Abstract
Objective:
Dentin hypersensitivity (DH) is a common oral disease with approximately 41.9% prevalence. Reconstruction of dental hard tissues is the preferred treatment for relieving DH. Here, we applied biomineralization method using oligopeptide simulating cementum protein 1 (CEMP1) to regenerate hard tissues on demineralized dentin.
Methods:
The self-assembly and biomineralization property of the oligopeptide were detected by scanning electron microscopy (SEM), circular dichroism spectroscopy, and transmission electron microscopy. Oligopeptide’s binding capacity to demineralized dentin was evaluated by SEM and attenuated total reflection Fourier transform infrared spectroscopy (ATR-FTIR). Remineralization was characterized using SEM, ATR-FTIR, X-ray diffraction, and nanoindentation. Oligopeptide’s biocompatibility was evaluated using periodontal ligament cells.
Results:
Oligopeptides self-assembled into nano-matrix and templated mineral precursor formation within 24 h. Moreover, oligopeptide nano-matrix bound firmly on demineralized dentin and resisted water rinsing. Then, bound nano-matrix served as a template to initiate nucleation and transformation of hydroxyapatite on demineralized dentin. After 96 h, oligopeptide nano-matrix regenerated an enamel-like tissue layer with a thickness of 15.35 μm, and regenerated crystals occluded dentin tubules with a depth of 31.27 μm. Furthermore, the oligopeptide nano-matrix had good biocompatibility when co-cultured with periodontal ligament cells.
Conclusions:
This biomimetic oligopeptide simulating CEMP1 effectively induced remineralization and reconstructed hard tissues on demineralized dentin, providing a potential biomaterial for DH treatment.
Keywords
Introduction
Dentin hypersensitivity (DH) is a common oral pain condition, which is prevalent in approximately 41.9% of the adult population. 1 DH can be defined as a transient and sharp pain or discomfort in response to external stimuli such as temperature, chemicals, osmotic pressure, and mechanical effects. According to the hydrodynamic theory, the liquid in dentin tubules flows multi-directionally after exposure to external stimuli, thereby activating nociceptors in the pulp/dentin border area and causing pain. 2 For DH occurring, the dentin surface must be exposed, and many dentin tubules near to one another must be patent from the pulp to the oral environment. Thus, two treatment modalities are used to treat DH, namely, stabilizing the pulpal nerve and altering fluid flow in the tubules. 3 For the first category, the potassium salt is a common agent to reduce interdental nerve excitability and inhibit interdental nerve function by increasing the concentration of local extracellular potassium ions. However, potassium efficacy is currently debated with no strong evidence supporting its efficacy. 4 As for the second category, topically introducing functional compounds can form insoluble materials precipitating in the tubules or/and on its surface. Facilitating natural mineral formation is also considered to be effective. 5 At present, most of the compounds such as strontium, 6 calcium phosphate, 7 soluble oxalates, 8 and bioactive glasses 9 function by physically blocking open dentin tubules. However, their efficacy is limited, and the precipitating materials/or treating agents can be washed by saliva and are vulnerable to mechanical abrasion.
The main inorganic component of enamel and dentin is highly ordered hydroxyapatites, of which formation and growth are controlled and regulated by natural noncollagenous proteins (NCPs) in vivo. For example, amelogenins play major structural roles during the oriented growth of enamel. 10 Dentin phosphoprotein, one main NCP in the dentin extracellular matrix, plays a crucial role in dentin mineralization. 11 Cementum protein 1 (CEMP1), only expressed in the cementum, influences the formation process of hydroxyapatite during cementum formation. 12 Those natural NCPs are often highly anionic due to the presence of many charged amino acids, such as aspartic acid, glutamic acid, and phosphorylated serine, and thereby can attract calcium ions to template biomineralization. 13 Based on this, the biomineralization method has the potential to reconstruct a remineralized layer on exposed dentin and/or within dentin tubules, hence protecting the pulp nerve from external stimuli and altering its fluid flow. However, the wide application of natural NCPs in practice remains an unmet challenge because their extraction and purification are not readily obtainable in vitro. Biomimetic oligopeptides that contain repetitive sequences of functional amino acid residues can partially imitate the functions of natural NCPs to promote hydroxyapatite formation. According to this speculation, many peptides or oligopeptides have been fabricated to reconstruct dental hard tissues such as enamel-like tissue and/or induce dentin tubule occlusion. For example, an amphiphilic oligopeptide imitating amelogenin has been used to fabricate an enamel-like tissue on the dentin surface. 14 Eight repetitive nucleotide sequences of glutamate-serine-serine derived from dentin phosphoprotein have been proved to have potent potential in the DH treatment due to their ability to induce mineralization and the occlusion of dentin tubules. 15 Those biomimetic peptides with potent biocompatibility facilitate the natural mineral formation, offering the potential for DH treatment.
In the previous study, a novel amphiphilic oligopeptide simulating CEMP1 was demonstrated to be an active mineralization agent by inducing intrafibrillar mineralization of reconstituted collagen. Amphiphilic oligopeptide is a self-assembling peptide containing a hydrophobic alkyl tail attached to the bioactive peptide sequence. Through self-assembly, these bioactive peptides are repeatedly arranged along the surface of the assembled nano-matrix, thereby acting as a template to induce mineralization. 16 Therefore, this amphiphilic oligopeptide simulating CEMP1 has a potent potential to reconstruct enamel-like tissue and induce tubule occlusion via biomineralization. However, the previous study has only demonstrated oligopeptide’s ability to induce the mineralization of reconstituted collagen and focused on identifying the relevant mechanism of mineralization. The evidence for facilitating the reconstruction of dental hard tissues on demineralized dentin for the potential of DH treatment under a simulated oral environment is still limited. Therefore, this study focused on the effectiveness of biomimetic mineralization on the regeneration of dental hard tissues on demineralized dentin by using this biomimetic oligopeptide simulating CEMP1. It was hypothesized that the biomimetic oligopeptide can reconstruct an enamel-like tissue on demineralized dentin and induce dentin tubule occlusion.
Materials and methods
Synthesis of oligopeptide-imitating CEMP1
The amphiphilic oligopeptide was composed of a bioactive peptide sequence imitating CEMP1 (NNCCCCRRES(p)) and a stearic acid derivative (C16H31COOH). Oligopeptides were fabricated through a solid-phase peptide synthesis method based on the 9-fluorenylmethoxycarbonyl (Fmoc) protection chemistry 17 using a peptide synthesizer (PSI-200 model, Peptide Scientific Inc., New York, USA). In brief, the rink amide resin was used as the carrier in the synthesis process, and amino acid coupling reactions were performed using the Fmoc-capped amino acids. Afterward, a hydrophobic palmitic acid was conjugated to the free N-terminus of the synthesized peptides. After the alkylation reaction, the oligopeptide was cleaved from the resin. Then, the synthesized oligopeptide simulating CEMP1 was purified and characterized using high-performance liquid chromatography (ACTA Purifier, Amersham, Arlington Heights, USA) and mass spectrometry (LTQ Orbitrap XL, Thermo Fisher Scientific, Waltham, Massachusetts, USA).
Self-assembly and mineralization property of the oligopeptide
Two milligrams of oligopeptide powder was dissolved in 1 mL of deionized water to obtain the oligopeptide stock solution with a concentration of 2 mg mL−1. An equal volume of 1 M CaCl2 and oligopeptide stock solution was mixed to trigger self-assembly. The assembled oligopeptide was named as oligopeptide nano-matrix in the following text. Therefore, the concentration of the oligopeptide nano-matrix was 1 mg mL−1. The suspension was ultrasonically blended to improve calcium ion diffusion and then incubated at 37°C for 24 h.
The oligopeptide nano-matrix was first detected by far-UV circular dichroism spectroscopy (Chirascan qCD, Applied Photophysics, Surrey, UK). In brief, the oligopeptide nano-matrix (1 mg mL−1) was ultrasonically diluted into 0.1 mg mL−1 clear solution using phosphate buffer solution (pH = 7.4) and then detected three times at 190–280 nm using a spectropolarimeter in a cell with a 0.1 cm path length. 18 Afterward, the buffer spectrum was subtracted. Oligopeptide nano-matrix was then evaluated under a field emission scanning electron microscope (FE-SEM, Gemini 500, Zeiss, Oberkochen, Germany). Also, 0.5 mL of oligopeptide nano-matrix (1 mg mL−1), was diluted with 4.5 mL of ethanol. The final concentration of the diluted oligopeptide was 0.1 mg mL−1 and subsequently coated on transmission electron microscope (TEM) grids. All samples were washed with deionized water for 30 s to remove remnant salts and then dehydrated with alcohol (30%, 50%, 70%, 90%, 95%, 100%, and 100%, incubated in each gradient for 20 min) before examination.
TEM grids coating oligopeptide nano-matrix were incubated in 15 mL of artificial saliva 19 at 37°C for 24 h to evaluate its mineralization property. Afterward, all samples were washed with deionized water for 30 s and dehydrated with gradient alcohol. FE-SEM and TEM (JEM-2100, JEOL, Akishima, Japan) were used to study the templating mineralization property of oligopeptide nano-matrix. The mineral structure was studied by electron diffraction, and the calcium and phosphorus components were detected by energy-dispersive spectrometry (X-max 80, Oxford).
Preparation and characterization of the demineralized dentin
Human third molars were selected to prepare the demineralized dentin slabs. The freshly extracted molars were obtained with informed consent from patients according to a protocol approved by the Ethics Committee of Nanjing Stomatological Hospital, China (number: 2019NL-065(KS)). The extracted human tissue was the third molars that were without any caries and defects. The human third molars are common teeth to become impacted. The impacted third molars often fail to erupt or develop into the proper functional location. Moreover, they are often non-functional, abnormal, and related to the pathology. Therefore, the impacted third molars are often extracted and used as the clinical samples for the research studies.19,20 The freshly extracted molars were stored in 70% ethanol solution at 4°C for 1 month until use. Collected teeth were thoroughly cleaned and then stored in 70% ethanol solution at 4°C for 1 month until use. Coronal dentin (2 mm in thickness) that is perpendicular to the tooth long axis was cut from each tooth using a low-speed diamond saw (SYJ160, HF-Kejing, Shenyang, China). Then, the coronal dentin was sectioned into 3 mm × 3 mm slabs. The surface of dentin slabs was successively polished using 400, 800, 1200, 1600, and 2000-grit silicon carbide papers under running water. Afterward, the dentin slabs were ultrasonically washed using detergent water, anhydrous acetone, ethyl alcohol, and deionized water for 15 min in each solution. Demineralized dentin was acquired by using 37% phosphoric acid for 30 s and rinsed thoroughly with deionized water. Demineralized dentin slabs were gradient dehydrated and stored in a drying tower for use.
Binding property of oligopeptide nano-matrix to demineralized dentin
ATR-FTIR (Nexus870, Nicolet, Madison, Wisconsin, USA) and FE-SEM were used to study the binding property of oligopeptide nano-matrix to demineralized dentin. A total of nine demineralized slabs were prepared to assess the binding property with oligopeptide nano-matrix. Six demineralized dentin slabs were immersed in 5 mL of oligopeptide nano-matrix (100 µg mL−1) and then incubated at 37°C for 24 h. Afterward, half of the samples were rinsed with deionized water for 30 s to remove redundant salts and then air-dried. The remaining samples were rinsed with sufficient deionized water for about 5 min to study the anti-washing capacity of the oligopeptide nano-matrix and subsequently air-dried. Demineralized dentin and all the samples were then investigated under ATR-FTIR.
Remineralization in artificial saliva
Demineralized dentin slabs were randomly divided into the experimental and control groups. The experimental samples were coated with oligopeptide nano-matrix and incubated at 37°C for 24 h, whereas those in the control group were placed in deionized water for the same time. All samples were incubated with 20 mL of artificial saliva at 37°C for 24, 48, and 96 h. Artificial saliva was replaced every 24 h. After mineralization, remineralized dentin slabs were thoroughly rinsed with deionized water and dehydrated with gradient alcohol.
FE-SEM was applied to evaluate the morphology of remineralized dentin, including surface and cross-sectional views. For cross-sectional detection, samples were pre-cooled in liquid nitrogen for 30 s and sectioned perpendicular to the dentin surface. Energy-dispersive spectrometry was used to study the Ca/P ratio of remineralization. All samples were sputter-coated with gold to enhance electrical conductivity before the examination.
The composition and structure of remineralization were detected by X-ray diffraction (X’TRA, Thermo Fisher Scientific, Waltham, Massachusetts, USA) at 2θ ranging from 10° to 70°. The acquired data were analyzed using Origin 2020 (OriginLab, Northampton, Massachusetts, USA) and compared with the standard diffraction peak cards of hydroxyapatites (JCPDS No. 09-4032). ATR-FTIR was used to detect the surface functional groups of remineralization.
The mechanical properties of remineralization were determined under a nanoindenter (HV-1000, Shanghai Microcre Optics-Mech Tech, Shanghai, China). Firstly, three demineralized dentin slabs were selected randomly to record their Vickers hardness. After mineralization for 96 h, the Vickers hardness of the remineralized dentin slabs was detected. Three samples in the experimental group and the control group were selected randomly, and 10 sites in each sample were detected. The adjacent detecting site was kept at the same distance of 150 μm. Nano-indentations were tested in 4 × 4 mm slabs with a uniform load of 200 g for 15 s loading time. Mean values with standard deviation for the Vickers hardness of each group were recorded and calculated. The data was then analyzed using Origin 2020 with one-way ANOVA (p < 0.05).
Biocompatibility analysis
Biocompatibility of the oligopeptide nano-matrix was evaluated using periodontal ligament cells (PDLCs). Cell Counting Kit-8 (CCK-8) assay (Dojindo Laboratories, Kumamoto, Japan) was used to determine the viability and proliferation of PDLCs on the oligopeptide nano-matrix. In brief, the oligopeptide nano-matrix was mixed with Dulbecco’s Modified Eagle’s Medium (DMEM) containing 20% fetal bovine serum and 1% penicillin/streptomycin. The final concentrations of the oligopeptide nano-matrix were 0, 5, 25, 50, and 100 μg mL−1. PDLCs were seeded into 96-well plates at a density of 4 × 104 cells per mL, and the DMEM mixture was added for 1, 3, 5, and 7 days of culturing. At each time interval, the 96-well plates were gently washed with phosphate buffer solution and then incubated in 100 μL DMEM containing 10 μL of CCK-8 solution for 1.5 h. Afterward, the absorbance microplate reader (SpectraMAXM3, Molecular Devices, San Jose, California, USA) was used to determine each group’s absorbance at 450 nm. Origin 2020 was used to analyze the optical density of each group and compared statistically using the t-test (p < 0.05).
The live/dead viability kit (Dojindo Laboratories, Kumamoto, Japan) was used according to the manufacturer’s instructions. In preparing the working solution, calcein and propidium iodide stock solutions were diluted with phosphate buffer solution into final concentrations of 2 and 4.5 μM, respectively. PDLCs at a density of 4 × 104 cells were seeded in laser confocal petri dishes. The experimental group was cultured with the DMEM mixture containing 100 μg mL−1 of the oligopeptide nano-matrix, whereas the control group was cultured with pure DMEM. After culturing for 7 days, the cells were washed with phosphate buffer solution and then incubated in the working solution for 15 min at 37°C. Then, all samples were observed under a laser scanning confocal microscope (Nikon Ti, Nikon, Tokyo, Japan). Viable, intact cells on the oligopeptide nano-matrix were identified by green fluorescent calcein at 490 nm, whereas dead or damaged cells were labeled with propidium iodide at 545 nm.
Results
Self-assembly and mineralization property of the oligopeptide
In this study, the CaCl2 solution was used to trigger the self-assembly of oligopeptides. Figure 1(a) and (b) were respectively SEM and TEM images of the amphiphile oligopeptide simulating CEMP1 after self-assembly. The oligopeptide self-assembled into the supermolecular nano-matrix. Figure 1(c) was the size distribution of the oligopeptide nano-matrix with a diameter of approximately 20.57 ± 4.50 nm. Figure 1(d) was the circular dichroism spectrum of the oligopeptide nano-matrix. The spectrum exhibited a negative band at 205 nm and a positive band at approximately 194 nm, indicating a helix structure of the oligopeptide nano-matrix.

Self-assembly of the oligopeptide simulating CEMP1: (a) SEM, (b) TEM image, (c) diameter distribution, and (d) circular dichroism.
Figure 2(a) was the SEM image of the oligopeptide nano-matrix after mineralizing for 24 h. Pearl necklace-like particles were anchored over the host oligopeptide nano-matrix. Figure 2(b) was the size distribution of pearl necklace-like particles with an average diameter of 39.34 ± 8.64 nm. Figure 2(c) and (d) were the elemental analysis, which showed the clear distribution of Ca and P corresponding to the pearl necklace-like particles. Figure 2(e) and (f) were the TEM image and electron diffraction of the mineralized area, respectively. The mineralized nano-matrix exhibited an almost diffused ring diffraction pattern.
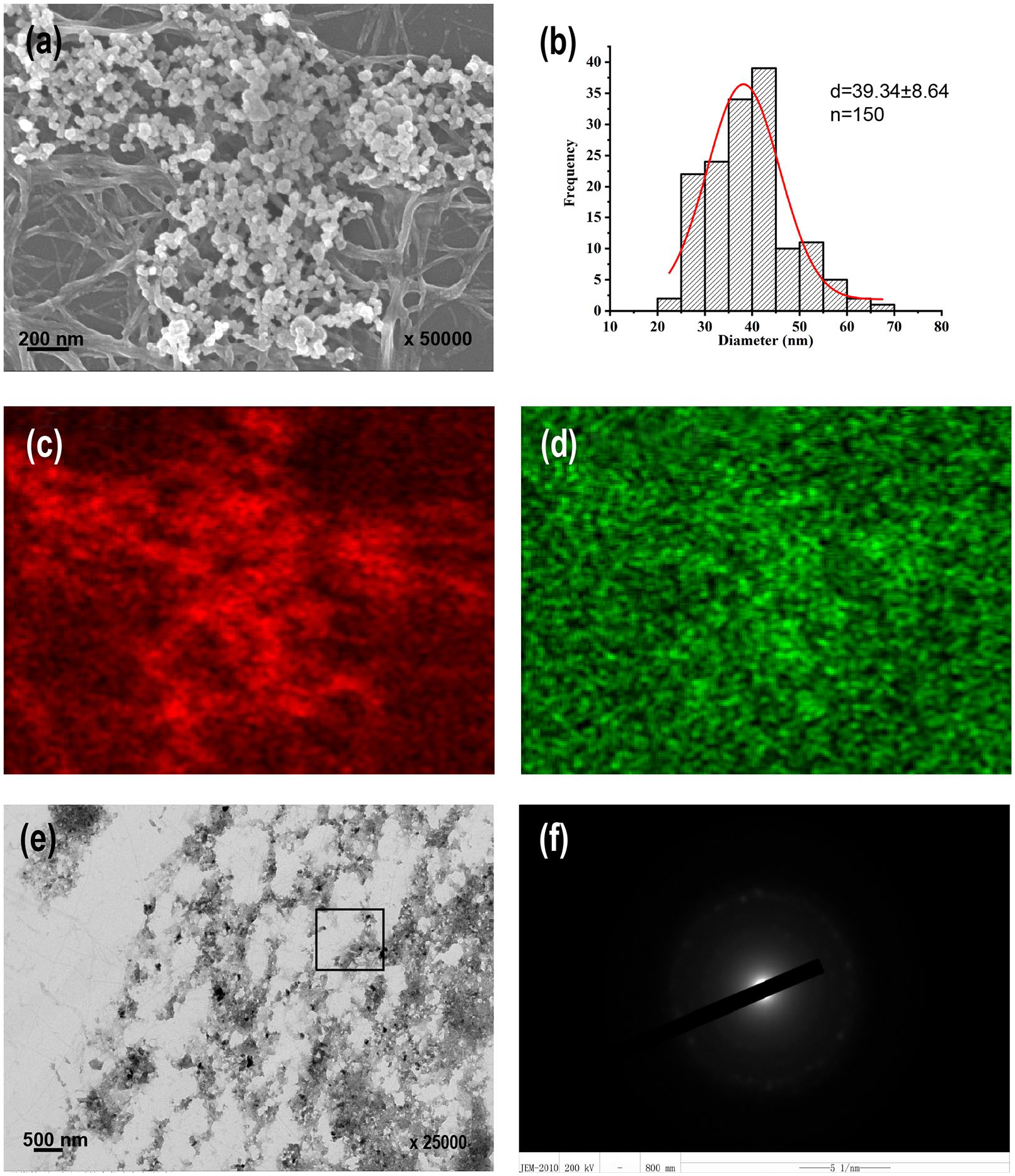
Templating mineralization property of the oligopeptide nano-matrix: (a) SEM image, (b) diameter distribution of pearl necklace-like particles in (a), (c) Ca, (d) P distribution in (a), (e) TEM image, and (f) electron diffraction of the selected area in (e).
Characterization of the demineralized dentin
The surface and cross-sectional views of demineralized dentin were evaluated using FE-SEM with elemental analysis. Figure 3(a) was the surface view of the demineralized dentin, and Figure 3(b) was the magnification of Figure 3(a). The dentin collagen fibrils were exposed, which seemed porous because of hydroxyapatite loss. All dentin tubules were open and empty after acid demineralization. Figure 3(c) was the cross-sectional view of the demineralized dentin, showing that the thickness of the demineralized dentin was approximately 6 μm. Figure 3(d) was the elemental composition of the demineralized dentin. The demineralized dentin exhibited a great amount of C, N, and O distribution. Besides, the elemental analysis displayed a few Ca and P ascribing to residual hydroxyapatites on demineralized dentin.

Characterization of the demineralized dentin: (a) surface view of the SEM, (b) magnified image of (a), (c) cross-sectional view of the SEM image, and (d) elemental analysis. White arrows indicate dentin tubules.
Binding property of the oligopeptide nano-matrix to demineralized dentin
Figure 4(a) was the ATR-FTIR analysis of the binding property between oligopeptide nano-matrix and demineralized dentin. The demineralized dentin displayed peaks at approximately 3299, 1631, 1542, and 1452 cm−1, which were obtained from the amide A, I, II, and III groups of demineralized dentin, respectively (Blueline). After coating the oligopeptide nano-matrix, the peaks of the amide A, I, II, and III groups decreased to 3226, 1581, 1496, and 1402 cm−1, respectively. Moreover, the absorption band at amide A broadened compared with demineralized dentin (Blackline). After thorough rinsing with deionized water for 5 min, the characteristic absorption changed slightly compared with the oligopeptide-coated dentin (rinsing for 30 s, Redline). Figure 4(b) was SEM image of the oligopeptide-coated demineralized dentin that was thoroughly rinsed with deionized water for 5 min. Figure 4(c) was the magnification of Figure 4(b). Those images showed that the oligopeptide nano-matrix remained on the demineralized detin after thorough rinsing.

Binding property of the oligopeptide nano-matrix to demineralized dentin: (a) ATR-FTIR, (b) SEM image after thorough rinsing, and (c) magnified image of (b). Blackline: oligopeptide-coating dentin after rinsing for 30 s; redline: oligopeptide-coating dentin after rinsing for 5 min; blueline: demineralized dentin.
Remineralization in artificial saliva
Figure 5 was the SEM of the oligopeptide-coated demineralized dentin, which was mineralized in artificial saliva for 24 and 48 h. Figure 5(a) was the surface view image of mineral sediments for 24 h. A layer of pearl necklace-like particles that were anchored over the oligopeptide nano-matrix was observed on demineralized dentin, obscuring dentin tubules. Figure 5(b) was the magnification and elemental analysis of Figure 5(a). The mineral sediments exhibited a great amount of C and O and a little Ca and P. Figure 5(d) was the surface view image of mineral sediments mineralized for 48 h. Compared with that in 24 h, the sediment layer became denser and completely covered the dentin tubules. Mature rod-like crystals developed from the irregular sediments. Figure 5(e) was the magnification and elemental analysis of Figure 5(d). The composition of Ca and P components increased compared with that in 24 h. Figure 5(c) and (f) were the cross-sectional view images of mineral sediments mineralized for 24 and 48 h, respectively. The sediments partially interweaved to the porous structure of demineralized dentin in a disorderly manner. Furthermore, a layer of dentin collagen fibrils was exposed between sediments and the underlying dentin. In the control group, no evident sediments were observed on demineralized dentin after mineralizing for 24 and 48 h (Figure S3).

Remineralization on the demineralized dentin after 24 and 48 h: (a) surface view image after mineralizing for 24 h, (b) magnified image of (a), (c) cross-sectional view of (a), (d) surface view image after mineralizing for 48 h, (e) magnified image of (d) and (f) cross-sectional view of (d).
Figure 6 was the SEM of oligopeptide-coated demineralized dentin that was mineralized for 96 h. Figure 6(a) to (c) were the surface view images of the remineralized layer. Figure 6(a) showed that a layer of homogeneous rod-like crystals was regenerated on the demineralized dentin. The Ca/P ratio of the remineralization was approximately 1.67. Figure 6(b) and (c) were the magnification of Figure 6(a). Oligopeptide nano-matrix remnants were observed on the regenerated crystal layer. Figure 6(d) to (f) were the cross-sectional view images of the remineralized layer. Figure 6(d) showed that the mature crystals regenerated a dense remineralization layer with a thickness of approximately 15.35 μm. The remineralized layer was perpendicular to the underlying dentin. Figure 6(e) was the magnification of Figure 6(d), which showed that the mature crystals were arranged in parallel. Figure 6(f) showed that the remineralized layer exhibited tight connection and fusion with the underlying dentin. Figure 6(g) to (i) were the SEM images that the mature crystals occluded dentin tubules. Figure 6(g) showed that the mature crystals precipitated on the inner wall of dentin tubules in the deep dentin, and extended toward the center of the tubules. Tubules’ depth, which was occluded by precipitated crystals, was greater than 31.27 μm. Figure 6(h) and (i) were the magnification of Figure 6(g), which showed that crystals even completely occluded the dentin tubules, causing the space between tubular walls to disappear.

Remineralization on the demineralized dentin after 96 h: (a) surface view image, (b) and (c) magnified images of (a), (d) cross-sectional view image, (e) and (f) magnified images of (d), (g) occlusion of dentin tubules, (h) magnified image of (g), and (i) magnified image of (h).
Figure 7(a) was the X-ray diffraction of remineralization. The demineralized dentin exhibited a relatively flat peak because of the hydroxyapatite loss (Blueline). After mineralizing for 96 h, the experimental group showed an increased diffraction peak intensity with 2θ at 25.88, 32.16, 39.96, 46.72, 49.56, and 53.22 (Blackline). The control group showed a diffraction peak intensity that was between the demineralized dentin and experimental group (Redline). Figure 7(b) was the ATR-FTIR of the samples. The demineralized dentin exhibited characteristic peaks at 3309, 1631, 1540, and 1452 cm−1, which were obtained from the amide A, I, II, and III groups of dentin collagen (Blueline). After mineralizing for 96 h, the experimental group displayed a decreased intensity of amide peaks and enhanced absorption peak of phosphate (Blackline). The control group exhibited a similar peak intensity to the demineralized dentin (Redline).

X-ray diffraction and ATR-FTIR of remineralized dentin after mineralizing for 96 h: (a) diffraction peaks in XRD, (b) ATR-FTIR. Blackline: experimental group; redline: control group; blueline: demineralized dentin.
Figure 8 was the result of the surface microhardness in all groups. Vickers hardness (HV) of the demineralized dentin was approximately 53.39 ± 4.80. After mineralizing for 96 h, the hardness of the experimental and control groups significantly increased to 110 ± 11.45 and 72.80 ± 7.55, respectively (p < 0.05). Compared with the control group, the hardness of the experimental group also exhibited a significant increase (p < 0.05).

Micro-hardness (HV) of remineralization after 96 h.
Figure 9(a) was the result of PDLCs’ proliferation incubated with oligopeptide nano-matrix. After 1 and 3 days, the oligopeptide nano-matrix did not inhibit PDLCs’ proliferation compared with the control group. After 5 days, 75 and 100 μg/mL oligopeptide nano-matrix improved the proliferation of PDLCs. The oligopeptide nano-matrix promoted PDLCs’ proliferation at a minimum concentration of 50 μg/mL after 7 days. Figure 9(b) displayed cell viability on the oligopeptide nano-matrix after 7 days, and no evident dead cells were observed in the experimental and control groups.

Biocompatibility of the oligopeptide nano-matrix: (a) effects of oligopeptide nano-matrix at various concentrations (0–100 μg/mL) on the PDLCs proliferation and (b) live/dead staining assay after culturing for 7 days.
Discussion
The present study determined the capability of a CEMP1-derived oligopeptide reconstructing hard tissues on demineralized dentin. The oligopeptide nano-matrix that bound well to demineralized dentin reconstructed enamel-like tissue on demineralized dentin and induced strong dentinal tubule occlusion, which provided a potential agent for DH treatment.
The oligopeptide simulating CEMP1 self-assembled into the nano-matrix with a helix structure when calcium ions were introduced, thereby exposing its bioactive peptide sequence to form the hydroxyapatite precursors within 24 h. For DH occurrence, the dentin must be exposed with enamel or cementum removal because of attrition, abrasion, or erosion.21,22 Therefore, the reconstruction of dental hard tissues is an important research area for DH treatment. 23 In vivo, the mineral formation including nucleation, 24 phase transformation, 25 and orientation, need the regulation of organic biomacromolecules. Thus, the biomimetic mineralization method has attracted scientists’ attention in designing organic molecules as templates to control the nucleation and growth of minerals in vitro. 26 This biomimetic oligopeptide consisted of a bioactive peptide sequence derived from CEMP1 (NNCCCCRRES(p)) and a hydrophobic tail (C16H31COOH). When introducing calcium ions, the oligopeptide self-assembled into the supermolecular nano-matrix, thereby, exposing bioactive peptides on the surface of the nano-matrix. In addition, the circular dichroism results indicated that the oligopeptide nano-matrix exhibited a typical helix structure, 27 which could affect mineral nucleation and morphology.28,29 Oral saliva contains calcium, phosphate, and fluoride in a supersaturated state, which can compensate for the mineral loss in an initial erosive lesion. 30 Artificial saliva was used to mimic oral conditions and to imitate the release of calcium and phosphate ions. 31 The oligopeptide nano-matrix can induce the formation of pearl necklace-like minerals after incubating in artificial saliva for 24 h. Moreover, element analysis and diffused ring diffraction pattern indicated that the formed minerals were comparable to the precursors of hydroxyapatites. 14
The oligopeptide nano-matrix exhibited a considerable binding capacity to demineralized dentin and resist water rinsing in vitro. Demineralized dentin was applied to imitate the DH condition. 32 FE-SEM with elemental analysis indicated that 37% phosphoric acid treatment can lead to effective dentin demineralization and dentin tubule exposure, which was used in this study as a simple simulation of DH. The binding property of natural NCPs to collagen is important for its biomineralization. Osteocalcin, which binds with a specific region of the bone extracellular matrix, regulates bone mineralization. 33 After crosslinking to collagen, polyacrylic acid, an analog of NCPs, can promote intrafibrillar mineralization in vitro. 34 Moreover, flowing saliva of the oral environment can wash off the mineralization agent once it does not tightly bind with dentin. In our study, dentin collagen was exposed after treating with phosphoric acid; hence, binding of the oligopeptide nano-matrix to demineralize dentin is important for the oligopeptide nano-matrix to achieve its function. ATR-FTIR indicated that demineralized dentin exhibited typical amide A, I, II, and III bands, which was attributed to the exposed dentin collagen after demineralization. After coating the oligopeptide nano-matrix, the aforementioned amide bands red shifted to lower peaks, indicating the hydrogen bindings between amino/hydroxyl of the oligopeptide nano-matrix and amino/hydroxyl of the demineralized dentin. 35 After thorough rinsing, the characteristic absorption bands of the oligopeptide-coating dentin almost no change, indicating that a mass of oligopeptide nano-matrix remained on the demineralized dentin.
The bound oligopeptide nano-matrix then served as a template to induce mineralization and reconstruct hard tissues on demineralized dentin. The oligopeptide induced the formation of hydroxyapatite precursors and prevented its transformation to crystals within 24 h. The precursor structure on demineralized dentin was similar to those formed by the oligopeptide nano-matrix on TEM grids. This observation indicated that the oligopeptide nano-matrix maintained its mineralization property on demineralized dentin. After 48 h, mature crystals were developed from those precursors. The mesophase transformation of precursors may be mediated by the oligopeptide nano-matrix. 26 In addition, the interface between the sediment layer and the underlying dentin was creviced, which indicated that the early sediment layer may not resist chewing pressure. After 96 h, mature crystals with a Ca/P ratio of approximately 1.67 developed into a homogeneous dense mineral layer on demineralized dentin. This Ca/P ratio indicated that the mature crystals were comparable to hydroxyapatites. 36 The hydroxyapatite crystal layer was oriented and perpendicular to the underlying dentin, of which the structure was similar to natural enamel. 37 The biological function of enamel was to protect the underlying dentin and pulp from mechanical forces and chemical abrasion from the oral environment, which was ascribed to the arrangement and orientation of hydroxyapatites. 38 Moreover, the enamel-like tissue exhibited tight connection and fusion with the underlying dentin. These observations indicated that the reconstructed enamel-like tissue may resist mechanical forces such as chewing pressure and chemical abrasion in the oral environment. Oligopeptide nano-matrix remnants were observed on the surface of enamel-like tissue, which indicated that the nano-matrix could migrate between the enamel-like tissue and underlying dentin. Natural enamel formation can be concluded as follows: (a) In the beginning, highly polarized ameloblasts secreted an organic hydrogel-like matrix primarily composed of amelogenin and non-amelogenin. (b) Afterward, calcium and phosphate ions secreted by ameloblasts were attracted to the organic matrix and dentin surface to crystallize. (c) Along with crystal growth, the organic matrix was squeezed out of the crystal space because of the generated pressure and absorbed by ameloblasts. 39 In this study, the oligopeptide nano-matrix can be a simulation of the organic matrix secreted by ameloblasts during enamel formation. With the development of remineralization, the oligopeptide nano-matrix was pushed out by the newly formed hydroxyapatites and exposed on the surface.
These hydroxyapatite crystals effectively occluded the dentin tubules with a depth greater than 31.27 μm after mineralizing for 96 h. The oligopeptide nano-matrix can stabilize mineral precursors from transformation into crystals within 24 h; thus, the hydroxyapatite crystals that are directly nucleated on demineralized dentin may be delayed. Therefore, the amorphous precursors have a greater opportunity to infiltrate into the dentin tubules and crystallize on the tubule wall to occlude dentin tubules. In conclusion, after 96 h, the oligopeptide nano-matrix can regenerate enamel-like tissue on demineralized dentin with a thickness of 15.35 μm and occlude dentin tubules with a depth of 31.27 μm. Based on the hydrodynamic hypothesis, the treatments that can decrease dentinal fluid conductance, such as forming insoluble sediments on the dentin surface or occluding dentin tubules, 15 are considered effective in decreasing the clinical symptomatology of DH. Therefore, oligopeptide nano-matrix simulating CEMP1 shows great application potential for DH treatment.
Some limitations should also be considered in this study. For example, the micro-hardness of enamel-like tissue formed by oligopeptide nano-matrix significantly increased compared with demineralized dentin and control groups after mineralizing for 96 h. However, the hardness of enamel-like tissue was smaller than the natural enamel. 40 The reasons could be partially ascribed to the limited mineralizing time, and the effectiveness of oligopeptide simulating CEMP1 was lower than natural NCPs. In the future, extending the mineralizing time or increasing the oligopeptide concentration could increase the hardness of enamel-like tissue. In this study, the oligopeptide nano-matrix spent 96 h to reconstruct hard tissues on demineralized dentin. The enamel-like tissue and the dentin tubule occlusion could not be immediately formed via the treatment of the oligopeptide nano-matrix. To relieve the pain of DH quickly, the drugs that can reduce interdental nerve excitability could be incorporated into the oligopeptide nano-matrix to form a multi-functional desensitization agent. However, this finding needs further study.
CCK-8 assay and live/dead staining indicated that the oligopeptide nano-matrix had great biocompatibility, and it improved the proliferation of PDLCs, indicating its safety in DH treatment. In this study, an oligopeptide nano-matrix was added to the culture medium to evaluate its biocompatibility using PDLCs. The oligopeptide nano-matrix can resemble the natural extracellular matrix and analogous scaffold for tissue engineering. For example, scientists have fabricated a biphasic scaffold containing a self-assembling peptide nano-matrix and hydroxyapatite nanoparticles for bone regeneration. 41 Therefore, the amphiphilic oligopeptide simulating CEMP1 can be used as a scaffold for tissue engineering such as periodontal tissue regeneration. However, this issue needs further study.
Conclusions
The possible mechanism of oligopeptide simulating CEMP1, which reconstructed the enamel-like tissue and induced tubule occlusion, was as follows. First, the oligopeptide simulating CEMP1 self-assembled into nano-matrix, thereby exposing its bioactive peptide with an active mineralization property. Then, the oligopeptide nano-matrix could bind firmly to demineralized dentin by hydrogen bindings. Afterward, the bound nano-matrix served as an organic template to induce and stabilize the hydroxyapatite precursors on demineralized dentin. Finally, those mineral precursors transformed into mature hydroxyapatite crystals, forming an enamel-like tissue on demineralized dentin and occluding dentin tubules. Based on the hydrodynamic hypothesis, the oligopeptide imitating CEMP1 shows great application potential for DH treatment.
Supplemental Material
sj-pdf-1-jbf-10.1177_22808000211005384 – Supplemental material for Biomimetic oligopeptide formed enamel-like tissue and dentin tubule occlusion via mineralization for dentin hypersensitivity treatment
Supplemental material, sj-pdf-1-jbf-10.1177_22808000211005384 for Biomimetic oligopeptide formed enamel-like tissue and dentin tubule occlusion via mineralization for dentin hypersensitivity treatment by Qing-qing Wang, Siqing Wang, Tian Zhao, Yan Li, Jie Yang, Yumei Liu, He Zhang, Leiying Miao and Weibin Sun in Journal of Applied Biomaterials & Functional Materials
Footnotes
Contributorship
Qing-qing Wang and Weibin Sun researched literature and conceived the study. Leiying Miao, Yan Li, and Jie Yang were involved in protocol development, gaining ethical approval, patient recruitment, and data analysis. Qing-Qing Wang wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Guarantor
Prof. Sun and Prof. Miao are the people who take full responsibility for the article, including the accuracy and appropriateness of the reference list. They are also authors of all correspondence about this paper.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (Nos. 51772144, 51972167, and 82001111); the Project supported by Medical Science and Technology Development Foundation, Nanjing Department of Health (ykk18126); the Project of Invigorating Health Care through Science, Technology, and Education Jiangsu Provincial Medical Youth Talent (QNRC2016120); Jiangsu Province Natural Science Foundation of China (No. BK20170143); Key Project supported by Medical Science and Technology Development Foundation, Nanjing Department of Health (No. YKK17144); and Nanjing Municipal Key Medical Laboratory Constructional Project Funding (Since 2016).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
