Abstract
High viscosity glass ionomer cements (GICs) are widely used in various clinical applications, being particularly effective in atraumatic restorative treatment (ART) due to the synergistic interaction between the material and the technique. However, the inadequate mechanical properties of GICs raise concerns regarding the predictability and longevity of these restorations in areas exposed to occlusal stress. Various modifications of the powder components have been proposed to improve the mechanical strength of GICs to withstand occlusal loading during mastication. In this in vitro study, we investigated whether the nanoparticles (NPs) added to commercially available GICs could fulfill this requirement, which would likely broaden the spectrum of their potential clinical applications. Two commercially available GIC powders (Fuji IX and Ketac Molar), modified by the addition of 5 wt.% TiO2, MgHAp100 or MgHAp1000 NPs, were incorporated into the corresponding liquid in an appropriate ratio, and the mixed cements were evaluated in terms of fracture toughness, flexural strength, Vickers microhardness and rheological tests and compared with the original material. Fuji IX containing 5 wt.% MgHAp100 NPs had lower flexural strength, while Ketac Molar with 5 wt.% TiO2 NPs showed increased fracture toughness. Vickers microhardness increased in Fuji IX following the addition of 5 wt.% TiO2 and MgHAp100 but decreased in Ketac Molar comprising 5 wt.% MgHAp100 (
Introduction
Tooth structure is supposed to withstand a considerable number of cycles of masticatory forces that can exceed 700 N in value. 1 When dental structure is lost or damaged, usually due to caries or trauma, dental materials used for restoration are expected to preserve the overall tooth integrity and function. The glass ionomer (polyalkenoate) cements (GICs) are particularly well-suited for this purpose, as these tooth-colored restorative materials exhibit not only anticariogenic properties but also bond chemically to the tooth structure. 2 However, as these materials have low mechanical strength, overcoming this shortcoming has been a subject of extensive research. Various modifications of their powder3–5 and liquid6–8 components have been proposed to improve the mechanical properties and fill the voids between the particles in the GIC matrix. Generally, the powder component contains calcium or strontium fluoro-alumino-silicate glass particles, while the liquid represents aqueous solution, mainly consisted of polyalkenoic acid. The acid-base curing reaction lasts for 24 h after mixing these two components and this prolonged effect is made possible by the presence of an aqueous reaction medium. The setting process commences with a reaction between the components, allowing gel formation after which the unreacted glass particles, which serve as fillers, are bound to the silica (SiO2) gel matrix. Subsequently, the cement hardens as a result of cross-linking between the polymeric chains of the polyacid component, forming a cross-linked acrylate matrix with the calcium and aluminum ions in the powder component. 9
Resin-modified GIC was developed with the aim of controlling the setting reaction and improving the esthetic and mechanical properties of conventional GIC. This light-curable GIC contains resin and a light-curing agent to accelerate the setting process and enhance the mechanical durability of the restoration. However, despite its advantages, resin-modified GIC may cause adverse biological effects such as cytotoxicity and pulp irritation, which are not observed with conventional GIC. 10 With regard to possible formulation changes in the powder component of GIC, hydroxyapatite (HAp) and tricalcium phosphate (TCP) are proposed as naturally occurring minerals, commonly referred to as “bone minerals.” They possess properties that are highly beneficial for dental restorations because their structure bears a remarkable resemblance to that of a natural tooth. Available evidence shows that the use of HAp nanoparticles (NPs) in dental applications leads to an improvement in the surface hardness, toughness, and biocompatibility of the restored tooth. 11 In addition, inclusion of TiO2 NPs into the GIC has been found to improve its mechanical behavior. As this inorganic photocatalytic additive is considered chemically stable and has antimicrobial properties, 12 TiO2 NPs are expected to fill the gaps between the microparticles in the ionomer matrix, making it more compact and denser. 13
The size and distribution of particles within a material can have a significant impact on its mechanical resistance. If the particles are large, unevenly distributed, or do not achieve a predictable bond with the matrix, the material may be weak and prone to fracture or deformation.14,15 Conversely, if the particles are small, uniform and evenly distributed, their more even incorporation into the ionomer matrix will increase the resistance to deformation. 16 Occlusal loading during mastication can restrict movement within the material when the constituent particles are tightly packed, preventing its deformation or failure. Therefore, to obtain the best possible outcome, when working with these cements, the manufacturer’s instructions for mixing must be strictly followed. In contrast, mixing to achieve the consistency desired by the clinician may result in a weaker structure and poorer mechanical performance. In the study conducted by Baig et al., 17 mixing GIC with different proportions of powder and a constant volume of liquid decreased the material’s compressive strength, highlighting the importance of following the manufacturer’s instructions regarding the powder/liquid ratio to achieve the optimal rheological properties of the cement paste and more predictable characteristics of the final material.
Although the modification of GICs with various NPs has been studied extensively, the application of magnesium-doped HAp NPs or spherical shaped TiO2 NPs in order to evaluate and compare the mechanical properties of two different commercially available GICs has not yet been investigated. The aim of this in vitro study was to compare the effects of TiO2 and HAp modifications on two types of conventional hand-mixed GIC, focusing on fracture toughness, flexural strength, surface microhardness, and rheological properties, in comparison to the respective original formulation. It was hypothesized that the above-mentioned properties of the cements would improve after the incorporation of TiO2 or MgHAp NPs.
Methods
Grouping of specimens
Powder of both commercially available high-viscosity GIC materials—Fuji IX (Fuji IX GP, GC International, Japan) and Ketac Molar (Ketac Molar EasyMixTM-3 M/ESPE, Maple-wood, Minnesota, USA) was incorporated in proportion of 5 wt.% with either TiO2, MgHAp100 or MgHAp1000 NPs. Four different experimental groups were obtained per each material, in general there were eight groups: G1. Fuji IX (FIX) = control; G2. FIX + 5 wt.%TiO2; G3. FIX + 5 wt.% MgHAp100; G4. FIX + 5 wt.%MgHAp1000; G5. Ketac Molar (KM) = control; G6. KM + 5 wt.%TiO2; G7. KM + 5 wt.%MgHAp100; G8. KM + 5 wt.%MgHAp1000. Flexural strength, Vickers microhardness, fracture toughness and rheological properties were evaluated. Furthermore, the fracture morphology of specimens was examined.
Specimen preparation
TiO2 nanoparticles were produced through a sol-gel technique utilizing titanium IV butoxide (Ti(OBu)4, Sigma Aldrich, 97%), isopropanol (C3H8O, Fluka Chemicals, 99.8%) glacial acetic acid (C2H4O2, Lach Ner Chemicals, 99.8%) and distilled water and as starting materials. The synthesis was carried out with an excess of water for complete hydrolysis of the butoxide, using Ti(OBu)4:C3H8O:C2H4O2:H2O molar ratio of 1:5:0.1:10. Following uninterrupted magnetic mixing for 2 h at room temperature, a white solid formed. This solid was cleansed through centrifugation using distilled water (2 times at 10,000 rpm/1 min) and ethanol (1 time at 10,000 rpm/1 min), then subjected to 70°C oven drying for 24 h and ethanol, then subjected to 70°C oven drying for 24 h. Ultimately, the resultant powder underwent calcination at 600°C for 2 h to facilitate the crystallization of TiO2.
MgHAp100 powder was obtained starting from calcium nitrate tetrahydrate (Carl Roth Gmbh, Karsruhe, Germany), magnesium nitrate hexahydrate (NRK Inzenjering, Belgrade, Serbia), ethylenediaminetetraacetic acid disodium salt dihydrate and urea (Sigma-Aldrich Chemie GmbH, Taufkirchen, Germany), sodium dihydrogen phosphate dihydrate (VWR Chemicals Prolabo, Leuven, Belgium). Starting solution for hydrothermal synthesis of Mg-doped hydroxyapatite was made under the stoichiometric Ca/P molar ratio of 1.67, whereby 5 mol.% of calcium was replaced with magnesium ions. Mg-doped hydroxyapatite (MgHAp) powder was synthesized by hydrothermal method at 160°C for 3 h18,19 using Ca(NO3)2•4H2O (11.21 g), Mg(NO3)2•6H2O (0.6390 g), Na2H2EDTA•2H2O (11.18 g) NaH2PO4•2H2O (4.68 g) and (NH2)2CО (12.0 g) as precursors. The precursors were dissolved in 1.5 L deionized water, and the synthesis was performed in an autoclave by heating of Erlenmeyer flask with precursor solution under the pressure of 8 bars. After slow cooling, the precipitate was filtered, washed with deionized water and dried at 105°C. MgHAp1000 was obtained by calcination of dried Mg-doped powder at 1000°C for 2 h in air atmosphere, using the heating rate of 10°C min−1.
Five percent by weight based on the weight of the GIC powder per scoop was replaced with either TiO2, MgHAp100, or MgHAp1000 powder. Applied powder was weighed on a precision balance to an accuracy of 0.0001 g and manually added to the GIC powder of the two materials tested, encapsulated, and shaken in an amalgam mixer for 30 s.20,21 Immediately thereafter, the prepared powder was fully incorporated into the appropriate liquid based on polyalkenoic acid with the spatula on the glass plate, at an appropriate ratio according to the manufacturer’s instructions. The recommended powder/liquid (P/L) ratio for Fuji IX is 3.6/1, while for Ketac Molar it is 3.0/1.
Within 60 s of mixing, the cement from each group was placed in one increment into polymer molds that had the same dimensions: length (L) × width (b) × thickness (h) of 25 mm × 6 mm × 3 mm for different tests. The molds were pressed between a glass plate and a polyester strip for 6 min and stored at 37°C and relative humidity for 24 h before each test. Only the samples that showed no visible defects in the macroscopic examination were used for the upcoming tests.
Flexural strength
Samples (

where
Fracture toughness
Fracture toughness KIC (MPa·m1/2) was measured using the single edge V-notched beam (SEVNB) method as described in ASTM D5045-14. Testing was also performed under three-point bending on a universal testing machine AT-L-118A (Toyoseiki, Japan) with the same settings as for flexural strength testing. The dimensions and sample size of the specimens were identical to those used in the bending test. First, a sharp notch was created by machining, and then a sufficiently sharp crack was created by sliding (or sawing) a new razor blade over the originally machined notch root. The generated crack length (a) was measured using an Orthoplan light microscope (Leitz, Germany). The fracture toughness KIC (MPam1/2) was calculated according to the following formula 23 :
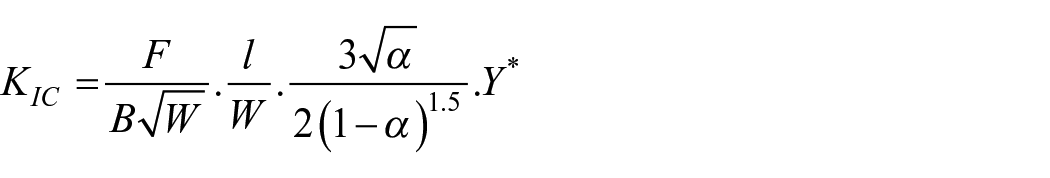
where
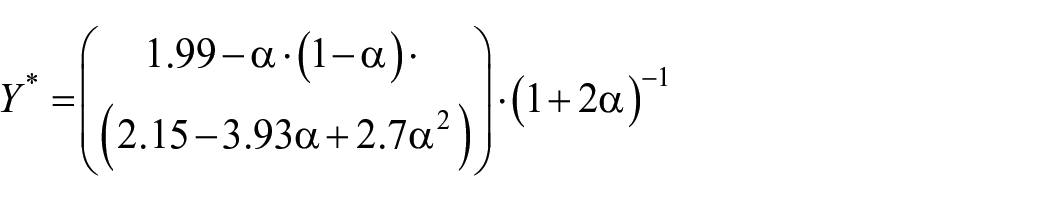
Vickers microhardness test
The Vickers surface microhardness was tested using a microhardness tester Wilson Tukon 1102 (Buehler, IL, USA) with a low-force load of 0.5 kgf (4903 N) and a dwell time of 30 s, according to procedure described in ISO 6507-1:2018. The results were reported as Vickers hardness number (HV 0.5). The measurement was performed on the fractured samples (
Rheology test
Rheological measurements were performed using a controlled-stress HAAKE Mars Rheometer (Thermo Fisher Scientific) at 23°C ± 0.1°C with parallel plates (PP35 Ti) and a 1 mm gap. Amplitude sweep tests determined the linear viscoelastic region (LVR) by recording storage (Gʹ) and loss (Gʺ) moduli versus shear stress (0.01–100 Pa) at 1 Hz. The midpoint of the LVR was used as the constant shear stress in frequency sweep tests (0.1–10 Hz) to determine the complex viscosity (η*), Gʹ, and Gʺ moduli.
Structural characterization—X-ray diffraction analysis
The crystallinity and the phase composition of the obtained TiO2 NPs were determined by X-ray diffraction (XRD) using Rigaku MiniFlex 600 diffractometer (CuKα radiation, λ = 1.54 Å). X-ray diffraction pattern was recorded in the range 20°–50° at a scan rate of 0.02°s−1 Similarly, the phase composition of HAp based powders was also investigated using XRD analysis performed on a Rigaku Smartlab diffractometer with CuKα radiation (1.54 Å) in the 2θ angle ranging from 20° to 50° with a scan rate of 0.02°s−1.
Morphological and microstructural characterization by scanning electron microscopy
Impact of added particles to the GIC powders on mechanical properties of modified GIC cements was observed by SEM analysis of the fracture surface obtained in the previously applied testing methods. Morphology of used inorganic fillers and microstructural characteristics of fractured surfaces of the GIC samples was observed after deposition of an atomic gold layer by MIRA 3 XMU FE-SEM (TESCAN, Brno, Czech Republic), operated at 20 keV.
Statistical analysis
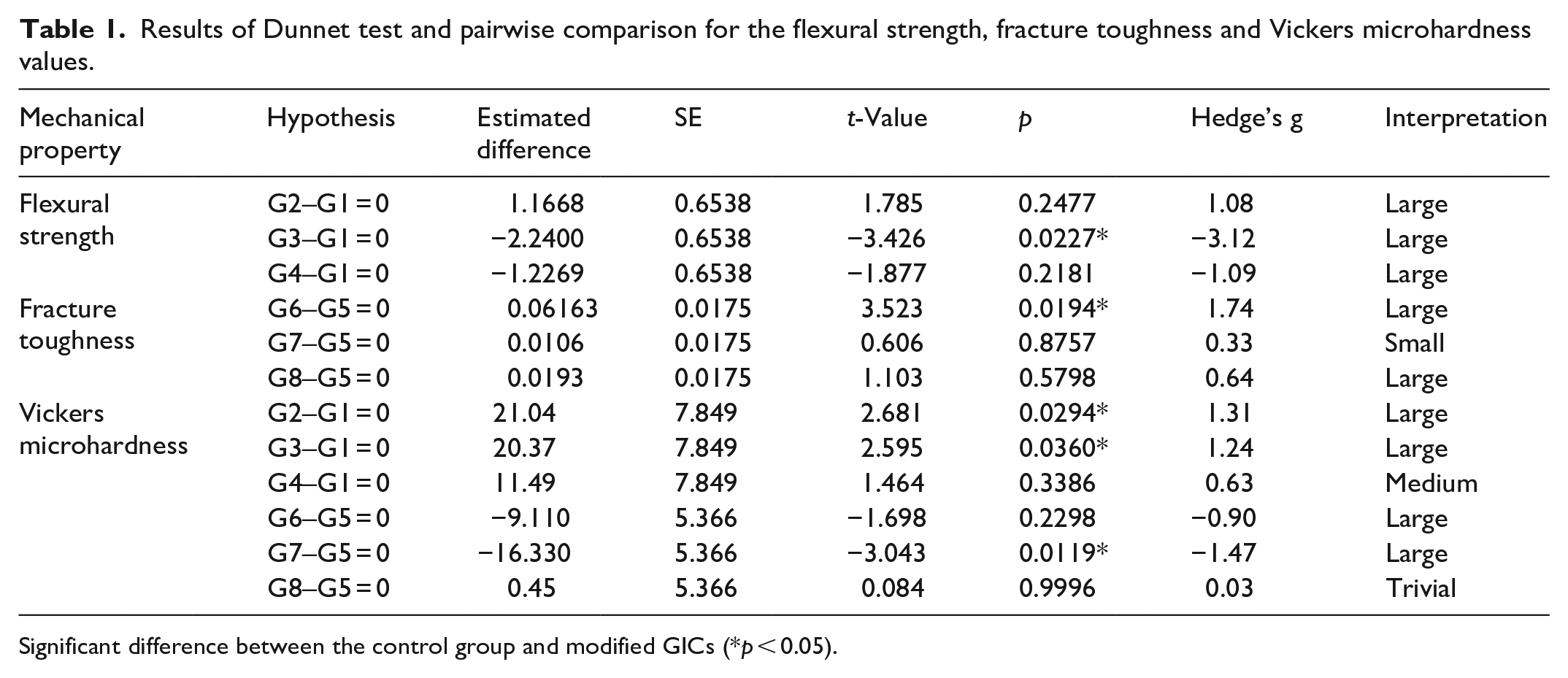
To assess the differences in the mean Flexural strength, Fracture toughness, and Vickers hardness between four samples in two different materials, six one-way ANOVA (one-way analysis of variance) were conducted, followed by post hoc Dunnet tests. Effect sizes were determined as Hedge’s g (Cohen’s
Results
Characterization of the powder samples
XRD analysis
All diffraction peaks correspond to the anatase phase with tetragonal crystal structure (JCPDS 21-1272). No peak corresponding to rutile phase was observed (Figure 1).
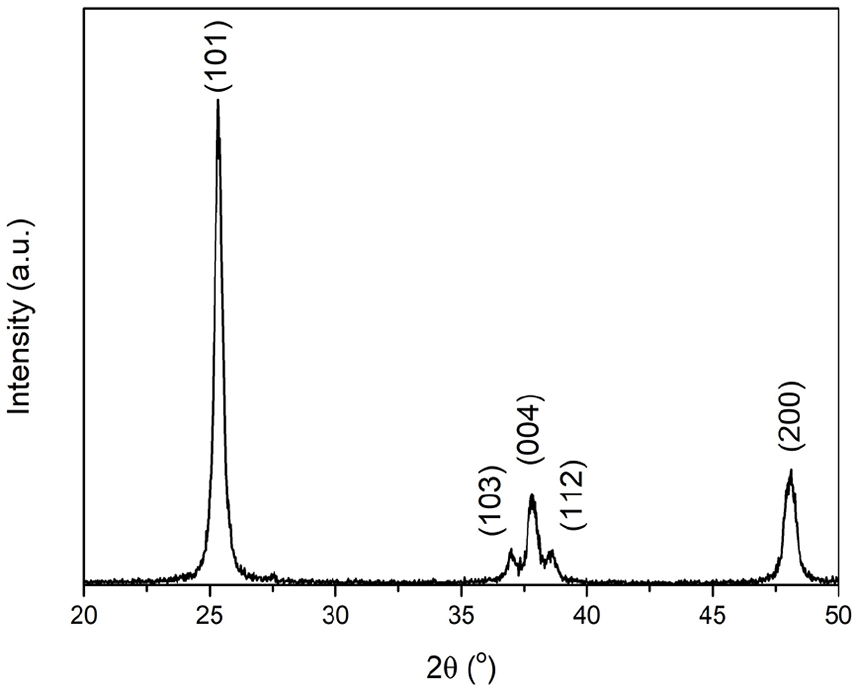
XRD pattern of the TiO2 NPs calcined at 600°C.
In the case of MgHAp100 powder, all diffraction peaks correspond to the HAp. The presence of Mg ions in the HAp crystal structure promotes its phase transformation to the β-TCP phase, as shown in Figure 2. On the other hand, β-TCP is the dominant phase in the case of calcinated MgHAp1000 powder. The main peaks at the showed XRD patterns were compared with Standard cards, JCPDS 09–0169 and JCPDS 09–0432 for β−TCP and HAp respectively, in order to confirm crystal phases.
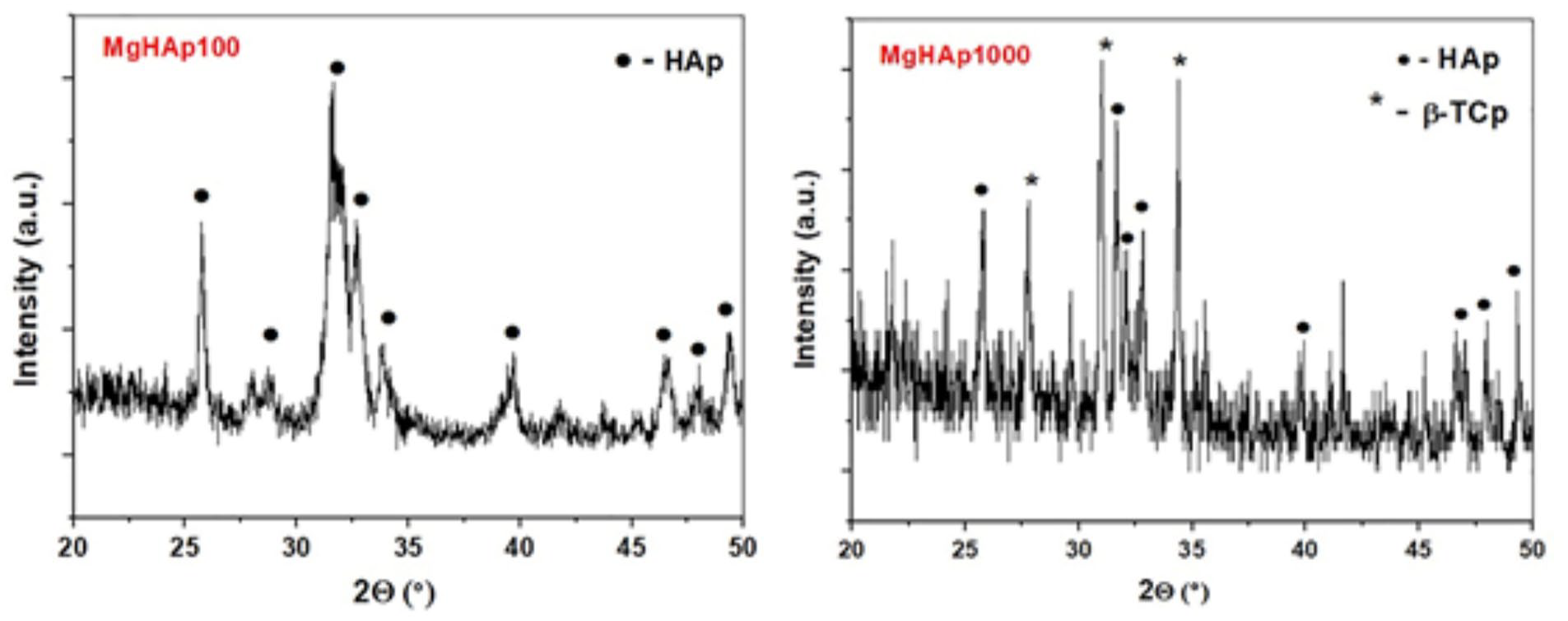
XRD patterns of the MgHAp100 and MgHAp1000 powders.
The elemental composition of MgHAp100 powder was determined previously by Energy Dispersive X-Ray (EDX) analysis performed by Tescan Vega TS 5130MM SEM coupled with INCaPentaFETx-3 detector. The results of the EDX analysis confirmed follow atomic percents: Ca—18.19 at.%, P—14.25 at.% and Mg—0.44 at.%, confirming the presence of Mg as a dopant in HAp structure. 25
SEM analysis
Representative SEM micrographs of TiO2, MgHAp100 and MgHAp1000 particles revealed their shape and cluster/agglomerate formation tendency. Particle size were determined by image analysis of SEM micrographs. As can be seen from Figure 3(a), TiO2 NPs were predominantly of spherical shape and uniform size in the range of 30–80 nm, whereas MgHAp100 NPs formed spherical agglomerates composed of nano-rods with a size of 20–30 nm in width and 50–100 in length (Figure 3(b)). In the case of MgHAp1000 NPs, these rods were more rounded and densely packed, forming cauliflower-like agglomerate particles around 1 micron in size (Figure 3(c)).

Representative SEM micrographs of the obtained powders: (a) TiO2, (b) MgHAp100, and (c) MgHAp1000.
Flexural strength
Flexural strength values of the two modified GICs-Fuji IX (G1–G4) and Ketac Molar (G5–G8) are shown in Figure 4. As can be seen from Table 1, the group comprising of Fuji IX modified by incorporating 5 wt.% MgHAp100 NPs exhibited significantly lower flexural strength compared to other samples based on this GIC (
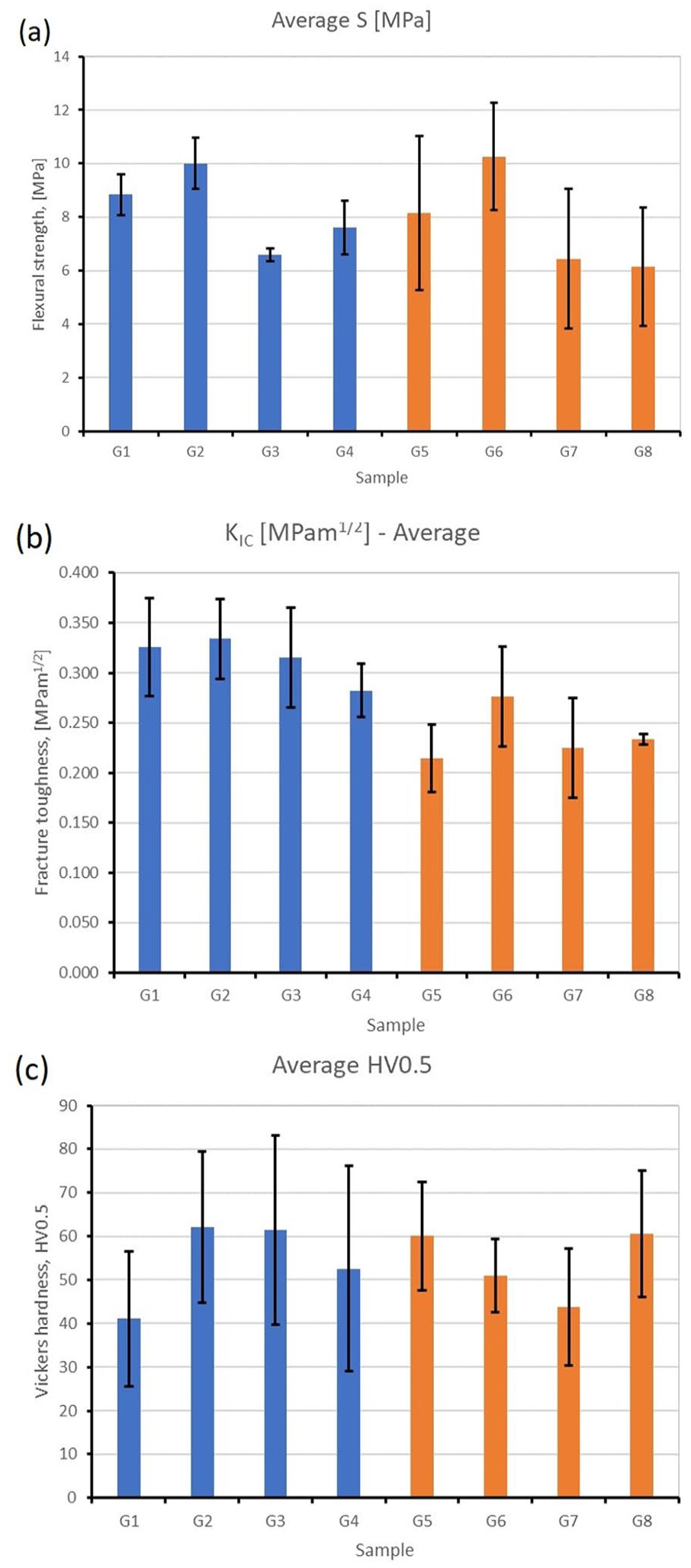
Graphs about comparisons of the mechanical properties of different groups. (a) fracture toughness, (b) flexural strength, (c) Vickers microhardness. G1–G4 Fuji IX and its modifications; G5–G8 Ketac Molar and its modifications.
Results of Dunnet test and pairwise comparison for the flexural strength, fracture toughness and Vickers microhardness values.
Significant difference between the control group and modified GICs (*
Fracture toughness
Fracture toughness values of the modified GICs are shown in Figure 5. As indicated in Table 1, only the addition of 5 wt.% TiO2 NPs to the Ketac Molar GIC led to a statistically significant increase in fracture toughness compared with the control group (
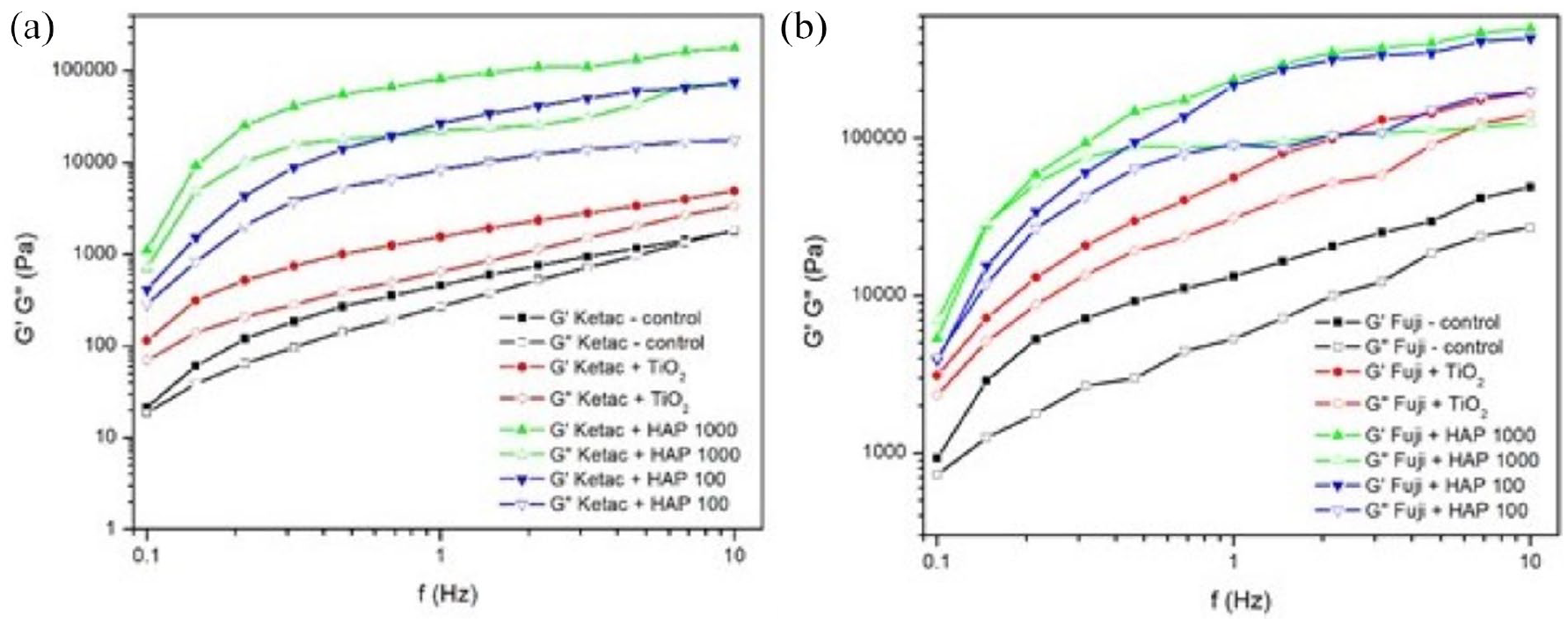
Storage (Gʹ) and loss (Gʺ) moduli versus frequency of: (a) Ketac Molar and its modified alternatives and (b) Fuji IX and its modified alternatives.
Vickers microhardness
Vickers microhardness values of the modified GICs are shown in Figure 6. As can be seen from Table 1, a significant increase in surface microhardness (VHN) was observed in Fuji IX + 5 wt.% TiO2 and Fuji IX + 5 wt.% HAp100 compared to the compatible control group (
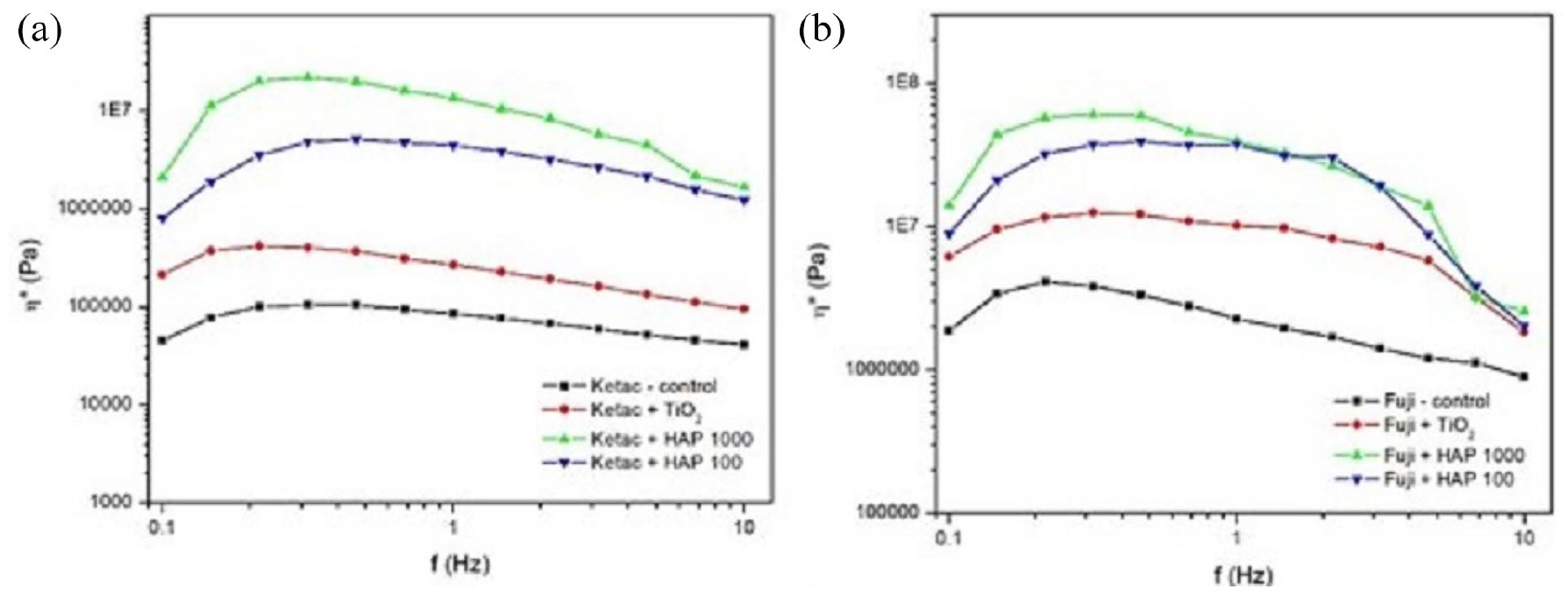
Complex viscosities η* versus frequency of: (a) Ketac Molar and its modified alternatives and (b) Fuji IX and its modified alternatives.
Rheological properties
The viscosity of all samples gradually increased during the setting process. Additionally, the viscosities of the modified GICs increased linearly upon the addition of TiO2 and MgHAp NPs. The highest viscosity was detected in samples modified with MgHAp1000 NPs, followed by those containing MgHAp100 NPs, as shown in Figure 5, where Gʹ represents the elastic component or the solid-like behavior of fluids.
The studied materials were subjected to the frequency sweep tests and the results are shown in Figure 5. It can be noted that, for both tested GIC materials with and without NPs, storage modulus values exceeded viscosity measurements across the entire range of applied frequencies. The complex viscosity curves for all materials decreased with the increase in frequency, indicating non-Newtonian flow behavior (Figure 6).
Addition of TiO2 and MgHAp NPs to Ketac Molar and Fuji IX affected the rheological characteristics of these materials differently, with MgHAp1000 being the most influential, as it increased the values of the storage moduli as well as the complex viscosity of the entire system. Moreover, the rheological parameters were least affected by the addition of TiO2.
SEM observation of the fracture surface
In preparation for the SEM analysis, after the flexural strength tests, representative fracture surfaces for each GIC group were dried and gold-coated, and the SEM micrographs were captured as shown in Figures 7 and 8. In general, all Ketac Molar specimens exhibited a rougher fractured surface texture due to the presence of wider cracks compared to the Fuji IX specimens (Figure 7, G5–G8). Presence of TiO2 agglomerates (which may influence the direction of crack formation) was confirmed in both GICs under 1000× magnification. The SEM analysis also revealed a weakened bond between these agglomerates and the matrix, leading to the appearance of cracks along their interface (Figure 7, G2, G6). In contrast, a more uniform distribution of HAp-based particles was observed in the samples containing both 5 wt.% MgHAp100 and 5 wt.% MgHAp1000 (Figure 7, G3, G4, G7, G8).

Representative SEM micrographs of Fuji IX and Ketac Molar samples after flexural strength testing.

SEM micrographs of the fractured surface of Fuji IX and Ketac Molar samples after flexural strength testing at 30,000× magnification.
SEM micrographs obtained at a higher magnification revealed very good adhesion of MgHAp100 and MgHAp1000 spherically agglomerated particles with polymeric matrix, but also confirmed the presence of deagglomerated TiO2 NPs which were evenly distributed throughout the matrix in both cases (Figure 8, G2, G6). Unmodified Ketac Molar control sample (Figure 8, G5), unlike Fuji IX (Figure 8, G1), contained spherical particles measuring a few 100 nm in diameter as a filler, but the morphology of these particles was clearly different compared to the modifiers used in this study. While some MgHAp100 spherical agglomerates were fractured through the central void, in some cases spherical agglomerates remained unbroken at the fractured surface of modified Fuji IX samples (Figure 8, G3). As can be seen from Figure 8, G4, calcinated MgHAp1000 spherical particles at the fractured surfaces remained unbroken in all cases, while thin polymer layers formed over the surface of MgHAp100 and MgHAp1000 particles (Figure 8, G7 and G8, respectively). These differences in fracture type within the polymer matrix could be attributed to the strong bonding between the calcium phosphate particles within the Ketac Molar material.
Discussion
The chemical composition and the powder/liquid ratio are the most significant factors affecting the GIC material properties upon setting. Increasing the glass filler content by adding a greater amount of powder to a constant volume of liquid can result in greater resistance to occlusal forces. 26 However, increasing the material viscosity in this manner will render its handling more difficult. The Ketac Molar Easy Mix powder has a highly granulated texture, which makes it less dense and more absorbent. For this reason, the manufacturer recommends a higher powder/liquid ratio when using this product. When curing the cement, it is difficult to avoid voids, especially in hand-mixed products, as air is inevitably trapped during mixing, which makes the material less homogeneous and reduces its mechanical strength. 27 The addition of NPs to the GIC restorative material is expected to decrease the gaps between the GIC particles, thereby improving its uniformity and solidity, enhancing its mechanical properties. 28 However, in the present study, SEM analysis revealed that the incorporation of NPs in both examined GICs did not alter the structure of the GIC, as shown in Figure 7. The dark areas indicate voids in the cement structures caused by the manual mixing of the samples. It can be seen that all groups with added different NPs had a similar number and depth of voids as the control group.
The key variations among calcium phosphates are mainly based on their Ca/P ratio and their solubility in the natural environment. For example, HAp has the highest Ca/P ratio (1.67) and is the least soluble in a physiological setting, whereas β-tricalcium phosphate (β-TCP) with a lower Ca/P ratio (1.50) is a more soluble HAp phase characterized by excellent biocompatibility. 29 Previous studies have shown that the addition of Mg2+ to HAp increases the likelihood of HAp undergoing the phase transformation into a more biologically active ß-TCP phase during the formation of biphasic calcium phosphate (BCP) within bioceramic materials.18,19 In the present study, two distinct powders based on HAp were produced using precursor solutions with the 1.67 (Ca + Mg)/P molar ratio by a hydrothermal method, followed by drying and calcination at 100°C for MgHAp100 and 1000°C for MgHAp1000. Generally, when calcium-deficient HAp is exposed to temperatures above 800°C, it transforms partially into β-TCP which has denser particles with grained morphology that are more soluble in water and bodily fluids than hydroxyapatite. Furthermore, the substitution of 5 wt. mol.% of calcium ions with magnesium ions in the material prevents the transition from β- to α- phase, which could weaken its mechanical properties.18,27 Instead, as shown in the present study, a biphasic structure called BCP is obtained, which contains HAp and β-TCP phases in roughly equal amounts. This type of structure is often desirable for biomedical applications because BCPs are highly bioactive due to the increased solubility of β-TCP. 30
As a part of the present investigation, two GIC materials were modified by adding either 5 wt.% TiO2 or MgHAp NPs, given that Cibim et al. 31 found this percentage optimal as there was a sufficient amount of GIC particles available to bind with the TiO2 NPs, minimizing the possibility of attachment failure. Similarly, Gu et al. 27 observed good distribution and dispersion of HAp NPs as well as their low tendency to form agglomerates, which resulted in superior mechanical properties of the restorative material. Additionally, in the same study, at 5 wt.% concentration, HAp NPs have been shown to provide better chemical bonds with the GIC, resulting in fewer pores inside the material.
Flexural strength was in focus of the present study because this test permits the simulation of clinical loading conditions by providing an appropriate estimate of a material’s tensile strength, while improvements in flexural strength are closely linked to the microstructure integration. 17 In general, the size of and interconnections among NPs during the mixing process play a crucial role in enhancing the GIC flexural strength. Their small size likely allows NPs to fill larger voids in the material, thereby increasing its overall strength. 32 Generally, as the amount of TiO2 NPs increases, so does their number and surface area. However, when the amount of GIC particles is insufficient to bind with the TiO2 NPs, a larger number of free and unattached TiO2 nanoparticles will remain within the restorative material, reducing its flexural strength. 33
Unlike the results obtained in similar investigations,34,35 in the current study, the addition of TiO2 NPs failed to produce any noticeable strengthening effect in flexural strength. As can be seen in SEM micrographs under higher magnifications, a certain amount of TiO2 NPs was well distributed within the matrix, while the remaining TiO2 NPs agglomerated during the formation of the modified GICs. This phenomenon is evident in the SEM micrographs shown in Figure 7, G2 and G6 where the agglomerate formation affects the direction of adjacent fracture lines. Conversely, flexural strength values declined with the addition of MgHAp NPs, and this reduction was statistically significant in the case of Fuji IX modified with MgHAp100. Furthermore, MgHAp-modified GIC specimens were characterized by lower mean values of both flexural strength and fracture toughness compared to the controls. This might be explained by the fact that MgHAp NPs may compete with the GIC particles for the available liquid, as shown in Figure 8, G7 and G8, leading to an incomplete polysalt bridge formation and weaker overall structure.3,36 The presence of highly soluble and bioactive β-TCP molecules may affect the long-term maturation of GICs, potentially leading to poor mechanical performance of these experimental materials, although their use in previous studies has not compromised biocompatibility.20,29 This highlights the importance of a material fulfilling both biological and mechanical requirements to be suitable for clinical use.
Measuring the fracture toughness of dental materials is important for several reasons. First, dental restorative materials are constantly subjected to masticatory forces, which can cause cracks and fractures within their structure. Therefore, the ability of a dental material to resist crack propagation and fracture is crucial for its long-term clinical success. Second, fracture behavior of selected materials can help identify any weaknesses in their structure, which can be addressed through material design or processing improvements. 17 In our study, neither of the two tested materials exhibited significant changes in fracture toughness following the HAp modifications. Conversely, in line with the results reported by Elsaka et al., 35 5% wt. TiO2 modification of Ketac Molar led to a statistically significant improvement in its fracture toughness. This finding might be partially explained by the fracture surface micrographs presented in Figure 7, G6. Namely, TiO2 NP agglomerate formation might arrest the crack propagation, contributing to the greater fracture toughness of this tested material. 37 Undoubtedly, it is more likely that a large number of deagglomerated and well-dispersed TiO2 nanoparticles incorporated into the matrix (Figure 8, G2, G6) have a dominant effect on improving fracture toughness and have a more positive effect than the negative effect caused by agglomerate formation. As relatively wide variations in the fracture behavior of modified GIC materials are reported in the available literature, direct comparisons are difficult, given that these differences likely arise from different testing methods, structural inhomogeneities, microstructure disparities 34 and other intrinsic material properties.
In the present study, modifications to the Fuji IX material contributed to the increase in Vickers microhardness values. However, the difference was statistically significant only in the case of samples modified with TiO2 and MgHAp100, likely because these NPs are packed more closely on the cement surface compared to its core. 34 In contrast, all modifications of Ketac Molar material resulted in a decrease in microhardness values, which was statistically significant only in the samples containing MgHAp100. According to the manufacturer’s brochure, 5% of this GIC powder contains polycarbonate acid, which increases the overall acid content in order to improve cross-linking and consequently mechanical properties of this cement. It can thus be speculated that the addition of NPs could interfere with the cross-linking of the GIC matrix as the NPs may act as barriers to the formation of strong chemical bonds between the GIC components, leading to a weaker matrix and decreased microhardness. 38 This is particularly noticeable in the Ketac Molar sample micrographs shown in Figure 8, G7 where unreacted glass cores are clearly visible.
Restorative materials used as a base to protect the pulp from the potentially toxic effects of dental composites should have optimal flowability and desirable adaptation to an irregular cavity floor. In this regard, the risk of gap formation would be lower and the likelihood of postoperative sensitivity after placement of composite restorations would be reduced. The rheological profiles obtained in the present study showed the transition from liquid paste to elastic solid (gelation process). The increasing Gʹ value may be attributed to the cross-linking of the polyacrylic acid chains by Ca2+. It is assumed that this chemical step is the rate-limiting step for cement hardening. 39 Although the addition of all tested NPs to the Ketac and Fuji materials led to the changes in their original rheological characteristics, MgHAp1000 NPs were most beneficial, as more viscous materials with a more pronounced elastic component were obtained compared to additive-free samples as well as those containing MgHAp100 or TiO2 NPs. In addition, as viscosity decreased with frequency, the handling properties of the GICs subjected to the aforementioned modifications are expected to remain unchanged by the mechanical stress induced during their application and adaptation to the cavity walls.
The results of this study provide an understanding of the complex interactions between the added NPs and the GIC matrix and help to identify possible modifications of the microstructure to improve the mechanical properties. These results could extend the clinical application of conventional GICs to areas exposed to greater occlusal stress, particularly during their use in atraumatic restorative treatment (ART). However, due to methodological limitations, these in vitro results cannot be directly transferred to clinical settings. Not taking into account specific intraoral factors such as tooth tissue, moisture, temperature fluctuations and the different masticatory habits of patients may limit these results. While the current literature supports the application of NPs in dental materials, further studies are needed to determine the optimal conditions for their predictable and long-term incorporation into clinical performance.
Another limitation of this study worth mentioning is the small sample size per group in the flexural strength test. Therefore, further investigations with a larger number of samples are required to confirm the results obtained in this study. Future studies could additionally investigate the incorporation of NPs of different sizes, shapes, weight ratios and with functionalized surfaces for better dispersion and bonding with GIC particles and include a long-term evaluation of the mechanical properties. Finally, it is also highly recommended to standardize the size and geometry of the samples per test as well as the laboratory conditions to ensure that the results obtained are more comparable.
Conclusions
Based on the presented results and the study’s limitations, we can draw the following conclusions:
The addition of 5 wt.% MgHAp100 NP to Fuji IX GIC significantly reduced the flexural strength, while the addition of 5 wt.% TiO2 NP to Ketac Molar GIC resulted in a significant increase in fracture toughness.
The modified GICs showed different effects on surface microhardness: Fuji IX with 5 wt.% TiO2 or HAp100 exhibited significantly higher microhardness, while Ketac Molar with 5 wt.% HAp100 exhibited significantly lower microhardness compared to the respective control groups.
The SEM analysis showed a tendency of TiO2 NPs to form agglomerates in mixed GIC materials, which may affect their mechanical properties. Nevertheless, SEM micrographs revealed the good adhesion of MgHAp100 and MgHAp1000 spherically agglomerated particles with the polymeric matrix, along with a large quantity of non/reacted original glass-particles, with adverse effects on their mechanical properties.
Footnotes
Acknowledgements
The results presented in this paper were realized as a part of the project no. 142-451-3138/2023-01, titled “Nanotechnology in dentistry: Improvement of biological and physical properties of dental materials incorporated with titanium dioxide and hydroxyapatite nanoparticles,” funded by the Provincial Secretariat for Higher Education and Scientific Research, Autonomous Province of Vojvodina, Republic of Serbia.
Author contributions
BR, MC, BB, and MD researched the literature, conceived the study and were involved in protocol development. BR wrote the first draft of the manuscript. ĐV, VK, MM, and DR performed the endpoints. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
