Abstract
Plain Language Summary
People living with human immunodeficiency virus (HIV) infection who have hepatitis B or C infection have a higher chance of developing advanced liver disease than those who do not have either of the hepatitis infections. This finding highlights the importance of awareness, screening, and treatment of the hepatitis viruses in HIV programs in order to reduce the risk of liver disease in this population.
Background
Following the success of antiretroviral treatment (ART), liver disease has become an important cause of morbidity and mortality in people living with the human immunodeficiency virus (HIV) (PLWH). While overall death rates have declined, there has been a corresponding increase in the proportion of deaths due to liver disease and other non-AIDS-related causes. 1 Liver disease is now the most common non-AIDS-related cause of death among PLWH in Europe and the United States, accounting for up to 14% of all deaths and 50% of all in-hospital deaths.2,3
Liver fibrosis and cirrhosis are common in PLWH in sub-Saharan Africa and other low- and middle-income countries, occurring in about 9% to 17%.4–6 In high-income countries, the most common causes of liver fibrosis and cirrhosis in PLWH are chronic viral hepatitis B and C (HBV and HCV), nonalcoholic fatty liver disease (NAFLD), and excess alcohol. 7 Among PLWH co-infected with HBV and HCV, rates of liver fibrosis are up to 5 times higher than in those with HIV alone.8,9 Progression of liver fibrosis is also faster in those with HIV/HCV co-infection than in those with HCV alone.10,11 In persons with HIV/HBV co-infection, liver fibrosis has been shown to progress in about 30% despite effective ART and HBV and HIV virological suppression.12,13
In Nigeria, there is a high prevalence of HBV and HCV among PLWH (7.8% and 4.7%, respectively). 14 Results from one study showed that HBV infection was associated with advanced liver fibrosis in PLWH in Nigeria. 9 In contrast, chronic HCV has not been well characterized, neither has its association with liver disease been well described in PLWH in Nigeria because of the lack of virological testing to confirm active infection in most studies.15,16 Chronic HCV prevalence rates ranging from 6% to 8.2% have been reported from the few studies that used confirmatory virological testing.17,18 Triple infection has also been reported, with a prevalence ranging from 0.6% to 2.5% in Nigeria.14,16,19 High rates of both HCV and HBV in PLWH in Nigeria put persons with HIV at substantial risk of liver fibrosis and other liver complications. 14 Several other local factors exist in Nigeria that can exacerbate that risk including fatty liver, hyperlipidemia, consumption of alcohol and herbs, cigarette smoking, and HBV/hepatitis D virus (HDV) co-infection.20–22
Our objective was to compare demographic, clinical, and laboratory characteristics in PLWH with and without significant liver fibrosis/cirrhosis in Nigeria. We also examined the association between viral hepatitis co-infection and significant liver fibrosis/cirrhosis, particularly chronic HCV, which has not been reported in this setting.9,23
Methods
This cross-sectional study was conducted among PLWH in Nigeria who were enrolled in a longitudinal study examining the epigenomic biomarkers of HIV-associated hepatocellular carcinoma (HCC) in Nigeria (grant number U54CA221205). 24 Participants included in this analysis were from 2 HIV control groups without HCC: (i) participants with HIV without HCC or significant liver fibrosis/cirrhosis (transient elastography [TE] < 9.3 kPa); (ii) participants with HIV with significant fibrosis/cirrhosis (TE ≥ 9.3 kPa). The TE cutoff for significant liver fibrosis/cirrhosis (≥9.3 kPa) was chosen from a previous validation study that was conducted in a population of predominantly black Americans. 25 Participants were recruited from the Gastroenterology and HIV clinics of 2 large tertiary hospitals located in the southwestern and northcentral regions of Nigeria, between July 2018 and August 2022. They were included in this analysis if they were: (a) documented HIV seropositive and (b) >18 years. Exclusion criteria were: (a) clinical and/or radiological evidence of HCC, other liver diseases such as autoimmune hepatitis, cholestatic liver diseases, or other primary or secondary malignancy; (b) unable to consent; and (c) currently pregnant. Ethical approval was received from the Health Research Ethics Committees of College of Medicine, University of Lagos, Lagos (approval number CMUL/HREC/01/18/326) and Jos University Teaching Hospital, Jos (approval number JUTH/DCS/ADM/127/XXVII/629), and all participants provided written informed consent.
Sampling Technique and Sample Size Determination
Convenience sampling technique was used, in which all consecutively presenting and consenting participants attending the HIV clinics were enrolled if they fulfilled the eligibility criteria.
Data Collection
All study participants were provided with information about the study and asked to sign an informed consent. At enrollment, participants underwent a physical exam by the study physician and laboratory testing for HIV (using enzyme-linked immunosorbent assay and western blot), complete blood count, comprehensive chemistry panel including albumin, total bilirubin, aspartate transaminase (AST) and alanine transaminase (ALT); viral serology including hepatitis B surface antigen (HBsAg) and antibody (anti-HBs) and HCV antibody (anti-HCV) using Lumi Quick diagnostics rapid test kits, and alpha-fetoprotein. Plasma samples were also collected for methylomic analysis. Patients who were confirmed HIV seropositive also had CD4+ T cell count (flow cytometry (Partec GmbH, Munster, Germany), and HIV RNA (Roche Ampliprep TaqMan, Roche Diagnostics Germany; lower limit of detection of 20 copies/mL) testing performed and were referred immediately to the HIV care and treatment clinic at their respective institutions for further treatment and care. Confirmatory HBV and HCV viral load testing on stored specimens was performed on all HBsAg and anti-HCV positive participants using the Cepheid GeneXpert® system.
The HIV clinics of the 2 hospitals provided comprehensive care and treatment services to participants with HIV infection based on Nigerian guidelines. 26 Participants were evaluated every 3 to 6 months in the clinic and at each visit were provided with ART, counseling and support to encourage medication adherence. All participants are initiated on ART immediately after diagnosis. The preferred ART regimen at the time of study was the combination of tenofovir, lamivudine, and dolutegravir according to Nigerian guidelines, which most study participants were receiving. 26 Participants diagnosed with HBV and/or HCV infection with clinical, laboratory, or radiological evidence of liver abnormalities, such as features of decompensated liver disease or HCC, are typically referred to the Gastroenterology clinic for additional evaluation.
Data included in this analysis included: age, sex, weight (kilograms), height (meters), highest educational level, marital status, consumption of local herbs and alcohol, tobacco use, family history of HCC, history of diabetes (self-reported), duration of ART, and ART regimen. The following laboratory data were included: platelet count, ALT, AST, HBsAg, anti-HCV, HIV RNA, HBV DNA, HCV RNA, and TE score.
Statistical Analysis
Data were analyzed using R Statistical software version 4.1.2. Baseline characteristics were described using the median and interquartile range (IQR) for continuous variables such as age, and counts and percentages for categorical variables such as sex. Body mass index (BMI) was calculated as weight (kilograms)/height2 (meters2) (kg/m2). The outcome variable was the presence or absence of significant liver fibrosis/cirrhosis, which was defined as TE ≥ 9.3 or <9.3 kPa, respectively. 25 We also analyzed the data using a lower cutoff for advanced fibrosis/cirrhosis of TE ≥ 7.6 kPa, which was chosen from a previous validation study. 27 We compared participants with and without significant liver fibrosis/cirrhosis using the Mann-Whitney U test for continuous variables, and the χ2 test or Fisher's exact test for categorical variables. For logistic regression, we dichotomized age (>50 vs ≤50 years), BMI (≥25 vs <25 kg/m2), and duration of ART (>5 vs ≤5 years). Univariate logistic regression was performed to evaluate the unadjusted associated factors for significant liver fibrosis/cirrhosis. Thereafter, we performed multivariable logistic regression to determine the factors that were independently associated with significant liver fibrosis/cirrhosis after adjusting for clinically significant predictors (BMI, duration of ART, HBsAg) and variables with a univariate P-value of <.2 (age, sex, consumption of herbs or alcohol, diabetes and viremic HCV [defined as anti-HCV seropositivity and HCV RNA > 10 IU/mL]). We presented results in tables as odds ratios and corresponding 95% confidence intervals.
Results
Characteristics of the Study Participants
Five hundred and thirty-seven participants of which 119 (22.2%) with significant liver fibrosis/cirrhosis (TE ≥ 9.3 kPa), and 418 (77.8%) without significant liver fibrosis/cirrhosis (TE < 9.3 kPa) were included in this analysis. The median age was 46 years (IQR = 40, 53), 34.3% were male and median duration of ART was 10.8 years (IQR = 5.6, 13.5). The majority were married (347 of 537, 64.6%), 362 of 534 (67.8%) had at least secondary school education, while 8 of 503 (1.6%) reported a family history of HCC. Fifty-seven of 507 (11.2%) were HBsAg seropositive, 59 of 502 (11.8%) were anti-HCV seropositive and 28 of 489 (5.7%) had viremic HCV (Table 1). Participants with significant liver fibrosis/cirrhosis were older than those without (48 vs 46 years), and consumed herbs (29.7% vs 15.4%) and alcohol (25.2% vs 13.7%) more frequently than those without. More participants with significant fibrosis/cirrhosis had viremic HCV infection (15 of 93 [16.1%]) than those without (13 of 396 [3.3%]), however, the frequency of HBsAg seropositivity was similar in those with and without significant fibrosis/cirrhosis. Diabetes was more common in those with significant fibrosis/cirrhosis (11 of 118 [9.3%] vs 8 of 412 [1.9%]). ALT and AST were higher in those with significant liver fibrosis/cirrhosis (25 vs 15 U/L and 44 vs 29 U/L, respectively).
Demographic, Clinical and Laboratory Characteristics of the Participants.
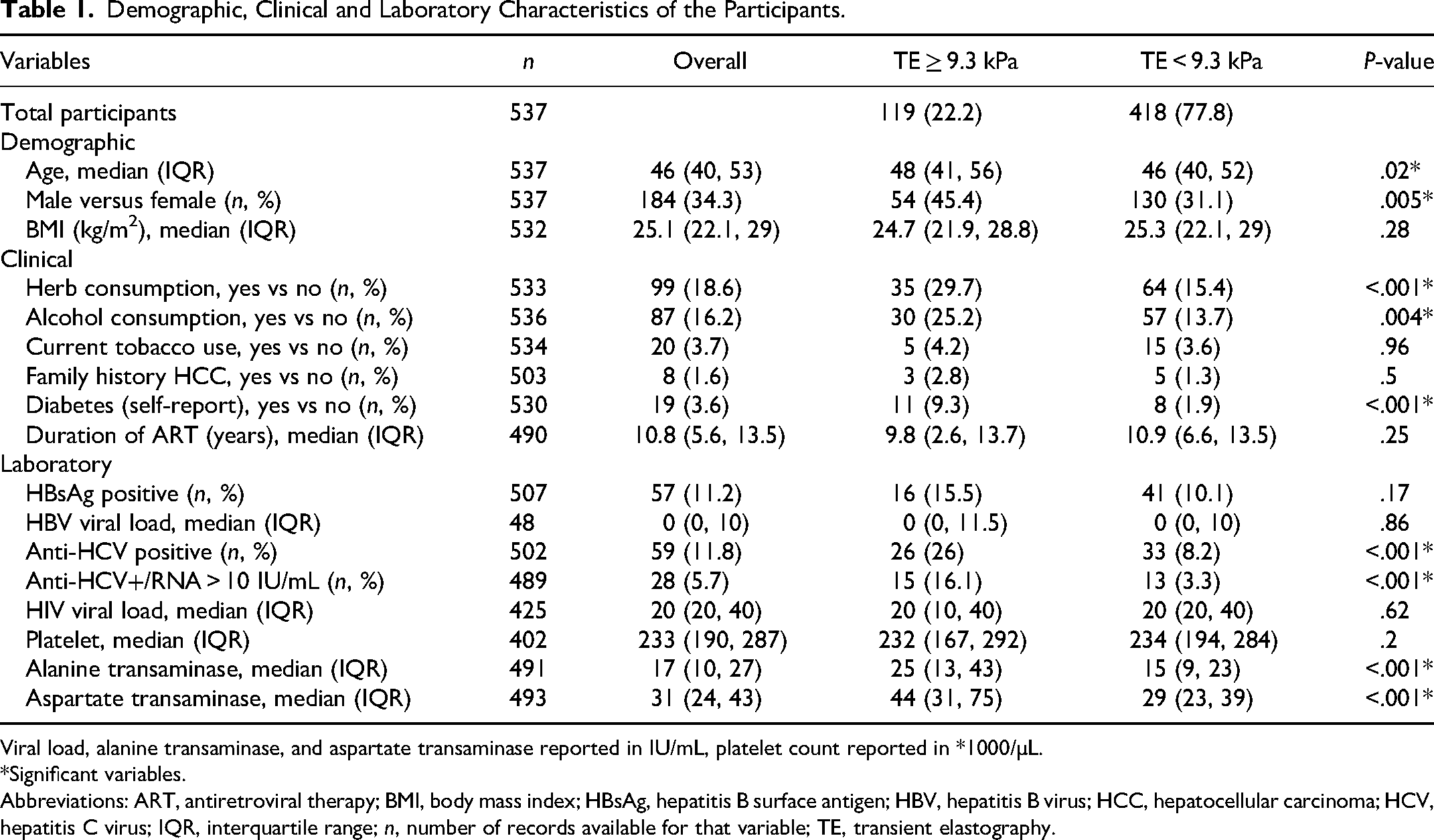
Viral load, alanine transaminase, and aspartate transaminase reported in IU/mL, platelet count reported in *1000/µL.
*Significant variables.
Abbreviations: ART, antiretroviral therapy; BMI, body mass index; HBsAg, hepatitis B surface antigen; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; IQR, interquartile range; n, number of records available for that variable; TE, transient elastography.
Factors associated with significant liver fibrosis/cirrhosis (TE ≥ 9.3 kPa). In multivariable analyses, factors independently associated with significant liver fibrosis/cirrhosis were male sex, diabetes, ART use, HBsAg seropositivity, and viremic HCV infection after adjusting for known risk factors for significant liver fibrosis/cirrhosis. ART use for more than 5 years reduced the odds of significant liver fibrosis/cirrhosis by 50%. HBsAg seropositivity and viremic HCV infection increased the odds by greater than 2 and 7 times, respectively. Age, BMI, and consumption of herbs or alcohol were not associated with significant liver fibrosis/cirrhosis after adjusting for other risk factors (Table 2). Using the lower TE cutoff value of ≥7.6 kPa obtained from a prior validation study, 27 only HBV and HCV were associated with liver fibrosis/cirrhosis, while ART use for more than 5 years reduced the risk of liver fibrosis/cirrhosis by 40%.
Factors Associated With Significant Liver Fibrosis/Cirrhosis.
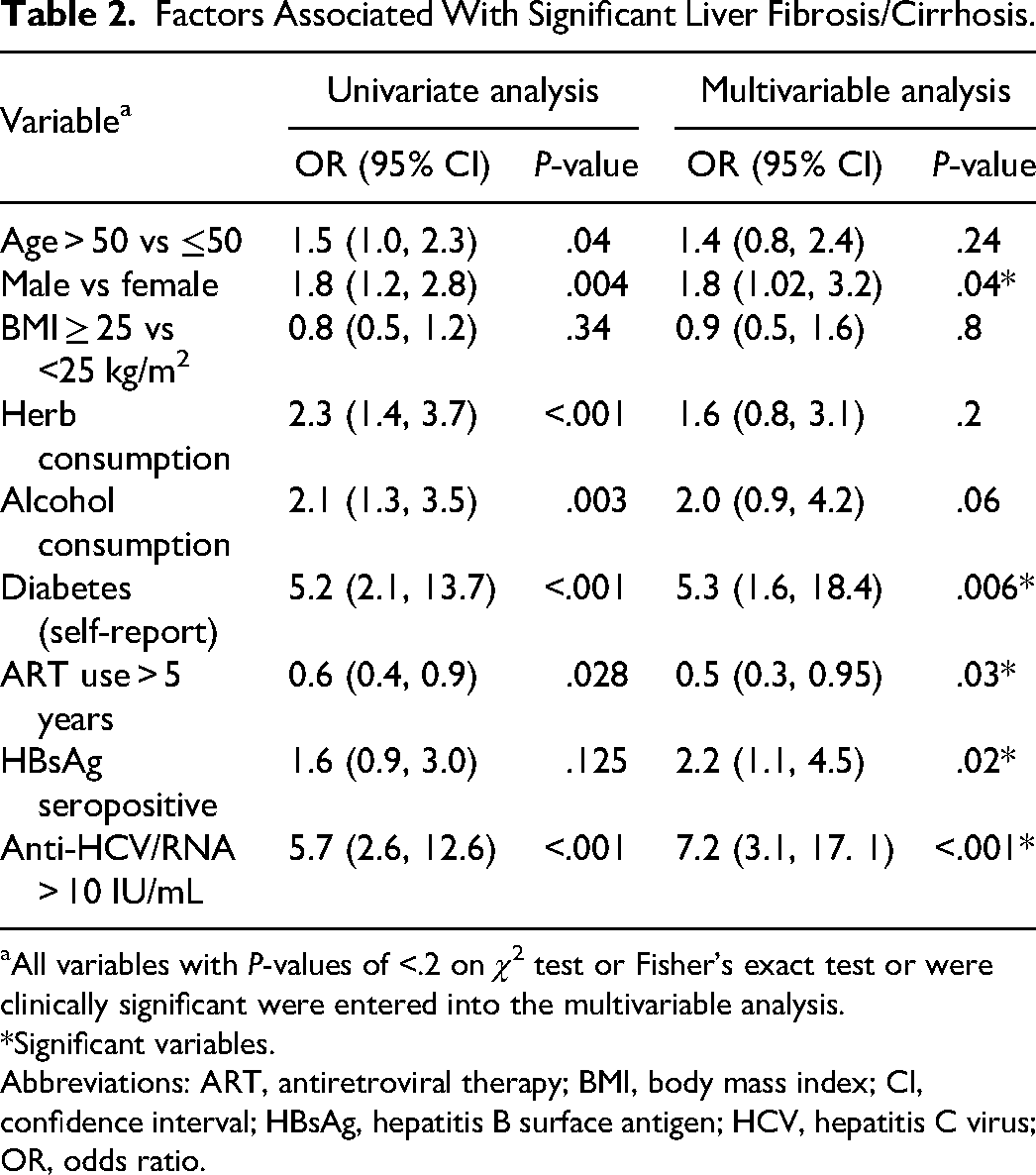
aAll variables with P-values of <.2 on χ2 test or Fisher's exact test or were clinically significant were entered into the multivariable analysis.
*Significant variables.
Abbreviations: ART, antiretroviral therapy; BMI, body mass index; CI, confidence interval; HBsAg, hepatitis B surface antigen; HCV, hepatitis C virus; OR, odds ratio.
Discussion
In this study, we compared demographic, clinical, and laboratory characteristics in PLWH with and without significant liver fibrosis/cirrhosis, examining more closely the associations between viral hepatitis co-infection and significant liver fibrosis/cirrhosis.
To our knowledge, this is the first study from Nigeria and one of the few from Africa to report an association between chronic HCV infection confirmed with molecular testing, and significant liver fibrosis/cirrhosis in PLWH. Most studies to date have only measured and reported the prevalence of antibodies to HCV which does not distinguish between active and past infection. In this study, 6% had chronic HCV. Our data is similar to 2 other Nigerian studies17,18 of PLWH where virologic testing was also performed, and higher than that reported in the general Nigerian population, 28 where only anti-HCV serological testing was performed. HCV has been reported to be highly prevalent (8%-28%) in other at-risk groups such as those with HCC or chronic liver disease,15,24,29,30 though in these studies, molecular testing was not done. The high prevalence of chronic HCV in this study may be explained by the shared routes of transmission of HCV and HIV which may include contaminated blood, needle sharing, scarification, and sexual activity.31,32 In this study, HCV increased the odds of significant liver fibrosis/cirrhosis more than 7 times. Other studies from outside Africa have also reported a strong association between HCV and significant liver fibrosis/cirrhosis in patients with HIV infection, using different TE cutoffs (≥7.1 and ≥9.4 kPa, respectively).33,34 Globally, direct-acting antiviral (DAA) therapy for the treatment of HCV has been available since 2013, resulting in cure rates of over 90%, regression of liver fibrosis, and improved survival.35–37 In Nigeria, DAA agents are available, however, access is limited because the drugs are mostly paid for out of pocket. 38 This data emphasizes need for more widespread access to DAA in Nigeria. Expansion of health insurance access and availability of government subsidies for HCV laboratory testing and treatment are some of the ways to help mitigate high costs.
In this study, we also found that chronic HBV infection increased the odds of liver fibrosis in PLWH, similar to several other studies from African settings.6,9 Chronic inflammation from HBV leads to repeated injury and activation of the fibrogenic pathways, resulting in chronic activation of fibrogenesis. This leads to increased synthesis and decreased degradation of fibrogenic collagen, resulting in its deposition in the extracellular matrix that surrounds the lobules, which is the hallmark of fibrosis. 39 Interestingly, HBsAg seropositivity was independently associated with fibrosis despite high rates of HBV viral suppression and a relatively long duration of ART (median 11 years) that included 2 active HBV agents. Fibrosis regression has been reported widely among PLWH with HBV on long-term ART in African and non-African settings.6,40,41 Vinikoor et al 41 reported a reduction in the prevalence of significant fibrosis from 14% to 6.7% and cirrhosis from 2.2% to 1.1% following a year of ART in persons with HIV and HIV/HBV from Zambia. 41 However, other studies have reported fibrosis progression in up to 30% of patients with HIV/HBV co-infection despite effective ART, 42 and persistently higher rates of fibrosis during follow up compared to those with HIV alone. 12 In Nigeria, and other countries with high HBV prevalence, HBV infection is frequently acquired in early childhood, thus predating HIV infection. 43 It is therefore possible for fibrosis to already be present at the time of HIV acquisition, and this may progress despite effective ART. While some studies have reported an independent association between HBV viremia and liver fibrosis, others have reported only associations with HIV-related factors such as, HIV viremia, and low CD4T cell counts, particularly before and early after ART initiation.9,12,13 This suggests multiple factors play a role in fibrosis progression which may differ according to stage of HBV and HIV infection, and is something that requires further evaluation. Nevertheless, it is clear that the large majority benefit from HBV active ART, and thus immediate screening of PLWH for HBV, treatment with HBV active ART and assessment for liver damage remain a priority in this population as well as a search for more effective HBV therapies to improve outcomes in these people. 43
In this study, we report other factors that were associated with fibrosis/cirrhosis, although only when the higher cutoff of 9.3 kPa was used. The association between male sex and liver fibrosis has been reported in other studies in the general population and in PLWH.41,44,45 Studies show that, independent of age, younger women, with viral hepatitis but without HIV infection, have less severe liver fibrosis than age-matched male peers.46–48 This may be due to the protective effect of estrogen in younger women via its suppression of the secretion of tumor necrosis factor-alpha and other cytokines that are involved in fibrogenesis.46,47,49,50 Other studies have found no association between male sex and liver cirrhosis.6,9,51 Different participant characteristics, such as higher rates of overweight/obesity and consumption of herbs in our study, may explain some of the discrepancy in findings.
Diabetes, albeit not very common and self-reported, was also independently associated with significant liver fibrosis/cirrhosis in this population. Diabetes has been reported commonly as a risk factor for fibrosis in several other studies both in persons with and without HIV.52,53 Insulin resistance is associated with progression of hepatic fibrosis, and is a central component of NAFLD in diabetes. 54 Several mechanisms have been proposed including genetic factors, hepatic fat accumulation, alterations in energy metabolism, and inflammatory signals derived from various cell types. 54 Hyperglycemia stimulates the hepatic stellate cells which are involved in fibrogenesis. 55 Our data, although preliminary, suggests that persons with diabetes may benefit from more routine targeted liver fibrosis screening particularly if other risk factors are present. 56
Our study had some limitations. A relatively small number of participants were included and were recruited from only 2 tertiary centers in Nigeria which may limit generalizability to other settings. Participants with and without liver fibrosis were intentionally enrolled and this could have introduced bias. The TE cutoffs used in this study to distinguish between those with and without significant fibrosis/cirrhosis were based on a previous validation study that was conducted in a population of predominantly black Americans, and reported a sensitivity of 86% for TE ≥ 9.3 kPa for the diagnosis of significant fibrosis/cirrhosis. 25 It is possible that some participants with more advanced stages of fibrosis could have been included in the TE < 9.3 kPa group. However, to account for this possibility, we repeated the analysis with a lower TE cutoff (from another validation study) and viral hepatitis remained associated with significant fibrosis/cirrhosis. Despite these limitations, our study has significant value that addresses research gaps in Africa, including the strong association of chronic hepatitis C with significant liver fibrosis/cirrhosis.
Conclusion
Chronic viral hepatitis (HCV and HBV) are associated with liver fibrosis in PLWH in Nigeria, while long-term ART use may reduce this risk. Strategies to improve HBV and HCV awareness and screening among Nigeria PLWH and promote access to treatment among those who are co-infected, are all critical to prevent further transmission, reduce morbidity, and mortality associated with these infections.
Footnotes
Acknowledgements
We thank Professor Folasade Ogunsola for her contributions to this work.
Author Contribution
OL, EO, AA, WA, FA, GI, AS, RM, LH, and CH conceptualized and designed the study. EO, GK, MO, MD, PD, DN, OL, and EO collected the data. EO, GO, HA, OO, BJ, IK, AC, and CH analyzed and interpreted the data. EO and CH drafted the work. All authors critically revised the manuscript for important content. All the authors approved the final version of the manuscript, including the authorship.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Division of Cancer Prevention, National Cancer Institute, grant no.: NIH/NCI U54CA221205 (PI Robert Murphy, Lifang Hou).
Ethical Statement
This study was approved by the Health Research Ethics Committees of the College of Medicine, University of Lagos, Lagos, Nigeria (approval number CMUL/HREC/01/18/326) on March 12, 2018 and Jos University Teaching Hospital, Jos, Nigeria (approval number JUTH/DCS/ADM/127/XXVII/629) on November 27, 2017. All participants provided written informed consent prior to enrollment in the study.
