Abstract
Background:
Recently, urinary extracellular vesicles (uEVs) have emerged as promising biomarkers for early diagnosis, prognosis, and treatment monitoring in urothelial carcinoma (UC). uEVs encapsulate nucleic acids, proteins, and other bioactive molecules that reflect the tumor microenvironment, potentially offering a non-invasive approach for real-time cancer assessment.
Methodology:
A comprehensive literature review was conducted, focusing on recent studies evaluating uEVs in UC, particularly regarding their molecular contents, such as microRNA (miRNA), long non-coding RNAs (lncRNAs), circular RNA (circRNA), messenger RNA (mRNA), and proteins. Studies that assessed the clinical utility of uEVs for diagnosis, prognosis, and individualized treatment guidance in UC were emphasized.
Results:
Studies have identified a variety of EV-RNAs and EV-proteins as potential diagnostic and prognostic biomarkers, with some showing promise for treatment response. However, challenges in validation, limited cohort sizes, and inconsistent findings have hindered their clinical application. Liquid biopsies using uEVs are advancing UC precision medicine by improving diagnostic accuracy, identifying molecular subtypes, and potentially predicting therapeutic responses.
Conclusions:
UEVs are promising for UC management, offering a minimally invasive, accessible source of biomarkers for diagnosis, prognosis, and treatment monitoring. Although further research and large-scale validation are needed, the integration of uEVs into clinical practice has the potential to transform UC patient care by providing precise personalized management strategies. Continued advances in EV research and biomarker discovery may ultimately lead to more effective targeted UC therapies.
Introduction
Bladder cancer (BCa) is the 9th most common cancer worldwide and the second most common genitourinary malignancy. 1 BCa is generally categorized into non-muscle-invasive (NMIBC), muscle-invasive (MIBC), and metastatic disease based on the extent of tumor invasion. While NMIBC is not typically life-threatening at initial diagnosis, its management is complicated by high rates of recurrence and the risk of progression to more advanced disease. 2 For instance, patients with high-risk NMIBC face significant long-term challenges; one large study reported 10-year rates of recurrence, progression, and disease-specific mortality to be approximately 74%, 33%, and 12%, respectively, 3 underscoring the need for regular rigorous follow-up of these patients, as early detection of recurrence or progression significantly affects management and outcomes. Upper tract urothelial carcinoma (UTUC), another form of UC, follows a similar classification system and presents as muscle-invasive in 50–60% of cases at the time of diagnosis.4,5 Its diagnosis poses unique challenges compared to BCa, often requiring more invasive and complex evaluation procedures.
Given these challenges, there is a critical need for more sensitive early detection methods and more convenient tools for monitoring patients with UC. Current diagnostic and surveillance methods for UC, such as cystoscopy and ureteroscopy, are effective yet invasive, uncomfortable, and expensive. Although non-invasive tests like the UroVysion have been developed to aid in diagnosis and surveillance, 6 urine cytology remains the most widely used clinically validated method. However, the clinical utility of urine cytology is limited by its low sensitivity, particularly for low-grade tumors (16%).7,8 Additionally, its performance is even less effective for the detection of UTUC compared to BCa. 9
Furthermore, there are no reliable biomarkers for prognostication and treatment stratification, particularly for chemotherapy candidates, underscoring the urgent need for non-invasive biomarkers that can aid in diagnosis, prognosis, and treatment decision-making across various stages of UC.
Recently, liquid biopsy techniques have emerged as a promising solution to overcome some of the limitations in the current management of UC. These non-invasive approaches offer the potential to improve diagnostic precision and patient monitoring, ultimately leading to better patient outcomes. In addition, by sampling molecular information from the entire tumor mass, liquid biopsies may offer a more representative snapshot of tumor characteristics than a single tissue biopsy, thereby providing a non-invasive means to capture the molecular evolution of cancer across various stages of its progression. 10 Therefore, liquid biopsies should play a critical role in the evolution of precision medicine for UC.
This review focuses on urinary extracellular vesicles (uEVs) aimed at advancing early diagnosis, guiding individualized treatment, and predicting prognosis in UC. Specifically, we highlight nucleic acids and proteins, both encapsulated within and presented on the surface of uEVs, providing an overview of current findings. We also explore the future potential of these approaches for enhancing the management of UC.
What are extracellular vesicles?
Extracellular vesicles (EVs) are small, membrane-enclosed particles released by all cell types. Once thought to be mere cellular waste, their role as key mediators in intercellular communication was established in 2007 when Valadi et al. 11 demonstrated that EVs transfer messenger RNA (mRNA) and microRNA (miRNA) between cells. EVs transport a diverse array of bioactive molecules, including miRNAs, long non-coding RNAs (lncRNAs), circular RNAs (circRNAs), mRNAs, proteins, DNA fragments, and lipids, from their cells of origin to recipient cells, thereby influencing various biological and pathological processes. The protective lipid bilayer that surrounds EVs shields their cargo from degradation in bodily fluids, making EVs promising candidates for liquid biopsy. 12
EVs are a diverse group of particles that are categorized by their size and method of biogenesis. Exosomes (30–120 nm) are generated within multivesicular bodies and are released when these structures fuse with the plasma membrane. Microvesicles (100–1000 nm) are larger, and arise from outward budding of the cell membrane. Apoptotic bodies, 500–2000 nm in size, are released by cells undergoing programmed cell death. 13
In line with the guidelines of the International Society for Extracellular Vesicles, the term “EVs” is used here to encompass all subtypes, recognizing their functional diversity and significance in both physiological and disease contexts. 14
Recently, EVs have gained increasing attention as a form of liquid biopsy due to their stable presence in bodily fluids such as blood, saliva, urine, bronchoalveolar fluid, breast milk, and semen. These vesicles carry molecular cargo that reflects the cells from which they originate, including miRNAs, lncRNAs, circRNA, mRNAs, proteins, and lipids, which provide valuable molecular insights into the state of various diseases.13,15 The non-invasive nature of EV sampling, combined with their accessibility and stability, makes them an attractive option as biomarkers for disease management.
Clinical utility of uEVs in urothelial carcinoma
Diagnosis
EVs derived from BCa have been reported in both blood and urine; however, most EV biomarker research on BCa focuses on uEVs. This is because, in the context of UC, tumor cells are in direct contact with urine, providing a direct route for the shedding of EVs into this biofluid. 16 It is hypothesized that these uEVs carry molecular signatures of the parent tumor cells, offering a real-time, non-invasive insight into the cancer's status. This direct anatomical pathway makes urine a particularly suitable biofluid for biomarker discovery in BCa. Given these advantages, uEVs are promising tools for early diagnosis and for monitoring recurrence. Among uEV-based diagnostic markers, RNAs (miRNAs, lncRNAs, circRNAs, and mRNAs) are the most extensively studied, followed by proteins.
EVs derived from BCa have been reported in both blood and urine. However, most EV biomarker research in BCa has focused on urinary EVs (uEVs). This is because, in the context of urothelial carcinoma, tumor cells are in direct contact with urine, providing a direct anatomical pathway for the shedding of EVs into this biofluid. 16 It is therefore hypothesized that uEVs carry molecular signatures of the parent tumor cells, offering a potential real-time, non-invasive insight into the cancer's status.
Given this direct access to tumor-derived material, urine is considered a highly suitable biofluid for the discovery of biomarkers for early diagnosis and recurrence monitoring. Among the potential uEV-based diagnostic markers, RNAs (miRNAs, lncRNAs, circRNAs, and mRNAs) and proteins are the most extensively studied.
EV-RNAs
RNAs are the molecules most frequently investigated as uEV-based markers for BCa. Since Valadi et al. identified RNA in EVs,
11
extensive research has focused on uEV-RNAs as promising diagnostic markers for BCa. Typically, studies involve isolating RNA from uEVs of BCa patients and healthy controls (HCs), followed by microarray or RNA sequencing (RNA-Seq) analyses to identify candidate markers. Other studies have focused on specific RNAs in uEVs based on tissue RNA-Seq data or the literature. Although some studies lacked thorough validation, most performed follow-up validation with RT-qPCR in independent cohorts to evaluate diagnostic performance.
Urinary EV-RNA diagnosis markers for urothelial carcinoma.
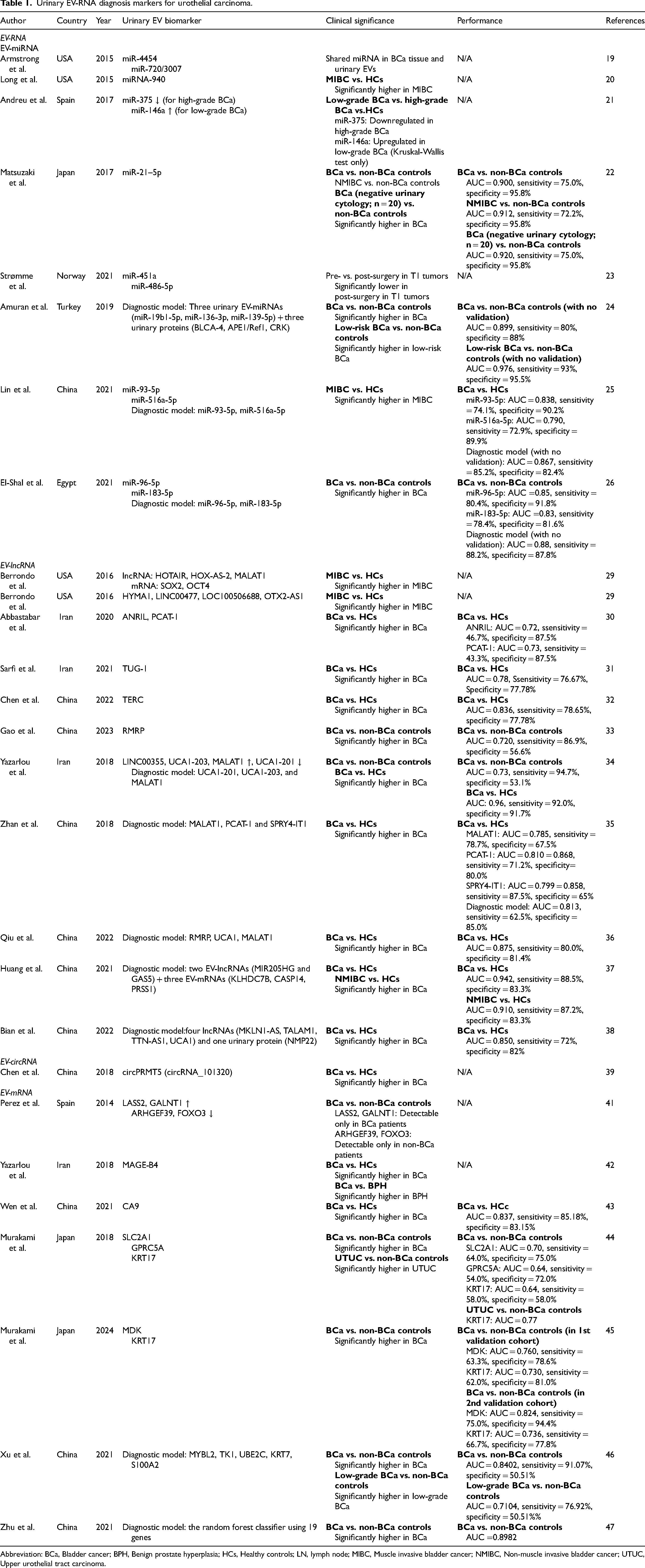
Abbreviation: BCa, Bladder cancer; BPH, Benign prostate hyperplasia; HCs, Healthy controls; LN, lymph node; MIBC, Muscle invasive bladder cancer; NMIBC, Non-muscle invasive bladder cancer; UTUC, Upper urothelial tract carcinoma.
・EV-miRNA
MiRNAs are short (20–25 nucleotides) non-coding RNAs (ncRNA) that regulate gene expression at the post-transcriptional level by binding to the 3’ untranslated region (UTR) of target mRNAs, suppressing translation or promoting mRNA degradation. 17 They are involved in various biological processes, and dysregulation of miRNAs in cancer has been linked to tumorigenesis and metastasis. Consequently, miRNAs in body fluids are promising non-invasive cancer biomarkers. In uEVs, miRNAs are shielded from degradation by a phospholipid bilayer, ensuring their stability in body fluids and making them ideal candidates for BCa diagnosis and monitoring recurrence. 18
Initial studies focused on determining whether miRNAs observed in BCa tissues and in urine samples from BCa patients could also be detected in uEVs. Armstrong et al. investigated 16 BCa tissues and corresponding uEVs and identified miR-4454 and miR-720/3007 (the latter was reclassified as a transfer RNA fragment; tRF) as miRNAs that were highly expressed in both tissues and uEVs. 19 These findings were validated within the same cohort using droplet digital PCR. Long et al. reported that levels of miRNA-940, which was identified as a urinary miRNA marker for BCa detection, were also elevated in uEVs from 30 patients with MIBC compared to 35 HCs. 20 Following these initial reports, studies have shifted toward directly identifying miRNAs specifically elevated in uEVs from BCa patients. Andreu et al. compared the uEV miRNA profiles of 34 BCa patients (16 low-grade BCa, 18 high-grade BCa) and nine HCs using microarray analysis, followed by RT-qPCR validation within the same cohort. 21 They identified EV-miR-375 as a potential marker for high-grade BCa and EV-miR-146a for low-grade BCa. Matsuzaki et al. performed miRNA microarray analysis with uEVs from six patients with BCa and three HCs and identified miR21-5p as a potential diagnostic marker. 22 Validation in a larger cohort (36 BCa patients, 24 non-BCa controls) showed an AUC of 0.90 (sensitivity 75%, specificity 95.8%), with similar performance in the subgroup with negative urine cytology with an AUC of 0.92. Strømme et al. adopted a different approach, comparing miRNAs in the uEVs of 41 NMIBC patients (27 Ta, 14 T1) before and after surgery. 23 This led to the identification of miR-451a and miR-486-5p, which were significantly downregulated in uEVs in T1 tumors after surgery, as possible markers for T1 BCa recurrence. However, since no significant differences were observed between pre-surgery BCa and non-BCa patients, their clinical potential may be limited.
Given the heterogeneous nature of BCa, several studies have emphasized the importance of using multiple miRNAs instead of a single miRNA to improve diagnostic accuracy. Amuran et al. constructed a diagnostic panel combining three uEV-miRNAs (miR-19b1-5p, miR-136-3p, and miR-139-5p) and three urinary proteins (BLCA-4, APE1/Ref1, and CRK; note that these are not uEV-proteins) using samples from 55 BCa patients, 34 HCs, and 10 follow-up patients without recurrence. This panel discriminated BC patients from non-BCa individuals with an AUC of 0.899 (sensitivity 80%, specificity 88%). 24 Similarly, El-Shal et al. developed a diagnostic panel using miR-96-5p and miR-183-5p in uEV samples from 51 BCa patients, 21 patients with urinary tract infections, and 28 HCs, achieving 88.2% sensitivity and 87.8% specificity. 25 They also found that the miR-96-5p and miR-183-5p levels in uEVs were significantly higher in patients with high-grade BCa, MIBC, and BCa with lymph node (LN) metastasis. Lin et al. performed RNA-Seq, identifying elevated miR-93-5p and miR-516a-5p in uEVs from 12 BCa patients compared to four HCs. 26 They validated this with RT-qPCR in a larger cohort (53 BCa patients, 51 HCs), and constructed a combined panel with an AUC of 0.867, sensitivity of 85.2%, and specificity of 82.4%. They also evaluated the value of EV-miR-93-5p for distinguishing MIBC (n = 21) and NMIBC (n = 32), with an AUC of 0.769 (sensitivity 90.5%, specificity 60.6%).
However, each of the above diagnostic EV-miRNA panels represented only a model report, and none has been validated in an independent cohort. Therefore, prospective validation is essential before applying these potential panels in clinical settings. Currently, a large-scale prospective cohort study (n = 3000) is underway to evaluate the diagnostic utility of the miR Sentinel™ BCa test, which measures up to 280 miRNAs in uEVs. The study aims to assess its diagnostic capability for both an initial diagnosis in patients with hematuria and recurrence monitoring after surgery in patients with BCa (NCT04155359).
・EV-lncRNA
LncRNAs are ncRNAs more than 200 nucleotides long that regulate gene expression at multiple levels, including the epigenetic, transcriptional, and post-transcriptional stages. 27 They also regulate miRNA expression. They play crucial roles in the development and progression of cancer in the early stage, making them promising biomarkers. 28 In addition to miRNAs, many lncRNAs in uEVs have been studied as potential biomarkers for BCa diagnosis.
Berrondo et al. examined eight tumor-associated lncRNAs and mRNAs and identified three lncRNAs (HOTAIR, HOX-AS-2, and MALAT1) and two mRNAs (SOX2 and OCT4) as significantly enriched in uEVs from eight MIBC patients compared to those from five HCs. 29 In the same study, they identified four additional lncRNAs (HYMA1, LINC0047, LOC100506688, and OTX2-AS1) elevated in uEVs from MIBC patients thorough RNA-Seq analysis, with validation by RT-qPCR in a separate cohort (10 BCa patients, 7 HCs). However, specific diagnostic performance metrics were not provided. 29 Abbastabar et al. reported that two lncRNAs, ANRIL and PCAT-1, were significantly elevated in uEVs from BCa patients (n = 30; T1 = 20, T2 = 10) compared to HCs (n = 10). ANRIL had an AUC of 0.72 (sensitivity 46.7%, specificity 87.5%), while PCAT-1 had an AUC of 0.73 (sensitivity 43.3%, specificity 87.5%), indicating the relatively low sensitivity of these markers despite all cases being T1 or higher. 30 However, they later reported another uEV-lncRNA, TUG-1, which showed better diagnostic accuracy for distinguishing 30 BC patients (20 Ta-T1, 10 T2) from 10 HCs with an AUC of 0.78 (sensitivity 76.7%, specificity 77.8%). 31
Chen et al. performed RNA-Seq on uEVs from four BCa patients and three HCs and identified a lncRNA, TERC, upregulated in uEVs from BCa patients. In RT-qPCR validation using urine samples from 89 BCa patients and 63 HCs, uEV-TERC had an AUC of 0.836 (sensitivity 78.65%, specificity 77.78%). Furthermore, uEV-TERC was significantly upregulated in 108 high-grade BCa patients compared to 20 low-grade BCa patients. 32 In another study, they also reported that RMRP was the most highly expressed uEV-lncRNA in BCa patients relative to HCs in the same RNA-Seq data. 33 In the RT-qPCR validation using an independent cohort (99 BCa patients, 50 patients with benign urinary lesions, and 84 HCs), uEV-RMRP had an AUC of 0.720 (sensitivity 86.9%, specificity 56.6%) for BCa diagnosis. They also found that uEV-RMRP expression was higher in 26 MIBC patients than in 73 NMIBC patients.
Similar to miRNA studies, several researchers have reported diagnostic models with multiple uEV-lncRNAs, focusing on those highlighted as promising in the literature. Yazarlou et al. evaluated five tumor-associated lncRNAs in uEVs and found that LINC00355, UCA1-203, and MALAT1 were significantly higher in 59 BCa patients than in 49 non-BCa controls, while UCA1-201 was lower. 34 Their model using UCA1-201, UCA1-203, and MALAT1 in uEVs showed improved accuracy over individual RNAs, with an AUC of 0.73 (sensitivity 94.7%, specificity 53.1%), although it was not validated, and the low specificity implied false positives from non-malignant urinary conditions. Zhan et al. assessed eight tumorigenesis-related lncRNAs in uEVs from 104 BCa patients and 104 HCs and developed a logistic regression model comprising three lncRNAs (MALAT1, PCAT-1, and SPRY4-IT1). 35 In the validation set (80 BCa patients, 80 HCs), the model achieved an AUC of 0.813 (sensitivity 62.5%, specificity 85.0%). They also found that PCAT-1 and MALAT1 upregulation in uEVs was associated with a poor recurrence-free survival of NMIBC, with uEV-PCAT-1 overexpression being an independent prognostic factor. Qiu et al. also selected seven lncRNAs from The Cancer Genome Atlas (TCGA) database and literature, constructing a support vector machine (SVM) model with three lncRNAs (RMRP, UCA1, and MALAT1) in the training cohort (22 BCa patients, 20 HCs) and validating it in an independent cohort (33 BCa patients, 23 HCs). 36 The AUC of this lncRNA SVM model was 0.875 (sensitivity 80.0%, specificity 81.4%).
Some studies have combined lncRNAs with other molecules, such as mRNAs and proteins. Huang et al. identified tumor-progression-related lncRNAs and mRNAs by comparing deeply infiltrating tumor regions with superficial tumor regions in three MIBC tissues via RNA-Seq. 37 They validated these findings with the TCGA dataset and developed a diagnostic model based on the expression of these RNAs in uEVs, using a training cohort of 10 BCa patients and 10 HCs. The model, incorporating two EV-lncRNAs (MIR205HG and GAS5) and three EV-mRNAs (KLHDC7B, CASP14, and PRSS1), was validated in a separate cohort (80 BCa patients, 80 HCs) with RT-qPCR, achieving an AUC of 0.942 (sensitivity 88.5%, specificity 83.3%). Furthermore, higher KLHDC7B, CASP14, PRSS1, and MIR205HG expression in uEVs correlated with tumor stage and grade. However, this expression also correlated with the degree of hematuria, implying the need to confirm that hematuria does not lead to false positives. Bian et al. performed RNA-Seq on uEVs from seven BCa patients and seven controls; they identified differentially expressed lncRNAs and constructed a logistic regression model with four lncRNAs (MKLN1-AS, TALAM1, TTN-AS1, and UCA1) and one urinary protein (NMP22) in the training cohort (50 BCa patients, 50 controls). 38 In the validation cohort (43 BCa patients, 43 HCs), the panel had an AUC of 0.850 (sensitivity 72%, specificity 82%).
・EV-circRNA
CircRNAs are small ncRNAs with covalently closed loop structures without 5′ caps and 3′ poly tails. Studies have implied that circRNAs play vital roles in tumorigenesis and metastasis, functioning as miRNA and RNA-binding protein sponges, protein translation and gene transcription regulators, and competing products during pre-mRNA splicing. 39 Like miRNA and lncRNA, circRNAs are also reported to be enriched and stable in EVs, and could be used as tumor biomarkers. However, there have been few studies of uEV-circRNAs as biomarkers for BCa.
Chen et al. screened circRNA expression profiles using a circRNA microarray in three paired BCa and normal tissues, and identified circRNA_101320 (called circPRMT5) as the most highly upregulated circRNA in BCa tissue, with validation using RT-qPCR (n = 119). 40 They demonstrated that uEV-circPRMT5 was significantly higher in 18 BCa patients than in 14 HCs, and higher in 10 patients with metastasis than in 8 without metastasis.
・EV-mRNA
Although limited in number compared to ncRNAs, mRNAs in uEVs have also been reported to be diagnostic markers for BCa.
Perez et al. published the first report on the potential diagnostic value of the transcriptome in uEVs for BCa in 2014. 41 They evaluated mRNA expression through microarray analysis of uEVs from five BCa patients and six non-BCa patients with various urinary tract conditions, and identified differentially expressed mRNAs. The results were validated using RT-qPCR in two BCa patients and two non-BCa patients, revealing that LASS2 and GALNT1 were detectable only in BCa patients, while ARHGEF39 and FOXO3 were found only in non-BCa patients. Yazarlou et al. analyzed the expression of seven cancer-testis antigens (CTAs) and NMP22 using RT-qPCR in uEVs from 59 BCa patients, 25 patients with benign urologic diseases, and 24 HCs. 42 This showed that MAGE-B4 expression in uEVs was significantly higher in BCa patients compared to HCs, but lower in BCa patients than in eight with benign prostatic hyperplasia (BPH), indicating a potential cause of false positives. Wen et al. demonstrated that the expression of CA9 mRNA in uEVs was significantly higher in 168 BCa patients than in 90 HCs, with an AUC of 0.837 (sensitivity 85.18%, specificity 83.15%). 43
Murakami et al. performed RNA-Seq analysis on uEVs from four BCa patients, two patients with no residual BCa after resection, and four healthy and disease controls, identifying 12 mRNAs for BCa detection. 44 In the validation cohort (131 BCa patients, 32 UTUC patients, and 35 non-BCa controls) using RT-qPCR, three uEV-mRNAs (SLC2A1, GPRC5A, and KRT17) performed diagnostically with an AUC of 0.70 for SLC2A1 (sensitivity 64.0%, specificity 75.0%), 0.64 for GPRC5A (sensitivity 54.0%, specificity 72.0%), and 0.64 for KRT17 (sensitivity 58.0%, specificity 58.0%). Notably, they also showed that KRT17 had diagnostic performance for detecting UTUC (n = 32), with an AUC of 0.77. Based on this study, the same group conducted a prospective multicenter study to validate the diagnostic value of these three uEV-mRNAs along with two additional markers from the literature, MDK and CXCR2, in two settings: diagnosis (236 BCa patients, excluding UTUC, and 42 non-BCa controls) and longitudinal monitoring for NMIBC recurrence (n = 189). 45 In the diagnosis setting, MDK had the highest AUC (0.760) for detecting BCa (sensitivity 63.3%, specificity 78.6%), followed by KRT17 (AUC 0.730, sensitivity 62.0%, specificity 81.0%). Notably, in the longitudinal monitoring setting, KRT17 expression in uEVs remained elevated in patients with recurrence before confirmation of recurrence and gradually decreased over time in those without recurrence, implying that uEV-KRT17 is a potential biomarker for BCa detection and recurrence prediction.
Diagnostic panels combining multiple uEV-mRNAs for detecting BCa have also been reported. Xu et al. selected candidate mRNAs based on tissue RNA-Seq data from the TCGA database and developed a diagnostic model incorporating five mRNA markers (MYBL2, TK1, UBE2C, KRT7, and S100A2) using uEVs from a training cohort (126 BCa patients, 242 negative controls). 46 With the validation set (56 BCa patients, 99 non-BCa controls), the model had an AUC of 0.8402 (sensitivity 91.07%, specificity 50.51%). Zhu et al. analyzed mRNA from uEVs from 35 BCa patients and 35 controls with other urinary diseases using RNA-Seq, establishing a random forest classifier using 19 mRNAs for BCa diagnosis. 47 This model had an AUC of 0.8982 in an internal validation cohort (seven BCa patients, seven controls). Notably, this study revealed that while uEVs predominantly expressed tissue-specific genes for the bladder, the genes upregulated in the uEVs of BCa patients were more closely associated with immune-related signatures than with BCa-specific genes. This underscores the potential of immune-cell-derived EV-mRNAs as valuable markers for BCa diagnosis.
EV-protein
Proteins in uEVs are also promising diagnostic markers for BCa. Although they have been estimated to constitute only around 3% of all urinary proteins,
48
advances in mass spectrometry have enabled comprehensive analysis of uEV-proteins. Since the initial report of uEV proteins by Pisitkun et al. in 2004,
49
numerous uEV-proteins have been reported to be potential diagnostic markers for BCa. However, most early studies highlighted only differences in the expression of specific proteins between uEVs from BCa patients and controls, without providing details on their diagnostic performance.
Urinary EV-protein diagnosis markers for urothelial carcinoma.
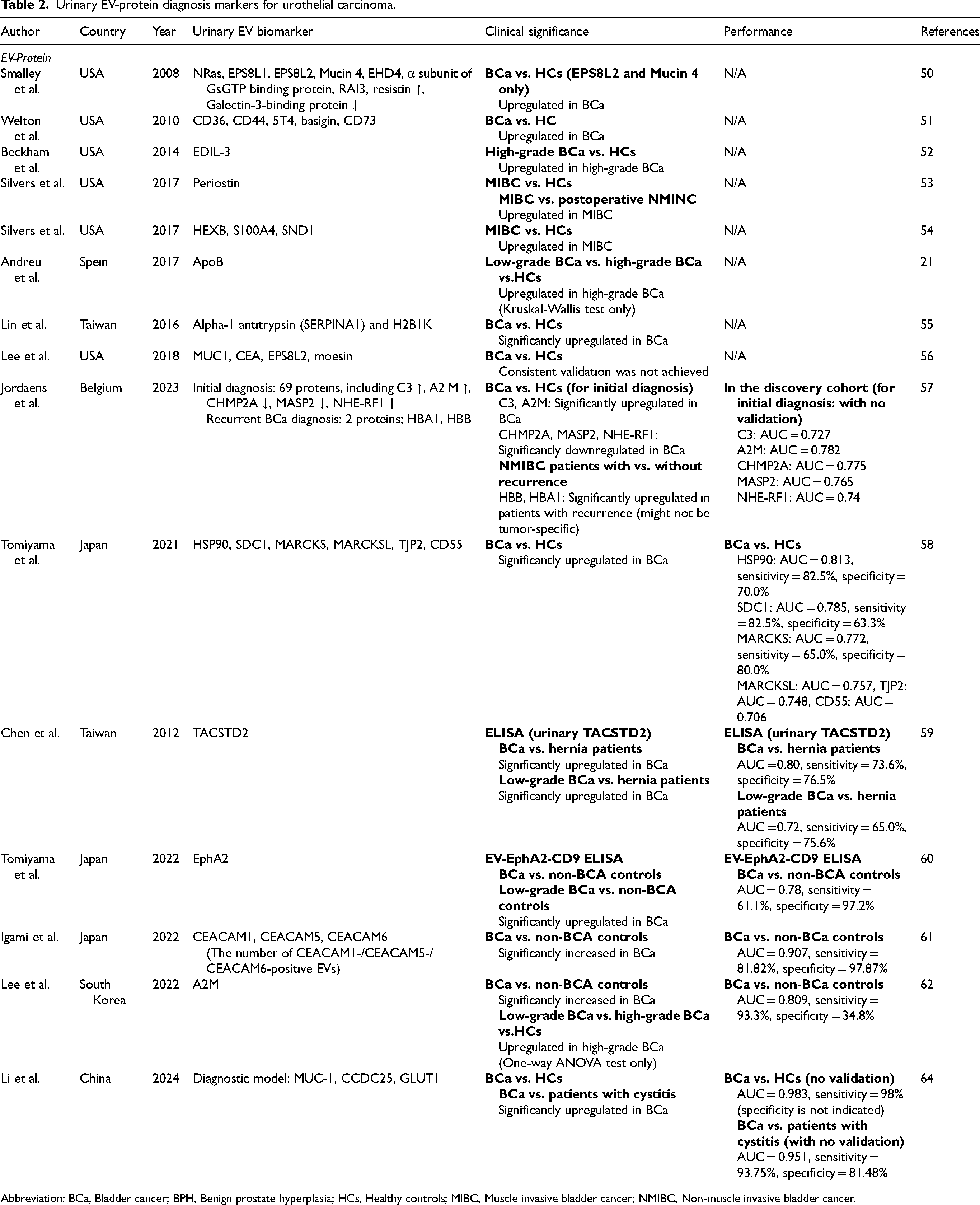
Abbreviation: BCa, Bladder cancer; BPH, Benign prostate hyperplasia; HCs, Healthy controls; MIBC, Muscle invasive bladder cancer; NMIBC, Non-muscle invasive bladder cancer.
Smalley et al. conducted the first analysis in this field, using liquid chromatography-tandem mass spectrometry (LC−MS/MS) on uEVs from four BCa patients and five HCs. 50 Notably, they excluded urine samples with hematuria from both groups, as the presence of blood was found to alter the uEV protein profile. Consequently, they identified eight uEV proteins upregulated in BCa patients, including components of the epidermal growth factor (EGF) pathway (NRas, EPS8L1, EPS8L2, Mucin 4, and EHD4), the α subunit of the G protein Gs, resistin, and retinoic acid protein 3. Following this study, Welton et al. performed proteomic analysis using liquid chromatography-matrix-assisted laser desorption/Ionization time-of-flight/time-of-flight mass spectrometry (LC-MALDI-TOF/TOF MS) on EVs derived from a high-grade BCa cell line (HT1376), rather than uEVs, to avoid interference of EVs from other cell types, like renal and urothelial epithelial cells, commonly found in urine samples. 51 To minimize contamination from non-EV proteins, they used ultracentrifugation with a sucrose cushion for EV isolation. They confirmed that these high-purity BCa cell-derived EVs expressed proteins associated with cancer, and some of these proteins (CD36, CD44, 5T4, basigin, and CD73) were detected at higher levels in uEVs from one BCa patient than in one HC. These two initial studies underscore the potential of uEV-protein analysis for BCa diagnosis and provide a foundation for research in this field.
Using a similar approach to Welton et al., another group has published several studies exploring diagnostic markers for BCa through proteomic analysis of BCa cell-derived EVs. Beckham et al. also performed LC-MS/MS on EVs from a high-grade BCa cell line (TCC-SUP) and compared them with EVs from SV-HUC, identifying 108 differentially expressed proteins. 52 Among these, they highlighted EDIL-3 and confirmed that its levels were significantly higher in uEVs from 12 high-grade BCa patients than in 12 healthy controls on Western blot analysis. From the 108 identified proteins, the same group focused on periostin and demonstrated that it was more abundant in uEVs from 9 MIBC patients than in 13 HCs and 6 postoperative NMIBC patients. 53 Furthermore, they re-analyzed their proteomic data from EVs from TCC-SUP and SV-HUC, selecting six proteins based on GO term annotations and published BCa tissue microarray data. 54 In Western blot analysis, three of these six proteins (HEXB, S100A4, and SND1) were expressed more in uEVs from six MIBC patients than in six HCs.
Most studies have conducted comparative proteomic analyses of uEVs, rather than BCa cell-derived EVs, to identify diagnostic markers for BCa. Andreu et al. compared the uEV protein profiles of four high-grade BCa patients versus five HCs using LC-MS/MS and identified ApoB in uEVs as a potential biomarker for high-grade BCa patients with validation using dot-blot analysis in an independent cohort (16 low-grade BCa patients, 16 high-grade BCa patients, and 6 HCs). 21 Lin et al. conducted MALDI-TOF analysis of uEVs from 129 UC patients and 62 non-UC patients, identifying alpha-1 antitrypsin (SERPINA1) and H2B1 K in uEVs as diagnostic markers for UC. These uEV-proteins were also associated with a higher risk of UC recurrence and progression in the internal validation cohort (137 UC, 65 non-UC), with H2B1 K an independent predictor of UC recurrence. They validated these EV-proteins as diagnostic markers using LC-MS/MS, and found that uEVsERPINA1 and H2B1 K levels were significantly higher in 5 UC patients than in 10 non-UC patients. 55 Lee et al. performed LC-MS/MS analysis of uEVs from 10 BCa patients and 10 HCs, identifying 56 proteins that were significantly upregulated in BCa patients. 56 Four proteins (MUC1, CEA, EPS8L2, and moesin) were selected for Western blot validation using samples from seven BCa patients and six HCs. However, consistent validation was not achieved due to variability among the urine samples. Jordaens et al. also conducted LC-MS/MS analysis of uEVs from BCa patients and controls in the initial diagnosis setting (50 NMIBC patients, 64 HCs) and in a follow-up setting (46 NMIBC patients with recurrence vs. 109 patients without recurrence). 57 They identified 69 differentially expressed proteins, including C3, A2 M, CHMP2A, MASP2, and NHE-RF1, as potential markers for the initial diagnosis, and two proteins (HBB and HBA1) as specific detection markers for recurrent BCa. However, given that HBB and HBA1 are blood-related proteins, these proteins might not be tumor-specific, and these study findings need validation.
Taking a unique approach to identify BCa-specific proteins in uEVs, Tomiyama et al. combined LC-MS/MS analysis of uEVs (seven BCa patients, four HCs) and tissue-exudative EVs (Te-EVs), which were isolated from cultured medium of freshly resected viable BCa tissues (n = 7). 58 They revealed that most of the proteins identified in Te-EVs were also present in uEVs (82.4%), with 55 upregulated in uEVs from BCa patients. In validation through target proteomic analysis (selected reaction monitoring/multiple reaction monitoring: SRM/MRM) of uEVs from 40 BCa patients and 30 HCs, six proteins (HSP90, SDC1, MARCKS, MARCKSL, TJP2, and CD55) were validated as significantly upregulated in BCa patients with an AUC of 0.813 for HSP90 (sensitivity 82.5%, specificity 70.0%), 0.785 for SDC1 (sensitivity 82.5%, specificity 63.3%), and 0.772 for MARCKS (sensitivity 65.0%, specificity 80.0%).
Importantly, focusing on membrane proteins located on the EV surface offers a major clinical advantage, as these proteins can be detected using common techniques like ELISA or FACS. Chen et al. conducted LC–MS/MS of uEVs from nine BCa patients and nine controls (hernia patients), identifying proteins with differential expression. 59 The validation analysis using SRM/MRM confirmed 24 EV proteins as differentially expressed in 28 BCa patients versus 12 in hernia patients. Among these, TACSTD2 was selected as a promising diagnostic marker, not significantly upregulated in patients with hematuria or urinary tract infections (n = 8). They quantified urinary TACSTD2 levels using ELISA in 221 samples (140 BCa patients, 81 hernia patients), yielding an AUC of 0.80 (sensitivity 73.6%, specificity 76.5%). However, they measured TACSTD2 in raw urine samples rather than specifically from uEVs. Notably, TACSTD2, also known as Trop-2, is highly expressed in many cancers, including UC, and is targeted by the antibody-drug conjugate, sacituzumab govitecan. The clinical relevance of any link between urinary TACSTD2 levels and the efficacy of sacituzumab govitecan in UC patients is of interest.
In another study, Tomiyama et al. selected 13 candidate proteins based on prior LC–MS/MS analysis 58 and validated them using SRM/MRM in uEVs from 49 BCa patients and 48 non-BCa controls (36 HCs, 12 hematuria patients). 60 EphA2 on the surface of uEVs was identified as a diagnostic marker capable of distinguishing BCa patients from both HCs and hematuria patients. Then, they developed an ELISA (EV-EphA2-CD9 ELISA) for quantifying EphA2 in uEVs without ultracentrifugation and evaluated uEVs from 36 BCa patients and 36 non-BCa controls (26 HCs, 10 hematuria patients), achieving an AUC of 0.78 (sensitivity 61.1%, specificity 97.2%). Igami et al. also performed LC-MS/MS analysis of uEVs from 18 BCa patients and 29 HCs, but they focused on “medium EVs”, which were pelleted at intermediate speeds without ultracentrifugation, enabling simpler, more reproducible isolation. 61 They identified CEACAM1, CEACAM5, and CEACAM6 on the surface of uEVs as diagnostic markers for BCa. Subsequently, they analyzed uEVs from 31 BCa patients, 18 patients with benign urologic conditions, and 29 HCs to evaluate the diagnostic performance of the number of EVs expressing CEACAM1, −5, and −6 using flow cytometry. This approach yielded an AUC of 0.907 (sensitivity 81.82%, specificity 97.87%).
Lee et al. took a different approach. 62 Instead of using proteomics, they performed protein array analysis, intending to develop an antibody-based diagnostic method. They identified two proteins that showed significant changes in uEVs from two high-grade BCa patients between pre- and post-surgery, selecting A2 M in uEVs as a novel diagnostic marker for BCa. Furthermore, they validated its diagnostic value in an independent cohort of 83 urine samples [28 low-grade BCa patients; 32 high-grade BCa patients; 23 non-BCar controls (17 patients with BPH, 6 with hematuria)] by analyzing uEVs with ELISA; uEV-A2 M had an AUC of 0.809 (sensitivity 93.3%, specificity 34.8%). However, it remains unclear whether A2 M, a secreted protein rather than a membrane protein, 63 can be detected on the surface of EVs using ELISA.
Furthermore, there have been attempts to measure marker proteins directly in raw urine samples without isolating EVs. Li et al. developed an immunoassay based on a choline phosphate-grafted platinum nanozyme (Pt@CP). 64 This assay captures uEVs by antibodies immobilized on the plate and detects them using Pt@CP, which functions as a universal EV probe by binding to phosphatidylcholine on the EV membrane. Using the Pt@CP-based ELISA, three uEV-protein markers (MUC-1, CCDC25, and GLUT1) were identified, and a logistic regression model was constructed using measurements from 99 urine samples (48 BCa patients, 27 patients with cystitis, and 24 HCs). This model achieved an AUC of 0.983 and a sensitivity of 98% in the same cohort.
Prognosis
Cancer-derived EVs play a crucial role in tumor progression by facilitating intercellular communication. Consequently, molecules in uEVs hold promise not only as diagnostic markers but also as prognostic markers for risk stratification of UC. Several studies have assessed the aggressiveness of BCa using uEVs, with miRNAs and lncRNAs frequently highlighted as prognostic markers due to their functional significance.
Urinary EV-based prognosis markers for urothelial carcinoma.
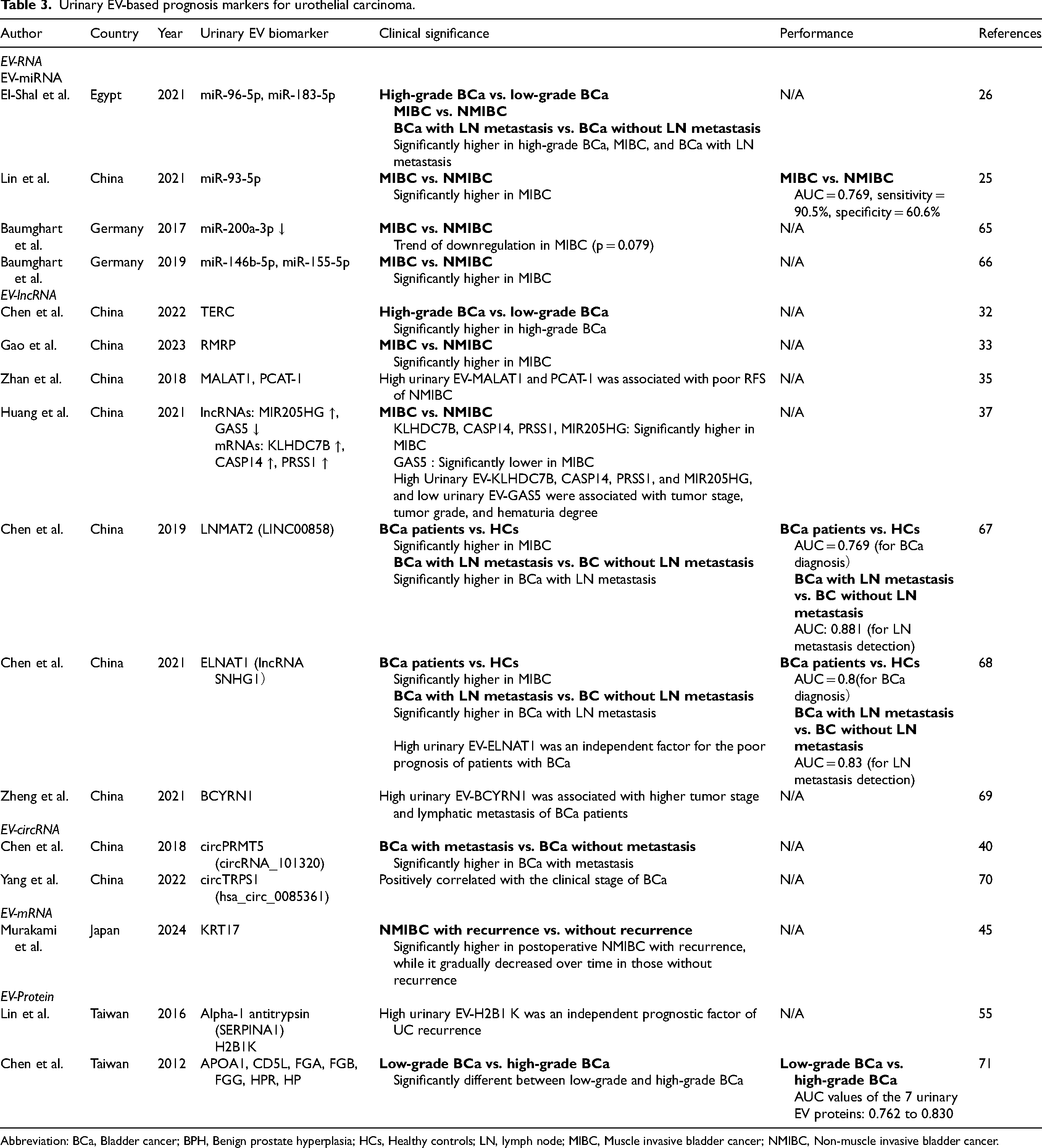
Abbreviation: BCa, Bladder cancer; BPH, Benign prostate hyperplasia; HCs, Healthy controls; LN, lymph node; MIBC, Muscle invasive bladder cancer; NMIBC, Non-muscle invasive bladder cancer.
EV-RNAs
・EV-miRNA
Baumghart et al. aimed to differentiate NMIBC from MIBC by analyzing uEVs. 65 Initially, they identified six invasion-associated EV-miRNAs using microarray analysis of BCa cell-derived EVs and their parental BCa cell lines [MIBC cells (T24, J82, and 253J-BV) versus NMIBC cells (RT112 and 5637)]. Subsequent validation by RT-qPCR showed a downregulation trend (p = 0.079) of uEV-miR-200a-3p in 14 MIBC patients compared to 7 NMIBC patients. Following this study, they extended their investigation to 24 clinical BCa tissue samples. 66 Microarray analysis identified two miRNAs (miR-146b-5p and miR-155-5p) that were significantly upregulated in MIBC tissue compared to NMIBC tissue with RT-qPCR validation in a larger tissue cohort (n = 56). Finally, they assessed the expression of these miRNAs in uEVs, finding that expression of both miR-146b-5p and miR-155-5p was significantly higher in uEVs from 20 MIBC patients compared to those from 17 NMIBC patients.
・EV-lncRNA
Chen et al. explored using uEVs to diagnose LN metastasis in BCa. 67 They analyzed lncRNA profiles in five pairs of MIBC and adjacent normal tissues, as well as in five pairs of LN-positive and LN-negative BCa tissues using RNA-Seq and validated the results with RT-qPCR in 266 BCa tissues. They identified LINC00858, termed lymph node metastasis-associated transcript 2 (LNMAT2), as lncRNAs overexpressed in BCa tissues and linked to LN metastasis. 68 They then found that uEV-LNMAT2 was elevated in BCa patients and positively correlated with LN metastasis in a validation cohort (206 BCa patients [52 LN+, 154 LN-] and 120 HCs), achieving an AUC of 0.769 for BCa diagnosis and 0.881 for LN metastasis detection. The same group also conducted RNA-Seq using uEVs from five MIBC patients and five healthy controls and combined their previous RNA-Seq data from BCa tissue, identifying lncRNA SNHG16, termed EV-mediated LN-associated transcript 1 (ELNAT1) as a lncRNA associated with LN metastasis. UEV–ELNAT1 that was significantly upregulated in BCa patients and associated positively with LN metastasis in a validation cohort (242 BCa patients [50 LN+, 192 LN- and 166 HCs), achieving an AUC of 0.80 for BCa diagnosis and 0.83 for LN metastasis detection, and uEV-ELNAT1 was an independent factor for the poor prognosis of patients with BCa. Furthermore, in another study, the same group identified uEV-lncRNA, BCYRN1, which is correlated with the activation of the VEGF-C/VEGFR3 signaling, with validation in an independent cohort (210 BCa patients [53 LN+, 157 LN-] and 112 HCs) with uEV-BCYRN1 overexpression positively correlated with higher tumor stage and lymphatic metastasis of patients with BCa. 69
・EV-circRNA
Yang et al. performed RNA-Seq using four paired BCa tissues and normal tissues to identify circRNAs with significant biological functions in BCa, identifying circTRPS1 (hsa_circ_0085361) as one of the most abnormally expressed circRNAs in BCa tissues. 70 They found that circTRPS1 expression in uEVs was positively correlated with the clinical stage of BCa (n = 20; T1 = 6; T2 = 7; T3 + 4 = 7).
EV-protein
Chen et al. performed MRM/MS analysis on uEVs from 28 BCa patients and identified seven proteins (APOA1, CD5L, FGA, FGB, FGG, HPR, and HP) that were differentially enriched in uEVs between 7 low-grade and 21 high-grade BCa, serving as potential grade discriminators with AUCs ranging from 0.762 to 0.830. 71
Future directions of urinary extracellular vesicles in urothelial carcinoma
There has been marked interest in the study of uEVs in UC, with numerous reports highlighting their potential as biomarkers. However, inconsistencies in findings and the diverse range of targeted molecules imply that clinical implementation may take time. To date, the only FDA-approved uEV biomarker in urology is ExoDx, 72 which targets mRNA in uEVs to detect prostate cancer and has demonstrated efficacy through multiple studies, including prospective trials.73–75 Given that UC cells are continuously bathed in urine, identifying specific molecular markers within EVs could yield effective clinical biomarkers, akin to ExoDx. However, most current studies of uEVs in UC are based on relatively small sample sizes, which may introduce institutional biases and overfitting, potentially affecting results. Moving forward, large-scale, multicenter studies are essential to validate these findings and minimize such biases.
At present, circulating tumor DNA (ctDNA) is the biomarker closest to clinical application. CtDNA, which consists of fragmented DNA shed into the circulation by apoptotic and necrotic tumor cells, provides direct insights into genomic alterations within a tumor. 76 Recent research, often as part of clinical trial ancillary studies, has explored the utility of ctDNA as a prognostic marker. 77 For example, in the IMvigor010 study, ctDNA positivity in post-surgical UC patients indicated a poor prognosis, and adjuvant atezolizumab treatment improved both disease-free and overall survival in ctDNA-positive patients. 77 Furthermore, studies have evaluated urinary ctDNA as a diagnostic tool for UC. For instance, Tamura et al. demonstrated that urinary ctDNA monitoring could detect intravesical recurrence after radical nephroureterectomy for UTUC earlier than standard methods such as cytology or cystoscopy. 78 While ctDNA holds significant clinical promise, the high cost associated with sequencing technologies is a barrier to its widespread use. By contrast, focusing on specific molecules within uEVs will offer a more stable and potentially cost-effective approach to biomarker discovery. Recently, Woo et al. evaluated whether the ratio of PD-L1 present on the membrane of uEVs to the conventional EV protein Alix can predict treatment efficacy for Tecentriq, an anti-PD-L1 antibody). 79 Although no cutoff values were established due to the limited sample size, the study reported distinct PD-L1/Alix ratio behaviors between responders and non-responders, highlighting the potential of PD-L1 on uEV as a biomarker for predicting treatment response.
A recent study of prostate cancer showed that DNA and RNA within EVs can provide valuable insights into disease status and could serve as high-quality biomarkers. 80 In UC, one study extracted DNA from the urine of 78 patients with UTUC and found that 82.2% had genetic alterations identical to those in tumor tissue. 81 We have summarized the presence of various RNAs encapsulated within uEVs. These raise important questions about the potential of uEVs, particularly whether the nucleic acids contained within these vesicles could offer comparable diagnostic value and aid in molecular subtype classification for UC. To date, only a few studies have explored the clinical relevance of uEVs in predicting BCa subtypes. Notably, Dong et al. investigated whether EVs accurately reflect the molecular subtypes of BCa. 82 Their research involved whole transcriptome RNA sequencing of formalin-fixed paraffin-embedded (FFPE) tumor tissues, alongside EVs isolated from both tissue explants and urine samples from patients with BCa. They found a strong correlation between molecular subtype classifications from FFPE tumor tissue and tissue-derived EVs. Notably, all uEVs were consistently classified as the luminal subtype, implying that uEVs may not capture the full spectrum of subtype-specific features compared to tissue-derived EVs, which might be more reflective of disease-specific biological processes. Based on these findings, the authors recommended modifications to the current algorithm for EV extraction and expression evaluation. Although significant challenges remain in the clinical implementation of uEVs, sustained research efforts hold the potential to uncover clinically effective biomarkers for UC. With continued advances in technology and collaborative research, uEVs will likely play a critical role in the personalized management of UC in the near future, offering clinicians novel tools for diagnosis, prognosis, and treatment.
This review summarized the current status of uEVs in UC. While the future of uEV research in UC is promising, overcoming existing hurdles will be essential for successful clinical translation. Continued research, technological innovation, and multidisciplinary collaboration are crucial to unlocking the full potential of uEVs, paving the way for personalized and precise management of UC in clinical practice
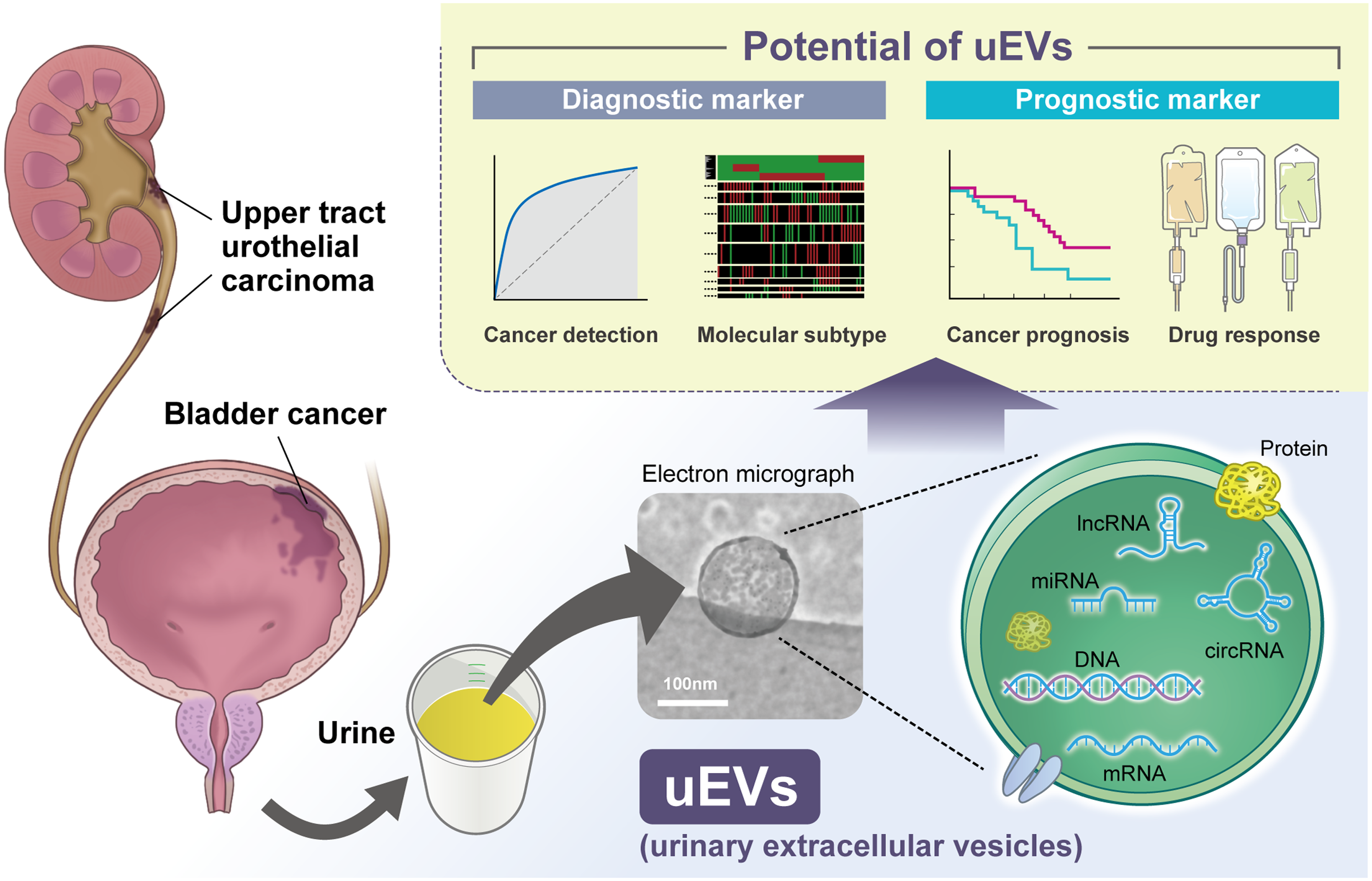
Clinical application of urinary extracellular vesicles in urothelial carcinoma management. Urinary extracellular vesicles (EVs) are emerging as valuable clinical biomarkers for urothelial carcinoma. EV-associated biomolecules, such as nucleic acids (miRNA, lncRNA, circRNA, mRNA, and DNA) and proteins, have been identified. Targeting these molecules holds promise for their clinical application as diagnostic and prognostic markers, paving the way for improved patient management. EVs, extracellular vesicles; miRNA, microRNA; lncRNA, long non-coding RNAs; circRNA, circular RNA; mRNA, messenger RNA.
Supplemental Material
sj-docx-1-blc-10.1177_23523735251395361 - Supplemental material for Urothelial carcinoma and the potential clinical application of urinary extracellular vesicles: Current Status and prospects
Supplemental material, sj-docx-1-blc-10.1177_23523735251395361 for Urothelial carcinoma and the potential clinical application of urinary extracellular vesicles: Current Status and prospects by Eisuke Tomiyama, Fumihiko Urabe, Kazutoshi Fujita, Takahiro Kimura, Norio Nonomura and Peter C Black in Bladder Cancer
Footnotes
Acknowledgments
The authors have no acknowledgements
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Translational Research Grant of Urological Oncology provided by Japanese Urological Association.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Peter C Black serves as an Editorial Board Member for Bladder Cancer and a corresponding author of this article. The other authors have no conflict of interest to report.
Author contributions
ET: Performance of work, interpretation of data, writing the article.
FU: Performance of work, interpretation of data, writing the article.
KF, TK, NN: Supervision of the article.
PB: Project administration, writing – review and editing
All authors have access to the data presented in the study
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
