Abstract
Purpose:
The prognosis of hepatocellular carcinoma (HCC) has recently improved, and so clinicians have greater opportunity to treat HCC-derived spinal metastases. Therefore, predicting life expectancy is important for determining the optimal treatment strategy for such tumors. This study aimed to investigate the prognostic factors for HCC-derived metastatic spine tumors and to develop a scoring system for predicting life expectancy in such cases.
Methods:
The posttreatment survival time and factors that might influence it were investigated in 62 patients with HCC-derived metastatic spine tumors who were treated at our department (surgery: 27 patients and conservative treatment: 35 patients), and a prognostic scoring system for predicting life expectancy was developed by combining the factors that significantly influenced survival.
Results:
In the univariate analyses, sex, the patient’s general condition, the presence/absence of major internal organ metastasis, the total revised Tokuhashi score, the serum albumin level, Child-Pugh class, spinal surgery, and bone-modifying agent (BMA) treatment were found to influence the posttreatment survival time. These factors were subjected to multivariate analysis, and a novel scoring system for predicting life expectancy based on the patient’s general condition, the serum albumin level (or Child-Pugh class), and BMA treatment was developed. In the retrospective analysis, the concordance rate between the patients’ life expectancy and actual survival times was 90.3%.
Conclusion:
The patient’s general condition, the serum albumin level (or Child-Pugh class), and BMA treatment influenced the posttreatment survival times of patients with HCC-derived metastatic spine tumors. A prognostic scoring system based on these factors was proposed.
Keywords
Introduction
The prognosis of hepatocellular carcinoma (HCC) is very poor, and the life expectancy of HCC patients was previously reported to be about 3–4 months. 1 {Tralhao, 2007 #454}{Tralhao, 2007 #454} With recent advances in surgical treatment and transcatheter arterial chemoembolization (TACE), the life expectancy of HCC patients has increased to more than 2 years. 1 As a result, epidural spinal cord compression due to HCC-derived spinal metastasis is encountered increasingly frequently even in cases in which the primary lesion is under control. 2 {Zhang, 2013 #455}{Zhang, 2013 #455}{Zhang, 2013 #455} However, the prognosis of HCC-derived spinal metastasis is still poor. 3 Accordingly, many researchers consider that the surgical indications for such tumors should be carefully investigated 4 and being able to accurately estimate life expectancy is important for determining the optimal treatment strategy for such tumors. 2 Thus, we investigated the factors that influence the post-treatment survival time in patients with HCC-derived metastatic spine tumors.
Materials and methods
Study design
This was a retrospective study.
Subjects
At our hospital, 562 HCC patients were treated between April, 2000 and December 2016. The cases of 62 patients with HCC-derived metastatic spine tumors who were examined and treated at our department between 2000 and December 2016 (surgical treatment: 27 patients and conservative treatment: 35 patients) were investigated.
At our institution, the surgical indications for HCC-derived metastatic spine tumors are as follows: (1) pain and/or paralysis caused by spinal instability or (2) spinal cord invasion by the tumor. However, surgery (posterior decompression and fusion, or posterior fusion alone) is only indicated for patients with a Karnofsky’s performance status (KPS) of >50% and a Child-Pugh score of 5–8 points. In addition, a life expectancy of >6 months is considered ideal. Patients with encephalopathy, moderate ascites, a platelet count of <100,000 mL−1, or an international normalized ratio of >1.7 were indicated for conservative treatment. The combination of treatments (adjuvant therapy) was not restricted in either the surgical treatment group or the conservative treatment group, and multidisciplinary treatments were employed as often as possible.
In the surgical treatment group, 13 patients underwent posterior decompression and fusion and 14 patients underwent posterior fusion alone. There were no patients who underwent decompression alone in this study. In the conservative treatment group, two patients received care for their symptoms alone, and adjuvant therapy was administered to the other 33 patients. Several patient-requested treatments were administered after it had been confirmed that the patient was in a suitable condition. The chemotherapy included TACE, cisplatin, and sorafenib, a molecular-targeting drug. Sorafenib was introduced in 2008. As for radiotherapy, local fractionated radiotherapy involving a dose of 24–30 Gy was normally administered. Regarding bone-modifying agent (BMA) treatment, zoledronic acid, a bisphosphonate, or denosumab, an anti-receptor activator of nuclear factor kappa-B ligand (RANKL) antibody, was used from 2009 onward. BMA treatment has become an almost routine treatment for solid cancers at our institution, provided there are no specific contraindications. A breakdown of the administered adjuvant treatments is provided as follows: posterior stabilization surgery with decompression (n = 13): chemotherapy, 3; radiotherapy, 9; BMA treatment, 3; and no adjuvant therapy, 1. Posterior stabilization surgery (n = 14): chemotherapy, 7; radiotherapy, 7; BMA treatment, 8; and no adjuvant therapy, 1. Conservative treatment (n = 35): chemotherapy, 12; radiotherapy, 29; BMA treatment, 10; and no adjuvant therapy, 2. The mean and median post-treatment (for the spinal metastasis) survival times were 7.0 (±7.7) and 4.0 months (interquartile range: 2.0–11.0), respectively.
Regarding the factors that might influence the posttreatment survival time, sex, age, the affected vertebral level (symptomatic level), the parameters of the revised Tokuhashi score (the patient’s general condition, the number of extraspinal bone metastases, the number of vertebral metastases, the presence/absence of major internal organ metastasis, and the patient’s paralysis state) (Table 1), 5 the total revised Tokuhashi score, the serum levels of albumin and α-fetoprotein (AFP), and the Child-Pugh class were retrospectively investigated as pretreatment factors. As therapeutic intervention-related factors, the presence/absence of chemotherapy before the development of metastasis, surgical treatment for spinal metastasis, BMA treatment, and sorafenib treatment were also investigated. Based on our findings, a novel scoring system for predicting life expectancy was designed by combining the factors that were found to significantly influence the posttreatment survival time.
A revised Tokuhashi score.

KPS: Karnofsky’ s performance status.
All procedures performed in studies involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Institutional review board approval was obtained from Nihon University Itabashi Hospital (approval number: RK-11209-8).
Statistical analysis
Univariate analyses of the factors that might influence the posttreatment survival time were performed using the t-test, Welch’s method, or analysis of variance. The factors that exhibited significance in the univariate analyses were subjected to multivariate analysis using Cox regression analysis. All statistical analyses were carried out using StatMate V® (Atoms Co., Tokyo, Japan). The p-values of <0.05 were considered significant.
Results
Univariate analyses of the factors that might influence the posttreatment survival time
Sex
Fifty-six and six patients were male and female, respectively (Table 2). The mean survival times of the males and females were 8.0 ± 8.6 (range 0.3–36) and 3.9 ± 3.4 (range 0.5–9) months, respectively (p = 0.04341).
Significant differences of survival time (months).

a Mean p-value was <0.05 and considered statistically significant.
Age
The mean survival times of the patients aged ≤69 years (n = 37) and ≥70 years (n = 25) were 7.3 ± 7.4 and 7.9 ± 9.6 months, respectively (p = 0.7945).
Affected vertebral level (symptomatic level)
The cervical, thoracic, and lumbosacral vertebrae were affected in 13, 32, and 17 patients, respectively. The mean survival times of these three groups were 4.8 ± 3.9, 8.3 ± 8.5, and 8.4 ± 10.3 months, respectively (p = 0.3932).
The revised Tokuhashi score parameters
Each factor included in the Tokuhashi score 5,6 was investigated (Table 2).
General condition (performance status)
The patients were divided into three groups based on their general condition according to the KPS; that is, into poor (KPS: 10–40%) (n = 5), moderate (KPS: 50–70%) (n = 17), and good (KPS: 80–100%) (n = 40) groups. The mean survival times of the poor, moderate, and good groups were 1.7 ± 0.9, 3.9 ± 3.3, and 9.9 ± 9.4 months, respectively, and a significant difference was noted between the moderate and good groups (p = 0.01036).
Number of extraspinal bone metastases
The patients were divided into three groups based on the number of extraspinal bone metastases that they possessed; that is, into those with ≥3 (n = 35), 1–2 (n = 12), and no (n = 15) extraspinal bone metastases. The mean survival times of these groups were 6.0 ± 7.7, 7.7 ± 10.1, and 11.1 ± 7.7 months, respectively (p = 0.1401).
Number of vertebral metastases
The patients were divided into three groups based on the number of vertebral metastases that they possessed; that is, into those with ≥3 (n = 33), 2 (n = 12), and 1 (n = 17) vertebral metastases. The mean survival times of these groups were 7.0 ± 8.7, 6.5 ± 5.9, and 9.4 ± 9.2 months, respectively (p = 0.5346).
Major internal organ metastasis
The mean survival times of the patients with (n = 48) and without (n = 14) metastasis to the major organs were 5.3 ± 6.2 and 15.2 ± 10.2 months, respectively (p < 0.001).
Paralysis
Paralysis was graded based on the Frankel classification. The mean survival times of the patients with complete (Frankel A or B, n = 7), incomplete (Frankel C or D, n = 34), and no (Frankel E, n = 21) paralysis were 5.3 ± 3.8, 8.1 ± 9.2, and 7.5 ± 8.0 months, respectively (p = 0.0699).
Total revised Tokuhashi score
When the patients were classified according to their life expectancy based on the total Tokuhashi score. The mean survival times of the patients with life expectancies of <6 months (score: ≤8) (n = 53) and ≥6 months (score: 9–10) (n = 9) were 6.2 ± 7.5 and 15.8 ± 8.6 months, respectively (p < 0.001).
Laboratory data
Regarding blood test parameters, the serum albumin level, serum AFP level, and the Child-Pugh classification were investigated.
Serum albumin level
The patients were divided into those with serum albumin levels of <3.5 g/dL (n = 34) and ≥3.5 g/dL (n = 28) based on the cut-off point used in the Child-Pugh classification. The mean survival times of these groups were 3.5 ± 3.0 and 12.5 ± 9.9 months, respectively (p < 0.001).
Serum AFP level
The patients were divided into those with serum AFP levels of >32 ng/mL (n = 43) and ≤32 ng/mL (n = 19). The mean survival times of these groups were 6.5 ± 6.4 and 9.9 ± 11.4 months, respectively (p = 02338).
Child-Pugh classification
The patients were divided into those with class B (n = 28) and class A (n = 34) disease according to the Child-Pugh classification. The mean survival times of these groups were 3.0 ± 2.5 and 11.4 ± 9.5 months, respectively (p < 0.001).
Factors related to the therapeutic intervention
The presence/absence of chemotherapy before the development of spinal metastasis, surgical treatment for spinal metastasis, BMA treatment, and sorafenib treatment were investigated as therapeutic intervention-related factors.
Chemotherapy before the development of spinal metastasis
The mean survival times of the patients that were (n = 54) and were not (n = 8) treated with chemotherapy before the development of spinal metastasis were 8.1 ± 8.6 and 4.3 ± 5.3 months, respectively (p = 0.1069).
Surgical treatment for spinal metastasis
The patients were divided into those who received surgical (n = 27) and conservative (n = 35) treatment for spinal metastasis. The mean survival times of these groups were 10.4 ± 8.5 and 5.4 ± 7.6 months, respectively (p = 0.01671).
BMA treatment
Regarding the BMA used, zoledronic acid, a bisphosphonate (n = 7), or denosumab, an anti-RANKL antibody (n = 20) was administered. The patients were divided into those that were (n = 27) and were not (n = 35) treated with BMA. The mean survival times of these groups were 10.6 ± 9.2 and 5.3 ± 6.8 months, respectively (p = 0.01598).
Sorafenib treatment
The patients were divided into those that were (n = 6) and were not (n = 56) treated with sorafenib. The mean survival times of these groups were 8.9 ± 7.9 and 7.4 ± 8.4 months, respectively (p = 0.6889).
Multivariate analysis of the factors that might influence the posttreatment survival time
The factors that might influence the post-treatment survival time and the results of the univariate analyses are shown in Table 2. Seven items were extracted as factors that influenced the post-treatment survival time in the univariate analyses: the patient’s general condition, the presence/absence of major internal organ metastasis, the serum albumin level, the Child-Pugh class, surgical treatment for spinal metastasis, and BMA treatment. Of these factors, the serum albumin level is included in the Child-Pugh classification (Table 3). Therefore, we created two sets of six factors, which included the serum albumin level or the Child-Pugh classification and the other factors that exhibited significance in the univariate analyses. Multivariate analyses of the relationships between each of the six items in each set and the posttreatment survival time were then performed using Cox regression analysis.
Multivariate analysis of the prognostic factors affecting survival.

KPS: Karnofsky’ s performance status.
a Mean p-value was <0.05 and considered statistically significant.
As a result, two sets of three significant factors were extracted: (1) the patient’s general condition, the serum albumin level, and BMA treatment and (2) the patient’s general condition, the Child-Pugh class, and BMA treatment (Table 3).
A novel scoring system for predicting life expectancy in patients with metastatic spine tumors derived from HCC
Based on the hazard ratios for each set of three factors, a new scoring system for predicting the life expectancy of patients with HCC-derived metastatic spine tumors was created (Table 4), in which the maximum scores for the patient’s general condition, the serum albumin level or the Child-Pugh classification, and BMA treatment were set to 2, 2, and 2 points (Table 4), respectively, and a significant relationship between the total score and survival time was detected regardless of whether the serum albumin level (Figure 1; y = −2.289 + 2.955x, r = 0.6263, r 2 = 0.3922, p < 0.001) or the Child-Pugh classification (Figure 2; y = −2.169 + 2.759x, r = 0.5932, r 2 = 0.3519, p < 0.001) was used. Based on the relationship between the patients’ 6-point predictive scores and their actual survival times, the following criteria for predicting life expectancy were postulated: Total scores of 0–2 points, 3–5 points, and 6 points indicate a life expectancy of <6, <12, and ≥6 months, respectively (Table 4 and Figure 3).
A novel scoring system for predicting life expectancy in patients with metastatic spinal tumors derived from hepatocellular carcinoma.
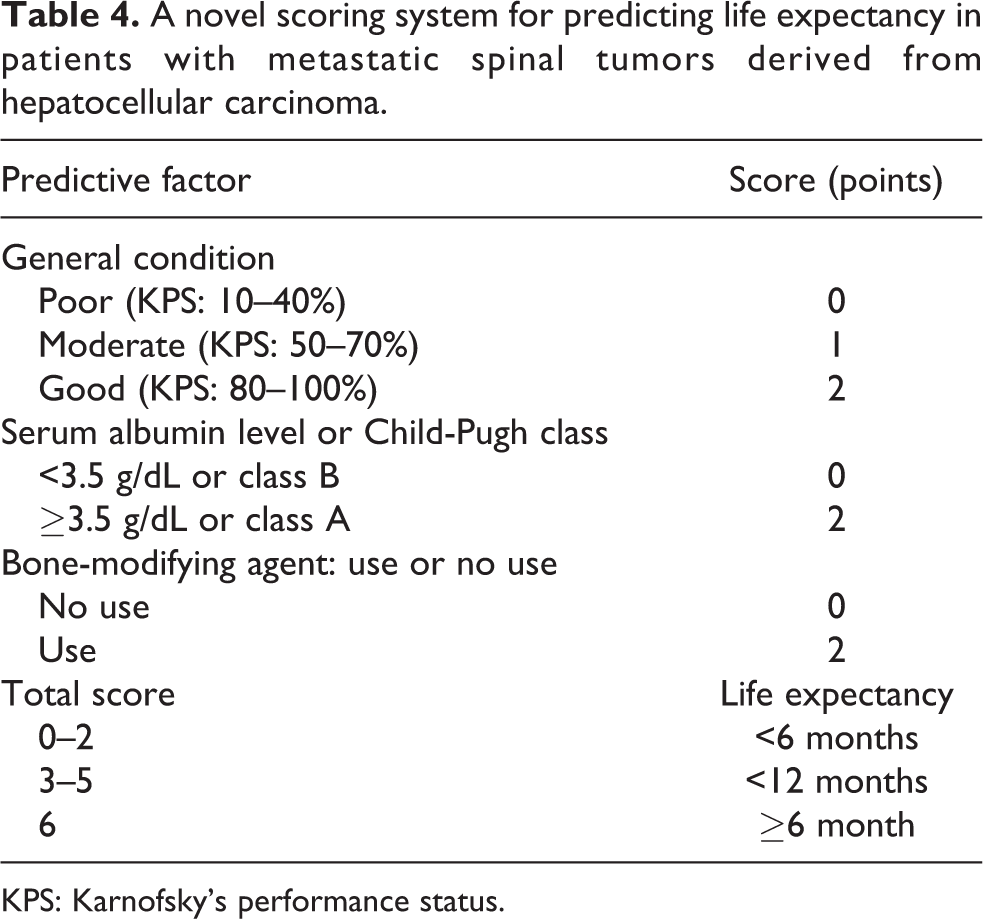
KPS: Karnofsky’s performance status.

The relationship between the total score and survival time in the scoring system including the serum albumin level.

The relationship between the total score and survival time in the scoring system including the Child-Pugh classification.

A six-point scoring system for predicting the prognosis of patients with metastatic spine tumors derived from hepatocellular carcinoma.
When the cases of the 62 patients were retrospectively investigated, the concordance rate between the life expectancy predictions based on the abovementioned criteria and the patients’ actual survival times was 90.3% (Table 5). These results were compared with those obtained using the revised Tokuhashi scoring system criteria. According to these criteria, scores of 0–8 points and 9–11 points are associated with prognostic predictions of <6 and ≥6 months, respectively. The concordance rate between the prognostic predictions based on the revised Tokuhashi score and the patients’ actual survival times (69.4%) was significantly lower than that obtained using the scoring system developed in this study (Table 5; p = 0.0167 when the serum albumin level was used and p = 0.0317 when the Child-Pugh classification was used).
Six-point predictive score vs. survival time (months).

Discussion
There are regional variations in the incidence of HCC and it is particularly high in Southeast Asia as well as in Taiwan, Korea, China, and Japan in East Asia. 7 HCC is associated with hepatitis C virus infections and alcohol intake. 8 The survival times of patients with primary HCC have increased with recent advances in treatment, but similar improvements in life expectancy have not been achieved in patients with spinal metastasis. It has been reported that the outcomes of patients with spinal metastasis derived from HCC are very poor (median overall survival time: 108 days). 3 The median survival time after the diagnosis of HCC-derived bone metastasis is generally about 7–10 months, 9 and it was 7.0 ± 7.7 months in the present study. However, 9 (18.0%) of the 50 patients survived for >1 year. Therefore, the effects of surgery on paralysis and patients’ ability to perform activities of daily living can only be assessed in some cases, and so an appropriate system for predicting the outcomes of HCC patients with spinal metastasis is required.
Performance status was the first factor that was reported to influence the prognosis of HCC patients with spinal metastasis, 10 and the severity of paralysis has also been frequently reported to affect the prognosis of such patients. 11 –13 However, using multivariate analysis, Zhang et al. found that the severity of paralysis was not a significant predictor of the prognosis of HCC patients with spinal metastasis, although it was identified as a significant prognostic predictor in their univariate analyses. 2 Similarly, the severity of paralysis was not identified as a significant prognostic factor in our study.
Zhang et al. highlighted the importance of the Tomita score 14 as a prognostic factor in HCC patients with spinal metastasis. The Tomita score is based on the grade of malignancy of the primary tumor, visceral metastases, and bone metastases. HCC is considered to be a highly malignant, rapidly growing form of cancer, and these characteristics influence its prognosis. However, the presence/absence of major internal organ metastasis was not identified as a significant prognostic factor among our patients.
The presence/absence of ascites is another possible prognostic factor for HCC. 15 Normally, it is judged based on descriptions in patients’ medical records and abdominal computed tomography findings, but there are no clear criteria for diagnosing ascites. For this reason (and problems caused by missing data), we did not examine this factor in the present study.
Regarding laboratory test data, the serum albumin level, 16,17 lactate dehydrogenase level, 16,18 –20 serum AFP level, platelet count, and serum calcium level 21 have been proposed as prognostic factors for HCC patients with spinal metastases. In the current study, we examined the serum albumin level and the serum AFP level, and the former was identified as a significant prognostic factor.
The Child-Pugh score has been statistically and clinically validated as a means of quantifying hepatic function and predicting overall survival in patients with HCC in numerous studies. 9,22 According to our surgical indications for metastatic spine tumors, which are based on the Child-Pugh classification, patients with encephalopathy, moderate ascites, a moderately high bilirubin level, or a high international normalized ratio are excluded from such procedures. In the multivariate analysis of potential prognostic predictors conducted in the present study, the Child-Pugh classification was also identified as a significant prognostic predictor.
Various therapeutic intervention-related factors have been investigated as prognostic factors for HCC-derived spinal metastasis. We examined the following four items: the presence/absence of chemotherapy before the development of spinal metastasis, surgical treatment for spinal metastasis, BMA treatment, and sorafenib treatment. Surgical treatment for spinal metastasis and BMA treatment were extracted as significant factors in the univariate analyses, but neither of them was found to be significant in the multivariate analysis. The effects of bisphosphonates on HCC have recently been attracting attention. Bisphosphonates have been demonstrated to inhibit the proliferation and migration of HCC cells, 23 and they are considered to be effective at preventing bone resorption, hypercalcemia, and pathological fractures and inhibiting local recurrence. 24 –27 According to the multivariate analysis, the use of BMA significantly influenced survival among our study population. BMA are now routinely used to treat spinal metastases from all types of cancer. Thus, their prognostic effects might be an important issue for further investigation. In addition, stereotactic radiotherapy has the potential to affect the prognosis of patients with spinal metastases derived from HCC. 10 It is known to result in both longer survival times and better functional prognoses than conventional radiotherapy. 10,28 Therefore, its increasing use might represent a therapeutic paradigm shift.
The prognostic factors for spinal metastases derived from HCC have been investigated in many East Asian studies (Table 6). 9,10,13,16,29 –32 Recently, Rim et al. reported prognostic factors for spinal metastases derived from HCC in a study of 255 cases. As a result, they established a novel disease-specific scoring system and performed a validation study of it. However, new prognostic factors, that is, the AFP level and Child-Pugh class, were newly identified in the validation study. 31 This indicates that patients’ backgrounds might affect the prognostic factors for spinal metastases derived from HCC. For example, patients tend to survive longer in studies involving many surgical cases than in studies in which many patients are treated with radiotherapy (Table 6). In fact, patients’ backgrounds might vary considerably within the same study population. Although Rim et al. used a “fuzzy” prognostic factor, that is, the control of the primary HCC, there were many uncontrolled patients in the validation group, and the patients’ backgrounds varied considerably. Therefore, we must always consider patients’ backgrounds when evaluating reported prognostic factors for spinal metastases derived from HCC.
Prognostic factors derived from clinical outcomes of hepatocellular carcinoma patients with spinal metastasis.

R: retrospective study; M: meta-analysis; V: validation study; RT: radiotherapy; OP: operation; CT: chemotherapy; OS: overall survival; ECOG: Eastern Cooperative Oncology Group performance status; LDH: lactate dehydrogenase; KPS: Karnofsky’s performance status scale; HCC: hepatocellular carcinoma; AFP: alpha-fetoprotein; BMA: bone-modifying agent.
a No definitive description of median overall survival.
Furthermore, it is very likely that treatment interventions influence prognosis, even in cases of spinal metastasis. 9 Therefore, further studies aimed at identifying prognostic factors for HCC-derived spinal metastases and the development of novel prognostic scoring systems that include treatment intervention-related factors are required. From this point of view, the prognostic utility of BMA or sorafenib treatment, which are specific therapies for spinal metastases, needs to be evaluated.
The limitations of this study include the fact that it was a retrospective study rather than a prospective randomized study. At our hospital, 562 HCC patients were treated between 2000 and December 2016. However, only 62 patients were treated for HCC-derived metastatic spine tumors. It is possible that asymptomatic patients or patients with poor performance statuses were not included in the study. On the other hand, it should be noted that patients with HCC-derived metastatic spine tumors usually have poor prognoses, and few of them are eligible for systemic treatment. Furthermore, we must examine whether the use of new treatments, including stereotactic radiotherapy, has resulted in a therapeutic paradigm shift. Despite these limitations, the predictive value of our novel scoring system was high, even though it was comprised of a relatively small number of parameters. Therefore, we will repeat this study with a larger number of patients.
Conclusion
In conclusion, the patient’s general condition, the serum albumin level (or Child-Pugh class), and the administration of BMA influenced the post-treatment survival times of patients with HCC-derived metastatic spine tumors. A prognostic scoring system based on these factors was proposed. Further follow-up will be necessary to validate and monitor the relationship between the predicted prognoses and actual survival times of patients with HCC-derived metastatic spine tumors.
Supplemental material
Supplemental Material, ToreviewersHCCscorerevision - Prognostic scoring system for metastatic spine tumors derived from hepatocellular carcinoma
Supplemental Material, ToreviewersHCCscorerevision for Prognostic scoring system for metastatic spine tumors derived from hepatocellular carcinoma by Hiroshi Uei and Yasuaki Tokuhashi in Journal of Orthopaedic Surgery
Footnotes
Author contributions
HU designed this study, YT collected the data, HU and YT analyzed the data, HU edited the article, and YT approved the final version of the manuscript.
Availability of data and materials
The data sets obtained in this study are available from the corresponding author on reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
All procedures performed in studies involving human participants were conducted in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Institutional review board approval was obtained from Nihon University Itabashi Hospital (approval number: RK-11209-8).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
