Abstract
Background:
Previous research has studied the rate of hepatitis C virus (HCV) coinfection among HIV-positive people regardless of their antiretroviral therapy (ART) status. Our objectives were to measure the seroprevalence of HCV both in HIV-positive people receiving ART and in those not receiving ART and to describe the characteristics of coinfected people in the Kathmandu Valley, Nepal.
Methods:
We conducted a cross-sectional survey of 319 HIV-positive people residing in the Kathmandu Valley, Nepal. We screened the participants’ serum samples for HCV antibodies using the Latex Photometric Immunoassay based on third-generation assay.
Results:
A total of 138 (43.3%) participants were HCV positive. The prevalence of HCV coinfection was 96.2% (125 of 130) among participants with lifetime injection drug use (IDU). Among participants not receiving ART, the coinfection rate was 58.1% (50 of 86) compared with 37.8% (88 of 233) among those receiving ART. In multivariable analysis, participants who reported lifetime IDU and were current smokers were more likely to have HCV coinfection. The adjusted odds ratio (AOR) of HCV coinfection for ART was decreased, although it was not statistically significant (AOR = 0.45; 95% confidence interval = 0.13-1.48).
Conclusion:
The high prevalence of HCV coinfection among both individuals receiving ART and those not receiving ART suggests that screening for HCV among HIV-positive people would be useful, in particular, for those with lifetime IDU and soon after their HIV diagnosis.
Introduction
Coinfection of the hepatitis C virus (HCV) and HIV is an important public health concern. Of the 33.4 million HIV-positive people globally, 1 around 20% are coinfected with HCV. 2 Coinfection of HCV and HIV accelerates liver disease progression. 3 Among coinfected patients, cirrhosis of the liver is 6 times more likely compared with those infected solely with HCV. 4 The provision of antiretroviral therapy (ART) has dramatically increased the life span as well as the quality of life for HIV-positive people both in developed and in resource-limited countries. However, HCV-induced liver disease has emerged as a major cause of morbidity and mortality, particularly among HIV-positive people coinfected with HCV. 3,5
The management of patients coinfected with HCV and HIV is challenging. Coinfected patients are more likely to develop liver enzyme abnormalities and liver toxicity when treated with ART. 3 The successful treatment of HCV has been associated with a regression of liver fibrosis 6,7 and with a reduced risk of ART-related liver toxicity. 8 HIV-positive people, therefore, are encouraged to get screened for HCV, and those with positive results are considered for HCV treatment. Knowing that patients are coinfected informs practitioners to provide specific education on HCV transmission and treatment, factors influencing liver disease progression, risk reduction strategies, and risk of reinfection.
Previous studies have assessed the prevalence of HCV coinfection among HIV-positive people in resource-limited countries, where the overall burden of coinfection is disproportionately high. 9 –13 The rate of coinfection is highly variable in these countries, ranging from 1.9% in South Africa 14 and 2.4% in northern India 15 to 36.2% in Brazil. 9 In areas where injection drug use (IDU) is a major route of HIV transmission, coinfection rates of over 90% have been reported. 11,15 –17
Despite the high burden of coinfection, HCV screening is not a routine practice among HIV-positive people particularly among those who are not eligible for ART initiation, as is the case in most resource-limited countries. The national guidelines of these countries recommend HCV screening before the initiation of ART. 18 However, the provision of ART is still low in some resource-limited countries 19 and is even worse among IDUs. 20 Late HIV diagnosis and delayed initiation of ART are common in these countries, resulting in elevated morbidity and mortality. 21 Since HIV and HCV share a common route of transmission, we assume that the rates of coinfection are similarly high among those who have initiated ART and those who have not. However, previous studies have reported the rate of HCV coinfection among HIV-positive people without regard for their ART status. 9,10 Gaining knowledge about HCV coinfection among HIV-positive people, who have not yet initiated ART, would be useful to inform the development of an HCV-screening strategy for this group at high risk of liver disease.
We conducted this study in Nepal, a resource-limited country in South Asia. In Nepal, the first case of HIV/AIDS was reported in 1988. Since then the HIV epidemic in the country has advanced from “low prevalence” to a “concentrated epidemic,” with prevalence over 5% within a subgroup of most-at-risk populations. In particular, HIV prevalence among IDUs has remained high at 6.3% in 2011 in the Kathmandu Valley, though declining gradually over the years. HIV prevalence among IDUs in the Kathmandu Valley was 68% in 2003 and 20.7% in 2009. 22
Similar to HIV, previous studies have reported high prevalence of HCV among IDUs in the Kathmandu Valley. In a study in 1998, 23 for example, the prevalence of anti-HCV was 94% among IDUs. In 2009, another study in the Kathmandu Valley assessed the prevalence of anti-HCV among 118 IDUs enrolled in opioid substitution therapy (OST) and 82 IDUs who were not in the OST program. In that study, the prevalence of anti-HCV among the OST users and nonusers was 80.5% and 57%, respectively. 24 Hepatitis C virus is known to be more rapidly transmitted by IDU than HIV. 25 This suggests that the prevalence of HCV coinfection should be high particularly among HIV-positive people with lifetime IDU in the Kathmandu Valley, regardless of their ART status. However, to date no studies have been conducted to assess the rate of HCV coinfection among HIV-positive people or the characteristics of coinfected people in the Kathmandu Valley. National ART guidelines for Nepal 26 recommend screening for HCV among HIV-positive people before ART initiation. However, specific guidelines are still lacking for HCV screening among those HIV-positive people who are not yet eligible for ART.
The objectives of this study were to measure the seroprevalence of HCV in HIV-positive people who are receiving ART as well as those who are not receiving ART and to describe the characteristics of coinfected people in the Kathmandu Valley, Nepal.
Methods
Study Area
We conducted this cross-sectional study in the Kathmandu Valley, Nepal, from February to March 2010 in the wider context of a baseline survey of a prospective healthy living intervention study headed by the first and the last authors. 27 –29 The Kathmandu Valley consists of 3 districts, namely Kathmandu, Lalitpur, and Bhaktapur, with an estimated population of 1.9 million in 2006. In 2012, 6 of the 39 ART sites within the country were located in the Kathmandu Valley.
Participants
We recruited a total of 322 participants for this study. The inclusion criteria were age 18 years or older, self-reported diagnosis of HIV infection, currently living in the Kathmandu Valley, and giving written informed consent to participate in the study. We recruited the participants through the staff of 5 nongovernmental organizations working with HIV-positive people in the Kathmandu Valley. The procedures of the study were reviewed and approved by the ethical committees of the Nepal Health Research Council, Kathmandu, Nepal; National Center for Global Health and Medicine, Tokyo, Japan; and Waseda University, Tokyo, Japan.
Procedures
We used a structured Nepali version of the questionnaire for data collection. First, we developed the questionnaire in English, translated it into the Nepali language, and then back translated it into English. After the back translation, we revised the Nepali version again. Finally, we pretested it with 30 HIV-positive people and made additional modifications based on the pretest results.
We hired 4 interviewers for the data collection. The first author provided a day-long training session on the questionnaire and interview technique. The interviewers first informed each of the participants individually about the study procedures using an information sheet. The participants then signed the informed consent form agreeing to participate in the study. Subsequently, the interviewers conducted face-to-face interviews in a private setting using the structured questionnaire. To enhance confidentiality, the interviewers reassured the participants that codes would be used in all records instead of their names or other identifying information. Each interview took about 45 minutes. Participants were reimbursed 100 Nepali rupees (about US$1.35) for their transportation costs to and from the interview venues.
Laboratory technicians hired for the study collected approximately 10 mL of venous blood from each participant using an evacuated tube. To separate the serum, the blood was centrifuged immediately at the interview site for 15 minutes. The first and last authors supervised the fieldwork.
Measures
Measures included sociodemographic and health characteristics, including age, sex, marital status, education, employment status, months since testing HIV positive, ART status, hepatitis C status, and assessment of drug use, smoking, and alcohol use.
We asked participants about their recent and lifetime use of several substances including alcohol, marijuana, and others. Alcohol use was assessed by asking participants whether they had consumed any alcohol in the past 30 days. To assess lifetime IDU, we asked participants whether they had ever used drugs by injection. In addition, we asked about IDU in the past 6 months. We assessed smoking status and categorized participants as
We performed the anti-HCV test with the Latex Photometric Immunoassay (LPIA) using test kits from Ortho HCV Abs LPIA test III based on third-generation assay (Ortho-Clinical Diagnostics, Tokyo, Japan).
Data Analysis
A total of 322 participants completed the structured interview. However, 3 participants did not provide a blood sample; thus, we included 319 participants in our analysis. For the analysis, first, we calculated the frequencies and descriptive statistics to summarize the general characteristics of the participants. Second, we used the chi-square test to assess the differences in sociodemographic and health characteristics between participants who reported lifetime IDU and those who did not and between participants who were receiving ART and those who were not. Third, we examined the bivariate associations between each independent variable and HCV coinfection among the participants. Finally, we performed multivariable logistic regression analysis to explore the factors of HCV coinfection among the participants. We analyzed the data using SPSS version 15.0 (SPSS Inc, Chicago, Illinois) for Windows.
Results
General Characteristics
The mean age of the participants was 35.6 years (SD = 6.9 years). Of the total (n = 319), 57.4% of participants were men, 68.7% were currently married, 31.7% were not employed, 73.1% were receiving ART, 40.8% reported lifetime IDU, and 8.5% (n = 27) reported IDU during the past 6 months (Table 1). The median duration since testing HIV positive is 53 months.
Sociodemographic and Health Characteristics of the Participants (n = 319).

a Two participants did not respond to this question.
Characteristics of the participants by IDU are shown in Table 2. A higher proportion of male participants (68.3%) reported lifetime IDU than female participants (3.7%). Similarly, educational level of the participants, antiretroviral status, current smoking status, alcohol use, and history of sexual intercourse in the past 6 months were statistically associated with lifetime drug use.
Characteristics of the Participants by Their Lifetime Injection Drug Use (n = 319).

Abbreviation: IDU, injection drug user.
a Two participants did not respond to this question.
Characteristics of the participants by their ART status are shown in Table 3. Fewer participants between 20 and 34 years of age (65.4%) were receiving ART than those between 30 and 60 years of age (83.2%). Other characteristics of the participants that were statistically associated with their ART status included sex, education level, lifetime drug use, current smoking, and alcohol use. Of the 27 participants who had endorsed IDU in the past 6 months, 15 were not receiving ART.
Characteristics of the Participants by Their Antiretroviral Therapy (n = 319).

a Two participants did not respond to this question.
Prevalence of HCV Coinfection
A total of 138 (43.3%) participants were found to be HCV positive. The prevalence of HCV coinfection was 96.2% (125 of 130) among participants with lifetime IDU, while it was 6.9% among those without lifetime IDU. The prevalence of HCV coinfection was 37.8% (88 of 233) among participants receiving ART, while it was 58.1% (50 of 86) among those not receiving ART. The prevalence of HCV coinfection was 93.3% (14 of 15) among those participants who were not receiving ART and had reported IDU in the past 6 months.
Factors Associated with HCV and HIV Coinfection
Factors associated with HCV coinfection among HIV-positive participants in the bivariate analysis are shown in Table 4. In the multivariable analysis, participants who reported lifetime IDU were more likely to have HCV coinfection than those who did not. Similarly, participants who were current smokers were more likely to have HCV coinfection than those who were not. The adjusted odds ratio (AOR) of HCV coinfection for ART was decreased, although it was not statistically significant (AOR = 0.45; 95% confidence interval = 0.13-1.48). Of the 13 participants who were coinfected with HCV but did not report lifetime IDU, 8 reported having had sexual intercourse in the past 6 months.
Factors Associated with HCV Coinfection among HIV-Positive People in the Kathmandu Valley, Nepal.
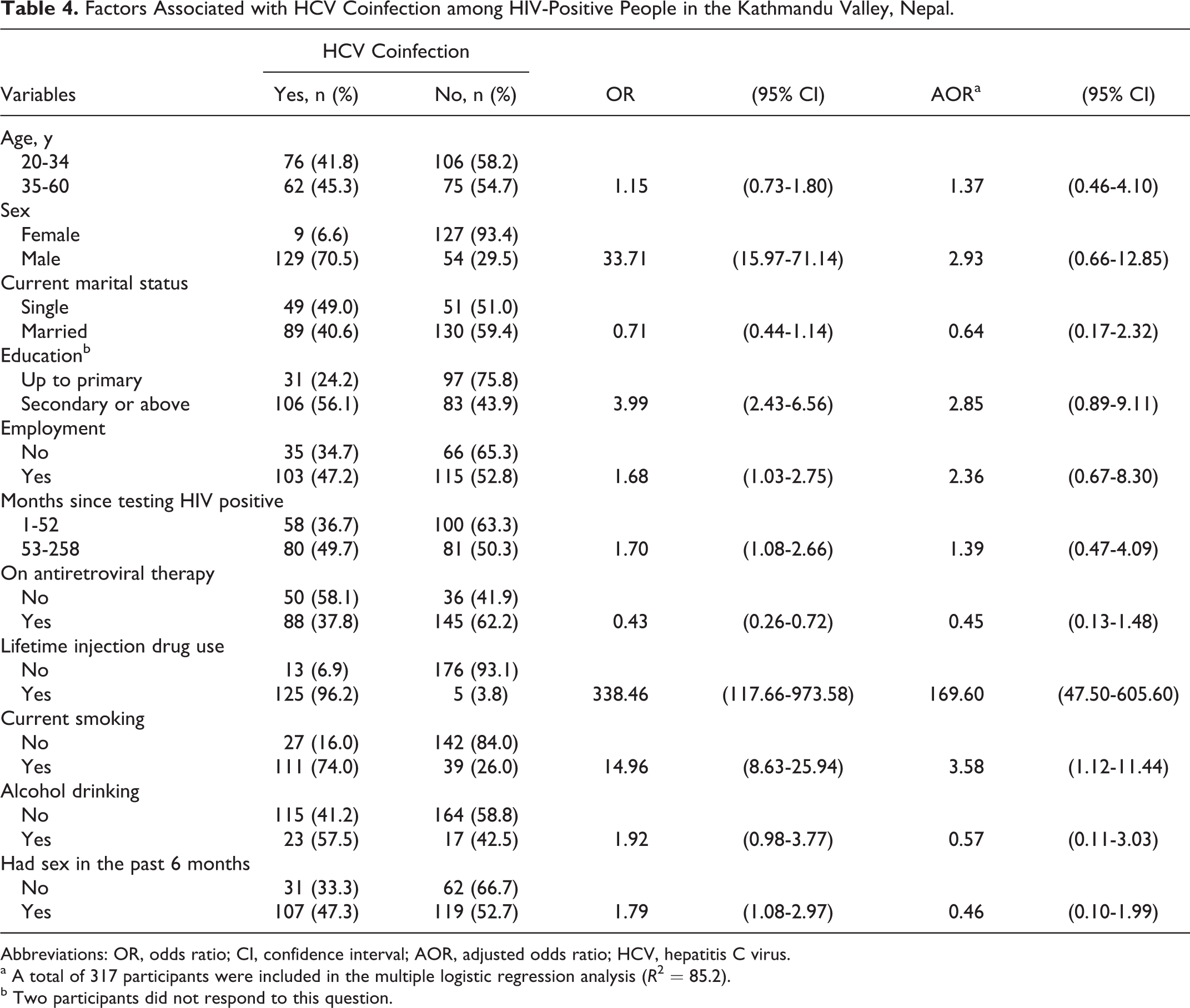
Abbreviations: OR, odds ratio; CI, confidence interval; AOR, adjusted odds ratio; HCV, hepatitis C virus.
a A total of 317 participants were included in the multiple logistic regression analysis (
b Two participants did not respond to this question.
Discussion
This study revealed high prevalence of HCV coinfection among HIV-positive people in the Kathmandu Valley, Nepal. Our data indicate that more than 2 of 5 HIV-positive people had HCV coinfection. The prevalence of coinfection was higher among participants not receiving ART than those receiving ART though the difference was not statistically significant in multivariable analysis. Participants, who endorsed lifetime IDU and were current smokers, were more likely to be coinfected with HCV.
A high proportion of HIV-positive people in this study were coinfected with HCV. At 43.3%, the HCV coinfection rate in our study far exceeded rates reported in India (2.4-3.5%) 10,13 and Brazil (36.2%). 9 Over 40% of participants reported a lifetime history of IDU. The HCV coinfection in this study was markedly associated with IDU similar to previous studies 31,32 ; almost all (96.2%) participants who reported the lifetime history of IDU were coinfected with HCV. According to previous research, HCV is known to be more rapidly transmitted by IDU than HIV. 25
Similarly high levels of HCV coinfection among participants receiving ART and those not receiving ART have potential implications in designing HCV-screening strategies for HIV-positive people and in counseling those with positive test results. Rarely do either acute or chronic HCV present with significant symptoms. 33 Consequently, those infected are unlikely to visit an HIV clinic or receive an HCV test without having other medical problems. Infected people, who are not aware of their HCV status, might unknowingly spread HCV to others. In our study, for example, 15 participants who reported IDU in the past 6 months were not receiving ART, and all except 1 were coinfected with HCV. These individuals could inadvertently transmit HCV to those with whom they share drug paraphernalia.
In total, about 6 of the 10 participants, who were not receiving ART in our study, were coinfected with HCV. Without innovative HCV-screening interventions, these participants are unlikely to receive HCV testing until ART initiation. Although HCV treatment is rare in Nepal 26 like in many other resource-limited countries, 34 timely diagnosis of HCV would be beneficial in order to educate patients regarding HCV transmission, risk of reinfection, and liver disease progression.
HIV-positive current smokers in our study were more likely to be coinfected with HCV than nonsmokers. The high rate of smoking among HCV coinfected HIV-positive people in our study is a cause for concern because smoking has been identified as a risk factor for hepatocellular carcinoma. 35,36 Moreover, a meta-analysis demonstrated evidence of a synergistic effect between smoking and HCV infection on the risk of hepatocellular carcinoma. 37 It is, therefore, important to encourage HCV-coinfected HIV-positive people to quit smoking.
An important question is how participants, who denied lifetime IDU, contracted HCV. In our study, a total of 13 HCV-coinfected HIV-positive participants did not report lifetime IDU. Eight of these participants had had sexual intercourse in the past 6 months. Although sexual transmission of HCV is still controversial, traumatic intercourse may lead to a loss of mucosal barriers and may facilitate the sexual transmission of HCV. 38,39 Unfortunately, we did not measure lifetime sexual behaviors and other important information, such as the history of sharing razors or toothbrushes, blood transfusion, dialysis, tattoo, and other potential modes of transmission. Further studies would be needed to address this issue.
Our study has 2 limitations. First, the diagnosis of HCV in our study was based on anti-HCV antibody testing; HCV RNA analysis was not performed to confirm the diagnosis. However, it is likely that the HCV prevalence will remain high and that the characteristics of the coinfected people will remain similar even after confirmation of the diagnosis. This is because HCV and HIV coinfection is associated with a reduced rate of spontaneous clearance of HCV RNA, with over 80% of patients developing chronic infection. 40 Next, as we relied on self-reported data, the responses of our participants might have been influenced by social desirability bias, although our methodology attempted to minimize this effect.
Despite these limitations, our study documented high prevalence of HCV coinfection both among HIV-positive people receiving ART and those not receiving ART in the Kathmandu Valley, Nepal. It is likely that IDU was the major route of HCV transmission among our study participants. Screening for HCV among HIV-positive people, particularly those with lifetime IDU and soon after their HIV diagnosis, would be useful for the timely identification of HCV coinfection as well as education and possibly treatment.
Footnotes
Acknowledgments
We would like to thank all the participants of this study for their valuable information, cooperation, and participation. We thank the interviewers, laboratory assistants, and research assistants for their efforts to carry out the fieldwork. We would also like to gratefully acknowledge the 5 nongovernmental organizations – SPARSHA Nepal, Sneha Samaj, Srijansil Mahila Samuha, Shakti Milan Samaj, and Youth Visition Nepal –for their valuable support during our fieldwork.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was partly supported by a Grant for Research on Global Health and Medicine (21A-2) from the National Center for Global Health and Medicine, Japan, a Grant-in-Aid for Young Scientists (B 22790581), Japan Society for the Promotion of Science, The Ministry of Education, Culture, Sports, Science and Technology, Japan, and by a Waseda University Grant for Special Research Projects (2012A-101).
