Abstract
Study Design:
Systematic review.
Objective:
Surgical decompression and reconstruction of symptomatic spinal metastases has improved the quality of life in cancer patients. However, most data has been collected on cohorts of patients with mixed tumor histopathology. We systematically reviewed the literature for prognostic factors specific to the surgical treatment of prostate metastases to the spine.
Methods:
A systemic review of the literature was conducted to answer the following questions: Question 1. Describe the survival and functional outcomes of surgery or vertebral augmentation for prostate metastases to the spine. Question 2. Determine whether overall tumor burden, Gleason score, preoperative functional markers, and hormonal naivety favor operative intervention. Question 3. Establish whether clinical outcomes vary with the evolution of operative techniques.
Results:
A total of 16 studies met the preset inclusion criteria. All included studies were retrospective series with a level of evidence of IV. Included studies consistently showed a large effect of hormone-naivety on overall survival. Additionally, studies consistently demonstrated an improvement in motor function and the ability to maintain/regain ambulation following surgery resulting in moderate strength of recommendation. All other parameters were of insufficient or low strength.
Conclusions:
There is a dearth of literature regarding the surgical treatment of prostate metastases to the spine, which represents an opportunity for future research. Based on existing evidence, it appears that the surgical treatment of prostate metastases to the spine has consistently favorable results. While no consistent preoperative indicators favor nonoperative treatment, hormone-naivety and high Karnofsky performance scores have positive effects on survival and clinical outcomes.
Introduction
Prostate cancer is the most commonly diagnosed cancer and the second leading cause of cancer-related death in men in the United States. 1 Almost 35% of prostate cancer patients have metastatic disease at time of death. 2 Osseous metastases are the most common, 3,4 occurring in more than 80% of patients with hormone-refractory prostate cancer, 5 with the spine being the most common site. 6 –8 Roughly one third of prostate metastases to the spine become symptomatic 4 from spinal cord compression or mechanical instability. 9
The goal of treatment is to improve quality of life through symptomatic relief. 10 –12 Since the study by Patchell et al 12 was published, the surgical treatment of spinal metastases has been guided by the superiority of direct surgical decompression and radiation to radiation alone in the treatment of symptomatic spinal cord compression. This solitary Level 1 study and 3 Class II studies demonstrated increased survival and ambulatory function in mixed-histology series 11,13,14 and support the surgical intervention in a broad range of spinal metastases. 12,15 –18 However, studies focusing on prostate-specific data point to better decisions regarding interventions.
Conforming to PRISMA
19
guidelines, this article systematically reviews the literature to determine the indications and outcomes of the surgical treatment of metastatic prostate cancer of the spine (MPCS) to address key clinical questions:
Methods
Electronic Literature Search
A comprehensive search of major databases (Ovid MEDLINE In-Process & Other Non-Indexed Citations, Ovid MEDLINE, Ovid EMBASE, Ovid Cochrane Database of Systematic Reviews, Scopus) from inception to October 2013 was conducted by a librarian and the study’s principal investigator using controlled vocabulary and keywords to search for studies explicitly designed to evaluate patient-specific outcomes of surgery or vertebral augmentation for MPCS. The actual strategy is available from the corresponding author on request. Nonhuman studies, case reports, meeting abstracts, review articles, and editorials were excluded. Mixed tumor series were reviewed to extract disease-specific information. The patient-intervention-control-outcome (PICO) summary of inclusion and exclusion is described in Table 1.
Selection Criteria.
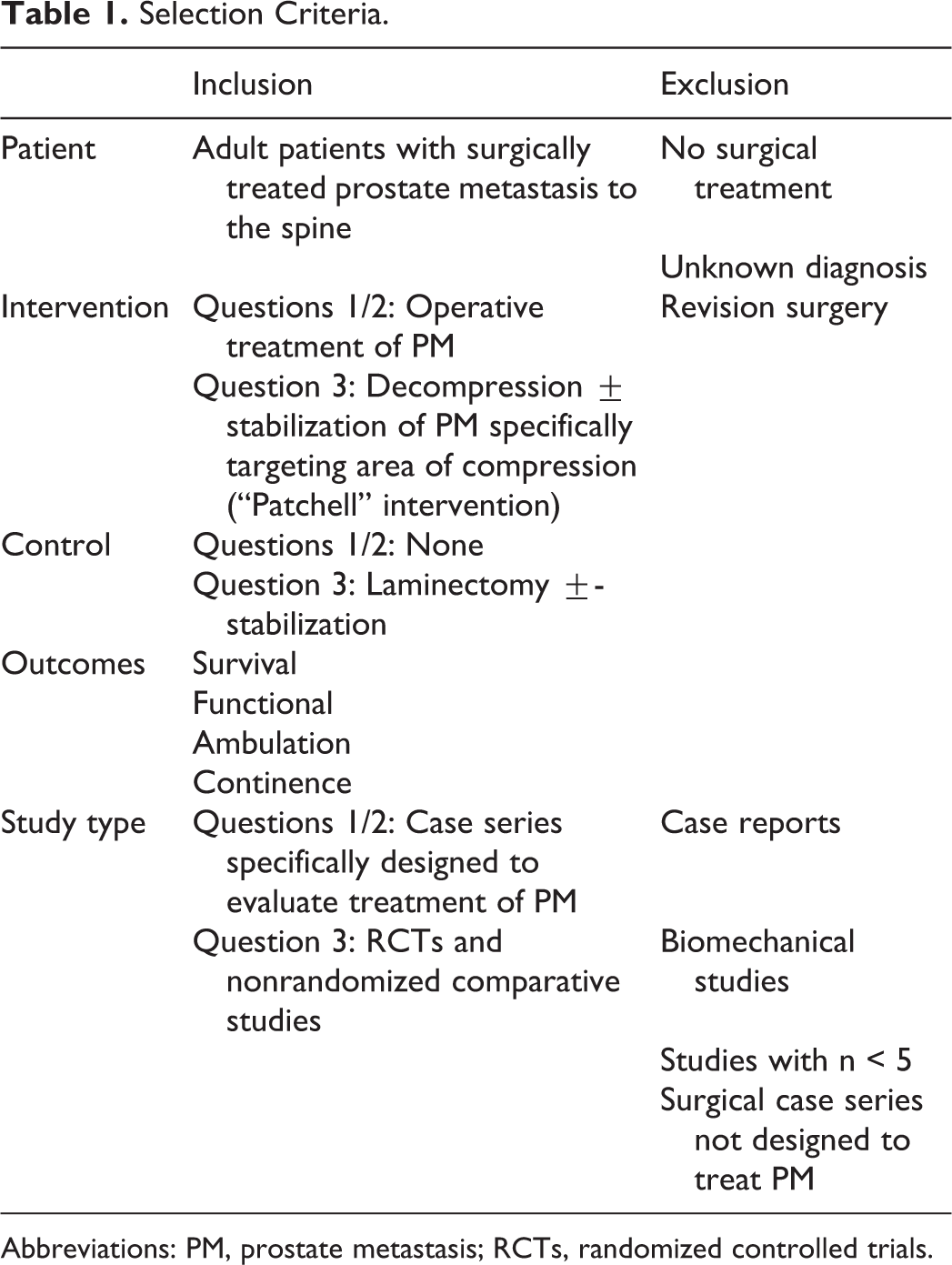
Abbreviations: PM, prostate metastasis; RCTs, randomized controlled trials.
Data Extraction
Full versions of the articles meeting the inclusion criteria were reviewed. Data extracted included study design, demographics, presentation, treatment, and outcomes, including postoperative survival and neurologic parameters (motor weakness, sensory dysfunction, ambulatory status, and bladder continence).
Study Quality and Overall Strength of Evidence
Class-of-evidence (CoE) ratings were assigned to each article by 2 reviewers (MJC, DMS) accounting for methodological quality and sources of bias based on recommendations of the Agency of Healthcare Research and Quality (AHRQ). 20 –23
The overall strength of the body of evidence was based on the Grading of Recommendations Assessment, Development and Evaluation (GRADE) Working Group 24 –26 and AHRQ 20,22 and is a measure of our confidence that the effect size closely matches the true effect and is stable. 27 The overall strength of evidence is considered “Strong” if the majority of studies were Class I or II and indicates that the true effect lies close to the estimated effect. It is considered “Weak” if the majority of studies were Class III or IV and indicates that the true effect may be very different from the estimated effect. A rating of “Insufficient” indicates there was too little evidence to estimate an effect. Overall strength was downgraded (inconsistent results, indirect evidence, imprecise estimates) or upgraded (large magnitude of effect, dose-response gradient) 27 as warranted.
Clinical Recommendations and Consensus Statements
Expert consensus opinion was obtained using a modified Delphi process involving subspecialty spine tumor surgeons at the AO Knowledge Forum Tumor section. Recommendations were made using the GRADE/AHRQ criteria that separates the strength of evidence from the strength of recommendations as described elsewhere. 28
Results
Study Selection
The search yielded 360 relevant citations: 323 were excluded based on title/abstract, and 37 articles were reviewed. Fourteen were rejected because they had no outcomes of interest (9), did not involve postprocedural outcomes (2), or had <5 cases (3). All 5 mixed-histology articles on vertebral augmentation were rejected as they had <5 patients. Data sets overlapped in articles by Chaichana, 29 Ju, 3 and Crnalic 5,30 ; thus, the less comprehensive article was excluded. Of the remaining 16 studies, 10 studies 31 –40 contained only disease-specific data on overall survival. Thus, 16 articles 3 –5,31 –43 were studied for overall survival (Figure 1A) and 6 studies 3 –5,41 –43 for all nonsurvival questions (Figure 1B). These are summarized in Table 2.
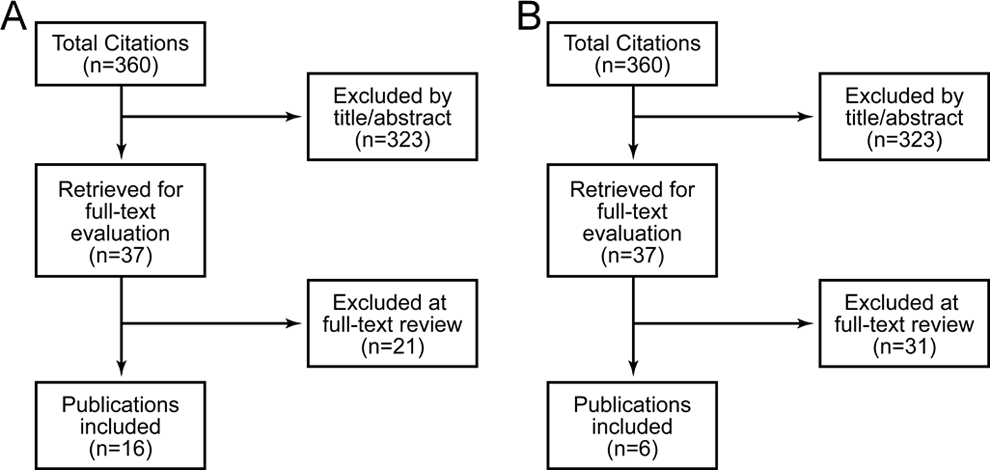
Study selection for (A) survival and (B) remaining questions.
Included Studies.
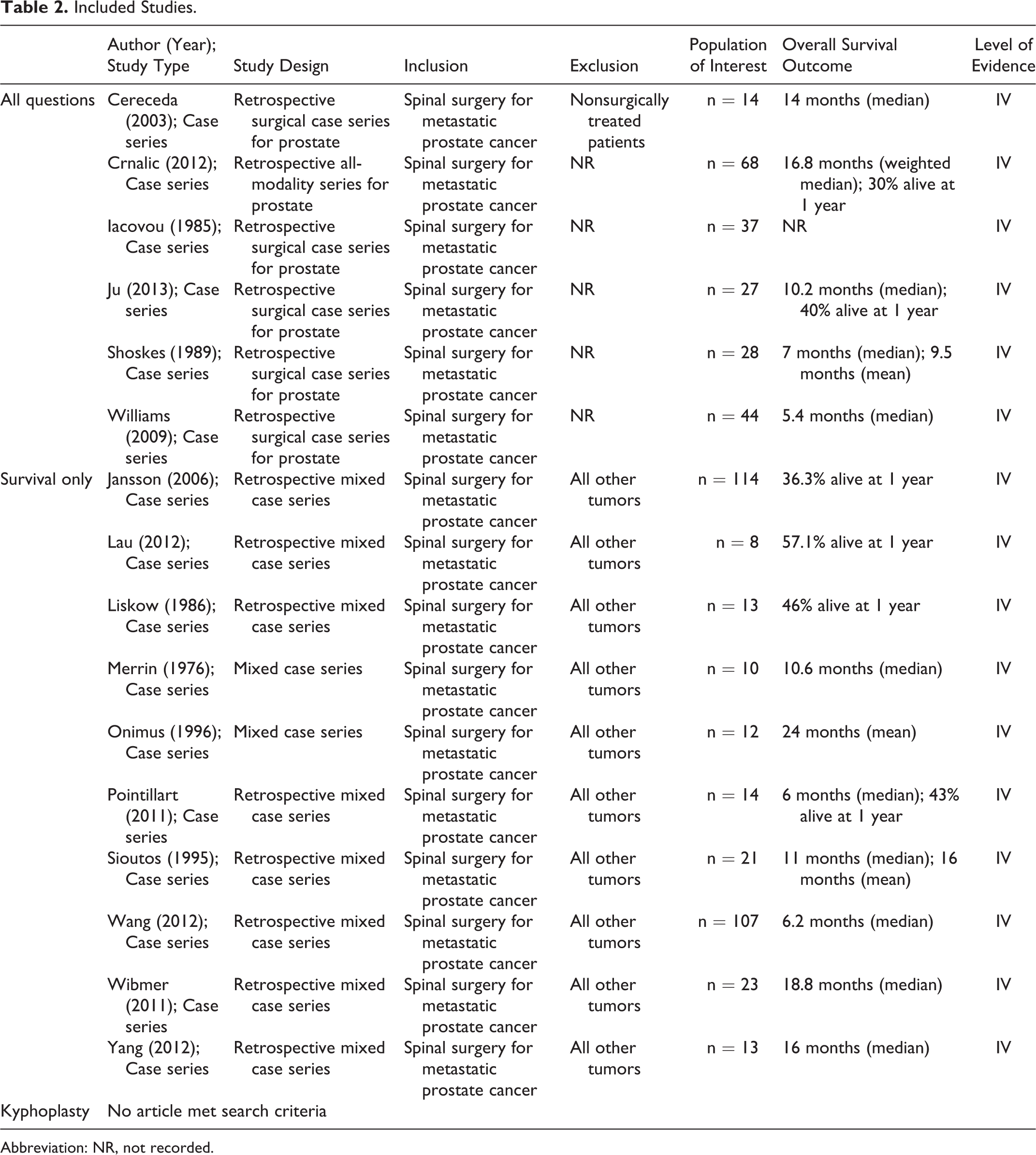
Abbreviation: NR, not recorded.
Survival
Survival rates following intervention were recorded in 16 studies using various methods, often mixing hormone-naive and hormone-refractory patients (Table 2). Four studies consistently demonstrate longer survival in hormone-naive patients (Table 3).
Hormone Naivety.
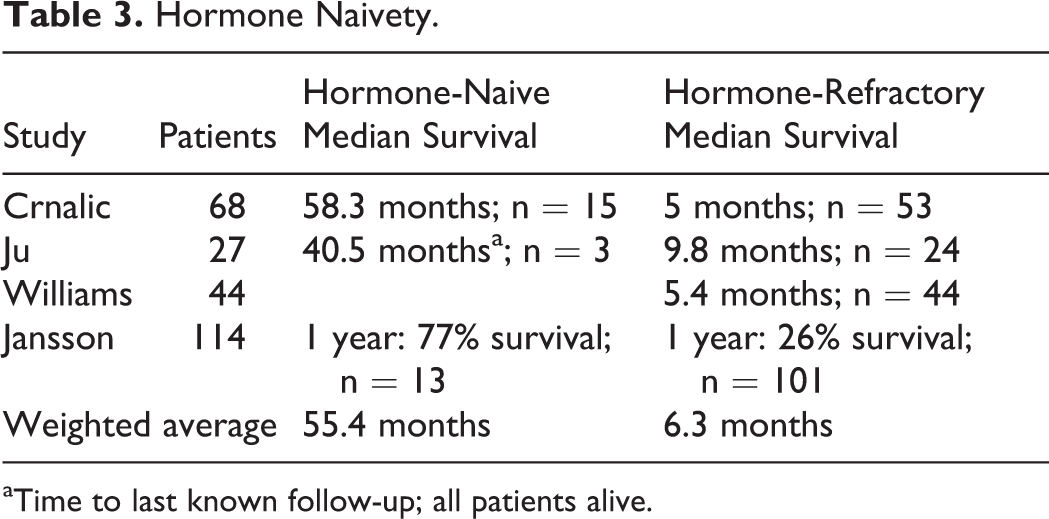
aTime to last known follow-up; all patients alive.
Other prostate-specific variables were reviewed for prognostic value. Elevated prostate-specific antigen (PSA) had a significant negative impact on survival in 2 studies. 3,5 Gleason score of ≥8 had a statistically significant difference in survival in one study 4 and not in another. 3 Overall tumor burden was reviewed. There was a statistically negative impact with visceral metastases 4,5 and lymph node involvement. 4 Ju noted no significant difference in complications for surgically treated patients with either visceral metastases or lymph node involvement. 3
Overall survival was affected by preoperative Karnofsky Performance Status (KPS) score. In the largest series, Crnalic noted a median survival of 22 months (n = 9) in patients with KPS ≥80, and 4.8 months (n = 44) in patients with KPS <80. 5 Ju also found a statistically significant difference in survival. 3 Although a similar trend was seen in the Cereceda study (11.5 vs 8 months for KPS ≥ 80), this study was hampered by small numbers. 41
Pain and Functional Outcome
The goals of palliative surgery are the improvement or preservation of ambulation, bowel/bladder continence, and pain relief (Table 4). Across 4 studies, 94% of patients maintained or improved strength at 1 month and 71% of patients were ambulatory. Of patients who were nonambulatory preoperatively, 55% regained ambulation. Spinal cord function outcomes were outlined in 3 studies by pre- and postoperative Frankel scores 3 –5 (Figure 2). Forty-three percent (52/122) had useful motor function (Grade D or E) preoperatively, increasing to 75% (92/122) postoperatively; 57% (40/70) without useful function regained it postoperatively. The number of patients improving 1 or more grades was 48% (59/122) while only 3% (4/122) worsened.
Other Factors With Statistically Significant Negative Effect on Survival.
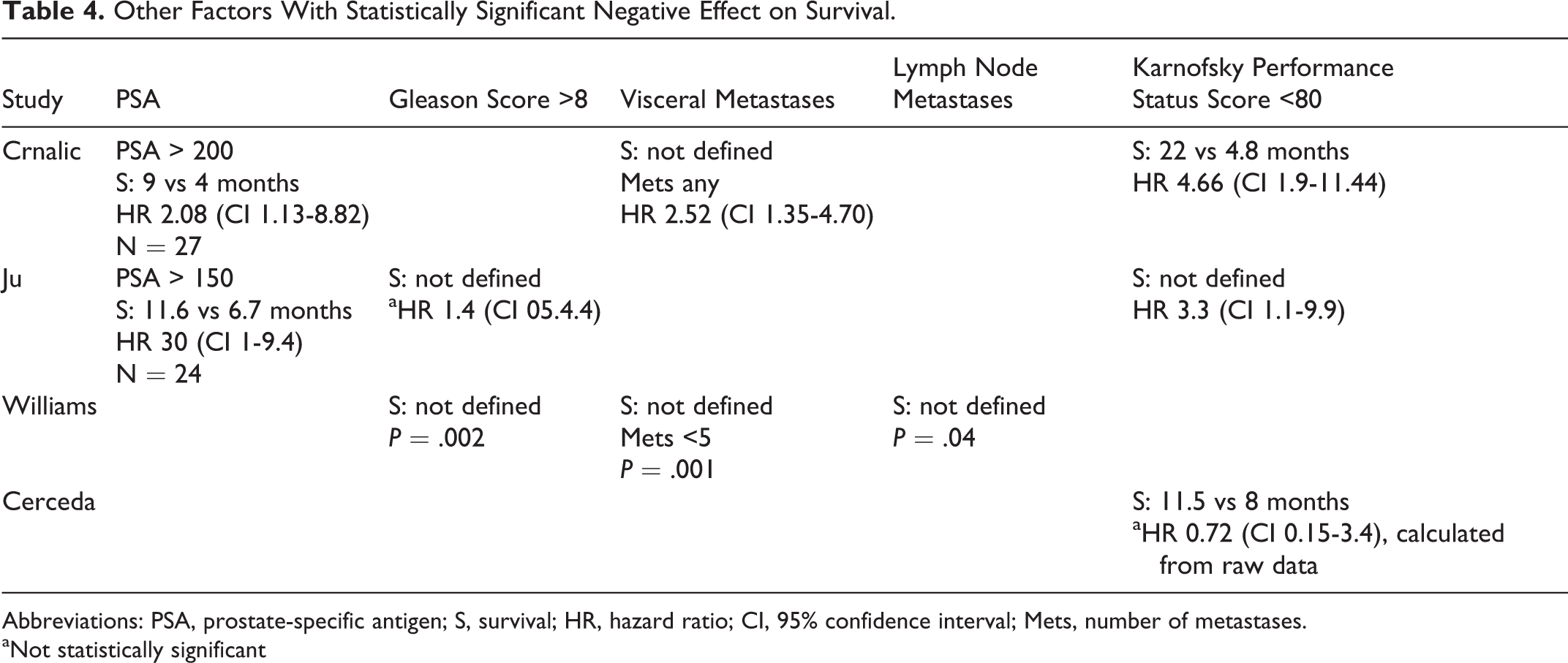
Abbreviations: PSA, prostate-specific antigen; S, survival; HR, hazard ratio; CI, 95% confidence interval; Mets, number of metastases.
aNot statistically significant
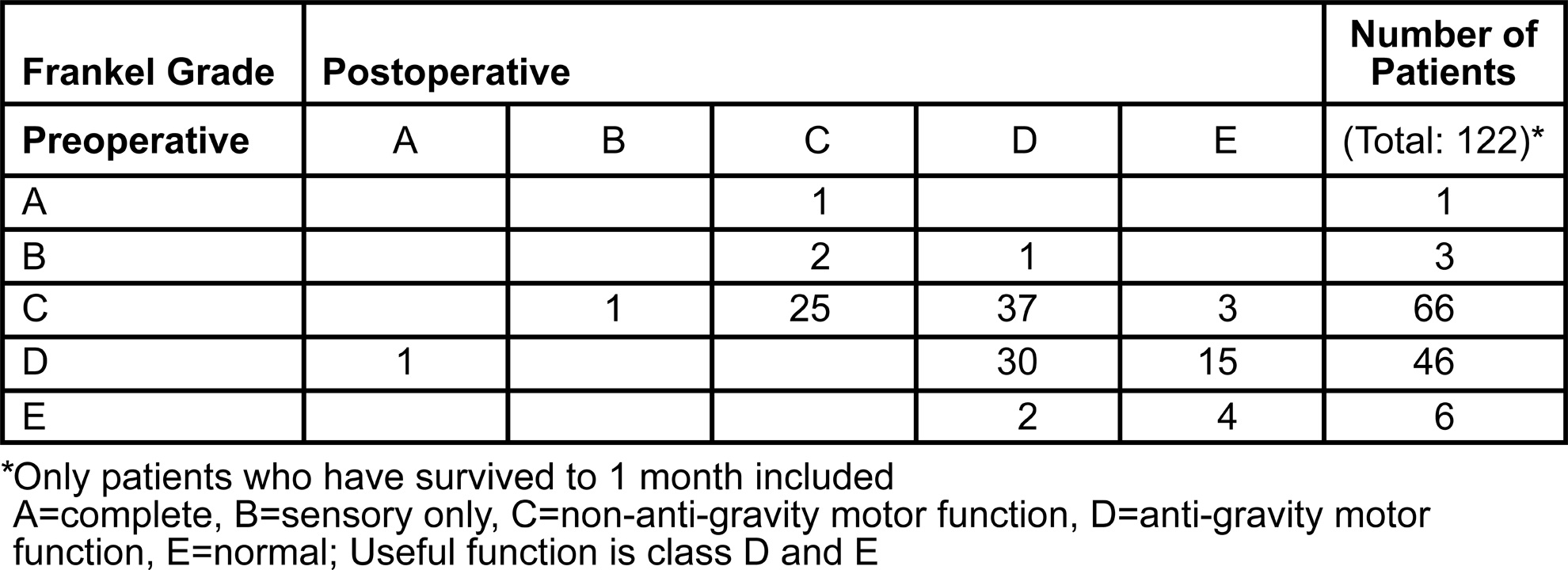
Frankel classification improvement at 1 month.
Forty-nine percent of patients with bladder dysfunction related to spinal cord compression regained continence. Seventy-four percent of patients with preoperative pain noted improvement 1 month postoperatively based on studies by Cereceda et al 41 and Iacovou et al. 42 Both Ju and Williams noted a decrease in median narcotic use (40 mg/day to 10.7 mg/day morphine equivalents, and 10 mg/day to 4 mg/day) 3,4 and a decrease in steroid use (24 mg/day to 0 mg/day dexamethasone equivalents). 3
Surgical Outcome
Overall, 204 patients underwent a surgical procedure for prostate metastases to the spine in 5 studies. 5,25,36,42,44 Fifteen died in the perioperative period for an overall mortality of 7% (15/204). Three studies noted an overall complication rate of 32% (34/106) 25,36,44 and a major complication rate of 18% (19/106). 25,36,44
No study delineated surgical approach based on compressive pathology as advocated by Patchell et al. 12 It is possible that many patients underwent laminectomy in the setting of anterior compressive pathology. Anterior and combined anterior-posterior approaches were only noted in studies by Williams (55%) 4 and Ju (61%). 3 Patient numbers were low, and survival and functional outcomes based on surgical approach were not calculated; however, Ju noted no statistical difference in surgical complications. 3
Study Quality and Overall Strength of Literature
All 12 articles were retrospective case series without control groups; thus, all had a baseline CoE of Level IV. Due to large, consistent effects, hormone-naivety and pain/functional outcome data were upgraded. Based on limited information and data, PSA, Gleason score, and metastatic disease data were downgraded. Based on the CoE and the quality of data, the overall strength of evidence is “Low” to “Insufficient” as outlined with each summary (Table 5).
Functional Outcome.
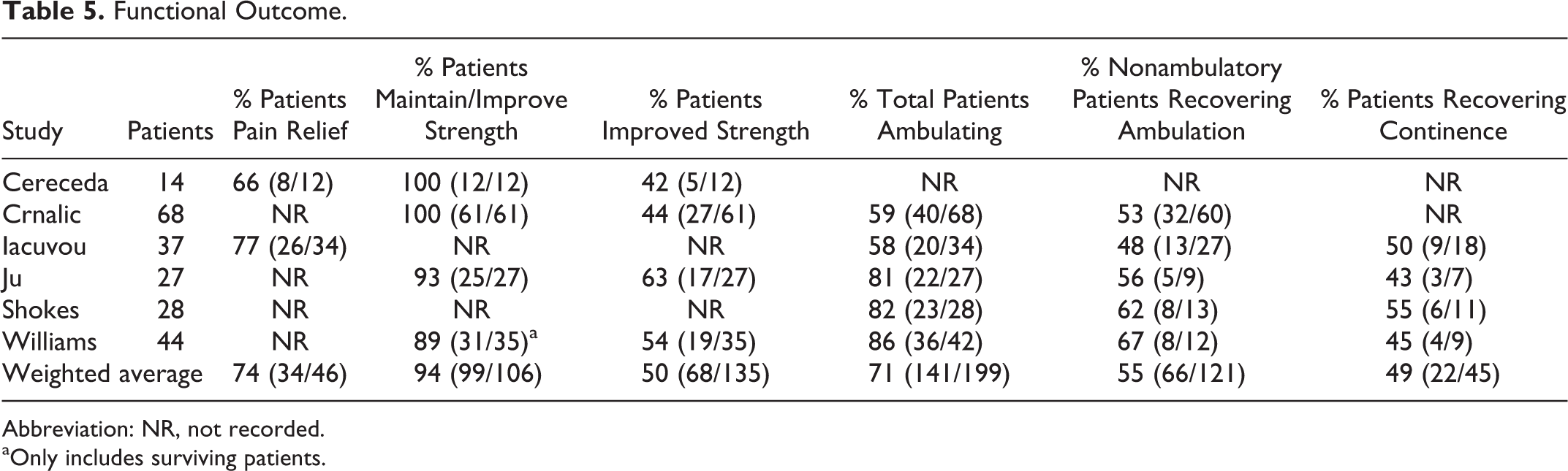
Abbreviation: NR, not recorded.
aOnly includes surviving patients.
Clinical Recommendations and Consensus Statements
Although there is scant prostate-specific literature, the information gleaned from this systematic review, data from mixed tumor histology studies, and expert opinion were felt sufficient to provide clinical recommendations and consensus statements: Experts agreed that decompressive/reconstructive spinal surgery is beneficial in appropriately selected patients with MPCS. Although low in strength, evidence suggests that surgery improves quality of life, improves/preserves motor strength, ambulation, and bowel/bladder function with relatively low morbidity and mortality. (Level of recommendation: “Weak”) No markers definitively precluded patients from surgical intervention. However: There was moderate evidence that hormone resistance has a large negative impact on overall postoperative survival. These patients had an average survival of over 6 months. In patients with MPCS, the extent of systemic metastases, medical comorbidities, and functional status must be considered when determining surgical candidacy. The evidence supporting the negative impact of hormone-refractory tumor status and low KPS on survival should be considered. These patients have a higher risk of limited postoperative survival; however, they may still be appropriate surgical candidates as each represents a unique combination of systemic considerations. (Level of recommendation: “Weak”) In patients requiring spinal cord decompression, surgical options including instrumented spinal stabilization can be considered. Circumferential decompression may be indicated. Experts felt the blastic nature of most MPCS may lessen the need for corpectomy and stabilization compared with lytic histologies. In scenarios where laminectomy alone is indicated (direct or indirect decompression, stability is not in question, adjunctive treatments are available), a non-destabilizing laminectomy is performed. (Level of recommendation: “Weak”) Based on expert opinion gleaned from the Delphi process, in patients with mechanical instability without neural compression, cement augmentation, percutaneous or open instrumented spinal stabilization represent potential treatment options. (Level of recommendation: “Weak”)
Discussion
Prostate cancer is a variable disease ranging from indolent to aggressive. 45 While localized disease may be cured with surgery and radiation, in 20% to 30% of patients the disease recurs. 46,47 Since androgens promote prostate cancer survival and growth, 44,48 androgen synthesis pathway ablation is the primary treatment for advanced disease. 49 Unfortunately, many patients develop androgen deprivation therapy insensitivity, also known as castration-resistant prostate cancer or hormone-refractory disease 50,51 with survival ranging from 1 to 2 years. 47 Up to 80% of patients with hormone-refractory prostate cancer develop spine metastases, 5 and up to one third of these will become symptomatic requiring intervention. 4
The Patchell study outlines the basic surgical selection criteria applied to most metastatic spine tumor patients: patients must be healthy enough to tolerate the intervention, they should have an expected survival over 3 to 6 months, and they must have some expectation of neurologic recovery. Refining the Patchell selection criteria by incorporating pathology-specific prognostic markers has the potential to improve surgical decision making.
A major consideration for surgical selection is anticipated survival. It is thought that patients should have an expected survival of 3 to 6 months to reap the benefits of the intervention. As survival in MPCS is dependent on tumor responsivity to treatment, patients with hormone-naive tumors have a significant survival advantage over patients with hormone-refractory tumors. Of importance, both hormone-naive and hormone-refractory patients have an expected survival of over 6 months. Similarly, although PSA, Gleason score, and KPS scores demonstrate a statistically and temporally significant difference in survival, no marker definitively demonstrates a survival below the expected criteria of 3 to 6 months. This must be interpreted with caution, as expected survival is borderline, the strength of studies is insufficient, and none discuss prognostic markers in combination. It is possible that multiple negative prognostic indicators may predictably shorten survival to a degree that decompressive surgery will not offer a satisfactory result. However, we found no prognostic marker that definitively rules out MPCS patients from having surgery.
Additionally, the tenet that patients should have a 3- to 6-month anticipated survival may be flawed. Less invasive options than the 360-degree decompression Patchell advocated, including “separation surgery” 52 and minimally invasive surgical techniques, 53,54 may produce similar results with less morbidity and convalescence, potentially expanding surgical indications to patients with shorter anticipated survival. Also, Patchell focused on neurologic deficit, yet local and mechanical pain can limit mobility and quality of life. Less invasive options such as percutaneous fixation and vertebral augmentation may be useful palliative options in patients with limited life expectancy.
Evidence supports the use of vertebral augmentation in the treatment of metastatic disease and is agreed upon by expert consensus. Although excluded from this study, 4 mixed-histology Level 4 retrospective cohorts reported sustained analgesia in over 80% of patients with minimal complications. 55 –58 In 2 studies, functional status improved due to increased mobility from pain relief. 57,58 Prostate cancer usually causes sclerotic lesions until late in the disease, often once the patient has hormonal-resistant cancer, when osteolytic lesions occur. 59,60 Thus, vertebral augmentation may be most useful in patients with advanced disease.
Predictability of postoperative functional status is outlined by our study. The majority of patients were ambulatory postoperatively, and approximately 50% of nonambulatory patients regain the ability to walk. Operative selection criteria were poorly outlined, however, including the preoperative ambulatory status and temporal course of neurologic decline. In our pooled data, only one Frankel Class B patient had a good outcome (Class D or E), although good outcomes occurred in the majority of patients with preoperative Class C, D, or E function. Selection bias is noted in this combined cohort, however, as most cases were Class C and D. More complicated is data about urinary continence. Although almost 50% of patients regained continence following surgery, this information must be approached with caution as incontinence may be a direct result of the disease process and treatment in prostate cancer and not spinal cord compression. Surgical treatment provided significant pain relief. In 2 studies, 74% of patients noted significant pain relief, 41,42 and 2 separate studies demonstrated a large reduction in narcotic medication requirements. 3,4 Thus, surgical treatment reliably improves/maintains motor function, may allow well-selected patients to regain ambulation and urinary continence, and can reduce the pain of spinal metastases.
Finally, patients must be able to medically tolerate decompressive surgery. The included studies had a 20% perioperative morbidity and 10% mortality. Anecdotally, Iacovou notes that hormone-refractory patients recovered from surgery more slowly; but no further information is available. 42 Ju notes that extent of surgery (number of levels) and age were significant predictors of surgical complications. 3 Although this information is consistent between studies, the surgical techniques are dated. All of the studies include large numbers of laminectomy-only patients, while the more contemporary 360-degree decompressive procedure is underrepresented making it difficult to apply contemporaneously. That said, as a predominantly osteoblastic tumor, laminectomy alone may be viable as the structural integrity of the affected vertebral body may not be compromised. Additionally, prostate cancer has a predilection for the lumbar spine, 61 resulting in cauda equina as opposed to spinal cord compression, making Patchell less relevant. This underscores the importance of histologically specific studies on spine metastases.
Surgical treatment of prostate metastasis to the spine has the potential to reliably decrease pain, maintain or improve ambulation, and maintain urinary continence. Systematic review of the literature is limited and the strength of data relatively poor; however, there is no definitive marker that would preclude consideration for surgery. Despite these limitations, this review does provide prostate cancer–specific data that will enable improved shared decision making for patients presenting with symptomatic spinal metastasis. Further high-quality prospective studies on this important topic are warranted.
Conclusion
There is a dearth of literature on the treatment of prostate metastases to the spine. All studies have a level of evidence of IV, and consistencies across studies correspond to the more robust mixed-histology literature. The overall results of the original questions are the following:
Footnotes
Acknowledgments
AOSpine funds were received in support of this work. AOSpine is a clinical division of the AO Foundation—an independent, medically guided, nonprofit organization. The AOSpine Knowledge Forums are pathology-focused working groups acting on behalf of AOSpine in their domain of scientific expertise. Each forum consists of a steering committee of up to 10 international spine experts who meet on a regular basis to discuss research, assess the best evidence for current practices, and formulate clinical trials to advance spine care worldwide. Study support is provided directly through AOSpine’s Research Department and AO’s Clinical Investigation and Documentation unit.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Fisher reports personal fees from Medtronic, personal fees from Nuvasive, grants from OREF, outside the submitted work. Dr Gokaslan reports grants from AO Spine North America, personal fees from AO Foundation, grants from AO Spine, outside the submitted work. Dr Rhines reports personal fees from Stryker, personal fees from Globus, outside the submitted work. Dr Laufer reports personal fees from SpineWave, personal fees from Globus, personal fees from DePuy/Synthes, outside the submitted work. Dr Patel reports grants from Eisai, grants from Jannsen Research Development, grants from Morphotek, personal fees from Novartis, personal fees from EMD Serono, personal fees from Johnson and Johnson, personal fees from CytRx, personal fees from Daiichi Sankyo, outside the submitted work. Dr Rampersaud reports other funding from Medtronic, outside the submitted work. Dr Reynolds reports personal fees from Globus, personal fees from DepuySynthes, personal fees from AOSpine, personal fees from Medtronic, outside the submitted work. Dr Chou reports other funding from Globus, Medtronic, and Orthofix, outside the submitted work. Dr Sciubba reports other funding from Medtronic, Depuy-Synthes, and Globus, outside the submitted work. Dr Clarke, Dr Molina, Dr Fourney, Dr Schmidt, Dr Fehlings, Dr Bettegowda, Dr Mendel, and Dr Weber have no conflicts of interest to disclose.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: AOSpine funds were received in support of this work.
