Abstract
Background
The antimicrobial activity of restorative materials is clinically relevant because all dental materials are subject to an environment containing bacteria. This study aimed to investigate the use of 2 methodologies referred to in the literature to assess antimicrobial properties of restorative materials and investigate whether material properties alter results of these traditional methodologies.
Methods
A number of dental restorative materials – namely, Chemfil Superior®, Spectrum®, Heliobond®, Ionoseal®, Dyract Extra®, Smart Dentin Replacement (SDR®) and Biodentine® – were characterized by scanning electron microscopy (SEM), energy-dispersive spectroscopy (EDS), X-ray diffraction (XRD) analysis, Fourier transform infrared (FT-IR) spectroscopy and pH analysis. Antimicrobial activity was assessed using agar diffusion and biofilm accumulation tests. Key factors affecting results were assessed using analysis of covariance.
Results
Biodentine after immediate mixing and Ionoseal aged for 6 weeks resulted in an inhibition zone, while significantly higher McFarland readings were observed in the presence of barium when using materials Ionoseal, Dyract and SDR at 24-hour aging. Through analysis of covariance it was shown that material properties affected methodology results.
Conclusions
Properties of materials affect results of antimicrobial testing, but this may not directly reflect the antimicrobial potential of the material in question. Careful choice of methodology and interpretation of results is important.
Keywords
Introduction
The rate of dental caries following restorative treatment is claimed to reach values of up to 50%-60% (1). Secondary caries is the main reason for failure of glass-ionomer cement (GIC) and composite resin restorations (1, 2). Composites result in more plaque accumulation than other materials, which may also be the cause of the increased rates of recurrent decay experienced with this material (3). Nonetheless, resin composites are nowadays the most commonly used dental restorative material (4). Adherence of cariogenic bacteria to restorative materials is considered an important step in the progression of secondary caries and is influenced by the restorative material's topography and surface chemistry (5). Therefore a restorative material with antimicrobial activity would be in the ideal location to prevent secondary decay (2). The use of such materials would also theoretically suppress residual infection and increase the survival of the restored tooth especially in minimally invasive approaches (6, 7).
Several methodologies have therefore been employed to assess antimicrobial properties of dental materials. Methodologies used in the literature vary significantly and there are no current standards for antimicrobial assessment of restorative materials. This study aims to assess whether 2 methods used in the dental literature are still clinically relevant methods for assessing antimicrobial properties of materials. With emphasis on the possibility that material properties may alter the results of methodologies used.
Methods
Seven restorative materials used for dentin replacement were included in this study as shown in Table I. The materials were mixed according to the manufacturers’ instructions.
Materials tested in the study

Assessment of Chemical Properties
Material chemical and physical properties were assessed through scanning electron microscopy (SEM), energy-dispersive spectroscopy (EDS), X-ray diffraction (XRD) analysis, Fourier transform infrared (FT-IR) spectroscopy and pH assessment in artificial saliva.
Microscopy and EDS Analysis
The set materials were characterized using SEM. Polished sections of set materials impregnated in resin were prepared, mounted on an aluminium stub and carbon coated. The specimens were viewed under a scanning electron microscope (Merlin Field Emission SEM; Carl Zeiss NTS GmbH, Oberkochen, Germany) at different magnifications in back-scatter electron mode, and EDS analysis of the different phases was carried out.
Phase Analysis
Phase analysis was carried out using XRD. The diffractometer (Bruker D8 Advance; Bruker Corp., Billerica, MA, USA) used Cu Kα radiation at 40 mA and 45 kV. Set materials were crushed into powder form using a mortar and pestle prior to testing, and the detector was rotated between 5° and 60°. A step of 0.02° 2θ and a step time of 1 second and spinning at 15 rpm were used. Phase identification was accomplished with search-match software utilizing the ICDD database (International Center for Diffraction Data, Newtown Square, PA, USA).
FT-IR Spectroscopy
Materials were crushed using a mortar and pestle and desiccated for 24 hours. And the resultant powders were assessed using FT-IR spectroscopy. Infrared spectra of the ground powders were obtained by mixing with KBr (2 mg powder/300 mg KBr) and analyzed with a FT-IR spectrometer (Shimadzu IRAffinity-1; Shimadzu Corp., Kyoto, Japan) using transmitted infrared spectroscopy with frequencies between 4,000 and 400 cm−1 (wavenumbers).
Assessment of pH in Artificial Saliva
The pH meter was calibrated using 3 standard calibrating solutions (pH 4, 7 and 10) after which the materials were immersed in Fusayama-Meyer artificial saliva (8), and pH of the leachate (n = 3) was monitored after 24 hours, 1 week and 6 weeks by a pH/mV/ISE meter (Hanna HI 3221; Hanna Instruments, Woonsocket, RI, USA) with a single-junction (Ag/AgCl) ceramic pH electrode (Hanna HI 1131). The results were recorded, and the mean and standard deviation of each was calculated.
Investigation of Antimicrobial Tests
Bacterial strain Streptococcus mutans (NTCC 10449/ATCC 21575; LGC Standards, Wesel, Germany) stored from stock cultures in culture beads at −20°C was used to assess the antibacterial properties of the test materials. Although dental decay progression is dependent on several species of bacteria (7, 9), S. mutans was chosen as it is the most common species associated with dental decay (2, 9), with strain NTCC 10449/ATCC 21575 being one of the most commonly tested in the literature (6, 10).
Material diskettes (10-mm diameter, 2-mm height) were disinfected in 70% alcohol in an ultrasonic bath (which also has the added advantage of removing any uncured monomer, which would later have dissolved into solution and altered the biofilm accumulation test results especially (11)). Specimens were then dried using sterile gauze. Sterilized samples were then aged in 1 mL of sterile artificial saliva (8), in sealed sterile polycarbonate containers in an incubator at 37°C for 24 hours, 1 week or 6 weeks. After aging, the samples were rinsed with sterile water, dried with sterile gauze and subjected to antimicrobial testing. Several conditions for aging of materials prior to antimicrobial testing have been documented in the literature. Most common aging ranged from 24 hours (11), to 1 month (2). In in vitro studies, aging was for longer periods, ranging from 3 months (12) to 6 months (13, 14).
Changes were assessed after immediate mixing, 24 hours, 1 week and 6 weeks to assess a span of time, which was longer than 1 month. Aging of materials is generally carried out in distilled water a 37°C (10); this was modified to sterilized artificial saliva, using the Fusayama et al formulation (8), to better mimic the conditions present inside the mouth (8).
Agar Diffusion Test
The strain was cultivated in brain-heart infusion (BHI) broth at 37°C, and a loopful (10 µL) of inoculum was transferred to 10 mL of BHI broth (Biolab, Zrt, Hungary). S. mutans was incubated for 24 hours, and 350 µL of bacterial suspension was placed on a BHI agar plate (Biolab, Zrt, Hungary) for 30 minutes at room temperature. Each sample (n = 4) was placed on the prepared agar plate, more than 20 mm away from the other samples, with the sample surface touching the agar. Plates without bacterial suspension were used as controls. The plates were then incubated at 37°C for 24 hours.
Incubation conditions for S. mutans used in antimicrobial analysis of dental materials also varies in the literature. Incubation times vary between 24 and 48 hours (6, 15), in either aerobic (6), anaerobic or CO2-enriched conditions (15).
Anaerobic conditions at 24-hour incubation were chosen to mimic conditions under restorations, since the most common area for secondary caries is the gingival area, where availability of oxygen is generally deprived (16). S. mutans has also been shown to exhibit more efficient growth by organisms in conditions deprived of oxygen (17). The presence of any zone of inhibition was recorded.
Biofilm Accumulation Test
S. mutans was cultured overnight at 37°C in BHI broth in an anaerobic atmosphere. The bacterial suspension obtained was adjusted to 0.5 McFarland (McF) standard for further usage. Specimens (10-mm diameter) were placed in 2 mL volume of BHI supplemented with 1% sucrose inoculated with 20 µL of bacterial suspension in sterile Bijoux bottles (n = 5). Specimens without inoculum were used as a control. After incubation at 37°C for 24 hours in an anaerobic chamber, each specimen was vortexed (Vortex Genie 2; Scientific Industries, New York, USA) for 30 seconds in 1 mL of distilled water to detach adherent bacteria and disperse congregated cells. After removing the sample from the tube, the turbidity (McF) of the bacterial suspension was measured using a densitometer (Densichek Plus; Biomeriuex, Boston, USA).
Statistical Analysis
The data were evaluated using PASW Statistics 18 (SPSS Inc., Chicago, IL, USA). One-way analysis of variance and Tukey post hoc tests were used to perform multiple comparison tests. The chi-square test was used to assess differences between categorical variables, while analysis of covariance (ANCOVA) was used for interaction analysis between turbidity and variables at a significance level of a p value equal to 0.05.
Results
Assessment of Chemical Properties
Microscopy and EDS Analysis
Scanning electron micrographs are shown in Figure 1. Table II summaries the elemental composition observed in the general EDS of the materials. All materials contained an organic component as shown by their carbon and oxygen content. Heliobond contained the organic phase only, without any filler particles, Chemfil, Spectrum, Ionoseal, Dyract and SDR all exhibited glass particles, with Chemfil, Ionoseal and SDR showed larger glass particles than Dyract and Spectrum. Chemfil was composed only of glass in a matrix, while the other materials also incorporated a radiopacifier phase, achieved through high atomic weight elements such as strontium and barium. Biodentine on the other hand was composed of particles of calcium, carbon and oxygen in a matrix rich in calcium and silicon. Smaller zirconium oxide particles exhibiting a high electron contrast were also present.

Back scatter electron micrographs of polished materials. SDR = smart dentin replacement.
Summary of general elemental analysis results
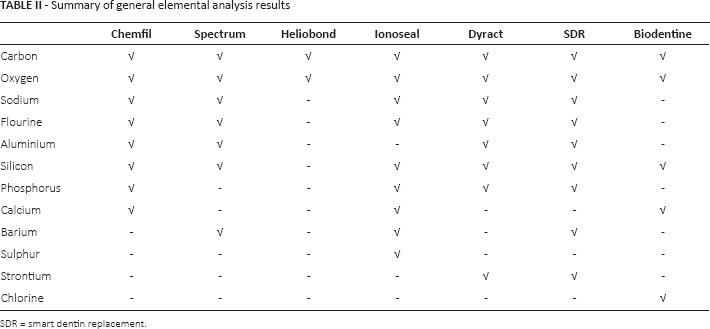
SDR = smart dentin replacement.
Phase Analysis
The phase analysis of the test materials is shown in Figure 2. The XRD plots demonstrated the absence of crystalline structures for SDR, Heliobond, Spectrum and Chemfil, as no specific peaks could be identified. Biodentine exhibited peaks for the tricalcium silicate phase (ICDD: 31-0301) with peaks at 32.461° and 34.27° 2θ. In addition Biodentine exhibited a Portlandite (ICDD: 44-1481) peak at 18.05° and 34.09° 2θ and peaks for the zirconium oxide phase (ICDD: 37-1484) at 28.17° and 31.47° 2θ and calcium carbonate (ICDD: 85-1105) at 29.459° 2θ. The Dyract Extra exhibited peaks for strontium fluoride (ICDD: 06-0262) at 26.572°, 44.118° and 52.264° 2θ. Ionoseal exhibited multiple peaks for barium sulphate (ICDD: 78-1833) at 20.255°, 22.887°, 25.002°, 26.041°, 27.015°, 28.947°, 31.671°, 32.77°, 42.772°, 42.981° and 43.328° 2θ.
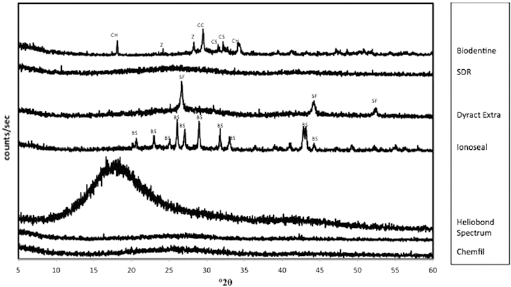
X-ray diffraction (XRD) plots of test materials showing the main crystalline phases present. SDR = smart dentin replacement. BS = barium sulphate, CC = calcium carbonate, CS = tricalcium silicate, CH = calcium hydroxide, SF = strontium fluoride, Z = zirconium oxide.
FT-IR Spectroscopy
FT-IR plots (Fig. 3) exhibited similar patterns with the high-frequency and intermediate regions of the spectrum showing no absorption bands. The low-frequency region was dominated by a strong band in the 800-1,200 cm−1 region. This is typical of Si-O stretching vibrations. All of the materials were composed of a silicate glass, as shown by the EDS analysis (Tab. II). Furthermore, bands in the region of 1,600-1,400 cm−1 due to H-O-H bending vibrations were also present.
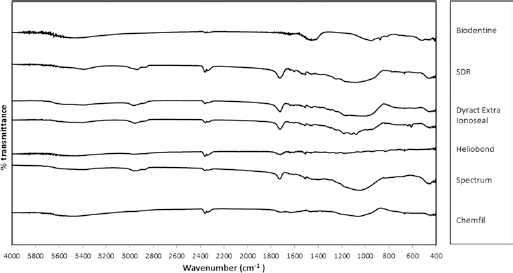
Fourier transform infrared spectroscopy scans of test materials. SDR = smart dentin replacement.
Assessment of pH in Artificial Saliva
The results for the pH of the storage solution of the test materials is shown in Figure 4. At all time intervals tested, all materials tested exhibited slightly acidic pHs, apart from Biodentine which exhibited alkaline pH values ranging from 11.33 to 12.13. Statistically significant differences in pH were observed between materials after immersion in artificial saliva for the same time periods. After 24 hours of aging, Chemfil and Dyract exhibited a reduction in pH, while Biodentine exhibited a very alkaline pH (p<0.05). At 1 week, Ionoseal was more acidic and Biodentine more alkaline than the control (p<0.05), while at 6 weeks, Chemfil and Ionoseal were more acidic than the artificial saliva control and Biodentine more alkaline (p<0.05). Differences were also noted between pH of the same material at different aging times. Heliobond's pH at 24 hours decreased after 1 week (p = 0.005), while Biodentine pH increased at 1 week (p = 0.02). Ionoseal's pH did not change over time (p<0.05)
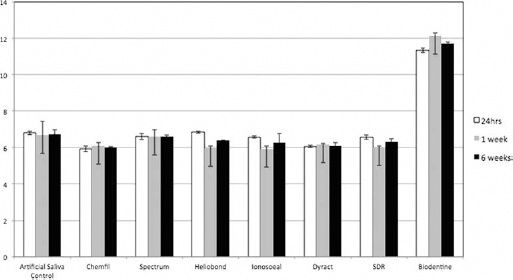
The pH of artificial saliva and storage solutions over a period of 6 weeks. Values are means, with whiskers representing standard deviations. SDR = smart dentin replacement.
Investigation of Antibacterial Properties
Agar Diffusion Test
A lighter area around the sample was observed around all Biodentine control samples and to a lesser extent around some Ionoseal control samples possibly due to diffusion of the material through the agar (Fig. 5), indicated by arrows. All of the samples tested apart from Biodentine did not produce an inhibition ring after immediate mixing (p = 0.003), and a less defined inhibition area was present around Ionoseal aged for 6 weeks (p = 0.003) in artificial saliva.
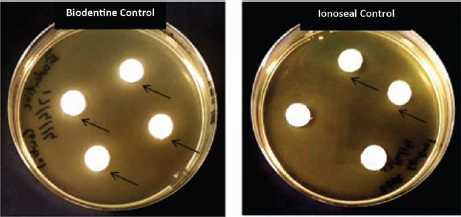
Agar plates showing presence of a lighter region in Biodentine and Ionoseal control samples without bacterial inoculation, indicating increased solubility of these materials.
Biofilm Accumulation Test
The results of the biofilm accumulation test are shown in Figure 6. Turbidity (McF) increased with aging of the material in Chemfil, Spectrum, Heliobond and SDR, and decreased with aging of the material in Biodentine. On the other hand, Ionoseal's McF started increasing with time and then decreased again at 6 weeks, while Dyract's readings increased up to 24 hours of aging, then decreased at 1 week and increased again at 6 weeks. Differences between McF of controls and materials at different aging times were not statistically significant.
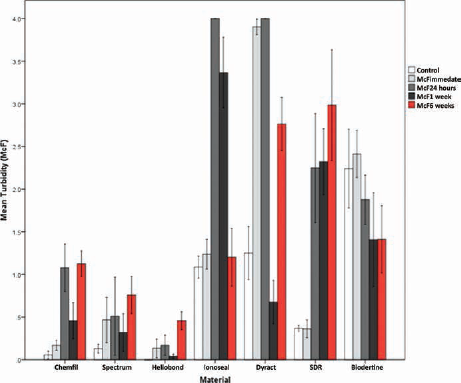
Biofilm accumulation test for mean McFarland (McF) of materials, for control and after different aging periods. SDR = smart dentin replacement.
Statistically significant differences versus controls were observed in Chemfil 24 hours (p = 0.004) and 6 weeks (p = 0.003), Heliobond 6 weeks (p = 0.011), Ionoseal 24 hours and 1 week (p<0.001), all Dyract samples (p<0.05) and SDR 1 and 6 weeks (p<0.05). An increase was also observed from Chemfil immediate to 24 hours aging and 6 weeks aging (p<0.001), as well as between 1 and 6 weeks of aging Heliobond (p = 0.022). Dyract McF decreased between immediate and 1 week (p = 0.00) and 6 weeks (p = 0.018), although an increase in McF was shown between 1- and 6-week samples (p<0.001). McF increased significantly in SDR between the immediate and 1-week (p = 0.039) and 6-week samples (p = 0.009). None of the changes observed in Spectrum and Biodentine were statistically significant (p<0.05).
Statistical Analysis and Correlation of Results
ANCOVA was carried out to identify relationships between the variables (elemental composition, material, aging and pH) and turbidity values observed (Tab. IIIA). After removing the weaker predictors only 5 explanatory variables were retained (aging, material, aluminium, barium and pH) (Tab. IIIB); however aluminium and pH had a p value greater than 0.05, indicating that they were not significant explanatory variables (Tab. IIIC). After removing these 2 predictors (1 at time) from the model, the resulting parsimonious model included 3 significant predictors. Material was the best predictor, because it had the lowest p value (p = 3.09e-17). This was followed by barium (p = 1.52e-8) and aging (p = 0.001) (Tab. IIID). Higher levels of turbidity were observed in the presence of barium, when using materials Ionoseal, Dyract and SDR and at 24-hour aging (Tab. IV).
Progression of analysis of covariance to identify the parsimonious model identifying relationship between the variables and turbidity values
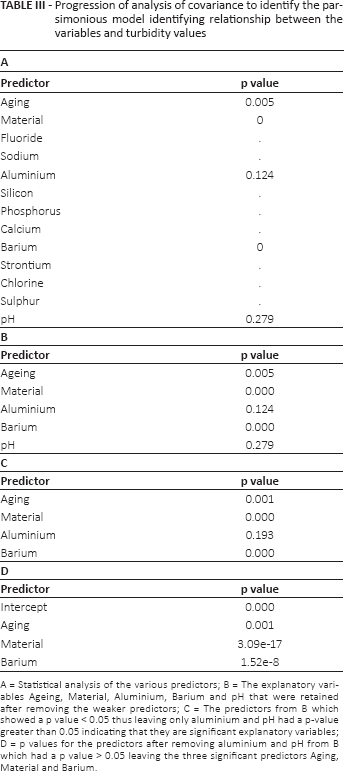
A = Statistical analysis of the various predictors; B = The explanatory variables Ageing, Material, Aluminium, Barium and pH that were retained after removing the weaker predictors; C = The predictors from B which showed a p value < 0.05 thus leaving only aluminium and pH had a p-value greater than 0.05 indicating that they are significant explanatory variables; D = p values for the predictors after removing aluminium and pH from B which had a p value > 0.05 leaving the three significant predictors Aging, Material and Barium.
Descriptive statistics for barium, aging and materials
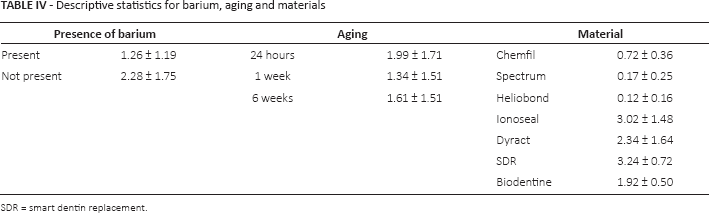
SDR = smart dentin replacement.
Discussion
The importance of antibacterial properties has also gained popularity in the field of dental materials (18), including antibacterial restorative fillings, due to their potential in secondary decay prevention (14). All materials chosen for this study included commercially available materials used as restorative and dentin replacement materials. Tooth-colored materials are nowadays most commonly used as restorative materials, especially resin composite-based materials (4, 14). Therefore this study focused on a conventional composite and bonding agent, Spectrum and Heliobond; a composite that is relatively new to the market, SDR; a conventional glass-ionomer cement, Chemfil Superior; and 2 hybrids, Ionoseal and Dyract Extra, a compomer and Biodentine from the calcium silicate cement class.
The literature shows conflicting evidence on the antimicrobial properties of glass ionomer cements and hybrid materials (19). No published data were found on antimicrobial properties of SDR. Also there are no currently published data comparing the differences in antimicrobial properties of currently used materials after aging, in relation to chemical and physical properties. Furthermore, there is also no published data comparing results achieved by different methods of antimicrobial testing. To our knowledge, no other study has directly shown how material properties directly affect agar diffusion tests and biofilm accumulation tests.
The agar diffusion test and the biofilm accumulation test are the most commonly used methodologies used in the literature to assess antimicrobial properties of restorative materials (14). The agar diffusion test is the most commonly used method (3), probably due to this method's simplicity and cost-effectiveness (12, 20). In this study, the agar disc diffusion modification of this test was used, which has the added simplicity of only placing discs of the sample on the agar plate (21).
Biofilm accumulation testing through assessment of optical density was also used as a quantitative analysis in this study. A biofilm consists of microbial cells and their extracellular material accumulated on substrata (22). Assessment of bacterial growth via changes in optical density is commonly quoted in the dental literature to involve several methods, including spectrophotometry (15). The material sample is incubated in a broth, which would allow biofilm formation around samples which do not possess antibacterial properties. The sample is then agitated in a solution to detach any biofilm around the sample in solution, which would increase the turbidity of the solution, which can be quantified as a value of optical density. Optical density is measured as intensity reduction of a light beam transmitted through a biofilm, and it correlates with biofilm mass, measured as total carbon and as cell mass (23). In this study, a densitometer was used to assess turbidity of the resultant solution. Densitometers assess turbidity in McF units, which is a measurement proportional to microorganism concentrations, in colony forming units (CFU) per milliliter present in the solution. One unit of McF is equivalent to 3 × l08 CFU/mL (24). The major advantage of this method is its simplicity and in theory the immediate estimation of CFU.
The materials tested showed significant differences between their chemical components and physical properties. This was mirrored in the different results for antimicrobial testing and the results of the statistical analysis showing that the results obtained in the turbidity analysis were strongly related to material type. Freshly mixed Biodentine and 6-week aged Ionoseal showed signs of antimicrobial potential due to the significant formation of inhibition halos in the agar diffusion test. Antibacterial properties of tricalcium silicate cements (7), as well as those of resin-modified glass ionomer cements (RMGICs), have been documented in the literature (25). Although antimicrobial properties of RMGICs have been attributed to the initial drop in pH, pH was not shown to be a significant contributor to decreased turbidity in this study. Release of strontium and fluoride ions may also produce an antibacterial effect in RMGICs (26), but Ionoseal does not contain strontium (as confirmed by the chemical analysis), and no reports in the literature were found for the rate of fluoride release from Ionoseal over time. It is possible that rather than increased antimicrobial properties, what these materials truly possess is increased solubility. The increased solubility would allow the materials to diffuse more through the agar and react with bacteria on the agar quicker than the other materials. In fact in the control samples, both Biodentine and Ionoseal resulted in a difference in shade of the agar surrounding the samples, indicating that the samples diffused into the agar. Biodentine has been shown to have high washout values (27), while Ionoseal has shown extensive hydrolytic degradation (28).
In the biofilm accumulation test, significantly higher McF readings were observed in the presence of barium when using the materials Ionoseal, Dyract and SDR with 24-hour aging and this therefore showed the accumulation of more bacteria. The ability of RMGICs (such as Ionoseal) and compomers (such as Dyract) to prevent biofilm accumulation has been disputed in the literature. While some authors claim that a combination of fluoride release, low initial pH (25), and when present, strontium release result in antibacterial properties (26), others claim that these properties are not enough to prevent bacterial growth and biofilm attachment (15). SDR, on the other hand, is a composite; and composites have been shown to allow considerable adherence of bacteria and allow more plaque than other restorative materials (3). No data were found which related barium to increased biofilm accumulation on dental materials. Barium has been shown to leach into solution from dental materials (29), which would in turn explain the increase in turbidity in Ionoseal and SDR, since in this study, it has been confirmed through EDS analysis that these materials contain barium. In Ionoseal, it is in the form of barium sulphate, while in SDR it is in the form of barium-aliminum-borosilicate glass. Barium and its salts are generally used as a radiopacifier, due to its high atomic number (30). Therefore if leached into the solution tested, it could lead to increased turbidity of the solution, leading to possible misinterpretation of the antimicrobial properties of the material tested. This would explain the ANCOVA results, which showed that barium was a strong predictor of turbidity. Dyract has not been shown to contain barium, but it contains strontium, which also has a high atomic number and is used as a radiopacifier (30). Increased turbidity associated with Dyract samples may be related to leaching of ions used as radiopacifiers into the solution rather than higher rates of biofilm accumulation on this material.
Conclusion
Although the antimicrobial properties of restorative materials are clinically relevant, antimicrobial testing results may be altered by the material properties and therefore may not be extrapolated clinically. Further assessment of the methodologies used is required to be able to compare materials of different physical and chemical properties. More emphasis on methods which assess actual bacterial presence rather than turbidity analysis is required – such as a method using assays (fluorescent assays). These are more useful as they are less affected by material properties and provide information on whether the bacteria are dead or alive. Methodologies should also mimic more of the conditions present inside the mouth, including methods using multispecies infection.
Footnotes
Acknowledgements
The authors wish to acknowledge the assistance of Dr. Christopher Barbara, Head of Department of Pathology, Mater Dei Hospital for access to equipment, Ing. James Camilleri of the Department of Metallurgy and Materials Engineering, Faculty of Engineering and Mr Jonathan Spiteri from the Department of Chemistry, Faculty of Science, University of Malta for their help with the testing.
Financial support: ERDF (Malta) financed the testing equipment through the project Developing an Interdisciplinary Material Testing and Rapid Prototyping R&D Facility (Ref. no. 012).
Conflict of interest: None of the authors has any financial interest related to this study to disclose.
