Abstract
Objective:
To evaluate the effect of COVID-19 preventive mouthwashes on the surface hardness, surface roughness (Ra), and color change (ΔE) of three different polymer-based composite CAD/CAM materials (Vita Enamic (ENA), Grandio Block (GB), Lava Ultimate (LU)).
Methods:
A total of 100 rectangular-shaped specimens with dimensions of 2 mm × 7 mm × 12 mm were obtained by sectioning three different CAD/CAM blocks and randomly divided into five subgroups according to the 30 days of mouthwash immersion protocol as follows: Control: artificial saliva, PVP-I: 1% povidone-iodine, HP: 1.5% hydrogen peroxide, CPC: mouthwash containing 0.075% cetylpyridinium chloride, EO: mouthwash containing essential oils. Microhardness, Ra, and ΔE values were measured at baseline and after 30 days of immersion protocols. Data were analyzed using the Wald Chi-square, two-way ANOVA, and post hoc Tukey tests.
Results:
The independent factors (materials and solutions) significantly influenced the microhardness and color (
Conclusion:
Within the limitations of this study, it was found that the surface hardness and color of tested polymer-based composite CAD/CAM materials are susceptible to degradation and change after 30 days of immersion in 1% PvP-I and 1.5% HP mouthwashes.
Keywords
Introduction
COVID-19 is an infectious disease that is caused by a virus with a single-stranded RNA. The virus known as SARS-CoV-2 belongs to the coronavirus family and it may spread from one person to another through direct contact or respiratory droplets released by an infected person when coughing or sneezing. 1 This virus, for a long time the cause of a global pandemic, is still a cause of health problems today due to its mutations. 2 The oral mucosa, gums, tongue epithelium, and salivary glands serve as reservoirs for SARS-CoV-2 in the mouth, so dentists and dental patients are at risk of contracting COVID-19 disease through regular exposure to aerosols.3,4 Various mouthwashes have been recommended to reduce the transmission risk by decreasing the viral load in the oral cavity.5,6
Povidone-iodine (PVP-I) is an antiseptic agent with broad-spectrum effectiveness against bacteria, fungi, protozoans, and viruses. It is a reliable solution for disinfecting various surfaces to protect against various harmful microorganisms. The PVP-I reduces inflammation by releasing free iodine, which destabilizes the virus’s lipid envelope, degenerates its nucleoproteins, and oxidizes the viral nucleic acids. 7 PVP-I is available in various forms, including ointments, sprays, and mouthwashes, with concentrations ranging from 0.23% to 10%. 8 A study reported that 99.8% of SARS-CoV-2 was inactivated by incubation with 1% PVP-I for 30 s. 9
Cetylpridinium chloride (CPC) is a reliable and secure quaternary ammonium compound suitable for use in treatment. 10 The use of CPC has been shown to have antiviral effects in influenza patients, resulting in a significant reduction in the duration and severity of cough and sore throat. 11 Currently, CPC is utilized in mouthwashes at a concentration ranging from 0.02% to 0.075%. 12 Komine et al. 13 stated that incubation with mouthwashes containing 0.04%–0.075% CPC can inactivate over 99.99% of SARS-CoV-2 in 20–30 s.
Essential oils (EO) are volatile compounds extracted from plants. EO mouthwashes contain terpenes, terpenoids, and phenylpropanoids as their main ingredients, with ethanol as an inactive component. 14 Mouthwashes that contain EOs are commonly used both professionally and at home and have been found to exhibit virucidal effects. 15 Meister et al. 16 stated that after 30 s of exposure to EOs, SARS-CoV-2 was eliminated to undetectable levels.
Since the beginning of the century, hydrogen peroxide (H2O2- HP) has been used in dentistry either alone or combined with salts. 17 When used as a mouthwash, this liquid is clear, colorless, and odorless. 18 Two studies have reported that SARS-CoV-2 can be inactivated by incubating it with 1.5% hydrogen peroxide for 30 s. One study reported an inactivation rate of 90%, 16 while another reported an inactivation range of 90%–99%. 19
Computer-aided design and computer-aided manufacturing (CAD/CAM) technology was first introduced to the dental industry in 1980. 20 In CAD/CAM technology, polymer-based composite materials combine the advantages of composite and ceramic materials. These materials have an elastic modulus similar to dentin, making them ideal for intraoral milling, conforming, and polishing. 21 Due to their comparable mechanical performance, they can be used as an alternative to ceramics. Polymer-infiltrated ceramic network (PICN), which has both ceramic and polymer properties, is one of the materials used with CAD/CAM technology. PICN has high flexural strength and elasticity similar to dentin, and superior wear resistance compared to composite resins. 22 Nanoceramics, one of the polymer-based CAD/CAM materials, have a microstructure similar to resin composites but with different proportions. These materials contain nanoparticles made of either traditional ceramics, polycrystalline ceramics (such as zirconia), or a hybrid mixture of the two. 23 Resin composite blocks (RCBs) used for CAD/CAM are manufactured by the incorporation of filler particles into a mixture of monomers that are cured under high temperature and pressure. Compared to traditional resin composites, this polymerization method results in reduced water absorption, and improved mechanical properties of the material. 24 These polymer-based CAD/CAM composite materials mentioned above can be used in dentistry for inlay, onlay, veneer, partial crown, and full crown restorations. 25 In restorative dentistry, restorative materials are expected to retain their mechanical and optical properties for a long time. Despite the benefits mentioned above, mouthwashes can affect the surface properties of polymer-based composite CAD/CAM materials. 26 Surface properties of restorations are important for esthetic properties, patient comfort, and lifespan of the material. 27 There are studies in the literature evaluating the degradation of resin composites when immersed in mouthwashes used to prevent COVID-19.20,21 However, no comparative study has yet been conducted on the mechanical and optical properties of polymer-based composite CAD/CAM restorative materials when immersed in mouthwashes used for the prevention of COVID-19, to the best of our knowledge.
This study aimed to evaluate the effects of mouthwashes (1% povidone-iodine, 1.5% hydrogen peroxide, 0.075% cetylpridinium hydrochloride, and essential oils) used for COVID-19 prevention on the surface hardness, surface roughness, and color changes of a PICN, a nanoceramic and a resin composite CAD/CAM material. The first null hypothesis was that the surface hardness of CAD/CAM materials tested would be affected after immersion in mouthwashes tested. The second null hypothesis was the mouthwashes tested would affect the surface roughness of the CAD/CAM materials. The third null hypothesis was the color of the CAD/CAM materials would change after immersion in the mouthwashes.
Methods
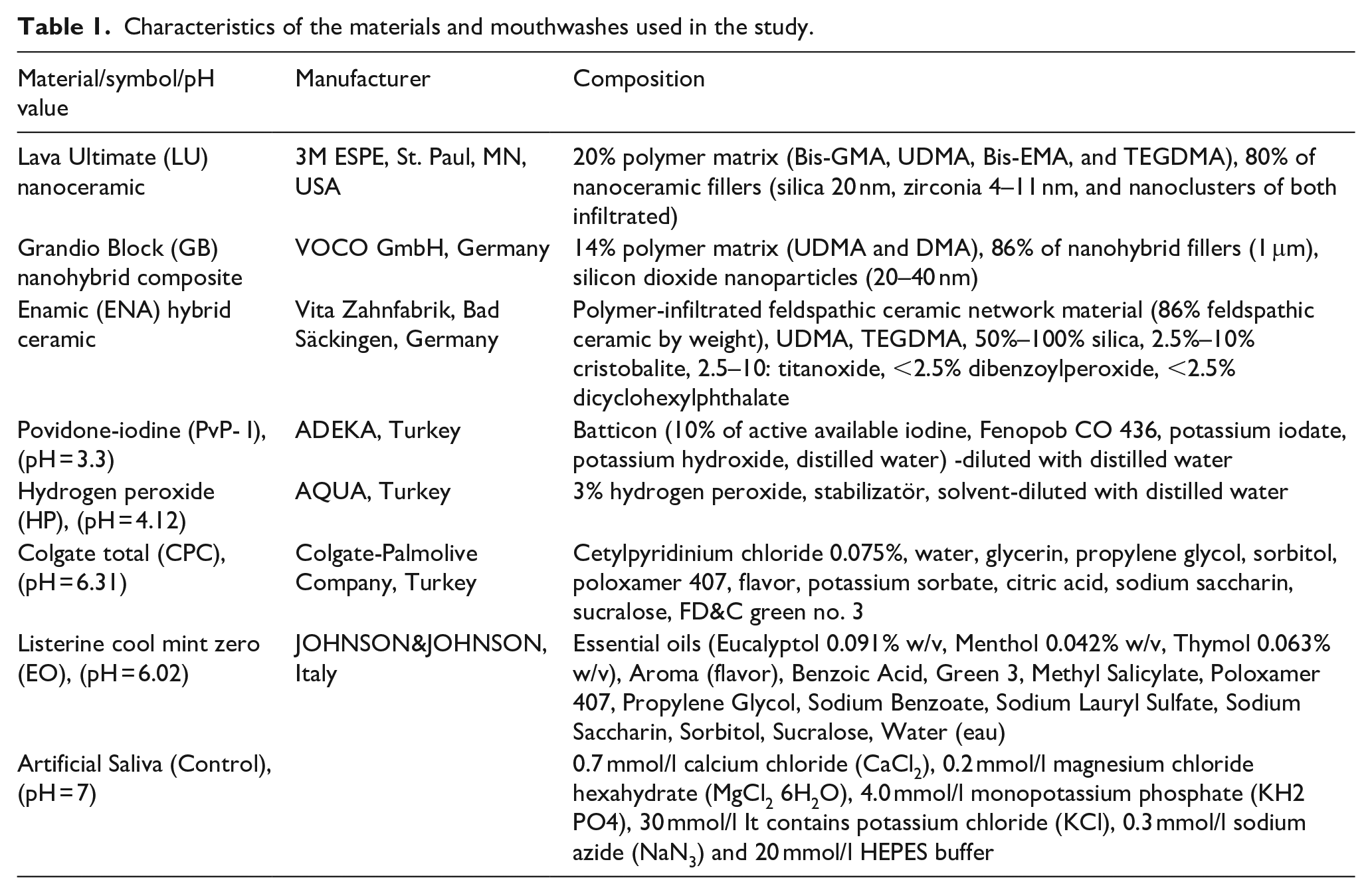
Table 1 presents information about the materials, mouthwashes, and artificial saliva used in this study.
Characteristics of the materials and mouthwashes used in the study.
Specimen preparations
Three polymer-based composite CAD/CAM materials (Vita Enamic, Lava Ultimate, Grandio Block) were selected for this study. Rectangular specimens with dimensions of 2 mm × 7 mm × 12 mm were obtained from CAD/CAM blocks using a water-cooled diamond saw (Micracut 201; Bursa, Turkey). A total of 300 specimens were prepared, 100 for each group. The specimens were polished using silicon carbide paper discs ranging from P600 to P1200 on a grinding machine (Buehler, Lake Bluff, IL) to achieve a standard surface.
Mouthwash and artificial saliva preparations
In this study, 1% povidone-iodine and 1.5% hydrogen peroxide mouthwashes were freshly prepared every day for 30 days by diluting with distilled water. The artificial saliva solution was prepared according to the formula of Pashley et al. 28 The pH of the artificial saliva was adjusted to seven and renewed weekly. The pH of each mouthwash and artificial saliva was measured using a pH meter (Seven Compact pH-meter (Mettler-Toledo 135 International Inc., Columbus, OH, USA).
Mouthwash immersions
The prepared resin CAD/CAM specimens were divided into five subgroups for mouthwash immersion (1% povidone-iodine, 1.5% hydrogen peroxide, cetylpyridinium chloride and essential oils) and artificial saliva (control), with 20 specimens in each group. Half of the specimens were used for hardness testing and the other half for color measurements and surface roughness testing. Surface roughness, hardness, and color measurements of the specimens were taken before mouthwash immersion. Then, specimens were immersed in 25 mL of mouthwash for 1 min twice a day, and this process was repeated every day for 30 days. The specimens were stored in artificial saliva at 37°C when they were not immersed in mouthwash. The solution container was sealed with paraffin wax to prevent evaporation and contamination. The mouthwashes were changed, and the pH was measured daily throughout the study. After the immersion process was completed, the specimens were rinsed with deionized water, and dried, then their final surface hardness, surface roughness, and color measurements were taken.
Surface hardness testing
The surface Vickers hardness of each specimen was measured using a microhardness tester (HMV-G20S, Shimadzu Corp., Kyoto, Japan) with a Vickers diamond indenter. Three indentations were made by applying a 1000 g load on the top surface of each specimen for 15 s. The diagonal lengths of each indentation were measured using a microscope at ×40 magnification. Microhardness values were calculated using the formula HV = 1.854 P/d 2 , where HV represents micro-hardness in kg/mm2, P is the load in kgf, and d is the average length of the diagonals in mm. The mean hardness values were recorded by calculating the average of the three hardness values for each specimen. The indentation images of each group were taken initially and after the mouthwash immersion.
Surface roughness testing
The surface roughness values (Ra) of specimens were measured using a profilometer (SURTRONIC 25, Taylor-Hobson, Leicester, UK) equipped with a stylus featuring a 5 μm tip radius and a 90° tip angle. The stylus traversed a distance of 4.0 mm at a constant speed of 0.5 µ/s and a measuring force of 4 µN. The surface roughness cut-off value was set at 0.8 mm. Initially and after immersion in mouthwash, three measurements were taken from the surface of each sample. The average of the measurements was then calculated and recorded.
Colour measurements
A spectrophotometer (Vita Easyshade, VITA Zahnfabrik, Germany) was used to measure the color values (L*, a*, and b*) of each specimen under standardized lighting conditions (CIE D65 illumination). The spectrophotometer was calibrated according to the manufacturer’s instructions before each measurement. Mean values were calculated from three measurements taken from the central areas of the specimens initially and after immersion protocols. The color change values (ΔE) were calculated using the following formula (CIEDE2000).
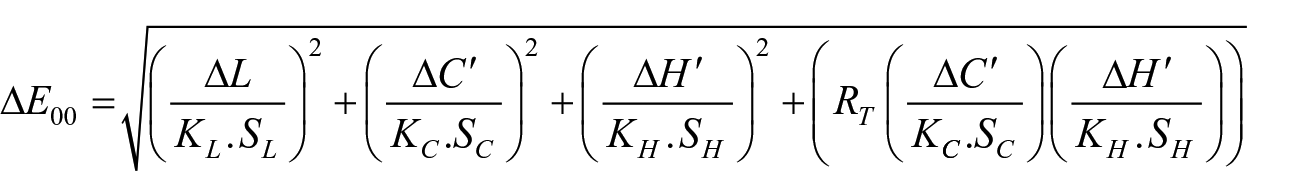
The parametric factors of the CIEDE2000 color difference formula were set as 1. The perceptibility threshold was taken as ΔE00 = 0.8 and the acceptability threshold as ΔE00 = 1.8. 29
Statistical analysis
The data were analyzed using IBM SPSS V23 (Chicago, USA). The conformity of the values to normal distribution was examined by Shapiro Wilk test. The comparison of hardness and roughness values of the groups was analyzed using the Wald Chi-square test. The mean percentage of microhardness and surface roughness change were calculated for each CAD/CAM material. The mean percentage hardness and roughness change, as well as the color change between the groups, were compared using a two-way ANOVA and post hoc Tukey HSD tests. The level of significance was set at 0.05.
Results
The mean and standard deviation of the surface hardness values are shown in Figure 1. Considering the initial surface hardness values of the materials tested, the values reported for the ENA were significantly higher than those of the other materials (
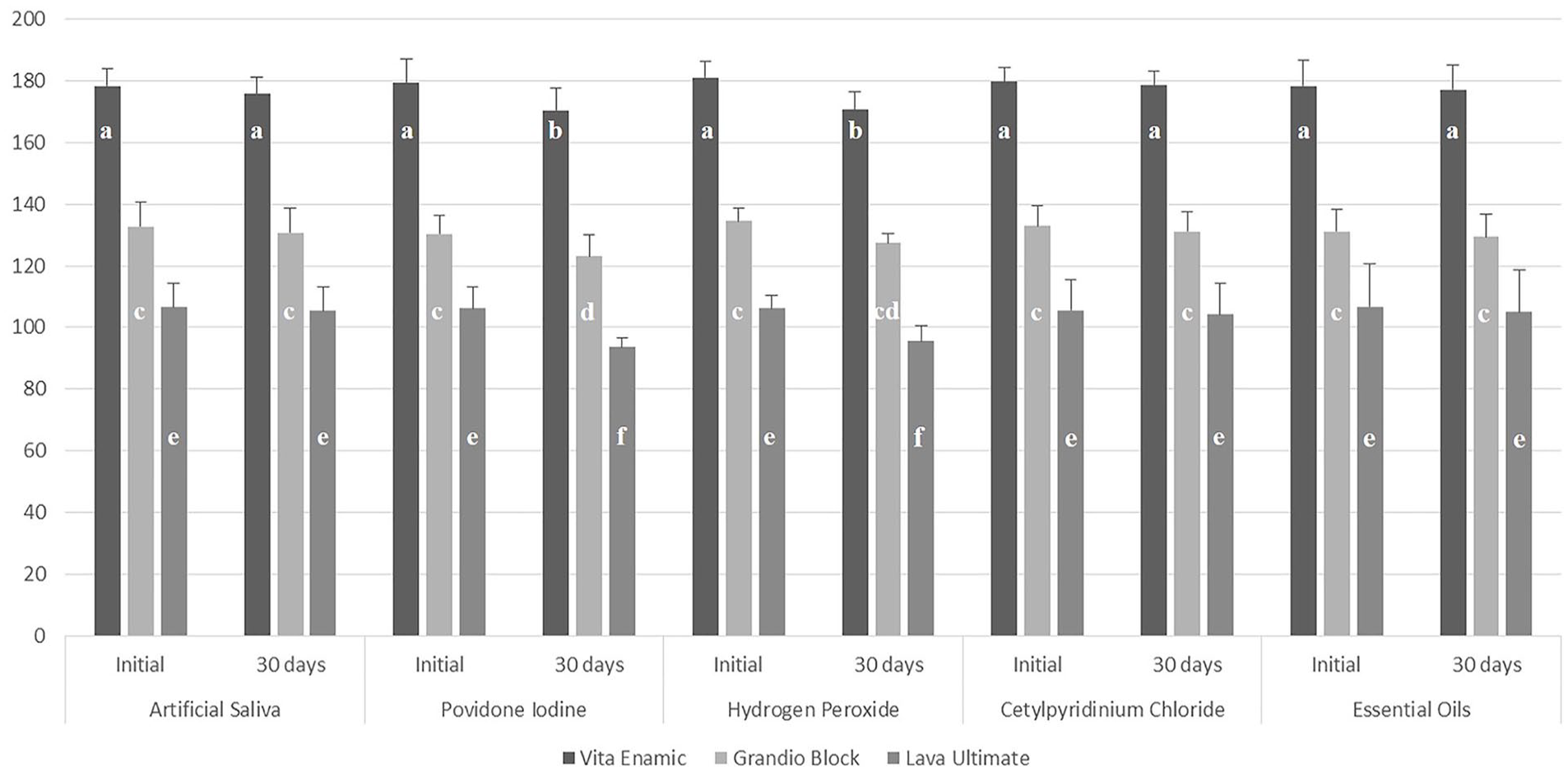
Mean and standard deviation of microhardness values and intergroup comparison for different immersion protocols. (a–f) Different lowercase letters indicate significant differences between groups.
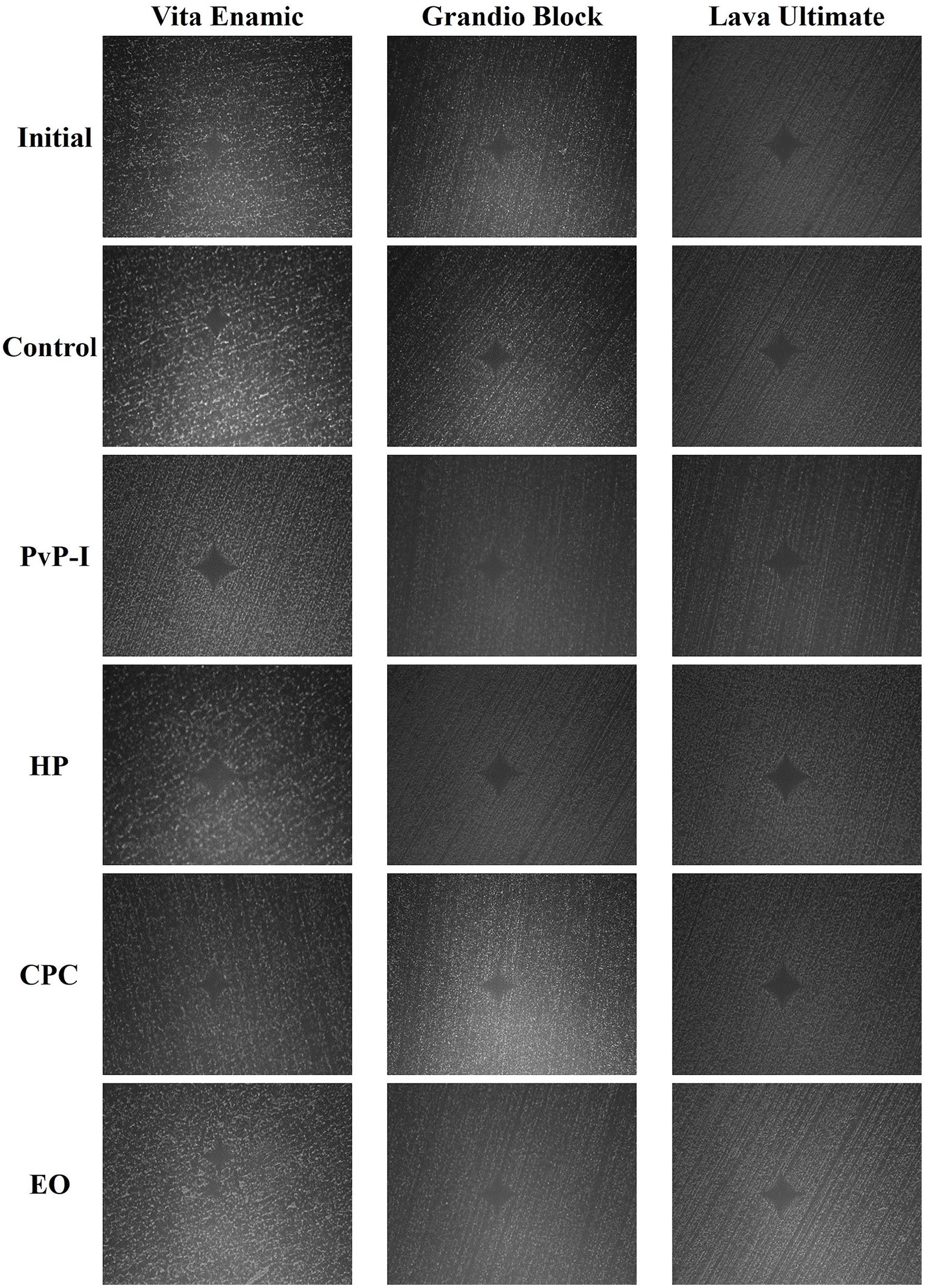
Representative Vickers indentation images at ×40 magnification of the CAD/CAM materials initially and after immersion in artificial saliva and mouthwashes.
The mean and standard deviation of the surface roughness values are shown in Figure 3. Given the initial surface roughness values of the materials, the Wald Chi-squared test showed that ENA had the highest values among the groups (
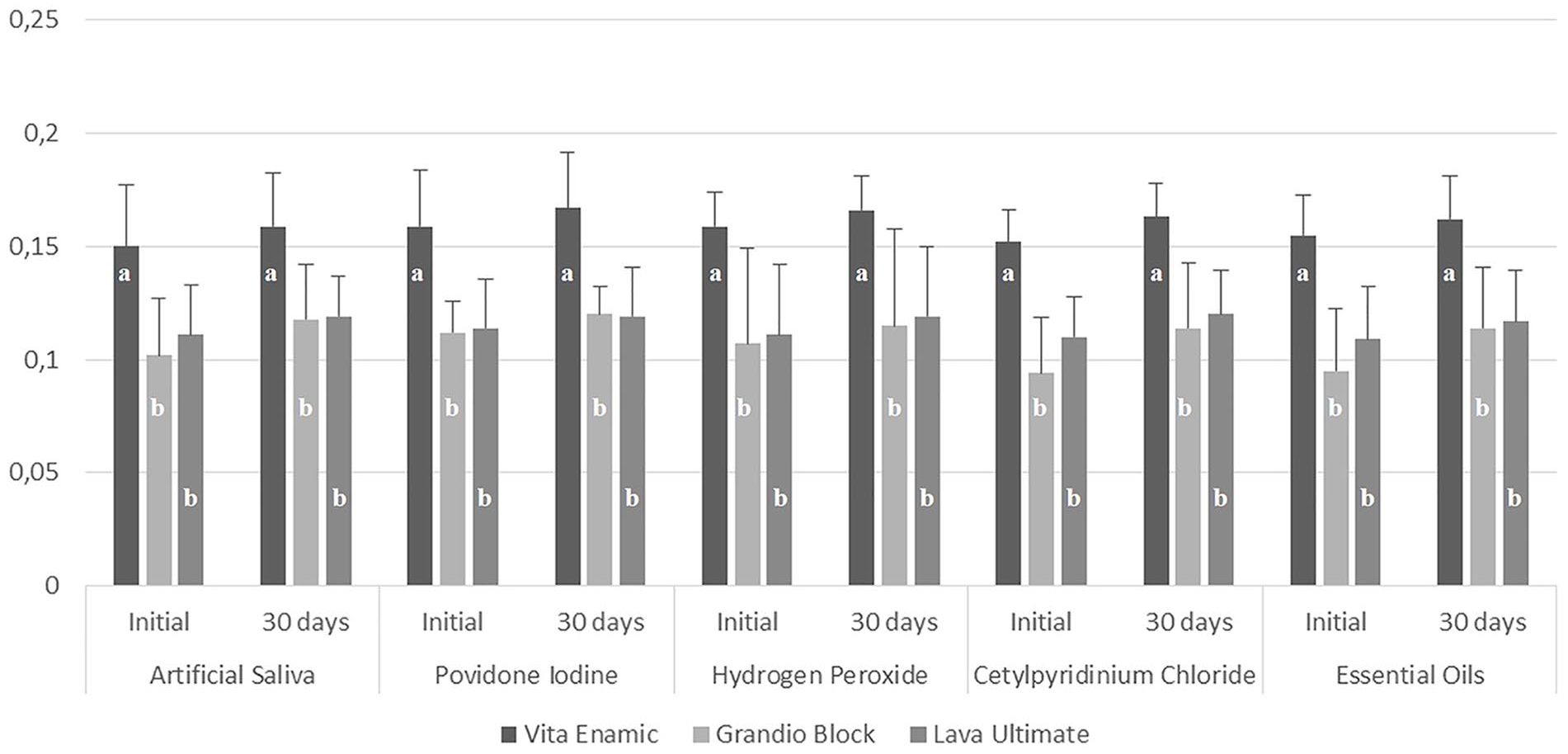
Mean and standard deviation of surface roughness values for different immersion protocols. (a, b) Different lowercase letters indicate significant differences between groups.
Mean percentage microhardness loss values and intergroup comparison for different immersion protocols.
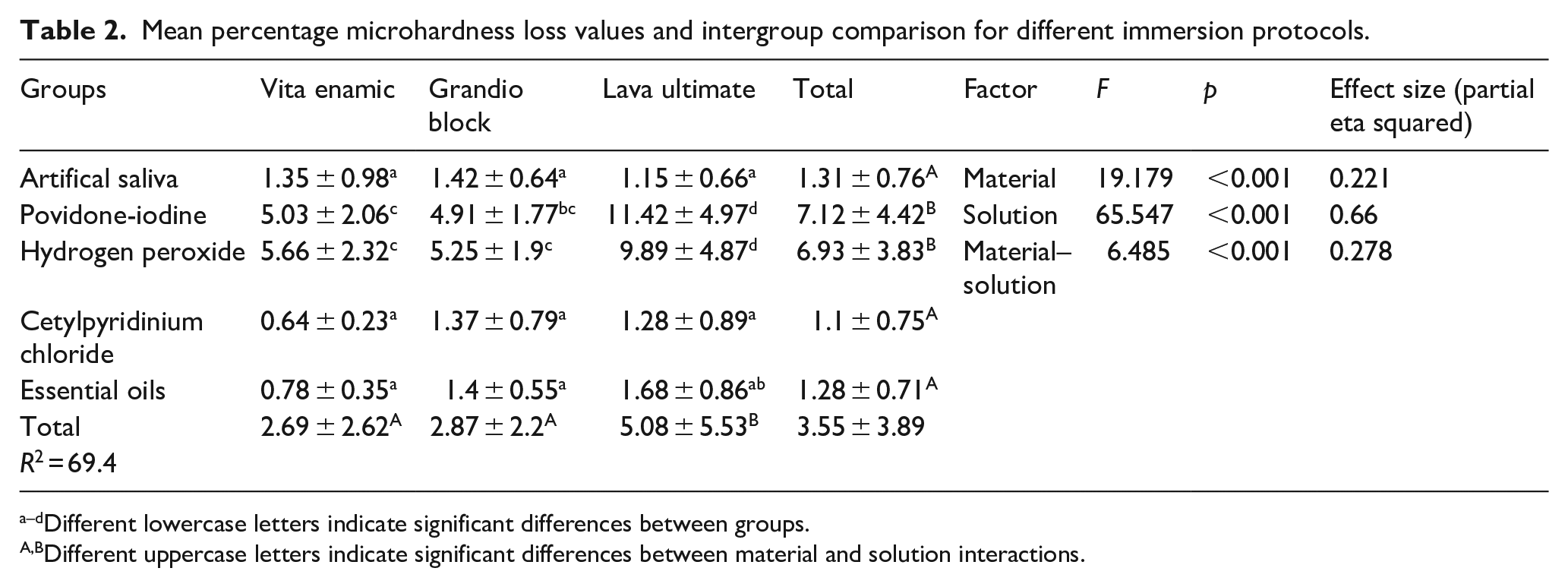
Different lowercase letters indicate significant differences between groups.
Different uppercase letters indicate significant differences between material and solution interactions.
Mean percentage roughness change values and intergroup comparison for different immersion protocols.
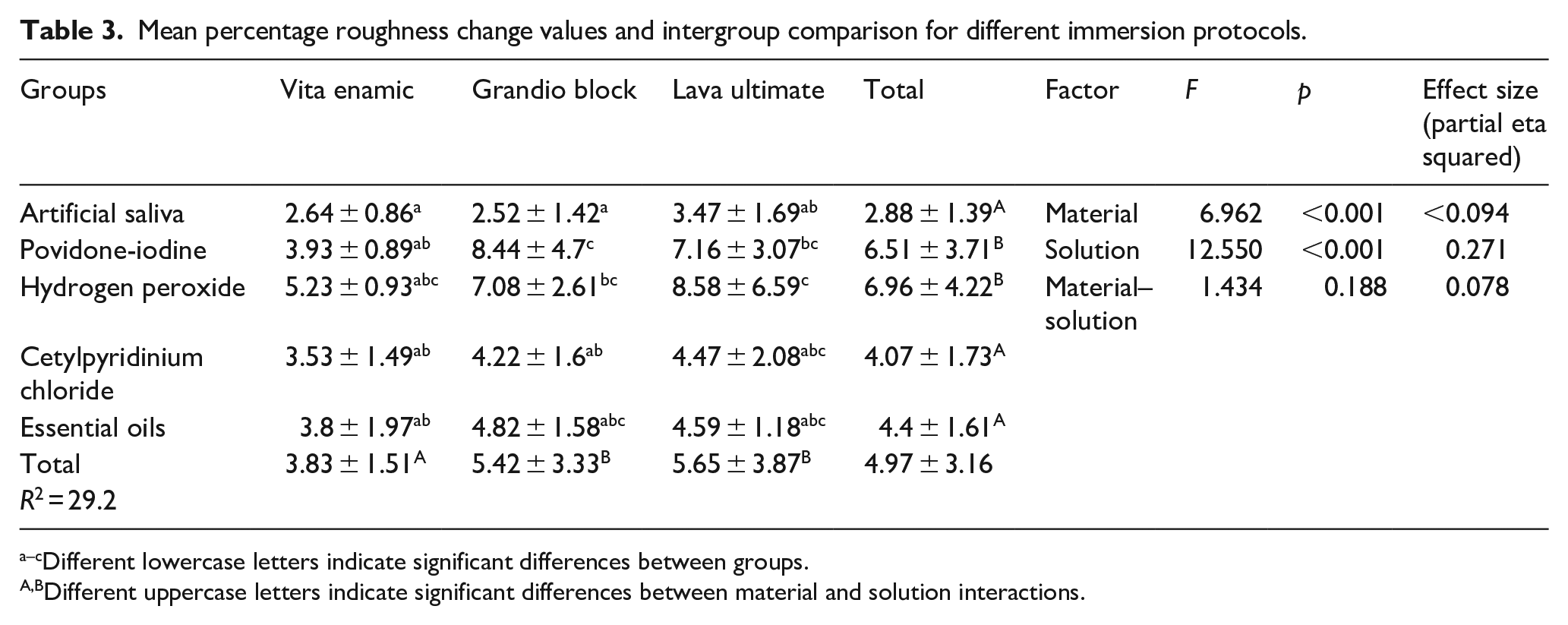
Different lowercase letters indicate significant differences between groups.
Different uppercase letters indicate significant differences between material and solution interactions.
The mean and standard deviation of ΔE00 values and intergroup comparison for different immersion protocols.
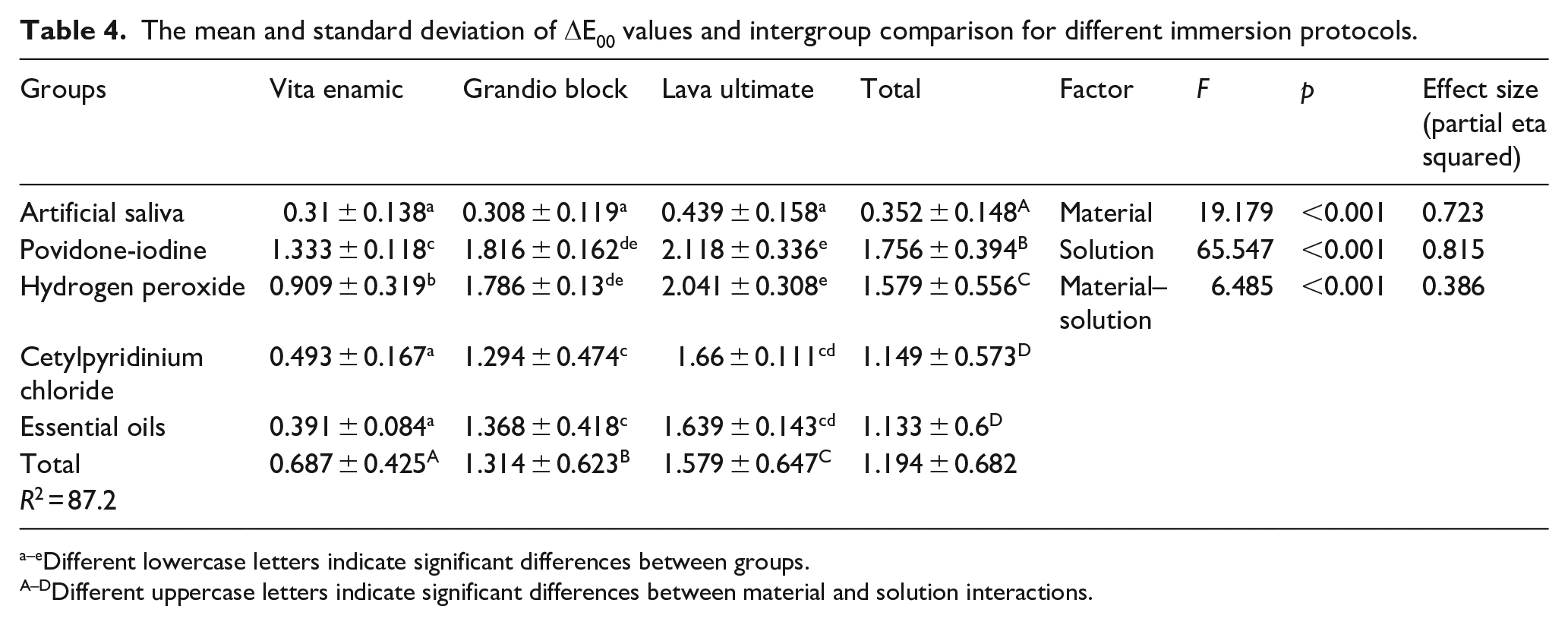
Different lowercase letters indicate significant differences between groups.
Different uppercase letters indicate significant differences between material and solution interactions.
Discussion
One of the objectives of the present study was to evaluate the microhardness of three CAD/CAM restorative materials immersion in various mouthwashes used to prevent COVID-19. Surface microhardness is a crucial factor that ensures the longevity of materials by enabling them to resist forces applied to them or abrasive effects. Maintaining mechanical properties is crucial to prevent micro-fractures on the surface of the resin material, which may lead to pigment retention and secondary caries formation. 30 According to the Vickers hardness values obtained in this study, the GB (RCB) and LU (nanoceramic) composite resin blocks have lower surface hardness than the ENA (PICN) blocks. This result is consistent with other studies indicating that polymer-infiltrated feldspathic ceramic networks have better microhardness properties than CAD/CAM resin composites.31,32
The initial Vickers hardness test results are similar, but not identical, to the inorganic filler ratio ranking of the materials. The results show that although the inorganic filler ratio of GB is the same as that of ENA, its strength is lower than that of ENA. According to the manufacturer, GB is a resin-based nanohybrid composite, while ENA is a polymer-infiltrated feldspathic ceramic network material. 31 This difference may be due to the hardness of the materials being affected by the inorganic filler content. Even if the GB and LU have similar filler content, the resin-based nanofill composite GB (86% wt) showed better results than the nanoceramic composite LU (80% wt). This finding suggests that the surface hardness may be affected by the filler size and the filler content, as well as the inorganic filler rate. LU was the most affected CAD/CAM material regarding surface hardness after mouthwash immersion. The fact that LU has a higher mean percentage of microhardness loss than the other materials may be due to the lower inorganic content of the material.
The findings obtained in this study showed that the microhardness of materials tested was significantly affected only after being immersed in PVP-I, and HP among the mouthwashes. Therefore, the first null hypothesis was partially accepted that the surface hardness of CAD/CAM materials tested would change after immersion in mouthwashes used for COVID-19 prevention.
The pH of the mouthwash is one of the factors that can alter the organic resin matrix of the materials. 33 Cengiz et al. 34 stated that exposure to simulated gastric juice or citric acid negatively affected the polymeric network of various composite resins. In addition, Catelan et al. 35 stated long-term immersion in acidic conditions affected the microhardness properties of resin-based materials. ENA contains a feldspathic ceramic matrix infiltrated with a low-viscosity copolymer, besides GB and LU also have polymeric matrix. Therefore, the microhardness of all materials was significantly affected after immersion in 1% PVP-I (pH = 3.3) and 1.5% HP (pH = 4.12) mouthwashes, which have the lowest pH among the mouthwashes tested was possibly due to the dissolution and softening the feldspathic, and polymeric matrix of the materials by the acidic environment.
Hydrogen peroxide belongs to the oxygen-oxygen single bond family of peroxides and is a strong oxidizing agent due to its chemical composition and unpaired electrons. 17 HP is an unstable molecule that is easily degraded by unstable peroxide bonds. This leads to the degradation of the polymer chain of the composite material, breaking double bonds and resulting in a non-durable surface, which affects the hardness of the material. 36 Thus, the microhardness loss in materials after being immersed in 1.5% HP mouthwash can be attributed to the HP content and the acidity.
Despite the acidic nature of CPC and EO mouthwashes, there was no significant alteration in the microhardness of the materials after immersion in these mouthwashes. These results may be because the acidic pH of these mouthwashes is not at a level that affects the surface degradation of the resin CAD/CAM materials. 37
The initial and the mean percentage of surface roughness change values after being immers in the aforementioned mouthwashes of the CAD/CAM restorative materials were also compared in this study. Surface roughness is an important material parameter because it may lead to plaque accumulation. 38 There was a significant difference in initial surface roughness values between the CAD/CAM materials tested. As the inorganic filler size increases, the surface roughness of the materials also increases. 39 Thus, the higher surface roughness of ENA compared to LU and GB could be attributed to the microstructured SiO2 fillers in ENA and the nanostructured SiO2 and ZrO2 fillers in LU and GB. 32
According to the results of this study, the mouthwashes used to prevent COVID-19 had no significant effect on the surface roughness of the CAD/CAM materials tested. Therefore, our second null hypothesis was rejected. To the best of our knowledge, there is no study evaluating the effect of the mouthwashes used in this study on the surface roughness of CAD/CAM materials. Although not directly comparable because of the evaluation of different materials in parallel with the results of this study, Bayraktar et al. 20 reported that mouthwashes containing 1% povidone-iodine, 1.5% hydrogen peroxide, and 0.075% cetypyridinium hydrochloride did not affect the surface roughness of a nanofil resin composite after 3 weeks immersion. In addition, similar to the results of this study, Urbano et al. 40 reported that mouthwashes containing CPC and HP did not affect the surface roughness of a nanofill resin composite. In contrast to these findings, a study reported that mouthwashes containing EO and CPC affected surface roughness of composite materials but this contradiction may be due to the different methodology of the studies, as da Silva et al. 41 subjected the materials to brushing simulation.
It has been reported that a Ra value of 0.2 μm is the highest critical threshold for bacterial adhesion and that a Ra value of 0.25–0.5 μm can be noticed by the patient’s tongue. 42 According to the results of this study, in all mouthwashes tested, the surface roughness values of all materials remained below these threshold values at initial measuring or after 30 days of immersion.
Regarding the mean percentage of roughness change in materials, the values increased after immersion in all the mouthwashes tested. The acidity of the mouthwash may cause a separation of the polymer matrix of the resin composites by hydrolysis of the monomers and the fillers to extrude from the resin matrix easily. 43 In the present study, the increase in the mean percentage surface roughness of the materials after immersion in 1% PVP-I and 1.5% HP mouthwashes was higher than that after immersion in other mouthwashes, which can be attributed to the acidity of the mouthwashes.
Although the CIELAB formula is commonly used to assess the discoloration of dental materials after immersion in staining solutions, Herrera et al. 44 found that the CIEDE2000 formula, developed by the International Commission on Illumination, showed a better adaptation to assess the discoloration of dental materials in terms of PT and AT compared to the CIELAB formula. 45 In this study, the CIEDE2000 formula was used to determine the color changes in parallel with the literature.
LU showed a higher color change after mouthwash immersion than ENA and GB. The main reason materials show different color changes after being immersed in solutions is the difference in the chemical structure and composition of the materials. 46 Khokhar et al. 47 found that resin-based materials containing urethane dimethacrylate (UDMA) in the resin matrix were more resistant to discoloration than materials containing other types of dimethacrylates. Although all materials tested contain UDMA, LU also contains bisphenol A glycidyl methacrylate (Bis-GMA) monomers, which are more hydrophilic than UDMA or triethylene glycol dimethacrylate (TEGDMA). In addition, GB and ENA are Bis-GMA-free materials. 48 Gajewski et al. 49 stated that Bis-GMA caused the most water absorption compared to UDMA, TEGDMA, and bisphenol A ethoxylate dimethacrylate (BisEMA). Therefore, the reason for more discoloration in LU compared to the other materials tested may be due to the Bis-GMA monomer content.
In dentistry, the 50:50% perceptibility threshold and 50:50% acceptability threshold are 0.8 and 1.8, respectively. 50 Low pH can adversely affect the mechanical and optical properties of resin-based materials through the collapse of the polymer matrix and degradation of the resin surface. 51 This may explain why the discoloration of the tested materials after immersion in PVP-I and HP mouthwashes, which have the lowest pH of the mouthwashes used, was always above the clinical perceptibility threshold of 0.8. These results are consistent with other studies showing that PVP-I and HP mouthwashes were more likely to cause discoloration of the materials.20,52 Therefore, the third null hypothesis was rejected. In the present study, the materials tested revealed color changes below the acceptability threshold of 1.8 after immersing in artificial saliva, and mouthwashes containing CPC and EO. The fact that mouthwashes do not contain alcohol may explain this result.
According to the results of this in vitro study, the use of PVP-I and HP antiviral mouthwashes caused changes in the mechanical and optical properties of the polymer-based composite CAD/CAM materials tested. Clinicians may need to warn their patients who use these mouthwashes about the conditions that may occur in their restorations and suggest not to delay their regular dental check-ups.
Clinically, the effects of mouthwashes on CAD/CAM materials may vary based on several factors, including biofilm, diet, and oral care products, which cannot be replicated in vitro. These factors, either in combination or separately, may affect the mechanical and physical properties of the materials and thus the durability of the restorations by changing the temperature and pH of the environment. To evaluate the effects of these antiviral mouthwashes on CAD/CAM materials as if they were in the oral environment, a brushing simulator that can simulate the daily oral care of patients, a thermal cycle aging procedure that can simulate the temperature change or a chewing simulator that can simulate the abrasion that may occur as a result of the contact of the materials with the teeth can be used in future in vitro studies. Also, further in vivo studies should be performed in the oral cavity, including testing different periods related to different types of dental procedures, to provide results that examine the effect of mouthwash on CAD/CAM materials.
Conclusion
It was concluded within the limitations of this in vitro study that immersion in mouthwashes to prevent COVID-19 transmission increased color change and reduced surface hardness of the materials tested, and did not cause a significant change in the surface roughness of the materials. Among the mouthwashes tested, those containing 1% povidone-iodine and 1.5% hydrogen peroxide were found to greatly affect the hardness, roughness, and color change of CAD/CAM materials.
The CAD/CAM material containing polymer-infiltrated feldspathic ceramic network (ENA) has better mechanical properties, especially in microhardness, than resin CAD/CAM materials and is less affected by mouthwashes used for COVID-19 transmission prevention. LU, which had the lowest filler rate among the materials tested, showed the highest degradation after immersion in mouthwashes used to prevent COVID-19.
Footnotes
Acknowledgements
The authors thank Dr. Seyit Çağlar at the ZBEÜ Department of Metallurgical and Materials Engineering for providing technical support to the surface hardness and roughness tests of the CAD/CAM materials.
Author contributions
AH and EH researched the literature and conceived the study. AH was involved in protocol development, prepared the specimens, and performed the experiments. AH analyzed the data. AH wrote the first draft of the manuscript. AH and EH reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
