Abstract
Background:
Glass ionomer cements (GICs) are widely used in dentistry because of their remineralizing and cariostatic potential induced by fluoride. In vitro studies have reported cell toxicity triggered by GICs; however, the influence of hydroxyapatite (HAp) must be considered. The aim of this study was to evaluate the effect of HAp in decreasing the cytotoxicity of the GIC 3M Vitrebond in vitro.
Methods:
Samples of 3M Vitrebond (powder, liquid and light-cured) were incubated in Dulbecco’s modified Eagle’s medium–Ham’s F12 (DMEM-F12) for 24 hours at 37°C. Subsequently, the light-cured medium was treated with 100 mg/mL of HAp overnight. Toxicity of conditioned media diluted 1:2, 1:4, 1:8 and 1:20 was analyzed on human gingival fibroblasts (HGFs) using light microscopy and the fluorometric microculture cytotoxicity assay. The amounts of calcium fluoride (CaF2) were determined by the alizarin red S method.
Results:
The exposure of HGFs to light-cured induced cell death and morphological changes such as chromatin condensation, pyknotic nuclei and cytoplasmic modifications. Exposure to light-cured treated with HAp, significantly increased cell viability leading to mostly spindle-shaped cells (p<0.001). The concentration of CaF2 released by the light-cured was 200 ppm, although, in the light-cured/HAp conditioned medium, this quantity decreased to 88 ppm (p<0.01).
Conclusions:
These data suggest that HAp plays a protective role, decreasing the cytotoxic effect of 3M Vitrebond induced by CaF2.
Introduction
Currently, there are a wide range of dental materials used for clinical purposes, whose biocompatibility must be proven before by in vitro assays. These biomaterials are expected to not cause injury to the surrounding structures in the oral cavity; however, this does not always happen (1).
Glass ionomer cements (GICs) are a type of dental material developed a few decades ago by Wilson and Kent. They are composed of a calcium aluminosilicate powder, which contains calcium fluoride (CaF2), aluminum fluoride (AIF3), sodium fluoride (NaF) and aluminum phosphate mixed with a polyacrylic acid (2). An important technological advancement of GICs is the development of resin-modified glass ionomer systems. The GIC 3M Vitrebond, a resin-modified glass ionomer liner/base supplied in a powder-liquid form was introduced in 1988. The liquid polyacid component includes a photopolymerizable resin that hardens the material substantially when a visible light beam is applied. This material is known for preventing dental sensitivity and for its antimicrobial activity (3). GICs have certain advantages such as resistance, rigidity and aesthetic translucency. In addition, these biomaterials are characterized by other features that include adhesion to calcified tooth tissue and long-term fluoride release; nevertheless, their correlation with in vitro toxic effects remains controversial (4).
Due to the importance of fluoride in dental caries prevention, GICs have been considered therapeutic materials because this electronegative element has a strong binding affinity to enamel and the dentin inorganic matrix, in particular to hydroxyapatite (HAp) (Ca10(PO4)6(OH), a ceramic commonly used for biomedical applications because of its biocompatibility (5). HAp is the principal inorganic component of teeth and bones. This mineral has been successfully applied for bone regeneration in periodontics, implantology and maxillofacial surgery (6).
The cytotoxicity of GICs depends mainly on components eluted during the setting process (6). After the powder-liquid component is mixed, significant amounts of fluoride ions are released to the extracellular matrix (7, 8). Previous reports have shown how these biomaterials induce cytotoxic effects in vitro; however, the influence of HAp has not yet been investigated (9, 10).
The goal of this study was to evaluate the effect of HAp to decrease the cytotoxicity of 3M Vitrebond in vitro. We hypothesized that HAp exerts a positive influence on the viability of human gingival fibroblasts (HGFs). Our results provide evidence that HAp is responsible for CaF2 uptake due to ion exchange. These observations indicate that HAp plays a protective role by decreasing the cytotoxic effect of 3M Vitrebond induced by CaF2.
Materials and methods
Samples preparation
To obtain the conditioned media, 3M Vitrebond (3M Dental Products, St. Paul, MN, USA) (powder, liquid and light-cured), was prepared according to the manufacturer’s instructions (1 scoop and 1 drop of powder and liquid, respectively, per portion) and then incubated in 400 μL of growth medium for 24 hours at 37°C in a humidified atmosphere with 5% CO2. Additionally, light-cured medium was treated with 100 mg/mL of lyophilized-bone HAp for 24 hours at room temperature. Growth medium was used as a negative control. After incubation, the samples were centrifuged at 10,000 rpm for 5 minutes, and eluates were obtained.
Alizarin red S assay
The CaF2 content of light-cured medium and light-cured/HAp conditioned medium was measured by the alizarin red S method (11). Growth medium was used as a negative control. A solution of 2% alizarin red S was prepared and adjusted to pH 4.2 with 0.5% ammonium hydroxide (Sigma-Aldrich Co., St. Louis, MO, USA). CaF2 was used to perform a standard curve in a range of 0-200 ppm. The amount of CaF2 was quantified in a 96-well scanning spectrophotometer at 595 nm.
Cell culture
HGFs were donated by Higinio Arzate, PhD (UNAM, Mexico) and then grown as previously described with slight modifications (12-14). The HGFs (passage 16) were cultured in Dulbecco’s modified Eagle’s medium–Ham’s F12 (DMEM-F12) supplemented with 10% fetal bovine serum (Gibco- Invitrogen, Carlsbad, CA, USA), 100 U/mL penicillin, 100 μg/mL streptomycin and 0.25 μg/mL amphotericin B (Sigma-Aldrich) at 37°C in a humidified atmosphere with 5% CO2.
Fluorometric microculture cytotoxicity assay
HGFs were seeded onto 96-well plates at a density of ~2 × 104 cells per well, maintaining these in growth medium for 24 hours (Corning Inc., Corning, NY, USA). The conditioned media of powder, liquid, light-cured and light-cured/HAp treated medium were added at dilutions of 1:2, 1:4, 1:8 and 1:20. Growth medium was used as a negative control. After 24 hours of incubation, the medium was removed, and the cells were washed 3 times with phosphate-buffered saline (PBS). The cytotoxicity assay was performed as described by Larsson and Nygren (15, 16). Briefly, fluorescein diacetate (FDA; Sigma-Aldrich) was dissolved in dimethyl sulfoxide (DMSO; Sigma-Aldrich) and kept frozen at -20°C as a stock solution (10 mg/mL). FDA was diluted in PBS at 10 μg/mL, and 200 μL was added to each well. The plates were then incubated for 30 minutes at 37°C in the dark. A 96-well scanning fluorometer, GloMax® Multi + Microplate Multimode (Promega, Madison, WI, USA) was used at 495 nm. Data were analyzed to determine cell viability (%).
Crystal violet staining
Treated HGFs were washed and then fixed with 10% neutral-buffered formalin (BDH Chemicals, Ltd, London, UK) for 30 minutes at room temperature. Fixed cells were stained with a 1% crystal violet (Sigma-Aldrich) solution for 1 minute and then washed 2 times using PBS. Cells were analyzed in a Zeiss Axiovert 200 M inverted microscope (Carl Zeiss, Göttingen, Germany).
Statistical analysis
Data were analyzed using ANOVA and the Tukey test; a p value <0.01 was considered statistically significant in all treatments. The analysis was performed using SPSS for Windows (SPSS Inc., Chicago, IL, USA). All experiments were done in triplicate.
Results
CaF2 concentration
CaF2 was measured by the alizarin red S method. The light-cured sample had the highest quantity of CaF2 (200 ppm per portion); however, in light-cured/HAp treated medium, a concentration of 88 ppm was found. In the negative control, CaF2 was not detected (p<0.01) (Fig. 1).
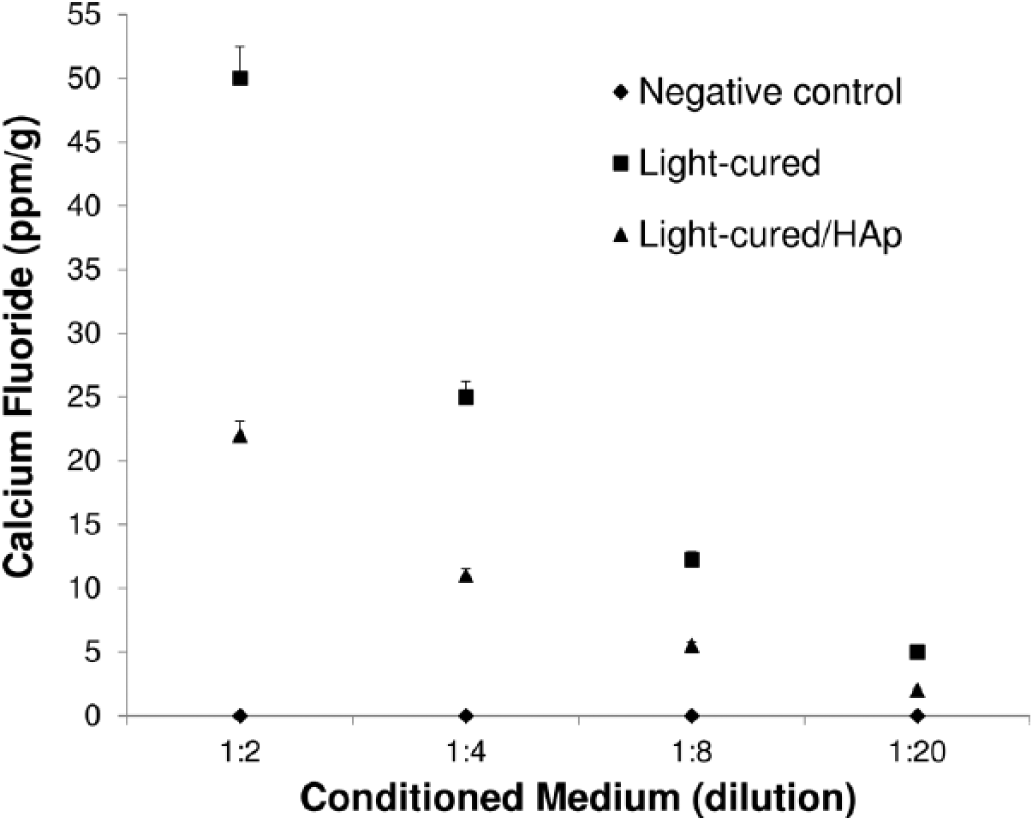
Mean calcium fluoride (CaF2) concentrations in 3M Vitrebond conditioned media. CaF2 content was quantified using the alizarin red S assay. The amount of CaF2 in light-cured medium significantly decreased at all dilutions after exposure to hydroxyapatite (light-cured/HAp). CaF2 was not detected in the negative control (p<0.01).
Comparison of cell viability
Cytotoxicity of HGFs exposed to conditioned media at different dilutions (1:2, 1:4, 1:8 and 1:20) was evaluated using a fluorometric assay. Overall, the cells treated with powder, liquid and light-cured media suffered damage or dose-dependent cell death. After exposure to powder, a gradual increase of cell viability was maintained (0, 23%, 50% and 63%). In contrast, the liquid was capable of drastically decreasing the number of living cells, although, in the 1:20 dilution, viable cells were detected (55%). The light-cured medium was highly toxic to the cells except in the lowest dilution (83%); however, the light-cured/HAp conditioned medium showed the highest quantity of living cells, including at the greatest dilution (49%, 65%, 76% and 98%) (p<0.001) (Fig. 2).
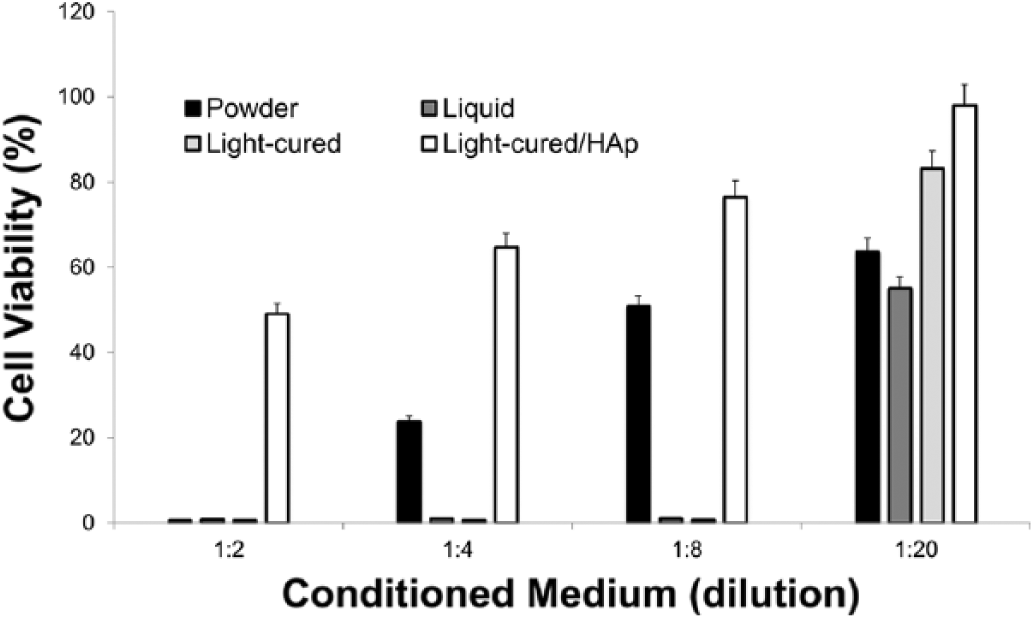
Cytotoxicity of 3M Vitrebond conditioned media on human gingival fibroblasts (HGFs). Toxicity was determined by fluorometric microculture cytotoxicity assay. Each column represents the mean percentage of cell viability where the cytotoxic effect of light-cured/HAp conditioned medium was significantly less compared with that of powder, liquid and light-cured media at all concentrations (p<0.001). Error bars indicate SD (n = 3).
Analysis of cell morphology
The morphological changes in HGFs after exposure to conditioned media were analyzed using crystal violet staining. The negative control cells showed a typical spindle shape with spherical nuclei, cytoplasmic extensions and a clear tight adherence to the surface of the well. However, in the powder, liquid and light-cured media, a lower number of cells with chromatin condensation, pyknosis and low cytoplasmic content were observed. In contrast, in the light-cured/HAp conditioned medium, a significant increase of cell confluence as well as a similar morphology to the negative control was evident (Fig. 3).
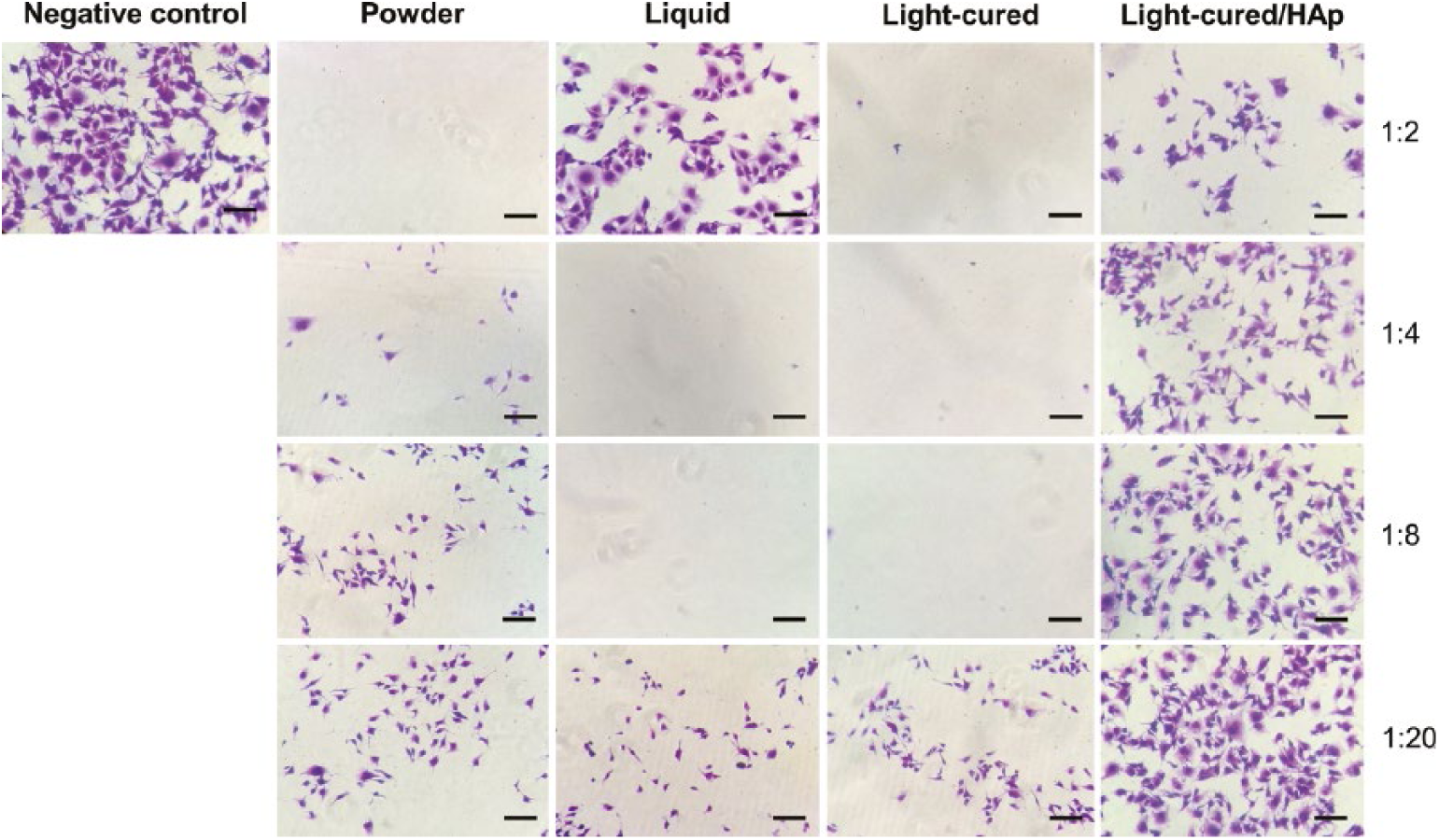
Cell morphology after exposure to 3M Vitrebond conditioned media. Human gingival fibroblasts (HGFs) were stained with crystal violet and then analyzed by bright-field microscopy. Representative images of light-cured/HAp conditioned medium showed a major cell confluence without apparent cytoplasmic changes compared with powder, liquid and light-cured media at all dilutions. (Original magnification ×10, scale bar = 100 μm.)
Discussion
GICs are recognized for their wide range of applications in dentistry based on their excellent properties (2, 17, 18). Despite these characteristics, the biocompatibility of GICs is not always satisfactory, with several reports describing their cytotoxicity (19-22).
Our results provide evidence that HAp is capable of decreasing the cytotoxicity of 3M Vitrebond induced by CaF2 in vitro. The results for exposure of HGFs to light-cured/HAp conditioned medium suggest an important protective mechanism mediated by this mineral. A possible explanation for this toxicity reduction is a chemical interaction that occurs between HAp and CaF2, triggering an ion exchange and consequently the removal of fluoride from the local environment. This phenomenon confirms that GICs have the ability to bind chemically with tooth structures after a setting reaction (23-25).
It is known that GICs produce cell damage, because these biomaterials are capable of releasing fluoride, an element associated with systemic toxicity. In agreement with our experiments, a previous study suggested that 3M Vitrebond induces high cytotoxicity in human cells because it releases a greater amount of fluoride compared with other GICs (26). Based on our findings, the quantity of CaF2 was significantly lower after treatment with HAp, confirming their fluoride uptake capacity.
This study was mainly based on the cytotoxicity caused by CaF2, although it was not the unique responsible factor. The liquid conditioned medium also significantly decreased cell viability in all analyzed dilutions. Interestingly, when cell morphology after treatment was observed, the cells did not show any apparent changes due to the fixing effect of the liquid, demonstrating a higher cytoplasmic content in comparison with the negative control, although with a lack of metabolic activity.
Despite the fact that our results support the general concept that GICs are biocompatible materials during in vivo circumstances, there are some shortcomings associated with this method. In vitro tests for the evaluation or comparison of cytotoxicity have attractive features (27); however, the absence of microorganisms, and of interactions with growth factors or enzymes included in the dentin matrix may alter the cells’ fate. Therefore, a direct extrapolation of these assays to clinical situations is not possible (28).
GICs have been claimed to have low dental pulp toxicity in clinical use (29). These cements exhibit cytotoxicity in the freshly set state, but this decreases substantially and is time-dependent. The buffering and protein-binding effects of saliva appear to mitigate the cytotoxic effects (30). Early reports of studies performed with human teeth have demonstrated that 3M Vitrebond applied as a liner in deep class V cavities causes no inflammatory dental pulp response, supporting the lack of a toxic effect in clinical outcomes (31). In this study, the light-cured conditioned medium tested would not appear to be a desirable material for placement in direct contact with human cells; however, we observed that HAp is an excellent inorganic buffer that could be used for evaluating the cytotoxicity of GICs in vitro, with the objective of partially mimicking the biological response that occurs in animal models.
To our knowledge, this is the first report that evaluates the cytotoxic effect of 3M Vitrebond in contact with HAp in vitro. Although we have found promising results, to evaluate the influence of other factors such as bacteria interaction, the effects of the immunological system and organic molecules, further studies are required.
In conclusion, this study found evidence to support the hypothesis that CaF2 correlates with the cytotoxic effect of 3M Vitrebond in vitro. Moreover, HAp is capable of significantly reducing this toxicity due to its protective role in the uptake of fluoride ions. Altogether these data explain the high biocompatibility of GICs observed in clinical practice.
Footnotes
Acknowledgements
C. C.-R. wants to thank the CONACyT for financing the project (grant no. 183825). All authors are grateful to Andrés Mendiola-Jiménez for his technical support. S. M.-M. also wants to thank the CONACyT for her scholarship.
Disclosures
Financial support: Funding was received from the CONACyT to finance this project (grant no. 183825).
Conflict of interest: The authors declare that they have no conflicts of interest.
