Abstract
Background:
Cell-free fat extract (CEFFE) has shown promising therapeutic effects on osteoarthritis (OA) in preclinical studies, but its clinical role remains unclear.
Purpose:
To evaluate the safety and efficacy of intra-articular autologous CEFFE injections for symptomatic early- and mid-stage knee OA.
Study Design:
Randomized controlled trial; Level of evidence, 2.
Methods:
A total of 24 participants were randomly allocated to 1 of 4 groups to receive 5 weekly injections of (1) hyaluronic acid (HA, active comparator), (2) low-dose CEFFE (containing 1 mL CEFFE), (3) medium-dose CEFFE (containing 1.5 mL CEFFE), or (4) high-dose CEFFE (containing 2 mL CEFFE). Autologous CEFFE was prepared by spin, emulsification, and filtration after abdominal liposuction, effectively removing lipid and cellular components. Information regarding adverse effects was collected. Routine blood tests, C-reactive protein levels, liver function tests, and kidney function tests were performed at baseline and at the 24-week follow-up to assess systemic toxic effects of CEFFE injection. The clinical effectiveness of CEFFE injections was evaluated using the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) and visual analog scale (VAS) scores. Magnetic resonance imaging (MRI) was used to detect changes in bone marrow lesions (BMLs) and cartilage loss at baseline and at the final follow-up.
Results:
All patients received the corresponding injections and were followed up for 24 weeks without serious adverse events or clinically significant laboratory abnormalities. At the final follow-up, the mean WOMAC pain subscale score was 5 ± 1.3 in the HA group, compared with 1.5 ± 1.5, 1.7 ± 1, and 1.8 ± 1.7 in the low-, medium-, and high-dose CEFFE groups, respectively. All 3 CEFFE doses were more effective than an intra-articular HA injection for pain relief and recovery of knee function, as assessed by WOMAC and VAS scores. Semi-quantitative MRI analysis indicated greater improvements in BML in CEFFE-treated participants (57.1%) than in controls (16.7%). None of the research groups exhibited a significant improvement in cartilage loss compared with baseline.
Conclusion:
CEFFE is an alternative biosafety method for the biological treatment of knee OA, with symptom improvement and BML reduction, and has promising potential for clinical application.
Knee osteoarthritis (OA) is a major cause of disability that affects millions of older adults worldwide.8,21,27 Although arthroplasty is the gold-standard treatment for end-stage knee OA, the optimal treatment approach for early to mid-stage knee OA remains unknown. Traditionally, nonsurgical options—including lifestyle modifications, physical therapy, use of nonsteroidal anti-inflammatory drugs, and corticosteroid injections—primarily aim to alleviate pain and reduce synovial inflammation. 29 However, these nonsurgical methods always fail to attenuate knee OA progression, and their long-term use is associated with adverse effects. These limitations have spurred a growing interest in regenerative medicine approaches that seek not only to alleviate joint pain but also to restore cartilage and subchondral bone structure.
In recent years, with ongoing efforts by orthopaedists and scientists, many biological therapies have been applied in clinical settings. 7 Platelet-rich plasma (PRP) has been widely promoted and is considered a regular conservative approach for knee OA despite controversy regarding its efficacy.1,26 In addition, mesenchymal stem cells (MSCs) have emerged as a promising therapeutic approach for knee OA, owing to their unique multipotency and tissue-regenerative capacity.13,33 However, the lack of standardized preparation and production protocols has limited cell-based therapies to small-scale applications, which have shown varied efficacy.4,9,36 The development and clinical integration of cell-based products have been substantially constrained by heterogeneous regulatory frameworks across different geographic regions, further hindering their consistency and accessibility. Furthermore, the high cost of cell-based therapies and logistical challenges remain significant barriers to their widespread clinical adoption. Therefore, cost-effective biological therapies for early- and mid-stage knee OA remain elusive.
Adipose tissue is a vital resource in regenerative medicine, as it is enriched with adipose-derived stem cells, growth factors, and exosomes, offering unparalleled potential for tissue repair, immunomodulation, and anti-inflammatory therapies. In previous studies, we have developed the cell-free fat extract (CEFFE) from adipose tissue. Through physical methods, such as centrifugation, emulsification, and filtration, CEFFE effectively purifies bioactive proteins from adipose tissue while preserving their biological activity. 18 CEFFE is rich in various growth factors—including insulin-like growth factor-1, transforming growth factor-β, and vascular endothelial growth factor. 35 Owing to its cell-free design, CEFFE offers several potential advantages for clinical use. The absence of intact cells simplifies storage requirements and enables a single extraction to support multiple injections, aligning with current requirements for knee OA treatment. The nonimmunogenic nature of CEFFE theoretically permits allogeneic injection, thereby increasing donor availability and reducing treatment costs. Finally, the cell-free formulation eliminates concerns regarding postinjection cell fate and behavior, thereby ensuring enhanced safety and predictability in clinical applications. In our previous studies, CEFFE promoted cartilage structure restoration and reduced synovial inflammation in animal models with monosodium iodoacetate-induced knee OA, highlighting its promising clinical potential.16,17
Therefore, this study aimed to evaluate the safety and efficacy of intra-articular autologous CEFFE injections in symptomatic early- and mid-stage knee OA. We hypothesized that autologous CEFFE is a safe clinical strategy for knee OA that provides superior outcomes at 24 weeks compared with hyaluronic acid (HA).
Methods
Study Design and Participants
This was a single-center, patient-blinded, positive-control, exploratory, randomized clinical trial. The procedure used in this study is shown in Figure 1A. Before enrollment, the trial was registered at www.chictr.org.cn (ChiCTR2100051039) and approved by the institutional review board of our institution. Patients were recruited from the hospital's outpatient orthopaedic clinics. Written informed consent was obtained from all patients before participation. The study was performed in accordance with CONSORT (Consolidated Standards of Reporting Trials) guidelines and ethical principles of the Declaration of Helsinki.2,34

(A) Schematic overview of the present clinical trial. (B) Schematic illustration of the preparation of autologous CEFFE. CEFFE, cell-free fat extract; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
The eligibility criteria were as follows: (1) age 20 to 80 years; (2) symptomatic Kellgren-Lawrence grades 1 to 3 knee OA with recurrent knee pain over the past month; (3) a body mass index within the range of 18.5 to 30 kg/m2; (4) a Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) pain subscale score between 7 to 17 points at baseline (discontinuation of analgesic medication for at least 48 hours before assessment); (5) discontinuation of all analgesic medication 7 days or 5 half-lives before the first injection, excluding acetaminophen; (6) ability to walk >50 meters without the help of crutches or walking sticks; (7) no pregnancy plans and voluntary adoption of effective contraceptive measures during the trial and within 90 days after the last administration; and (8) voluntary provision of a written informed consent.
The exclusion criteria of this study mainly include (1) infection; (2) secondary OA; (3) malalignment >15°; (4) flexion contracture >5°; (5) surgery planned for the lower extremity during the trial; (6) intra-articular injection within 6 months before the trial; (7) chronic or recent use of oral corticosteroids and glucosamine; (8) contraindications for magnetic resonance imaging (MRI) examination; (9) abnormal liver and renal function; and (9) serious medical conditions (such as cardiovascular events, malignancy, human immunodeficiency virus [HIV] infection, or uncontrolled hypertension) that could be contraindications to participation.
Randomization
Computer-generated randomization was achieved using SPSS software (IBM Corp) and provided to the clinical research center at our hospital. Participants were randomized (1:1:1:1) to receive 5 once-weekly intra-articular injections of: HA, low-dose CEFFE, medium-dose CEFFE, or high-dose CEFFE. Participants were blinded to their group assignment.
CEFFE Preparation
Patients in both HA and CEFFE groups underwent abdominal liposuction and autologous CEFFE preparation as previously described. 35 First, liposuction was performed by a senior plastic surgeon (Z.Y.), and approximately 200 mL of adipose tissue was obtained under local anesthesia with lidocaine in an outpatient operating room. CEFFE was prepared using a physical method. Briefly, adipose tissue was rinsed with saline to remove blood cells (approximately 5-10 minutes). After standing briefly to allow sedimentation (about 2 minutes), the blood and fluid were removed by decantation and degassing. The fat was rinsed with physiological saline to remove blood and debris and then centrifuged at 300g for 3 minutes. The retained fat was emulsified by shifting between two 10-mL syringes connected by a 2- mm 3-way stopcock, repeated approximately 30 passes (about 5-10 minutes) until a uniform white emulsion was formed. The emulsion was centrifuged at 1200g for 5 minutes, and the third layer was collected and filtered through a 0.22-μm membrane. As a result, the final CEFFE product contained no viable cells and was stored at −20°C (Figure 1B). Microbiological testing was performed to confirm sterility. Excluding the liposuction procedure, the entire CEFFE preparation process typically took 40 minutes.
Study Intervention
Intra-articular injection was performed via a superolateral approach with the patient in the supine position, and synovial fluid was aspirated before medication injection. Patients in the control group were treated with 2 mL of HA (20 mg; Shandong Bausch & Lamb Freda Pharmaceutical Co, Ltd). Three CEFFE intervention groups were established based on dose: low-, medium-, and high-dose. To minimize potential confounding effects of injection volume, the total injection volume was fixed at 2 mL across all CEFFE groups. Specifically, the low-dose group received an injection of 1 mL of CEFFE mixed with 1 mL of normal saline. The medium-dose group was administered 1.5 mL of CEFFE and 0.5 mL of normal saline. The high-dose group received 2 mL of CEFFE alone. For patients in the 3 intervention groups, autologous CEFFE was prepared by thawing naturally at room temperature before intra-articular injection. All injections were administered once a week for 5 consecutive weeks.
Toxicity Monitoring and Safety Assessment
Information on adverse effects was collected to evaluate their correlation with intra-articular CEFFE injections. The participants’ vital signs were systematically documented at baseline, before each injection, half an hour after each injection, and at each follow-up. All patients underwent routine blood tests, including C-reactive protein levels, liver function, and kidney function tests, at baseline and at the 24-week follow-up to further evaluate possible systemic toxic effects of CEFFE.
Clinical Outcome Evaluation
The WOMAC Version 3.1 questionnaire—including the pain and function subscales—was administered at baseline and at 6-, 12-, and 24-week follow-ups to assess pain relief and functional recovery after intra-articular injection. 32 The minimal clinically important difference (MCID) for the Pain subscale (range, 0-20) and Function subscale (range, 0-68) are 3.94 and 6.66, respectively. 23 Visual analog scale (VAS) scores with an MCID of 1.4 were collected at baseline and at 6, 12, and 24 weeks. 3
MRI Assessment
MRI assessments were performed on all participants at baseline and at the 24-week follow-up. All data were obtained with the participants in the supine position using a 3 T superconducting MRI scanner (Koninklijke Philips NV) with a dedicated knee coil. A musculoskeletal radiologist (C.L.) performed a semiquantitative evaluation of knee OA using the MRI Osteoarthritis Knee Score (MOAKS). 14 Improvements in bone marrow lesions (BMLs) and cartilage loss were evaluated using MRI, as previously described. 28 Improvement in Hoffa's synovitis and joint effusion was defined as a decrease in the MOAKS score at 24 weeks compared with baseline. 1
Statistical Analysis
Between-group differences in patient characteristics, VAS scores, WOMAC Pain subscale scores, WOMAC Function subscale scores, and WOMAC (total scores) at baseline and at the 24-week follow-up were assessed using 1-way analysis of variance. Statistical significance was set at P < .05 (2-tailed). SPSS Version 23 (IBM Corp) was used for all statistical analyses.
Results
The baseline participant characteristics are presented in Table 1. The 24 participants enrolled in this study were randomized (1:1:1:1) into 4 different research groups. All participants received 5 intra-articular injections and completed the final follow-up.
Patient Characteristics a

Data are presented as mean ± SD or n/n. BMI, body mass index; KL, Kellgren-Lawrence; VAS, visual analog scale; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Safety of CEFFE Intra-articular Injection
No serious adverse events related to CEFFE—such as septic arthritis, infection, and allergic reactions—were reported during intra-articular injection and at the 24-week follow-up. No patient had impaired liver function (defined as aspartate aminotransferase and alanine aminotransferase levels greater than twice the upper limit of normal) or kidney function (defined as serum creatinine >1.5 times the upper limit of normal) at the final visit, according to blood biochemical examination. One patient in the HA group and 1 in the medium-dose CEFFE group reported injection site pain, which resolved spontaneously within 72 hours without intervention.
Clinical Outcome
At the 24-week follow-up, CEFFE injection was significantly more effective than HA injection for pain relief and recovery of knee function (Figure 2 and Table 2). Based on previously reported MCID values, 23 each of the 3 CEFFE groups showed improvement in the WOMAC Pain and Function subscales that exceeded the MCID. However, the mean change in the WOMAC Pain subscale score was less than that in the control group. Consistent with the WOMAC Pain subscale, VAS scores also demonstrated improvement across all CEFFE groups. At the final follow-up, the reductions in VAS scores for all 3 CEFFE groups exceeded the established MCID of 1.4 for knee OA.
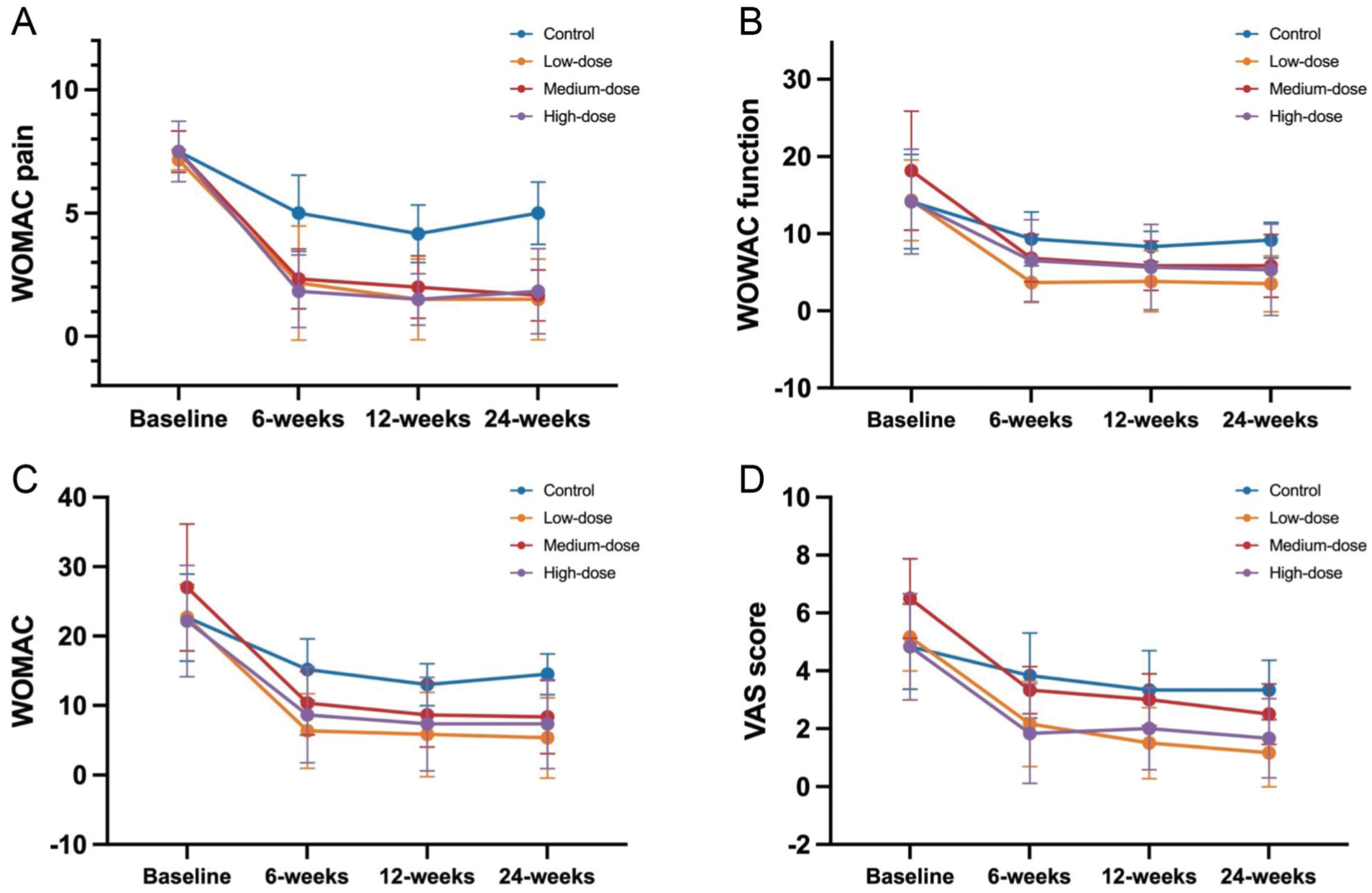
(A) Measurement of the WOMAC Pain subscale. (B) WOMAC Function subscale. (C) WOMAC. (D) VAS over time. VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
Clinical Outcome of Participants at 24-Week Follow-up a

ANOVA, analysis of variance; VAS, visual analog scale; WOMAC, Western Ontario and McMaster Universities Osteoarthritis Index.
MRI Assessment
Baseline and final follow-up MRI scans were available for all patients included in this study. CEEFE injections were not more effective than HA injections in treating cartilage loss, Hoffa's synovitis, and knee effusion (Table 3 and Figure 3).
Baseline Prevalence and Improvement of MOAKS Features at 24-Week Follow-up a

Data are presented as n/N (%). CEFFE, cell-free fat extract; MOAKS, MRI Osteoarthritis Knee Score; MRI, magnetic resonance imaging; BMLs, bone marrow lesions.

(A) MRI of a 63-year-old female patient with left knee pain in the medial tibiofemoral joint. She had BMLs in the medial tibiofemoral joint and received medium-dose CEFFE injection. The patient reported significant pain relief in the left knee after the third injection. The WOMAC Pain subscale decreased from 9 at baseline to 3 at the final follow-up. (B) MRI showed significant improvements in BMLs of the left tibiofemoral joint after 24 weeks.
Discussion
In this prospective, exploratory, and randomized clinical trial, we reported that CEFFE intra-articular injection, given once weekly for 5 consecutive weeks, provided pain relief and improved knee function at 24-week follow-up in patients with early- and mid-stage knee OA. The primary results of this small-sample exploratory trial indicated the translational potential of CEFFE in the treatment of knee OA.
This study had some limitations. The primary limitation of the present study was the relatively small sample size. To the best of our knowledge, the present study is the earliest clinical study to evaluate the therapeutic effect of intra-articular CEFFE injection for knee OA. Therefore, our institution's institutional review board mandated cautious, limited enrollment for this exploratory application. Given the absence of previous clinical data, a formal sample size calculation was also unavailable. This exploratory study helped confirm the safety and preliminary efficacy of CEFFE injection in the treatment of early- and mid-stage knee OA, and the results provide a basis for sample size calculation in future studies. Large-sample, randomized controlled trials are necessary to clarify the findings of the present study. Second, the total follow-up period in the present study was 24 weeks; thus, we could not assess the long-term efficacy of CEFFE injections. Another limitation of this study is that, although originally planned, analysis of synovial inflammatory factors could not be completed. Many patients lacked detectable synovial fluid at baseline. In those with effusion, complete aspiration before injection, combined with subsequent reduction in effusion, left insufficient synovial fluid for reliable cytokine assessment. As a result, we were unable to estimate intra-articular biological responses to CEFFE, which should be determined in future investigations.
Most established biological therapies for knee OA are cell-based, and the potential of MSCs for tissue regeneration is considered a fundamental factor in their treatment. Bone marrow aspirate concentrate, stromal vascular fraction, and umbilical cord tissue MSCs have been injected into patients with symptomatic knee OA.6,15,22,25,31 Despite the reports of clinical efficiency and debate regarding the most beneficial cellular treatment, cell-based strategies have several disadvantages for knee OA treatment. In general, quality control for cellular therapy has not been established, and the lack of standardized manufacturing procedures may account for the heterogeneity in outcomes reported across studies. However, performing protein content and composition analysis for each product batch is a feasible quality control measure. Unlike stromal vascular fraction and microfragmented adipose tissue, which retain viable cells and tissue structures, CEFFE is defined as a cell-free, adipose-derived extract obtained through a purely physical preparation process. The benefit of CEFFE's cell-free design eliminates the requirement for cryoprotectants and ultra-low-temperature storage, and our stability study data16,35 indicate retained bioactivity for at least 6 months at −20°C, enabling long-term preservation for repeat injections. Moreover, the removal of intact nucleated cells during the physical preparation process renders the CEFFE product immunologically inert. While all injections in this trial were autologous, the cell-free design of CEFFE provides a theoretical foundation for safe allogeneic injection in the future, which could significantly improve accessibility and reduce costs. Based on the comprehensive advantages outlined above, we assert that CEFFE represents a more economically viable and readily deployable biotherapeutic approach than cellular therapies and is particularly well-suited for developing nations.
For autologous injections, the pain caused by liposuction under local anesthesia is an important factor affecting the medical experience of the participants, especially in thin male patients who lack abdominal fat. According to our survey, pain caused by liposuction was the main factor preventing patients—even those with excellent clinical outcomes—from choosing autologous CEFFE treatment again. Given the substantial costs and potential complications associated with general anesthesia, minimizing procedural pain during local anesthesia-assisted liposuction is critical for both the patient experience and health care economics. In previous investigations, a minimum adipose tissue volume of 200 mL was required to produce sufficient CEFFE for 5 standardized intra-articular injections. However, reducing the liposuction volume can significantly decrease procedural duration and pain. Therefore, our study design included 3 experimental groups with low, medium, and high CEFFE doses, based on the protein concentration used in a previous animal study. 16 Our primary rationale for this approach was to reduce the volume of adipose tissue removed during liposuction, thereby minimizing invasiveness and enhancing patients’ comfort and overall experience. In this small exploratory cohort, low-dose CEFFE injection appeared to produce clinical improvements similar to those observed in the higher-dose groups. This finding suggests the feasibility of a reduced harvest volume, which may improve patient tolerance and satisfaction during treatment. Further investigation into batch-level cytokine quantification may help refine dose-response interpretation and support the optimization of CEFFE dosing strategies in future studies.
Preclinical studies have demonstrated that CEFFE exhibits significant anti-inflammatory and chondroprotective properties, effectively reducing synovitis and promoting cartilage matrix synthesis in animal models.16,17 Based on the MRI semi-quantitative statistical results, we did not observe the repair effect of CEFFE on human cartilage in the present study. The discrepancy between the cartilage regeneration effects of CEFFE observed in animal models and those in our clinical trial highlights the critical challenges in translating regenerative therapies to knee OA. Several factors are likely to have contributed to this discrepancy. First, the clinical reality of advanced human knee OA presents a more complex, chronic pathological environment distinct from that of animal models of acute injury. 24 Second, the inherently limited proliferative capacity of adult human chondrocytes compared with that of rodent cells may limit the regenerative potential of CEFFE. 5 Finally, we did not quantify cartilage thickness before and after treatment, and semi-quantitative analysis may not have accurately evaluated the repair effect of CEFFE on the cartilage.
Recently, emerging evidence has demonstrated that BMLs are a fundamental component of OA pathology rather than merely an incidental MRI finding.10,11 The presence of BMLs correlates strongly with pain severity and radiographic progression, serving as both a marker of disease activity and a direct contributor to structural deterioration through altered joint biomechanics and bone remodeling.12,30,37 The observed mitigation of BMLs 24 weeks after intra-articular CEFFE injection is a finding of potential significance for OA therapeutics. We must acknowledge that the pathophysiological mechanisms underlying the formation and resolution of BMLs in knee OA remain incompletely understood. Some studies suggest that BMLs can be self-limiting and may improve under certain conditions—such as reduced weightbearing or temporary load modification— even without pharmacological treatment.3,30 In addition, the limited sample size of the present study restricts our ability to establish a definitive association between observed changes in BMLs and CEFFE treatment. Therefore, any apparent improvement in BMLs should be regarded as an exploratory finding rather than a definitive treatment-attributable finding. Importantly, although structural cartilage regeneration was not observed, CEFFE maintained clinically meaningful benefits. This pattern mirrors the clinical experience of patients who received PRP injections reported in previous articles, where symptomatic improvements often occur without significant radiographic cartilage restoration.19,20 Despite the controversy regarding the efficacy of PRP, CEFFE still has practical advantages, notably in storage and its potential for allogeneic injection. These characteristics enable multiple injections and are more consistent with standardized clinical workflows for knee OA injection therapy. Future randomized controlled trial studies are necessary to directly compare the clinical efficacy and cost-effectiveness of CEFFE and PRP in a well-defined population of patients with OA.
Conclusion
The present study demonstrated that the intra-articular autologous CEFFE injection was well tolerated, with no evidence of serious adverse events or abnormally elevated laboratory test results. In addition, our preliminary results indicated that intra-articular CEFFE injection significantly improved symptoms in patients with symptomatic early- and mid-stage knee OA compared with HA.
Footnotes
Final revision submitted January 19, 2026; accepted January 31, 2026.
One or more of the authors has declared the following potential conflict of interest or source of funding: This work was supported by the National Natural Science Foundation of China (12272232 and 82202680), the Science and Technology Commission of Shanghai Municipality (22YF1422900 and 23S31905900), the Natural Science Foundation of Shanghai (20ZR1432000), the Shanghai Collaborative Innovation Program on Regenerative Medicine and Stem Cell Research (2019CXJQ01), the Cross-disciplinary Research Fund of Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine (JYJC202201), the Open Project of Translational Medicine of SJTU (TMSK-2024-121), and the Fundamental Research Funds for the Central Universities (YG2025QNA04 and YG2025LC12).
Ethical approval for this study was obtained from Institutional Review Board of Shanghai Ninth People's Hospital, Shanghai Jiao Tong University School of Medicine (SH9H-2021-T88-6).
