Abstract
Objectives:
Dentin hypersensitivity (DH) is a notable painful condition with a negative impact on quality of life. This paper evaluates the occluding abilities of nanohydroxyapatite synthesized from the eggshell waste (EnHAp) in the treatment of DH.
Methods:
EnHAp was synthesized following the wet chemical precipitation method. The purity and phase change were characterized using Fourier transform infrared (FTIR) and X-ray diffraction (XRD). Field emission scanning electron microscopy (FESEM) and high-resolution transmission electron microscopy (HRTEM) were used to observe the morphology of EnHAp. The occluding capabilities were evaluated in vitro using the bovine model. Twenty-five simulated dentin specimens were randomly assigned to five groups: Group 1, untreated; Group 2, eggshell powder alone; Group 3, calcined eggshell powder; Group 4, Colgate Sensitive treated, EnHAp; and Group 5, calcium dihydrogen phosphate (n = 5). The specimens were then subjected to remineralization protocol. The tubule occluding capabilities were observed using FESEM. Analysis of variance was used to analyze the occluding ratio values for all treated specimens. Post hoc test with Bonferroni correction was used to identify the mean differences among the five test groups (α = .05).
Results:
The FTIR and XRD results confirm that synthesis of EnHAp occurred. HRTEM and FESEM revealed an irregular rod structure with a particle size of 65 nm. The occluding area ratios observed for the EnHAp specimens were significantly higher than other test groups (p < 0.001).
Conclusions:
The application of EnHAp resulted in an efficient occlusion of dentin tubule. This study suggests that EnHAp is an effective therapy to treat DH.
Introduction
Dentin hypersensitivity (DH) is a notable painful condition among dental patients. The condition occurs by the loss of the outer enamel layer and the exposure of the dentinal tubules. It has been reported in the literature that enamel covering the dentin could be lost through attrition, abrasion, erosion, and abfraction.1,2 Other causes for enamel loss includes gingival recession, which can be due to toothbrush abrasion, pocket reduction, tooth preparation for a crown, excessive flossing, or secondary periodontal diseases. 3 In South Africa, there is a high consumption of acidic drinks which could increase enamel surface erosion and the likelihood of DH.4,5
From an epidemiological viewpoint, DH is a common non-communicable public health disease affecting more than 80% of children and up to 43% of the adult population. 1 Several hypotheses have been postulated in the past decades to explain the mechanism of DH. Currently, the hydrodynamic theory proposed by Brannstrom is the most extensively used theory to explain the mechanism of DH. 6 According to this theory, any decrease in the dentinal fluid movement should result in a reduction of DH. 7 The aforementioned theory is widely supported by scanning electron microscope (SEM) evidence, which revealed that dentinal tubules in the sensitive tooth are eight times more numerous, twice larger in diameter and are open, whereas tubules in the non-sensitive tooth are less numerous, smaller, and usually blocked.8–10 Thus, and as reported by Yu et al., 11 the ideal strategy in the management of DH would be to effectively occlude the dentinal tubules to prevent fluid flow.
Significantly, the occluding abilities of different materials, such as potassium oxalates,12–15 sodium fluoride, 16 strontium salt, 1 amorphous calcium phosphate containing casein phosphopeptide, 17 calcium glycerophosphate, 18 and stannous fluoride, 19 have been well documented. However, the dentin tubules occluded by some of these materials are superficial with limited infiltration depth, showing poor acid resistance. 20 Owing to these drawbacks, the use of nanomaterials as dentin occluding material has been predicted to be the future of treating DH.21,22 Among these materials, mesoporous silica and nanohydroxyapatite (nHAp) are projected to revolutionize the treatment of DH. 11 In particular, the biocompatibility and bioactive nature of nHAp have promoted its acceptance in medicine and dentistry. 23 For example, numerous in vitro studies have reported on the potential of nHAp to effectively occlude dentin tubules.24–26 This is supported by clinical studies which indicate that nHAp provides quick relief from DH.27,28
In this present study, nHAp was synthesized from eggshell waste following the wet chemical precipitation method; 29 its dentin occluding abilities were evaluated in vitro using a bovine dentin model. Eggshell is a bioceramic composite that is highly rich in calcium carbonate (95% in the form of calcite). Recently, there has been an increase in research in the process of extracting value-added products from eggshell waste for a variety of medical and dental applications. Kattimani et al. demonstrated that eggshell-derived nanohydroxyapatite (EnHAp) showed early bone regeneration and values of density measurements equal to or more than that of the surrounding normal bone. 30 Their work supports Abdulrahman et al.’s inference that EnHAp would have excellent biocompatibility and mechanical strength that would facilitate effective tissue repair and regeneration. 31 In addition, eggshell provides a cost-effective, renewable, and sustainable source of material for EnHAp. 31 Despite the enormous potential of eggshell and its high bioavailability of calcium, there is limited research in the use of EnHAp to treat DH. This study aimed to synthesize and evaluate in vitro the dentin tubule occluding capabilities of EnHAp. The hypothesis tested was that EnHAp would significantly occlude open dentinal tubules.
Materials and method
Preparation and synthesis of EnHAp from eggshell waste
The preparation and synthesizes of EnHAp from eggshells waste followed three steps. In the first step, eggshell waste collected from fast food outlets within Durban, South Africa was prepared according to the procedure reported by Onwubu et al. 32 The eggshells were washed with deionized water to remove surface impurities. Thereafter, they were disinfected by storing them in a dilute solution of household sodium hypochlorite for 6 h, and subsequently vacuum dried for 6–9 min at 250°C. Using a planetary ball mill (Retsch PM 100), the eggshells were dry milled for 50 min to obtain a fine powder. The conditions of the milling included a 500 mL stainless steel jar (inner diameter of 100 mm) and 50 stainless steel balls of 10 mm diameter. The weight of eggshells milled was 30 g. In the second step, the prepared eggshell powder was calcined in a furnace at a different temperature to obtain a snow white powder. In the first heating temperature, eggshell powder (10 g) was heated for 1 h at 300°C at a heating rate of 3°C/min. The temperature of the furnace was raised to 900°C and the powder was further calcined for 3 h. The temperature of the furnace was allowed to cool down to room temperature. In the third step, the calcined snow white powder (2 g) was mixed with calcium dihydrogen phosphate (1.36 g) in a 150 mL beaker containing 100 mL deionized water. Subsequently, the beakers were placed in the heating chamber which was kept fixed at 37°C. The reaction was allowed to complete for 1 week. The pH of the solution was constantly monitored to maintain a constant pH of 12. After 1 week of reaction, the white precipitate obtained was separated from the solution by washing with deionized water and centrifuged for 30 min. The resulting powder was dried in the oven at 37°C for a week. The white powders obtain thereafter were characterized to establish the successful synthesis of EnHAp.
Methods of characterization
Fourier transform infrared spectroscopy
The Fourier transform infrared (FTIR) spectra were measured using a Perkin Elmer Universal ATR spectrometer to identify the functional group constituents of eggshell powder, the calcined eggshell powder, and EnHAp. A very small amount of each sample was placed in the sample holder. An initial background check was performed before scanning in the range 400–4500 cm−1 at a resolution of 4 cm−1.
X-ray diffraction
X-ray diffraction (XRD) analysis was performed to observe the possible changes in crystallinity between the eggshell powder, calcined eggshell powder, and EnHAp. The XRD patterns were recorded using a diffractometer (PANalytical-Empyrean instrument; Co radiation 1.54056 Å) and analyzed between 0 and 90° (2θ). The voltage, current, and pass time used were 40 kV, 40 mA, and 1 s, respectively.
Field emission scanning electron microscopy
An SEM (field emission, Carl Zeiss) operating in controlled atmospheric conditions at 20 kV was used to examine the surface morphology of the samples. Prior to SEM observation, the surface was coated with a thin, electric conductive gold film to prevent a build-up of electrostatic charge.
High-resolution transmission electron microscopy
A transmission electron microscope (TEM) was used to observe the particle size, shape, and distribution of EnHAp. Very small quantities of the EnHAp powder were dispersed in 10ml ethanol and sonicated at 10 kV for 10 min. Subsequently, thin cross-sections of cryo-microtomed specimens were prepared using a Leica microtome (South Africa) and placed on carbon copper grids. The analysis was conducted using a TEM (Philips CM 120 model) operating at 120 kV.
pH test
1.5 g of each sample (Table 1) were placed in a beaker containing 50 mL deionized water. The solution was constantly agitated at a low speed of 600 r/min and the pH measured at 1 min intervals for a duration of 10 min. A pH meter (Starter 300, Ohaus Incorporation, USA) equipped with a temperature sensor was used to record changes in the pH reading. Similarly, the samples were exposed to 2 mol L−1 hydrochloric acid (HCl) and the pH recorded after 10 min.
Sample groups.
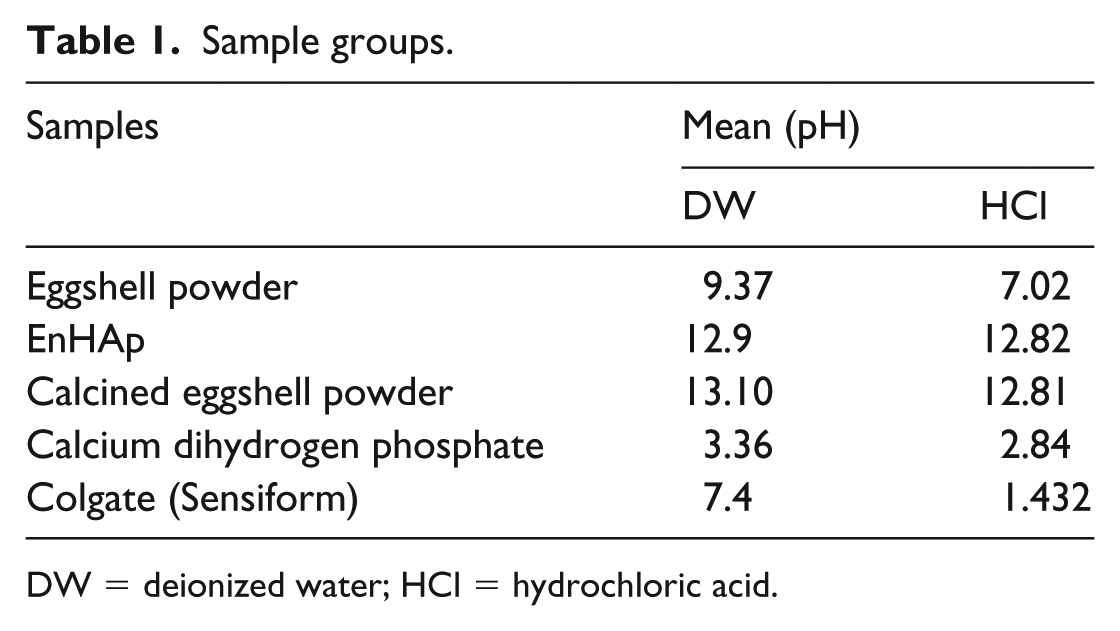
DW = deionized water; HCl = hydrochloric acid.
Dentin tubule occlusion test
Twenty-five freshly extracted bovine-enamel anterior teeth were obtained from a slaughterhouse, South Africa. The collected teeth were subsequently cleaned and disinfected in 10% chloroxylenol solution. Dentin discs measuring 5 mm × 5 mm × 1 mm were prepared by sectioning perpendicular to the long axis of the teeth below the enamel-dentinal junction using a low-speed diamond saw under water cooling conditions. Subsequently, the prepared dentin disc was wetly grounded with silicon carbide polishing papers (600–1000 grits) for 60 s. Before simulating the sensitive model, the discs were mounted in a resin (AMT composite, South Africa). Thereafter, dentin tubules were opened by soaking the specimens in 1 wt % citric acid solution for 5 min. The specimens were randomly assigned into five groups (n = 5). The specimens were then agitated in a beaker containing 1g of the samples (Table 1) and 40 mL deionized water for 5 min. After exposure, the specimens were rinsed with deionized water and blot tried.
SEM evaluation of the occluded specimen
An SEM (field emission, Carl Zeiss) operating in controlled atmospheric conditions at 20 kV was used to evaluate the occluded dentin pre- and post-treatment. Prior to SEM observation, the surface was coated with a thin, electric conductive gold film to prevent a build-up of electrostatic charge. Additionally, the ratios of occluded and opened tubules were computed using ImageJ software (National Institute of Health USA, http://imagej.nih.gov./ij). This was calculated by dividing the area of occluded tubules by the total tubules area using ×3000 magnification images (n = 5).
Statistical analysis
The mean values of the occluded area ratio in SEM study were evaluated with one-way analysis of variance (ANOVA) with statistical software (IBM SPSS Statistics v24; IBM Corp), This was followed by a multi-comparison test with Bonferroni correction (α = .05).
Results
Characterization
The FTIR spectra of eggshell powder, calcined eggshell powder, and EnHAp in Figure 1 revealed several bands from 500 cm−1 to 4000 cm−1. Visible differences were observed in the band aspect of the FTIR spectra difference between the eggshell powder (Figure 1(a)), calcined eggshell powder (Figure 1(b)), and EnHAp (Figure 1(c)). The prominent absorption peak at 1411 cm−1 observed in Figure 1(a) is associated with the presence of carbonates in the eggshell powder. Furthermore, the FTIR spectra show the absorption peak of calcite at around 711 and 873 cm−1. These were attributed to asymmetric and symmetric stretching, out-of-plane bending and in-plane bending vibration modes for calcium carbonate (CO32−) molecules. Although the carbonates peaks were identified in the calcined eggshell powder in Figure 1(b), they were, however, reduced in intensity. In addition, the prominent peak at 3450 cm−1 corresponding to the O–H bending was found in the calcined eggshell powder.
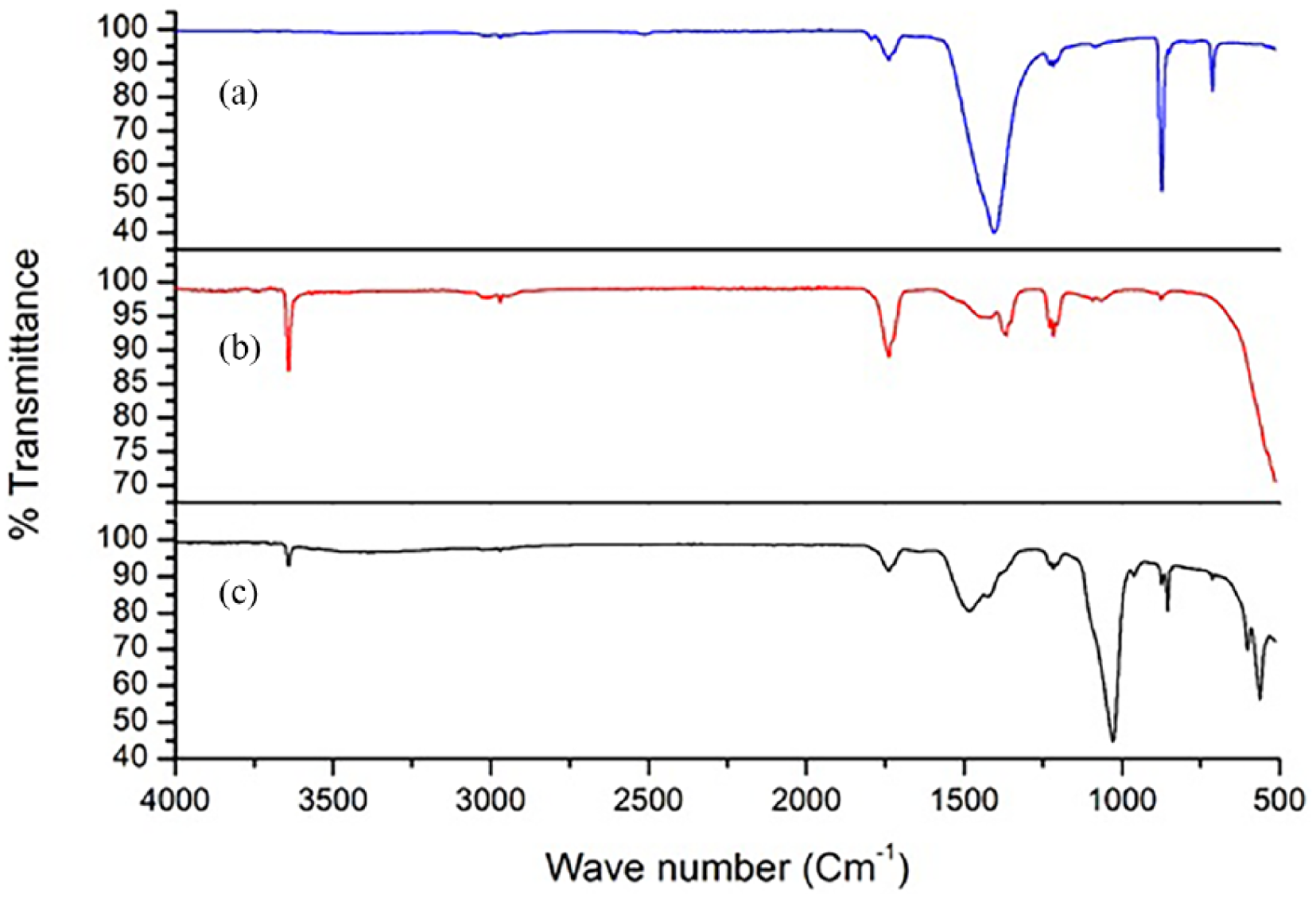
FTIR spectra of (a) eggshell powder, (b) calcined eggshell powder, and (c) EnHAp.
On the other hand, and as shown in Figure 1(c), the peak observed around 500 cm−1 corresponded to the PO4 bending in EnHAp. Similarly, the absorption peak found at 980 cm−1 was attributed to the P–O asymmetrical stretching of a PO4 group. The hydroxyl O–H bending was identified around 3450 cm−1 and 870 cm−1 was attributed to water molecules. Added to these, the absorption bands that appeared at 1450 cm−1 were indicative of the presence of the carbonate ion, which could have resulted from the incomplete reaction of the carbonates present in the calcined eggshell powder. All the FTIR spectra observed for EnHAp were in agreement with those reported in the literature for HAp.33–35
The XRD patterns of eggshell powder, calcined eggshell powder, and EnHAp are shown in Figure 2. At a glance in Figure 2(a), the characteristic peaks marked at around 27.5°, 34.5°, 42.5°, 46.5°, 50.5°, 56°, 57°, 67°, 68°, and 72° (2θ) confirm the presence of calcite in the eggshell powder. No other crystalline phase was observed. On the other hand, the structure of eggshell powder appeared to change upon calcination at 900°C showing different diffraction angles marked at 20.8°, 40°, 39.7°, 55.3°, 59.8°, and 64.1° (Figure 2(b)). It was observed that the calcite peak (calcium carbonate) decreased while the intensity of a new peak marked at around 40° (2θ) increases. This new peak could be attributed to the decomposition of the calcium carbonate constituent of eggshell powder to form calcium oxide. 36 In terms of the XRD pattern observed in Figure 2(c), the diffraction angles marked at 20.9°, 30°, 30.5°, 31.7°, 32.6°, and 35.8° suggest the formation of EnHAp. 36 Notwithstanding this, another crystalline peak observed at 33.4° and 34.5° corresponds to the aragonite and calcite phase of calcium carbonate, respectively. The presence of both aragonite and calcite in EnHAp could be attributed incomplete reaction of the calcined eggshell powder and calcium dihydrogen phosphate. This aligns with other EnHAp powders produced by other research works,37,38 where traces of unreacted Ca(OH)2 and H3PO4 were observed.
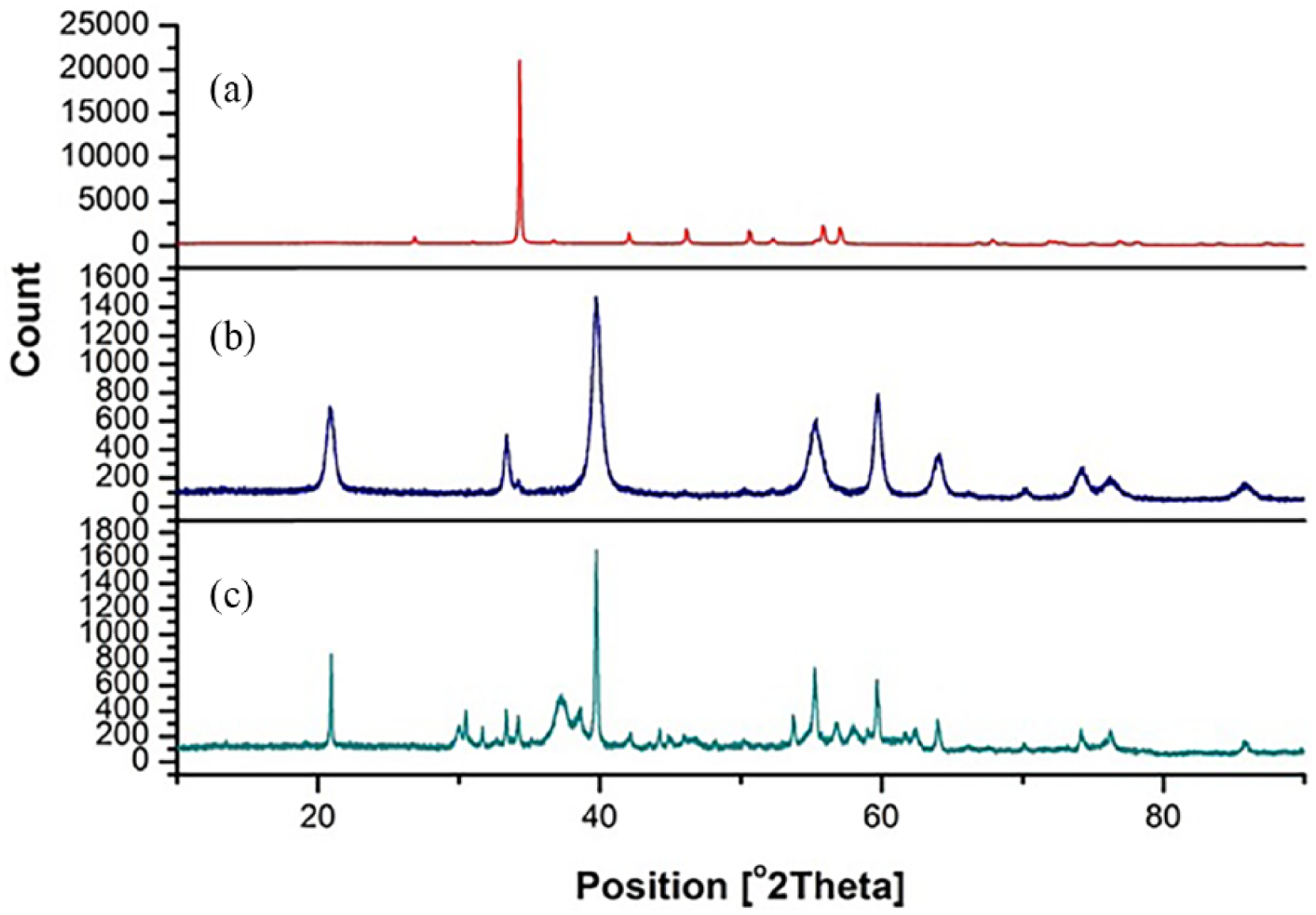
XRD pattern of (a) eggshell powder, (b) calcined eggshell powder, and (c) EnHAp.
The mineralogical content derived from the XRD analysis further revealed differences in the mineral composition of eggshell powder, calcined eggshell powder, and EnHAp (Table 2). While the calcined eggshell powder was mainly portlandite (100%), different mineral content was found in eggshell powder and EnHAp. The eggshell powder contains calcite (97.9%), apatite (0.8%), and graphite 2H (2.1%), whereas EnHAp contains calcite (2.5%), apatite (74.8%), aragonite (8.9%), and portlandite (13.8%).
Mineralogical composition of eggshell powder, calcined eggshell powder, and EnHAp, as determined by XRD analysis.
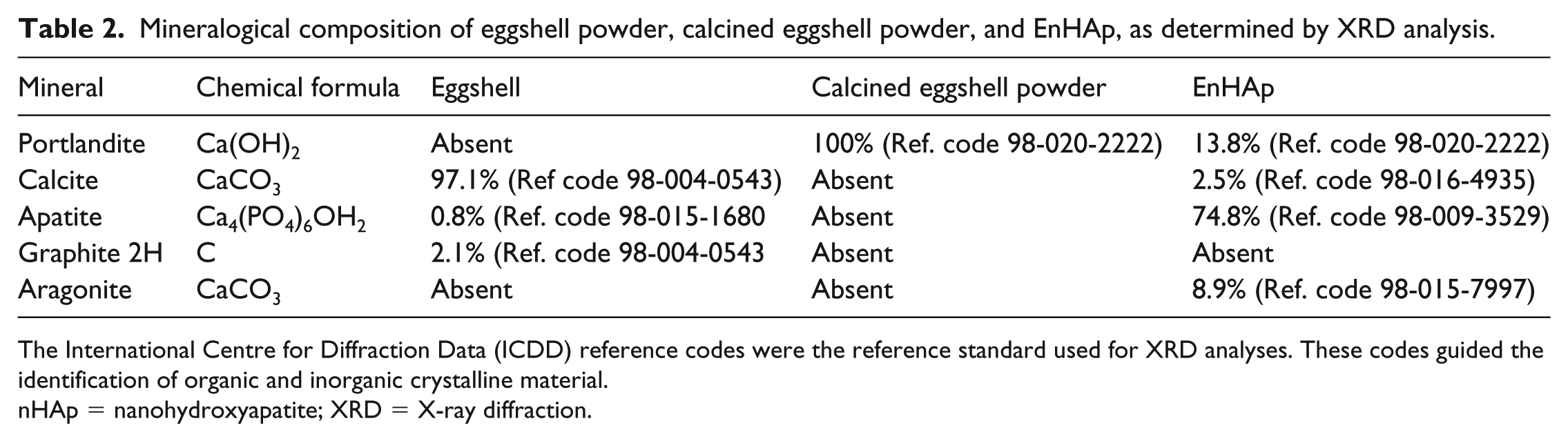
The International Centre for Diffraction Data (ICDD) reference codes were the reference standard used for XRD analyses. These codes guided the identification of organic and inorganic crystalline material.
nHAp = nanohydroxyapatite; XRD = X-ray diffraction.
The SEM images of the eggshell powder, calcined eggshell powder, and EnHAp are given in Figure 3. As shown in Figure 3(a), the presence of an irregularly shaped particle was observed for eggshell powder. In contrast, and in the case of the calcined eggshell powder (Figure 3(b)), regular rod-like particles was observed, and that of calcium dihydrogen triphosphate was rectangular in shape (Figure 3(c)). However, it can be seen from the SEM image (Figure 3(d)) of EnHAp that the particles are nearly round with a diameter of about 63 nm.

SEM micrographs for (a) eggshell powder, (b) calcined eggshell powder, (c) calcium dihydrogen triphosphate, and (d) EnHAp.
A TEM image of EnHAp is shown in Figure 4. An irregular-rod like structure was observed. It was also noted that the particles assembled into short chains, and then some chains stick together into clusters with a dimension of 80–100 nm in length. The morphology of EnHAp observed is in agreement with that reported by Alobeedallah et al. 33

TEM image of the synthesized EnHAp.
Occluding test comparison
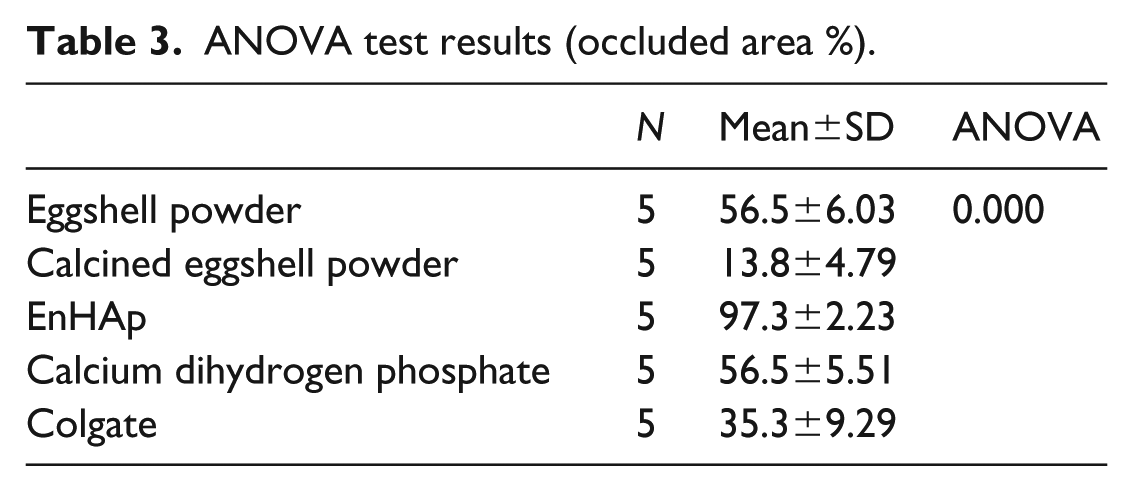
The one-way ANOVA, mean, and standard deviation of the dentin specimens after treatment are given in Table 3. Notably, the occluded area ratio (%) for the specimens treated with eggshell powder, calcined eggshell powder, EnHAp, calcium dihydrogen phosphate, and Colgate paste were statistically different (p < .001). The dentin specimens treated with EnHAp had the highest percentage tubule occluded area (97.3 ±2.23%), while the calcined dentin group treated with calcined eggshell powder had the lowest percentage tubule occluded area (13.8 ±4.78%).
ANOVA test results (occluded area %).
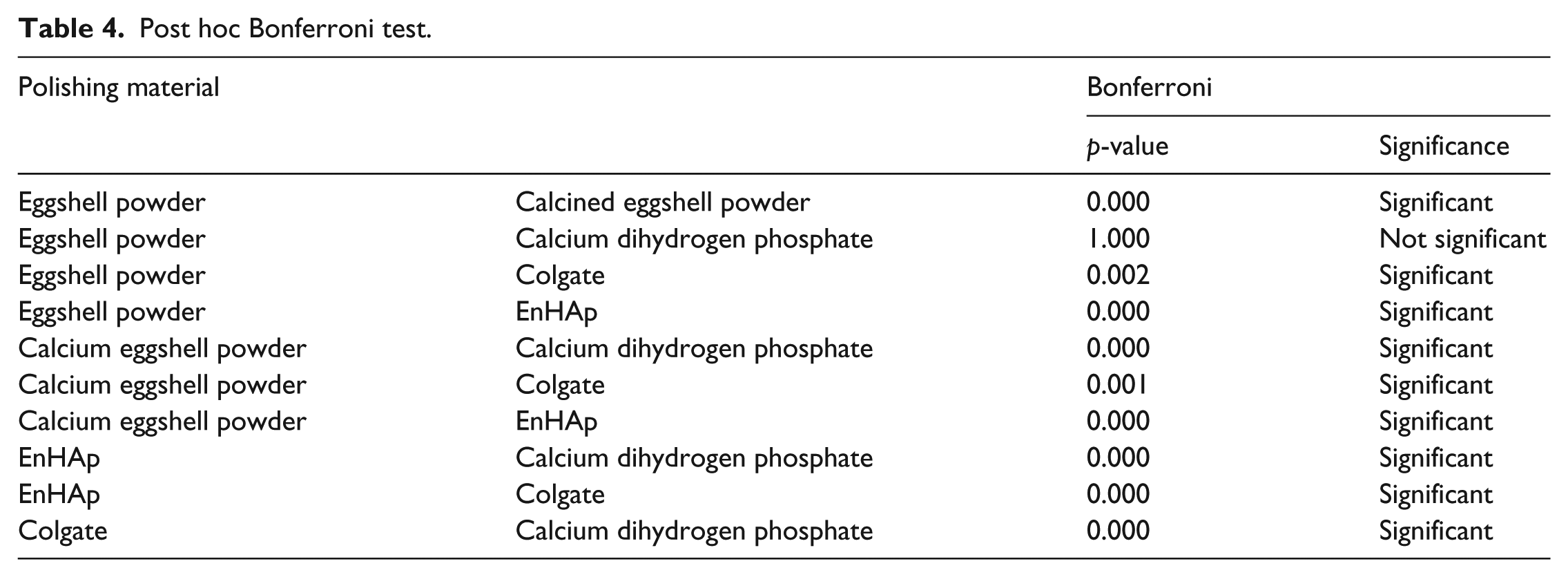
The results of the post hoc comparison test are given in Table 4. The % tubule occluded for the EnHAp group were significantly higher than those observed for other test groups (p < .001). No statistical differences were found between the % tubule occluded area for the groups treated with eggshell powder and calcium dihydrogen phosphate (p > .05). However, a statistical difference was found between the group treated with Colgate and calcined eggshell powder (p < .001). In addition, the groups treated with both eggshell powder and calcium dihydrogen phosphate were significantly higher than those treated with Colgate and calcined eggshell powder (p < .001).
Post hoc Bonferroni test.
The SEM images of the untreated and treated dentin specimens are seen in Figure 5. The images reveal occlusion differences between the EnHAp treated specimens with the other test groups (eggshell powder, Colgate, calcined eggshell powder, and dihydrogen phosphate). The dentin specimen treated with EnHAp revealed a remineralized surface with nearly all the tubules occluded (Figure 5(e)). In contrast, and as shown in Figure 5(c) and (f), open tubules were visible on the dentin specimen treated with calcined eggshell powder and Colgate. On the other hand, specimens treated with eggshell powder (Figure 5(b)) and calcium dihydrogen phosphate (Figure 5(d)) to some extent demonstrate tubule occlusion.

Representative SEM micrograph for the (a) untreated dentin specimen, (b) treated eggshell powder, (c) calcined eggshell powder, (d) calcium dihydrogen triphosphate, (e) EnHAp, and (f) Colgate paste (agitated for 5 min, ×5000 original magnification).
Discussion
DH is a public health concern with a high prevalence rate. This study evaluates the occluding capabilities of nHAp synthesized from eggshell waste (EnHAp) as a potential therapy material. Different characterization technique was used to evaluate the purity and phase formation of the synthesized EnHAp. The results confirmed the successful production of nanocrystalline hydroxyapatite from eggshell waste, albeit, with some impurities of unreacted calcium oxide and calcium dihydrogen phosphate (Figure 2(c)). The occluding capabilities were evaluated in vitro using bovine dentin specimens. The findings led to the acceptance of the research hypothesis as the dentin specimens after treatment were significantly occluded by the EnHAp (p < .001).
Evidently, the open dentin tubules treated with EnHAp achieved outstanding tubule occlusion (Figure 5(e)). In light these, it can be inferred that the synthesized EnHAp could be useful for the management of DH. This is according to the hydrodynamic theory that is widely accepted that any substance occluding dentin tubules invariably reduces the clinical symptom of DH. 39 Given the statistical differences in the % occluded area values between the EnHAp other test groups (p < .001), the observed differences could be attributed to particle sizes and the reactivity of EnHAp. According to Besinis et al., 40 the size and high biological activity and reactivity of nHAp particles facilitate the binding of the particles to dentin apatite which subsequently infiltrates the dentinal tubules. Moreover, the occluding capabilities observe for EnHAp (Figure 5(e)), were comparable to those reported by Baglar et al. 41
Furthermore, the result of this study demonstrated that eggshell powder (Figure 5(b)) and calcium dihydrogen (Figure 5(d)) showed equal effectiveness in occluding dentin tubules as well as the formation of layer over and within the dentin tubules, while the occluding capabilities of Colgate paste (Figure 5(f)) and the calcined eggshell powder (Figure 5(c)) were inferior. The comparable occluding capabilities observed for eggshell powder with that of calcium dihydrogen phosphate could be attributed to the calcium and phosphate content in eggshell powder (Table 2). This is in agreement with the results of Mony et al., 42 which showed that the high bioavailability of calcium in eggshell together with the phosphate content supports its remineralization capabilities. On the contrary, the absence of phosphate in the calcined eggshell powder (Table 2) could have contributed to its inferior occluding abilities. It was noted that a new mineral portlandite was created when eggshell powder was calcined at 900°C (Table 2). As highlighted in the Hand Book of Mineral Data, 43 portlandite occurs naturally as calcium hydroxide and is known to be soluble in water. This compound may have been formed following the moisture absorption the calcined eggshell powder.
Equally important, the inferior occluding capabilities Colgate paste could be attributed to the absence of occluding agents in the tested brand. This supports the report by Schiff et al. that desensitizing toothpastes like Colgate toothpaste are designed to deliver potassium ions that acts as a nerve depolarizing agents in the treatment of DH. 44 This notwithstanding, Colgate brands like calcium–arginine combination have been reported to be highly effective in occluding dentin tubules.45,46
Conclusion
Although EnHAp was successfully synthesized, there was, however, the presence of other crystalline carbonate structure in the material. Despite this, the EnHAp showed outstanding occluding capabilities that suggest its potential in the treatment of DH. More importantly, this study anticipates that using EnHAp in the treatment of DH will contribute massively to environmental management as well as reducing the carbon footprint which is high in global agenda.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of South Africa (grant number 104824).
