference (Table 2).
Comparison between the mean values of CBC in negative control group, positive control group, and captopril group by ANOVA test.
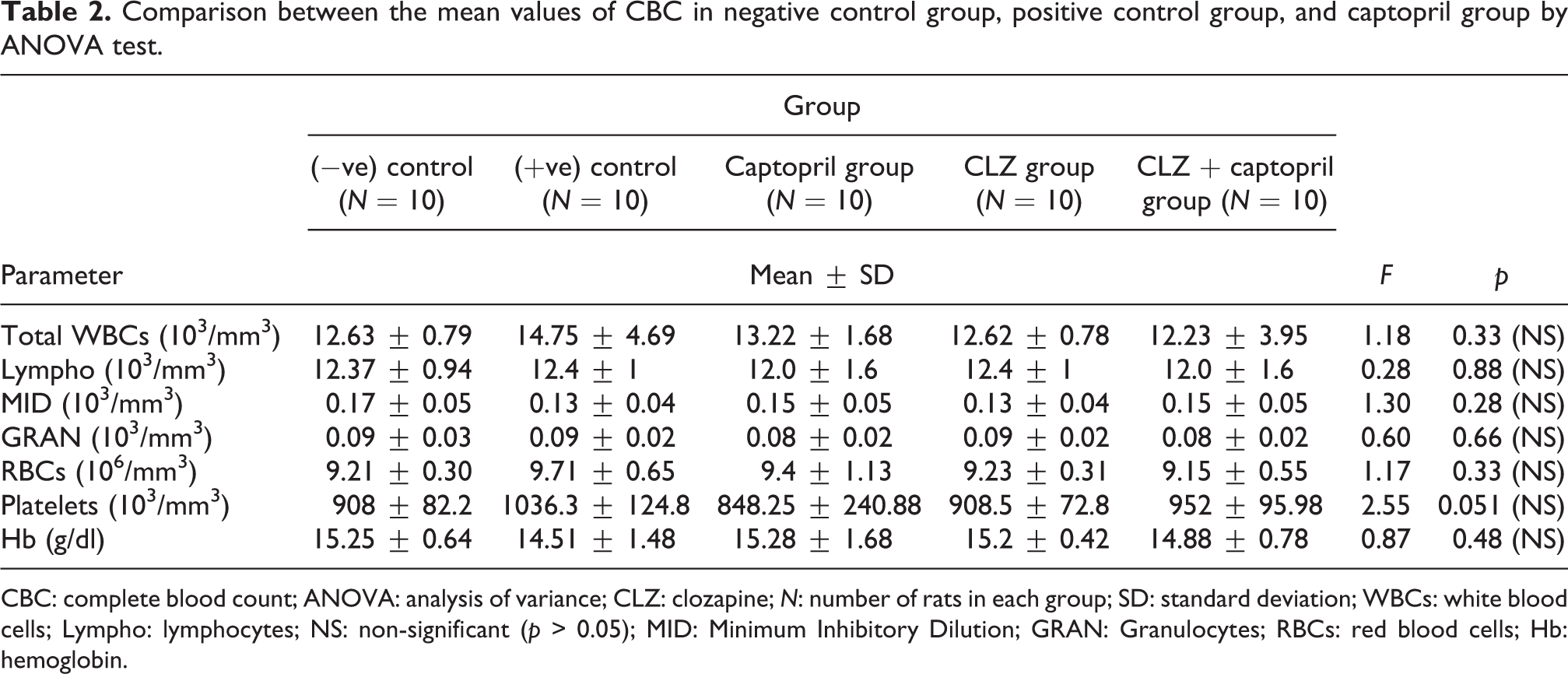
CBC: complete blood count; ANOVA: analysis of variance; CLZ: clozapine; N: number of rats in each group; SD: standard deviation; WBCs: white blood cells; Lympho: lymphocytes; NS: non-significant (p > 0.05); MID: Minimum Inhibitory Dilution; GRAN: Granulocytes; RBCs: red blood cells; Hb: hemoglobin.
Histolopathological and immunohistochemical results
Histopathological results
Microscopic examination of the heart specimens of the rats of the negative control, positive control, and captopril-treated group showed normal myocardial tissue in the form of branching and anastomosing cardiac muscle fibers with acidophilic sarcoplasm and centrally located nuclei (Figure 1(a)), while microscopic examination of the heart specimens of the rats of the CLZ-treated group showed interstitial edema (Figure 1(b) and (c)), evident fibrosis (Figure 1(d)), and disorganization and degeneration of the myocardium (Figure 1(c)). Consistent with myocarditis, inflammatory aggregates of lymphocytes were found in both the left and right ventricles, mainly in the myocardium (Figure 1(b) and (d)).
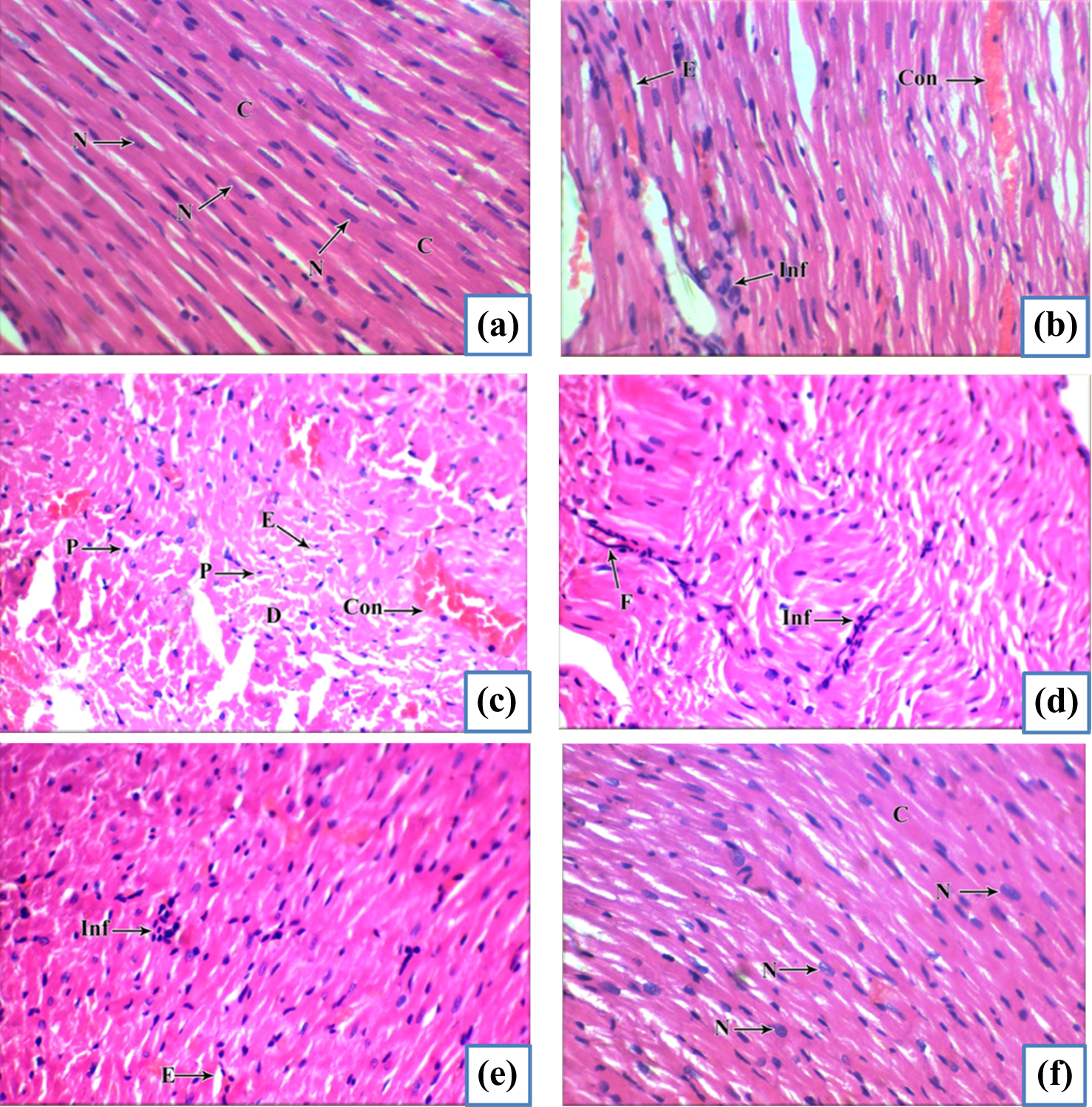
(a) Myocardium of rats of the control and captopril-treated groups showing normal branching anastomosing myocardial fibers with acidophilic cytoplasm (C) and centrally located, large, pale, vesicular nuclei (N). (b–d) Myocardium or rats of CLZ-treated group showing congestion (Con), inflammation (Inf), and edema (E) in (b), degeneration (D), pyknotic nuclei (P), congestion (C), and edema (E) in figure (c), fibrosis (F) and inflammation in (d). (e, f) Myocardium of rats of CLZ + captopril-treated group showing minimal inflammation and edema in (e) and near normal architecture in (f). CLZ: clozapine.
Histological evidence of myocarditis was classified in terms of the degree of cellular infiltration, degeneration, edema, and fibrosis and graded on a 5-point scale ranging from 0 to 4 with normal tissue score 0. 34
According to this grading, there was highly significant difference in inflammation, interstitial edema, myocardial cell degradation, and focal fibrosis between control group, captopril group, CLZ group, and CLZ + captopril group by χ 2 analysis (Table 3).
Comparisons between degree of inflammation, degeneration, edema, and fibrosis in the different studied group by χ 2 analysis.
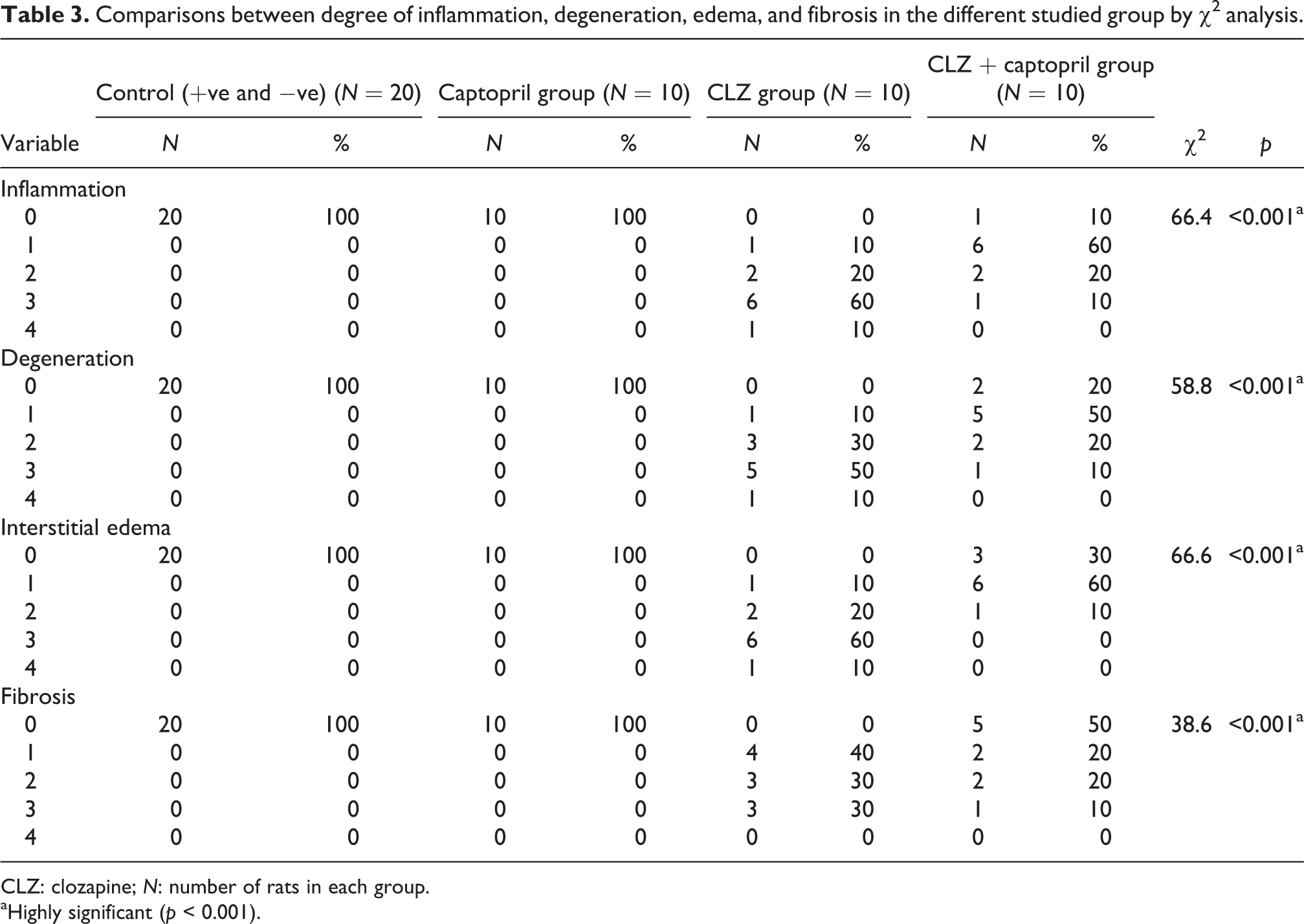
CLZ: clozapine; N: number of rats in each group.
aHighly significant (p < 0.001).
Microscopic examination of the heart specimens of the rats of CLZ + captopril-treated group revealed significant improvement of all histopathological changes compared with tissues from rats treated with CLZ alone (Figure 1(e) and (f)).
Immunohistochemical examination of the heart
8-OHdG serves as an established marker of oxidative stress as it is produced by the oxidative damage of DNA by reactive oxygen and nitrogen species. Immunohistochemical staining of 8-OHdG in myocardial section from a control rat and captopril-treated rat showed negative immunostaining (Figure 2(a)), while immunohistochemical staining of 8-OHdG in CLZ-treated rats showed strong and moderate positive brown staining indicating increased oxidative DNA damage in response to CLZ (Figure 2(b) and (c)). The immunohistochemical staining of 8-OHdG rats treated with CLZ and captopril shows attenuated expression of 8-OHdG with weak immunostaining (Figure 2(d)).
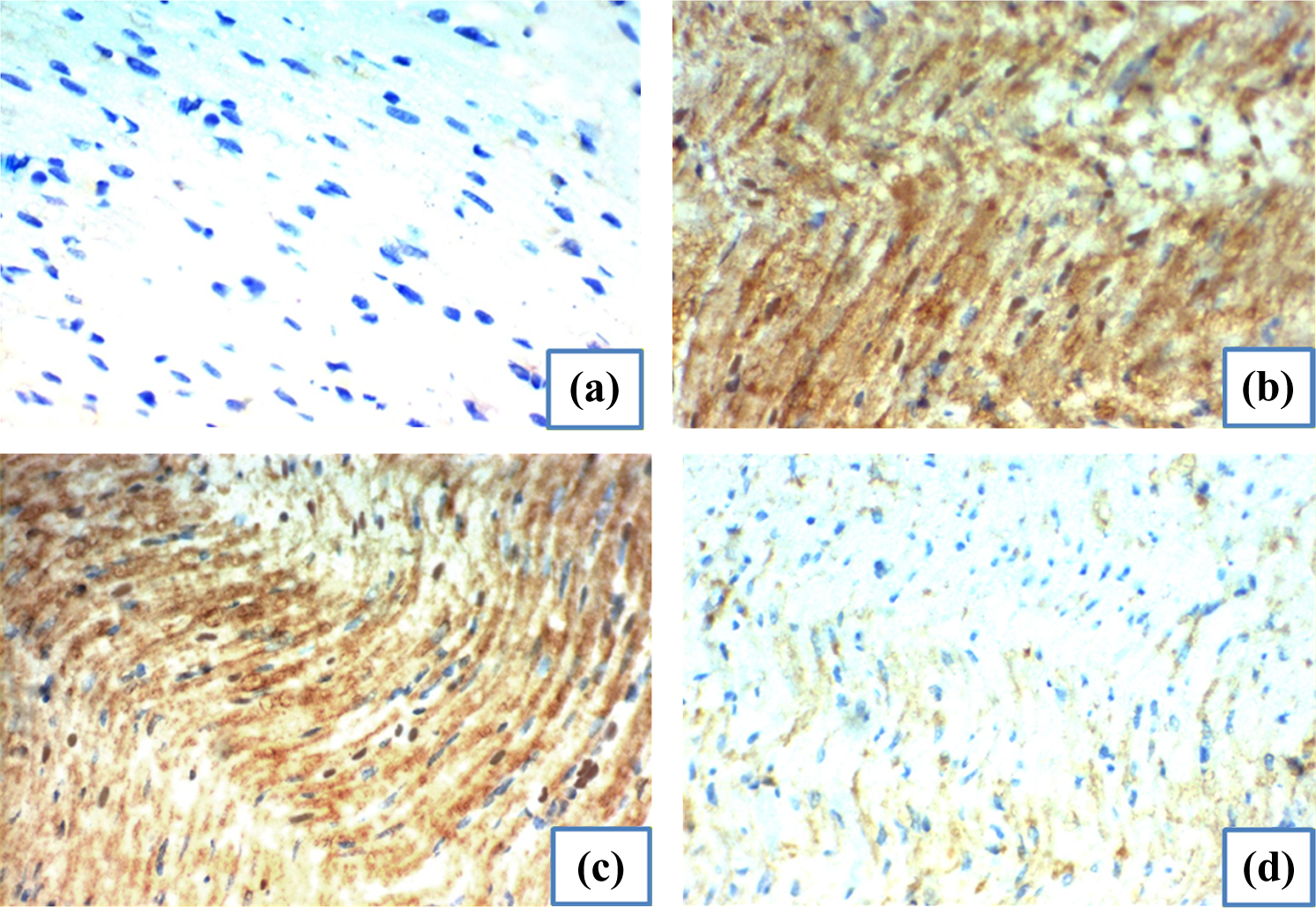
Immunohistochemical staining of 8-OHdG in myocardial section from: (a) control and captopril-treated group showing negative immunostaining. (b) CLZ-treated group showing strong nuclear and cytoplasmic immunostaining. (c) CLZ-treated group showing moderate nuclear and cytoplasmic immunostaining. (d) CLZ- and captopril-treated group showing weak immunostaining. Immunoperoxidase 400×. 8-OHdG: 8-hydroxy-2-deoxy guanosine; CLZ: clozapine.
Significant differences were observed in the degrees of 8-OHdG immunostaining among the tested groups by χ 2 analysis (Table 4).
Comparisons between degrees of immunohistochemical staining of 8-OHdG in the different studied groups by χ2 analysis.
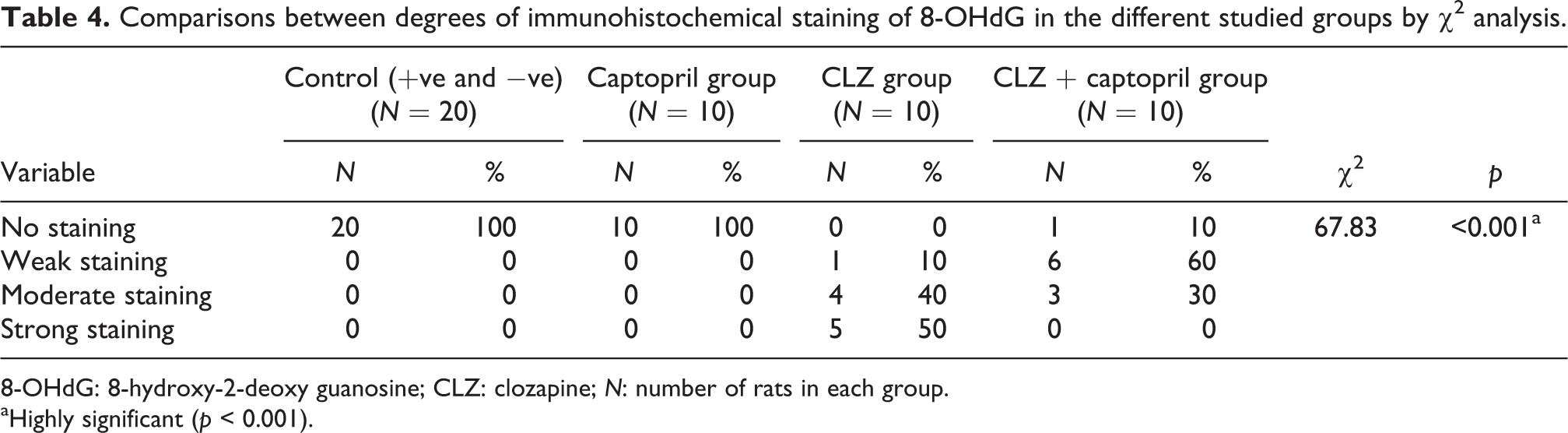
8-OHdG: 8-hydroxy-2-deoxy guanosine; CLZ: clozapine; N: number of rats in each group.
aHighly significant (p < 0.001).
Bone marrow evaluation results
Light microscopic examination of bone marrow smears (Figure 3(a)) and sections (Figure 3(b)) stained with H&E of all groups showed normal histological architecture with marked cellular crowdedness of various mature and immature hematopoietic cells with normal appearance including the characteristic of megakaryocytes, granulocytes, and few fat cells.
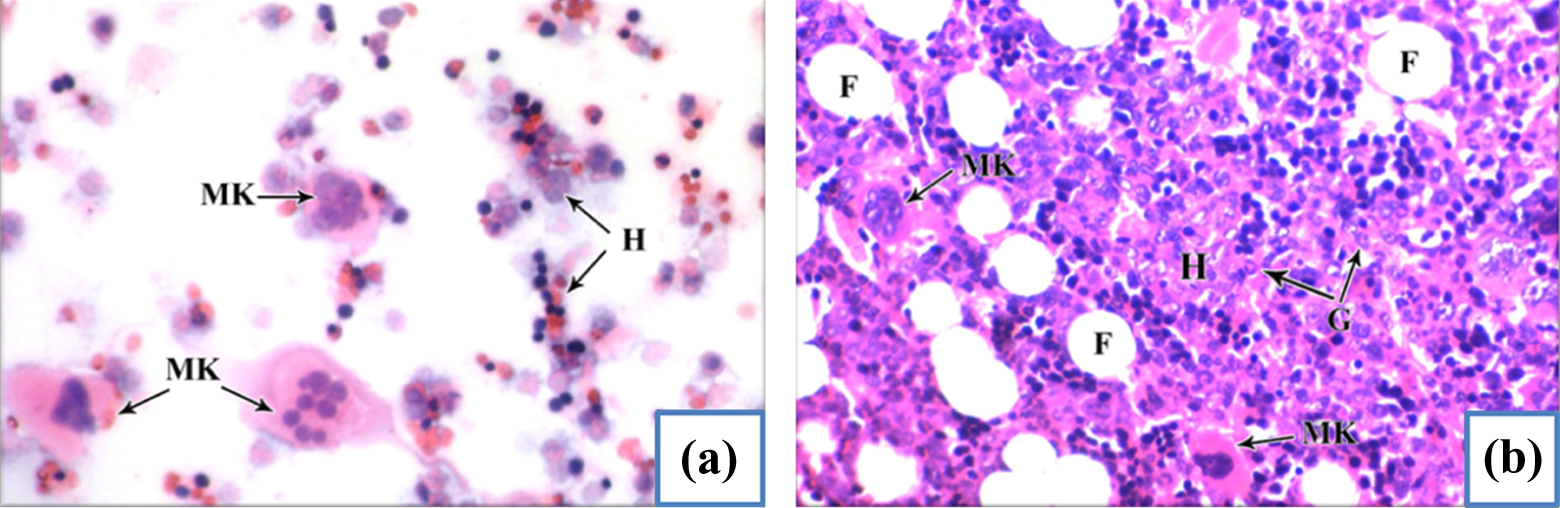
(a) Bone marrow smear of all treated group showing a variety of hematopoietic cells (H) with normal appearance and megakaryocyte cells (MK). (b) Bone marrow sections from all studied group showing multiple hematopoietic cells (H), granulocytes (G), megakaryocytes (MK), and normal fat cells (F).
Discussion
CLZ is the gold standard and the only evidence-based therapy for treatment of refractory schizophrenia. 35 It is superior in reducing suicidality and overall mortality compared to other antipsychotics. 3 However, a significant number of patients cease CLZ therapy. The main cause is potential cardiotoxicity in the form of myocarditis and cardiomyopathy. 36,37
Confirming this, the results of the present study showed a highly significant increase in the mean values of serum troponin I (p < 0.001) in CLZ-treated group when compared with the control group. Serum troponin I is a macromolecule found in cardiac myocytes and its appearance in the serum is indicative of myocyte injury. 38 Myocyte injury may be caused by myocardial infarction, myocarditis, trauma, unstable agina, cardiac surgery, or other cardiac procedures. 39
Walker 40 stated that troponin I is the most sensitive and specific marker of myocardial injury, which can provide information on the relative severity, extent, or duration of myocardial injury, while Wells and Sleeper 41 considered it the “gold standard” biomarker of myocardial injury.
Campobasso et al. 42 stated that cardiac troponin is a useful cellular marker for diagnosis of early myocardial damage as it is rapidly released following myocardial cellular injury. In patients with acute myocarditis, serum concentrations of troponin I and T are elevated more frequently than creatine kinase. 43
The results of present study showed a highly significant increase in the mean values of serum MDA (p < 0.001) and a highly significant decrease in the mean values of serum GPx (p < 0.001) in CLZ-treated group when compared with the control group. MDA is a widely accepted sensitive biomarker of lipid peroxidation; it is considered a useful measure of oxidative stress status. 44 However, GPx is an important enzyme in cellular antioxidant defense systems, detoxifying peroxides and hydroperoxides, using glutathione as the reducing substrate. 45
Since the heart tissue is very sensitive to free radical damage because of its highly oxidative metabolism and because of its fewer antioxidant defenses, 46 the results may indicate that the myocarditis process caused by CLZ is caused by oxidative stress in accordance with Heiser et al. 47 and Abdel-Wahab and Metwally. 19
CLZ induces free radical generation directly where it undergoes bioactivation to a chemically reactive nitrenium ion, which stimulates cellular injury, lipid peroxidation, and free radical formation, 48 or indirectly through increasing serum levels of catecholamines which increases myocardial oxygen demand by direct myocardial stimulation and increasing cardiac loads. In addition, catecholamines decrease myocardial oxygen perfusion, and these events can lead to myocardial ischemia and free radical generation. 49
The released free radicals and reactive oxygen species (ROS) can attack membrane phospholipids, increasing lipid peroxidation and damage to the myocardial cell membrane 50 ; this process is reflected as elevated MDA level observed in this study.
The decreased GPx activity shown in our study is due to excessive production of ROS in myocardial cells with a resultant exhaustion of cellular glutathione stores, thereby leading to the impairment of antioxidant defenses. 51
Light microscopic examination of sections of the myocardium of CLZ-treated group revealed the interstitial edema, evident focal fibrosis, congestion, disorganization, and degeneration of the myocardium. Consistent with myocarditis, as myocarditis is an inflammatory disease of the myocardium, 52 aggregates of inflammatory cells, mainly lymphocytes, were found in both the left and right ventricles, mainly in the myocardium. These histopathological lesions were also reported by Wang et al., 23 Abdel-Wahab et al., 53 and Abdel-Wahab and Metwally. 19
The degenerated cardiomyocytes with pyknotic nuclei detected in this work may be attributed to decreased cell activity or being apoptotic or proapoptotic features. 54 However, the wide spaces between cardiac muscle fibers which were also observed by Dyntar et al. 55 can be attributed to the lack of cellular coherence that contributes to cardiac dysfunction.
In the present work, focal fibrosis was evident in accordance with Cleutjens et al. 56 and Fedak et al. 57 They referred the fibrosis to replacement of degenerated cardiomyocytes by interstitial collagen and other extracellular matrix component.
Our results strongly indicate that the increased oxidative stress associated with impaired antioxidant defense status is responsible of CLZ-induced cardiotoxicity, in agreement with those reported by Heiser et al., 47 Abdel-Wahab et al., 53 and Abdel-Wahab and Metwally. 19
One of the widely recognized theories of CLZ-induced cardiotoxicity is acute IgE-mediated hypersensitivity reaction (type 1 allergic reaction), which was proposed by Kilian et al. 58 then several studies followed, 59,60 and they speculated that CLZ-associated myocarditis may be caused by hypersensitivity as evidenced by the rapid development of myocarditis (within 1–2 weeks of treatment). They noted eosinophilic infiltrates in the myocardium with peripheral eosinophilia. However, Ronaldson et al. 61 found in a clinical study that eosinophilia was delayed so it would not help in the early diagnosis of myocarditis.
However, Hägg et al. 62 criticized this theory by the absence of other type 1 reactions, such as urticaria and anaphylaxis, speculating that another mechanism seems more likely.
The results of our study disagree with this theory as the inflammatory infiltrate in myocardial sections consisted mainly of lymphocytes, didn’t include eosinophils, and there was no eosinophilia in the CBC results.
Also in support of the oxidative stress–induced myocarditis caused by CLZ, the immunohistochemical examination of 8-OHdG (an established marker of oxidative stress as it is produced by the oxidative damage of DNA by reactive oxygen and nitrogen species), the hearts of rats treated with the test compound CLZ showed positive brown staining of nuclei and cytoplasm of cardiomyocytes with levels were significantly increased in this group compared to the control group which showed negative reaction.
Under normal conditions, the free radicals attack nuclear and mitochondrial DNA, causing mutagenic DNA lesions; one of these lesions is 8-OHdG, the end product of the hydroxylation of guanine. The DNA lesions are consequently removed by the base excision repair (BER) pathway, which prevents replication of DNA lesions. However, ROS inhibit BER which may potentiate mutagenesis and DNA damage. 63
The results of our study proved that rats treated with CLZ and captopril showed a highly significant decrease in the mean values of serum troponin I (p < 0.001) when compared with the CLZ group. In addition, there was a significant improvement in all histopathological changes and the myocardial architecture of rats of this group appeared almost normal with large, central nuclei and acidophilic sarcoplasm but with minimal inflammation and edema. This indicates improvement in myocardial damage induced by CLZ.
The cardioprotective effect of captopril has been previously reported in CLZ-induced cardiotoxicity, 19,20 infection-induced cardiomyopathy, 64 drug-induced cardiomyopathy, 65,66 and autoimmune myocarditis. 67
Captopril acts as a free radical scavenger of various ROS and it has excellent antioxidant properties related to its sulfhydryl group. Sulfhydryl compounds are able to neutralize ROS by either a hydrogen donating or an electron-transferring mechanism. 68 Also, it attenuates the effect of Ang II which has been implicated in inflammation, endothelial dysfunction, myocarditis, and heart failure, 69 and many of the cellular effects of Ang II appear to be mediated by ROS. 70 The improvement in antioxidant status by captopril was reflected in this study by the decrease in the level of MDA and increased GPx level together with attenuated expression of 8-OHdG with weak brown staining, indicating that 8-OHdG levels were significantly reduced in this group compared to CLZ group. Captopril has also been shown to be immunomodulatory as it modulates chemotaxis, motility, adhesion, differentiation, activation, and cytokine and chemokine production of immune cells. 67
As regard to hematological results of the study, CBC showed an increase in the leukocyte count particularly the lymphocyte component of the WBCs in rats treated with CLZ when compared to control group, however, not reaching a statistically significant level (p > 0.05). This leukocytosis can be explained by myocarditis, consistent with several studies. 71 There was a decrease in white blood cells (WBCs) count in CLZ + captopril group when compared to CLZ group most probably due to improvements in the inflammatory process as WBCs in captopril group were within the normal range.
There was normal bone marrow histology in rats of all groups. The H&E stained specimens showed normal hematopoietic tissue with marked cellularity and few fat cells. The characteristic of megakaryocytic cells (large with multilobulated nuclei) were found in all specimens excluding any affection of the bone marrow. The agranulocytosis previously reported with CLZ did not appear in our study probably due to small number.
While some authors advised that CLZ shouldn’t be used with other drugs known to potentially affect bone marrow including carbamazepine, phenytoin, phenobarbital, captopril, and sulphonamides, 21,72 –74 our study didn’t prove increased risk of CLZ-induced bone marrow toxicity by concomitant captopril administration.
Conclusion
It can be concluded that CLZ has a toxic effect on the heart. Increased myocardial oxidative stress, cellular, and DNA damage with attenuation in antioxidant defenses are all contributing factors in the occurrence of CLZ-induced cardiotoxicity. Captopril can protect against CLZ-induced myocarditis. The protective effect of captopril was accompanied with a significant attenuation of CLZ’s effect on oxidative stress parameters (MDA), activation of antioxidant defenses (GPx), and protection from oxidative DNA damage. Captopril, in the protective dose given in our study, does not increase the risk of CLZ-induced hematotoxicity.
Footnotes
Acknowledgments
The authors express their sincere gratitude and appreciation to Dr Dina Sameh, lecturer of community medicine, Faculty of Medicine, Zagazig University, Egypt, for her assistance in performing the statistical analysis of this study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
