Abstract
Background
Prolonged hospital stay could lead to increased hospital-acquired infections, and unnecessary utilization of hospital beds, medications, and other resources. However, there is limited evidence regarding the length of hospital stay (LOS) and predictors of prolonged hospital stay in pediatric patients with severe pneumonia. Therefore, this study was conducted to fill the information gap on length of stay and predictors of prolonged hospital stay among pediatric patients with severe pneumonia, in southwest Ethiopia.
Objective
This study aimed to determine the LOS and predictors of prolonged hospital stay among pediatric patients with severe pneumonia, Southwest Ethiopia/2022.
Methods and Materials
A Prospective follow-up study was conducted on pediatric patients with severe pneumonia. Data were entered into Epi-data manager Version 4.4.2.1, for coding, editing, and cleaning, then exported to Stata Version 16 for analysis. Bivariate logistic regression analysis at a significance level of 0.25 and multivariate logistic regression analyses with a significance level of 0.05 were conducted to determine the factors associated with prolonged hospital stay among pediatric patients.
Results
In this study, the median LOS was 5 days and, approximately 38.22% (95% CI [33.66-43.01]) of patients with severe pneumonia had prolonged hospital stays. The presence of underlying comorbidity (adjusted odds ratio [AOR]: 2.64, 95% CI [1.65-4.26]), health insurance status (AOR: 2.22, 95% CI [1.4-3.55]), and incomplete vaccination status (AOR: 4.20, 95% CI [1.04-16.61]) were independent predictors of prolonged hospital stay among pediatric patients with severe pneumonia.
Conclusion
In this study, more than one-third of pediatric patients with severe pneumonia had a length of stay of more than 5 days, and incomplete vaccination status, insurance status, and underlying comorbidities were independent predictors of prolonged hospital stay. Therefore, healthcare providers, parents, and other stakeholders should work to improve the pneumococcal vaccination rate, timely initiation of advanced diagnosis, and patient management of comorbid diseases to reduce hospital stays for pediatric patients with severe pneumonia.
Keywords
Background
The World Health Organization (WHO) defines severe pneumonia as an infectious disease diagnosed in children with a history of cough or reported breathing difficulty and at least one of the following: low oxygen saturation severe respiratory distress, general danger signs, and age-specific increased respiratory rate.1,2
Pneumonia is associated with high health costs of hospitalization and death worldwide. 3 Globally pneumonia in children accounts for approximately 10 to 20 million hospitalizations with an annual incidence of 150 to 156 million cases, 4 and each year, almost 700 000 children die from pneumonia; every minute 2 children die from pneumonia: out of these, 80% of mortality occurs in low and middle-income countries including, Ethiopia. 5
There is wide variation in clinical practice in the treatment of severe pneumonia, which leads to different in-patient hospital stays among patients. Caring for a child with pneumonia in the case of prolonged hospitalization may be financially burdensome in resource-poor areas where parents need to pay for treatment at the point of care.6,7 Medical costs (direct and indirect) of care show a significant difference based on how long a child is hospitalized and depending on the charge for different conditions, and the total medical costs directly related to the costs of hospital admission and length of stay.8–10 Prolonged hospitalization for severe pneumonia exposes the patients to the development of adverse drug reactions and the chance of another hospital-acquired infection, takes time away from work for parents, and is a burden on time expenditure by clinical staff.11,12 Aside from case fatality, length of hospitalization is a commonly accepted indicator of pneumonia severity.7,13
Previous studies have shown that patients who stay longer in hospitals have poor and unfavorable treatment outcomes.6,14 Treatment outcomes and the length of stay for hospitalized pneumonia patients until recovery are influenced by various factors such as comorbidities, complications, severity, and type of drug prescribed.15,16 A study conducted in, Gambia revealed that approximately 25.0% of the children with pneumonia had a prolonged hospital stay while malnutrition severe wasting, and hypoxemia at presentation were independent predictors. 7
In Ethiopia, trials are underway to prevent the onset of pneumonia in children such as pneumococcal conjugate vaccines (PCVs), develop integrated treatment strategies for newborn and childhood illnesses to treat pediatric patients, and reduce morbidity and mortality. The Ethiopian 2015 health sector transformation planned to reduce the mortality rate of children to 31 and 14 per 1000 live births in 2025 and 2035 respectively. 17 Despite these plans, strategies, and initiatives, Ethiopia has a high incidence of pneumonia, a high rate of hospitalization, and a higher risk of mortality, and is categorized among the top five nations with the highest number of pneumonia deaths. 18
A retrospective follow-up study conducted in Benshangulgumuze, Ethiopia revealed that approximately 28.93% of the children with pneumonia had a prolonged hospital stay(>5 days) while having facility referral, nutritional status of wasting, anemia, no identified hemoglobin level and no identified blood film presentation were independent predictors. 19
Although studies have been conducted in different hospitals in Ethiopia on the time to recovery and mortality rate of pneumonia, there is not enough evidence on the length of hospital stay (LOS) and predictors of prolonged hospitalization among pediatric patients with severe pneumonia. Therefore, this study aimed to determine the LOS and predictors of prolonged hospitalization among pediatric patients with severe pneumonia admitted to public hospitals, in Southwest Ethiopia to provide evidence on length of stay and predictors of prolonged hospital stay among pediatric patients with severe pneumonia.
Methods and Materials
Study Design
An institution-based prospective follow-up study was conducted among pediatric patients with severe pneumonia admitted to public hospitals in the southwest region, of Ethiopia.
Study Area and Period
This study was conducted in public Hospitals, in the Southwest region, of Ethiopia, which is a newly established region. There are eight well-known public Hospitals: Out Of the eight hospitals; MizanTepi University Teaching Hospital (MTUTH), G/TsadikShawo General Hospital, Tepi General Hospital, and Wacha Primary Hospital were selected using a lottery method. The MTUTH is located 584 km from Addis Ababa. It served as a referral center. TheTepi General Hospital is located 622 km from Addis Ababa. The G/tsaddikShewa General Hospital is located in the Keffa zone Bongatown, which is 464 km from Addis Ababa, and the Wacha primary hospital is located in Wacha-maji town. All Hospitals provided outpatient care and inpatient services for pediatric problems including severe pneumonia. The study was conducted from June 1 to November 30, 2022, in 4 selected public hospitals.
Study Participants
All pediatric patients (aged 1 month to 15) years with severe pneumonia admitted to public hospitals, in Southwest Ethiopia were the source population. All sampled and selected pediatric patients with severe pneumonia admitted to selected public hospitals, in Southwest Ethiopia during the data collection period were the study population.
Eligibility Criteria
All pediatric patients with severe pneumonia who were in the age categories of 1 month to 15 years were eligible for participation, while pediatric patients with severe pneumonia who were below 1 month and above 15 years, readmitted patients and patients with incomplete observation during follow-up; and unknown dates of admission and discharge were excluded.
Sample Size Determination, Sampling Technique, and Procedure
The sample size was determined by using a single population proportion formula considering the following assumptions; 95% confidence level, margin of error (0.05), and proportion (P) of prolonged hospitalization for pneumonia as 50%, since there is no previous study in Ethiopia the sample was calculated as follows:
n = (Zα/2)2 × p (1-p)/(d)2
n = (1.96)2 × 0.5 × (1 − 0.5)/(0.05)2 = 384: After adding a nonresponse rate of 10% (19.2 ≈ 20), the final sample size was
Sample size distribution to each hospital was conducted using the formula:
Finally, 67 children from Wacha Primary Hospital, 114 from G/tsaddik Shewa, 118 from Tepi General Hospital, and 123 from MTUTH were obtained from 422 pediatric patients with severe pneumonia. After allocation, the sampling interval (kth) was calculated as the total number of children admitted with severe pneumonia divided by the sample size (791/420 = 1.8∼2). Then, at every interval, participants were selected after the first eligible patients were selected by the lottery method on the planned date of data collection.
All the selected respondent's medical record number was recorded in one single sheet to remove redundancy if the child came for readmission during our data collection period.
Among the 422 sampled patients with severe pneumonia, 6 patients were lost to follow-up, withdrew, and were referred to another hospital. Data from 416 patients with severe pneumonia were included.
Study Variables
Dependent variables: Prolonged hospital stay among severe pneumonia patients.
Independent variables: include the following subheadings.
✓ ✓ ✓ ✓
Operational Definitions
Prolonged Hospital Stay
A prolonged LOS was considered an outcome variable in the study, defined as the time from hospital admission to hospital discharge. It was dichotomized as ≤5 and >5 days based on the median time (length) of hospital stay. Patients who stayed in the hospital for more than five days were categorized as having “prolonged” hospitalization.7,19
Severe Pneumonia
Patients were diagnosed with severe pneumonia by pediatricians working in the pediatric emergency rooms of each hospital using standardized baseline assessment methods such as medical history, clinical examination, laboratory tests, and radiography.
Vaccination Status
Fully vaccinated is defined as being vaccinated according to the Ethiopian EPI guidelines, and partially immunized (vaccinated) is defined as not being vaccinated according to the Ethiopian EPI guidelines in the absence of certain vaccinations. 20
Comorbidity
Any disease condition (acute or chronic) present at admission in addition to severe pneumonia which includes childhood asthma, HIV, anemia, meningitis, bronchitis, and urinary tract infection. 21
Time Scale
The LOS was measured in days.
Starting Time Origin
The first day of admission of children at the pediatric ward or if the child acquired severe pneumonia in the hospital, the diagnosis date of severe pneumonia was considered as the first day of admission.
End of Follow-Up/Ending Time
Specific time of discharge from the hospital.
Follow-Up Time
This is the duration of time from admission to the hospital from severe pneumonia to discharge from the hospital.
Data Collection Tools and Procedures
The data collection tool was adapted and modified after reviewing other related literature,3,7,22,23 and prepared in English and translated into a local language, then retranslated back into English. Data were collected through face-to-face interviews with mothers and day-to-day patient follow-ups using medical charts and vital sign sheets. Two BSc nurses who had previous data collection experience were collecting the data, and 1 MSc nurse supervisor was assigned in each hospital to supervise the data collection process.
Data Processing and Analysis
Data were entered into Epi-data Manager Version 4.4.2.1, for coding, editing, and cleaning, then exported to Stata Version 16 for analysis. Predictors of a prolonged hospital stay were identified using a logistic regression model. The logistic regression model of the fitness test was assessed using the Hosmer and the Lemeshow test, and the model was fitted. Variables with a P-value < .25 in the bivariate logistic regression analysis were transferred to the multivariable logistic regression analysis. Then, variables with a P-value < .05 at a 95% confidence level in multivariate analysis were considered independent predictors of a prolonged hospital stay.
Data Quality Assurance
A pretest was conducted on 5% of the sample size before the actual data collection period then editions, wordings, and sequencing of questions were made on the data collection tool. One day of training was provided to the data collectors and supervisors on the techniques, objectives of data collection, and ethical research issues. Supervisors regularly checked the completed questionnaires and provided immediate feedback to data collectors.
Patient and Public Involvement
There was no patient/public involvement in the development of the research question or the design, conduct, reporting, implementation, or dissemination plans or evaluation of the study.
Results
From the total sample of 422 children diagnosed with severe pneumonia, only 416 completed the follow-up during the study period and gave a response rate of 98.58%.
Sociodemographic Characteristics of Pediatric Patients and Their Mothers
In this study, more than half (55.3%) of pediatric patients with severe pneumonia were male, and 246 (59.1%) pediatric patients were found in the 1 to 5 years age categories. most of the participants (57.5%) of pediatric patients were from urban residences (Table 1).
Sociodemographic Characteristics of Pediatric Patients With Severe Pneumonia Admitted In Public Hospitals, Southwest, Ethiopia, 2022/2023 (N = 416).

Nutritional Status of the Pediatric Patients
In this study, the majority 77.6% of pediatric patients with severe pneumonia had a history of exclusive breast-feeding. More than 90% of pediatric patients with severe pneumonia had normal nutritional status, whereas underweight, stunting, and wasting were in 38 (9.1%), 30 (7.2%), 34 (8.2%) pediatric patients with severe pneumonia respectively (Table 2).
Nutritional Status of Pediatric Patients With Severe Pneumonia Admitted in Public Hospitals, Southwest, Ethiopia, 2022/2023 (N = 416).

Clinical Characteristics of Pediatric Patients With Severe Pneumonia
In this study, one 191 (45.9%) pediatric patients with severe pneumonia had hypoxia during hospital admission, and 391 (94%) patients had danger signs during admission. Two hundred sixty-two (63.0%), 141(33.9%), and 71(17.1%) patients had respiratory distress signs, high-grade fever, and altered consciousness respectively (Table 3).
Clinical Characteristics of Pediatric Patients With Severe Pneumonia Admitted In Public Hospitals, Southwest, Ethiopia, 2022/2023 (N = 416).

Therapeutic/Interventional Characteristics of Patients With Severe Pneumonia
In this study, the majority (67.1%) of the pediatric patients with severe pneumonia had a history of taking antibiotics before admission to the hospital, and most of them were taking Ceftriaxone. Antibiotics had changed after admission in the hospital for 206 (949.5%) of pediatric patients. More than half of the children and their parents (54.6%) were not insured (did not have community health insurance), and only 150 (36.1%) pediatric patients had completed their recommended vaccination appropriate to their age (Table 4).
Therapeutic Characteristics of Pediatric Patients With Severe Pneumonia Admitted in Public Hospitals, Southwest, Ethiopia, 2022/2023 (N = 416).

Pediatric Comorbidity Among Pediatric Patients With Severe Pneumonia
In this study, approximately 15.1% of patients with severe pneumonia had other co-morbidities in addition to having severe pneumonia, and HIV infection, asthma, and malaria account for 3.4%, 4.3%, and 2.2% of comorbidities respectively (Table 5).
Pediatrics Comorbidity Characteristics of Pediatric Patients With Severe Pneumonia Admitted In Public Hospitals, Southwest, Ethiopia, 2022/2023 (N = 416).
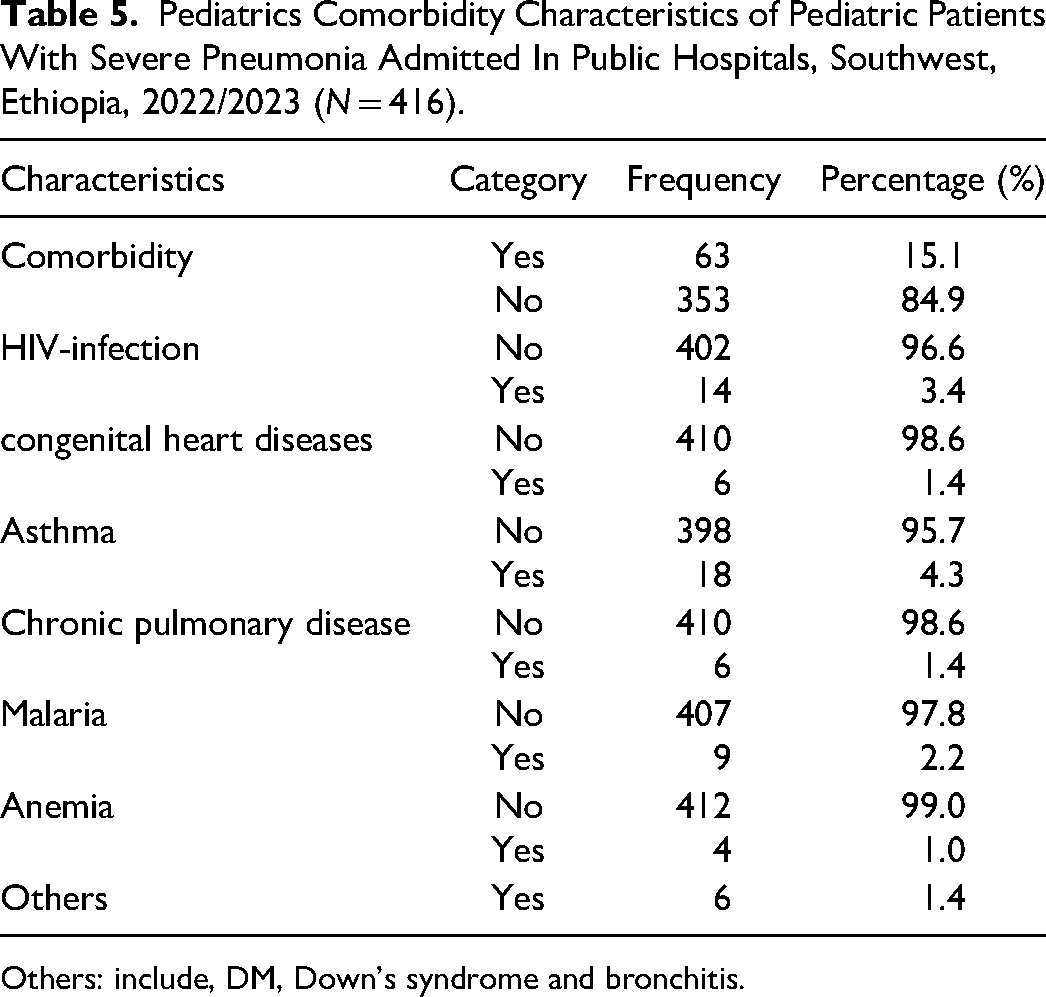
Others: include, DM, Down's syndrome and bronchitis.
Lengths of Hospital Stay Among Pediatric Patients With Severe Pneumonia
In this study, the median time of hospitalization was 5 days with an interquartile range (IQR) of 4 to 7 days. The majority of patients with severe pneumonia, 61.78% (95% CI [56.99-66.34]) had stayed in the hospital for less than five days, and approximately 38.22% (95% CI [33.66-43.01]) severe pneumonia patients were hospitalized for more than five days of duration (Figure 1).

Length of hospitalization among pediatric patients with severe pneumonia admitted in public hospitals, in southwest Ethiopia (N = 416).
Predictors of Prolonged Hospitalization
During the bivariate logistic regression analysis, the independent variables with a significance level of < .25 were transferred to multivariate logistic regression analysis, and variables with a significance level of 0.05 during multivariate logistic analysis were considered as predictors of prolonged hospital stay.
In this study, independent variables such as residency, nutritional status (WFA, WFH, and HFA), signs of respiratory distress, convulsion, comorbidity, history of previous respiratory infection, vaccination status, and insurance were significant in the bivariate logistic regression analysis. Finally, vaccination status (incomplete vaccination), insurance status (not insured), and having comorbidity diseases, were independent predictors of prolonged hospital stay in pediatric patients with severe pneumonia. Pediatric patients with severe pneumonia and comorbidity (additional disease) had 2.64 times higher odds of prolonged hospital stay than those who were only diagnosed with severe pneumonia (adjusted odds ratio [AOR]: 2.64, 95% CI [1.65-4.26]).
Additionally, in this study severe pneumonia pediatric patients who were insured with community health insurance had 2.22 times longer odds of hospitalization than those who were not insured with community health insurance (AOR: 2.22, 95% CI [1.4-3.55]). Severe pneumonia pediatric patients who did not complete the recommended vaccination had 4.2 times higher odds of hospitalization than pediatric patients with severe pneumonia who had completed their recommended vaccination (AOR: 4.20 (95% CI [1.04-16.61]) (Table 6).
Predictors of Prolonged Hospitalization Among Pediatric Patients With Severe Pneumonia Admitted in Public Hospitals, Southwest Ethiopia (N = 416).

Note: *, significant at P < .05:1.00, considered as reference categories; COR-crude odds ratio and AOR-adjusted odds ratios.
Discussion
This prospective follow-up study was conducted to determine the LOS and to identify the predictors of prolonged hospitalization among pediatric patients with severe pneumonia.
In this study, the median time of hospitalization was 5 days with an IQR of 4 to 7, which was similar to the results from previous studies conducted in Benshangulgumze, Ethiopia, 19 and comprehensive health at Masse Major Health center, Gambia, 7 Where the median hospital stay was 5 and 4.5 days respectively.
However, the LOS among pediatric patients with severe pneumonia in this study was lower than that in previous studies conducted in Japanese health centers, 22 and hospitals in Switzerland, 24 where the LOS was 9.8 days and 11 (IQR, 8-17) days respectively. A possible reason for these differences might be variations in the level of quality of care, sample size difference, sociodemographic, and other parameter variations among studies.
The variation in the study conducted in Japan might be because of the varying methods of data collection using different sources of data (primary/secondary). This study used primary data sources during follow-up and had data that were more accurate and reliable regarding the specific/actual time of admission and discharge of patients. However, the Japanese study used secondary data/care review, which makes it difficult to obtain actual data on their charts; therefore, the actual LOS of patients might be overreported.
The study showed that there was a significantly high level of prolonged hospitalization 38.22% (95% CI [33.66-43.01]) in pediatric patients with severe pneumonia. This was higher than the studies conducted at Benshangulgumze, Ethiopia, 19 and at Basse Major Health Center, Gambia, 7 where 28.93% and 25.0% of the patients had a prolonged hospital stay respectively. however, this was lower than the study conducted on patients community community-acquired pneumonia in a teaching hospital in Valencia (Spain) 25 where prolonged hospitalization was recorded in the other 68% of patients.
The possible reason for this difference with the study conducted at Benshangulgumze, Ethiopia
19
might be due to variations in the quality of care provided during hospitalization and the source of data
The variation in the study conducted at Gambia, and Valencia (Spain) 25 also might be because of varying methods of clinical practice and contextual factors such as laboratory equipment and sociodemographic differences of the population served between countries.
In this study, the prolonged hospitalization of patients with severe pneumonia was multifactorial, and vaccination status (incomplete vaccination), insurance status (not insured), and comorbidities were independent predictors of prolonged hospital stay.
Patients with severe pneumonia and comorbidities (having additional diseases) had 2.64 times higher odds of prolonged hospital stay than those who were only diagnosed with severe pneumonia, which is supported by other studies.3,26–28 This might be because the occurrence of one or more underlying comorbid diseases is associated not only with a greater risk of developing pneumonia but also worsens pneumonia prognosis, increases hospital stays, and may lead to patient death. However, this is open to future research to understand the impact of co-morbidities on the prognosis of severe pneumonia using clinical and diagnostic variables, and severity index scores in patients with severe pneumonia.
Patients with community health insurance had 2.22 times longer odds of hospitalization than those without community health insurance. This was supported by previous studies in the United States, California, 29 Washington DC, 30 and China, 31 whichis an expected result because patients who had insurance and the ability to pay out-of-pocket inpatient care had good health-seeking behavior and waited until the recommended discharge time, which affects the LOS. However, this result was in contrast to a study conducted in Uganda 32 which revealed that insured patients had a significantly shorter length of stay. However, this is open to further rigorous study to identify the effect of insurance on pediatric hospital stay and to differentiate the length of stay among the insured and not insured to solidify the evidence guiding future policy development in Ethiopia.
Pediatric patients with severe pneumonia who did not complete the recommended vaccination had 4.2 times higher odds of hospitalization than those who had completed their recommended vaccination for age, which is similar to the results of a study conducted in the Philippines. 33 This might be because there is an increased severity of pneumonia and other comorbidities in pediatric patients who have been partially or not vaccinated completely, and the effect of PCV on the reduction in the pneumonia hospitalization burden. 34 However, data to determine the contribution of PCV to the prognosis of pneumonia are missing. Therefore, the relationship between PCV and length of hospitalization for severe pneumonia requires further in-depth research. The study was conducted using prospective follow-up and primary data from multicenter public hospitals, which increased data accuracy and reliability. However, this study had some limitations. This study used a relatively small sample size; therefore, it is difficult to generalize the results to all pediatric patients with severe pneumonia, as this study was limited to patients admitted to public hospitals and it cannot be generalized to pediatric patients with severe pneumonia admitted to private health institutions.
Conclusion
This study indicates that approximately more than one-third of a pediatric patient with severe pneumonia had prolonged hospital stays of more than 5 days, and incomplete vaccination status, insurance status, having underlying comorbidities were independent predictors of prolonged hospital stay to pediatric patients with severe pneumonia. Therefore, advanced and special care should be given to pediatric patients with severe pneumonia with a special focus on patients with underlying comorbidity, and who did not complete the recommended vaccination for age during their hospital stay. This study may be helpful to healthcare providers and policymakers in developing strategies for the advanced management of patients with severe pneumonia at a higher risk of prolonged length of stay.
Footnotes
Acknowledgments
Our thanks also go to Mizan-Tepi University Teaching Hospital (MTUTH), Tepi General Hospital, G/tsaddik Shewa General Hospital, and Wacha primary hospital staff of the pediatrics ward, our appreciation extended to the study participants, data collectors, and supervisors.
Author Contributions
All authors made a significant contribution to the work reported; whether that is in the conception, study design, execution, acquisition of data, analysis, and interpretation; or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Before data collection, ethical clearance was obtained from the Mizan-Tepi University ethical review board with reference number HSC/00103/2014, and a formal letter of cooperation was written for each public hospital. After receiving an explanation of the objectives of the study, confidentiality, and benefits of the study, all parents gave informed consent for the participation of the pediatric patients in the study. Additionally, the names of participants were not used in the study. Participants (mothers) were informed that they had the right to participate in the study as well as to interrupt/withdraw/at any time during follow-up.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
