Abstract
In this study, we aimed to improve the properties of conventional glass ionomer cement (GIC), including mechanical properties, wear resistance, antibacterial properties and biological activity, by adding fluorinated graphene (FG). Composites of synthesised FG and GIC were examined after being combined at different mass proportions (0, 0.5, 1.0 and 2.0 wt%). The microstructure and morphology of FG prepared via the hydrothermal method was characterised using scanning electron microscopy (SEM) and X-ray photoelectron spectroscopy (XPS). The FG/GIC composite was obtained through the blending method and characterised using SEM. Then, the Vickers microhardness and the wear property of the FG/GIC composite-imitated brushing was measured. The plate count and dilution methods (10-fold) were adopted to investigate the antibacterial properties of FG/GIC by incubating Escherichia coli and Staphylococcus aureus. The biocompatibility of FG/GIC containing the adhesion and cytotoxicity of mouse fibroblast cells (L929) was estimated by the MTT and acridine orange (AO) fluorescent staining. Our results demonstrated that the hardness and abrasive wear resistance of the composites increased, and the microhardness parameter changes exhibited a gradual increase as the concentration continued to increase. A 2.0 wt% FG concentration could effectively improve the bacterial inhibition performance of GIC and was directly proportional to the concentration of FG. The composite materials showed no apparent cytotoxicity on normal L929 cells compared to the control group, and the materials exhibited no cytotoxic effect compared to traditional GIC. Thus, FG/GIC has potential therapeutic value in the field of dental treatment.
Keywords
Introduction
Dental caries, also known as tooth decay, is the most common disease among all oral conditions. Glass ionomer cement (GIC) has been widely used owing to its good chemical bonding, easy filling, low cost, low thermal expansion coefficient, self-adhesion and long-term fluoride-releasing properties.1,2 However, its applications are limited because of its brittleness, poor resistance to crack propagation and poor wear resistance, which may even lead to secondary caries.3,4 In clinical setting, when GIC is applied to class II mesio-occlusal-distal dental restorations, it faces high shrinkage stress influenced by the material composition after contrast elastic behaviour of different materials by 3D-finite element analysis (FEA). 5 Although alternative materials such as block resin and lithium disilicate have limited displacement and can absorb shrinkage stress resembling the natural tooth, 6 they do not possess better antibacterial properties and biocompatibility than fluorinated graphene (FG). 6
FG is widely used in material construction and has advantages such as high temperature and wear resistance, corrosion prevention, low surface energy, high mechanical strength, antibacterial activity and good stability. It is usually added to biobased composites to enhance the hardness of the substrate, wear resistance and antibacterial activity.7–9 Latest research shows that the addition of FG could increase the microhardness and decrease the wear volume of the composites markedly.10,11 With an increase in FG concentration, the coefficient of friction decreased, resulting in better biocompatibility. In addition, Wang et al. 12 found that guanidine-modified graphene (PHGH-G) nanosheets synthesised using FG had better antibacterial performance, while nanosheets applied at a concentration of 20 mg/mL may contribute to a 99.9% viability loss in Escherichia coli. It has become feasible to combine GIC to improve the mechanical and antibacterial properties of the filling material. Sun et al. 10 added FG to GICs to achieve better properties, including mechanical properties, wear resistance and antibacterial properties, of GICs.
Previous studies have primarily focused on antibacterial properties; however, few studies have reported the cellular biocompatibility of FG. According to Wang et al. 13 FG has a promotion effect on the differentiation of MSCs towards neuronal lineages. Another study reported that FG partially showed better antibacterial properties and cytocompatibility with rat bone marrow mesenchymal stem cells (rBMSCs) than pristine graphene and fluorographene. 9 Although the molecular mechanism of cellular coexistence is still not well understood, the cell results mentioned above provide a new direction for the research questions. The screening of appropriate concentrations of our research is based on data and sophisticated experimental methods established by previous studies. In this work, we aimed to examine the main properties of FG/GIC composites besides testing the biological properties of FG/GIC composites.
Methods
Preparation of FG
The graphene oxide (GO) solution (China Nanjing Nano Technology Co., Ltd) was prepared by dispersing 100 mg of GO powder in 100 mL deionised ultrapure water with polytetrafluoroethylene under ultrasonic treatment for 30 min, which resulted in the formation of a homogeneous suspension. The suspension was transferred to a 100 mL Teflon-lined autoclave, filtered with 10 mL hydrofluoric acid and the prepared mixture was placed in an oven at 180°C for 12 h. The reaction mixture was allowed to cool naturally to 24°C–26°C. After filtration, the obtained cake was washed with pure water until the pH reached 7. The final solid product was filtered and air-dried. 14 The surface morphology and chemical composition of FG and GO were observed using SEM and XPS.
Preparation of FG/GIC composites
Composite materials of GIC (purchased from Shanghai Rongxiang Dental Material Company Ltd.) and FG with different mass fractions of 0.5, 1.0 and 2.0 wt% were prepared by mechanical agitation. 10 The powder was weighed and added to the FG ethanol dispersion, ultrasonicated for 1 h and then ground until the ethanol was almost volatilised. The composite powder was mixed with liquid GIC at a solid-to-liquid mass ratio of 2:1. The specimens were fabricated into 1.0 cm × 1.0 cm × 0.1 cm shapes. After 24 h at 37°C at a relative humidity of 100%, the surface was sequentially fine-sanded with #400, #800, #1200, #1500 and #2000 sandpapers before testing. 10 The surface morphologies of the composites were characterised using SEM. The FG/GIC composites were cut into specimens (1.0 cm × 1.0 cm × 0.1 cm), and the sample surfaces were treated via ultraviolet disinfection (Table 1).
Experimental material component.

Mechanical property of FG/GIC composite
Microhardness
The Vickers microhardness of each surface from six randomly selected positions was measured using a micro-Vickers hardness tester (HV-1000; Guanghui Co., Ltd., Dongguan, Guangdong, China). The measurements for each material were averaged. Experiments were carried out using a diamond with a 120° prism shape. Six measurements of each sample were taken, and the average value of three measurements was considered (excluding the highest and lowest values). The corresponding microhardness value can be calculated using the hardness formula below:

where F is 200 g, load time is 15 s and d2 is the product of the indentation diagonal lengths d1, d2. 15
Wear test
Mechanical abrasion was applied to the same specimens using an electric toothbrush (D12.013, Oral B, Germany) with the speed set at 7600 rpm for 12 min, simulating 3-year-brush abrasion of the composite. The SEM images were evaluated to determine the wear resistance of the FG/GIC composite.
Biocompatibility of FG/GIC
Cell spreading and adhesion morphology
Briefly, L-929 cells in the logarithmic phase were digested with 0.25% trypsin to a single-cell suspension (1.0 × 104/mL, 0.1 mL making cell climbing). The substrate material was placed on a 24-well plate, and the above-mentioned cell suspension with 1 mL culture medium (10% foetal bovine serum, FBS) was added. After attachment for 4 h, the original medium was replaced with complete medium containing the corresponding concentration of the FG leaching solution. After 1, 2 and 4 days, the cells were washed three times with phosphate-buffered saline and then fixed in 95% alcohol for 15 min. The cells were stained with acridine orange (AO, Polyscience Inc.) for 1 min in the dark. Finally, the cells were observed under a fluorescence microscope (IX-71 Olympus, Japan). Each group was set in five parallel wells. The experiments were performed three times.
MTT assay
The specimens were soaked in 10 mL of low-glucose Dulbecco’s Modified Eagle’s medium (DMEM) cell culture for 1 week, and the product obtained from the liquid was designated as the leaching solution. The experiments were carried out at the same 24–26°C and leaching solution to preclude the influence of temperature and pH. The leaching solution was termed the experimental group and the control group contained no FG in the solution. The leaching solution was placed on a 24-well plate, and 1 mL cell suspension (1.0 × 104/mL) was added to each well and cultured for another 4 h. After incubation, the medium was replaced as mentioned above. After 1, 2 and 4 days, 100 μL of 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) was added to each well. After 4 h, the medium was removed, and 750 µL of dimethylsulfoxide (DMSO) was added. The plates were placed on a shaker for 10 min. Subsequently, 150 μL DMSO was added to a 96-well plate, and the absorbance of each well was read at 490 nm under iMark microplate reader (BioRad, America). Each experiment was performed three times, with at least three replicates for each concentration.
Antibacterial property
The strains of bacteria used in the present study to explore the antibacterial properties of materials were Staphylococcus aureus (ATCC 2592, JL7010) and Escherichia coli (ATCC8739, JL7085) by using 10-fold dilution and colony count method. The bacterial cultures (E. coli and S. aureus) were diluted 10-fold in 0.8% sterile aqueous solution of sodium chloride. The leaching solutions were prepared by placing 0.5, 1.0 and 2.0 wt% FG/GIC in bacterial suspension at room temperature for 30 min. The mixture was then spread onto nutrient agar plates, and each experiment was performed three times at 37°C for 12 h. The number of colonies was counted, and the average values were calculated. 16
Statistical analysis
Statistical analysis was performed using SPSS software (IBM, version 22.0). Values are expressed as the mean ± standard error of the mean (SEM). Differences between the mean values for the individual groups were assessed using one-way analysis of variance. Statistical significance was set at p < 0.05, unless otherwise stated.
Results
Characterisation of GO, FG and FG/GIC
FG was prepared using a hydrothermal method with GO. Under SEM, it can be seen that both FG and GO showed a transparent and thin structure. Compared to the single-layer sheet structure of GO, the edge of FG resembled a stair-like structure, whereas the micro-rough surface lost its metallic lustre. Along with the larger-sized (10 μm) lamellar, some small-sized (3 μm) lamellar that formed following the stripping process upon fluorination could be observed (Figure 1(a.i–a.iii and a.vi)). These structures with some folded parts existed owing to the reduced surface free energy stability during the reaction dispersion. XPS analysis of the surface elemental composition of FG revealed three major elements, namely fluorine (F), oxygen (O) and carbon (C), while that of GO had C and O elements. Two characteristic XPS peaks of FG also appeared at higher binding energies of 284.6 and 285.8 eV, corresponding to C-C and C-F, respectively. The binding energies of FG were lower than those of GO. Subsequently, the distance between the covalent C-F bond (687.6 eV) and the half-ionisation C-F bond (686.5 eV) decreased. This indicates that F successfully grafted atoms on GO and replaced O atoms through fluorination, indicating the formation of FG (Figure 1(b)). The surface morphology of the composites with 2.0 wt% FG, characterised using SEM, was compared with that of pure GIC (Figure 1(a.iv and a.v)).

Structural characterisation of GO, FG, FG/GIC and GIC: (a) SEM characterisation of GO (a.i and a.ii) and FG (a.iii and a.vi); SEM characterisation of 2.0 wt% FG/GIC (a.v) and comparison with GIC (a.vi); (b) survey C1s XPS spectra of FG (b.i–b.iii)) and GO (b.iv and b.v).
Mechanical properties
Microhardness
The 2.0 wt% FG showed the highest microhardness, followed by the 1.0, 0.5 wt% and the control group. The control group without FG had a microhardness value of 70 N/mm2, which increased to 93.9 N/mm2 when FG was doped with 0.5 wt% FG. The hardness value increased to 105.3 N/mm2 (p < 0.05) when 1.0 wt% FG was doped with the 2.0 wt% group, which was significantly higher than that of the control group (127.8 N/mm2) (p < 0.05). In addition, the microhardness parameter changes exhibited a gradual increasing trend as the concentration continued to increase (Figure 2(a)).
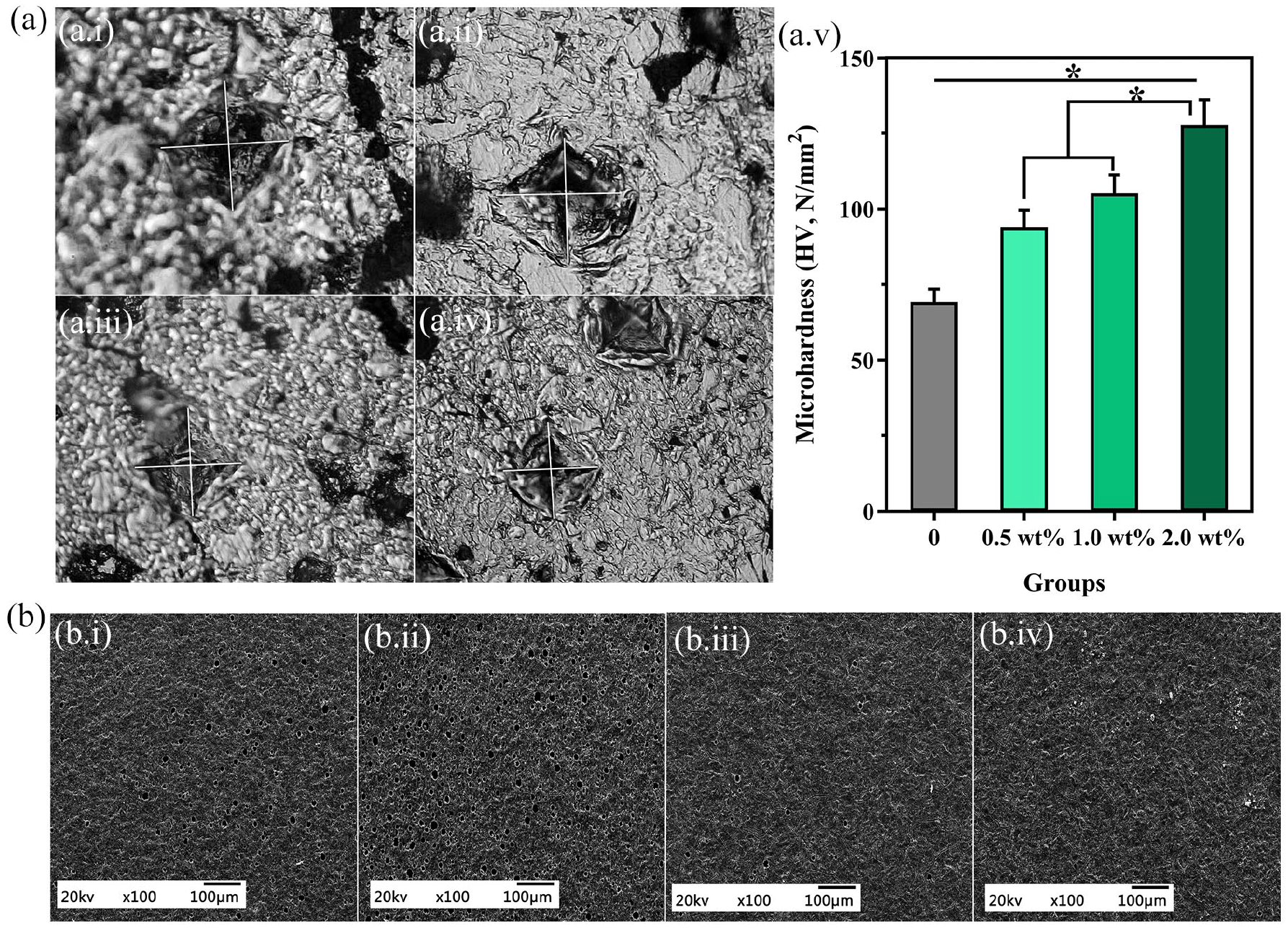
Microhardness and wear resistance of FG/GIC composites: (a) diamond shaped (~40 µm) hardness image (a.i–a.iv), and average values of microhardness (a.v); and (b) SEM image representing the wear resistance of different concentrations of FG. (b.i) 0 wt%, (b.ii) 0.5 wt%, (b.iii) 1.0 wt% and (b.iv) 2.0 wt%.
Friction and wear experiments
Figure 2(b) showed the rough surface morphology due to attrition wear observed at the electric toothbrush speed. The most obvious wear scar was found in the control group with rough mottled, even grinding, pits or cracks (Figure 2(b.i)). The wear crack diameter of the 0.5 wt% FG narrowed slightly (Figure 2(b.ii)). Surface grinding pits of 1.0 wt% FG were still present, although the crack diameter was substantially reduced (Figure 2(b.iii)). However, the cracks and pits were unremarkable and not obvious in the 2.0 wt% FG group. Usually, performance anti-friction properties are achieved by using different types and smaller size of fillers. 17 In the experimental group, when the gap inside the GIC particles was filled with FG, the surface asperities disappeared and the particles became smooth. As the FG ratio increased, the wear resistance of the FG/GIC composites increased significantly.
Antimicrobial properties
Compared to the control group, a significantly increased FG mass ratio was proportional to the bacteriostatic effect. In E. coli, the bacteriostatic rates were 8.33%, 23.33%, 66.67% and 90.00% from low to high concentrations, respectively (Figure 3(a)). Meanwhile, the inhibition rates of S. aureus were 8.89%, 32.65%, 56.12% and 83.67% at different concentrations, respectively (Figure 3(b)). Significant differences were observed between the experimental and control groups in terms of bacteriostatic activity (p < 0.05; Figure 3(c)). In special dentistry GIC, fluoride inhibits demineralisation, enhances remineralisation and inhibits cariogenic bacteria.18–20 A higher FG mass ratio has stronger antibacterial effects, and the local anti-caries effect was also enhanced.

Antimicrobial performance of FG/GIC composites. Escherichia coli (a) and Staphylococcus aureus (b) plate count chart. FG/GIC 0, 0.5, 1.0 and 2.0 wt% composites corresponding to (b.i–b.iv) in each series, respectively. (c) The colony-forming unit of E. coli and S. aureus, and the antibacterial rate of test groups.
Cytocompatibility
Morphological and adhesion observation of L929 cells
A day after the inoculation of L929 cells, more adherent cells were observed (Figure 4(a)). The cells entered the log phase at 24–96 h after inoculation and gradually increased in number. The cells exhibited a characteristic spindle shape and star shape. No significant changes were observed at any of the tested concentrations of infiltrates in the cell morphology. The corresponding proliferation rate can be calculated using the Proliferation Index (PI) formula as follows: PI (%) =

Cytocompatibility test of the composites: (a) MTT fluorescence result of L929 cells after culturing in the leaching liquid of FG/GIC for 1, 2 and 4 days and (b) cytotoxicity of FG/GIC composites determined using MTT assay after culturing for 1, 2 and 4 days.
Cell viability of FG/GIC composites (%) (x‾ ± s).

There was no significant difference between the 0.5, 1.0, 2.0 wt% groups, and the FG-free group, p > 0.05.
Discussion
In this study, we aimed to improve the properties of conventional GIC by adding FG and found that 2 wt% FG concentration effectively improved the bacterial inhibition performance of GIC, and the composite materials showed no apparent cytotoxicity on normal L929 cells.
Preparation of FG is generally divided into two main categories: chemical reactions and physical stripping.22–25 Traditional chemical reactions usually require costly reagents and are toxic, and the method and equipment are very sensitive. For the physical method, FG is separated from an ionic liquid solution using an expensive organic solvent.13,18,20,26 In comparison to these two methods, the hydrothermal process, which involves a special kind of chemical reaction without the need for any expensive ionic liquid media, shows improved safety and requires a relatively simple operation and low energy consumption. FG was obtained as a white powder. Adding FG to GIC makes FG/GIC yellowish, thus enhancing aesthetics.
In addition, conventional GIC can be further developed based on the unique properties of graphene nanomaterials, such as crystal structure and excellent physical properties.19,27,28 The addition of more FG into GIC results in a strong interface bonding between the FG and GIC matrix because the number of particles impacting the surface decreases the leading interreaction of acrylic acid. A recent review reported the characteristics of cracks during the preparation of GIC. 29 Previous studies have indicated that when some modified materials are added to GIC, the nature of the base material will not change.10,29,30 Our results are consistent with these findings. FG addition could not markedly diminish the cracks, although the crack of FG/GIC is less than that of GIC. Owing to the gap-filling effect of the particles, the high proportion of FG enhanced the microhardness of the composites.31–33 Furthermore, the fluorine content in FG significantly affects the bacteriostatic performance and is released through porosity or cracks. 34 F ions inhibits the growth of bacterial plaque formation 35 and oral Streptococcus mutans. 36 Fluorapatite is generated afterward and displays greater biocompatibility than pure hydroxyapatite. Introducing fluoride ions, C-F bonds and surface structure may improve the biocompatibility and antibacterial properties of FG, which are also related to its microstructure.9,24,37 As reported, graphene exhibits excellent antimicrobial properties because of cell membrane mechanical damage.31,32,38 Fluorination process can help determine the antibacterial activities of composites against E. coli (gram-negative) bacteria to some extent. 9 In addition, the unique two-dimensional structure of graphene facilitates strong dispersion interactions between bacterial membrane lipid molecules and graphene, resulting in bilayer lipid instability. 39 Meanwhile, previous studies have demonstrated that FG preserved its structure of graphene.9,40,41 Another potential explanation for the excellent antibacterial efficacy of FG/GIC resulting from the combined action of FG surface structure and F− is the damaged bacterial envelope caused by a surface structure that is more vulnerable to F ions. 9 Lastly, the biocompatibility of FG/GIC containing the adhesion and cytotoxicity of L929 cells was estimated using MTT and AO fluorescent staining. Research has shown that the strong polarity of C-F bonds indirectly appears to enhance cell stability.13,42–44 The available literature reports that it is easier to adsorb proteins driven on the surface of graphene owing to strong π-π stacking interactions with aromatic amino acid residues of proteins and graphene.9,45,46 Fibrinogen is associated with coagulation, and its distribution can serve as a predictor of platelet morphology/activation. 47 Massa et al. 47 illustrated the relationship between fibrinogen surface distribution and platelet adhesion. Wang et al. 13 revealed that the C-bonds in FG have good biocompatibility and low cytotoxicity towards early cell adhesion. All these findings are consistent with our experimental results.
However, there are some limitations to this study. First, the number of analysed groups was small. In this regard, additional animal model experiments are required for verification. Second, the precise friction cannot be guaranteed. Lastly, further innovation and randomised controlled clinical trials are required to elucidate the potential benefits.
Conclusion
In summary, we successfully prepared FG through a hydrothermal process and achieved a significant ratio of FG/GIC. The 2.0 wt% group exhibited extremely high hardness, superior wear resistance and enhanced bacteriostatic activity. No obvious cytotoxicity against L929 cells was detected in any of the groups. Thus, our study shows that FG/GIC materials, with a novel promising role, can replace traditional GIC in therapy of tooth decay.
Footnotes
Acknowledgements
We would like to thank the funder for his assistance and guidance in this research.
Author Note
Kailiang Zhang and Baoping Zhang are now affiliated to Gansu Provincial Key Laboratory of Maxillofacial Reconstruction and Intelligent Manufacturing, Lanzhou University, Lanzhou, PR China.
Contributorship
Baoping Zhang and Ruimin Liu researched literature and conceived the study. Errui Wang, Yumeng Guo and Qiaozhen Zhou were involved in protocol development and data analysis. Errui Wang, Yayuan Zheng and Junkai Zhai wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Lanzhou University’s ‘Double First Class’ Guided Project-Innovative Talents Training Project (316-561119101); Natural Science Foundation of Gansu Province (20JR10RA591; 20JR5RA258; 20JR5RA150); Fundamental Research Funds for the Central Universities (lzujbky-2020-cd03); General project of National Scientific Research Program of Gansu Province Hospital (19SYPYB-27).
Guarantor
Baoping Zhang.
