Abstract
Purpose:
We aimed to estimate the 3-year budget impact of replacing salmeterol/fluticasone with indacaterol/glycopyrronium for the treatment of chronic obstructive pulmonary disease patients in Italy.
Methods:
The budget impact model included patients with moderate, severe, and very severe chronic obstructive pulmonary disease and was developed using data from the FLAME study. The budget impact model considered direct medical costs from the National Healthcare System perspective over a 3-year time-horizon. Costs of drugs, adverse events, and exacerbations were estimated using public net prices for drugs and national (inpatient/outpatient) tariffs for patient management. Data on adverse events and exacerbations were derived from FLAME and other clinical trials. The model assumed an increasing uptake of indacaterol/glycopyrronium, substituting it for salmeterol/fluticasone. Two sensitivity analyses were conducted. In the first, every long-acting muscarinic antagonist/inhaled corticosteroid switch to indacaterol/glycopyrronium; in the second, drugs compliance varies from 20% to 60%.
Results:
The target population was estimated at 572,788 patients in the first year, increasing to 585,491 in the third year. The alternative treatment pattern, with a higher indacaterol/glycopyrronium uptake, was associated with a total cost of €1.69 billion over 3 years, while the total costs in the current treatment pattern amounted to €1.71 billion. Thus, increasing indacaterol/glycopyrronium use provided total savings of €17.3 million over 3 years. Sensitivity analysis confirmed robustness of the results.
Conclusion:
Increasing the uptake of indacaterol/glycopyrronium was estimated to be cost-saving for the Italian National Healthcare System, both when drug-related expenditures and total costs were considered. Savings increased with the number of patients switching to indacaterol/glycopyrronium.
Introduction
Chronic obstructive pulmonary disease (COPD) is a common, preventable, and treatable disease, characterized by persistent airflow limitation associated with a chronic inflammatory response. Chronic respiratory symptoms may precede the development of airflow limitation and can be associated with an acute worsening of respiratory symptoms called an exacerbation. 1 The Global Initiative for Chronic Obstructive Lung Diseases (GOLD) expert panel classified COPD into four groups based on severity, ranging from mild (group A), through moderate (group B) and severe (group C), to very severe (group D). 1
Although COPD is associated with a significant economic and societal burden, the true extent of its impact is frequently underestimated due to under-reporting of symptoms and/or poor disease management.1–3 According to GOLD strategy, therapy choice should be driven by COPD severity. 1 However, in clinical practice, inappropriate prescribing of available treatments is common, increasing COPD management costs. For instance, there is a high level of under-diagnosis/under-treatment and/or misdiagnosis/mistreatment in the early COPD stages, highlighting the problem of inadequate standards of care that may adversely impact on the whole spectrum of COPD management, from prevention to long-term care.4,5 Consequently, patients affected by COPD are managed in a discontinuous and non-integrated way, without well-coordinated care procedures. 5 Importantly, inappropriate diagnosis and management of COPD increase its economic burden.6,7 In Italy, observational data confirm the findings from international studies, revealing poor adherence of current clinical practice to GOLD international strategy. 8
There is also a direct relationship between the severity of COPD and the cost of care. Specifically, cost distribution changes with disease progression, increasing in more severe cases because of hospitalizations also prompted by exacerbations.9,10 Several economic studies reported that COPD exacerbations are associated with a high risk of hospitalization and death, worsening quality of life, and an increase in healthcare costs.2,10–13
In the Italian general population, the prevalence of COPD is estimated at 3.1%; this means that about 2 million people across the country are affected by the disease.14,15 The burden of COPD in Italy is very high—its direct costs have been estimated at approximately €7 billion. 16 Costs of lost productivity add further to this burden, contributing approximately a tenth of the total per-patient cost. 13
Several compounds with different mechanisms of action are currently available on the Italian market for the treatment of COPD. These include bronchodilators, such as short-acting muscarinic antagonists (SAMAs), short-acting β2-agonists (SABAs), long-acting muscarinic antagonists (LAMAs), long-acting β2-agonists (LABAs), and anti-inflammatory drugs—inhaled corticosteroids (ICSs) and phosphodiesterase-4 (PDE-4) inhibitors. These medications can be used as monotherapy and in flexible-dose combinations; several fixed-dose combinations are also available.17,18
Bronchodilators are the corner stone of COPD treatment. The most recent GOLD strategy report that combining bronchodilators with different mechanisms and durations of action may increase the degree of bronchodilation with a lower risk of side-effects compared to increasing the dose of a single bronchodilator used in monotherapy. 1 Indeed, combined treatments can be useful in patients whose disease is not adequately controlled by a single active agent, such as a LABA or a LAMA. 19 Combined LABA/LAMA treatment is indicated in all COPD patients apart from those with mild disease (group A). In particular, the GOLD strategy recommends initiating a LABA/LAMA combination in group D patients, as it has proven more effective in improving patient-reported outcomes (PROs) compared with long-acting bronchodilator monotherapy, and in both improving PRO and reducing exacerbations compared with the ICS/LABA combination. 1
Indacaterol (IND) is a LABA that provides prolonged bronchodilation lasting at least 24 hours.20,21 Glycopyrronium (GLY) is a LAMA, which—similarly to IND—induces rapid and prolonged bronchodilation.22,23 Since LABAs and LAMAs act via different mechanisms, when used together, they may be additive bronchodilating effects. 24 Thus, IND and GLY are used together to optimize bronchodilation in COPD patients. 19
The FLAME study evaluated the role of the LABA/LAMA regimen versus LABA/ICS in COPD patients with a history of one or more exacerbations in the previous year. 25 FLAME was a 52-week, randomized, double-blind, double-dummy, non-inferiority trial which compared dual bronchodilator therapy with IND/GLY versus an ICS/LABA combination (salmeterol/fluticasone) (SFC). The trial showed that IND/GLY was more effective than SFC in preventing COPD exacerbations in patients with a history of 1 or more exacerbation during the previous year, and that this result was independent of the baseline blood eosinophil count. Furthermore, the rate of pneumonia was significantly inferior in the IND/GLY group. 25
In Italy, the fixed-dose combination of IND/GLY is currently indicated and reimbursed by the National Healthcare System (NHS) as a maintenance bronchodilator treatment to relieve symptoms in adult patients with COPD. 26
Objectives
The objective of this study was to analyze, based mainly on the FLAME study results, the economic impact of increasing the uptake of IND/GLY in Italy over 3 years at the expense of SFC.
Methods
Model
A deterministic budget impact model (BIM) focused on direct healthcare costs was developed from the perspective of the Italian NHS, in accordance with the recommendations published by the International Society for Pharmacoeconomics and Outcomes Research (ISPOR) Task Force on Good Research Practices.27,28 The model compared the current treatment pattern in Italy with an alternative treatment pattern, in which a proportion of patients switched from SFC to IND/GLY over a time-horizon of 3 years (2018–2020).
Population
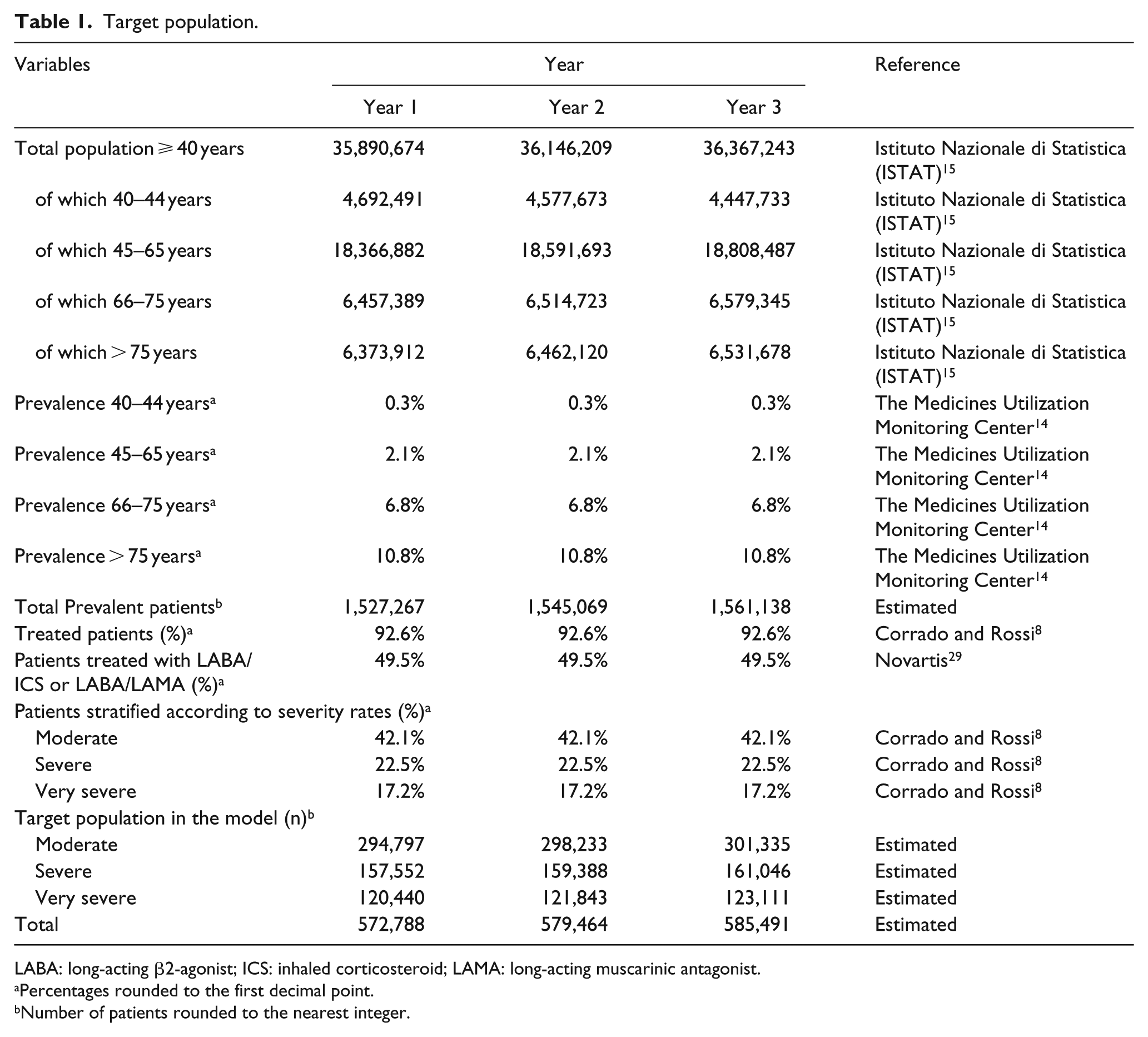
The population of interest included Italian COPD patients aged 40 years or older. Age-specific COPD prevalence rates were used: 0.3% at the age of 40–44 years, 2.1% at 45–65 years, 6.8% at 66–75 years, and 10.1% in patients aged over 75 years.14,15
In line with a previously published Italian study, 8 the BIM assumed 92.6% of COPD patients received treatment. Among those, 49.5% were treated with LABA/LAMA or LABA/ICS. 29 Patients were stratified by disease severity, and those with moderate-to-very-severe disease were included in the model (i.e. 81.8% of all COPD patients). 8 Patients with mild disease were excluded from the analysis, as treatment with a single bronchodilator is more suitable for this group. 1 Characteristics of the target patient population are reported in Table 1.
Target population.
LABA: long-acting β2-agonist; ICS: inhaled corticosteroid; LAMA: long-acting muscarinic antagonist.
Percentages rounded to the first decimal point.
Number of patients rounded to the nearest integer.
Treatments and market shares
All drugs investigated in the BIM are available and reimbursed by the NHS in Italy. The therapies considered in the analysis were:
LABA/LAMA: IND/GLY, vilanterol/umeclidinium (VUM), formoterol/aclidinium (FAC), and olodaterol/tiotropium (OTI).
LABA/ICS: SFC, formoterol/budesonide (FBD), vilanterol/fluticasone (VFL), and formoterol/beclomethasone (FBE).
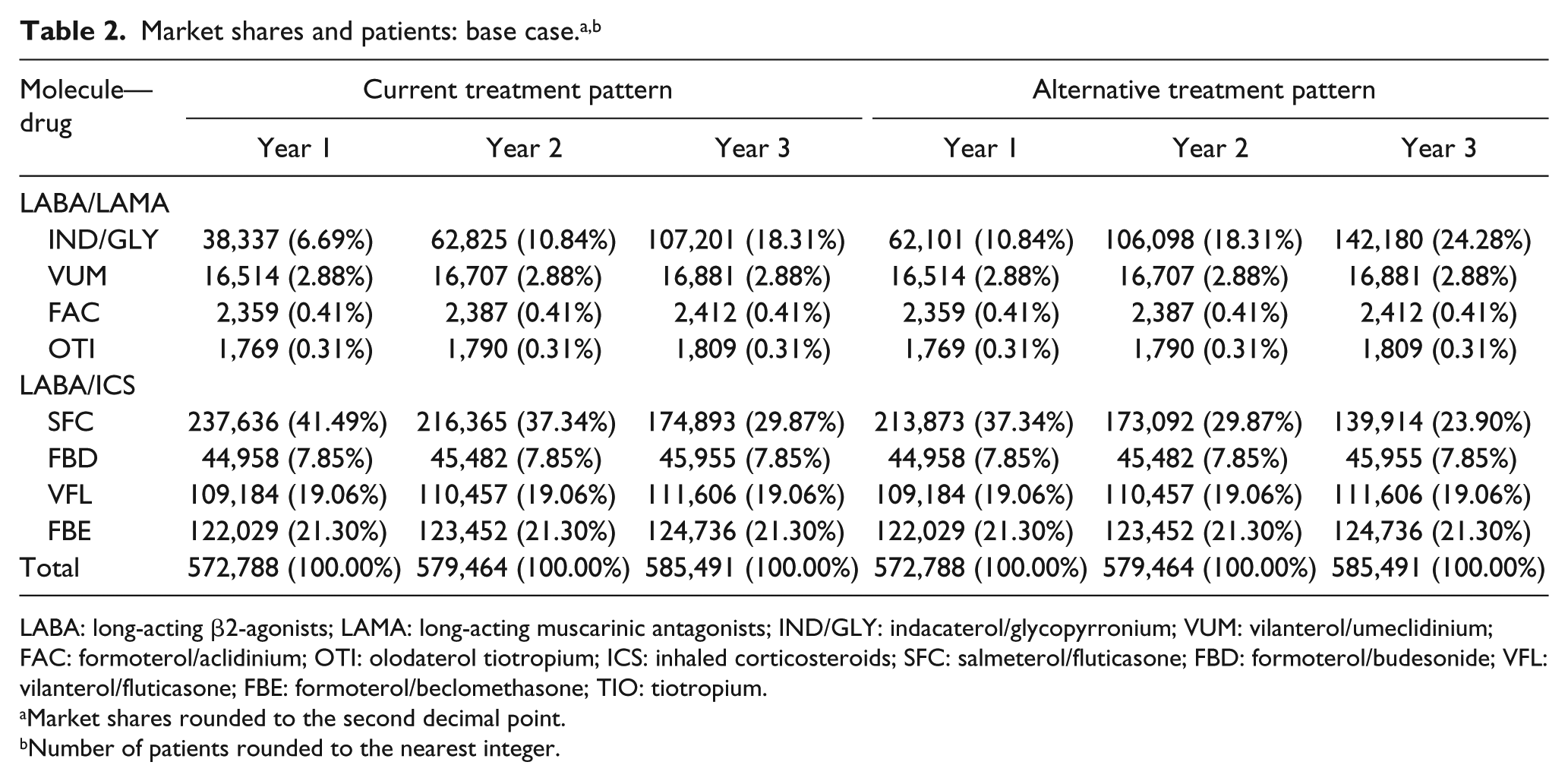
In the current treatment pattern, it was assumed that the market share of SFC would decrease by 10% in the first year (from 41.49% to 37.34%) and by 20% in the second and third years and that it would be replaced by IND/GLY (Table 2).
LABA: long-acting β2-agonists; LAMA: long-acting muscarinic antagonists; IND/GLY: indacaterol/glycopyrronium; VUM: vilanterol/umeclidinium; FAC: formoterol/aclidinium; OTI: olodaterol tiotropium; ICS: inhaled corticosteroids; SFC: salmeterol/fluticasone; FBD: formoterol/budesonide; VFL: vilanterol/fluticasone; FBE: formoterol/beclomethasone; TIO: tiotropium.
Market shares rounded to the second decimal point.
Number of patients rounded to the nearest integer.
Clinical data
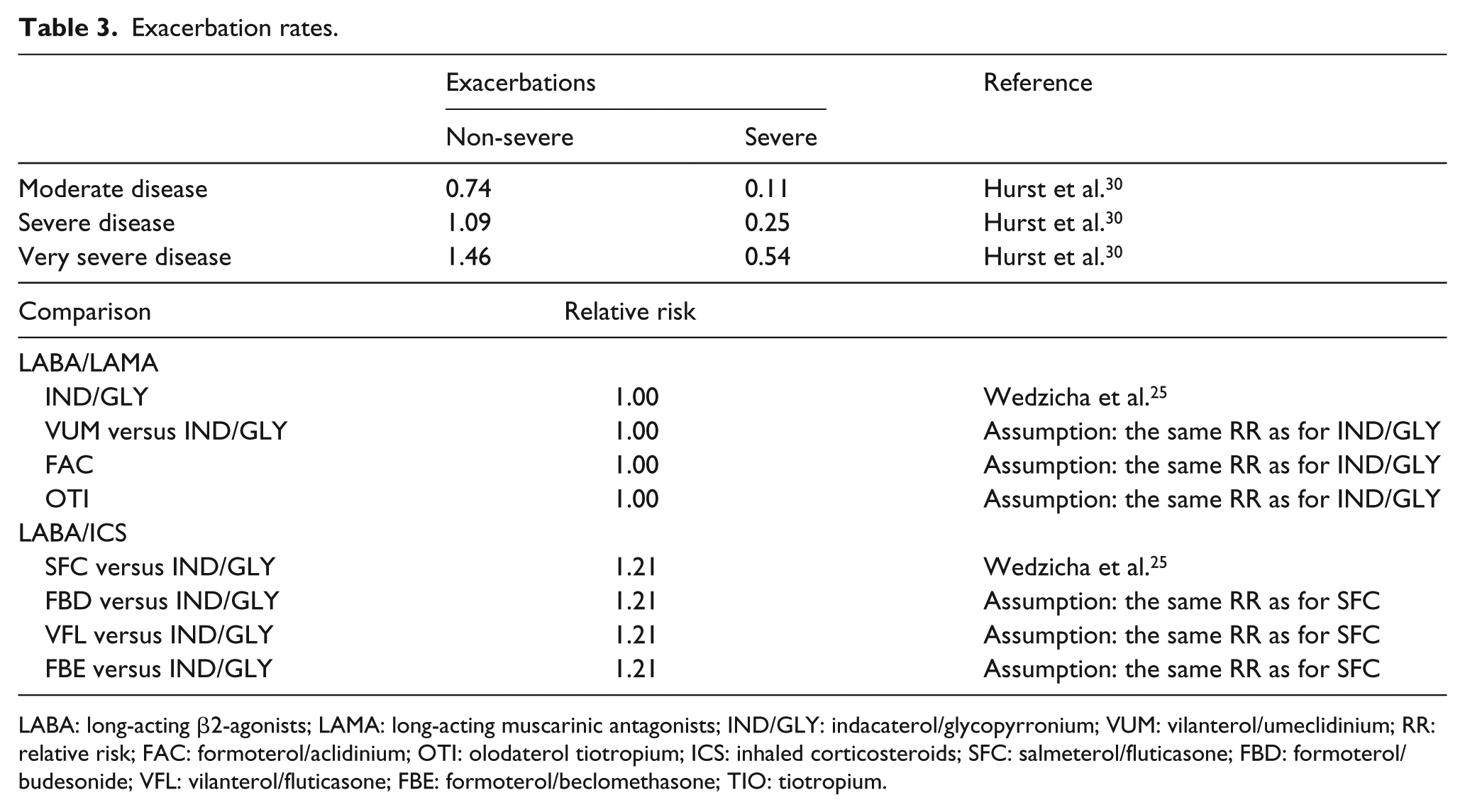
The BIM reflects patient characteristics in the FLAME study, which directly compared IND/GLY versus SFC. 25 The rates of severe and non-severe exacerbations and those of serious adverse events (SAEs) associated with the different compounds modeled were retrieved from the FLAME and ECLIPSE clinical studies, as well as from published the literature.25,30,31 In line with the FLAME and ECLIPSE trials, in the BIM, the per-patient exacerbation rate varied depending on disease severity.25,30 Details are reported in Table 3. In terms of SAEs, pneumonia and tuberculosis were included in the model. Published data showed IND/GLY, and LABA/LAMA in general, have a more favorable safety profile than LABA/ICS, including SFC.25,31 Consequently, in the model, the incidence of pneumonia was 3.20% with LABA/LAMA and 4.8% with LABA/ICS, while the incidence of tuberculosis was 0.06% and 0.15%, respectively.25,31
Exacerbation rates.
LABA: long-acting β2-agonists; LAMA: long-acting muscarinic antagonists; IND/GLY: indacaterol/glycopyrronium; VUM: vilanterol/umeclidinium; RR: relative risk; FAC: formoterol/aclidinium; OTI: olodaterol tiotropium; ICS: inhaled corticosteroids; SFC: salmeterol/fluticasone; FBD: formoterol/budesonide; VFL: vilanterol/fluticasone; FBE: formoterol/beclomethasone; TIO: tiotropium.
Costs
The BIM estimated the costs of drugs, exacerbations, and SAEs from the perspective of the Italian NHS. We used actual net public prices, according to the Italian market data, 18 for both COPD treatments and concomitant therapies (such as those used to alleviate SAEs); an additional discount of 5% was considered for drugs to which a payback agreement applies, in line with a previous Italian study. 32 To estimate the actual drug dose a compliance rate of 40% (i.e. the percentage of drugs’ daily dose taken by patients), according to Italian market data, 29 was applied to the recommended posology of each COPD drug. 33 The compliance rate affects only the COPD drugs cost. Overall daily drug costs, according to actual net prices, recommended posology and compliance rate, were estimated at €0.80 for LABA/LAMA; €0.86 for SFC; €0.79 for FBD; €0.66 for VFL; and €0.69 for FBE.18,29,33
National inpatient and outpatient tariffs and hospital drug prices were used to quantify the cost of both exacerbations and SAEs. 34 For non-severe exacerbations, we have taken into account the costs of specialist visits, antibiotic, and oral steroids;18,34 the consumption of these resources was estimated internally according to clinical expert opinion. For estimating the cost of both severe exacerbations and SAEs, the hospitalization cost according to the National inpatient tariffs was used; 33 integrating cost data for severe exacerbations retrieved from the literature.34,35 Especially, the BIM assumed that 10% of discharged patients required domiciliary oxygen and that this was administered for 1 year (Table 4). 35
Other healthcare costs.
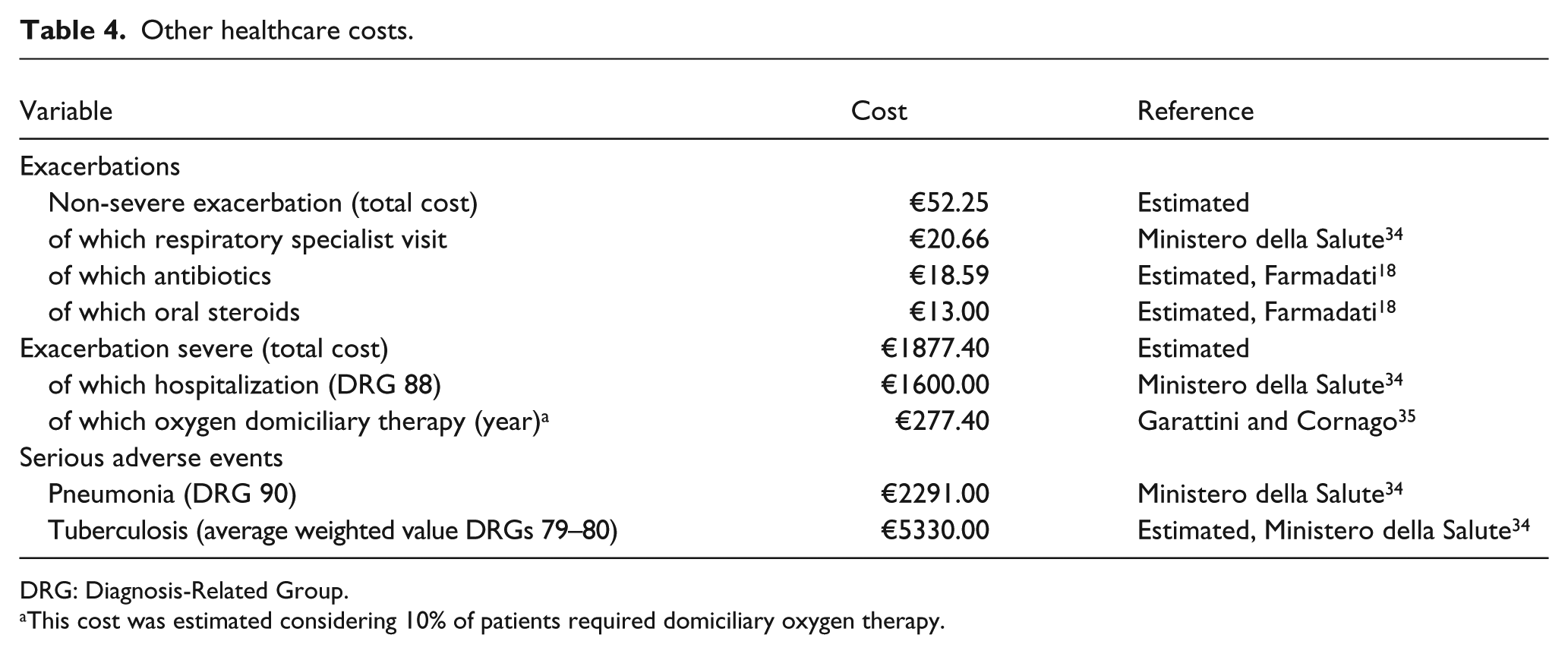
DRG: Diagnosis-Related Group.
This cost was estimated considering 10% of patients required domiciliary oxygen therapy.
Sensitivity analyses
To evaluate the robustness of the results, two sensitivity analyses were developed.
In the first sensitivity analysis (A), all LABA/ICS combinations were replaced with IND/GLY. We assumed 5% decrease, in the first year, and 8% in the second and third years for LABA/ICS and a complete replacement with IND/GLY. In the second one, we investigated two variants, one assuming a compliance of 20% for each drug (analysis B) and the other one a compliance rate of 60% (analysis C).
Results
The number of COPD patients aged ⩾ 40 years ranged from 1,527,267 in the first year to 1,561,138 in the third year. From this, the target population of patients treated with LABA/LAMA or LABA/ICS and suffering from moderate, severe, or very severe COPD was estimated at 572,788 in the first year, increasing to 585,491 in the third year (Table 1).
Base case analysis
In the current treatment pattern, the market shares of IND/GLY were estimated at 6.7% in 2017 and 18.3% in 2019, corresponding to 38,337 and 107,201 treated patients, respectively. For SFC, the market shares decreased from 41.5% (237,636 patients) to 29.9% (174,893 patients) over the same time period. In the alternative treatment pattern, the market share of IND/GLY increased from 10.8% to 24.3% over the model time-horizon so that the number of treated patients increased from 62,101 in the first year to 142,180 in the third year. Conversely, the estimated market share of SFC decreased from 37.3% (213,873 patients) in the first year to 23.9% (139,914 patients) in the third year. Details of market shares are reported in Table 2.
Comparing the alternative and current treatment pattern over the full time-horizon, we saw that increased utilization of LABA/LAMA (IND/GLY) as opposed to LABA/ICS (SFC) resulted in a reduced number of exacerbations and SAEs. In the alternative treatment pattern, the number of non-severe exacerbations was lower by 21,591, and that of severe exacerbations by 5,223, than in the current treatment pattern. Similarly, the alternative treatment pattern resulted in 1632 fewer cases of pneumonia and 92 fewer cases of tuberculosis than the current treatment pattern.
In terms of costs, SFC was associated with the highest drug cost, followed by LABA/LAMA (including IND/GLY), while VFL and FBE were least expensive. However, the reduced SAE incidence with LABA/LAMA and the lower number of exacerbations observed with this drug class provided cost offsets so that when the costs of SAEs and exacerbations were also considered, the total costs of all LABA/ICS combinations were consistently higher than those of LABA/LAMA (Table 5).
Mean cost per patient/year: base case.
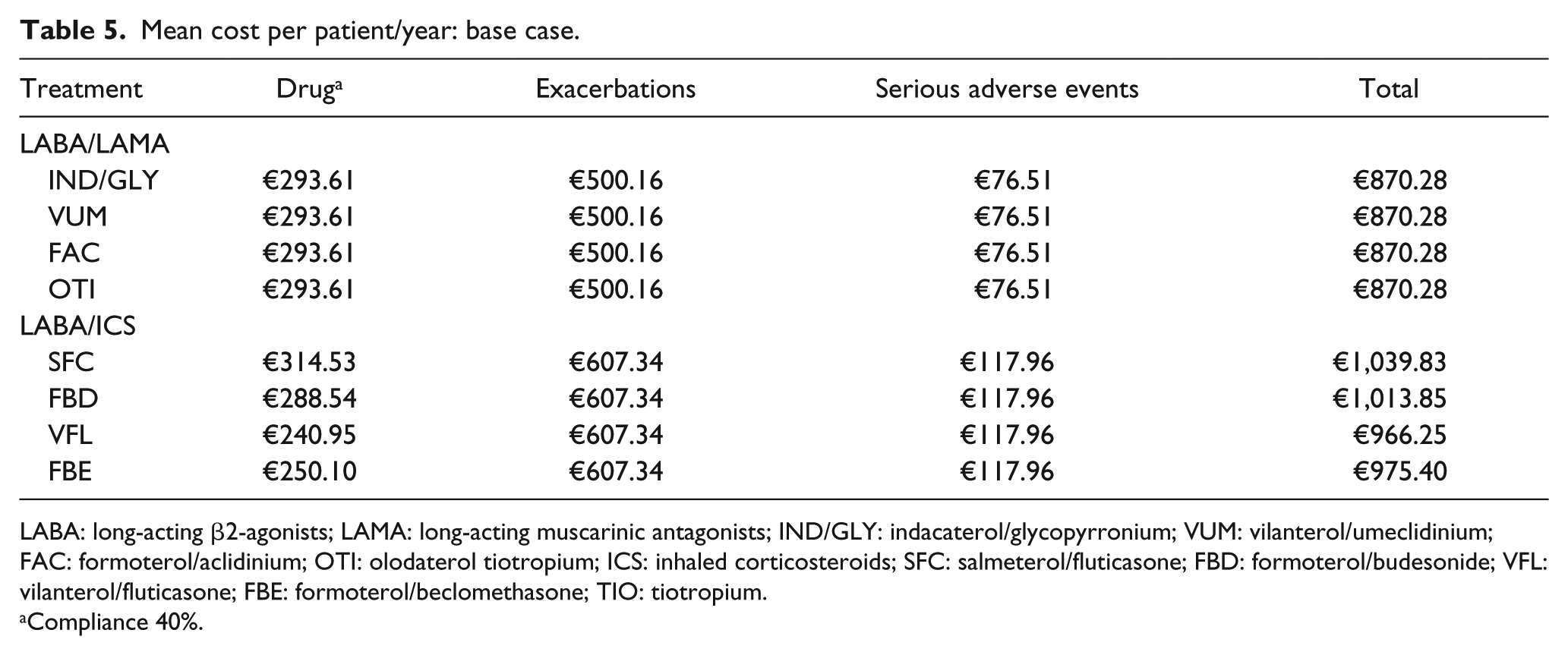
LABA: long-acting β2-agonists; LAMA: long-acting muscarinic antagonists; IND/GLY: indacaterol/glycopyrronium; VUM: vilanterol/umeclidinium; FAC: formoterol/aclidinium; OTI: olodaterol tiotropium; ICS: inhaled corticosteroids; SFC: salmeterol/fluticasone; FBD: formoterol/budesonide; VFL: vilanterol/fluticasone; FBE: formoterol/beclomethasone; TIO: tiotropium.
Compliance 40%.
In the current treatment pattern, the total expenditure was estimated at €568.5 million in the first year and €569.6 million in the third year (Table 6). During that time, spending on IND/GLY increased from €33.4 to almost €93.3 million. In the alternative scenario with a higher uptake of IND/GLY, spending on this combination increased from €54.0 to €123.7 million over the 3-year time-horizon, but total expenditure decreased from €564.5 million in the first year to €563.7 million in the third year (Table 6).
Budget impact: base case. a
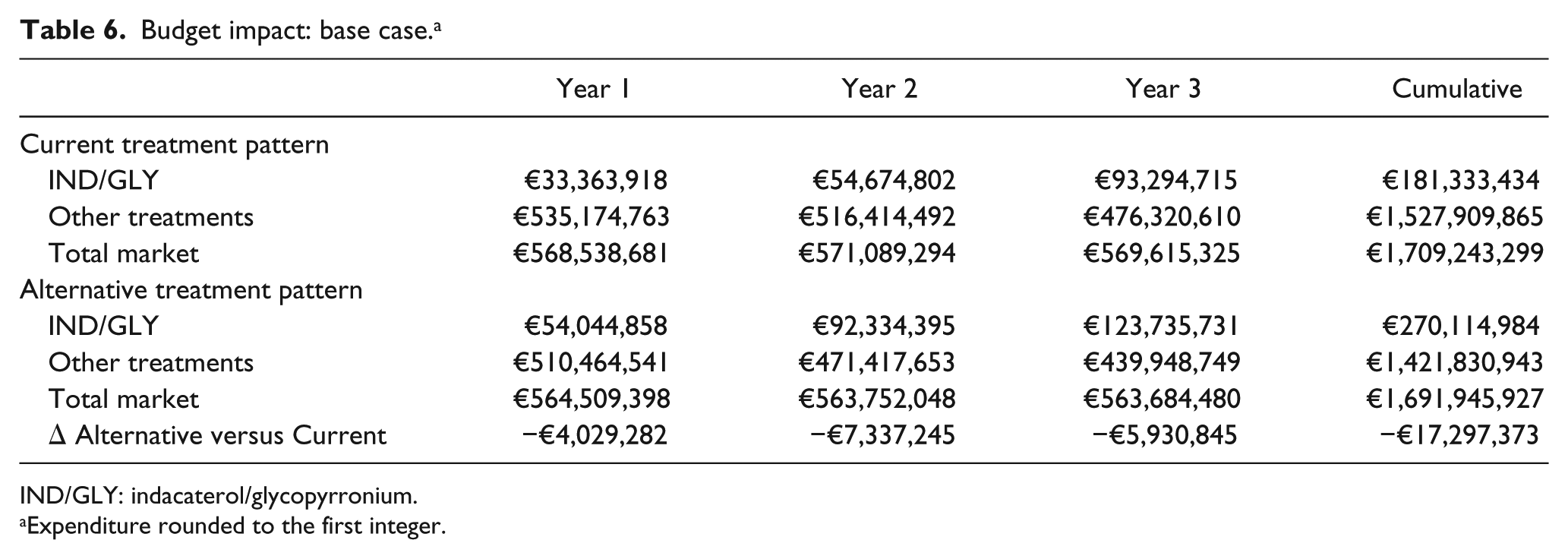
IND/GLY: indacaterol/glycopyrronium.
Expenditure rounded to the first integer.
To calculate the budget impact of increasing the uptake of IND/GLY, we compared the current and alternative treatment patterns over the 3-year time-horizon of the model. Replacing SFC with IND/GLY provided savings of almost €17.3 million (of which €9.8 million of severe exacerbations and €10.9 million of total ones) over 3 years, with total costs over the model time-horizon amounting to €1.71 billion with the current treatment pattern and €1.69 billion in the alternative treatment pattern (see Table 6 for details).
Sensitivity analyses
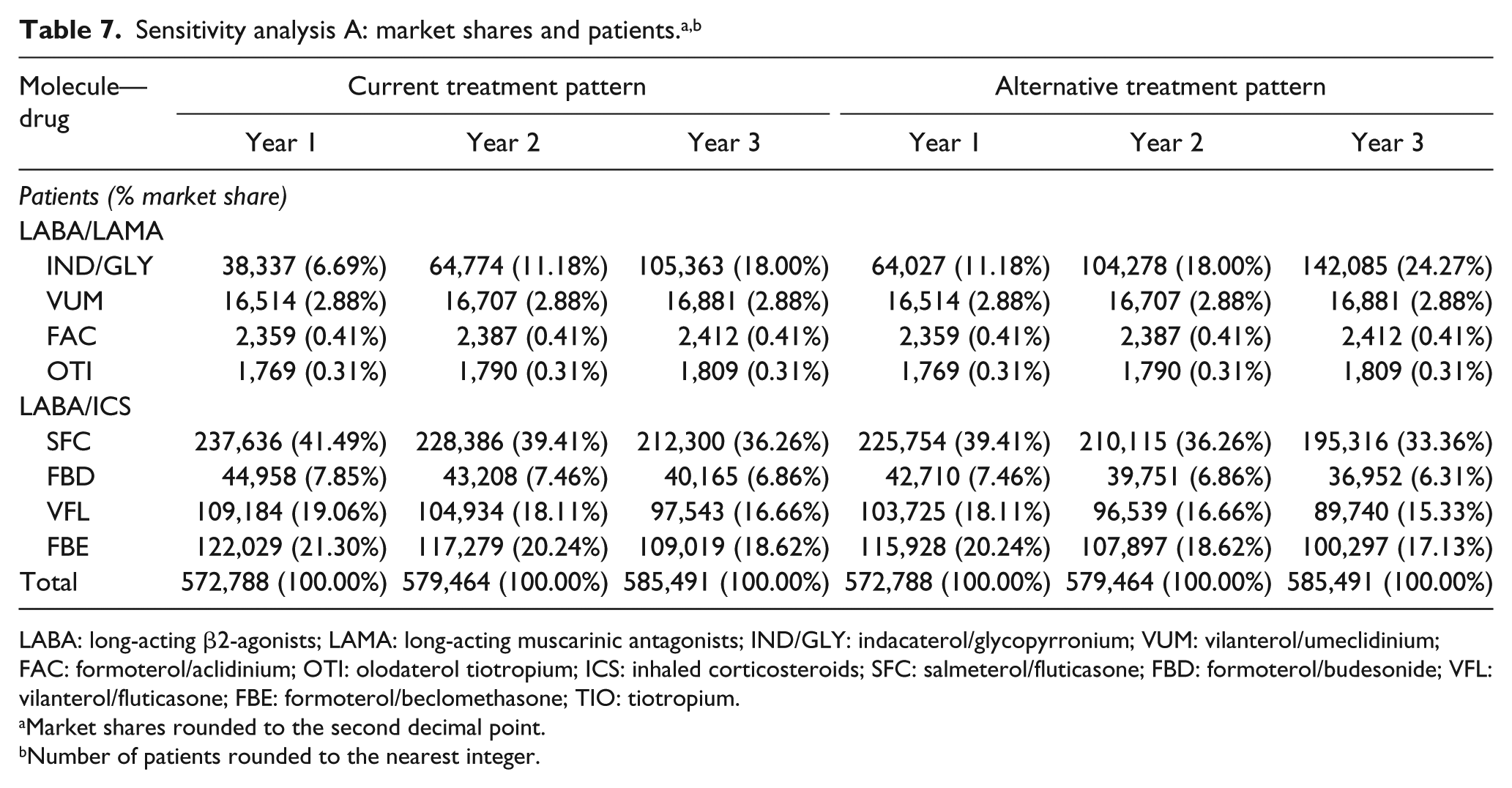
In the sensitivity analysis A, we modeled a switch from all LABA/ICS combinations to IND/GLY, equal to 5% in the first year and 8% in the second and third years (Table 7). In this scenario, due to differences in uptake and a lower cost of other LABA/ICS combinations compared with SFC, the total expenditure was higher, while savings associated with increasing IND/GLY usage were lower than in the base case. Total savings over the whole time-horizon were estimated at €13.9 million; detailed results are reported in Table 8.
LABA: long-acting β2-agonists; LAMA: long-acting muscarinic antagonists; IND/GLY: indacaterol/glycopyrronium; VUM: vilanterol/umeclidinium; FAC: formoterol/aclidinium; OTI: olodaterol tiotropium; ICS: inhaled corticosteroids; SFC: salmeterol/fluticasone; FBD: formoterol/budesonide; VFL: vilanterol/fluticasone; FBE: formoterol/beclomethasone; TIO: tiotropium.
Market shares rounded to the second decimal point.
Number of patients rounded to the nearest integer.
Sensitivity analyses: budget impact. a
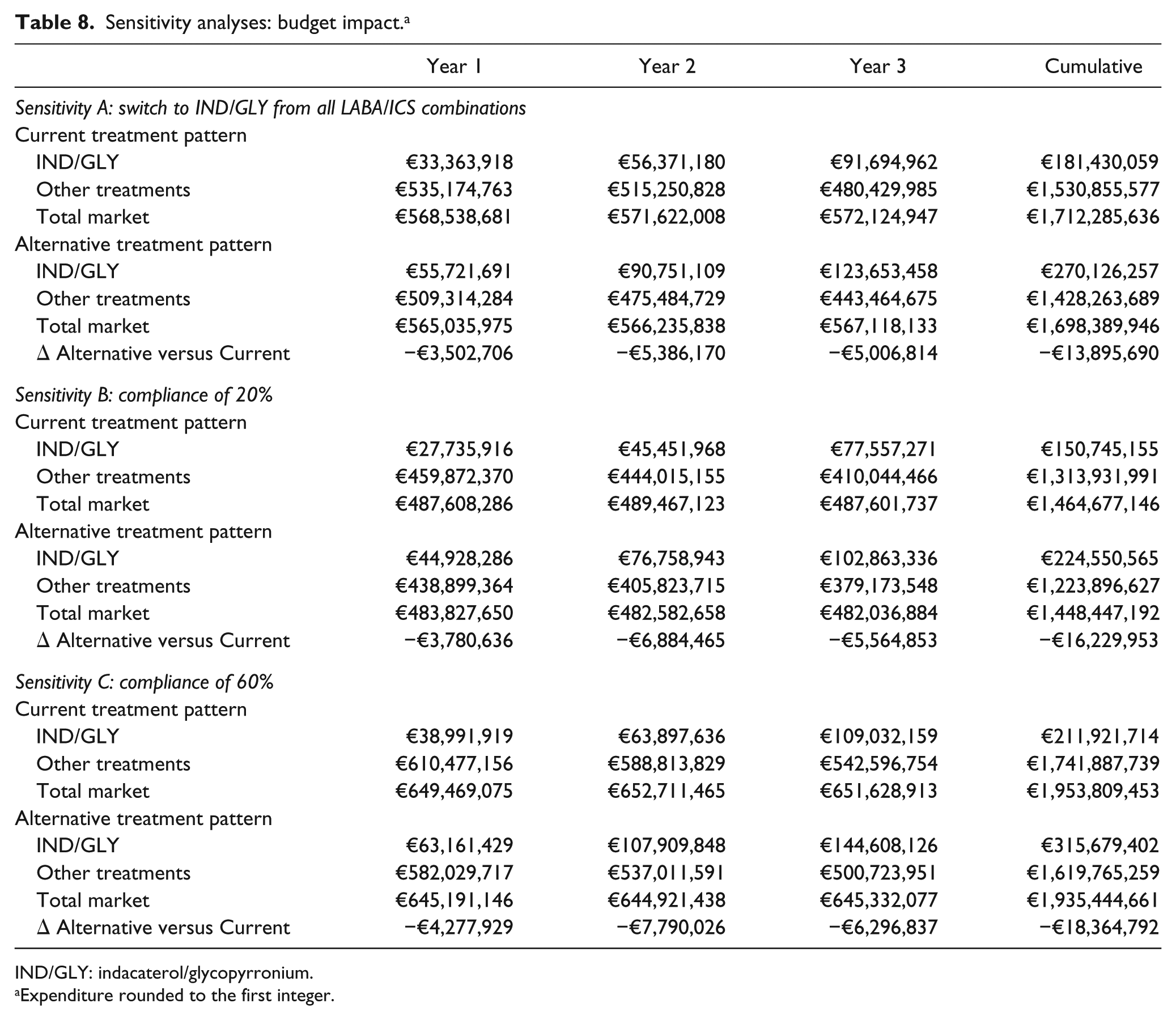
IND/GLY: indacaterol/glycopyrronium.
Expenditure rounded to the first integer.
In the second sensitivity analysis, we investigated two variants (Table 8). In variant B, we assumed a compliance rate of 20%. The yearly cost per patient treated was €723.47 (of which €146.80 were drug cost) for the LABA/LAMA group, €882.57 (drug costs: €157.27) for SFC, €869.57 (drug costs: €144.27) for FBD, €845.78 (drug costs: €120.47) for VFL, and €850.35 (drug costs: €125.05) for FBE. The model estimated total savings of almost €16.2 million over 3 years (Table 8), €1.1 million less than in the base case.
In variant C (Table 8), we assumed a compliance rate of 60%. The yearly cost per patient treated amounted to €1,017.08 (drug costs: €440.41 were drug cost) for the LABA/LAMA group, €1,197.10 (drug costs: €471.80) for SFC, €1,158.12 (drug costs: €432.82) for FBD, €1,086.72 (drug costs: €361.42) for VFL, and €1,100.45 (drug costs: €375.15) for FBE. This scenario resulted in total savings of almost €18.4 million over 3 years, providing higher cost savings than observed in the base case.
Discussion
Our study demonstrated that IND/GLY is a cost-saving option for the Italian NHS. SFC proved to be the most expensive therapy, being associated with the highest drug and total costs. Treatment with IND/GLY over three years was cost-saving compared with LABA/ICS thanks to fewer exacerbations and SAEs (especially pneumonias) being observed with this regimen. Compared with the current treatment pattern, the alternative treatment pattern provided total cost savings ranging between €16.2 and €18.4 million, with the costs of managing COPD exacerbations being the main cost drivers.
Previous studies have shown that COPD has a very relevant impact on healthcare resource consumption and related expenditures, which tend to increase sharply in the most severe cases.15,36,37 In particular, avoiding exacerbations is important both from the clinical and economic point of view, since they are not only associated with accelerated worsening of respiratory function but also the main cause of hospital admissions and resource consumption in Italy, accounting for 50% of the total of direct costs.10,13,36 IND/GLY is more effective than SFC in preventing COPD exacerbations in patients who experienced them during the previous year, as seen in the FLAME study. 25 These data were used in the present BIM to investigate how the reduced risk of exacerbations with IND/GLY could translate into economic benefits over other pharmacological treatments.
The main limitation of the present analysis is the fact it did not consider indirect costs. As COPD is a chronic disease, its impact on work productivity is likely to be substantial so that investigating indirect costs could be of interest for future studies. Another limitation was the assumption that the relative risk of exacerbations was the same for SFC and all other LABA/ICS combinations investigated, as well as for IND/GLY and the other LABA/LAMA combinations. However, the main objective was to compare IND/GLY to SFC, based on the direct clinical comparison reported in the FLAME study. 25 Finally, we used data from randomized clinical trials, which—due to a relatively narrow patient population and diligent care adherent to the trial protocol—may underestimate the number of adverse events and exacerbations in comparison with routine clinical practice. Consequently, our study shows lower average annual per-patient cost than previously published similar reports.10,13,37 However, the clinical evidence reported in the FLAME study can be considered robust, making it a valuable source of clinical data for the model.
Conclusion
Treating patients with moderate-to-very severe COPD with the dual bronchodilator combination of IND/GLY rather than SFC (a LABA/ICS combination) exemplifies a fundamental change in COPD treatment, moving away from the use of ICS. In the present model, this shift in treatment approach proved to be both clinically and economically favorable, reducing the number of COPD exacerbations and SAEs, and providing cost savings of almost €17.3 million over 3 years. Especially, the cost savings related only to severe exacerbations were €9.8 and €10.9 million considering the total one over 3-year time-horizon.
Footnotes
Declaration of conflicting interests
A.A. is an employee of Creativ Ceutical which received a grant from Novartis Farma S.p.A. for conducting the study and outside the submitted work. D.R. and C.P. are employees of Novartis Farma S.p.A., Italy.
Funding
This research was made possible by an unrestricted educational grant from Novartis Farma S.p.A., Italy.