Abstract
Study Design
Systematic review and clinimetric analysis.
Objectives
Frailty and sarcopenia predict worse surgical outcomes among spinal degenerative and deformity-related populations; this association is less clear in the context of spinal oncology. Here, we sought to identify frailty and sarcopenia tools applied in spinal oncology and appraise their clinimetric properties.
Methods
A systematic review was conducted from January 1st, 2000, until June 2022. Study characteristics, frailty tools, and measures of sarcopenia were recorded. Component domains, individual items, cut-off values, and measurement techniques were collected. Clinimetric assessment was performed according to Consensus-based Standards for Health Measurement Instruments.
Results
Twenty-two studies were included (42 514 patients). Seventeen studies utilized 6 frailty tools; the three most employed were the Metastatic Spine tumor Frailty Index (MSTFI), Modified Frailty Index-11 (mFI-11), and the mFI-5. Eight studies utilized measures of sarcopenia; the three most common were the L3-Total Psoas Area (TPA)/Vertebral Body Area (VBA), L3-TPA/Height2, and L3-Spinal Muscle Index (L3-Cross-Sectional Muscle Area/Height2). Frailty and sarcopenia measures lacked or had uncertain content and construct validity. Frailty measures were objective except the Johns-Hopkins Adjusted Clinical Groups. All tools were feasible except the Hospital Frailty Risk Score (HFRS). Positive predictive validity was observed for the HFRS and in select studies employing the mFI-5, MSTFI, and L3-TPA/VBA. All frailty tools had floor or ceiling effects.
Conclusions
Existing tools for evaluating frailty and sarcopenia among patients undergoing surgery for spinal tumors have poor clinimetric properties. Here, we provide a pragmatic approach to utilizing existing frailty and sarcopenia tools, until more clinimetrically robust instruments are developed.
Introduction
The NOMS (neurological, oncological, mechanical, systemic) framework was developed to provide a common language across medical and surgical disciplines and simplify the complicated decision-making process inherent to spinal oncology. 1 Evaluation of systemic condition remains an unaddressed pillar in the framework, which vaguely advises an estimation of patients’ physical reserve and the ability to tolerate surgery. Complexity in this process largely owes to the numerous pre-operative systemic variables that influence postoperative clinical outcomes in this patient population. 2 Given that older patients with multiple comorbidities constitute a substantial portion of patients with cancer, further research should strive to clarify the systemic component of the NOMS framework.
Frailty is an attractive surrogate for systemic condition and has been widely studied in medicine and surgery.3,4 Frailty may be operationalized as a phenotypic definition, or a composite index of accumulating deficits and diminished physical reserve.4–6 Increased frailty predicts worse clinical outcomes among patients undergoing spinal surgery for degenerative and deformity-related conditions,3,7 however, this association is less clear in the context of spinal oncology.8–11 An existing Metastatic Spinal Tumor Frailty Index was found to have poor clinimetric properties (e.g. content and predictive validity), 12 and discrimination for clinical outcomes. 9 Until recently, controversy existed regarding the definition and determinants of frailty among patients with spinal tumors. Consensus was attained among members of the AO Spine community, regarding a phenotypic definition of frailty and the association between 14 preoperative variables and frailty in the context of spinal metastatic disease, respectively. 13 This represents an important first step toward the development of a clinimetrically robust tool for use in this patient population.
Sarcopenia is an alternative indicator of frailty and surrogate for systemic condition, defined as a progressive loss of skeletal muscle mass and power. 10 Like frailty, sarcopenia has been found to predict functional decline and poor clinical outcomes in surgical populations,14–16 but not consistently among patients with spinal tumors.8,17,18 Various measurement techniques have been described, including the measurement of psoas area on axial CT imaging. Body composition analysis (BCA) expands on such traditional measurement techniques and may better capture total body sarcopenia.19–21 BCA may include a skeletal muscle index (SMI; paraspinal, psoas, and abdominal cross-sectional skeletal muscle quantity) and radiodensity (SMQ; skeletal muscle quality), as well as visceral and subcutaneous adiposity. 22 Recent advancements in the utilization of artificial intelligence and automated imaging analysis across surgical disciplines may offer improved feasibility and uptake of techniques like BCA into the clinical setting; however, the association with clinical outcomes remains to be determined.18,22,23
Further research should clarify the role of frailty and sarcopenia as surrogates for systemic condition in the context of surgical spine oncology. Here, we sought to gain insight regarding existing knowledge gaps by reviewing existing frailty and sarcopenia tools applied in surgical spinal oncology and assessing their clinimetric properties. Our specific study objectives included: (1) to identify frailty and sarcopenia tools used in spinal oncology (primary and metastatic disease); (2) appraise the objectivity, feasibility, and clinimetric properties of these tools; and (3) to assess and summarize the application of these tools in the context of risk stratification and outcome prognostication. These findings may facilitate the development of a clinimetrically robust tool for estimating systemic condition, stratifying risk, and predicting clinical outcomes in patients undergoing surgery for spinal tumors.
Methods
Study Design
The AO Spine Knowledge Forum Tumor conducted a systematic review that is reported according to the 2020 Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement (PRISMA checklist: Supplemental Content A).24,25 This study design was utilized in order to comprehensively synthesize studies applying tools toward the measurement of frailty and sarcopenia in surgical spinal oncology populations. The protocol for this study was registered with PROSPERO international prospective register for systematic reviews (registration number: CRD42022367938). 26 This study was Institutional Review Board exempt and informed consent was not sought, given that no identifiable health information was collected and only data from published studies were incorporated.
Search Strategy and Selection Criteria
Inclusion and Exclusion Criteria.

Study Selection
Study selection was performed using a two-step screening process. Covidence systematic review software was used to screen all results from the database searches. Upon upload of RIS files from each database, duplicate articles were automatically excluded by the software. A single reviewer (MAM) ensured accuracy of the duplicate removal algorithm, and additional duplicates were identified during screening. Institution and database of published studies were used to avoid inclusion of duplicate patient data. Two reviewers (MAM, MG) independently screened titles and abstracts using the pre-specified inclusion criteria. Selected full texts were assessed for eligibility and screened in duplicate. Cases of disagreement were resolved via open discussion.
Data Collection
Study Demographics.
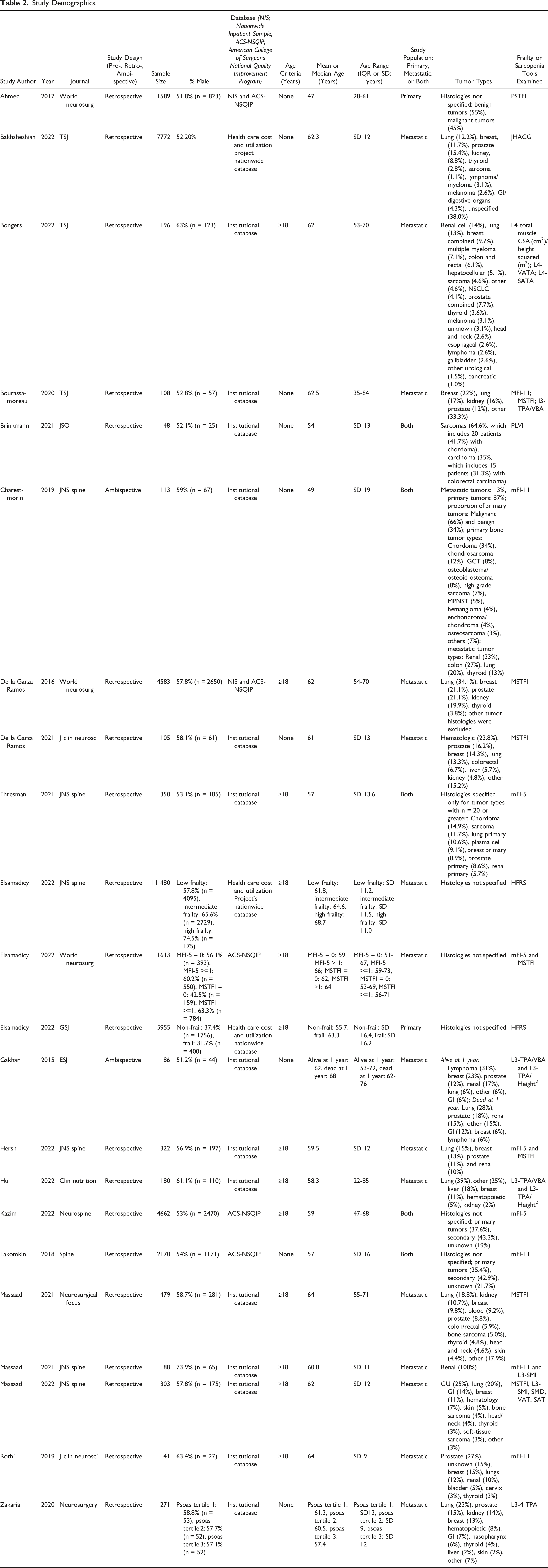
Tools for Assessing Frailty in Spinal Oncology.
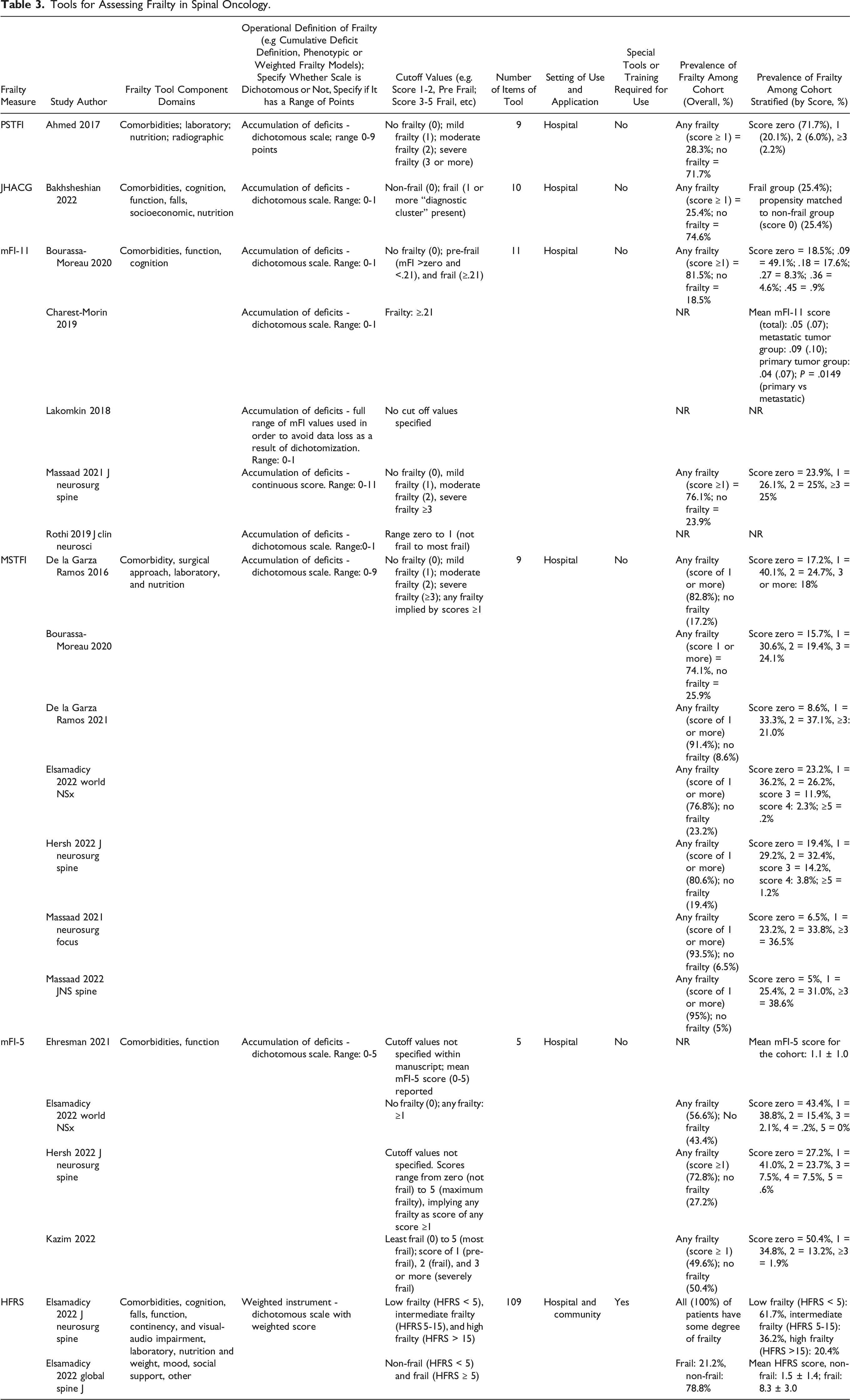
Tools for Measuring Sarcopenia in Spinal Oncology.
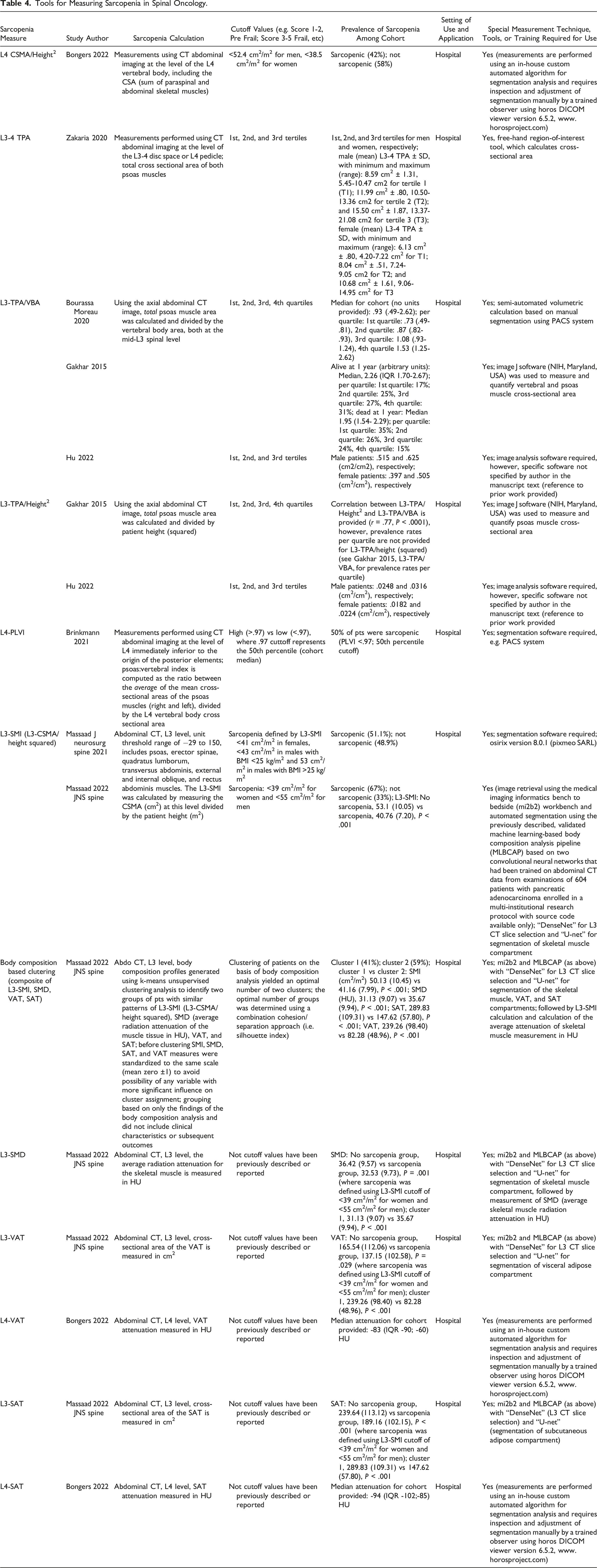
Cllinical Outcomes Frailty.
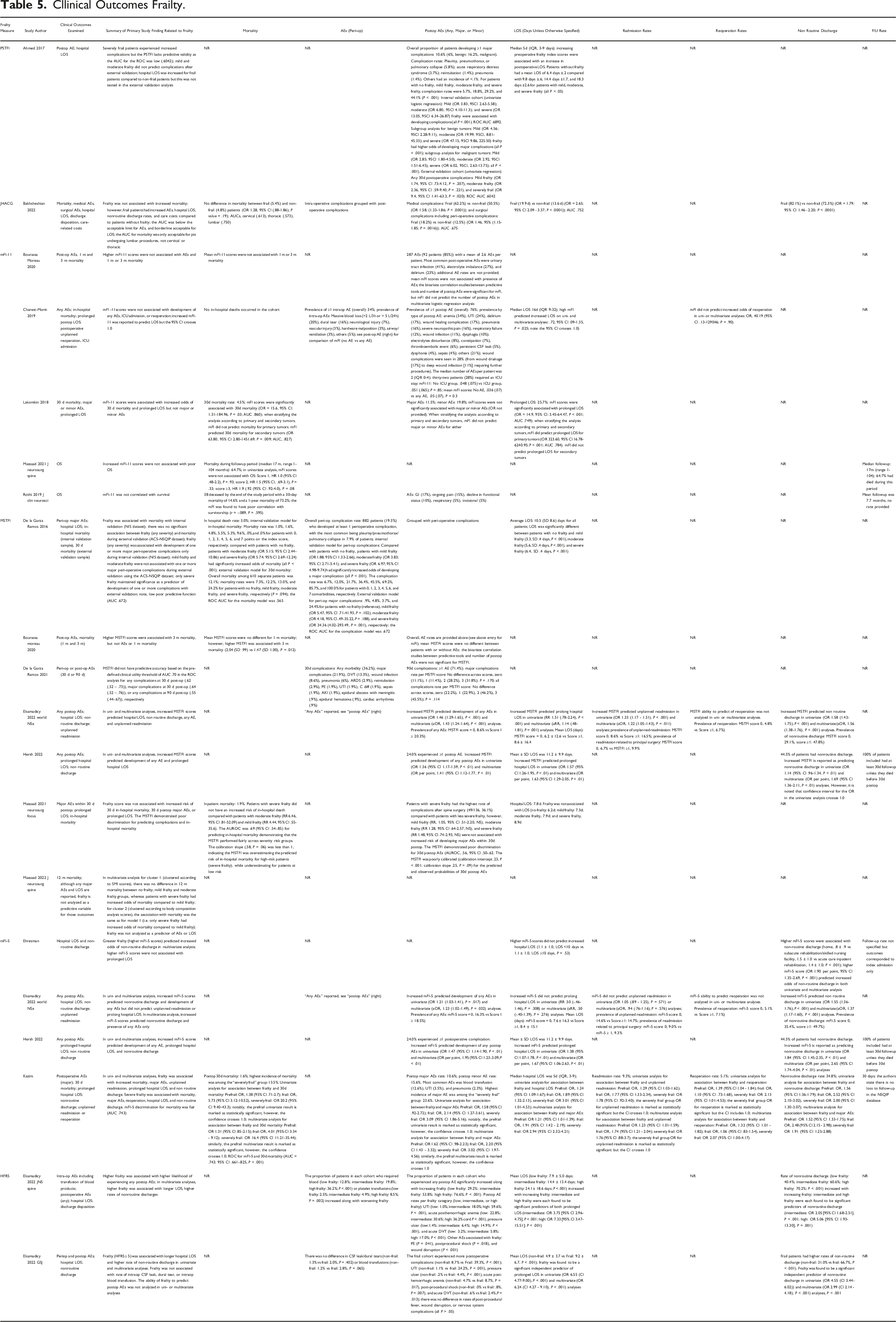
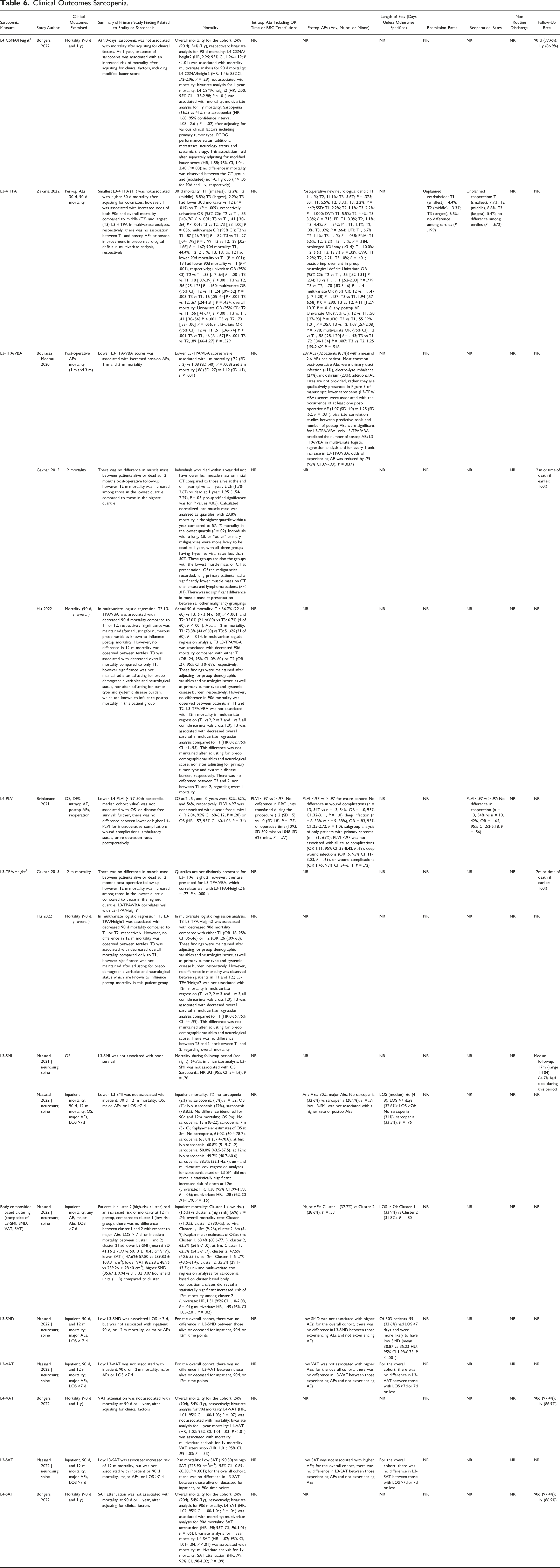
Clinical Outcomes Sarcopenia.
Clinimetric Assessment
Clinimetric Properties Frailty.
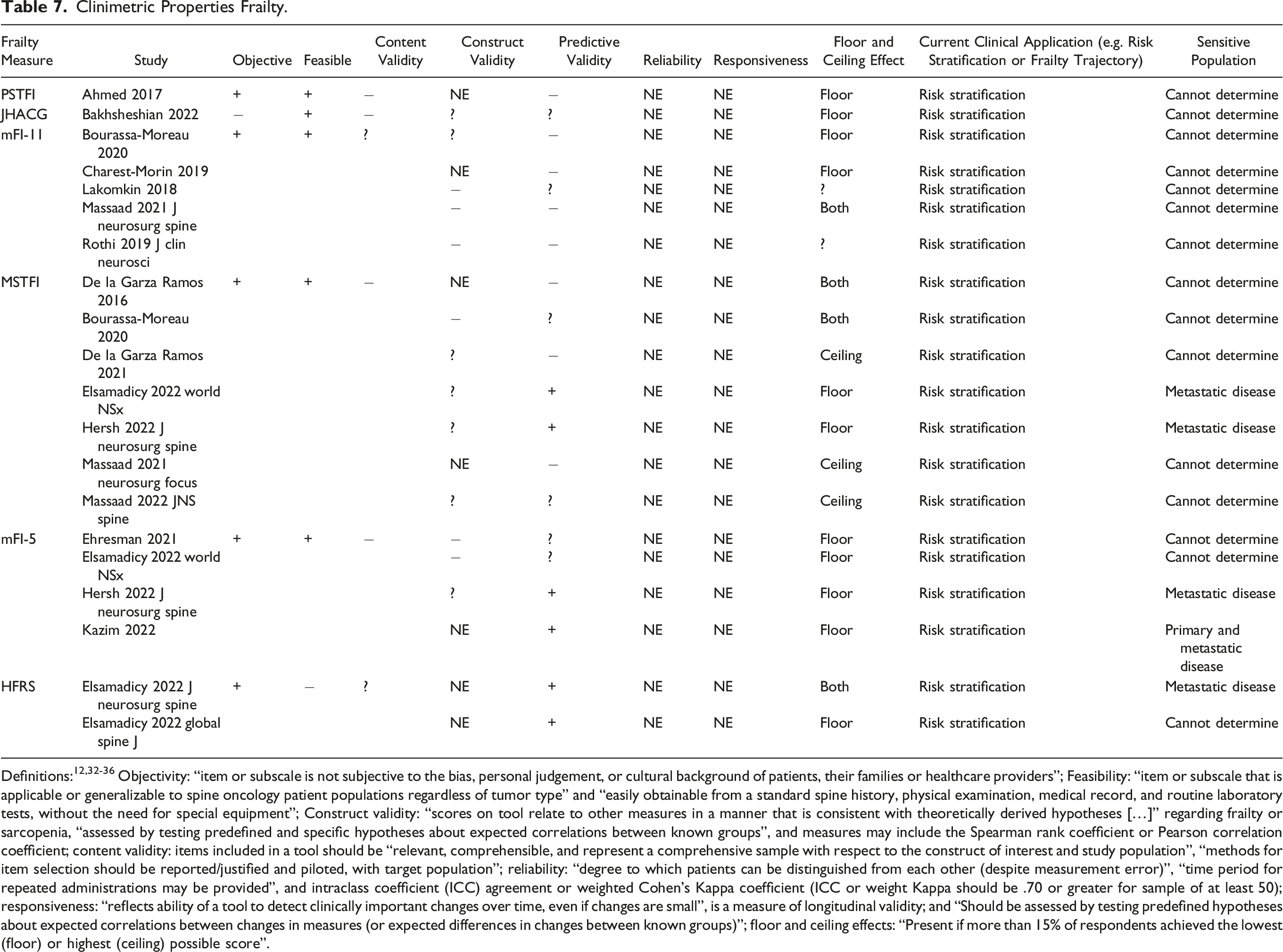
Definitions:12,32-36 Objectivity: “item or subscale is not subjective to the bias, personal judgement, or cultural background of patients, their families or healthcare providers”; Feasibility: “item or subscale that is applicable or generalizable to spine oncology patient populations regardless of tumor type” and “easily obtainable from a standard spine history, physical examination, medical record, and routine laboratory tests, without the need for special equipment”; Construct validity: “scores on tool relate to other measures in a manner that is consistent with theoretically derived hypotheses […]” regarding frailty or sarcopenia, “assessed by testing predefined and specific hypotheses about expected correlations between known groups”, and measures may include the Spearman rank coefficient or Pearson correlation coefficient; content validity: items included in a tool should be “relevant, comprehensible, and represent a comprehensive sample with respect to the construct of interest and study population”, “methods for item selection should be reported/justified and piloted, with target population”; reliability: “degree to which patients can be distinguished from each other (despite measurement error)”, “time period for repeated administrations may be provided”, and intraclass coefficient (ICC) agreement or weighted Cohen’s Kappa coefficient (ICC or weight Kappa should be .70 or greater for sample of at least 50); responsiveness: “reflects ability of a tool to detect clinically important changes over time, even if changes are small”, is a measure of longitudinal validity; and “Should be assessed by testing predefined hypotheses about expected correlations between changes in measures (or expected differences in changes between known groups)”; floor and ceiling effects: “Present if more than 15% of respondents achieved the lowest (floor) or highest (ceiling) possible score”.
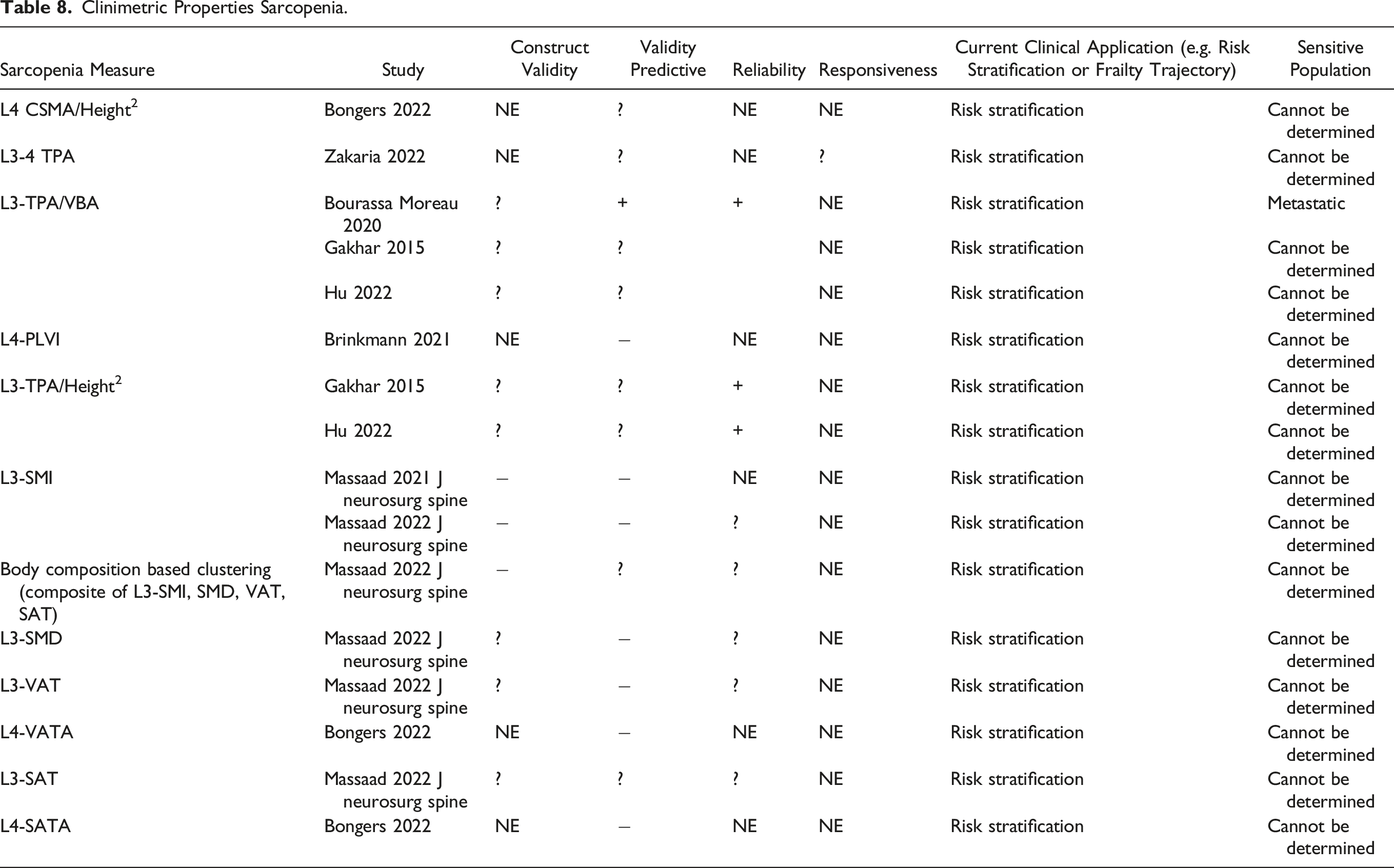
Clinimetric Properties Sarcopenia.
Study Bias and Evidence Appraisal
The Newcastle Ottawa Scale risk of bias assessment tool for observational studies was used to assess study bias (MAM, AC). 37 Included studies were also appraised by two reviewers (MAM, MG) using the “Oxford Levels of Evidence 2” grading system. 38 Disagreements were resolved via open, full-text review.
Results
Literature Search and Selection
Our initial database search yielded 1662 articles (Figure 1). After duplicates were removed, 1321 records were screened at the title/abstract level and 1273 articles were excluded. Full texts were obtained for 48 studies to assess eligibility. Twenty-six articles were excluded via full-text assessment and twenty-two articles were included for qualitative synthesis.8-11,17,18,22,39-53 PRISMA flow diagram.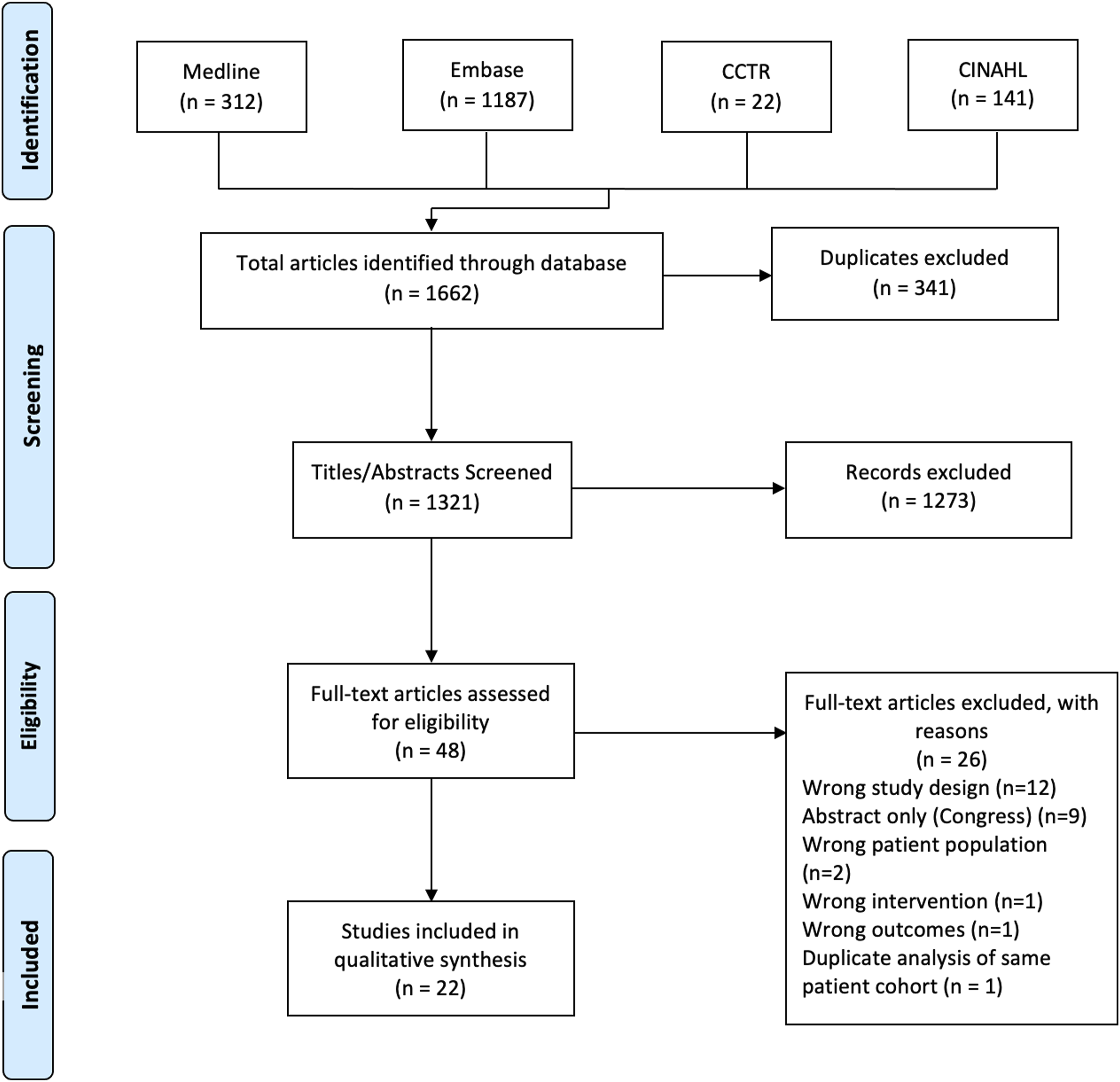
Study and Cohort Characteristics
Study descriptive characteristics are presented in Table 2. Study designs were either retrospective (n = 20; 91%) or ambispective (n = 2; 9%). The total patient sample across all studies was 42 514, including 23 786 (66%) and 18 728 (44%) male and female patients, respectively. The average age was 59.7 years. Fourteen (63%) studies analyzed institutional databases, whereas 3 (14%) studies used data from the Health care Cost and Utilization Project National Inpatient Sample (NIS) database, 3 (14%) used the American College of Surgeons National Surgery Quality Improvement Program (AC-NSQIP) database, and 2 (9%) used a combination of the NIS and the ACS-NSQIP database. Tumor types were metastatic (n = 15 studies; 68%), primary (n = 2 studies; 9%), or both (n = 5 studies; 23%). The three most common tumor histologies were lung, breast, and prostate.
Frailty Tools
Seventeen (77%) studies utilized frailty tools for risk stratification and outcome prognostication (Table 3). Tools included the PSTFI, JHACG, mFI-11, MSTFI, mFI-5, and HFRS. Components, domains, items, and their modifiability are described in Supplemental Content D. The MSTFI was the most employed frailty measurement tool (7 studies). The PSTFI and JHACG were each used in a single study, whereas the mFI-11, mFI-5, and HFRS were used in 5, 4, and 2 studies, respectively. Comorbidities was the most common component domain and was included in all tools (Table 3). Other common domains included function, cognition, nutritional status, and laboratory parameters. Frailty was most frequently operationalized according to an accumulation of deficits definition. Only the HFRS defines frailty using a weighted instrument. A single point score or more was categorized as either “pre-frail” or “frail” on the PSTFI, JHACG, and MTSFI. Cut-off values were either not reported or varied across studies examining the mFI-5 and mFI-11. The number of individual items per frailty tools ranged from 5-109. The overall mean prevalence of frailty across cohorts was 69%, and the range varied from 21%-100% (Table 3).
Sarcopenia Measures
Eight (36%) studies used various measures for sarcopenia (Table 4). These included the L4-Cross Sectional Muscle Area (CSMA)/height2, L3-4 Total Psoas Area (TPA), L3-TPA/Vertebral Body Area (L3-TPA/VBA), L3-TPA/height2, L4-Psoas:Lumbar Vertebra Index (L4-PLVI), L3-Spinal Muscle Index (L3-SMI) (L3-CSMA/height2), BCA-based clustering (i.e. composite of L3-SMI, L3-Spinal Muscle Quality (i.e. radiodensity) (SMQ), L3-Visceral Adipose Tissue (L3-VAT), L3-Subcutaneous Adipose Tissue (L3-SAT)), L4-VAT, and L4-SAT. The most common sarcopenia measure used was L3-TPA/VBA (3 studies) followed by both L3-TPA/height2 (2 studies) and L3-SMI (L3-CSMA/height2; 2 studies). All sarcopenia measures were employed in the hospital setting and required special techniques or training for use. The overall prevalence of sarcopenia widely varied among the cohort with a range of 155-67%; while some studies reported overall prevalence, others reported prevalence stratified by quartiles (Table 4).
Clinical Outcomes – Studies Employing Frailty Tools
Table 5 describes clinical outcomes, stratified by frailty tool. Nine studies examined mortality, 8 examined perioperative AEs, and 17 examined postoperative AEs. Furthermore, 14 studies examined hospital LOS, 3 examined readmission rates, 4 examined reoperation rates, and 9 examined NRD. The mFI-5 predicted mortality in a single study, the MSTFI in 1 of 3 studies, and the mFI-11 in 1 of 4 studies. The JHACG did not predict mortality in a single study examining it. The PSTFI and JHACG each predicted postoperative AEs in a single study. The mFI-5 predicted postoperative AEs in 3 of 3 studies, whereas the mFI-11 did not predict frailty in 3 of 3 studies. The MSTFI predicted postoperative AEs in 3 of 6 studies, and the HFRS in 1 of 2 studies. Seventy-nine percent of studies (n = 11 of 14) found increased frailty predicted prolonged hospital LOS, when employing the PSTFI, JHACG, mFI-11, HFRS, MSTFI (3 of 4 studies), and mFI-5 (2 of 4 studies). Eight of 9 studies found frailty predicts increased NRD rates, when employing the mFI-5, HFRS, JHACG, and MSTFI (1 of 2 studies). Only one of two studies employing the mFI-5, found increased frailty predicted an increased reoperation rate. Two of 3 studies found increased frailty predicted increased hospital readmission rates; increased MSTFI scores were associated with increased readmission in a single study, whereas mFI-5 predicted readmission in one of two studies. Only 27% (n = 6) reported the study cohort follow-up rate. No studies examined health-related quality of life, function, or cognition as clinical outcomes.
Clinical Outcomes – Studies Employing Measures of Sarcopenia
Table 6 includes a summary of clinical outcomes stratified by sarcopenia measure. All studies employing sarcopenia measures examined mortality and/or overall survival. Of these studies, a single study examined the L4-PLVI in the context of predicting perioperative AEs. Eight studies examined sarcopenia measures in the context of postoperative AEs, 5 examined hospital LOS, 1 examined readmission rates, and 2 examined reoperation rates. The L4 CSMA/height2, L4-PLVI, L3-SMQ, L3-VAT, L4-VAT, and L4-SAT did not predict mortality (each examined in one study, respectively). The following measures of sarcopenia predicted mortality: L3-4-TPA (1 of 1 studies), L3-TPA/VBA (2 of 3 studies), L3 TPA/height2 (1 of 2 studies), BCA (1 of 1 studies), and L3-SAT (1 of 1 studies). Only the L3 TPA/VBA predicted postoperative AEs in a single study. The L3-4 TPA, L4 PLVI, BCA, L3-SMI, L3-SMQ, L3-VAT, and L3-SAT did not predict postoperative AEs (each in a single study). Of the studies examining hospital LOS, 4 did not find an association between increased sarcopenia and hospital LOS; a single study found low L3-SMQ was associated with increased LOS. Neither study examining reoperation and sarcopenia found a significant association with L3-4 TPA or L4-PLVI. Similarly, a single study examining readmission rates did not find an association with sarcopenia (L3-4 TPA). Only 27% (n = 6) examined the follow-up rate. No studies examined health-related quality of life, NRD, function, or cognition.
Clinimetric Appraisal – Studies Examining Frailty Tools
The clinimetric properties of frailty tools are summarized in Table 7. The PSFTI, JHACG, mFI-11, MSTFI, mFI-5, and HFRS were found to be objective, but the JHACG was not. All tools were considered feasible, except the HFRS. PSTFI, JHACG, MSTFI, and mFI-5 tools lacked content validity, whereas the mFI-11 and HFRS had uncertain content validity. No tool was found to have construct validity. Only the HFRS had predicted validity in all studies employing it (n = 2; 100%). The mFI-5 had positive predictive validity in 2 of 4 studies and the MSTFI in only 2 of 7 studies. The mFI-11, PSTFI, and JHACG had either uncertain or negative predictive validity in all studies examining them, respectively. Neither reliability nor responsiveness were examined in any study. All frailty tools had either floor or ceiling effects. The most sensitive population for application of each respective frailty tool could not be determined given the clinimetric appraisal findings. All tools were studied in the context of risk stratification, and none were examined for their application toward assessing frailty trajectory.
Clinimetric Appraisal – Studies Examining Sarcopenia Measures
Clinimetric properties of sarcopenia measures employed in spinal oncology populations are described in Table 8. No tools had construct validity, although the L3-TPA/VBA, L3-TPA/height2, L3-SMQ, L3-VAT, and L3-SAT had uncertain construct validity. Predictive validity was positive for the L3-TPA/VBA in only 1 of 3 studies. All other measures lacked predictive validity. The L3-TPA/VBA and L3-TPA/height2 were reliable. All other measures had uncertain reliability or were not assessed for this clinimetric property. Most studies did not assess responsiveness, although L3-TPA was found to have uncertain responsiveness. All sarcopenia measures were studied in the context of risk stratification. All measures were studied in the context of risk stratification, and none were examined for their application toward assessing sarcopenia trajectory.
Study Bias Assessment
Study bias assignments are presented in Supplemental Content E. The most common sources of bias were related to inadequate follow-up duration for the outcome of interest to occur, no description of cohort follow-up duration (or rate), and analyses lacking adjustment for numerous pre-operative systemic variables known to influence post-operative clinical outcomes among patients undergoing surgery for spinal oncologic disease.
Study Quality Assessment
All studies were graded either “Level 4” (n = 15; 68%) or “Level 2b” (n = 7; 32%) evidence quality according to the Oxford Levels of Evidence 2 grading scale for prognostic studies. Quality assignments are provided in Supplemental Content F.
Discussion
Summary of Findings
This systematic review summarizes measures of frailty and sarcopenia applied in surgical spine oncology and critically appraises their clinimetric properties using a validated set of qualitative criteria and definitions from the Consensus-based Standards for Health Measurement Instruments (COSMIN). No measure of frailty or sarcopenia had positive ratings for all properties, all lacked or had uncertain content and construct validity. Frailty tools had floor or ceiling effects. The MSTFI was studied most frequently but lacked predictive validity. The HFRS had positive predictive validity but lacked feasibility for use in the clinical setting. Most measures of sarcopenia lack predictive validity, and all required special tools or training for use. None of the frailty tools have been assessed for reliability, responsiveness, or used to track frailty trajectory. Here, we discuss the data and highlight clinimetric limitations of existing measures of frailty and sarcopenia. We also recommend a pragmatic approach to utilizing existing measures of frailty and sarcopenia in spinal oncology until more clinimetrically robust tools are developed.
Towards the Development of a Clinimetrically Robust Measure of Frailty or Sarcopenia in Surgical Spine Oncology
We observed that all previously reported tools for assessing frailty were lacking or had uncertain content validity. This property may be optimized by including items that are comprehensible, relevant, and represent a comprehensive sample with respect to frailty in the setting of spine oncology. 28 The AO Spine Knowledge Forum Tumor (AOSKFT) recently conducted a systematic review to identify which systemic variables influence post-operative outcomes in the context of spinal metastatic disease. 2 Older age, low body mass index, comorbidity burden, non-ambulatory status, biochemical abnormalities, malnutrition, and systemic disease burden have been found to predict worse survival, increase adverse events, and worse health-related quality of life. 2 None of the current frailty measurement include a comprehensive sample of these variables. Tools such as the mFI-5, mFI-11, MSTFI, and PSTFI predominantly assess comorbidity status but do not account for the physiological effects of systemic disease, tumor burden, or use of adjuvant therapies, which influence physiological reserve and the ability to tolerate surgery.2,10,12,41 The mFI-11 and mFI-5 include only a limited number of deficits, rendering them susceptible to instability and imprecise index estimates. 6 The MSTFI includes case urgency and surgical approach, neither of which are factors related to patient frailty or would typically be included in a frailty index.5,6 The lack of predictive validity of the PSTFI was not surprising, given it was derived from the MSTFI, the values used to stratify frailty were arbitrarily chosen, and it predominantly assesses comorbidity status among patients that are typically younger, healthier, and have fewer comorbidities than those with spinal metastatic disease.
To better define the multidimensional nature of frailty in the surgical spine oncology context, the AOSKFT utilized a modified Delphi approach in combination with an international cross-sectional survey of the AO Spine community to assess perceptions of frailty in the context of spinal metastatic disease. 13 Pre-operative systemic variables were ranked for their association with frailty using weighted analysis. Consensus (defined as >70% agreement among respondents) was attained among respondents regarding the association between 14 pre-operative clinical variables and frailty. Examples included high risk cardiopulmonary disease, renal failure, liver failure, malnutrition, systemic disease burden, poor performance status, recent unplanned hospitalization secondary to complications of the underlying cancer, and ongoing cytotoxic chemotherapy and/or targeted therapy. Most respondents indicated they evaluate frailty based on general clinical impression, highlighting the outstanding need for a practical and objective frailty assessment tool. Their findings could be used to improve content validity of a future tool for measuring frailty in this patient population.
We observed that most measures of frailty lacked predictive validity. Similar findings were reported by Moskven et al 12 in their clinimetric appraisal of 14 frailty tools used in the setting of spinal surgery for degenerative or deformity-related conditions. Most tools had poor predictive validity, and no frailty tool received positive ratings for all clinimetric properties. The HFRS was found to be a validated risk stratification tool for predicting postoperative AEs following spine surgery for degenerative conditions. The HFRS has been used to predict AEs among patients with primary 47 and metastatic spinal tumors. 45 Although the HFRS has positive predictive validity, the 109-item tool lacks feasibility and could not be reasonably completed in clinical practice. Surgical decision-making particularly in the context of spinal metastatic disease often occurs after hours and on an urgent basis; a tool that can be quickly and easily applied is desirable. No study reviewed described the average time required to complete the frailty assessment. The MSTFI is the most frequently studied frailty tool in surgical spine oncology and is both feasible and objective; however, the predictive validity was positive in only 2 of 7 studies. Massaad et al 9 found the MSTFI overestimated the risk of postoperative AEs among severely frail patients and underestimated risk among mildly frail patients. Bourassa-Moreau et al 10 did not find the MSTFI predicts postoperative AEs. The construct validity, reliability, and responsiveness of both the HFRS and MSTFI remain unclear. Predictive validity was positive for the mFI-5 in 2 of 4 studies. All other frailty and sarcopenia tools had either uncertain or negative predictive validity.
Floor and ceiling effects were observed for all frailty tools. These effects limit the ability to differentiate patients with the lowest and highest scores and may partly explain poor predictive validity. Many existing frailty tools require only a single point to consider patients “pre-frail” or “frail”, resulting in a high cohort prevalence of frailty. Floor and ceiling effect can limit responsiveness (i.e. the ability of a tool to detect clinically important changes over time) and reliability (often measured with intraclass coefficient or weighted Cohen’s Kappa Coefficient analyses). Neither of these properties was directly examined for any frailty tool appraised. Moskven et al 12 reported similarly that reliability and responsiveness were not examined for most frailty tools included in their review. Responsiveness may also be reduced by the modifiability of items included in each tool. Except for the HFRS, most frailty tools contained non-modifiable constructs. None of the frailty tools included in this review were used to track the trajectory of frailty or sarcopenia, rather they were retrospectively applied for the purpose of risk stratification and outcome prognostication.
Sarcopenia has been associated with unfavourable clinical outcomes in patients with cancer.54,55 For example, the L3-TPA/VBA and L3-TPA/Height2 have been utilized in oncologic research and their reliability has been demonstrated.48,56,57 However, the construct and predictive validity of these tools remains unclear in the context of surgical spine oncology. Bourassa-Moreau reported a positive association between L3-TPA/VBA and postoperative AEs among patients with spinal metastatic disease, 10 however, predictive validity was unclear among all other studies reviewed. A potential explanation for these findings relates to traditional sarcopenia measurement techniques assessing psoas area, which may not represent total body sarcopenia. 19 BCA techniques may offer better capture total body sarcopenia and elucidate the relationship between body habitus and clinical outcomes in the surgical spine oncology. First, they often include more robust measures of cross-sectional muscle, such as the skeletal muscle index (i.e. the collective psoas, paraspinal and abdominal muscle mass at a spinal level divided by height (cm2/m2)). 22 Second, they may distinguish between distribution of muscle and adipose tissue, which may be differentially affected by adjuvant therapies.20,21 While muscle quantity has been associated with survival in cancer patients, muscle quality (e.g. radiodensity) has also been associated with survival, patients’ symptoms, and health care use. 58 BCA has been applied in the surgical spine oncology setting. Massaad utilized an automated segmentation technique using validated machine-learning based pipelines based on convolutional neural networks, trained on abdominal CT data from large oncologic cohorts. 22 In their retrospective analysis of institutional data, stratifying the cohort based on BCA phenotypes, the “high-risk” cluster had lower BMI, L3-SMI, SAT, VAT, and higher SMQ. In the analysis adjusted for age, sex, and frailty, there was an increased risk of death in the high-risk cluster (HR 1.45, 95% CI 1.05-2.01, P < .02). However, there was no difference in major AEs, LOS <7 days, or inpatient mortality between “low risk” and “high risk” clusters. Bongers et al 18 separately reported a retrospective review of 196 patients undergoing surgery for spinal metastatic disease and found the L4 CSMA/Height2 was associated with 1 year, but not 90-day mortality. Abdominal adipose and muscle attenuation were not independently associated with mortality. Studies utilizing BCA remain sparse. Whether BCA offers an advantage for risk stratification and outcome prognostication compared to traditional psoas measurement techniques remains to be determined.
Limitations
We recognize there are two study limitations that were introduced by our search strategy, including: (1) the use of an English language filter, which may have limited the inclusion of relevant evidence in other languages; and (2) our use of a date filter (2000-date of search), which was informed by our knowledge of frailty and sarcopenia tools in spinal oncology but may have omitted relevant evidence published prior to 2000.
A Pragmatic Approach to Utilizing Existing Frailty and Sarcopenia Measures in Spinal Oncology: AO Spine Knowledge Forum Tumor Working Group Recommendations
1. Clinical decision making regarding the ability to tolerate palliative surgery should account for the following variables: (a) Systemic disease burden (b) Severe comorbidities (high risk cardiopulmonary, renal failure, liver failure, malnutrition) (c) Reduced performance status (KPS < 70) 2. Until a clinimetrically robust, disease-specific frailty assessment tool is developed, the mFI-5 may be considered as a decision-making adjunct, supplementing factors described in recommendation 1:
Rationale: The mFI-5 is objective, feasible, and predicts non routine discharge (4 of 4 studies), any AEs (3 of 3), increased hospital LOS (2 of 4), and mortality (1 of 1). However, this tool has questionable content validity (includes 4 comorbidities and dependent functional status), a floor effect, and responsiveness is unknown. 3. The L3-TPA/VBA has the most positive clinimetric properties among sarcopenia measures applied in spinal oncology to-date, but requires further study before it can be recommended for use toward risk stratification given the paucity of studies evaluating its contextual utility:
Rationale: The L3-TPA/VBA is objective, feasible, has established reliability in surgical oncology, and predicts mortality (2 of 3 studies) and AEs (1 of 1 studies) in surgical spine oncology. However, this measure has been examined in few studies and responsiveness has not been determined. 4. There is insufficient data to support a recommendation regarding: (a) use of frailty tools or sarcopenia measures for assessing HRQoL, cognition, or trajectory of frailty and sarcopenia following surgery (b) use of body-composition analysis techniques for risk stratification in spinal oncology
Future Work
Future studies should conduct prospective external validation of frailty tools employed for the purpose of risk stratification and outcome prognostication in the surgical spine oncology setting. Several previously reported tools are composed of arbitrarily chosen items. While the MSTFI and PSTFI were developed as disease-specific frailty assessment tools, they lack key disease-specific variables. For example, the MSTFI does not capture systemic disease burden. Given the paucity of disease-specific frailty measures for use in spinal oncology, it is too early to determine whether disease-specific instruments may outperform general measures of frailty for risk stratification and outcome prognostication. Items included in future frailty tools may be drawn from those recently reviewed by the AOSKFT, 2 and ranked via weighted analysis, 13 to improve construct, content, and predictive validity. 6 Clinically relevant outcomes for future study in the context of frailty and surgical spine oncology may include neurological recovery, change in performance status, time-to-event regarding mortality, health-related quality of life, function and cognition. 13 Inclusion of modifiable items in frailty tools may allow testing of interventions to improve frailty in the pre-operative setting, or track frailty trajectory following intervention. In addition to the impact of surgery on frailty, the effect of spinal tumors and systemic disease on frailty remains unclear in this context. Standardizing cut-off thresholds for defining frailty in the context of future frailty indices may improve interpretability, consistency, and comparability across patient cohorts. All measures of sarcopenia identified required special measurement techniques or training for use. In most cases, this included volumetric image analysis capable software, PACS imaging systems. Future work should strive to improve the feasibility of sarcopenia measurement and BCA techniques, particularly through automation and use of machine learning or other artificial intelligence modalities. Collaboration with neuroradiologists may facilitate an improved understanding and appreciation for technical considerations pertaining to image acquisition, properties, and analysis.
Conclusion
Existing tools for evaluating frailty and sarcopenia among patients undergoing surgery for spinal tumors have poor clinimetric properties. While acknowledging these limitations, we provide pragmatic recommendations for utilizing existing frailty and sarcopenia measures in spinal oncology populations, until more clinimetrically robust tools are developed.
Supplemental Material
Supplemental Material - “A Critical Appraisal of the Application of Frailty and Sarcopenia in the Spinal Oncology Population”
Supplemental Material for “A Critical Appraisal of the Application of Frailty and Sarcopenia in the Spinal Oncology Population” by Mark A. MacLean, Antoinette J. Charles, Miltiadis Georgiopoulos, Jackie Phinney, Raphaële Charest-Morin, Rory Goodwin, Ilya Laufer, Michael G. Fehlings, John Shin, Nicholas Dea, Laurence D. Rhines, Arjun Sahgal, Ziya Gokaslan, Byron Stephens, Alexander C. Disch, Naresh Kumar, John O’Toole, Daniel M. Sciubba, Cordula Netzer, Tony Goldschlager, Wende Gibbs and Michael H. Weber, on behalf of the AO Spine Knowledge Forum Tumor in Global Spine Journal.
Footnotes
Author Contributions
All authors contributed to the study conception and design. Material preparation, data collection, data analysis, and data verification were performed by MAM, AC, MG, RCM, CRG, JP, and MHW. The first draft of the manuscript was written by MAM and AC. All authors edited the manuscript. All authors read and approved the final manuscript. MHW had full access to all data in the study. All authors had responsibility for the decision to submit for publication.
Declaration of Conflicting Interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was organized and funded by AO Spine through the AO Spine Knowledge Forum Tumor, a focused group of international Tumor experts. AO Spine is a clinical division of the AO Foundation, which is an independent medically-guided not-for-profit organization.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
Appendix
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
