Abstract
To measure the net benefit of a pharmacist-led medication review in acute public hospitals. To identify and measure the resources used when completing a pharmacist-led medication review, an observational study was conducted in an acute urban university teaching hospital. Health Information and Quality Authority guidelines were used to value resources used in a pharmacist-led medication review. Model inputs included demographic data, probability of adverse drug events associated with the pharmacist interventions, estimates of future discharges and cost data. The cost of a pharmacist-led medication review and savings generated from avoidance of adverse drug events were estimated and projected over a 5-year period, using hospital discharge rates taken from the hospital inpatient enquiry system and the census of population. Using the per-patient cost of a medication review, the annual cost of delivering a bi-weekly medication review is projected to vary between €6 m and €6.4 m over a 5-year period from 2017 to 2021. The per-patient net benefit of a bi-weekly medication review is €45.88. Therefore, the projected annual net benefit of a bi-weekly medication review is between €29.5 m and €31.2 m over the 5-year period of 2017 to 2021. Introducing a pharmacist-led medication review for each inpatient saves in the short and longer term. The results are consistent with previous findings. Substantial savings were estimated, regardless of variation in model parameters tested in sensitivity analysis.
Introduction
With the increased prevalence of chronic diseases predominantly driven by an increasing ageing population, healthcare demand in Ireland has grown. It is predicted that 40% of adults in Ireland will have one or more chronic conditions by 2020 1 while internationally chronic disease and multi-morbidity is set to increase.2,3 Given the challenges of scarce resources and constrained public finances, effective dissemination of a health budget is challenging. The growing demand in medication use, particularly by the elderly as well as the rising costs of medicines increases the importance of pharmacist services. Compounding this is the recent recommendation in Ireland that patients in acute hospital settings should receive pharmacist-led medication reviews in line with Health Service Executive (HSE) integrated care guidelines. 4
Medication is one of the most common medical interventions to prevent, treat and manage disease and illness. 5 There are numerous studies that have investigated the impact of inappropriate prescribing and potential adverse drug events (ADEs) which have cost implications for healthcare systems.6,7 In many hospitals, dedicated clinical pharmacy services are available to provide pharmacist interventions (PIs) to optimise medication therapy and potentially reduce the occurrence of ADEs. In Ireland and elsewhere, variations are reported in the level of frequency and availability of clinical pharmacy services, predominantly due to a lack of resources. 8 This may be compounded by the fact that within the Irish jurisdiction there are no national standards of practice in place for clinical pharmacy. 4
A clinical medication review is defined as, the process where a health professional reviews the patient, the illness, and the drug treatment during a consultation. It involves evaluating the therapeutic efficacy of each drug and the progress of the conditions being treated. Other issues, such as compliance, actual and potential adverse effects, interactions, and the patient’s understanding of the condition and its treatment are considered when appropriate. The outcome of the review will be a decision about the continuation (or otherwise) of the treatment.
9
In this study, a clinical medication review was a review of the patients’ prescribed medicines by a pharmacist. A medication review by a hospital pharmacist can result in PI(s). A previous study found variations in the current practice of reviewing medications among hospital inpatients, with some patients not receiving any formal medication review. 8 A systematic literature review has shown the benefits of clinical medication review with PIs leading to a reduction of; inappropriately prescribed medicines; risk of ADEs and hospital stays,8,10–12 but previous studies have highlighted the need for a more rigorous cost analyses using a more quantitative approach. 12 A budget impact analysis (BIA) is defined as a tool to predict the potential financial impact of the adoption and diffusion of a new technology into a healthcare system with finite resources. 13 Therefore, the aim of this study is to conduct a BIA to evaluate the net effect of a clinical medication review, conducted by a pharmacist. This article measures the costs associated with the delivery of a pharmacist-led medication review and estimates the savings generated from this intervention on a per-patient basis.
A BIA is then conducted from a health provider’s perspective using projected inpatient discharge rates in all acute public hospitals to determine the net benefit of a pharmacist-led medication review.
Methods
Data collection
An audit was conducted in an acute urban university teaching hospital, situated in the south of Ireland and serving a population of 417,211 with 350 beds. 14 Ethical approval was granted by the Clinical Research Ethics Committee of the Cork Teaching Hospitals, University College Cork. Data were collected over a 6-month time period from 01 May 2015 to 01 November 2015. For the purposes of this study, patients received a pharmacist-led medication review on average twice weekly. In this study, a total of 1216 patients received medication reviews over this period.
Clinical medication review
In this study the pharmacist-led medication review consisted of a patient drug kardex review (a drug kardex is equivalent to a hospital inpatient prescription) and, if required, was supported by the patient notes and laboratory data but did not involve the patient as a source of data. When a need for a PI(s) was identified, it was brought to the attention of the patient’s medical or surgical team for review. PIs identified were classified according to type, 15 and the medicines associated with the PIs were coded using the Anatomical Therapeutic Chemical (ATC) Classification System. 16
A multidisciplinary approach was adopted to determine the likelihood of an ADE occurring, using an expert panel with the inclusion of a general practitioner (GP) and an academic pharmacist, to the panel of hospital pharmacists. 15 This is similar to Gallagher et al. 10 who also employed an expert panel to determine the likelihood of an ADE occurring if the PI had not been implemented. The panel members independently assigned a probability score to each PI (Table 1). This score reflected the likelihood of an ADE occurring if the PI had not taken place. Five categories were used to score the likelihood of an ADE occurring 10 (Table 1).
PIs and assigned probabilities.
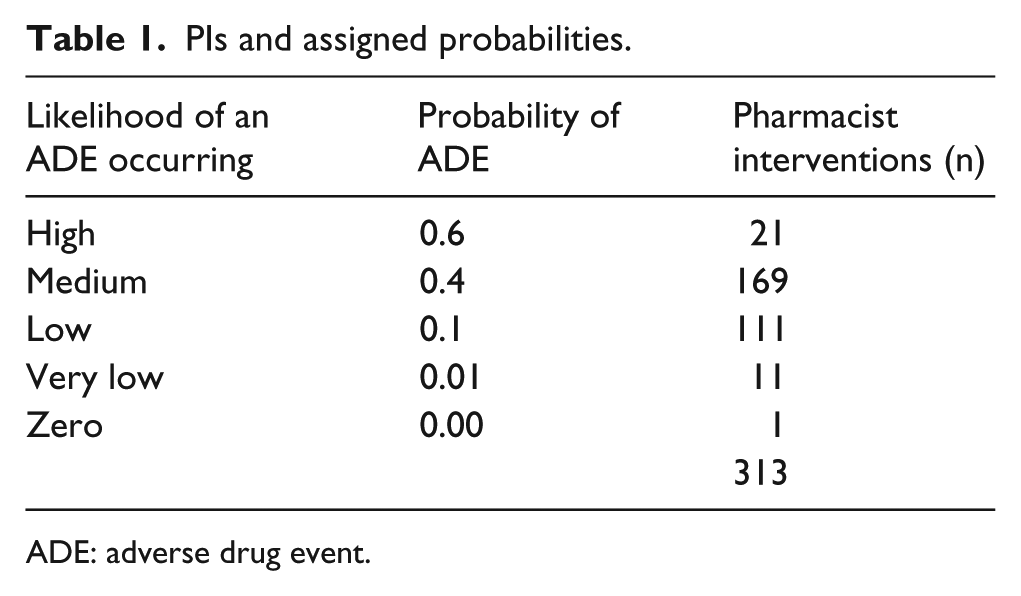
ADE: adverse drug event.
The median probability score for each PI was used in the analysis (Table 1). An inter-rater reliability (IRR) analysis using Cohen’s Kappa statistic was performed to determine agreement among raters. 15
Cost of clinical medication review
A micro-costing approach was adopted to identify, measure, and value the costs associated with providing a clinical medication review. In contrast to previous approaches, an observational study recorded the total time it took for the pharmacist to deliver the medication reviews and resulting PI(s) during the study period, and the average on a per-patient basis was then calculated. The hourly cost of employing a hospital pharmacist is calculated using the HSE salary scales. 17 The hourly cost of employing a hospital pharmacist was then used, and pay-related social insurance (PRSI), pension contributions and hospital overheads were included in the cost, in line with Health Information and Quality Authority (HIQA) guidelines. 18
Potential savings were then determined based on the opportunity cost of avoiding an ADE in direct healthcare which could have occurred in the absence of a clinical medication review. The associated costs of an ADE have been previously found to be €1057 on average per patient. 10 By using the cost of an ADE averted and applying a probability that an ADE would have occurred, without the medication review, potential savings from ADEs averted in the short term were calculated.
Once the costs of the delivery of medication reviews and potential savings from ADEs averted were calculated, the net benefit of medication reviews was determined using a BIA.
A BIA estimates the net benefits from a medication review clinical pharmacy service over time. When undertaking the BIA, a HSE perspective was taken as recommended by HIQA. 18 The target population were inpatients in an acute hospital in the south of Ireland. The age and sex of these inpatients are described in Table 3 in Appendix 1. Population and inpatient discharge data from the Hospital Inpatient Enquiry (HIPE) database 19 and projected population rates were calculated using data from the census of population. 14 These were used to determine and project population trends and predict discharge rates for the next 5 years. A top-down population approach was adopted to estimate the annual number of target inpatients. The resulting net benefit from medication reviews was then projected over a timeframe of 5 years using population estimates.
Results
A total of 1670 pharmacist-led medication reviews were delivered to 1216 patients, and PIs were identified in 213 patients (Figure 1). Patients aged ⩽17 years or those on speciality wards such as oncology were not included in the study. A total of 60.7% of PIs had a ‘medium’ or ‘high’ likelihood of causing an ADE.
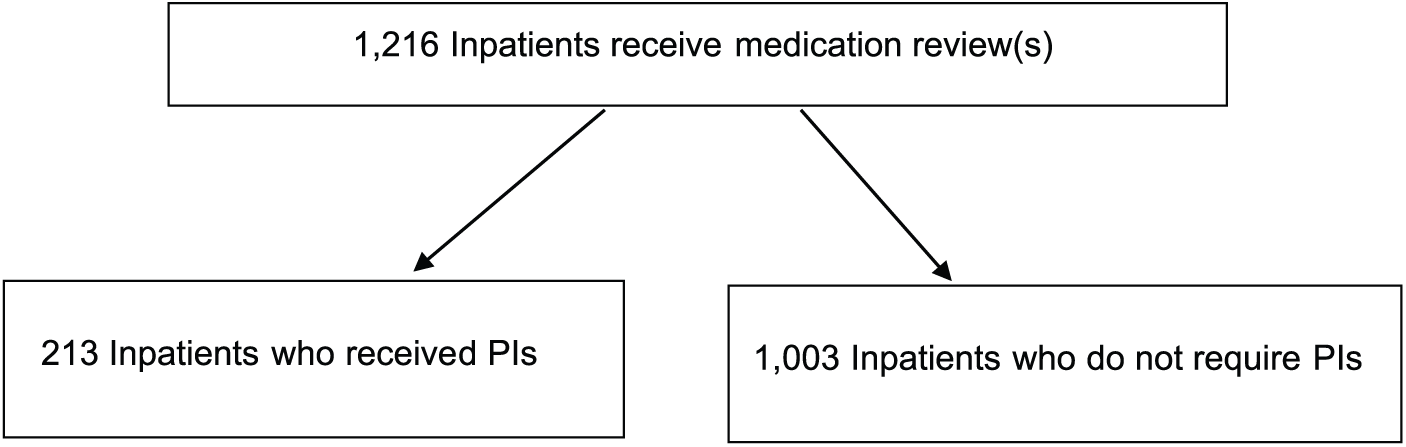
Medication review intervention flow diagram.
The majority of patients with PIs were female which is expected given higher utilisation rates of health service by female patients (refer to Table 3 in Appendix 1). The median age of patients with PIs was 74 years. The majority of patients (90%) with PIs were not undergoing a surgical procedure. The median length of stay for patients with PIs was 10 days. A total of 843 co-morbidities were identified in patients with PIs, which generated a median value of four co-morbidities per patient. The most common condition identified for patients was hypertension (n = 88).
The resulting PIs and their prevalence are displayed in Table 4 in Appendix 1. The IRR analysis using Cohen’s Kappa statistic determined agreement among raters and ranged from slight (0.114) to fair (0.324). 15 Duplication of drug/drug class, poor prescribing practice, frequency, dose and drug–drug interaction were the PIs predominantly identified.
It took 180 hours to deliver the medication reviews and resulting PI(s) to 1216 inpatients during the 6-month study period. From the observational study, the average time it took to deliver a medication review and resulting PI(s) was estimated at 6 minutes 28 seconds per patient. The cost of a pharmacist-led medication review and resulting PI(s) per patient in an acute public hospital was calculated as €4.70. Therefore, the cost of a bi-weekly medication review and resulting PI(s) was €9.40 per patient and used as the baseline.
Potential savings from ADEs averted were estimated by using the probability of an ADE which could have occurred in the absence of a clinical medication review. Gallagher et al. estimated that an ADE costs €1057 on average. There were 313 PIs identified in 213 patients. These 213 patients were from a sample size of 1216 patients. Therefore, the probability of a PI for the sample was estimated at 0.175. Duplication of drug/drug class, poor prescribing practice, frequency, dose and drug–drug interaction were found to be responsible for >70% of the PIs identified (Table 4 in the Appendix 1). Almost two-thirds (61%) of the total number of PIs were categorised by the expert panel as having a ‘medium’ to ‘high’ likelihood of an ADE occurring while the remaining 39% had a ‘low’ to ‘zero’ classification (Table 1). Taking into consideration the categorisation of patients (Table 1), the probability of an ADE occurring for those patients who required a PI is 0.293. The probability of an ADE (probability of an ADE for those who required a PI multiplied by the probability of a PI for the sample) for the total population is 0.051.
Assuming the cost of an ADE is €1084 (adjusted for inflation to 2017 prices) per patient, and given the probability of an ADE for the total population (0.051), the expected monetary benefit of an ADE avoided is €55.28 per patient (only taking into consideration the direct healthcare costs). Once the cost of the intervention is deducted (assuming all patients received a medication review bi-weekly is €9.40) the net benefit is €45.88 per patient.
To estimate the projected cost of service delivery, the cost of service delivery per patient was used as a baseline calculated at €9.40 per patient (bi-weekly medication review), then extrapolated and calculated over a 5-year period for inpatient discharge data. The projected number of inpatient discharges was estimated using the HIPE system (Table 2). 19 The estimated annual cost of delivery for the 2017–2021 period is projected to be between €6–€6.4 m for all acute public hospitals in Ireland.
To estimate the projected annual net benefit, the average saving from an ADE averted minus the cost of the intervention applied to the expected population is calculated. The expected monetary benefit of an ADE avoided is €55.28 per patient, and this is used as the baseline. The projected expected annual monetary benefit from an ADE avoided when direct healthcare costs are considered varies between €35.5–€37.6 m for the period 2017–2021 (Table 2). Whereas, the projected annual net benefit varies between €29.5–€31.2 m from 2017–2021.
Sensitivity analyses
To take into account uncertainty in the model, sensitivity analyses were applied. The length of stay in a public hospital, salary scales, the likelihood of an ADE, the time taken to complete a clinical medication review and the associated costs of an ADE were examined. Within the literature, it is recommended to complete a medication review of patients every day. 8 However, due to a lack of resources, a medication review of each patient could only be implemented in this study at most twice per week. If consideration was given to a daily medication review, this would have implications on the cost of delivery. Previous literature has found that patients with ADE stay marginally longer than the average stay, between 6.52–7.27 days with an average of 6.64 days.20,21 The direct annual cost of daily medication reviews for all patients in acute hospitals would be €20–€21.2 m in years 2017–2021(assuming 6.64 days; refer to Table 5 in Appendix 1). The resulting net benefit would be €98–€103 m in years 2017–2021. When variation in hospital stay is considered, the resulting net benefit changes but still remains positive between €96–€113 m.
For the purposes of this study, the midpoint on the pharmacist salary scale was used to calculate the cost of service delivery. However, if this was allowed to vary to reflect the lowest point on the scale and the highest point on the scale of a higher grade (senior pharmacist salary scale), cost would vary between €3.24 and €6.72 per patient (assuming bi-weekly medication reviews). Then the projected annual costs for bi-weekly medication reviews would increase or decrease depending on salary scale by approximately €2 m on average across the same time period (refer to Table 6 in Appendix 1). With the resulting net benefit on average €32.2 m for the lowest point on the scale (pharmacist salary scale) and €27.7 m for the highest point on the scale (senior pharmacist salary scale) across the same time period. This also resulted in a positive cost benefit ratio (see Figure 2 in Appendix 1).
The likelihood of an ADE avoided was varied each way by 50%, 10 and the expected benefit varies between €27.64 and €82.93 per patient. When costs per patient were considered, the expected net benefit was still positive, ranging between approximately €18 and €74 per patient (assuming delivery of bi-weekly medication reviews) with cost benefit ratios positive also (see Figure 2 in Appendix 1).
The time taken to complete the intervention was on average 6 minutes 28 seconds per patient. This reflects the time taken to deliver a drug kardex review and resulting PI(s) per patient. Previous literature in this area presents a wide variation in time taken to complete clinical pharmacy services. This suggests that the design of the study (i.e. what is included in the intervention) strongly influences the time taken to deliver the intervention. Therefore, it proved difficult to make comparisons across studies. For example, in some studies, a medication reconciliation was included in the intervention, while in others this was not the case. 10 Given previous literature, a range of between 5 and 30 minutes per patient was used.21–24 The expected cost varies between €7.49 and €44.93 per patient. The resulting net benefit is positive and varied between €10.35 and €47.79 per patient, with the overall cost benefit ratio remaining positive (see Figure 2 in Appendix 1).
This was a retrospective study, so a micro-costing of the associated costs of ADE was not possible. Replicating costs were the only data available to this study, as estimated costs of an ADE for hospital inpatients from the Irish Healthcare system were not available. Therefore, it was agreed that the estimated costs of an ADE for hospital inpatients was €1084 per patient, as it was the only cost cited in any Irish research. 10 It is a cost from another healthcare jurisdiction that has been converted to reflect cost of living differences. It is questionable as to whether it is in fact reflective of the Irish healthcare system. To demonstrate variation in cost, previously published ADE outcome measures ranging from €934–€5783 per case 10 were used. The diagnosis-related group (DRG) estimates measure exclusively allergic reactions to drugs and are generally less accurate than micro-costing studies, but these are captured in the range. In addition, more recent international studies were also examined but reported mean costs within the range €934–€5783 per case.20,21 Poudel et al. 20 highlighted that higher costs reported in earlier studies may be due to a lack of adjustment for length of stay or operative procedures. Therefore, this range seems to be the most appropriate. When associated costs of ADE vary, the expected benefit varies between €47.63 and €294.93 per patient. When costs per patient are considered, the expected net benefit would still be positive ranging between approximately €38.23 and €284.93 per patient (assuming delivery of bi-weekly medication reviews). This phenomenon results in positive cost benefit ratios (refer Figure 2 in Appendix 1).
Limitations
The study design and setting influenced the number of PIs identified. In this study, the pharmacist-led medication review consisted of a patient drug kardex review. A PI rate (number of PI(s) per patient who received a PI(s)) of 1.47 was reported in this study, which was lower than the rate of 1.98 reported in a similar study conducted in a university teaching hospital in Ireland. 10 However, in Gallagher et al, medicine omissions, relative to admission notes, represented 43% of the PIs in comparison to 4% of the PIs in this study. Medication reconciliation, identifying medication omissions relative to admission notes, was only opportunistically performed in this study.
In addition, in contrast to Gallagher et al., a multi-disciplinary approach was adopted in this study to determine the likelihood of an ADE, with the addition of a GP and an academic pharmacist to the panel of hospital pharmacists as recommended by previous research. 10 , 15 The composition of the panel and the classi-fication of PIs may have contributed to the probability of an ADE. Furthermore, two of the five most prevalent high-alert medicines as identified by the Institute for Safe Medication Practices (United States) were included, which would affect the probability of an ADE, while none of these medicines were included in the Gallagher et al. paper. 10 , 25
The associated costs of an ADE were found to be €1084 on average per patient 10 and was used in this study. However, it is important to acknowledge that taking an average cost across ADEs although beneficial can underestimate potential cost savings that could be made with certain illnesses. This can have an impact on cost savings. This was a retrospective study so the opportunity to collect patient records was not possible in order to complete a micro-costing of patient outcomes. In addition, there is a lack of published data costing an ADE in the Irish Healthcare system. The ADE cost from the Gallagher et al paper was used, and a sensitivity analysis around the ADE was presented.
Collecting information on patients who did not undergo a medication review was not possible, as this was a retrospective study. Therefore, this study could not determine whether there was a statistically significant difference between patients who had the intervention versus those that did not. This was a single-site study, and expanding the number of sites would have produced more data which would have increased the generalisability of the findings.
Gallagher et al (using results from a previous published study) reports a time of 15–30 minutes per PI. The study design has been found to influence the time taken to deliver the intervention. In this study, the intervention consisted of a patient drug kardex review, whereas in Gallagher et al., the definition of the intervention was much broader and included medicines reconciliation which can be time-consuming. 10 ,15,26 In contrast to Gallagher et al, this study observed the time to deliver this type of medication review and resulting PI(s) in an acute hospital in Ireland. The advantages of this approach is that it was not subject to bias, increased accuracy and provided real-world data.
Various approaches to eliciting probability distributions have been completed. 27 All are grounded in the SHeffield ELicitation Framework (SHELF). 28 There are five elicitation methods available online using the web-based MATCH Uncertainty Elicitation Tool. 27 Of the five elicitation techniques, the probability method was the most similar to this study. 27 Due to a lack of time and resources, these eliciting methods were not applied. However, for future research, the MATCH Uncertainty Elicitation Tool would be helpful to determine agreement between raters.
Conclusion
The projected annual net benefit is between €29.5–€31.2 m over this 5-year period based on the model assumptions outlined. The implementation of this type of clinical pharmacy service is likely to have a positive effect on health outcomes and reduce direct costs to the healthcare system. If this service was provided more widely in all Irish hospitals, there is scope to achieve further benefits.
The results of this study confirmed previous work and supplemented the body of evidence that medication reviews play a key role in preventing potential ADEs while generating savings.
Footnotes
Appendix 1
Projected cost for medication reviews.
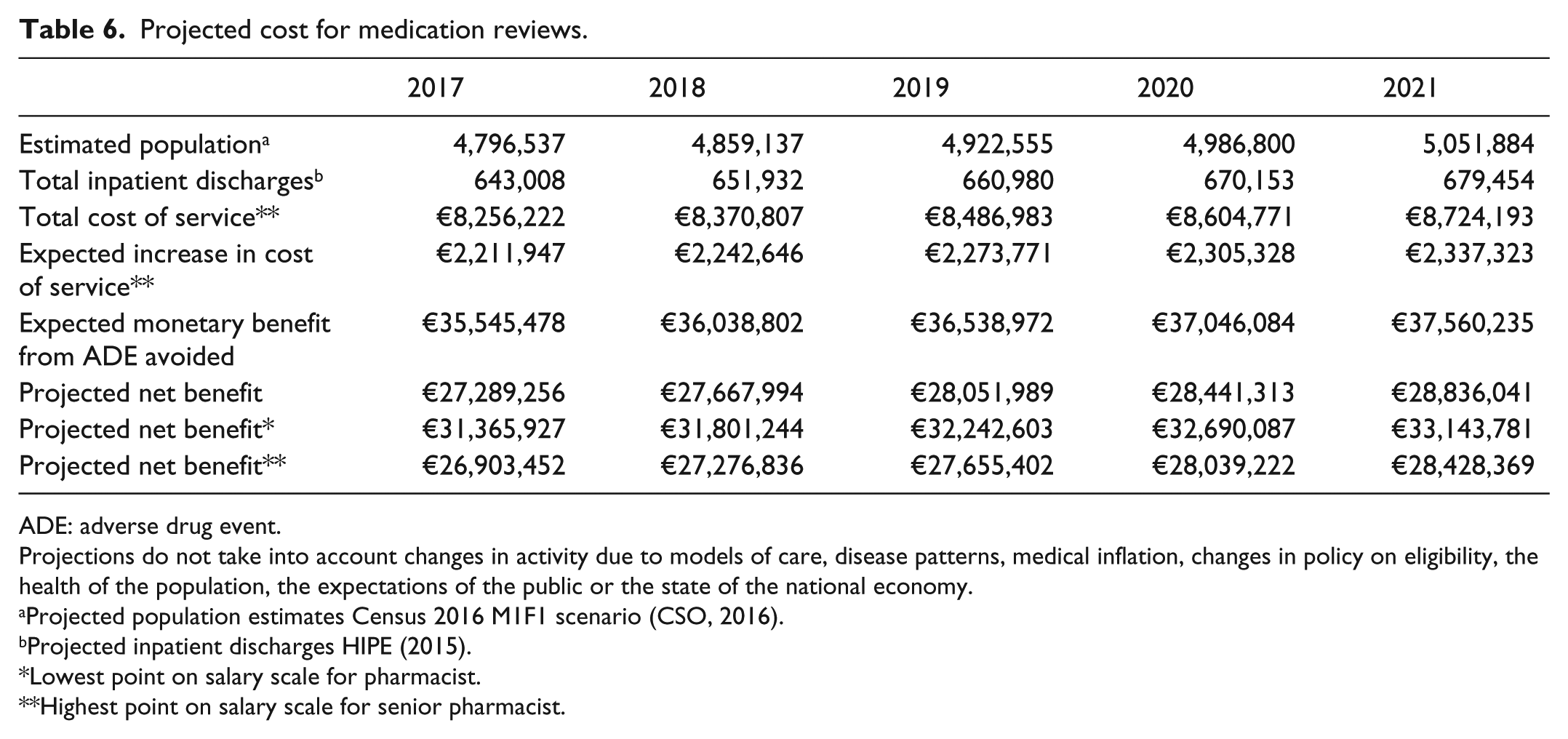
| 2017 | 2018 | 2019 | 2020 | 2021 | |
|---|---|---|---|---|---|
| Estimated population a | 4,796,537 | 4,859,137 | 4,922,555 | 4,986,800 | 5,051,884 |
| Total inpatient discharges b | 643,008 | 651,932 | 660,980 | 670,153 | 679,454 |
| Total cost of service** | €8,256,222 | €8,370,807 | €8,486,983 | €8,604,771 | €8,724,193 |
| Expected increase in cost of service** | €2,211,947 | €2,242,646 | €2,273,771 | €2,305,328 | €2,337,323 |
| Expected monetary benefit from ADE avoided | €35,545,478 | €36,038,802 | €36,538,972 | €37,046,084 | €37,560,235 |
| Projected net benefit | €27,289,256 | €27,667,994 | €28,051,989 | €28,441,313 | €28,836,041 |
| Projected net benefit* | €31,365,927 | €31,801,244 | €32,242,603 | €32,690,087 | €33,143,781 |
| Projected net benefit** | €26,903,452 | €27,276,836 | €27,655,402 | €28,039,222 | €28,428,369 |
ADE: adverse drug event.
Projections do not take into account changes in activity due to models of care, disease patterns, medical inflation, changes in policy on eligibility, the health of the population, the expectations of the public or the state of the national economy.
Projected population estimates Census 2016 M1F1 scenario (CSO, 2016).
Projected inpatient discharges HIPE (2015).
Lowest point on salary scale for pharmacist.
Highest point on salary scale for senior pharmacist.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
