Abstract
Introduction:
This article aims at investigating the 5-year budget impact of rituximab biosimilars in Italy.
Methods:
A budget impact analysis model was developed in accordance with the International Society For Pharmacoeconomics and Outcomes Research recommendations. Drug acquisition and drug administration costs were considered since the risk/benefit profile of biosimilars and the originator was assumed to be overlapping. The perspectives of hospitals and payers were used. Input data were retrieved from the literature and validated/integrated by an expert panel of seven clinicians from various Italian regions. A dynamic incidence-based approach was used.
Results:
From the hospital perspective, adopting a rituximab biosimilar would produce savings of €79.2 and €153.6 million over 3 and 5 years, respectively. The results are very similar if the payer perspective is considered, with a cumulated savings of about €153.4 million in 5 years. Lymphoma and chronic lymphocytic leukaemia would account for the most significant savings.
Discussion:
Despite its limitations, this study provides the first Italian evaluation of the financial impact of rituximab biosimilars and also incorporates the effects of biosimilars on the pricing strategies of the originator (dynamic impact). This dynamic effect is more relevant than the impact of the treatment shift from the originator to biosimilars. Our hope is that these savings will be used to cover new cost-effective drugs and not just for cost-cutting policies.
Introduction
Biosimilars may represent an important opportunity to enhance allocative efficiency in the market for biological products. Savings from price-competition could be invested in new cost-effective drugs and/or to broaden patient access to existing therapies. The availability of biosimilars is expected to be associated with allocative efficiency and decreased spending on medications, being one of the driving forces in budgetary savings. 1
At present, 44 biosimilar products have been approved by the European Medicines Agency (EMA) for 14 molecules, 2 and 16 are under evaluation. 3 In Italy, 32 biosimilars have been approved so far and, in the first 9 months of 2017, they had reached an 18% market share over total volumes (number of counting units) for the relevant markets (molecules with at least one available biosimilar). The biosimilar market share shows huge variations across regions in Italy, ranging from 62.6% in Piedmont to 6.5% in Puglia. 4 Regional variations can be ascribed to divergent procurement policies and actions on prescribing behaviour. 5 However, these differences could diminish in the future. A new regulation (Law 232/2016) only allows prescribing physicians to switch from the originator to the biosimilar (or from one biosimilar to another biosimilar). According to the same law, procurement should rely on a framework agreement if there are more than three available products for an off-patent biological molecule (originator and biosimilars).
Rituximab was the first monoclonal antibody approved for cancer in 1997, and is also approved for rheumatoid arthritis. It can be administered by both the intravenous (IV) and the more recently approved subcutaneous (SC) route as a monotherapy or in combination with chemotherapy regimens.
Rituximab is a chimeric monoclonal antibody targeting the CD20 antigen, which is present on the surface of B-lymphocytes. In 2016, rituximab was the third-largest selling hospital drug in Italy (€156 million), according to the Italian Medicines Agency (Agenzia Italiana del Farmaco – AIFA). 6
Since the patent for rituximab IV expired, EMA has approved two biosimilars so far: Truxima and Rixathon on 17 February 2017 and 23 June 2017, respectively. 2 The rituximab biosimilars were approved for the treatment of all indications approved for the reference biological (non-Hodgkin’s lymphoma, chronic lymphocytic leukaemia, rheumatoid arthritis, granulomatosis with polyangiitis and microscopic polyangiitis). AIFA subsequently also approved the use of biosimilar rituximab for the off-label indications previously identified for the originator (Determina AIFA 2107/2017) and included in the 648 List (i.e. the off-label use list covered by the Italian National Health Service – INHS).
Our study aims at investigating the financial impact of the introduction of rituximab biosimilars in clinical practice in Italy through a Budget Impact Analysis (BIA). In the present analysis, BIA of rituximab biosimilars is evaluated, including both approved and off-label indications, with a split per indication and adopting both the hospital and third-party payer point of view.
To the best of our knowledge, the budget impact of rituximab biosimilars has been investigated by only one study so far. 7 Although this article provided a general overview of the budget impact in European countries from the third-party payer perspective, it lacked information regarding the number of patients per approved indication and did not distinguish between specific off-label uses.
Methods
A BIA model was developed in accordance with the ISPOR Principles of Good Practice for BIA. 8
The model considers for both the rituximab originator and biosimilar as follows:
The approved clinical indications: follicular lymphoma (FL) III–IV stage (induction, associated with chemotherapy scheme CVP – cyclophosphamide, vincristine, prednisolone), recurrent/refractory FL III–IV stage (induction, associated with chemotherapy scheme CHOP – cyclophosphamide, hydroxydaunorubicin, oncovin, prednisolone), untreated or recurrent/refractory FL (maintenance therapy), recurrent/refractory FL III–IV stage (induction, mono-therapy), non-Hodgkin lymphoma (NHL) CD20+ large B cells in combination with chemotherapy CHOP, untreated or recurrent/refractory chronic lymphocytic leukaemia (CLL) in combination with chemotherapy, severe rheumatoid arthritis, granulomatosis with polyangiitis and microscopic polyangiitis.
The off-label use for the following indications: non-Hodgkin lymphoma CD20+ large B cells in combination with polychemotherapy for first-line or salvage treatment, CLL B cells in combination with polychemotherapy for first-line or salvage treatment, post-transplant lymphoproliferative disorder, acute or chronic graft versus host disease (GVHD), FL in patients not eligible for polychemotherapy (mono-chemotherapy), lymphocyte-predominant Hodgkin lymphoma, warm antibody autoimmune haemolytic anaemia, relapsing or refractory thrombotic thrombocytopenic purpura resistant to plasma exchange, immune thrombocytopenic purpura resistant to standard treatments, resistant acquired haemophilia.
The model was developed in MS Excel according to the following process:
Collection and analysis of epidemiological data (prevalence and incidence) relating to patients in Italy for whom treatment with rituximab is indicated (including off-label use);
Definition of the current scenario, that is, the number of prevalent and incident patients treated with the rituximab originator;
Definition of future scenarios (i.e. the penetration rate of the biosimilar) for the different indications;
Cost estimates for current and future scenarios.
Since biosimilars are approved through a comparability exercise, we assumed an overlapping risk/benefit profile between the originator and the biosimilars, the only difference being the actual unit price of the drug and the administration route (IV vs SC).
Two perspectives were used: (1) the hospital perspective, that is, the acquisition cost of the drug and the administration cost and (2) the payer perspective, that is, rituximab is administered either in ‘day hospital’ or in an outpatient setting, and hospitals are paid in principle on a fee-per-episode basis that should cover the full treatment cost but, in reality, hospitals are paid the drug acquisition cost and a ‘discounted’ fee 9 (Regional Laws).
Input data were validated and integrated by an expert panel of seven clinicians. Considering the large variability in biosimilar penetration rates across Italian regions, clinicians from different regions were invited to join the panel. A Delphi-driven approach was applied in the early stage of the data collection, since experts were asked to answer a questionnaire independently. 10 Afterwards, data gathered were discussed with the experts through a group meeting (11 October 2017). Hence, we have adopted a mixed method, integrating a Delphi approach with an experts’ panel approach. The panel was asked to: (1) validate the epidemiological data for each indication and the relevant proportion of patients treated with rituximab, (2) validate the therapeutic schemes per indication, (3) estimate the yearly and 5-year market share for the originator and the biosimilars per indication and (4) estimate time dedicated by healthcare professionals to patient care when the drug is being administered.
Market shares were applied to new, incident cohorts (naïve treatment) for almost all the indications, since the treatment lasts less than 1 year. The prevalent population was used only for maintenance therapy for FL (patients are treated every 2–3 months until progression, for a maximum of 2 years). We assumed that half of the prevalent patients are in the first year of treatment and half are in the second year of treatment. The expert panel reached a consensus of 12 cycles of therapy over 2 years for patients with untreated FL and 8 cycles of therapy distributed over 2 years for patients with relapsed or refractory FL. Survival curves for the different indications were consulted to obtain the percentages of patients still alive and treated at different times. As the focus was on the expected budget at each point in time, the financial streams were presented as undiscounted costs. 8
Clinicians expressed the opinion that biosimilars will be used only for drug-naïve patients (i.e. there will not be patients switching from the originator to the biosimilars) and that biosimilars will mostly substitute the IV rituximab originator. This assumption minimizes the impact of biosimilar market penetration on drug administration costs.
The unit cost of the rituximab originator was calculated as the ex-factory price net of the compulsory 5% + 5% discount and an additional discount, according to the agreement negotiated by AIFA and the company holding the marketing authorization in July 2017. 11 The unit cost of biosimilars was calculated as the ex-factory price of Rixathon net of the 5% + 5% discount. For future scenarios, we have adopted a dynamic approach looking at further discounts for the rituximab originator due to price-competition (5%, 10% and 15% for year 1, year 2 and years 3–5, respectively). Biosimilars are expected to be discounted to 30%, 35% and 45% on average for year 1, year 2 and years 3–5, respectively. These discounts were estimated on the basis of the most recent experience for infliximab (data on file).
As mentioned above, rituximab is administered in day hospital or in an outpatient setting. For both settings fees for drug administration are determined by the regional governments (payers). However, regions usually pay to the hospital the acquisition costs of the drugs and a ‘discounted’ fee. In the hospital perspective case, drug administration costs were estimated based on the consensus reached by the expert panel on time dedicated by nurses and physicians, that is, 50 and 20 minutes for an IV infusion, respectively, and 25 minutes spent by nurses on SC administration. The unit cost per minute for healthcare personnel was estimated using the gross annual salaries 12 of €32,518 for nurse and €73,050 for physician. These values correspond to a cost per hour of €19.64 and €44.11, respectively, considering 46 working weeks per year (36 working hours per week) and translate into €0.33 and €0.74 per minute, for nurse and physician, respectively.
Table 1 summarizes all unit costs used in the model.
BIA model input data.
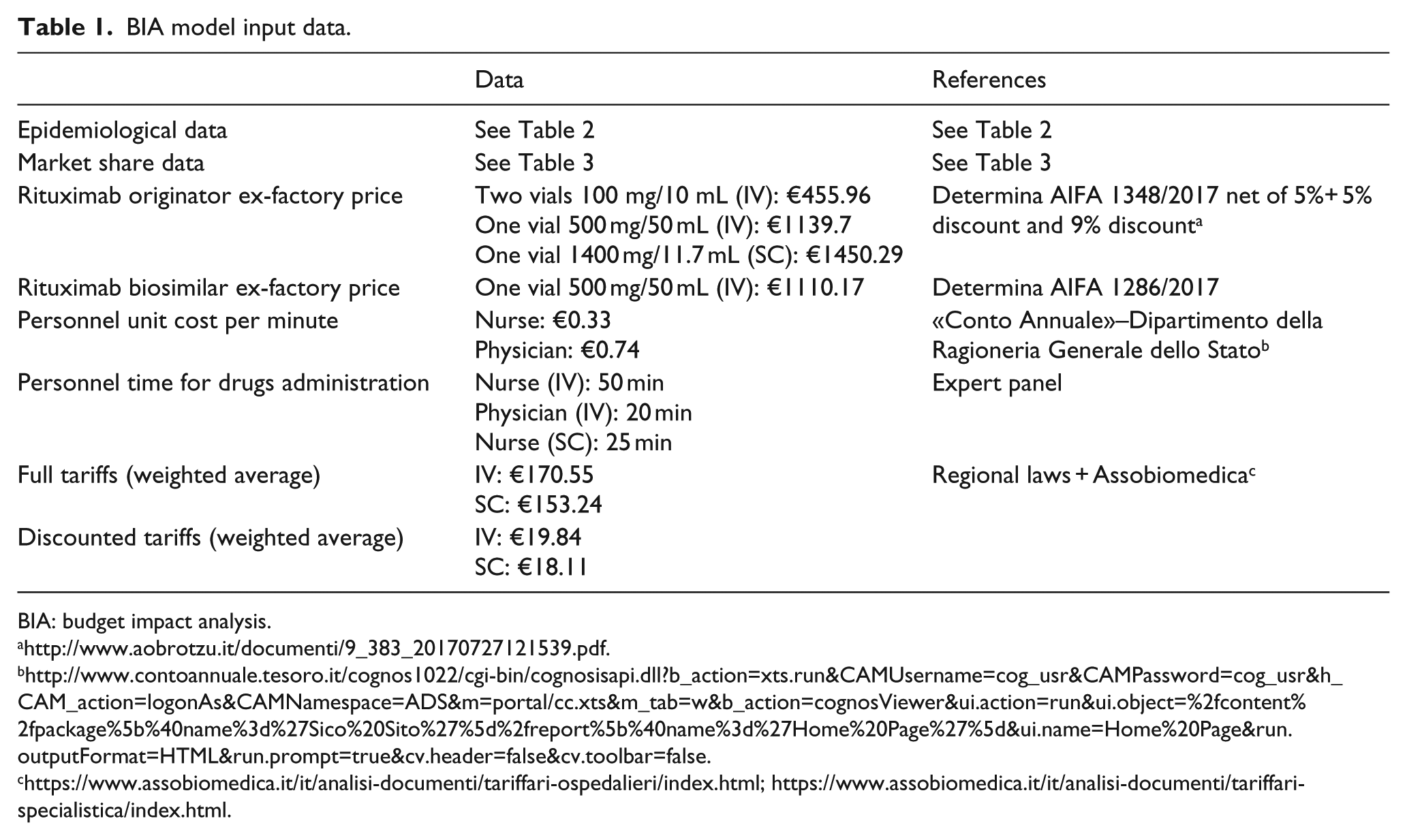
BIA: budget impact analysis.
The BIA model has been designed to allow for additional analyses. For example, a scenario analysis has been performed considering 7% savings if vial sharing is applied. 13
Results
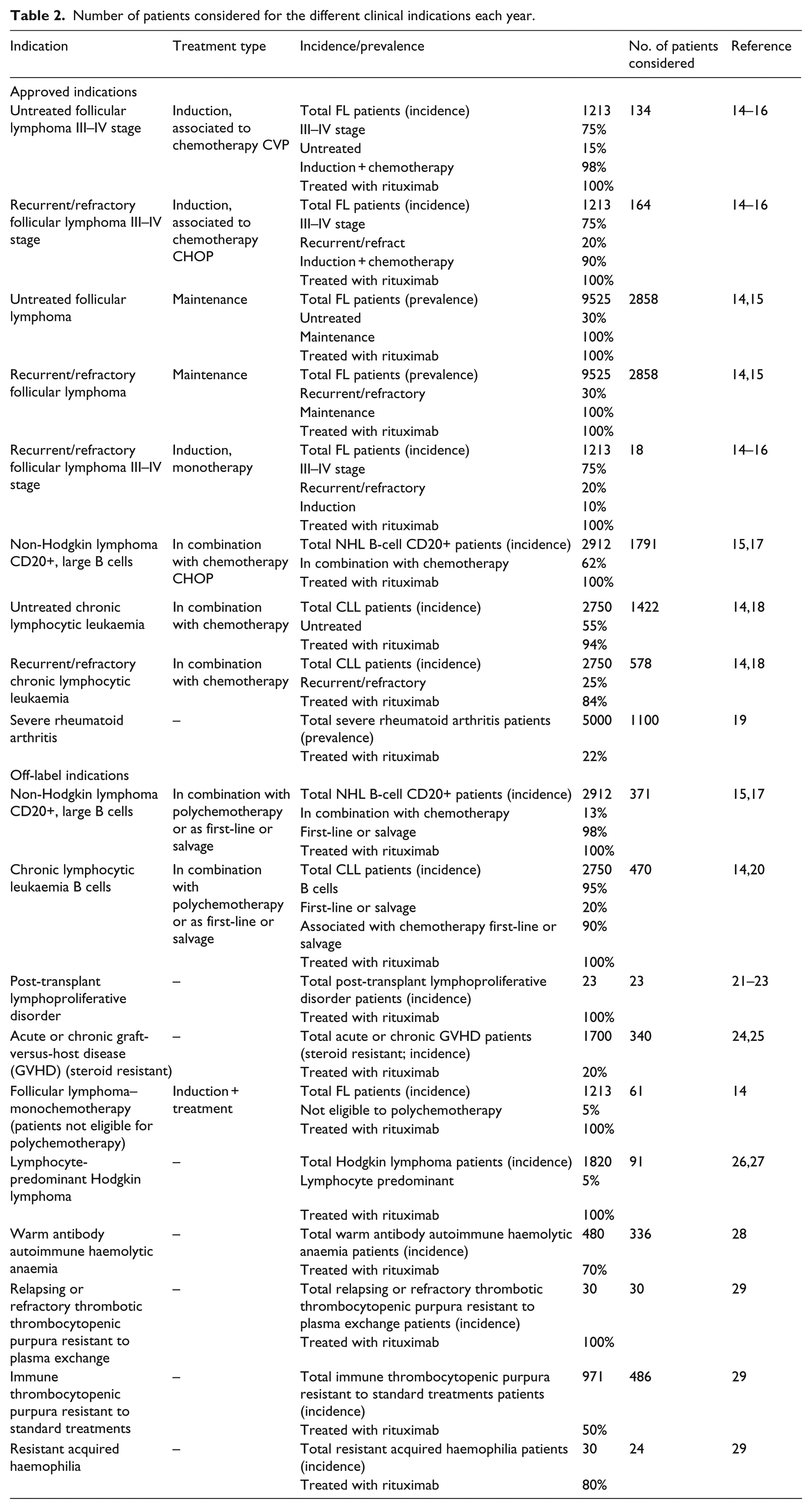
Table 2 illustrates the target population per indication. According to the expert panel, use of rituximab in patients with granulomatosis, polyangiitis and microscopic polyangiitis is very rare in Italy, and these three indications were excluded from the model.
Number of patients considered for the different clinical indications each year.
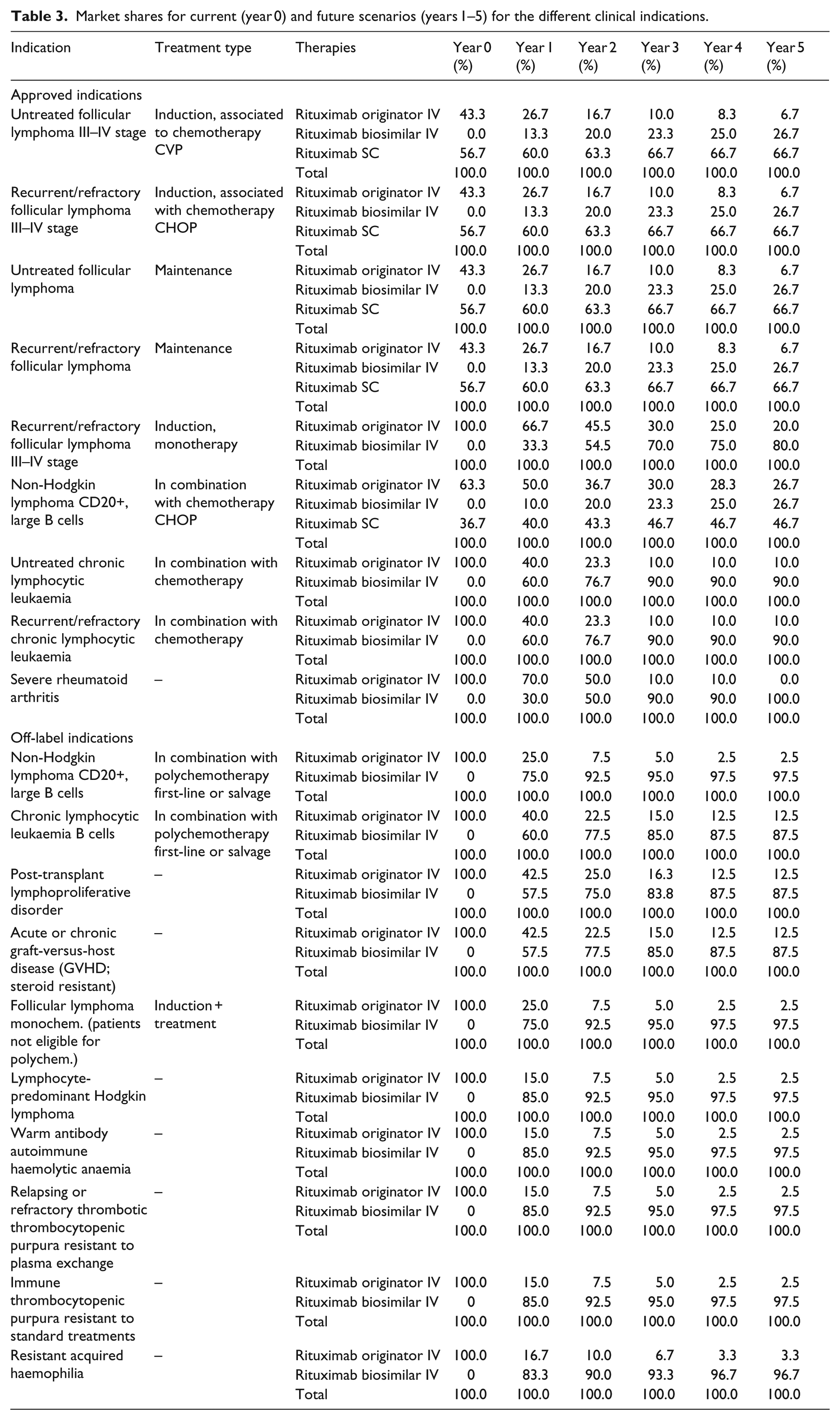
Current (year 0) and future scenarios (years 1–5) of market shares are presented in Table 3. As mentioned before, clinicians agreed that biosimilars will erode market share of the IV rituximab originator without large variations in the trend for the SC formulation and that regional policies (some more hostile to SC administration, others more favourable) will not change for the considered time horizon.
Market shares for current (year 0) and future scenarios (years 1–5) for the different clinical indications.
From the hospital perspective, rituximab would cost €123.8 million under the current scenario. Savings due to biosimilars would reach €16.9, €26.0, €36.2, €36.8 and €37.6 million in the 1st, 2nd, 3rd, 4th and 5th years, respectively, with cumulated savings of €153.6 million in the next 5 years (Figure 1). Results are very similar using the payer perspective, with a cumulated savings of about €153.4 million over the next 5 years. Detailed results are presented in Table 4.
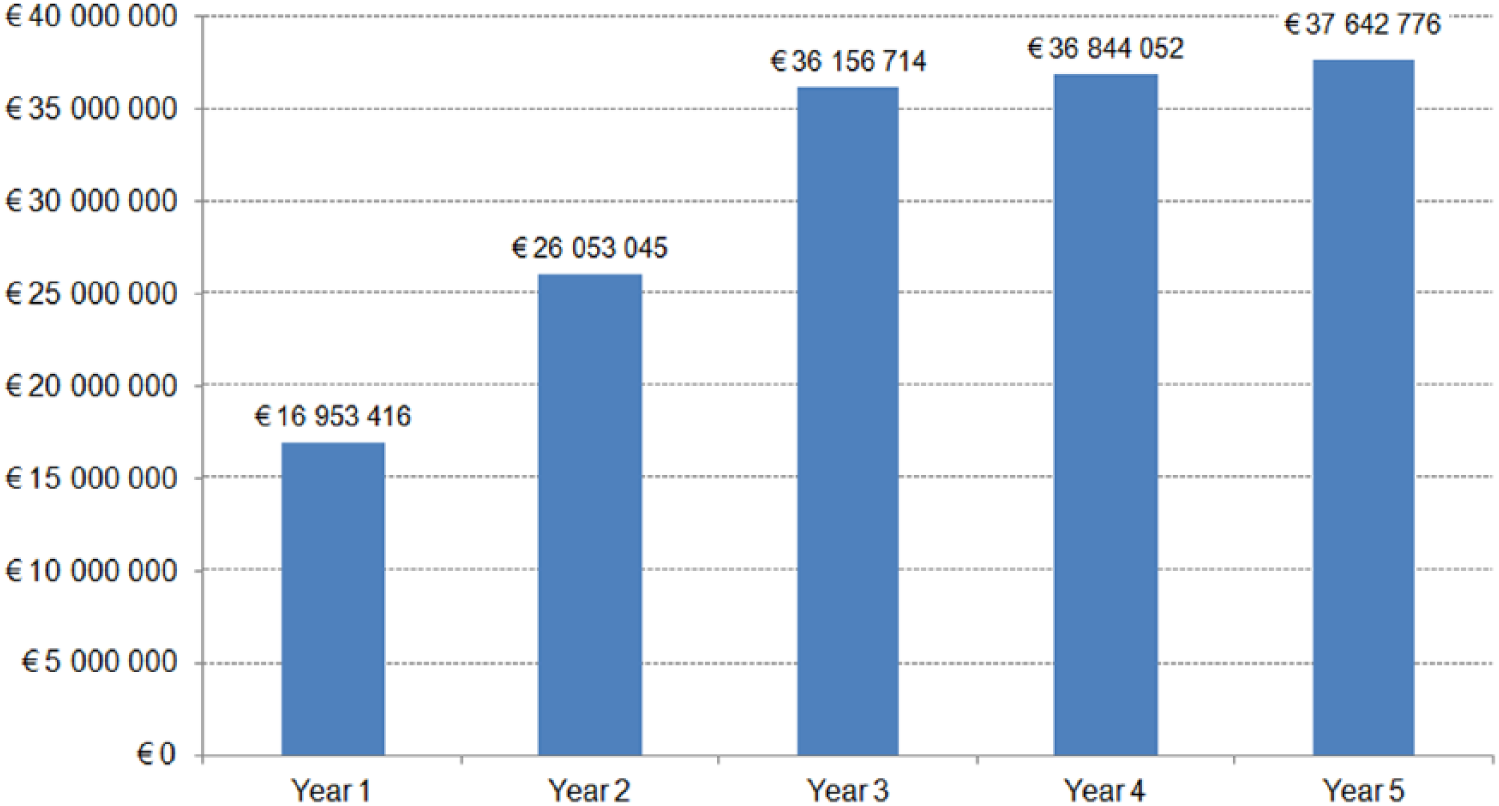
Savings in the future scenario compared to current scenario from the hospital perspective.
BIA according to the different clinical indications and perspectives considered.
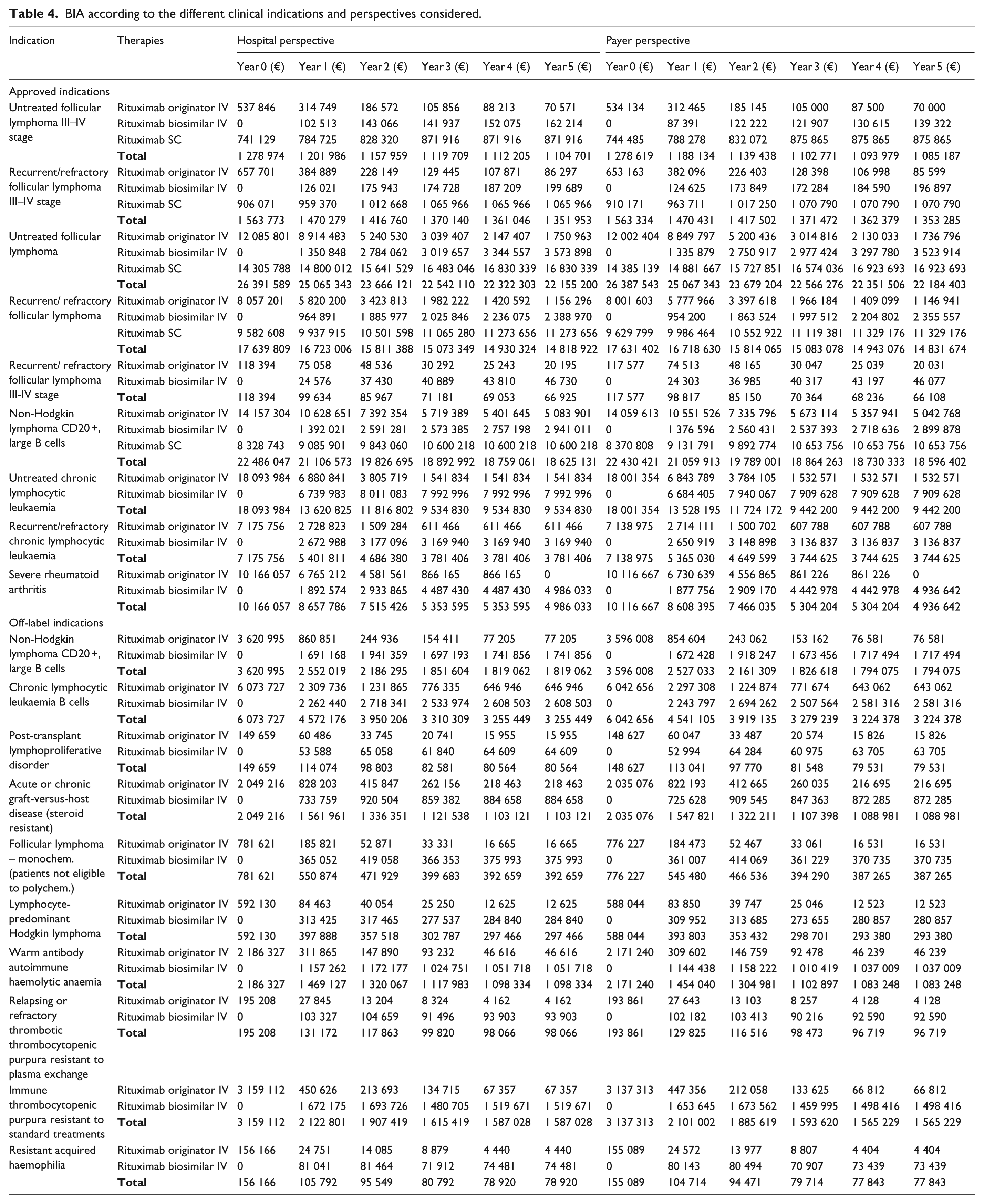
Figure 2 shows that the most important savings derive from the use of rituximab biosimilars for lymphoma (including follicular and non-Hodgkin lymphoma CD20+, large B cells) and chronic lymphocytic leukaemia.
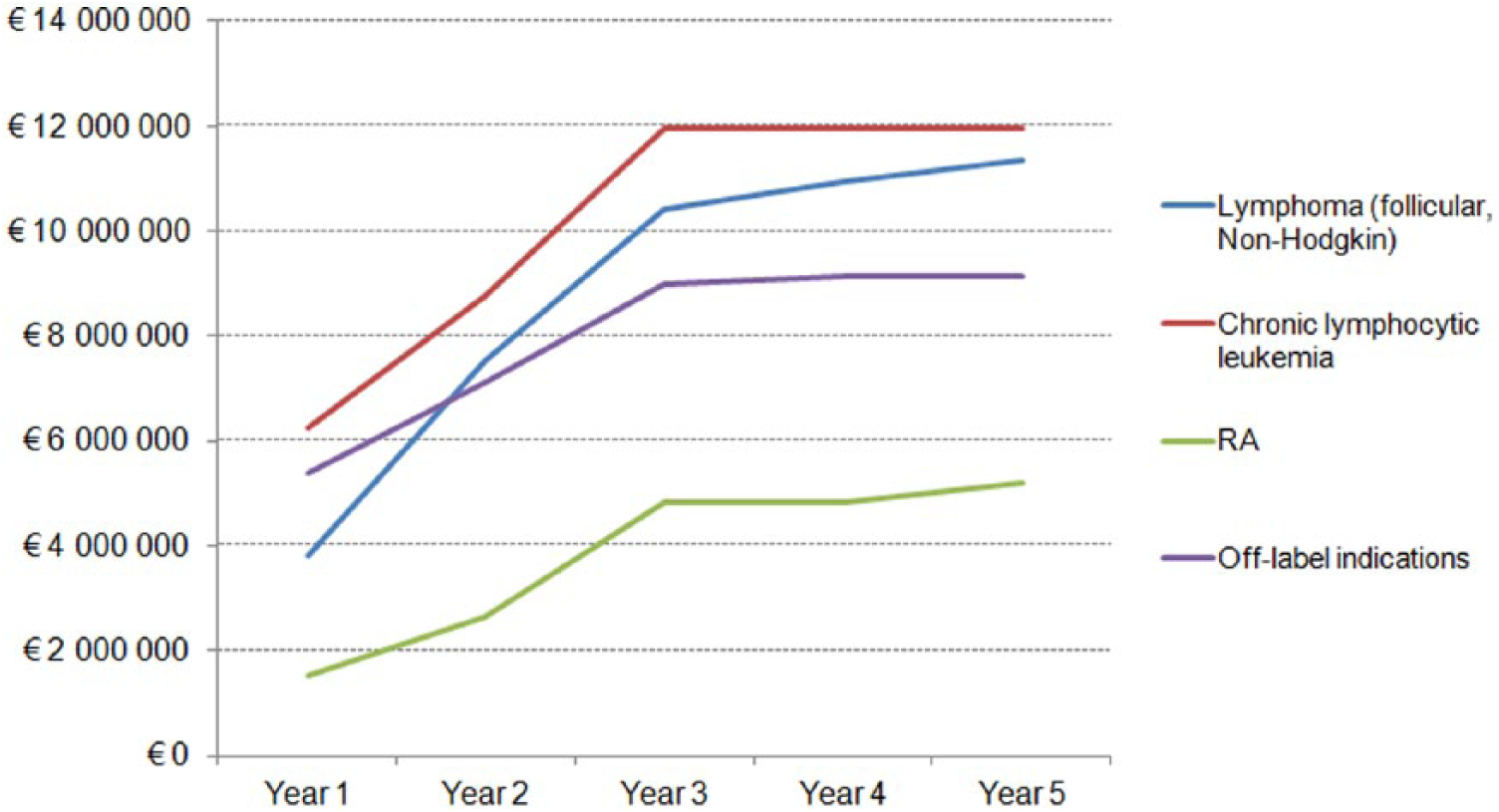
Savings in the future scenario compared to current scenario from the hospital perspective for different indications.
The scenario analysis assuming a 7% discount for vial sharing showed a total savings of €142.7 million for the period considered.
Discussion
This study, to the best of our knowledge, is the first BIA of rituximab biosimilars for all indications in Italy (both approved and off-label covered by the INHS). The BIA model was developed and populated with data retrieved from the literature and integrated and validated through expert opinions.
The model estimated a total savings in a 5-year time horizon of about €153.6 million if the perspective of hospital is used and very similar savings could be reached using the payer perspective.
Since we have adopted a dynamic approach, we were able to estimate savings derived from price competition on originators (67% of total savings) together with savings caused by the biosimilars uptake (33% of total savings). Hence, dynamic effects (i.e. price-competition induced by the biosimilar) seem to be more relevant than reallocation of market shares from the originator to biosimilars. However, since the penetration rate of biosimilars has been derived from expert opinions, it is worth to highlight that the reported values might be not fully representative of the real market shares and the consequent potential total savings.
Savings on drug costs will not be counterbalanced by an increase in drug administration costs, since, according to expert panel opinion, IV biosimilars are expected to gain market share at the expense of the IV rituximab originator and not the SC formulation, which is still under patent protection.
The study has some limitations. First, we relied on the opinions of clinical experts gathered initially through a Delphi approach and then through a collective discussion. We are aware that these ‘key informants’ do not necessarily represent the whole clinical community. However, since the clinicians’ home regions have adopted different policies on biosimilar drugs, our expectation is that they adequately represent the current situation across the country.
Second, we are aware that the Delphi method and experts’ panel approach could foment a propensity to eliminate extreme positions and force a ‘mean’ consensus. 30 Nonetheless, this method is very useful when the problem is not confined to precise analytical techniques, but can benefit from subjective judgements on a collective basis, considering diverse backgrounds with different experience and expertise.
Third, the total dimension of the current market for rituximab per indication was estimated on the basis of epidemiological data, for a total of about €124 million, which is lower than the 2016 INHS spending for rituximab (€156.3 million), according to AIFA. 3 However, we could not rely on the actual expenditure for rituximab since the split per indication is not provided.
Fourth, the BIA model does not incorporate the possible impact of new drugs launched for the same indications as rituximab. The longer the time horizon, the higher the level of uncertainty on our savings estimation. A more prudent 3-year time horizon would imply a €79.2 million savings in the hospital scenario.
Despite these limitations, the study provides for an Italian estimate of potential savings from rituximab biosimilars since it could rely on a per-indication analysis and a panel that represents the diverse policies of regional payers on line-extensions (SC vs IV) and biosimilars versus originators.
The main question is how the savings will be used. Savings can be used just to contain public pharmaceutical expenditure or to fund innovative and cost-effective drugs or can be allocated to fund other healthcare technologies and services. Our hope is that savings re-investment will be the option chosen by the healthcare system.
Footnotes
Acknowledgements
The authors would like to thank the clinicians who joined the expert panel: Professor Robin Foà and Dr Maurizio Martelli (Azienda Ospedaliero-Universitaria Policlinico Umberto I, Rome – Latium); Dr Massimo Gentile (Azienda Ospedaliera di Cosenza, Calabria), Dr Marco Picardi (Policlinico Federico II, Napoli, Campania), Dr Giuseppe Rossi (ASST Spedali Civili di Brescia, Lombardy), Dr Renato Zambello (Azienda Ospedaliera Padova, Veneto) and Dr Ennio Favalli (ASST Gaetano Pini, Milano, Lombardy).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was funded by Sandoz Italia Spa. No interferences occurred in carrying out the research project and in writing the manuscript, which is the sole responsibility of the authors.
