Abstract
The aim of the present study was to evaluate the shear bond strength (SBS) of bioactive cement (BAC) in comparison to conventional dual cure resin-modified glass ionomer cement (RMGIC) with Er,Cr:YSGG laser (ECL) dentin photo biomodulation.
Methods:
One hundred and twenty extracted human molars were allocated in eight groups (n = 15) based on surface conditioning and cement type. Specimens of groups 2 and 6 were conditioned with ECL whereas, groups 3 and 7 were treated with ECL + ethylenediamine tetra acetic acid (EDTA). Specimens in groups 4 and 8 were surface conditioned by ECL + EDTA + Tetric-N-Bond, and groups 1 and 5 were considered as control (non-surface treated). Cement build-ups were performed on the surface-treated dentin with BAC (groups 1–4) and RMGIC (groups 5–8). A universal testing machine was used to measure the SBS and the mode of failure was evaluated using a stereomicroscope. Statistical analysis was performed using an analysis of variance and Tukey’s post hoc test, at a significance level of p < 0.001.
Results:
The highest SBS values were observed in group 8, ECL + EDTA + Tetric-N-Bond + RMGIC (21.54 ± 3.524 MPa) and the lowest SBS values were displayed by group 1, with no surface treatment and BAC application (11.99 ± 0.821 MPa). The majority of failures were found to be mixed in lased dentin-treated dentin surfaces. BAC when bonded to dentin surfaces conditioned with ECL showed lower SBS in comparison to RMGIC.
Conclusion:
Conditioning of dentin with ECL and a bonding agent (Tetric-N-bond) improved bond strength scores for BAC and RMGIC. Use of EDTA improved bond strength values when bonded to BAC and RMGIC; however, this improvement was not statistically significant.
Introduction
One of the major challenges in dentistry is to find an ideal cementation material that has mechanical properties similar to the tooth structure, with no oral degradation, good dentinal adhesion, and low microleakage. 1 The clinical success of direct and indirect restorations requires a durable adhesive bond between the tooth dentin and adhesive restorative material. 2 Continued research has led to the formulation of resin-modified glass ionomer cement (RMGIC). RMGIC consists of 80% fluoro-aluminosilicate and 20% meth-acrylates, which improves the wettability of dentin and the adhesive bond strength. 3 RMGIC bonds to dentin both mechanically as well as chemically and has improved aesthetics, command polymerization, ease of handling, enhanced bond strength, and lower microleakage scores compared to glass ionomer cements (GICs).4,5
Innovation in cementation material development has introduced a new bioactive resin cement with good dimensional stability, aesthetics, antimicrobial properties, marginal fit, and bond strength.6,7 Bioactive cement (BAC) has the capacity to remineralize the tooth–restorative interface by exchange of fluoride, calcium, and phosphate ions with saliva and the tooth structure. The material stimulates mineral apatite crystal formation due to the presence of a bioactive glass (BAG) filler and bioactive resin matrix.6,8
Dentin surface treatment is highly critical for successful bond integrity between dentin and RMGIC due to complexity of the dentinal structure. 9 There are variety of methods that have been introduced to condition dentin prior to adhesive bonding to RMGIC. These conditioning methods include the use of phosphoric acid, ethylenediamine tetra acetic acid (EDTA), polyacrylic acid (PAA), and lasers.10,11 Recently, the use of laser application as a photo bio-modulating agent in treating dental and medical conditions has gained mounting interest.12–14 Laser therapy in the form of erbium, chromium-doped yttrium, scandium, gallium, and garnet (Er,Cr:YSGG laser (ECL)) causes the hydration of water and other organic components from the dentinal tissue, resulting in internal pressure causing ablation and destruction of surface before the melting point is reached. 15 ECL conditioning of dentin increases the concentration of calcium and phosphate ions, which form a more stable compound that is resistant to acid attack. 16
To our knowledge from the indexed literature, there is limited evidence on the bond strength of BAC and RMGIC to lased dentine using ECL. Moreover, evidence with regards to the ideal dentin surface treatment in the presence of other conditioning regimes (EDTA, PAA) is still debatable.4,17 Furthermore, the use of BAC on lased dentine with different conditioning regimes is still uncharted.15,18 Therefore, it is hypothesized that dentin conditioned with ECL and bonded to BAC will exhibit comparable shear bond strength (SBS) results compared to bonding to RMGIC. The aim of the present study was to investigate the SBS and the mode of failure of BAC to RMGIC to lased ECL dentin.
Methods
The present in vitro study was permitted by the ethical committee of King Saud University with ethical approval project no. E-18-3345. The study followed the Check List for Reporting In Vitro Study (CRIS) guidelines.
One hundred and twenty extracted non-carious, non-fractured, restoration-free human molars were collected and stored in 0.01% thymol solution for one week. Calculus, plaque, tissue remnants, and other organic debris were removed prior to tooth preparation with the help of a periodontal scaler (Sonic flex 2000, Biberach, Germany). The specimens were transferred to distilled water at 4°C until preparation. All samples were mounted vertically in acrylic resin (Meliodent, Kulzer, Hanau, Germany) within segments of polyvinyl pipe (4 mm radius) equal to the level of the cemento-enamel junction (CEJ), revealing only the clinical crown. To homogenize dentinal depth and to expose fresh dentinal tubules, the buccal surface of all molars were ground to a depth of 2 mm with an area of 3 mm using an Isomet saw (Buehler, Illinois, USA). The surfaces were polished with 300–500 grit silicon carbide paper (Buehler, Illinois, USA) on a rotary polishing machine (Aropol 2V, Arotec) (250 rpm) under water irrigation for 20 s. Specimens were randomly allocated into eight groups (n = 15) according to the type of surface conditioning (Table 1).
Material used in the study.

RMGIC: resin-modified glass ionomer cement; UDMA: Urethane Dimethacrylate; HEMA: 2-hydroxyethyl methacrylate; BISGMA: bisphenol A-glycidyl methacrylate.
Based on the type of cement, samples from groups 1–4 (n = 15) were bonded to BAC. BAC (Activa, Pulpdent Cooperation, Watertown, Massachusetts, USA) was dispensed in a polyether rubber mold with standardized dimensions (3 mm diameter and 3 mm height) applied on the dentin samples using a cement plunger and the samples were light cured for 20 s. Similarly, specimens from groups 5–8 (n = 15) were bonded to GC Fuji Plus (GC, Corp, Singapore) RMGIC. RMGIC was built-up using a standardized rubber mold and the dispensing method, as explained earlier. The complete tooth and cement build-up assembly is shown in Figure 1.
Specimen embedded in orthodontic resin with build-up using restorative material.
All the specimens were kept in an incubator (Memmert Universal Oven, Germany) at 37°C in a humid environment for 2 days. All specimens were assessed for SBS testing using a universal testing machine (Instron 8500 Plus, Canton). The samples were mounted in a metallic mold and were exposed to increasing load at a crosshead speed of 1 ml/min at the dentine restorative material interface. The shear strength that separated the test material was calculated. Similarly, 10 samples from each group were assessed for modes of failure (40× magnification) using a stereomicroscope (SZX7, Olympus, Hamburg, Germany) and classified into cohesive, adhesive, and admixed failure.
Data obtained through bond strength testing was tabulated using a statistical program for social science (SPSS version 21, Inc., Chicago, US). The normality of the data obtained was assessed using the Kolmogorov–Smirnov test. Means and standard deviations (SDs) were compared using an analysis of variance (ANOVA) and individual groups were compared using Tukey’s post hoc test, at a significance level of p < 0.05.
Results
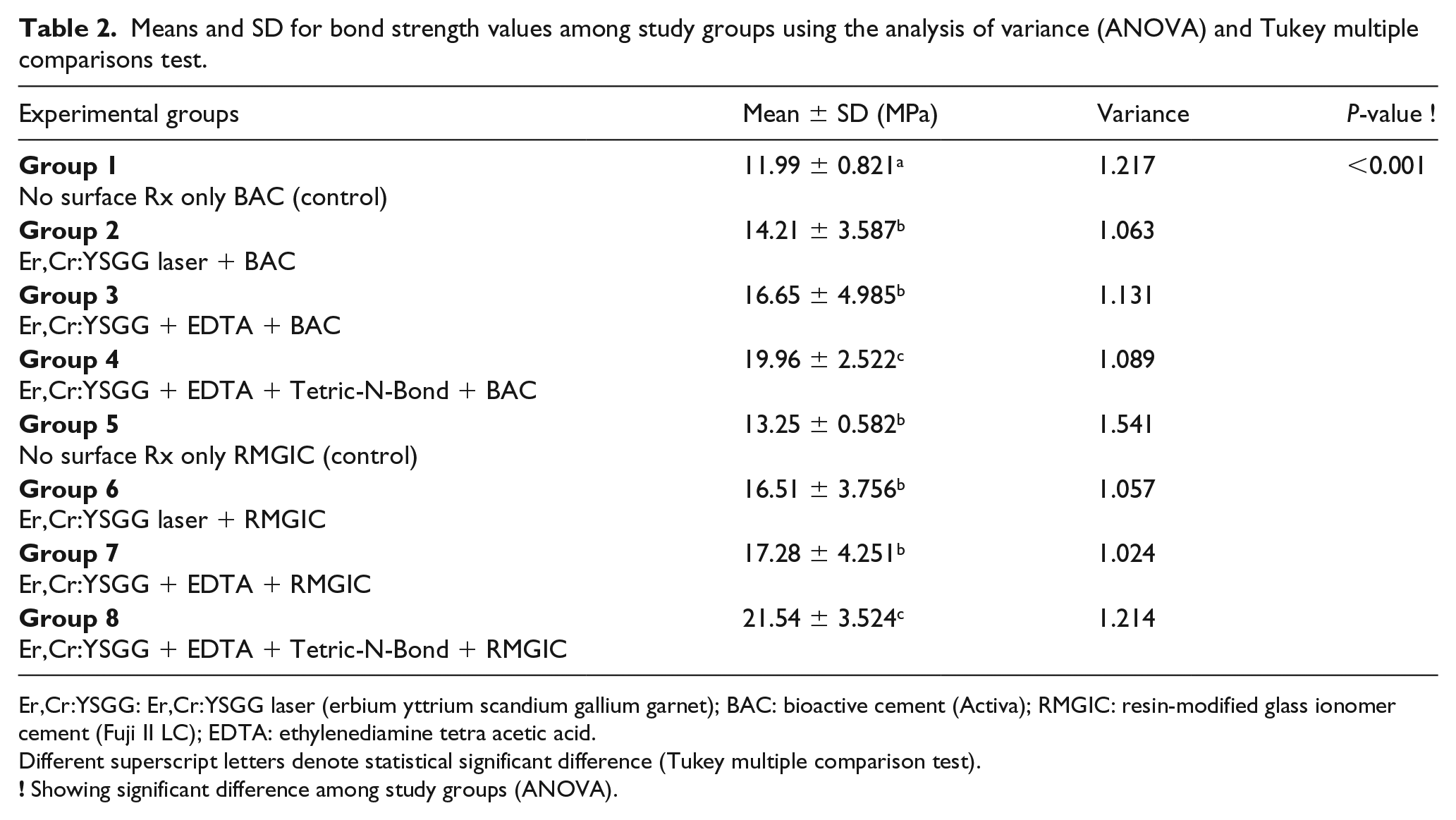
Table 2 presents mean and SD values for SBS within each experimental study group. In groups bonded with BAC, the highest bond strength was observed in group 4 (19.96 ± 2.522). Similarly, the lowest bond strength was exhibited in group 1 (11.99 ± 0.821). Moreover, the SBS values of group 2 (14.21 ± 3.587) were comparable to those of group 3 (16.65 ± 4.985) (p > 0.05).
Means and SD for bond strength values among study groups using the analysis of variance (ANOVA) and Tukey multiple comparisons test.
Er,Cr:YSGG: Er,Cr:YSGG laser (erbium yttrium scandium gallium garnet); BAC: bioactive cement (Activa); RMGIC: resin-modified glass ionomer cement (Fuji II LC); EDTA: ethylenediamine tetra acetic acid.
Different superscript letters denote statistical significant difference (Tukey multiple comparison test).
In experimental groups bonded with RMGIC, the maximum bond strength was displayed by group 8 (21.54 ± 3.524), whereas the minimum bond strength value was presented by group 5 (13.25 ± 0.582). SBS values in groups 5 (13.25 ± 0.582), 6 (16.51 ± 3.756), and 7 (17.28 ± 4.251) were comparable (p > 0.05).
Among all experimental groups, groups 2 (14.21 ± 3.587), 3 (16.65 ± 4.985), 5 (13.25 ± 0.582), 6 (16.51 ± 3.756), and 7 (17.28 ± 4.251) were comparable (p > 0.05). Furthermore, groups 4 (19.96 ± 2.522) and 8 (21.54 ± 3.524) were comparable (p > 0.05). For bond strength values, the ANOVA showed a significant difference among the study groups (p < 0.001) (Figure 2).
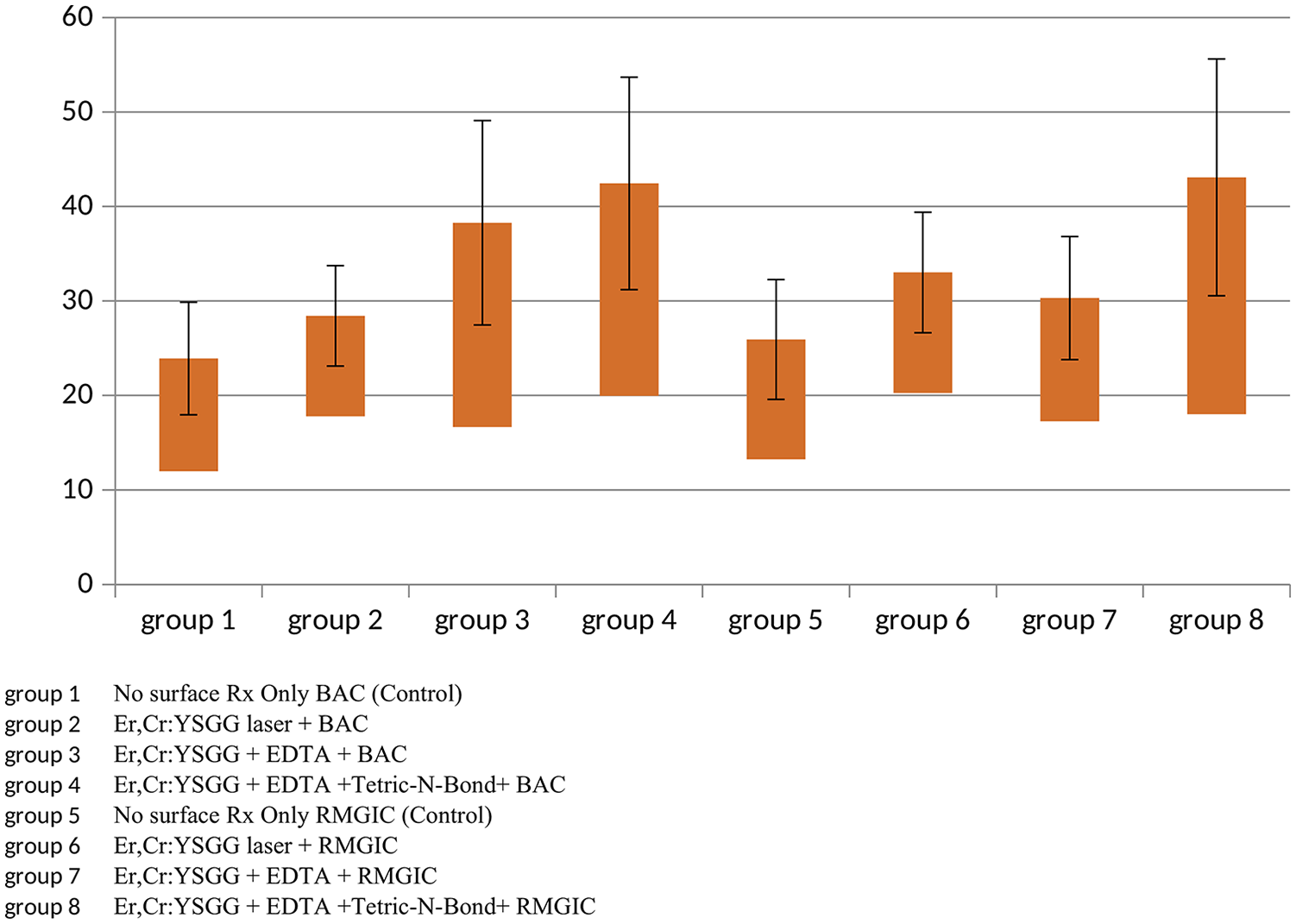
Box and Whisker plot demonstrating shear bond strength among experimental groups. BAC: bioactive cement; EDTA: ethylenediamine tetra acetic acid; RMGIC: resin-modified glass ionomer cement.
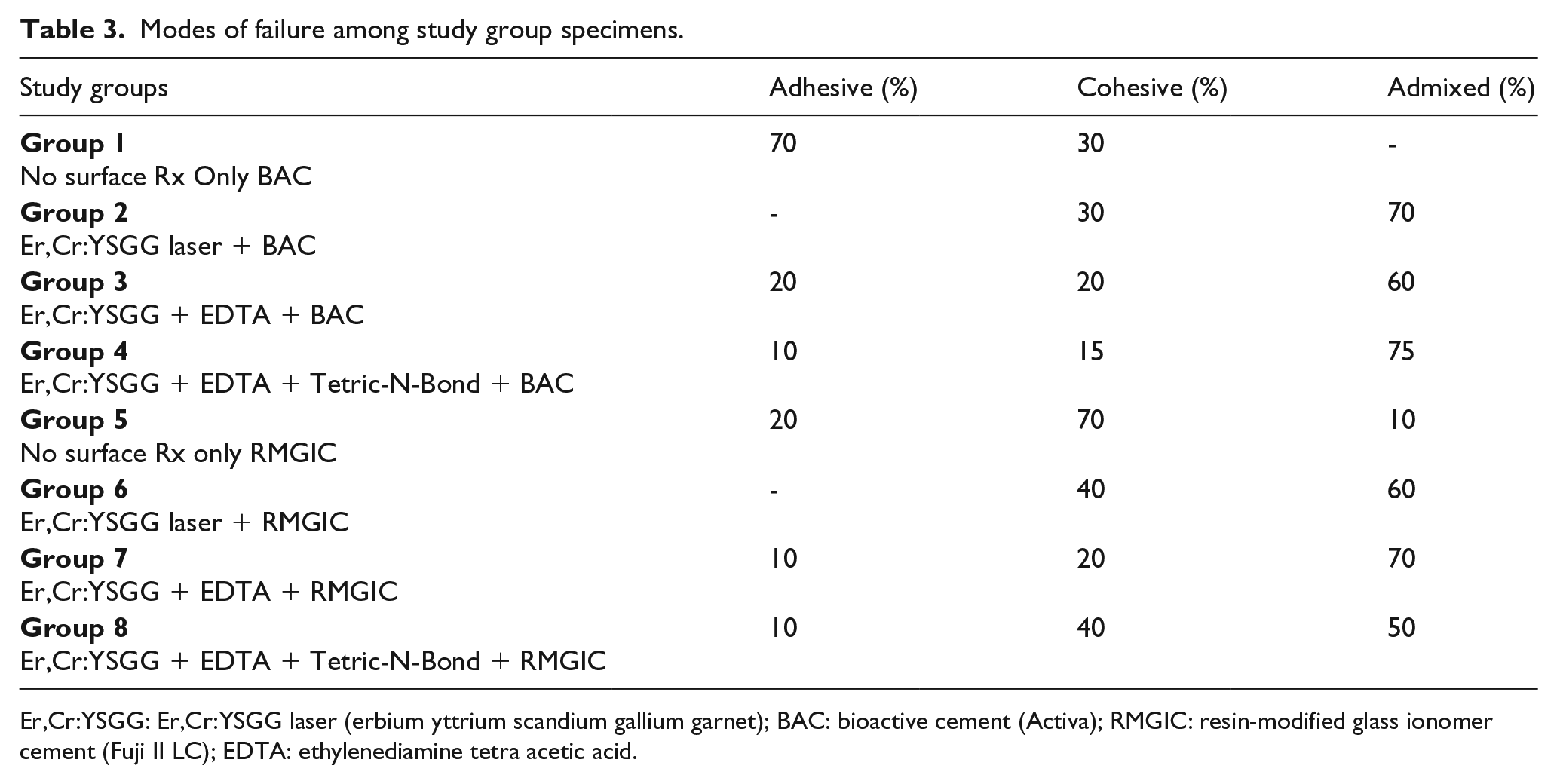
Table 3 presents the mode of failure among experimental groups. The majority of the failures in lased dentin were admixed, whereas group 1 displayed a high incidence of adhesive failure. Moreover, cohesive failure was more dominant in group 5 (Figure 3).
Modes of failure among study group specimens.
Er,Cr:YSGG: Er,Cr:YSGG laser (erbium yttrium scandium gallium garnet); BAC: bioactive cement (Activa); RMGIC: resin-modified glass ionomer cement (Fuji II LC); EDTA: ethylenediamine tetra acetic acid.
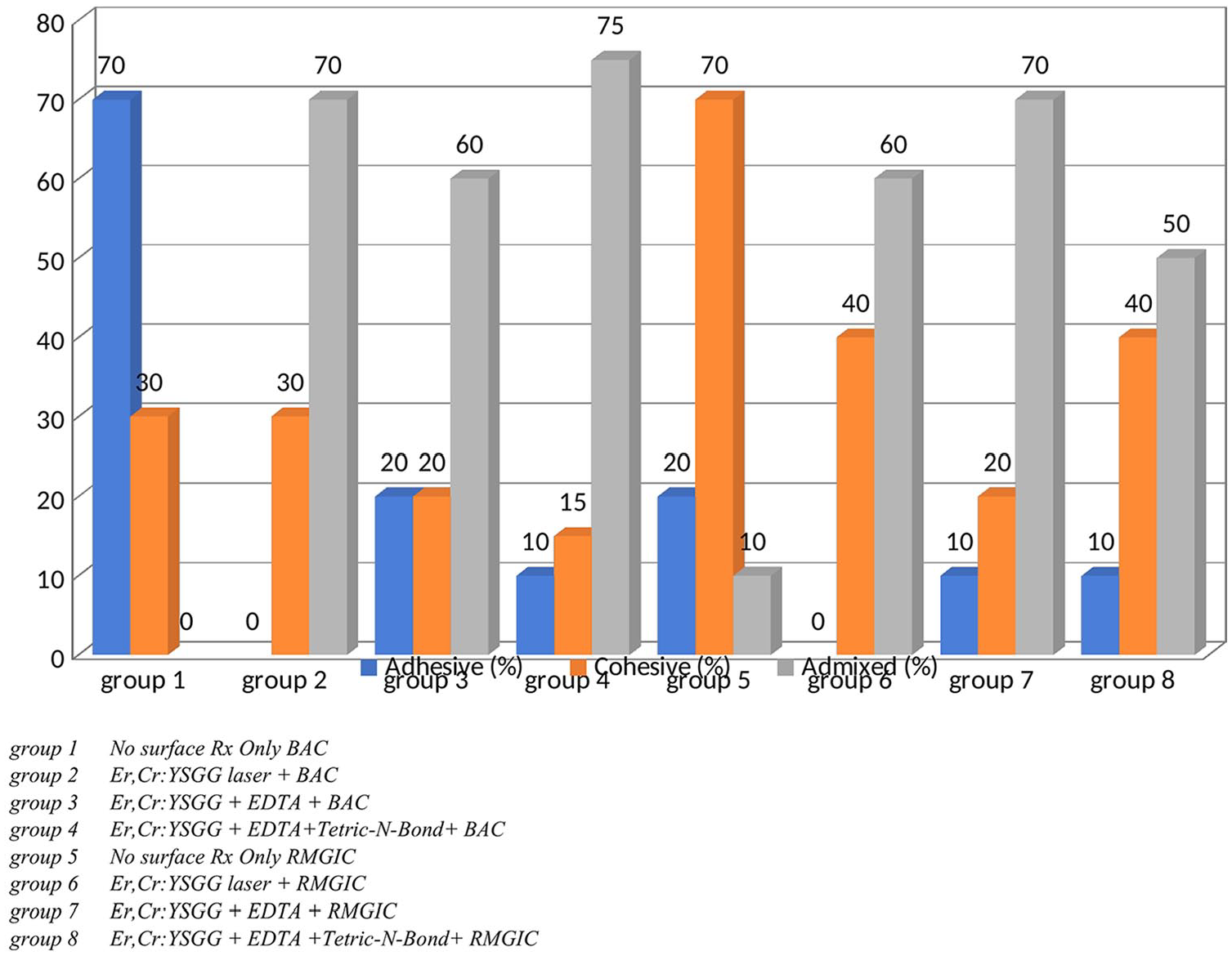
Bar chart presenting the mode of failure among different groups. BAC: bioactive cement; EDTA: ethylenediamine tetra acetic acid; RMGIC: resin-modified glass ionomer cement.
Discussion
The present study was based on the hypothesis that dentin conditioned with ECL and other surface treatment and bonded to BAC will exhibit comparable SBS results to RMGIC. Intriguingly, the hypothesis was rejected, as RMGIC showed better bond integrity when dentin was conditioned with ECL and other conditioning regimes. In the present study, bond strength was evaluated using a universal testing machine, as the method is standardized, homogenized, and consistent with previous studies.19,20 Secondly, the test is useful for RMGIC as it permits depth profiling and effective screening of the material. Moreover, the test can be used to collect and compare data among multiple experimental groups simultaneously.21,22
Bonding to the dentin structure is complex due to its histological characteristics, moisture physiognomies, the infiltration of resin monomers, hydrolytic degradation by water sorption, and its complex variations in composition. 23 To avoid failure in adhesive integrity it is advisable to condition the dentin surface, as it improves the dentine permeability and SBS. 24 In the present study, the lowest SBS scores (11.99 ± 0.821) were observed in BAC bonded to unconditioned dentin. A possible explanation for this finding is that BAC gains strength by exchange of ions from saliva and the oral environment; in the present in vitro study the environment in which specimens were stored may have limited its efficacy and effectiveness. 25 Secondly, bonding of BAC to dentin is mechanical and no adhesive or conditioner was applied in these specimens, compromising bond strength values.6,25 However, RMGIC bonded to dentin exhibited results similar to the findings of Yip et al. 10 (13.25 ± 0.582), explaining the presence of the interdiffusion layer between RMGIC and dentin for exchange of ions, making a strong bond.
ECL working at the wavelength of 2790 nm is well absorbed by mineralized tissues and exhibits strong affinity to water and hydroxyapatite. 26 In the current study, surface conditioning of dentin by ECL improved the bond strength remarkably when bonded to both RMGIC and BAC. ECL photosensitization on dentin causes thermomechanical ablation and evaporation of water and organic components, making the dentinal surface scaly, flaky, and more receptive to bonding. The inter-tubular dentin, due to its lower mineral and water content, is ablated more, causing protrusion of the dentinal tubules and generating a cuff-like appearance and hence improving the bond integrity.26,27 Our findings are in line with those of Gurbai et al. 28 and Ekworapoj et al., 29 who asserted that conditioning of dentin with ECL is advantageous and improves the bond strength with RMGIC. In contrast, a study by Jordehi et al. 16 declared that conditioning of dentin by ECL decreases or has no effects on the bond strength values of RMGIC. This contraction in studies may be explained due to different type of dentin specimens (bovine versus human), heterogenous laser parameters (power and frequency), irrigation, duration of laser treatment, distance, type of RMGIC, and conditioning regimes. Moreover, Sakr, 25 in his study, explained that the better bond strength (14.21 ± 3.587) of BAC to lased dentin can be attributed to the strong affinity of BAG to exposed collagen after surface treatment with ECL. Dentin samples conditioned with ECL exhibited high SD values (Table 2). A plausible explanation for this outcome is the manual control sweeping motion of the laser beam over the dentine surface during conditioning displaying weak etching patterns. 30
In the present study, EDTA was applied on a lased dentinal surface. EDTA is a chelating agent used to dissolve the mineral layer of dentine without altering the dentinal proteins. The material decalcifies dentine, removes the smear layer, widens the orifice of the dentinal tubules, and develops thin and long resin tags.4,31 EDTA improved bond strength values among experimental groups when bonded to BAC (16.65 ± 4.985) and RMGIC (17.28 ± 4.251); however, this improvement was not statistically significant in groups in which dentin was conditioned with ECL only. These results indicate that ECL application on dentin at 4.5 W and 30 Hz has a similar effect to that of EDTA, that is, removal of the smear layer, improved dentine surface wettability, and opened dentinal tubules without carbonization and melting, making the surface amenable and viable to bonding.13,26,27 Therefore, based on these observations, the authors do not recommend the clinical use of EDTA in the surface conditioning of lased dentin when bonded to BAC and RMGIC.
The application of Tetric-N-Bond adhesive for bonding to RMGIC (21.54 ± 3.524) and BAC (19.96 ± 2.522) improved the bond strength values. The authors speculate that the primary reason for this outcome is the presence of 2-hydroxyethyl methacrylate (HEMA) in Tetric-N-Bond, which enhanced the wettability with its hydrophilic nature promoting adhesion. 32 In addition, the extrinsic water in the adhesive agent along with the intrinsic moisture during photo-ablation of the dentine surface with the laser caused rehydration of the dentinal collapsed collagen matrix, improving SBS scores. 33 Furthermore, since the specimens were kept moist this resulted in easy penetration of the monomer. Moreover, Tetric-N-bond, which is a fifth generation ethanol-based adhesive, removed water from dentinal spaces, causing the hydrogel to collapse and thus enlarging the inter-tubular spaces and allowing more resin to infiltrate, hence improving the bond integrity. 34
It is not common practice to use dental adhesive with RMGIC and BAC, as this may prevent the exchange of ions and release of fluoride. However, evidence suggests that applying adhesive decreases but does not prevent the exchange of ions. 31 Furthermore, there is no minimal quantity of fluoride yet established that inhibits caries activity. 35 Moreover, significant evidence suggests the use of adhesive on freshly cut dentine before impression taking. This phenomenon, known as “Immediate Dental Sealing,” improves SBS, improves marginal adaptation, decreases dentinal sensitivity, and decreases bacterial leakage. 36
Adhesive failures were prevalent in group 1, whereas the most frequent type of failure in the lased dentinal groups was admixed. A credible cause of this failure is porosity within the cement, resulting in stress and fracture from the cement itself and with partial debonding. This outcome is similar to the findings of Ekworapoj et al., 29 although the type of failure mode concludes a strong affinity between RMGIC and dentin. In control group 5, cohesive-type failures were high; this failure form can be attributed to inappropriate adhesive, improper curing, and lack of elasticity of the material itself. 29
Due to the in vitro design of the investigation, the conclusions are only relevant to the type of materials used, bonding agent, conditioning techniques, and laser parameters. Further studies are required to extrapolate the findings of the present in vitro study in clinical settings. Scanning electron microscopy, surface profilometry, microleakage assessment, surface free energy measurements, and durability studies for BAC and RMGIC on lased dentin surfaces are recommended to support the findings of the present study. In addition, laser parameters should be optimized for clinical success.
Conclusions
BAC when bonded to a dentin surface conditioned with ECL showed lower SBS in comparison to RMGIC. Conditioning of dentin after ECL and application of DBA is preferable to improve bond strength scores when bonded to BAC and RMGIC. Use of EDTA improved bond strength values when bonded to BAC and RMGIC; however, this improvement was not statistically significant.
Footnotes
Acknowledgements
The authors extend their appreciation to the deanship of Scientific Research at King Saud University for funding the work through research group number (RGP-1438-028).
Declaration of conflicting interests
The authors declare that they have no conflict of interest and all authors have read and approved the final draft
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Deanship of Scientific Research at King Saud University (RGP–1438–028).
Guarantor
Fahad Alkhudhairy (FAK)
