Abstract
Objective:
To determine the effects of adding a quaternary ammonium methacryloxy silicate (K18) and K18-functionalized filler (K18-Filler) on the material and antimicrobial properties of a hard denture reline material.
Materials and methods:
30% K18 in methyl methacrylate (K18-MMA; 0–20 wt% of reliner) and K18-Filler (0–30 wt% of reliner) were incorporated into KoolinerTM hard denture reliner. KoolinerTM served as the control. The cure (Shore A hardness), hydrophilicity (contact angles), mechanical (3-point bend test), water sorption, and antimicrobial properties against Streptococcus mutans, S. sanguinis, and Candida albicans were determined.
Results:
Most K18 groups cured well and had comparable Shore A hardness values (range ~52 to 70 DHN) to that of controls (67.2 ± 1.8 DHN; Bonferroni corrected p > 0.0003). Even the softest group had hardness values within the range of commercial products. Half of the K18 groups had comparable contact angles to that of controls (range ~75° to 80° vs 83.41° ± 2.66°; Bonferroni corrected p > 0.0003), and most were within the range of commercial liners. K18-MMA and K18-Filler increased modulus but decreased ultimate transverse strength (UTS). All experimental groups had comparable or higher moduli than controls (range ~966 to 2069 MPa vs 1340 ± 119 MPa; Bonferroni corrected p < 0.0003), but only half of the experimental groups had comparable UTS to that of controls (range ~41 to 49 MPa vs 55.8 ± 1.5 MPa; Bonferroni corrected p > 0.0003). The 15% and 20% K18-MMA with 30% K18-Filler groups had significant antimicrobial activity against all three microbes (p < 0.05). However, the 15% and 20% K18-MMA with 30% K18-Filler groups had significantly higher water sorption at early time points (p < 0.05). After 8 weeks, they were comparable to each other (p > 0.05).
Conclusions:
K18-MMA and K18-Filler are promising antimicrobial additives that produce hard denture liners with material properties within the range of commercial products and significant antimicrobial properties against S. mutans, S. sanguinis, and C. albicans. Further development is needed to reduce water sorption.
Introduction
The U.S. Surgeon General’s report on oral health in 2000 reported that 32.2% of denture wearers suffer from Candida-associated denture stomatitis, characterized by edema and chronic erythema of the mucosa in contact with the denture.1,2 Furthermore, Candida albicans on the surface of denture acrylics forms biofilms that can harbor microorganisms associated with other systemic diseases and negatively affect systemic health. For example, elderly patients, who did not remove their dentures at night, had a 41% higher risk for pneumonia than those who did,3,4 and Felton 5 found that denture stomatitis can lead to various comorbid conditions, such as malnutrition, obesity, cardiovascular disease, rheumatoid arthritis, pulmonary diseases (including chronic obstructive pulmonary disease), cancer, and even mortality.
The association of stomatitis with denture material is due to the fact that conventional dentures are made of polymethyl methacrylate (PMMA), which lacks an ionic charge on the surface. This can inhibit the selective adsorption of salivary antimicrobials and cationic peptides that adsorb onto the tooth surface by electrostatic interaction and provide a protective pellicle coat. Thus, PMMA minimizes the adsorption of defense molecules on denture surfaces. In addition, attractive London-van der Waals forces facilitate the adherence of C. albicans and other microorganisms that form structured biofilms and lead to the development of the diseases mentioned above.6,7
Prevention of denture stomatitis is done via brushing of the denture, chemical disinfection, and/or the use of antimicrobial mouthwash. However, brushing or using an abrasive dentifrice introduces mechanical damage to the denture, brushing and using antimicrobial mouthwash have been shown to often be ineffective due to poor patient compliance, and chemical disinfection results in odd tastes, odors, color changes, and mechanical damage. 8
Treatments for denture stomatitis include soaking the denture in a chemical disinfectant and the use of antifungal ointments and lozenges. However, these treatments can have side effects. Many of these chemical disinfectants have persulfates that can cause tissue damage, rashes, hives, gum tenderness, breathing problems, and low blood pressure, and antifungal ointments and lozenges can cause nausea, vomiting, mild itching, and an unpleasant sensation in the mouth.9,10
Another treatment for denture stomatitis is the use of a hard denture reliner. Hard denture reliners are typically made from a heat- or chemically-cured acrylic resin, like PMMA and isobutyl methacrylate (IBMA). They are used to reline the surface of the denture to improve the fit for ill-fitting dentures, and to create a new, clean surface after an occurrence of denture stomatitis or inflammatory papillary epithelial hyperplasia. This prevents the need to create new dentures for the patients and reduces time and cost. They are durable and provide a stable and secure fit for the denture. They also help to maintain the vertical dimension of the face, which is crucial for proper facial aesthetics and functional harmony. 11 However, because they are made with PMMA and IBMA, they have the same propensity for denture stomatitis as denture material. Furthermore, as lining materials age, plasticizers can leach out and lead to a rougher, more porous, and harder surface that can further promote the growth of C. albicans. 3
Thus, these preventive interventions and treatments may not be adequate to control the development of denture stomatitis or other systemic diseases, and there is a need for an effective antimicrobial hard denture reline material to prevent or treat denture stomatitis.
Quaternary Ammonium Compounds (QAC) exhibit antimicrobial properties due to their positively charged nature, which enables them to interact with the negatively charged cell membrane of bacteria, ultimately disrupting its integrity. QACs have been used as antimicrobial agents in antiseptics and disinfectants dating back to the 1930s. 12 More recently, biocompatible quaternary ammonium methacryloxy silicates (QAMS), a type of QAC with a long lipophilic -C18H37 alkyl chain and acrylic groups have been developed. This unique structure enables deeper penetration into bacterial cell membranes leading to cell lysis. 13 K18 is a QAMS that can be used both as a coupling agent on glass filler (K18-Filler) through its silane functionality in its core, and as a monomer with its three acrylic functional groups that can react with acrylic groups in methacrylic dental resins to provide antimicrobial activity. A potential major advantage of using K18 would be that it significantly reduces leaching out of the K18 (depending on the degree of cure) to provide long-term antimicrobial property. This is unlike antimicrobial coatings that can be removed with surface wear or the releasing of antimicrobial agents. Any wear of materials containing K18 would result in the exposure of new surfaces containing K18 and persistently reduce biofilm formation. As such, K18 has been shown to have significant antimicrobial effects against both Streptococcus mutans, S. sanguinis, Lactobacillus acidophilus, and C. albicans.13,14 The long-term antimicrobial activity of a K18-containing hard denture reline material could help prevent and treat denture stomatitis.
Thus, the objective of this work was to determine the effects of adding K18 and K18-Filler on the cure, hydrophilicity, mechanical, water sorption, and antimicrobial properties of a hard denture reline material.
Material and methods
Fabrication of K18-containing denture liner
Different concentrations of 30% K18 QAMS in methyl methacrylate (K18-MMA; 0, 10, 15, and 20 wt% of total reline material substituting the liquid portion) and K18-functionalized barium glass filler (K18-Filler, 0, 10, 20, and 30 wt% of total reline material substituting the powder portion) were added to KoolinerTM hard denture reline material (GC America), which is supplied as a powder (poly(methylmethacrylate)) and liquid (isobutylmethacrylate) to form 16 groups. 2 The desired amount of K18-MMA was first mixed into the liquid portion of the reline material, and the desired amount of K18-Filler was mixed into the powder portion of the reline material by hand. Then the liquid and powder were mixed together by hand following manufacturer’s instructions. Proper mixing and homogeneity were confirmed visually to make sure that the mixed material appeared uniform in color and texture, without visible clumps or aggregates. Unmodified KoolinerTM (0 wt% K18-MMA and 0 wt% K18-Filler) was used as the control group. The liquid and powder were combined following manufacturer’s instructions and cured. The cure (Shore A durometer hardness), hydrophilicity (contact angle measurements), mechanical (three-point bend test), water sorption, and antimicrobial properties were determined.
Degree of cure (Shore A durometer hardness)
10 mm diameter × 2 mm thick disc specimens (n = 5) were used for measuring Shore A durometer hardness and contact angle. For hardness, three measurements were made on each specimen with a Shore A durometer (Shore Durometer Type A) as a quick, indirect measure of the degree of cure following ASTM D2240 guidelines.
Surface hydrophilicity (contact angle measurements)
Three 50 µL drops of deionized (DI) water were placed on each specimen (n = 5) and contact angles were measured after 15 s using a VCA 1000 video contact angle instrument (AST Products, Billerica, MA, USA). Measurements were made after 24 h of storage in 37°C deionized (DI) water as described previously and following ASTM 5946 guidelines. 15
Mechanical properties (three-point bend test)
2 mm × 2 mm × 25 mm bar specimens (n = 15) were mounted on an Instron/MTS 1125 ReNew universal mechanical test instrument (Instron Group, Norwood, MA, USA) and loaded using the three-point bending mode at 1 mm/min crosshead speed until material fracture following ISO 10477 guidelines. 15 Modulus and ultimate transverse strength were determined.
In vitro water sorption
Based on the combination of properties and the need to maximize K18 loading for adequate antimicrobial activity, KoolinerTM control, the 15% K18-MMA + 30% K18-Filler, and 20% K18-MMA + 30% K18-Filler groups were chosen for the rest of the experiments. For water sorption, 50 mm × 50 mm × 1 mm specimens were used, and a separate set of samples was used for each time point (n = 10). Water sorption was determined in DI water after 24 h, 4 weeks, and 8 weeks. Specimens were weighed before immersion in DI water. After 24 h in DI water, the specimens were blot-dried and weighed. The amount of water sorption was calculated by using the difference between the initial weight and the blot-dried weight in µg/mm3 following ISO 10139-2 guidelines.
In vitro antimicrobial activity
Antimicrobial activity against S. mutans, S. sanguinis, and C. albicans was determined using 10 mm diameter × 2 mm thick disc specimens (n = 5) as before. 14 S. mutans and S. sanguinis were cultured overnight, and then the bacteria density was adjusted to 0.2 at OD620 nm. Next, a 1:1000 dilution of the inoculum was used as the initial bacterial suspension, and 0.5 mL of that bacterial suspension was added to tubes with the resin specimens. Tubes were incubated in a Coy anaerobic chamber (5% CO2, 10% H2, 85% N2) at 30°C for 24 h and then 37°C for another 24 h under anaerobic conditions. To determine antimicrobial properties, the inoculation cultures were directly diluted in Trypticase soy broth in seven 10-fold (1:10−1) serial dilutions to 1:10−7- for all samples. Aliquots (20 µL) of each dilution were then plated using Trypticase soy agar plates. The colony forming units were counted to determine the growth inhibitory effect of the K18 containing hard denture reline materials.
For C albicans, a similar method was used, except the samples were inoculated at 30°C for 24 h and transferred in agar plates at room temperature for counting colonies.
Statistical analysis
Normally distributed and homoscedastic data was analyzed for statistical significance using analysis of variance (ANOVA) with Newman-Keuls post hoc test (Statistica, StatSoft) to determine significant differences between groups at a p < 0.05 level. For non-normally distributed or non-homoscedastic data, non-parametric analysis using Kruskal–Wallis with the Dunn multiple pairwise comparison and the Bonferroni correction was used to determine significant differences between groups at a p < 0.0018 level.
Results
Degree of cure (Shore A durometer hardness)
Figure 1 shows the Shore A durometer hardnesses of the control and K18-loaded hard denture reline materials. All groups cured within the manufacturer’s prescribed time specifications. Eleven of the 15 experimental groups had comparable hardness as the control group (67.2 ± 1.8 DHN). The four groups that had significantly lower hardness were 15% and 20% K18-MMA + 0% K18-Filler, 0% K18-MMA + 10% K18-Filler, and 20% K18-MMA + 20% K18-Filler groups (52.3 ± 6.2 DHN, 53.5 ± 7.4 DHN, 52.733 ± 8.5 DHN, and 55.2 ± 7.5 DHN, respectively; Bonferroni corrected p < 0.0003). No definite trends were identifiable, but increase in K18-MMA and K18-Filler concentrations tended to increase hardness except when K18-MMA concentration reached 20%. A combination of no K18-MMA but with K18-Filler also tended to produce specimens with lower surface hardnesses. This is consistent with our difficulty in curing liners with K18-MMA concentrations higher than 20 wt%. However, moderate amounts of K18-MMA tended to cure comparably to the control liners.
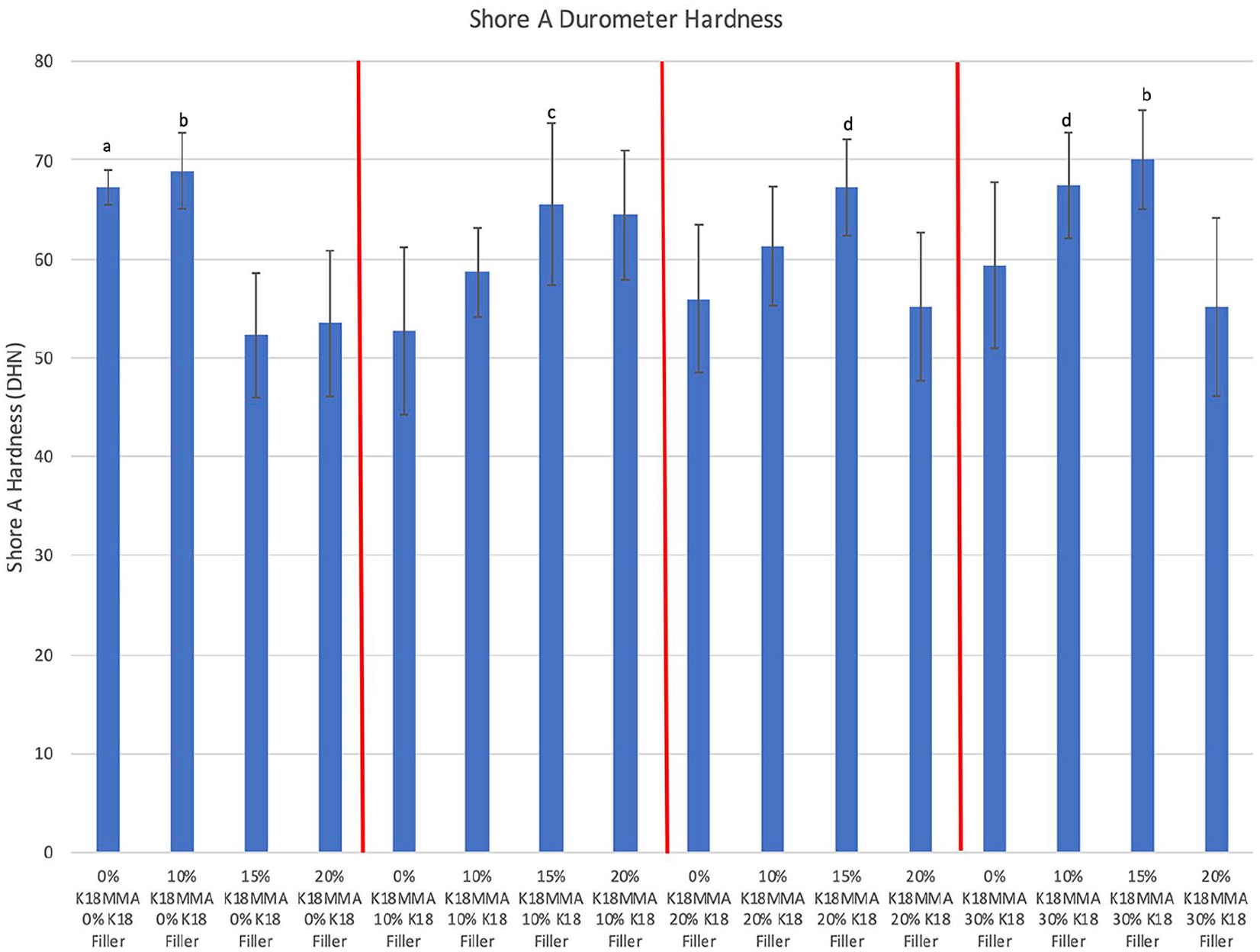
Shore A durometer hardness of control and K18-loaded Hard Denture Reline Material. Groups denoted with the same letter are statistically equivalent.
Surface hydrophilicity (contact angle measurements)
Figure 2 shows contact angles of the control and K18-loaded hard denture reline materials. Five of the 15 experimental groups had significantly lower contact angles than controls (range of ~69° to ~74° vs 83.4° ± 2.7°, respectively; Bonferroni corrected p < 0.0003). There were no definite trends, but groups without K18-MMA and groups with increased K18-Filler loadings tended to be more hydrophobic. As such, the 0% K18-MMA + 20% K18-Filler and 0% and 20% K18-MMA + 30% K18-Filler groups (signified with “b”) are significantly more hydrophobic than 10% K18-MMA + 0% K18-Filler and 10%–20% K18-MMA + 10% K18-Filler groups (Bonferroni corrected p < 0.0003).
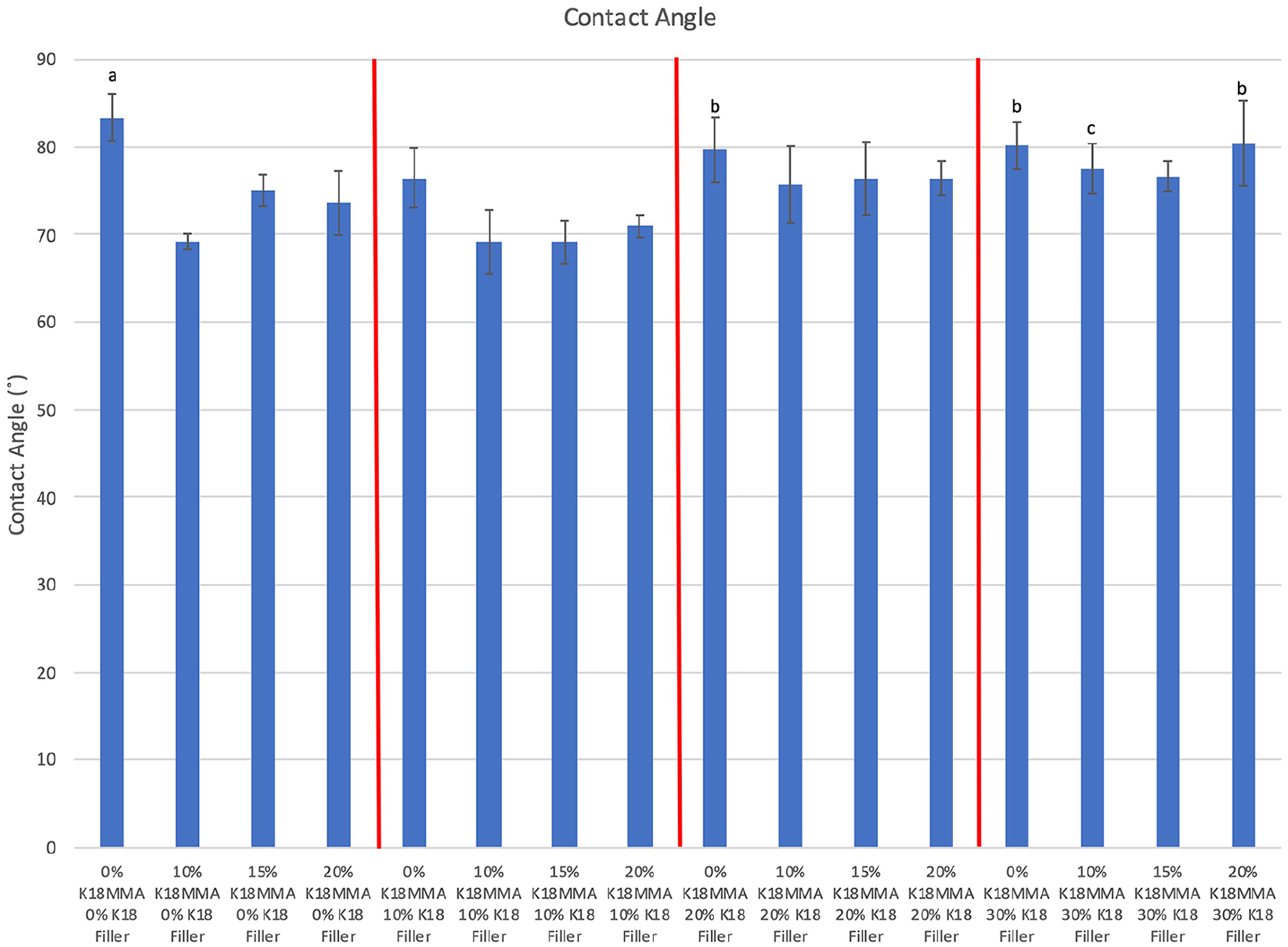
Contact angles of control and K18-loaded Hard Denture Reline Material. Groups denoted with the same letter are statistically equivalent.
Mechanical properties (three-point bend test)
Figure 3 shows the moduli of the control and K18-loaded hard denture reline materials. All experimental groups had comparable or higher moduli than controls (1340 ± 119 MPa). The two groups with significantly higher moduli were the 10% and 20% K18-MMA + 30% K18-Filler groups (1814 ± 220 MPa and 2069 ± 131 MPa, respectively; Bonferroni corrected p < 0.0003). There were no definite trends, but increase in K18-Filler loading tended to increase modulus.
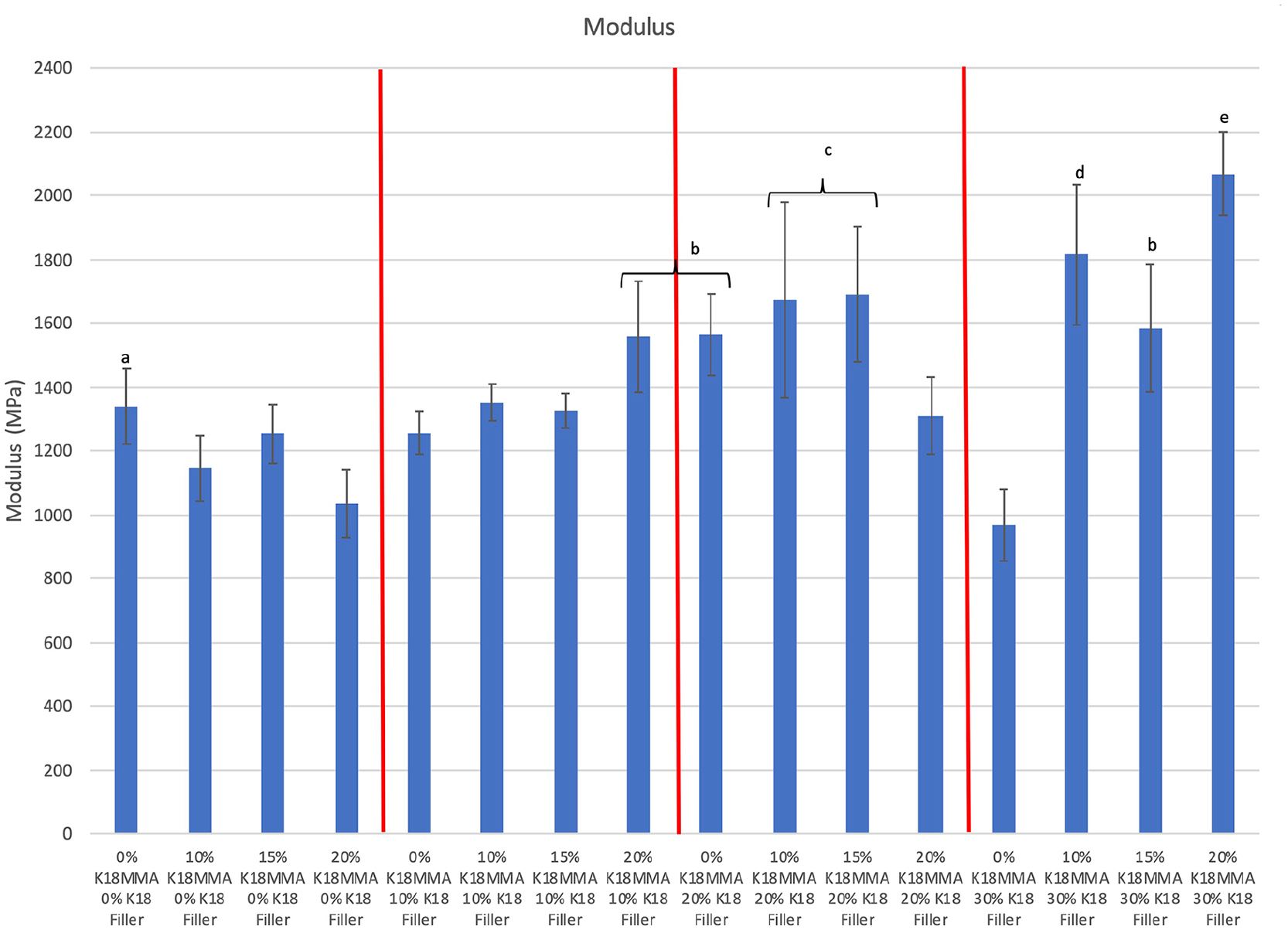
Moduli of control and K18-loaded Hard Denture Reline Material. Groups denoted with the same letter are statistically equivalent.
Figure 4 shows the ultimate transverse strengths (UTS) of the control and K18-loaded hard denture reline materials. Seven out of 15 experimental groups had comparable UTSs as controls (55.8 ± 1.5 MPa). There were no definite trends, but increases in K18-MMA concentration tend to decrease UTS, but increases in K18-Filler concentration tend to counter that and increase UTS. This may be due to the difficulty in curing at high K18-MMA and K18-Filler concentrations. Nonetheless, two groups with high K18-MMA and K18-Filler loadings had comparable UTS to controls, namely the 15% K18-MMA + 20% and 30% K18-Filler groups (UTS = 41.0 ± 1.6 MPa and 46.5 ± 2.8 MPa, respectively; Bonferroni corrected p > 0.0003).
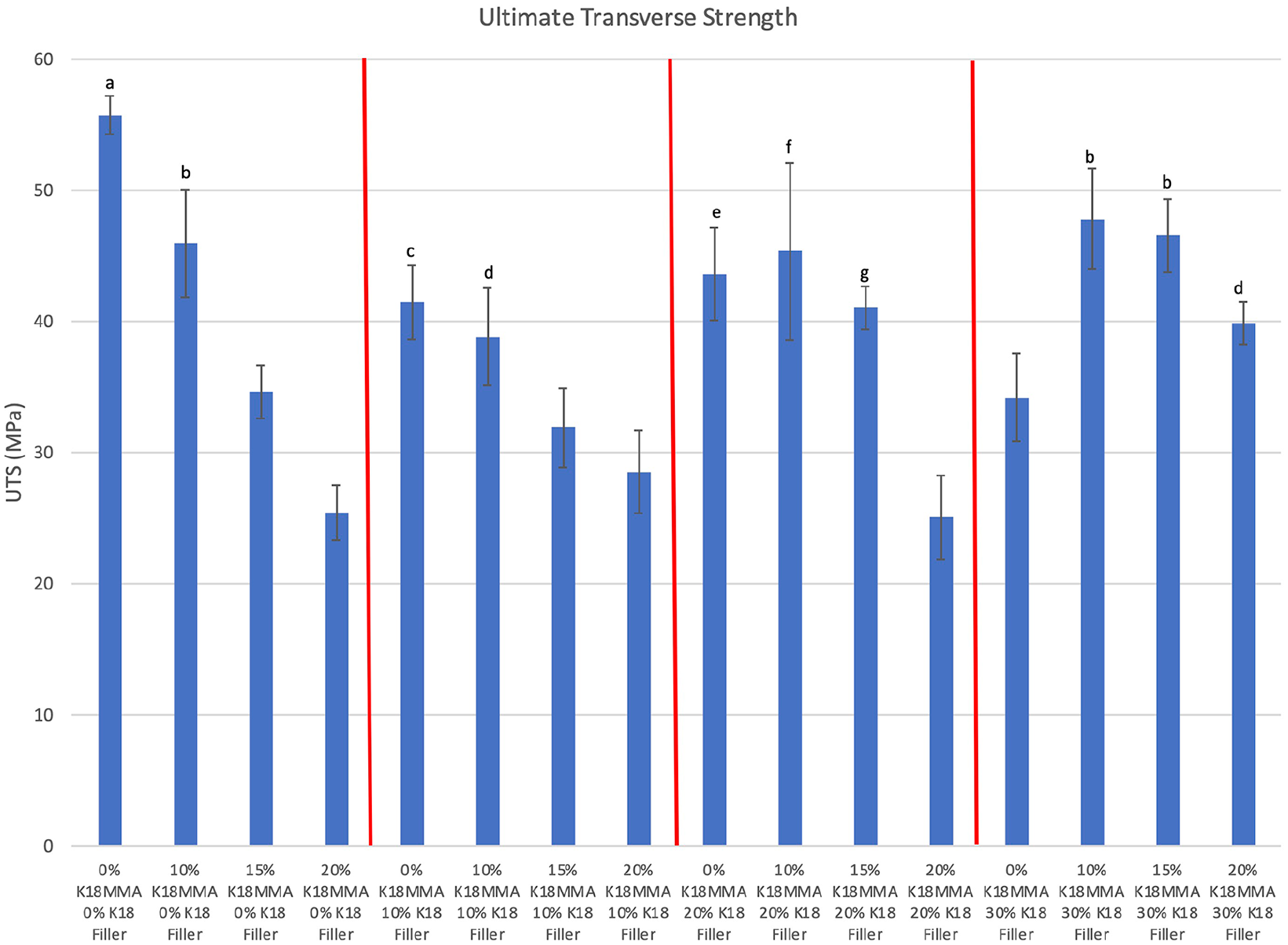
Ultimate transverse strengths of control and K18-loaded Hard Denture Reline Material. Groups denoted with the same letter are statistically equivalent.
In vitro water sorption
Based on the combination of properties and the need to maximize K18-MMA loading for antimicrobial activity, the control and the 15% and 20% K18-MMA + 30% K18-Filler groups were chosen for the rest of the experiments. The 15% K18-MMA + 30% K18-Filler group had comparable Shore A hardness, contact angle, modulus and UTS as controls. While the 20% K18-MMA + 30% K18-Filler group was weaker than the control group, this group was chosen because it had comparable Shore A hardness and contact angle as and higher modulus than the control group, and the addition of this group would elucidate any dose-dependent effect on antimicrobial properties.
Figure 5 shows percent water sorption after 24 h, 4 weeks, and 8 weeks for the control and the 15% and 20% K18-MMA + 30% K18-Filler groups. At 24 h, the control group absorbed significantly less water than the other two groups (0.81% ± 0.19% vs 2.70% ± 0.38% and 3.39% ± 1.40%, respectively; Bonferroni corrected p < 0.01667). The amounts of water sorption for the control and 15% and 20% K18-MMA + 30% K18-Filler groups were 10.3 ± 0.9 µg/mm3, 32.8 ± 3.2 µg/mm3, and 40.1 ± 16.4 µg/mm3, respectively. With increased time, the amount of water absorbed increased and the differences between groups decreased. At 4 weeks, the control group absorbed significantly less water than only the 15% K18-MMA + 30% K18-Filler group (3.16% ± 0.46% vs 5.43% ± 1.70%, respectively; Bonferroni corrected p < 0.01667) There was no difference between the groups at 8 weeks.
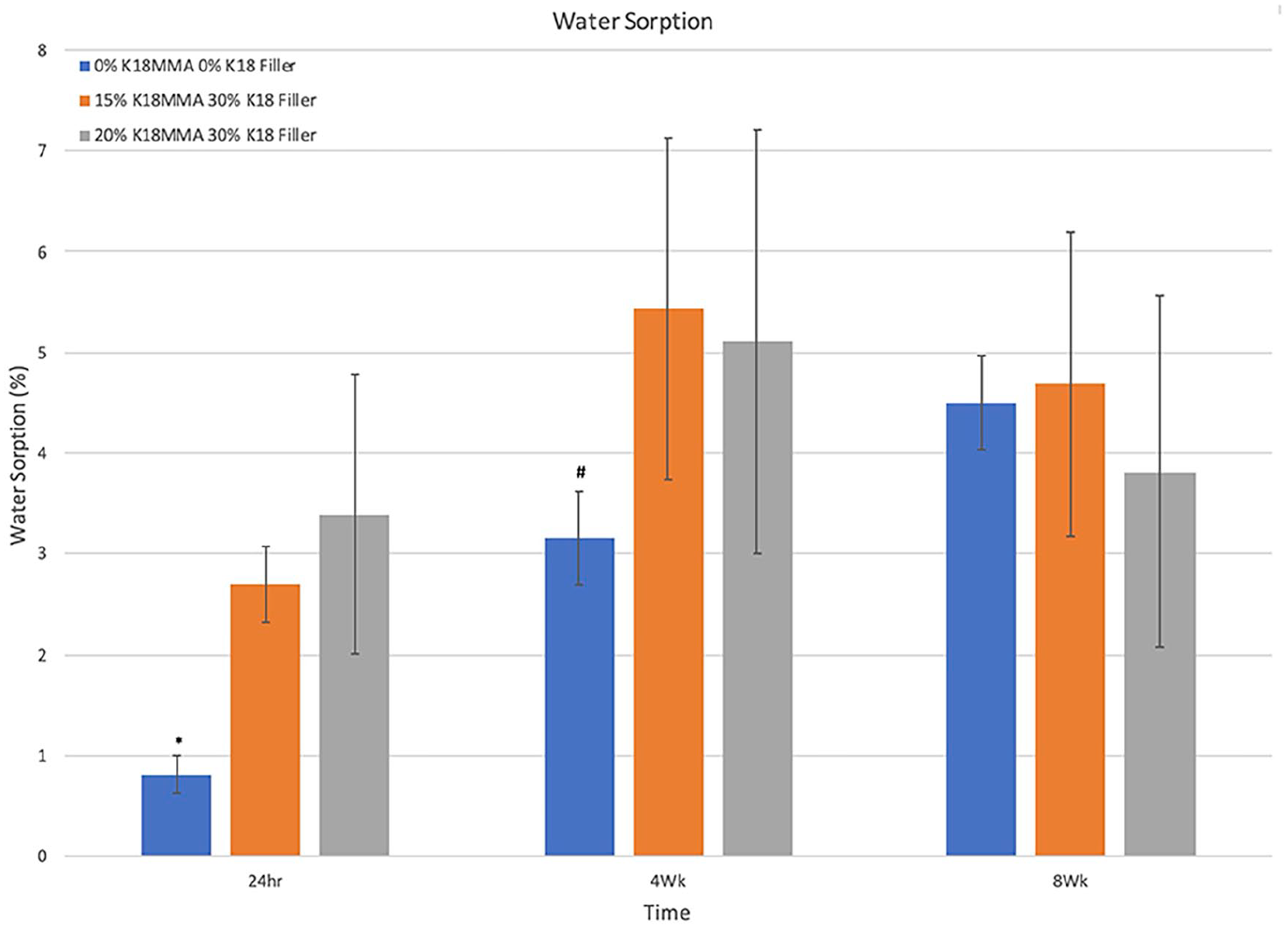
Percent water sorption of control and 15% and 20% K18-MMA with 30% K18-Filler-loaded Hard Denture Reline Material.
In vitro antimicrobial activity
Figure 6 shows the antimicrobial activity of the control and the 15% and 20% K18-MMA + 30% K18-Filler-loaded hard denture reline material against S. mutans. Both experimental groups had significantly more antimicrobial activity than the controls (181,6400 ± 196,267 CFU), and increase in K18-MMA concentration from 15% to 20% significantly increased antimicrobial activity (128,1600 ± 244,853 CFU and 749,600 ± 222,254 CFU, respectively) (p < 0.05).
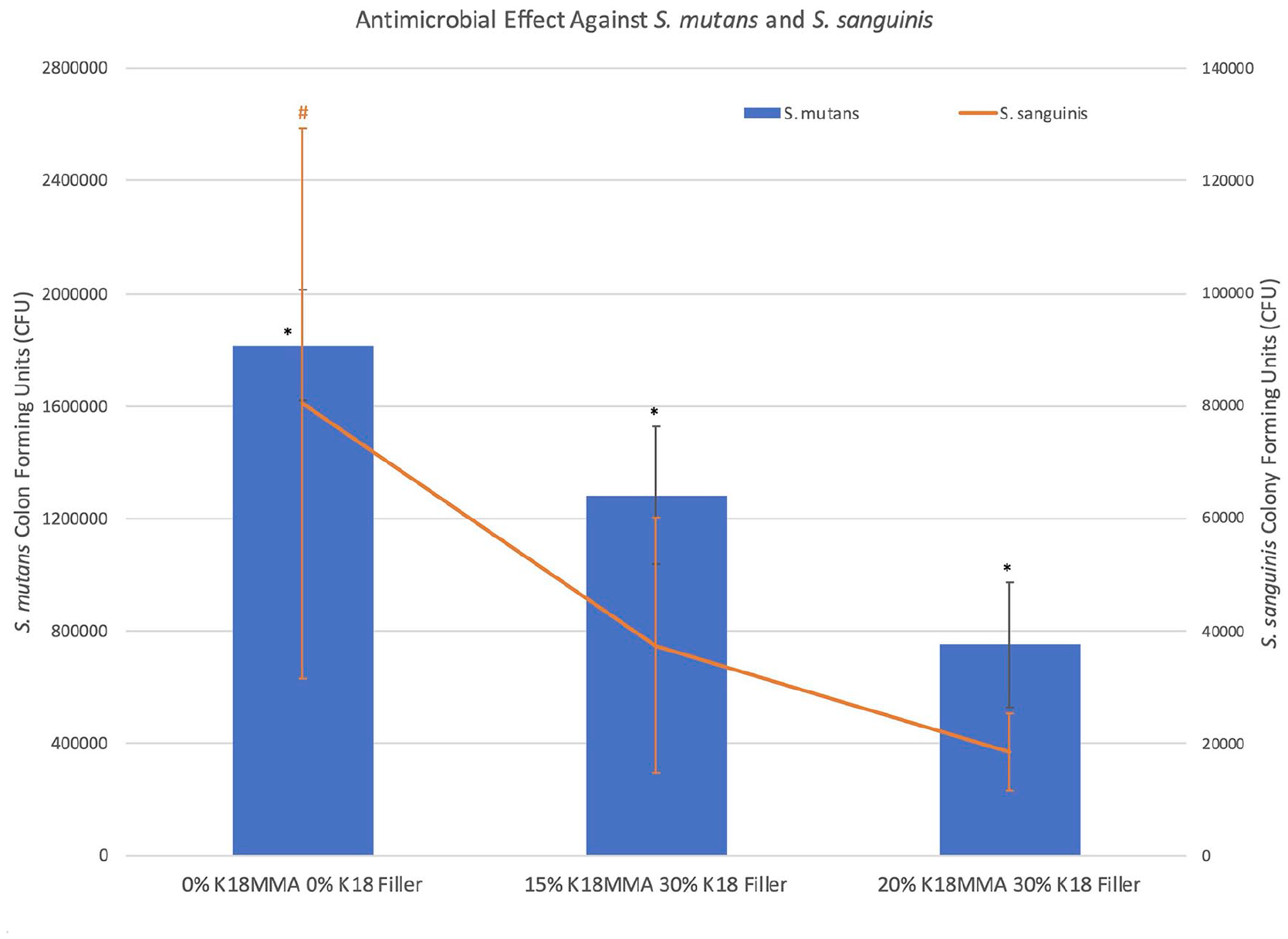
Antimicrobial activity of control and 15% and 20% K18-MMA with 30% K18-Filler-loaded Hard Denture Reline Material against S. mutans and S. sanguinis.
Figure 6 also shows the antimicrobial activity of the control and 15% and 20% K18-MMA with 30% K18-Filler-loaded hard denture reline material against S. sanguinis. Both the 15% and 20% K18-MMA + 30% K18-Filler groups (37,400 ± 22,711 CFU and 18,400 ± 6877 CFU, respectively) had significantly more antimicrobial activity than the controls (80,400 ± 48,932 CFU) (p < 0.05). No dose-dependent response was visible.
Figure 7 shows the antimicrobial activity of the control and 15% and 20% K18-MMA with 30% K18-Filler-loaded hard denture reline material against C. albicans. Both the 15% and 20% K18-MMA + 30% K18-Filler groups (99,200 ± 25,762 CFU and 84,600 ± 19,204 CFU, respectively) had significantly more antimicrobial activity than the controls (374,200 ± 65545 CFU) (p < 0.05). No dose- dependent response was visible. The effect against C. albicans was also much stronger than against the two bacterial strains. There was a 29% and 59% reduction in CFU for the 15% and 20% K18-MMA + 30% K18-Filler groups against S. mutans, respectively, a 53% and 77% reduction against S. sanguinis, respectively, and 73% and 77% reduction against C. albicans, respectively.
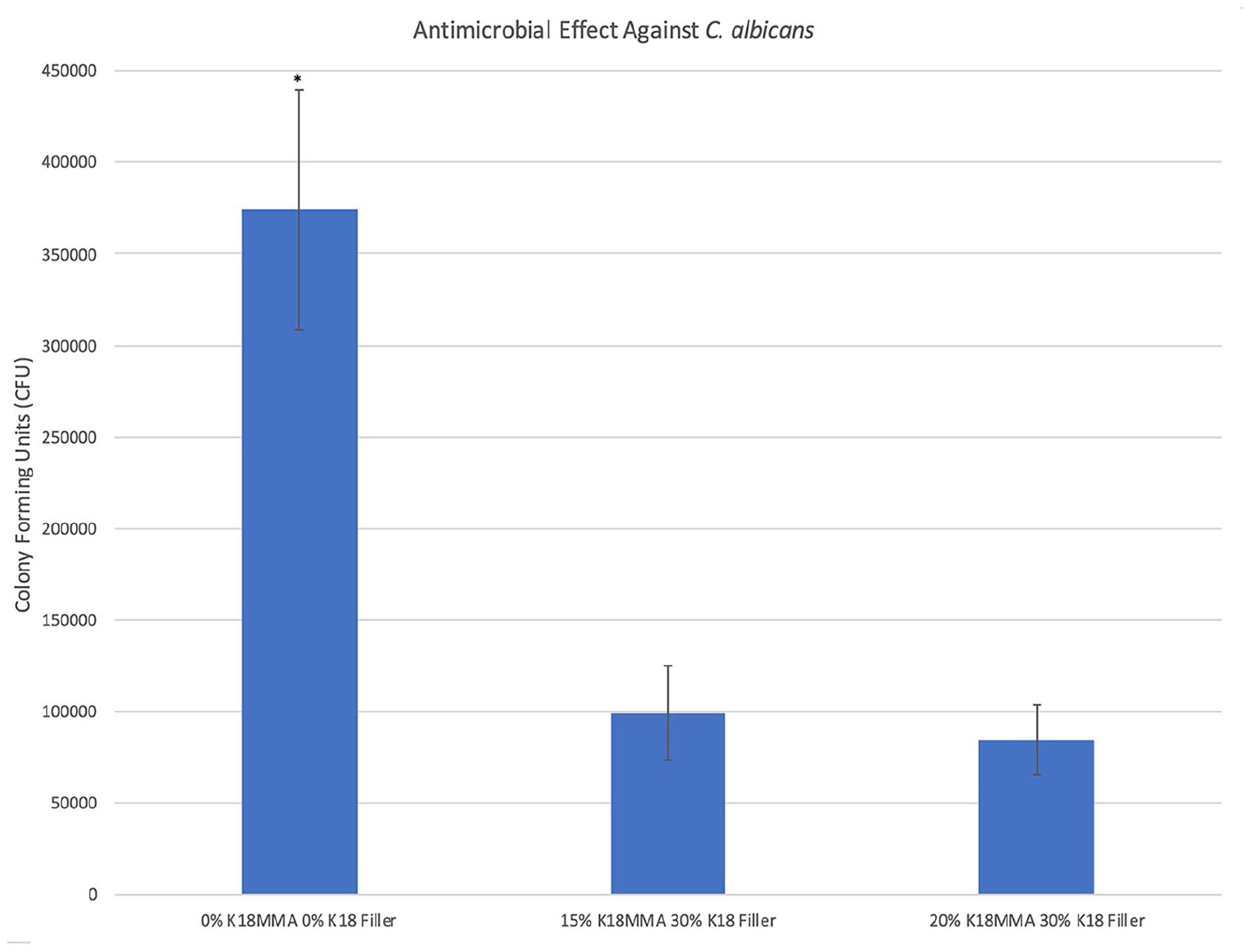
Antimicrobial activity of control and 15% and 20% K18-MMA with 30% K18-Filler-loaded Hard Denture Reline Material against Candida albicans.
Discussion
Current treatments for denture stomatitis involve the use of systemic or local antifungal therapy, or the fabrication of a new prosthesis once microbial balance is achieved. However, antifungal therapies can lead to resistant strains and fabrication of new prosthesis could be very costly to patients. 1 Thus, an antimicrobial hard denture reline material would be a cost-effective, long-term interim treatment approach to managing denture stomatitis.
Quaternary ammonium methacryloxy silicates (QAMS) have a long lipophilic -C18H37 alkyl chain that penetrates bacterial cell membranes and causes cell lysis. 16 K18, a QAMS, is capable of both functionalizing glass filler (K18-Filler) and copolymerizing with dental monomers to provide microbiocidal activity. K18 does not leach out and potentially provides long-term antimicrobial properties. Unlike coatings, surface wear would expose fresh K18, giving continuous microbiocidal activity and preventing denture stomatitis that may cause other systemic diseases. Thus, the use of K18 and K18-Filler in KoolinerTM hard denture reline material was investigated.
K18-MMA concentrations were chosen based on our previous work using K18-MMA in a dental composite system, which showed that at least 15% in conjunction with K18-Filler was needed for significant antimicrobial activity against S. mutans. 14 K18-MMA and K18-Filler concentrations were also limited to 20% and 30%, respectively, due to the difficulty in curing hard denture reline materials with concentrations higher than that. While additional initiators could have been used, the initiator system used in KoolinerTM hard denture reline material is proprietary and their concentrations were not known. All formulations of the K18-containing hard denture reline material cured within the manufacturer’s prescribed time specifications, and most experimental groups cured well and had comparable Shore A durometer hardness values as those of the KoolinerTM controls. Even the softest formulation (15% K18-MMA + 0% K18-Filler, 52.3 ± 6.3 DHN) was harder than the clinically acceptable minimum Shore A hardness level of 49 DHN. 17 KoolinerTM had a Shore A durometer hardness value of 67.2 ± 1.8 DHN, and most commercial chairside hard denture reline materials have Shore A durometer hardness numbers of 40–46 DHN. 18 Thus, the use of K18-MMA and K18-Filler did not adversely affect cure in a clinically significant way.
This is similar to the effects seen in other experimental antimicrobial acrylic materials and hard denture reliners. The generation of silver nanoparticles (AgNP) in orthodontic PMMA and the addition of chlorhexidine (CHX) into hard denture reline materials did not adversely affect surface hardness.19,24 However, the AgNP-containing resins required additional initiator to have comparable hardness as the controls while the experimental groups in this study did not use additional initiators. 19 The cure properties of K18-loaded hard denture liners, as far as hardness is concerned, should be within the range of commercially available products.
The incorporation of K18 made the surfaces of half of the experimental groups more hydrophilic. Despite the lack of clear trends, results indicate that the presence of K18-MMA tends to reduce contact angle since the K18-Filler would not be visible on the surface of the material. Most commercial hard and soft denture reliners have contact angles ranging from 75° to 111°, KoolinerTM control had contact angles of 83.4° ± 2.7°, K18-containing reliners had contact angles between 69° and 80°, and the two groups selected for antimicrobial testing (15% and 20% K18-MMA + 30% K18-Filler) had contact angles of 76.7° ± 1.7° and 80.4° ± 4.9°, respectively. 20 Most (10 of 15) experimental groups, including the two selected for water sorption and antimicrobial testing, had contact angles within the commercial range. This correlates somewhat with CHX-loaded denture reline materials, which tended to have significantly increased surface free energies and decreased contact angles. 24 Regardless, despite the statistically significant drop in the contact angles of the K18-containing liners from the KoolinerTM controls (up to 14°), the groups with the highest K18 loadings that would show the greatest antimicrobial activity are within commercial standards.
However, contact angle results did not correlate with water sorption. The 15% and 20% K18-MMA + 30% K18-Filler groups, which have comparable contact angles as the control group, had significantly greater water sorption than controls at 24 h, and the 15% K18-MMA + 30% K18-Filler group had significantly greater water sorption than controls at 4 weeks. At 8 weeks, there were no differences between groups. It is possible that the non-statistically significant slight increase in the contact angle of the control group was enough to reduce the water sorption rate, however, no such effect was observed between the 15% and 20% K18-MMA groups. Regardless, the 15% K18-MMA + 30% K18-Filler group barely exceeded ISO Standard 1567:1999 requirement for chairside hard denture reline materials to not exceed a water sorption of 32 μg/mm3 at 24 h, while the 20% K18-MMA group significantly exceeded it. 21 Efforts will need to be made to reduce the water sorption to meet ISO specifications.
Interestingly, the K18-MMA and K18-Filler increased reline material modulus but decreased ultimate transverse strength (UTS). All experimental groups had comparable or higher moduli, but only 7 out of 15 experimental groups had comparable UTS to those of KoolinerTM controls. No experimental groups had higher UTS than controls. The effect of K18-Filler loading seems to be stronger than that of the K18-MMA in increasing modulus. K18-Filler also seems to reduce the weakening effect of K18-MMA, with certain formulations in the 20% and 30% K18-Filler-loaded groups showing a synergistic positive effect on mechanical properties. The increase in modulus due to the use of filler is a well-known phenomenon in dental composites, but it should be noted that the total filler/powder loading in the K18-containing groups is the same as the powder loading of the control KoolinerTM specimens. It seems that the higher modulus of the glass filler may be contributing to the increased modulus over that provided by the KoolinerTM PMMA particles. However, the increase in strength usually seen with the use of glass filler was not observed in this work. Regardless, these UTS results are within the range of commercially available products, since the K18 reliners range in UTS from 25 to 48 MPa and most commercial hard denture reline materials fall within 5–27 MPa.22,23
These mechanical testing results are in contrast to AgNP-loaded PMMA, where AgNP loading decreased both modulus and transverse strength, and in line with results from adding CHX to Probase Cold hard denture reline material, which also had significantly reduced flexural strength.19,24 Gong et al. 16 also reported that as little as 2% QAMS decreased both flexural modulus and ultimate strength in polymethyl methacrylate (QAMS-PMMA). The K18-MMA used in this work contains 30% w/w K18 in MMA, and the 20% w/w K18-MMA group would have the equivalent of 6% w/w QAMS in the QAMS-PMMA. 16 Our results do not show a significant decrease in modulus when K18-MMA is added. However, the use of higher concentrations of K18-MMA did significantly reduce UTS and correlated with results from Gong et al. 16 Fortunately, K18-Filler increased modulus and reversed the decrease in UTS, and produced two formulations with high K18-loading and mechanical properties within the range of commercially available products.
Finally, the 15% K18-MMA and 20% K18-MMA + 30% K-18 Filler groups were selected for antimicrobial testing because both had comparable Shore A hardness, contact angle, and moduli as compared to those of KoolinerTM controls, and both had the highest K18 loading. Despite the 20% K18-MMA + 30% K-18 Filler group being weaker than controls, both groups also had higher UTSs than most commercial hard denture reline materials. Both experimental groups had significant antimicrobial activity. Against S. mutans, a dose-dependent relationship could be observed, but for S. sanguinis and C. albicans, the dose-dependent response was not as obvious, probably because the antimicrobial effects of the 15% K-18 MMA + 30% K18-Filler group were more potent against these microbes. The K18 was also more effective against C. albicans than the streptococcus strains.
In comparison, AgNP-containing PMMA had significantly higher antimicrobial effect of >99.9% bacterial inhibition, however, the bacteria tested are different from those tested here, and it can be seen that the strain affects how potent the antimicrobial effect is. Also, since Ag+ ions have to be released, the duration of the antimicrobial effect is unknown, and results show that there is a decrease in the antimicrobial effect over 4 weeks. 19
K18-loaded denture hard reliner antimicrobial results were more consistent with those of CHX-loaded hard denture reliners. CHX reliners showed antimicrobial activity with as little as 2.5% CHX in Kooliner, 5% CHX in UFI Gel Hard and 5% CHX in Probase Cold reliners. However, a dose-dependent effect was not observed, probably because the antimicrobial effects were limited by the diffusion of CHX, and the long-term antimicrobial efficacy is questionable, since it releases the antimicrobial agent. Also, these results cannot be directly correlated with those of K18-reliners because the antimicrobial assays used were different; colony counting assay vs. ring inhibition assay. Regardless, both results show enhanced activity against C. albicans. 24
QAMS-PMMA demonstrated significant antimicrobial activity against S. mutans with as little as 0.4% w/w QAMS and an enhanced activity against C. albicans. 16 The 15% and 20% w/w K18-MMA + K18-Filler groups used in this study also had significant antimicrobial activity against S. mutans and enhanced activity against C. albicans, despite requiring a higher QAMS loading. Therefore, the mechanism of action is expected to be analogous to that of the QAMS-PMMA, with the lipophilic -C18H37 alkyl chain penetrating bacterial cell membranes and leading to cell lysis. 16 Gong et al. 16 reported that while QAMS-PMMA did not prevent bacterial adhesion to pellicle-coated surfaces, notable decreases in live bacteria within the biofilms were observed. Regarding C. albicans, they noted that the enhanced antimicrobial effect appears to stem from biofilms being unable to form on the QAMS-PMMA surfaces causing the C. albicans cells to detach readily. In this work, K18-MMA and K18-Filler-containing hard denture liners demonstrated significant antimicrobial activity, and the mechanism of action is expected to be similar to that of QAMS-PMMA. However, additional research is warranted to ascertain their impacts on biofilm formation and more persistent bacteria.
Lastly, all components utilized in this investigation, including K18, have obtained FDA-cleared. A randomized clinical trial has corroborated the antimicrobial effectiveness and biocompatibility of QAMS-PMMA, with no adverse effects observed on the oral mucosa or overall systemic health of the patient. However, the biocompatibility of these liners containing K18 still requires assessment through various tests such as cytotoxicity, genotoxicity, delayed-type hypersensitivity, oral mucosa irritation, and evaluation of pulp and dentin response.
Conclusions
A novel antimicrobial hard denture reliner with K18 QAMS and K18-Filler has been developed for the prevention and treatment of oral candidiasis. Hard denture liners with K18 QAMS and K18-Filler had surface hardnesses, contact angles, moduli and ultimate transverse strengths within ISO requirements and ranges found among commercial hard reliners and significant antimicrobial properties against S. mutans, S. sanguinis, and C. albicans. However, water sorption increased and exceeded requirements in ISO Standard 1567:1999 for chairside hard denture reline materials, so efforts will need to be made to reduce that.
Footnotes
Contributorship
SB researched literature, developed the research proposal, conducted testing and data analysis, and wrote the manuscript. EF were involved in specimen fabrication, testing, and data analysis. LC conducted antimicrobial testing. KW conceived the study, oversaw data collection and analysis, and provided revisions to the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the FiteBac Foundation.
Guarantor
KW.
