Abstract
Introduction. Bone marrow lesions (BMLs) are MRI-visible subchondral bone alterations, highly correlated with symptoms in the knee. Subchondroplasty (SCP) is able to fill the subchondral defects associated with BMLs using an injectable bone substitute material. The aim of the present study is to evaluate the 12-month outcomes of the SCP in the treatment of symptoms of mild-to-moderate knee osteoarthritis (OA) patients with persistent BMLs of the knee. Materials and Methods. Subjects affected by BMLs of the femoral condyle or tibial plateau that were present for >3 months and not responsive to conservative treatments were enrolled in this prospective multicenter trial. All the patients underwent SCP. Follow-up was conducted at 1, 3, 6 and 12 months. All subjects completed Numerical Rating Scale (NRS) for pain, Knee Injury and Osteoarthritis Outcome (KOOS) score, Euro Quality of life-5 dimensions (EQ-5D) score, and a subject global satisfaction scale. Demographic information of the patients was also collected. Results. A total of 79 patients completed the 12-month follow-up. Statistically significant improvements on all clinical scales were registered from baseline to the 12-month follow-up. No severe adverse events were reported. Four patients were considered failed. A 12-month subgroup analysis was performed to evaluate the possible correlation between all the KOOS subscales and age, gender, number of BMLs, location of BMLs, and Kellgren-Lawrence grade: no statistically significant associations were observed. Conclusion. SCP is a safe and effective procedure for the treatment of symptoms related to persisting BMLs in mild-to-moderate osteoarthritic knees, with a low failure rate up to 12 months’ evaluation.
Introduction
The wide spectrum of pathological alterations caused by knee osteoarthritis (OA) reflects its modern definition as a whole joint disease.1 -3 Indeed, the modified load distribution during the disease progression along with the reduced healing capacity of the subchondral bone could be the etiology of bone marrow lesions (BMLs). 4 Found in up to 66% of symptomatic OA knees, 5 BMLs demonstrate histological characteristics of inflammation, fibrosis, cysts, trabecular thickening, and necrosis, testifying the ongoing degenerative changes of the subchondral bone.6,7 Radiologically, BMLs are characterized by poorly defined low signal intensity compared with the unaffected bone marrow on magnetic resonance (MR) T1-weighted images. 8 T2 images as well as proton density-weighted, fat-suppressed, and short tau inversion recovery images present BML as poorly defined subchondral areas of high signal intensity. 9 After contrast administration, there is evident enhancement of these signal alterations, which is indicative of hypervascularity and repair activity. 10
The presence of BMLs is associated with joint remodeling and increased load forces on the overhanging cartilage, ultimately leading to OA acceleration.11,12
It has been postulated that addressing the BMLs with appropriate treatments can improve patients’ pain and functional limitation,4,9,13 and ultimately delay or avoid arthroplasty procedures. 14
Even though BMLs may spontaneously resolve in certain situations, conservative treatments mainly centered on weightbearing restriction fail in more than one-third of patients. 15 Pharmacotherapy with the use of anti-inflammatories and, more recently, intravenous prostacyclin or bisphosphonates gave variable results.14,16 However, when the symptomatic BMLs persist, surgical options may be considered. Injection of biologic agents 17 directly into the subchondral bone has been suggested as a novel promising technique, but consistent evidence is still limited.18 -20
The treatment with Subchondroplasty® (SCP) involves intraosseous injections of synthetic calcium phosphate (CaP) which differs from other types of treatments because it constitutes an injectable bone substitute material. It aims to fill the void left by the BMLs, providing structural support and promoting the remodeling and regeneration of the surrounding subchondral bone. 21 SCP has been shown to be effective in providing pain relief and functional recovery.22 -25 Studies on the efficacy of SCP are limited by the small number of patients and short follow-up. 26 The aim of the present multicenter prospective clinical trial is to evaluate the safety and clinical efficacy (i.e. pain reduction and functional improvement) of the SCP procedure up to 12-month follow-up.
Material and Methods
Study Design and Patient Selection
The present prospective multicenter study was approved by the Hospital Ethics Committee and institutional Internal Review Board of each center. The trial was registered at clinicaltrials.gov (NCT03430219). The following centers have been involved in the clinical trial: Humanitas Research Hospital (Rozzano, MI, Italy), Universitätsklinikum Regensburg (Regensburg, Germany), Maastricht University Medical Centre+—MUMC+ (Maastricht, The Netherlands), and University Hospital of Vall d’Hebron (Barcelona, Spain). Informed consent for study participation was obtained from each patient. Inclusion criteria were as follows: (1) age ≥18 years old; (2) the presence of one or more BML(s) of the tibial plateau and/or femoral condyle confirmed on T2-weighted fat-suppressed or fat-suppressed proton density MR imaging; (3) concurrent presence of knee OA (Kellgren-Lawrence grade from 1 to 3); (4) persisting pain in the index knee (≥3 months), with a KOOS pain subscale below 70; (5) no significant mal-alignment (i.e. <6° varus or valgus); and (6) failed previous unsuccessful conservative non-surgical management (e.g. partial weightbearing, extracorporeal shockwave therapy). Exclusion criteria were as follows: (1) current acute or chronic infection at the surgical site; (2) MRI evidence of osteonecrosis in the affected compartment; (3) the presence of known systemic disorders or any systemic inflammatory condition (e.g. rheumatoid arthritis); (4) the presence of known metabolic bone disease; (5) patello-femoral joint OA; (6) Kellgren-Lawrence (K-L) grade 0 or 4 OA; and (7) any surgery performed on the index knee within 12 months prior to enrollment. During the screening visit, the patient brought his personal MRI which testified the presence of BMLs. Once the patient was enrolled, an x-ray was requested to assess the K-L grade, and a new MRI performed during the 30 days prior to the procedure to confirm the BML’s persistence.
Product
AccuFill® is a bone substitute material composed of 2 engineered CaP forms (amorphous CaP and dicalcium phosphate dihydrate). When injected, it rapidly interdigitates into the subchondral bone defects and undergoes an isothermic reaction at 37°C (body temperature). This chemical process triggered by sodium bicarbonate releases carbon dioxide during the setting process and forms pores within the material. The result is a nanocrystalline, macroporous, and osteoconductive scaffold with a chemical formulation and a 3-dimensional structure suitable for bone mechanical support and cell-mediated remodeling.
Procedure
All patients were treated by specialized orthopedic centers with a particular expertise in joint preservation surgery. Preoperative MRI was used to plan the access point, the trajectory, and depth of dedicated cannulas for reaching the BMLs. Spinal loco-regional anesthesia was administered prior to procedure. The patient was lying in supine position, and dedicated cannulas were placed intraosseously, under fluoroscopic guidance ( Fig. 1 ), into the BMLs. An appropriate volume of AccuFill® was prepared in the operating room (OR) by using a dedicated kit. The volume chosen was meant to fill the areas of BMLs taking care not to over-pressurize or overfill the defect. Based on previous preliminary experiences, a volume of 2-2.5 mL is sufficient to treat tibial plateau BMLs, whereas 2.5-3 mL is usually required for femoral BMLs. Multiple fluoroscopic images were taken during the intraosseous injection to check for any intra- or extra-articular leakage of the material. AccuFill® has a paste-like consistency at room temperature and it hardens when injected intraosseously in approximately 10-12 minutes: during the hardening process, cannulas were left in situ and then easily removed manually. Arthroscopy was performed after removing the cannulas to check for any microscopic leakage of tricalcium phosphate into the joint space. Any material found was removed from the joint using suction and lavage. No additional procedures were performed during the surgery. Skin portals were then sutured and an elastic bandage applied.
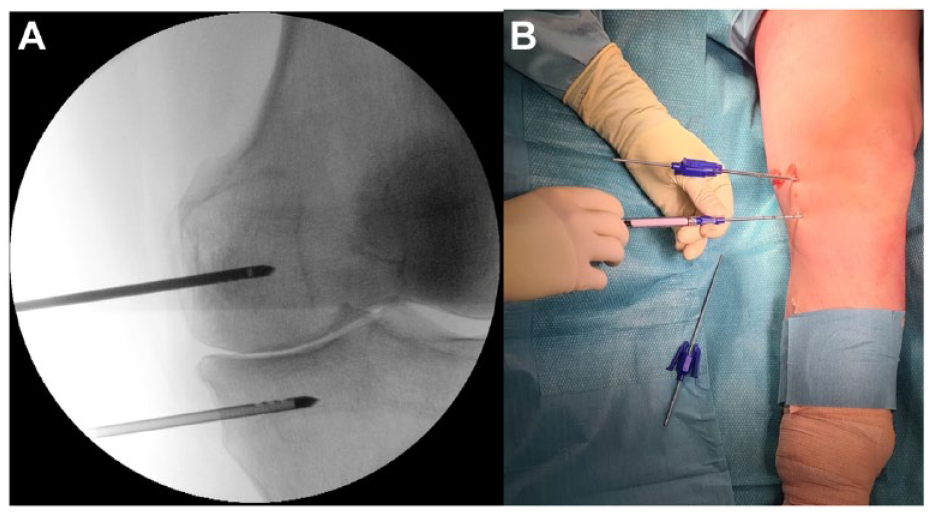
Subchondroplasty procedure, left leg: (
Patients were usually discharged the same day of the procedure, with a postoperative pain management protocol (Paracetamol 1,000 mg 1 tablet 3 times a day for 5 days then upon need + Ibuprofen 600 mg 1 tablet twice a day for 5 days then upon need) and a thromboembolic prophylaxis protocol (low-molecular-weight heparin for 2 weeks). Partial weightbearing was allowed for the first postoperative week and then progressive weightbearing was introduced until patients were able to discontinue crutches (usually after 2 weeks). Gradual range of motion (ROM) recovery exercises were allowed with no restriction and muscular strengthening exercises were suggested for 2 months. High-impact activities were forbidden for 3 months after the procedure.
Patients’ Evaluation
The primary outcome of the present study was the change in pain from baseline to 12-month follow-up, as measured by the Knee Injury and Osteoarthritis Outcome Score (KOOS) 27 pain subscale. Secondary outcomes assessed at 1, 3, 6, and 12 months included the change from baseline in the all KOOS subscales; EQ-5D 28 and Numeric Rating Scale (NRS) 29 for pain and the patients’ global satisfaction. Any adverse event was reported to assess the safety of the procedure. A serious adverse event was defined as an adverse event leading to a death, injury, or permanent impairment to a body structure or a body function. A minor adverse event was defined as the presence of tenderness, redness, and edema of the index knee reported at the first follow-up visit. Treatment failure was defined as any further treatment performed or prescribed (both injective or surgical) on the index knee due to persistent or worsening symptoms in the 12 months after SCP treatment.
Statistical Analysis
Continuous data were expressed as the mean and standard deviation; categorical variables were expressed as the frequency and percentages. The chi-square or Fisher test was applied to analyze categorical variables, while the Student’s t test, log-rank test, and Mann-Whitney test were used with continuous variables. The Pearson’s r test was used to extrapolate a correlation analysis in parametric setting. The Wilcoxon post hoc pairwise comparison was used for assessing group differences of not normally distributed scores. The analysis of variance (ANOVA) test and the Mann-Whitney test were used for continuous and normally distributed scores, respectively. A P-value <0.05 was considered statistically significant, and confidence intervals were constructed at the corresponding 95% level. For the sample size, a literature search on subchondroplasty performed in 2020 revealed one publication 30 that reported KOOS pain as the primary endpoint, the same of the present trial. Authors noted a 28-point improvement in KOOS pain at 1 year of follow-up. Based on this publication, to achieve 90% power and an alpha value = 0.05, assuming an expected change of 25 points from baseline for the KOOS pain subscale, 47 patients were required. Assuming a potential lost-to-follow-up rate of 20%, the minimum number of patients needed was set at 59.
Results
Patients’ Characteristics
A total of 80 patients were evaluated and 79 of them reached the 12 months of follow-up time-point. A total of 43 patients (53.8%) were males. Mean age was 54 ± 16 years and body mass index (BMI) was 28 ± 4.6. Fifty-two patients (65%) presented only 1 BML, and 28 patients had 2 BMLs (35%). The BML location was also assessed. In patients affected by 1 BML, the prevalent location was the medial femoral condyle (48.8%) followed by medial tibial plateau, lateral tibial plateau, and the lateral femoral condyle (26.3%, 15%, and 10%, respectively). A summary of patients’ demographic characteristics is reported in Table 1 .
Summarized Characteristics of Patients Included.

Clinical Scores
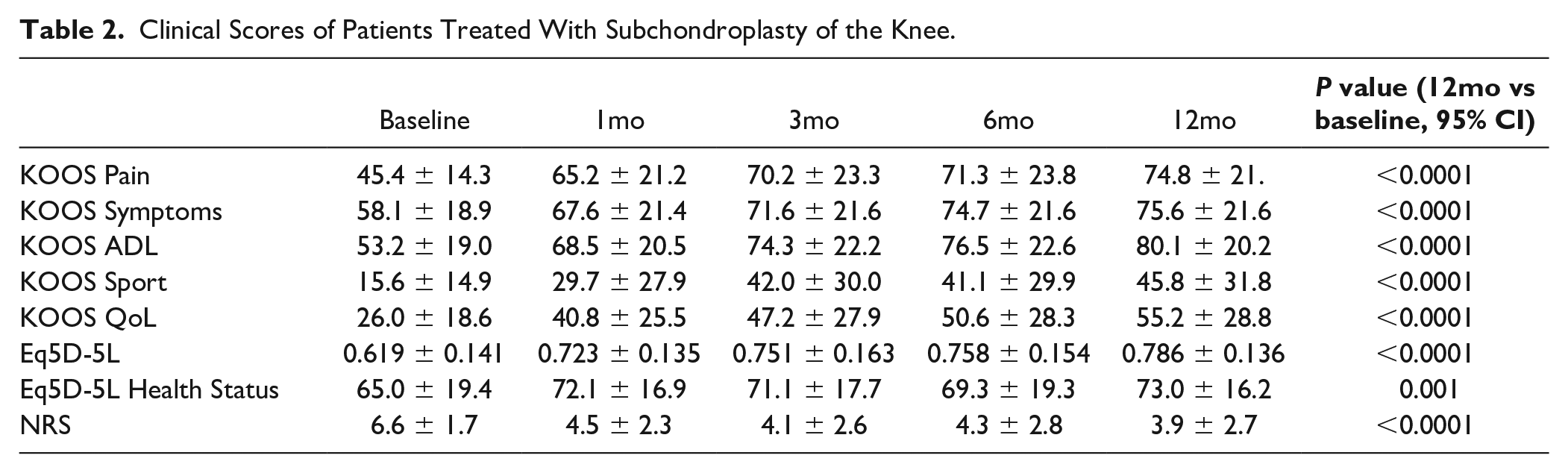
All the changes in the clinical scores evaluated during the study period are listed in Table 2 . In particular, when analyzing the primary outcome of the study, the mean basal KOOS pain was 45.4 ± 14.3 and it significantly increased at 1-, 3-, 6-, and 12-month follow-up time points (65.2 ± 21.2, 70.2 ± 23.3, 71.3 ± 23.8, and 74.8 ± 21.6, respectively, P < 0.0001 compared to baseline; Fig. 2 ). Pain was also assessed by the NRS scale, which showed a mean decrease from 6.6 ± 1.7 to 3.9 ± 2.7 (P < 0.0001) at 12-month follow-up compared to baseline.
Clinical Scores of Patients Treated With Subchondroplasty of the Knee.
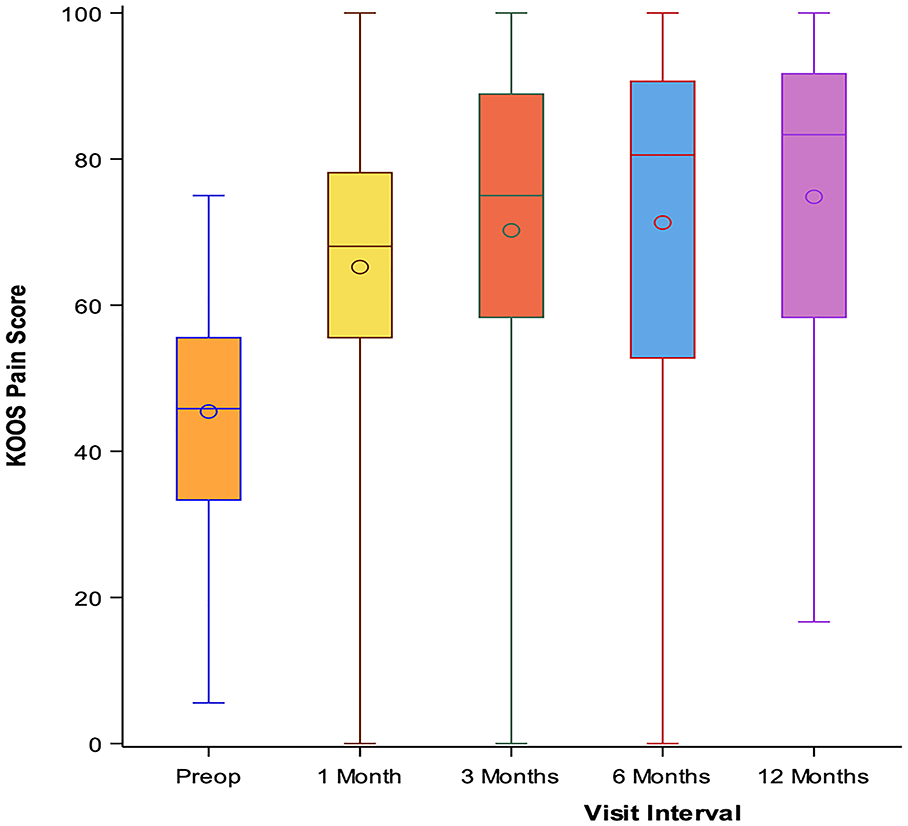
Evaluation of the primary outcome (KOOS pain) at 1-, 3-, 6-, and 12-month follow-ups.
Statistically significant improvements in the other KOOS subscales about symptoms and functionality were registered from baseline (Symptoms: 58.1 ± 18.9; Sports: 15.6 ± 14.9; Activities of daily living, ADL: 53.2 ± 19) to the 12-month follow-up (Symptoms: 75.6 ± 21.6; Sports: 45.8 ± 31.8; ADL: 80.1 ± 20.2; P < 0.0001; Fig. 3 ).
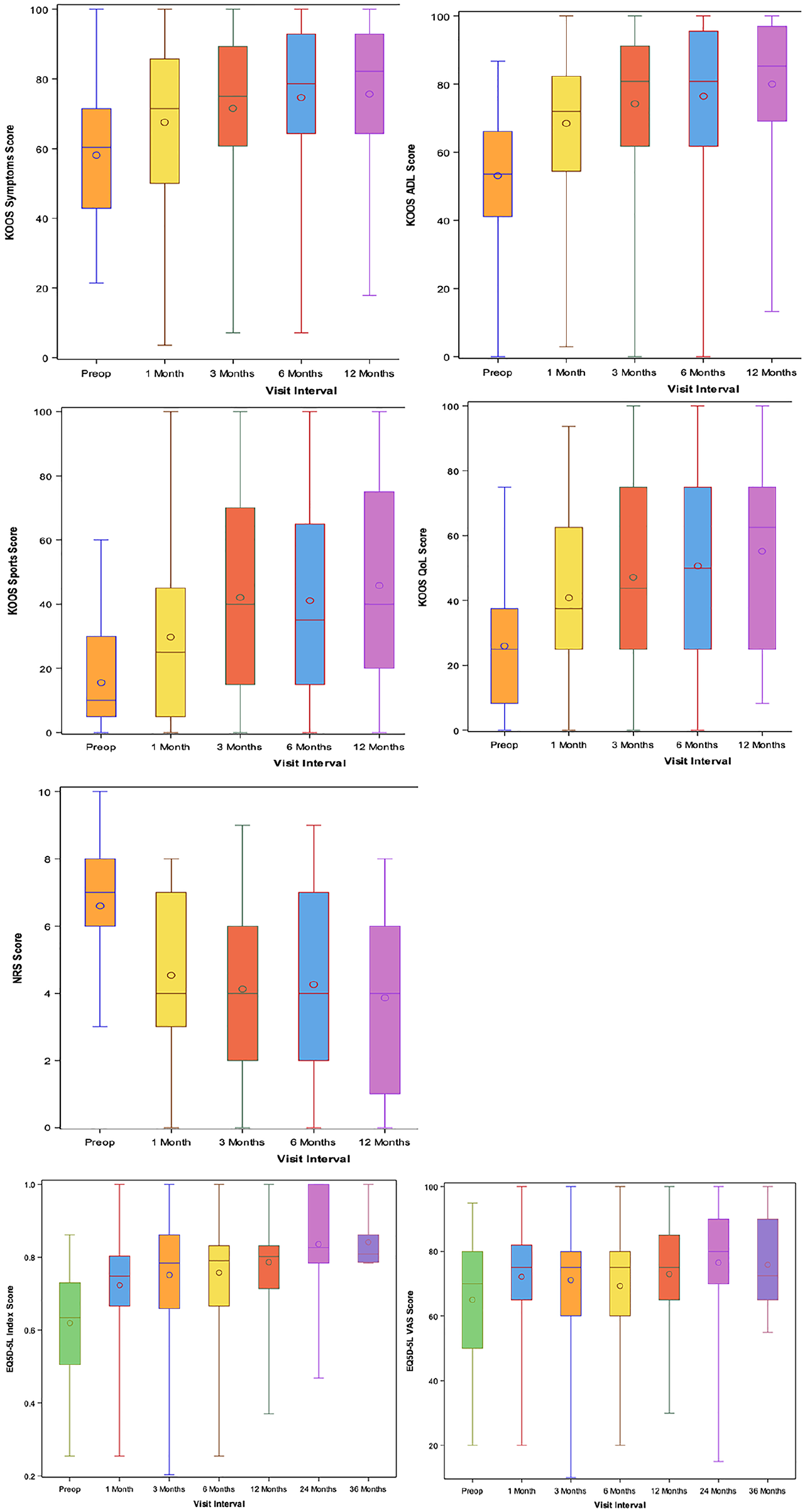
Evaluation of the secondary outcomes (KOOS subscales, NRS for pain, EQ5D-5L index, Eq5D5L VAS score) at 1-, 3-, 6-, and 12-month follow-ups.
A global assessment of the patients’ quality of life was also carried out. KOOS QOL showed a statistically significant increase from baseline (26 ± 18.6) up to 12-month follow-up (52.2 ± 28.8), P < 0.0001. EQ5D-5L increased from 0.62 ± 0.14 to 0.79 ± 0.14 at the last time point (P < 0.0001). When patients were asked if they would undergo again the procedure, 86.8% of them answered positively.
Given the results obtained, a further analysis was performed to assess if any outcome was influenced by concomitant individual characteristics. Hence, a subgroup analysis was conducted to evaluate the correlation between the KOOS subscales and age, gender, number of BMLs, location of BMLs, and Kellgren-Lawrence OA grade. No statistically significant associations were observed (Suppl. Appendix A).
Adverse Events
No serious adverse event was reported. A total of 9 patients reported minor adverse events:
1 patient experienced severe postoperative pain which was managed by Paracetamol/codeine phosphate 500 mg + 30 mg (oral) and then resolved gradually over time;
1 patient suffered from severe postoperative pain managed with Oxycodon, patient-controlled analgesia pump, and transcutaneous electrical nerve stimulation that led to symptoms resolution;
2 patients suffered from joint hemarthrosis during the 2-week following the surgery, which were drained;
1 patient experienced swelling which resolved with conservative measures (Ibuprofen 3 × 600 mg and cryotherapy);
1 patient experienced immediate postoperative pain which was resolved with a 2-day treatment of Oxycodone and an unloader brace;
1 patient had extra-articular leakage of tricalcium phosphate, which was removed through a small skin incision;
1 patient reported severe knee pain and swelling 15 days after the surgery, which was treated via arthrocentesis (50 mL) and methylprednisolone (40 mg) + lidocaine (10 mg) intra-articular injection;
1 patient experienced moderate knee pain and effusion after 1 month, which required an arthrocentesis that led to resolution of the symptoms.
Failures
We registered a total of 4 failures. Three of the treated patients required further surgical management of the symptoms related to the index knee. In particular, 1 patient underwent unicompartimental knee arthroplasty after 6 months, 1 patient underwent a cartilage treatment of the medial femoral condyle (while the SCP was performed on the lateral femoral condyle), and 1 patient was scheduled for total knee arthroplasty. Moreover, the patient who required arthrocentesis and subsequent steroid injection was considered failed as well.
Discussion
The main finding of this study is that SCP provided symptomatic and functional benefits in patients suffering from persisting BMLs associated with mild-to-moderate knee OA. All the scores evaluated showed statistically significant improvement from the first follow-up visit that was maintained up to 12-month follow-up. The importance of the treatment of OA-related BMLs lies in the central role played by the subchondral bone in OA progression and symptomatology. Indeed, OA does not only affect the articular cartilage but the whole joint environment. Moreover, recent evidence suggests the importance of bone-cartilage cross-talk, which has currently been investigated to develop new therapeutic approaches to cartilage degenerative disease. 31 Accordingly, it has been shown that the impairment in the articular environment can gradually alter the structure of the subchondral bone: OA progression is characterized by unbalanced load distribution that alters the cartilage structure, hence allowing the leakage of synovial fluid and inflammatory substances into the subchondral bone plate and thus affecting its whole integrity.13,32 The “unhappy interaction” between cartilage and bone also occurs in the opposite direction. Studies have shown how alterations in the subchondral bone may then cause further, progressive deterioration of articular surface.33,34 Moreover, OA symptoms have been correlated to the presence of BMLs: in fact, it has been shown that nerve growth factors are overexpressed in the subchondral bone affected by BMLs and they may be responsible for nerve sensitization and subsequent onset of pain flares. 35 These growth factors are able to reach the synovial fluid through fibrovascular channels resulting from chronic articular damage and may be the reason for symptom persistence. 32 Nowadays, the panorama of the available treatments for BMLs is extremely narrowed to a small amount of options: first, when addressing the BMLs, conservative options still deserve the role of first-line treatment.3,36 Off-loading, extracorporeal shockwave therapy, physiotherapy, non-steroidal anti-inflammatory drugs (NSAIDs), bisphosphonates, and prostaglandins may lead to complete symptom resolution in a few weeks. 37 However, when these nonoperative approaches fail, surgery may be indicated. Surgical treatment options include core decompression to stimulate spontaneous subchondral bone healing and, in case of mal-alignment associated with early OA, knee osteotomy may be also performed. 38 Biologic agents may nowadays represent an interesting alternative. Studies conducted by Hernigou et al. demonstrated how the subchondral injection of autologous bone marrow aspirate concentrate in BMLs of OA knees is able to postpone or avoid knee arthroplasty in the contralateral joint in patients affected by bilateral OA. 39 However, to our knowledge, this is the first study with a rigorous design and long-term follow-up. SCP might represent a valid, minimally invasive option in those not willing to receive knee arthroplasty or osteotomy with contraindications to such major surgery. The biological rationale of SCP lays in the ability of the macroporous mixture of 2 CaP forms to enhance spontaneous healing of the bone acting as an osteconductive material which is ultimately resorbed and replaced by new bone during the healing process. According to the most recent evidence available in the current literature, 26 the SCP procedure appears to be particularly effective in knee affected by BMLs and moderate OA.40,41
The evidence available so far on the SCP procedure has come from studies with a retrospective design and inconsistent methodology where inclusion and exclusion criteria were significantly diverse among the studies.24,40 -44 According to our knowledge, 2 other prospective studies have been published: Chua et al., 23 who evaluated the outcomes after SCP procedure in 12 patients, and the recent trial by Randelli et al., 45 who prospectively evaluated patients treated by SCP up to 2 years’ follow-up. In this trial, 45 patients were evaluated at 6 months, 39 patients at 12 months, and 37 patients at 24 months of follow-up. Both aforementioned studies appear to be limited by the small number of patients who reached the 1 year of follow-up, which we believe is one of the major strengths of our study. Moreover, the treatment of subchondral bone pathologies has recently expanded beyond the cement, using promising biologic injections targeted directly in the subchondral bone; however, their evidence is limited so far.18,46
In conclusion, we believe that the prospective design of our study, the inclusion and exclusion criteria, and the number of patients enrolled allowed us to obtain results that clearly support the use of SCP for the treatment of BMLs. Our data enabled us to perform a subgroup Pearson correlation analysis which showed that the treatment outcomes were not influenced by age, gender, number of lesions, locations of the lesions, or Kellgren-Lawrence grade. This is one of the greatest strengths of our study, considering the extreme variability of patients treated in previously published papers, where heterogeneous series of patients were evaluated. Furthermore, we believe that accurate patient selection prevented us from having high failure rates at short- to middle-term evaluation: this differs from previous studies, where failures reached up to 20% of treated patients.21,25,41,45
Nonetheless, the present study suffers from limitations as well. First, the lack of a control group treated with an alternative surgical procedure (such as subchondral perforations) strongly limits the reliability of our conclusions. Moreover, the lack of postoperative MRI data prevented us from documenting eventual subchondral bone remodeling, BMLs resolution, or OA progression following SCP. However, even in the presence of radiological data, the healing of BMLs could be fully assessed only by histological examination, which was not included in the study protocol due to ethical reasons. Finally, we must acknowledge that the current surgical technique involves the use of intraoperative x-rays to allow proper cannula placement into the subchondral bone: BMLs are only detected at MRI and therefore intra-op fluoroscopy does not guarantee the correct placement of the cannulas inside the lesions, thus exposing patients to the risk of administering the bone substitute material outside the damaged subchondral bone.
Conclusions
Subchondroplasty is a safe and effective procedure for the treatment of symptoms related to persisting BMLs in mild-to-moderate osteoarthritic knees. Further studies with randomized design and rigorous radiological data collection will be needed to recommend the SCP procedure in the current everyday practice for the management of OA-related BMLs.
Supplemental Material
sj-docx-1-car-10.1177_19476035241264011 – Supplemental material for Subchondroplasty® (SCP) Provides Resolution of Symptoms and Functional Improvements in Mild-to-Moderate Knee Osteoarthritis with Persistent Bone Marrow Lesions: 12-Month Follow-Up Results from a Multicentric Open-Label Prospective Clinical Trial
Supplemental material, sj-docx-1-car-10.1177_19476035241264011 for Subchondroplasty® (SCP) Provides Resolution of Symptoms and Functional Improvements in Mild-to-Moderate Knee Osteoarthritis with Persistent Bone Marrow Lesions: 12-Month Follow-Up Results from a Multicentric Open-Label Prospective Clinical Trial by Berardo Di Matteo, Giuseppe Anzillotti, Pietro Conte, Peter Angele, Pieter Emans, Joan Minguell-Monyart, Jennifer Woodell-May, Miguel Correa-Tapia and Elizaveta Kon in CARTILAGE
Footnotes
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: E.K. is a consultant for Zimmer Biomet. J.W.-M. and M.C.-T. are employees of Zimmer Biomet.
Acknowledgments and Funding
We give credit to Tianyu Gao, a statistician employed by Zimmer Biomet, who helped us with the whole statistical analysis. The presented study was fully sponsored by Zimmer Biomet.
Ethical Approval
The present prospective multicenter study was approved by the Hospital Ethics Committee and institutional Internal Review Board of each center. The trial was registered at clinicaltrials.gov (NCT03430219).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
