Abstract
Background:
A stable copper tetraamine fluoride (CTF) with low cytotoxicity has been developed for dental use.
Objective:
To investigate the antimicrobial effects of CTF against common microbes associated with dental caries and periodontal disease.
Method:
The minimum inhibitory concentrations (MIC) and minimum bactericidal/fungicidal concentrations (MBC/MFC) were used to evaluate the antimicrobial effects of CTF against eight common bacteria and one fungus associated with dental caries and periodontal disease. These nine microbes included cariogenic pathogens (
Results:
The MIC of CTF against nine microbes ranged from 80 ppm (
Conclusion:
CTF has antimicrobial effects against common oral pathogens and presents a promising antimicrobial agent to aid management of dental caries and periodontal disease.
Introduction
Dental caries is the localized destruction to the hard tissue of teeth caused by acids generated during the fermentation of sugars by microorganisms.
1
The primary pathogenic microorganisms responsible for tooth decay include
Among existing treatments, various antibacterial agents are widely used in dentistry to manage oral pathogens, with silver compounds being particularly common.5 –8 However, the oxidation of ionised silver can lead to the staining of enamel and dentin, resulting in patient dissatisfaction. 9 Additionally, some studies have shown that silver compounds exhibit high toxicity in human cells, underscoring the need for cautious use in dentistry. 10 These concerns highlight the growing need to investigate alternative chemical compounds that offer robust antibacterial properties, minimal toxicity, and no discoloration effects.
Using copper as an alternative to silver could potentially address these concerns associated with silver compounds. A bibliometric analysis indicates increasing interest in copper within medical research.
11
Copper exhibits antibacterial properties against oral bacteria, such as
Ammonia is antibacterial and acts as a stronger field ligand than water. Metal ammine complexes exhibit greater stability and reduced oxidation compared to their aquo counterparts. 15 Fluoride is commonly used for remineralization in caries management and can also provide some antimicrobial ability. 16 It promotes the crystal growth of fluorapatite on partially demineralised sub-surface crystals in carious lesions.
In our earlier research, we developed a novel copper tetraamine fluoride (CTF) using copper fluoride and ammonia solution.
17
Our results indicated that CTF can inhibit the growth of
Methods
Microorganisms
Nine common oral pathogenic strains were selected for this study. They are cariogenic pathogens including
Brain heart infusion (BHI) medium was used for the culture of
Minimum inhibitory concentrations and minimum bactericidal/fungicidal concentrations
The minimum inhibitory concentrations (MICs) and minimum bactericidal/fungicidal concentrations (MBC/MFCs) were used to evaluate the antimicrobial effects of CTF against these common oral pathogens. The broth (100 μL consecutive two-fold dilutions ranging from 5120 ppm to 10 ppm of CTF) was supplemented with a 10 μL bacteria/fungi culture (106 CFU/mL). The 96-well plates were then incubated at 37°C for 24 h. The optical density (OD) value was measured at a wavelength of 660 nm (bacteria) or 520 nm (fungi) both before and after the microorganisms were cultivated for 24 h. The MIC value is defined as the lowest concentration at which the OD values before and after incubation are consistent.
After the MIC determination, 10 μL fluid from each well, which showed the same OD values, was pipetted and seeded on agars, which were then incubated at 37°C for 48 h. The MBC/MFB is determined to be the lowest concentration at which no colonies are formed on the agar plate.
Microorganisms morphology
The transmission electron microscope (TEM) was used to examine the morphology of the microorganisms. The bacteria/fungi (108 CFU/mL) were added with the CTF of MBC/MFC concentration. 18 The bacteria/fungi were harvested after incubating at 37°C for 18 h and subjected to glutaraldehyde treatment. The semi-thin sections of the bacteria/fungi were contained in grids and observed with the TEM.
Results
MIC and MBC/MFC
Table 1 displays the MIC and MBC/MFC of CTF against
The minimum inhibitory concentrations (MICs) and minimum bactericidal/fungicidal concentrations (MBC/MFC) of CTF against common oral pathogens.
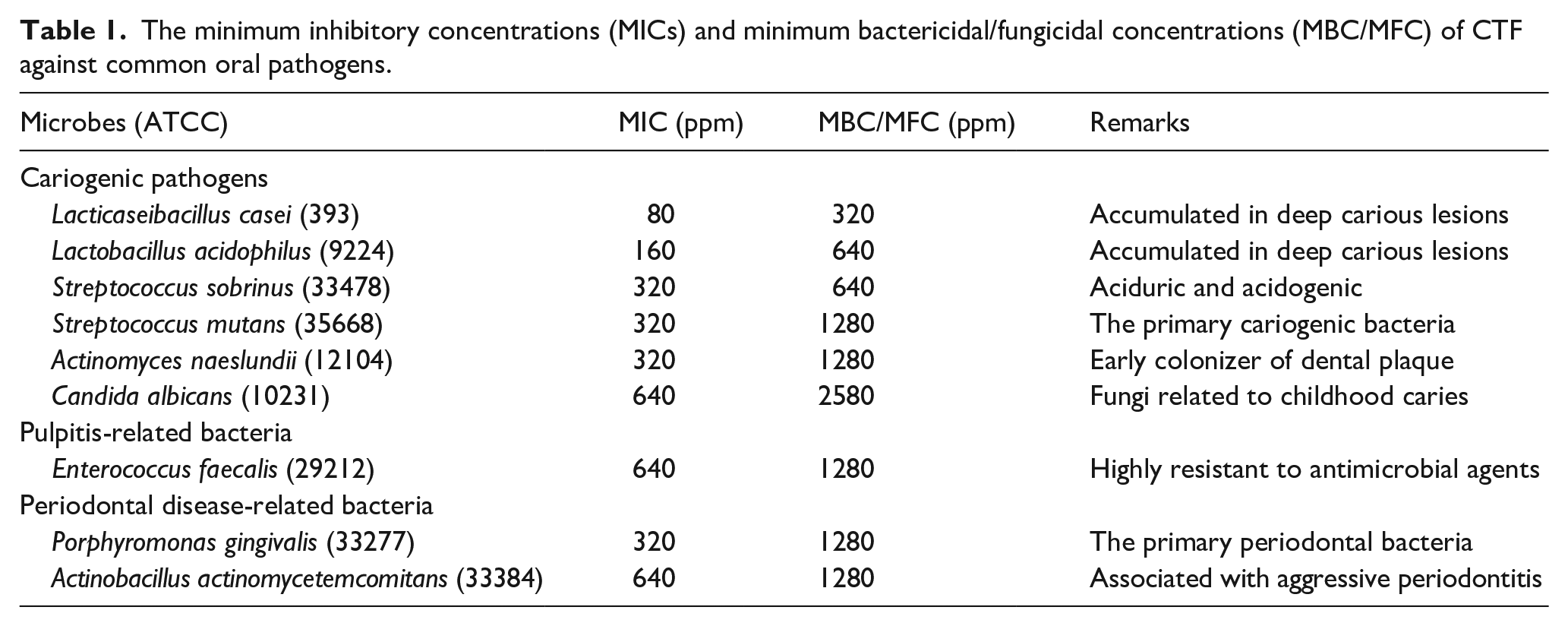
For cariogenic microbes, the MICs of the CTF against
Microorganisms morphology
Figure 1 presents TEM images of cariogenic pathogens with and without CTF treatment.

TEM images of cariogenic pathogens with and without CTF treatment.
Figure 2 shows TEM images of

TEM images of pulpitis-related bacteria with and without CTF treatment.
Figure 3 presents TEM images of periodontal disease-related bacteria with and without CTF treatment. The

TEM images of periodontal disease-related bacteria with and without CTF treatment.
Discussion
In recent years, copper has garnered considerable interest as a potential antimicrobial agent in dental treatments. Our previous study developed a novel CTF solution and examined the biocompatibility, antibacterial effect against
Cariogenic microorganisms are microbes that play a significant role in the development of dental caries. When the cariogenic microbes have colonised tooth surfaces, they can metabolise fermentable carbohydrates and generate organic acids, which can result in the demineralisation of tooth tissue.19,20 Constant mineral loss erodes the tooth structure, leading to dental caries. 21 To manage dental caries, it is important to manage bacterial activity and inhibit biofilm formation, which helps lower acid production and ultimately reduces demineralisation in dental caries.
For pulpitis-related bacteria,
Periodontal diseases are chronic inflammatory diseases marked by the degradation of gingival connective tissue and the resorption of the alveolar bone, often leading to eventual tooth loss.
39
These diseases are triggered by periodontopathogenic microbes present in the subgingival plaque, notably
Antimicrobial ability is a crucial factor in the selection of antimicrobial drugs, and it can be assessed using MIC and MBC. 44 The findings of the current investigation indicate that the CTF had notable antibacterial efficacy. The MIC and MBC of the CTF against the nine microbes were found to vary between 80 to 640 ppm and 320 to 2560 ppm, respectively.
Moreover, the TEM was employed to observe the morphological alterations in microbes treated with CTF, thereby providing insights into the antimicrobial mechanism of CTF. Researchers suggested that copper ions can interact with the bacterial cell membrane, causing structural damage. 45 This interaction can lead to increased membrane permeability, resulting in leakage of essential cellular contents and ultimately cell death. Copper ions can also catalyse the production of reactive oxygen species, which can cause oxidative damage to cellular components, leading to cell death. In addition, copper ions can bind to proteins, disrupting their three-dimensional structure and function. This can inhibit essential enzymatic activities and other protein functions, impairing the bacteria’s ability to survive. 46 Copper ions can also interact with bacterial DNA, causing strand breaks and other forms of genetic damage. This can lead to mutations, interference with replication, and ultimately cell death. 47 Furthermore, copper can disrupt various metabolic pathways within the bacterial cell. For example, it can inhibit enzymes involved in cellular respiration and other critical biochemical processes, leading to energy depletion and cell death. 48 Ammonia can increase the pH of its surrounding environment. Many oral microbes thrive in neutral to slightly acidic conditions, so the alkaline conditions created by ammonia can disrupt bacterial cell processes. Moreover, ammonia can diffuse across cell membranes and disrupt intracellular pH and metabolic activities. 49 Fluoride inhibits the enzyme enolase, which is crucial in the glycolytic pathway. 50
In this study, TEM images revealed that CTF treatment led to the disruption of the cell membrane, causing unusual membrane curvature, irregular cellular shapes, and intra-bacterial vacuolization. This disruption further resulted in the breakdown of the cytoplasmic membrane and leakage of cytoplasmic components from the microorganisms. These observations align with the TEM images captured in our prior study, where CTF exhibited similar effects on microbes. 17 However, this study primarily observed the antimicrobial mechanism of CTF through TEM images and did not involve a quantitative analysis of other potential factors contributing to CTF’s antimicrobial effects. These factors could include the interruption of DNA replication, denaturation of microbial proteins, and production of reactive oxygen species. In addition, the antimicrobial effect investigated in this study focused solely on single-species models. This approach provides simple, inexpensive, well-controlled, and repeatable experimental conditions. However, this strategy lacks complexity and does not account for the interactive effects between different microbial species. Future research initiatives should aim to provide a comprehensive analysis of the antimicrobial effects of CTF, including quantitative assessments of various contributing factors and investigations using multi-species models to better understand the antimicrobial effect of CTF.
Conclusion
This laboratory study showed that the novel CTF has antimicrobial effects against some common oral pathogens, specifically those associated with dental caries and periodontal disease. The CTF effectively inhibits the growth of these microbes and causes unusual cell morphology. CTF could be considered as a promising agent to manage oral infectious diseases including dental caries and periodontal disease.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study is supported by the RGC General Research Fund (GRF), Program No. 17100222.
