Abstract
Coinfection of hepatitis B virus (HBV) or hepatitis C virus (HCV) with HIV is common and associated with increased mortality and increased risk of progression to chronic liver disease. We aimed to study long-term liver diseases after initiation of antiretroviral therapy (ART) in HIV-infected patients with and without HBV or HCV coinfection. A retrospective cohort of 92 patients (32 patients with HBV and/or HCV coinfection) was analyzed. Overall mean age was 38.3 years, and 54.3% were males. Immunological and virological responses were similar between the 2 groups (
Introduction
Hepatitis B virus (HBV), hepatitis C virus (HCV), and HIV share the same routes of transmission, especially sexual contact and intravenous drug use. In HIV-infected patients, coinfection with HBV and/or HCV is common. 1 The global prevalence of HBV coinfection among HIV-infected patients is approximately 5% to 10%. However, this prevalence in Africa and Asia is higher to be at 10% to 15%. 2 In Thailand, HBV coinfection is observed at 8.7% to 9.7%. 3,4 For HCV, the prevalence of HCV coinfection among HIV-infected patients varies from region to region. The prevalence in the United States and Europe is as high as 25% among HIV-infected patients. 5 In South and Southeast Asia, this prevalence ranges from 1.2% to 98.5%. 6 The prevalence of HCV coinfection among HIV-infected patients in Thailand is 7.8% to 8.8%. 3,4
It is known that HIV accelerates the progression of liver disease and HBV or HCV coinfection with HIV results in increased morbidity and mortality associated with viral hepatitis. 1,7,8 Since the introduction of antiretroviral therapy (ART), liver-related mortality has become one of the most common causes of non-AIDS deaths among HIV-infected patients in developed countries. Complications of liver diseases due to chronic HBV or HCV coinfection are major reasons. 1,7 In Thailand and other developing countries, ART has been rapidly scaled up for a decade. AIDS deaths, such as opportunistic infections, have dramatically declined, and non-AIDS morbidity and mortality are emerging. 9 Although the burden of HBV and HCV coinfections is greatest in resource-limited settings, the long-term liver diseases among HIV-infected patients after initiation of ART are unclear. The primary objective of the present study was to compare liver diseases between HIV-infected patients with and without HBV or HCV coinfection. The secondary objectives were to determine the factors associated with liver diseases after initiation of ART and to determine the difference of liver diseases in a subgroup of patients with HBV or HCV coinfection between early and late ART initiation.
Materials and Methods
A retrospective cohort study was conducted in HIV-infected patients who were initiated ART at Ramathibodi Hospital, a medical school hospital in Bangkok, Thailand, between January 2006 and December 2006 and followed up until the end of December 2014. Inclusion criteria were HIV-infected patients who aged more than 15 years, had serology tests of HBV and HCV at baseline, had been initiated ART in 2006, and had regular monitoring for liver function test (LFT), CD4 count, and HIV RNA. Exclusion criteria were patients who had liver diseases prior to ART initiation. Patients were categorized into 2 groups: (1) coinfection group, including patients with HBV or HCV coinfection and (2) no coinfection group, including patients without HBV and HCV coinfections. Clinical characteristics and ART regimens, clinical outcomes, immunological response, virological response, and LFT including alkaline phosphatase (ALP), aspartate transaminase (AST), alanine transaminase (ALT), total bilirubin (TB), direct bilirubin, and albumin were retrieved from medical records.
“Liver diseases,” a composite outcome, was defined as the occurrence of abnormal LFT, clinical hepatitis, cirrhosis, or hepatocellular carcinoma. “Abnormal LFT” was the LFT result showing at least one of the following: ALP >1.5 times the upper normal limit (UNL) and AST, ALT, or TB >2 times the UNL. “Clinical hepatitis” was a new onset of jaundice with abnormal LFT. “Cirrhosis” was diagnosed if there were cirrhotic features in ultrasound or HAI fibrosis score of 3 or more on liver histology from liver biopsy.
Mean values (±standard deviations [SD]) and median values (with interquartile range [IQR]) were used to describe the continuous data with and without normal distribution, respectively. Categorical variables between the 2 groups were compared using χ2 or Fisher exact test where appropriate. Continuous variables between the 2 groups were compared using Student
Results
A total of 92 patients were included in the analysis. Of all, 32 (34.8%) patients had HBV and/or HCV coinfection. The number of patients with HBV, HCV, and HBV + HCV coinfections were 10 (10.9%), 19 (20.7%), and 3 (3.3%), respectively. Overall mean age was 38.3 (SD 8.0) years, and 54.3% of patients were males. Baseline characteristics of the 92 patients are summarized in Table 1. The proportions of male gender and patients with underlying diabetes mellitus were significantly higher in the coinfection group (
Baseline Clinical Characteristics of 92 Study Patients.

Abbreviations: ART, antiretroviral therapy; HBV, hepatitis B virus; HCV, hepatitis C virus; IQR, interquartile range; NS, not significant; NNRTI, non-nucleoside reverse transcriptase; PI, protease inhibitor; SD, standard deviation.
During a median (IQR) follow-up period of 6.1 (3.8-7.7) years, immunological and virological responses were similar over time between the 2 groups (
Immunological and Virological Responses after Initiation of ART in 92 Study Patients.

Abbreviations: ART, antiretroviral therapy; HBV, hepatitis B virus; HCV, hepatitis C virus; IQR, interquartile range; NS, not significant.
Liver Function Test after Initiation of ART in 92 Study Patients.

Abbreviations: ALB, albumin; ALP, alkaline phosphatase; ALT, alanine transaminase; ART, antiretroviral therapy; AST, aspartate transaminase; DB, direct bilirubin; HBV, hepatitis B virus; HCV, hepatitis C virus; IQR, interquartile range; NS, not significant; TB, total bilirubin.
Of the 92 patients, 12 (13.0%) had liver diseases and were exclusively observed only in the coinfection group. These included 10 (10.9%) patients with abnormal LFT and 7 (7.6%) patients with cirrhosis. Of note, 5 patients had both cirrhosis and abnormal LFT. There were no patients having clinical hepatitis or hepatocellular carcinoma. Liver diseases as composite outcomes, abnormal LFT, and cirrhosis were significantly higher among patients in the coinfection group (Table 4). Concordantly, Kaplan-Meier analysis also showed that patients in the coinfection group had a significantly higher probability of having liver diseases after ART (log-rank test,
Clinical Outcomes of 92 Study Patients.

Abbreviations: ALP, alkaline phosphatase; ALT, alanine transaminase; AST, aspartate transaminase; HBV, hepatitis B virus; HCV, hepatitis C virus; IQR, interquartile range; LFT, liver function test; NS, not significant; TB, total bilirubin.
aOccurrence of abnormal liver function test, clinical hepatitis, cirrhosis, or hepatocellular carcinoma.
bLFT result showing at least of one of the following—ALP >1.5 times the upper normal limit (UNL) and AST, ALT, or TB >2 times the UNL.
cA new onset of jaundice with abnormal LFT.
dCirrhotic features in ultrasound or histologic activity index (HAI) fibrosis score of 3 or more on liver histology from liver biopsy.

Kaplan-Meier analysis of liver diseases after initiation of antiretroviral therapy (ART) between patients with and without hepatitis B virus (HBV) or hepatitis C virus (HCV) coinfection.
Univariate and Multivariate Analyses of Factors Associated with Liver Diseases.
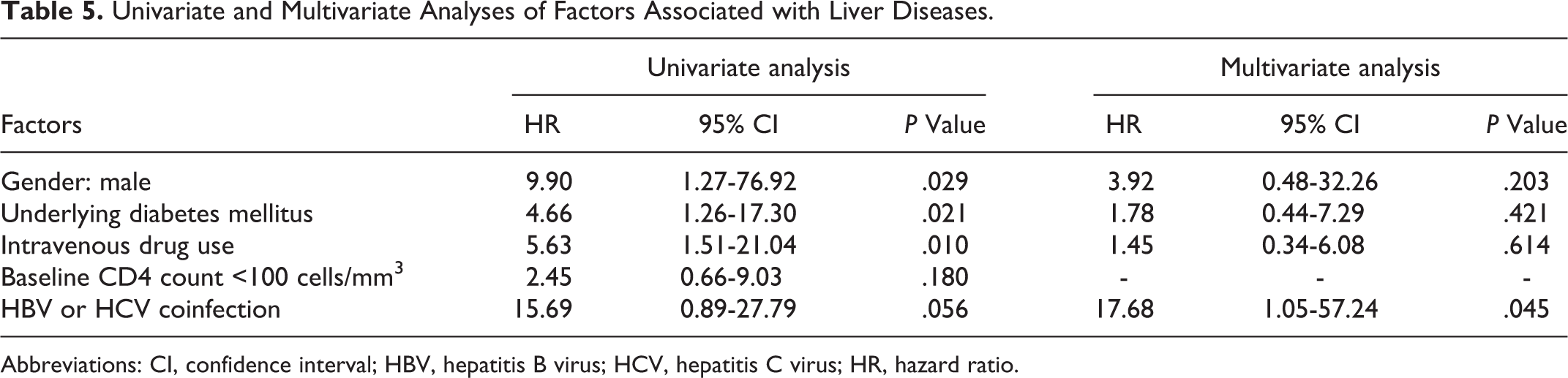
Abbreviations: CI, confidence interval; HBV, hepatitis B virus; HCV, hepatitis C virus; HR, hazard ratio.
In the subgroup of 32 patients with coinfection, patients who were initiated ART at CD4 count <200 cells/mm3 had a higher rate of liver diseases compared to those who were initiated ART at CD4 count ≥200 cells/mm3 (42.3% versus 16.7%;
Clinical Outcomes of 32 Patients with HBV or HCV Coinfection.

Abbreviations: ALP, alkaline phosphatase; ALT, alanine transaminase; ART, antiretroviral therapy; AST, aspartate transaminase; HBV, hepatitis B virus; HCV, hepatitis C virus; IQR, interquartile range; LFT, liver function test; NS, not significant; TB, total bilirubin.
aOccurrence of abnormal liver function test, clinical hepatitis, cirrhosis, or hepatocellular carcinoma.
bLFT result showing at least of one of the following—ALP > 1.5 times the upper normal limit (UNL) and AST, ALT, or TB > 2 times the UNL.
cA new onset of jaundice with abnormal LFT.
dCirrhotic features in ultrasound or histologic activity index (HAI) fibrosis score of 3 or more on liver histology from liver biopsy.
Discussion
Liver diseases from HBV or HCV coinfection have become a leading cause of morbidity and mortality among HIV-infected patients in developed countries. The progressive liver diseases may threaten the long-term success of ART. In developing countries where HBV or HCV coinfection with HIV is common, the emerging problem of liver diseases is a great concern. The results of the present study have demonstrated that long-term liver diseases after initiation of ART in HIV-infected patients with HBV or HCV coinfection were significantly higher than that in patients without coinfection. The Kaplan-Meier analysis also showed that liver diseases had occurred more frequently during the first 5 years after ART initiation and then occurred through 10 years after ART initiation. This finding emphasizes for physicians to regularly monitor liver function in long-term care of HIV-infected patients with HBV or HCV coinfection.
The results of the multivariate analysis in the present study have revealed that HBV or HCV coinfection was the only factor independently associated with the occurrence of liver diseases. A previous cohort study reported that coinfection with HBV is associated with hepatic outcomes similar to HCV. 10 The consequences of HBV or HCV coinfection are chronic hepatitis, cirrhosis, hepatocellular carcinoma, and hepatotoxicity from antiretroviral drugs. The latter is very important in HIV endemic regions with rapid scaling up of ART, especially in resource-limited settings where HBV and HCV coinfections are not routinely screened. 11 Elevation in liver enzyme levels after ART initiation is common, particularly in HCV coinfection because of impaired hepatocyte defense mechanisms. Hepatitis C virus coinfection is associated with 2- to 10-folds chance of developing elevated liver enzyme levels after ART initiation. 12 Although knowing HCV serology status may not change the decision regarding choices of antiretroviral drugs, it is beneficial to determine the patients who need close monitoring of liver function after ART initiation.
Initiation of ART in HIV-infected patients with HBV or HCV coinfection has improved survival and quality of life. For HBV coinfection, ART regimens also provide treatment for HBV as well. 13 For patients with HCV coinfection, ART has also improved liver function since CD4 count recovery on ART is associated with decreased progression to liver disease. 14,15 Among patients with HBV or HCV coinfection, the results from the present study have shown that late initiation of ART at CD4 count <200 cells/mm3 was associated with a higher chance of having liver diseases after ART initiation, when compared to patients who were initiated ART at CD4 count ≥200 cells/mm3. Liver flare after initiation of ART in advanced HIV-infected patients may play a role for this phenomenon. 16 –17 A previous cohort study also described that control of immunosuppression with ART and CD4 counts >200 cells/mm3 was associated with better hepatic outcomes. 10 Therefore, in resource-limited settings where ART is not available for all, HIV-infected patients with HBV or HCV coinfection should be one of the priority groups to initiate ART earlier.
There are some limitations to the present study. The nature of the retrospective study may lack some information. The study was conducted in resource-limited setting, and the results may not be applicable to resource-rich settings. The relatively small sample size of study patients may not be able to demonstrate the difference in clinical outcomes between patients coinfected with HBV and HCV.
In conclusion, HIV-infected patients with HBV or HCV coinfection have a higher chance of developing liver diseases after ART initiation. Close and regular monitoring of liver function after ART initiation is recommended. Screening of HBV or HCV coinfection prior to ART initiation is beneficial for long-term care of HIV-infected patients. To decrease the risk of liver diseases, ART should be initiated early in HIV-infected patients with HBV or HCV coinfection.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
