Abstract
Objective:
This paper reports on the buffering and acid-resistant properties of a modified eggshell–titanium composite against citric acid attack.
Materials and Methods:
Eggshell–titanium EB-TiO2 was prepared by ball-milling eggshell powder and titanium dioxide. Fourier Transform Infrared Spectroscopy (FTIR), X-ray Diffraction (XRD), and Transmission Electron Microscopy (TEM) were used to characterize EB-TiO2. The buffering property against citric acid at pH values of 2, 4, and 5 was measured using a pH meter. Five brands of toothpaste (Colgate, Colgate Sensitive, Aquafresh, Oralwise, and Sensodyne) were used to assess the acid-resistant properties of EB-TiO2. Enamel models were simulated by dissolving each brand of toothpaste with eggshell (control) and EB-TiO2. The samples were exposed to citric acid of pH 2. The average slope (kPa/s) was measured using a pressure sensor. An analysis of variance was used to analyze the kPa/s values (α =.05).
Results:
The FTIR and XRD analyses suggest the surface modification of EB-TiO2. The TEM image revealed spherical-shaped particles in EB-TiO2. The pH test results showed that the buffering properties of eggshell and EB-TiO2 were comparable. Significant differences were observed in the acid resistance properties of the samples exposed to citric acids (P < .05). The Colgate toothpaste infused with eggshell powder had the highest mean kPa/s values, whereas Sensodyne infused with EB-TiO2 had the lowest kPa/s values.
Conclusion:
The salient features of this study indicate that modification of eggshell with titanium dioxide does not affect its carbonate buffering properties. Connecting the kPa/s values to acid-resistant properties, EB-TiO2 effectively reduces erosive attacks when added to toothpaste.
Introduction
Enamel erosion is a common occurrence and, under extreme conditions when the enamel is exposed to a highly acidic oral environment, it will result to the dissolution of the enamel surface.1–3 Previously, enamel erosion, particularly in the cervical area, was mostly reported among elderly people. 4 Several studies4–7 alleged that a change in diets as well poor oral hygiene have significantly contributed to the frequency of enamel erosion among young people. More so, enamel erosion has become an important issue with the increased consumption of acidic drinks, such as sports drinks, soft drinks, and citric juices.3,5,8,9 More worrisome is that the afore-mentioned products have a pH value below the critical level for dissolving dental enamel.3,8 Gambon et al. 10 point out that citric acid, for example, is a complex acid with respect to erosive potential. They noted that when the pH is at a lower level, the citric acid releases [H]+ ions that attack the surface enamel, while at higher values of pH, the citrate anion is able to chelate calcium from the enamel surface.
Generally, human tooth enamel is predominantly hydroxyapatite in the form of phosphate ions (PO43−) and calcium ions (Ca2+).7,11 Fejerskov 12 and Featherstone 13 noted that a stable equilibrium exists between the crystalline hydroxyapatite making up the 96% of tooth enamel and the phosphate and calcium ions in saliva. The destabilization of the afore-mentioned equilibrium, particularly when the oral environmental pH drops below a critical level (5.5 for enamel and 6.2 for dentin), may result in the dissolution of tooth mineral (hydroxyapatite) in a process called demineralization. 14 In contrast, with the elevation of the oral environmental pH promoted by the natural buffer capacity of saliva, the mineral gets reincorporated into the tooth through the process of remineralization. 15 While saliva is considered an important biological factor that dictates the intraoral neutralizing effect of acid exposure, Neel et al. 2 stressed that some medication and asthmatic inhalers may reduce the flow of saliva, thereby inducing xerostomia. This, and according to Kargul et al. 16 and Sivasitamparam et al., 17 decreases the pH of saliva, weakening its overall buffering effect against intrinsic and extrinsic acids. Moreover, the loss of enamel material by erosion is a dynamic process occasioned with periods of demineralization and remineralization; hence, preventive measures against enamel dissolution from acid and potentially permanent damage should be a priority for oral health care providers.3,4
Importantly, toothpastes have been considered affective and accessible vehicles to improve enamel resistance against erosive oral environments. 18 In recent years, different ingredients have been incorporated into toothpastes to increase their protective effect against erosive attacks. Among these ingredients, the use of topical fluoride to modify the effects of erosion at the tooth surface is well documented.8,19–21 Bertassoni et al. 22 and Moretto et al., 23 in their asserted views, however, highlighted that fluoride toothpastes alone are not capable of completely inhibiting tooth wear. Resonating with them, Moron et al. 15 stressed that conventional fluoride-containing toothpastes lack the capacity to protect sufficiently well against erosive challenge. Further to this, it has been shown at below pH 3 that the protective effect of fluoride is diminished. 24 Consequently, the addition of calcium or calcium-containing material in toothpastes is suggested to have beneficial health effects against erosive attacks. 25 Davis et al. 26 reported that calcium-fortified juices were sufficient to prevent erosion occurring in human tooth enamel. In support of them, Magalhães et al. 27 found that the modification of carbonated fruit drink juice-based drinks with calcium reduced their erosive potential.
Of interest, eggshells are naturally composed of calcium carbonates (~97% in the form of calcite); 1% magnesium carbonates; 1% apatite (Ca3 (PO4)2; and ~5% organic matter.28,29 Several studies30–32 have shown that the calcium constituent in eggshell was similar to that found in bone and teeth, thereby supporting its use as an abrasive cleaner in toothpastes. Furthermore, Cutler 33 suggested that titanium dioxide and calcium carbonate-based abrasive agents when used together effectively reduce tooth sensitivity. Equally significantly, Lin et al. 34 and Tao et al. 35 have shown that the mechanical activation of calcium carbonates together with titanium dioxide improved the acid resistance properties when used in the paper making industry. Despite the benefits associated with the modification of calcium carbonates with titanium dioxide, limited evidence is available on its use in the prevention of erosive attacks. In this paper, we present a new product based on the modification of eggshell powder and titanium dioxide through ball-milling, as a potential ingredient in toothpastes for the protection of erosive attacks.
In recent years, the modification of eggshell waste through ball-milling to obtain a novel material has gained much attention amongst researchers, owing to its environmental friendliness, less use of harmful organic solvents and energy, and its reproducibility, with a high yield under simple and easy operating conditions.36,37 According to Baláž, 37 ball-milling is a tool of mechanochemistry that combines the solid state approach and mechanical energy input for various desired applications. The author alleged that ball-milling natural material such as eggshells offers the prospect to change their application potential to a new level. Significantly, Battistella et al. 38 advocated for the use of biomaterial obtained from natural sources in dentistry due to its outstanding healing properties, biocompatibility with natural tissues, abundant availability, and low cost. This paper therefore aimed to examine the buffering and acid-resistant properties of a ball-milled eggshell–titanium dioxide (EB-TiO2) composite against citric acid attack.
The formulated hypothesis was as follows: (1) EB-TiO2 is a suitable ingredient in toothpaste formulation as it improves acid resistance when added into toothpastes.
Materials and methods
Food grade anatase titanium dioxide (CAS No: 13463677) was purchase from Sigma-Aldrich (Germany). Citric acid monohydrate and lactic acid were supplied by Merck (South Africa). Five different brands (Sensodyne, Colgate sensitive toothpaste, Aquafresh, Oral-wise, and Colgate) of toothpastes were bought from a popular shopping mall located in Durban (South Africa).
Preparation of eggshell–titanium dioxide material
As previously described by Onwubu et al., 39 eggshells obtained from Durban fast foods were washed with regular detergent to remove impurities. Eggshells were then disinfected by storing the eggshells in a diluted solution of household sodium hypochlorite for 6 hours. Thereafter, eggshells were vacuum dried for ±6–9 minutes at 250°C. Modification of eggshells with titanium dioxide was achieved in two steps. In the first step, eggshells were ball-milled by placing 30 g of the eggshell in a 500 ml stainless jar (inner diameter of 100 mm), together with 10 stainless steel balls of 10 mm diameter, and dry-milled in a planetary ball mill (Retsch ® PM 100) at 400 rpm for 20 min. The low milling time of 20 min allows for the homogenization of the eggshell powder particle sizes. 37 The collected powder was then sieved to a particle size of ⩽25 µm using a mechanical sieving shaker (Retsch AS 200, Germany). Thereafter, the fine eggshell powder obtained in step 1 was modified according to the procedure described by Lin et al. 34 A total of 20 g of the fine eggshell powder was modified by adding 5 g of food grade anatase titanium dioxide (⩽15 µm) and subsequently ball-milled for 200 min to obtain a nanosized eggshell–titanium dioxide material (⩽80 nm). Lin et al. 34 suggest that a new material is capable of forming after ball-milling calcium carbonate-based material with titanium dioxide for above 120 min.
Characterization of EB-TiO2
Fourier Transform Infrared Spectroscopy analysis
The infrared spectra were measured using a Perkin Elmer Universal ATR spectrometer to identify the functional group constituents of EB-TiO2. A very small amount of sample was placed in the sample holder. An initial background check was performed before scanning in the range of 400–4500 cm−1.
X-Ray Diffraction analysis
The crystallinity of the modified Eb-TiO2 was assessed using X-Ray Diffraction (XRD). The XRD diffractometer (PANalytical-Empyrean instrument; Co radiation 1.54056 Ao) was calibrated with a voltage of 40 kV, current of 40 mA, and time of 1 s and analyzed between 0° and 90o (2 theta).
Transmission Electron Microscopy analysis
Transmission Electron Microscopy (TEM) was used to observe the particle size, shape, and distribution of EB-TiO2. Some amounts of the samples were dispersed in 5 ml ethanol and sonicated at 10 kV for 20 min. Then, thin cross-sections of cryo-microtomed specimens were prepared using a Leica microtome (South Africa) and placed on carbon copper grids. Analysis was conducted using a transmission electron microscope (TEM-Philips CM 120 model) at 120 kV.
Examining the buffering properties of EB-TiO2
The buffering properties of the EB-TiO2 and eggshell powder were examined against citric acid at pH values of 2, 4, and 5. A stock solution of citric acid was prepared by dissolving 2.1022 g of citric monohydrate in a 100 ml volumetric flask. Serial dilution was subsequently used to prepare pH values of 2, 4, and 5, respectively. A total of 1.5 g of each sample was placed in a beaker containing 50 ml of the prepared citric acid. The solution was constantly agitated at the low speed of 600 rpm for 30 min. A pH meter (Starter 300, Ohaus Incorporation USA) equipped with a temperature sensor was constantly used to monitor changes in the pH reading.
Preparation of the simulated enamel model
Each brand of toothpaste was used to assess the acid-resistant properties of EB-TiO2. The enamel model was simulated by dissolving each brand of toothpaste with eggshell (control) and EB-TiO2. This entails dissolved the substance in a beaker containing 100 cm3 deionized water. A total of 1 g of the prepared eggshell powders and EB-TiO2 was added to the beaker containing the dissolved toothpastes. These were further agitated for 8 h at a speed of 800 rpm. The mixtures were filtered and oven dried at 60 degrees for 3 h. The samples were exposed to citric acid of pH 2 for 30 min.
Evaluation of acid-resistant properties
The acid-resistant properties of the prepared samples were analyzed using a gas pressure sensor (Order Code GPS-BTA). Using an Erlenmeyer flask (250 ml) as the reaction vessel, the pressure sensor was connected to an interface system (Vernier LabPro, USA). The mean slope (kPa/s) was recorded using LoggerPro 3 software. Prior to the experiment, 0.5 g of each sample was placed in the Erlenmeyer flask, while 25 ml of the prepared citric acid was used as the reactant. All tests were performed in duplicate, and the mean slope (kPa/s) values were used for the statistical analysis.
Statistical analysis
Using a statistical package (IBM SPSS Statistics v 24; IBM Corp), the mean kPa/s values were evaluated and compared with one-way analysis of variance (ANOVA).
Results
Characterization of EB-TiO2
The Fourier Transform Infrared Spectroscopy (FTIR) spectra of eggshell powder and EB-TiO2 are given in Figure 1. As shown in Figure 1(a), the absorption peaks of the carbonate structures observed at 1411, and 711, and 873 cm−1 are attributed to the presence of calcite found in eggshell. 39 In contrast, the FTIR spectra in Figure 1(b) show a broad band below 800 cm−1, which corresponds to the Ti-o-Ti vibrations of the titanium dioxide.
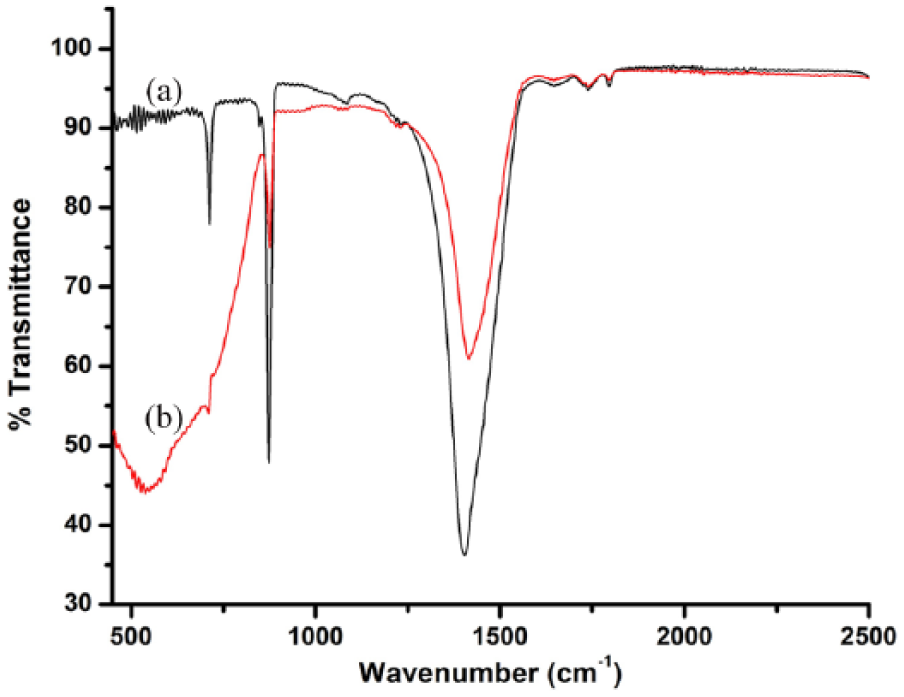
Fourier Transform Infrared Spectroscopy spectra of (a) eggshell powder and (b) EB-TiO2.
The XRD pattern of eggshell powder and EB-TiO2 showing characteristic diffraction peaks with diffraction angles is given in Figure 2. Consistent with Onwubu et al., 39 the characteristic peak marked around 34.5o indicates the presence of calcite (Figure 2(a)). Furthermore, the XRD pattern of EB-TIO2 given in Figure 2(b) reveals a characteristic diffraction peak with values lying at 2 theta (29.5o). This relates to the anatase phase of titanium dioxide and is confirmed by the International Centre for Diffraction Data (ICDD Ref: 98-009-6946). Furthermore, the shape, intensity, and location of the EB-TiO2 peaks suggest that TiO2 is deposited on the surface of the CaCO3 constituents of eggshell powder. The above assertion further supports the work of Tao et al., 35 who observed the deposition of pure TiO2 on the surface of CaCO3 at diffraction peaks with values around 27.5o.
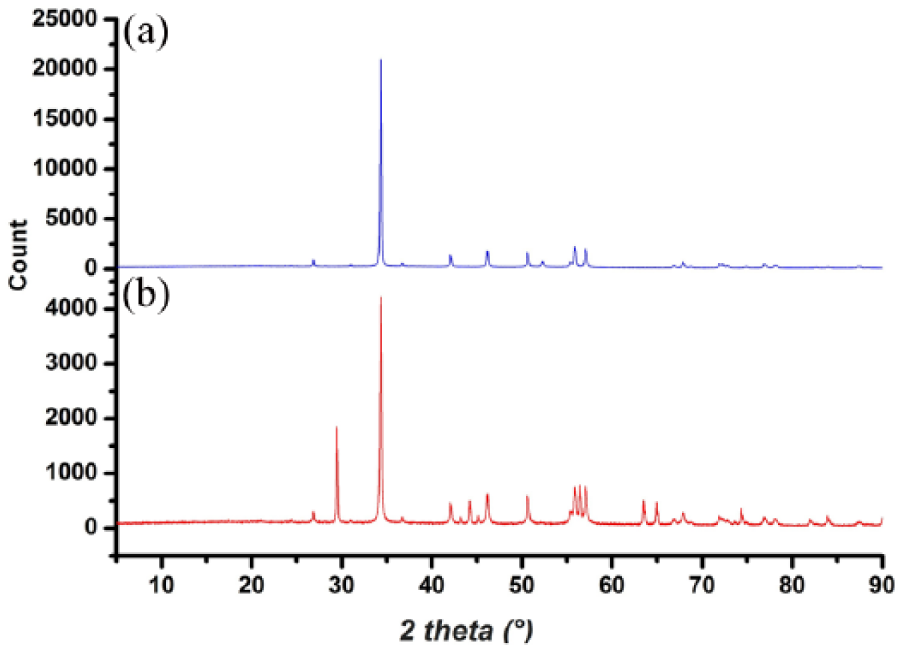
X-ray Diffraction pattern of (a) eggshell powder and (b) EB-TiO2.
Equally important, and contrary to the report by Baláž et al. 40 that ball-milling induces phase transformation of the calcite in eggshell to aragonite, the XRD pattern in Figure 2 suggests the absence of phase transformation. The differences in these studies could, however, be attributed to the milling conditions. According to Baláž, 37 ball-milling eggshell for 240–360 min causes phase transformation in the eggshell powder from calcite to aragonite and vice versa. A similar factor may have contributed to the absence of phase transformation of EB-TiO2, as the composite was ball-milled for 200 min.
The TEM micrograph of EB-TiO2 is given in Figure 3(a). The presence of nonhomogeneous particles with some agglomeration was observed. In addition, the TEM revealed that irregular-shaped particles coexist together with spherical-shaped particles. The presence of the irregular-shaped particles could be attributed to the calcite shape of the eggshell powder, whilst the spherical-shaped particles typified the presence of TiO2.

Transmission Electron Microscopy images of EB-TiO2 showing particle shapes.
The particle distribution of EB-TiO2 analyzed using ImageJ (National Institute of Health USA) reveals an average mean particle size of around 13 nm (Figure 4).
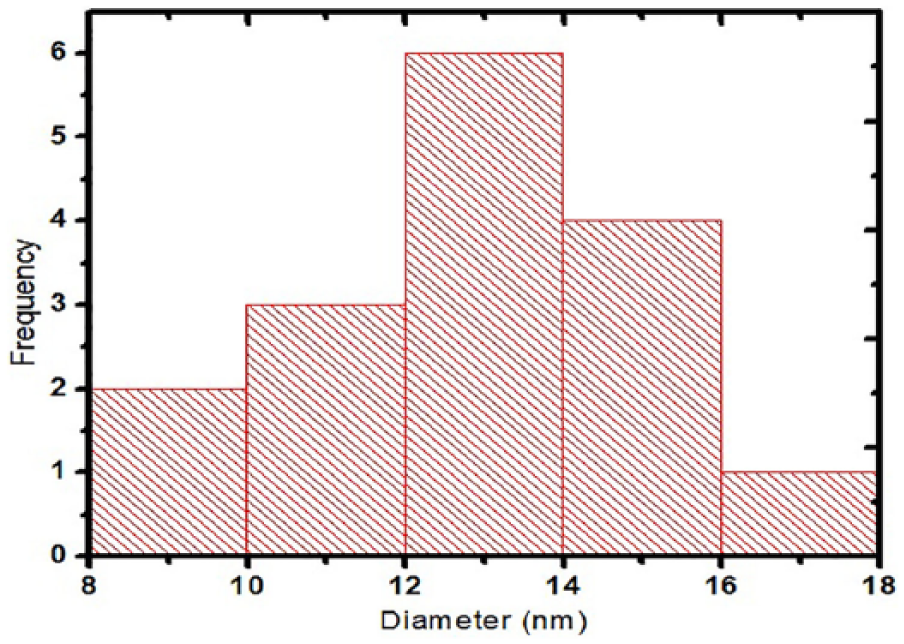
Histogram of EB-TiO2 showing the average mean particle sizes.
Buffering and acid-resistant analysis
Figure 5 illustrates the buffering properties of the tested samples against citric acids with different pH values of 2, 4, and 5, respectively. The results suggest that the buffering properties of eggshell and EB-TiO2 at each measured pH (2, 4, and 5) were comparable the Colgate brand of toothpaste.
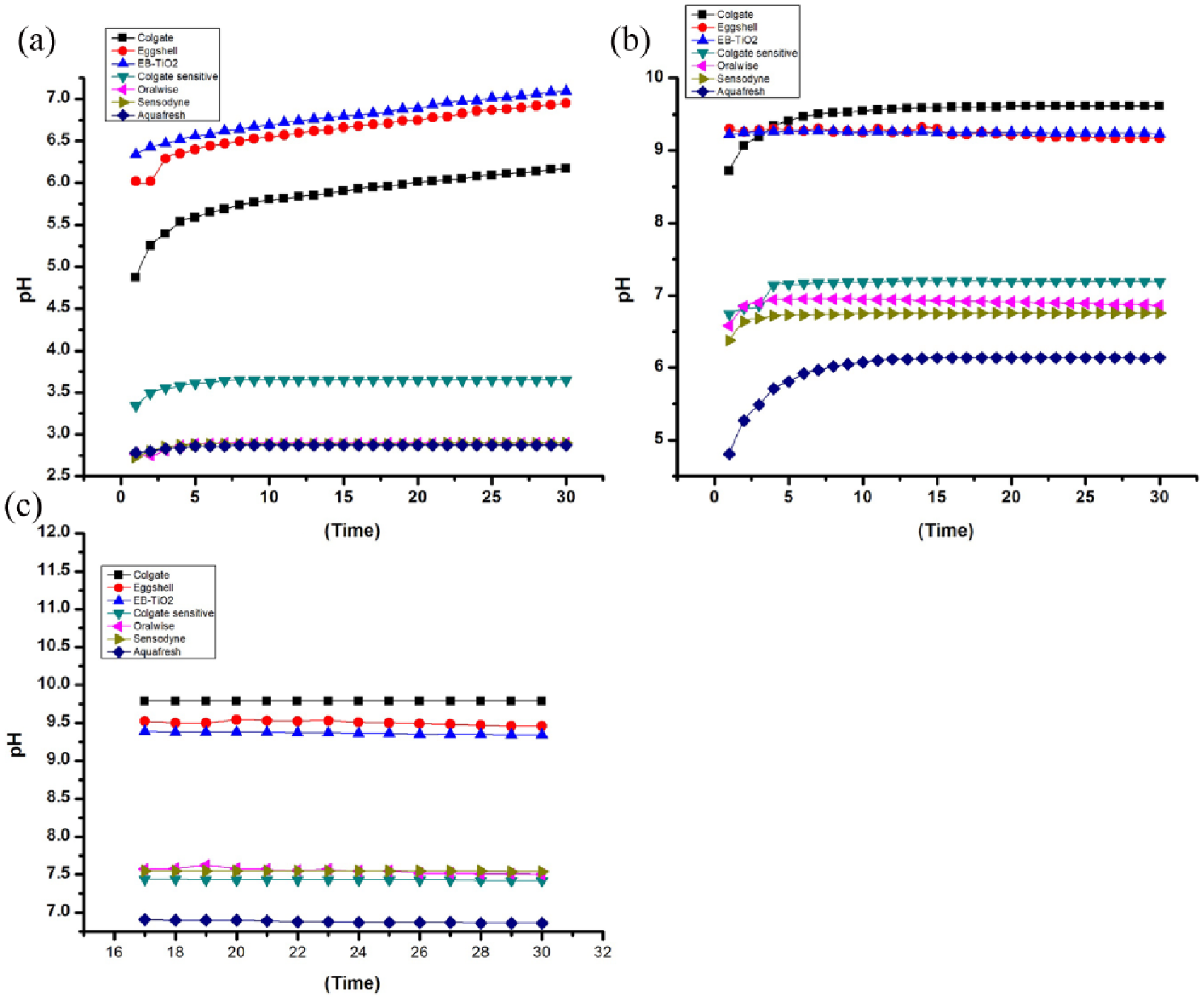
Buffering properties for (a) pH 2, (b) pH 4, and (c) pH 5 after 30 min exposure to citric acid.
The one-way ANOVA, mean, standard deviation, and standard error results for the resistance of the samples against citric acid exposure are illustrated in Table 1. Significant differences were found in the acid resistance properties of the samples and their exposure to citric acid (P < 0.05). The results of the post hoc comparison test are given in Table 1. The k/Pa/s values for the Sensodyne, Colgate, and Aquafresh toothpastes infused with eggshell powder were significantly higher than those for the toothpastes infused with the EB-TiO2 group (P < 0.05). No statistical differences were found between the k/Pa/s values for the Colgate Sensitive and Oralwise groups infused with both eggshell powder and EB-TiO2 (P > 0.05).
Analysis of variance (ANOVA) results showing sample resistance against citric acid.
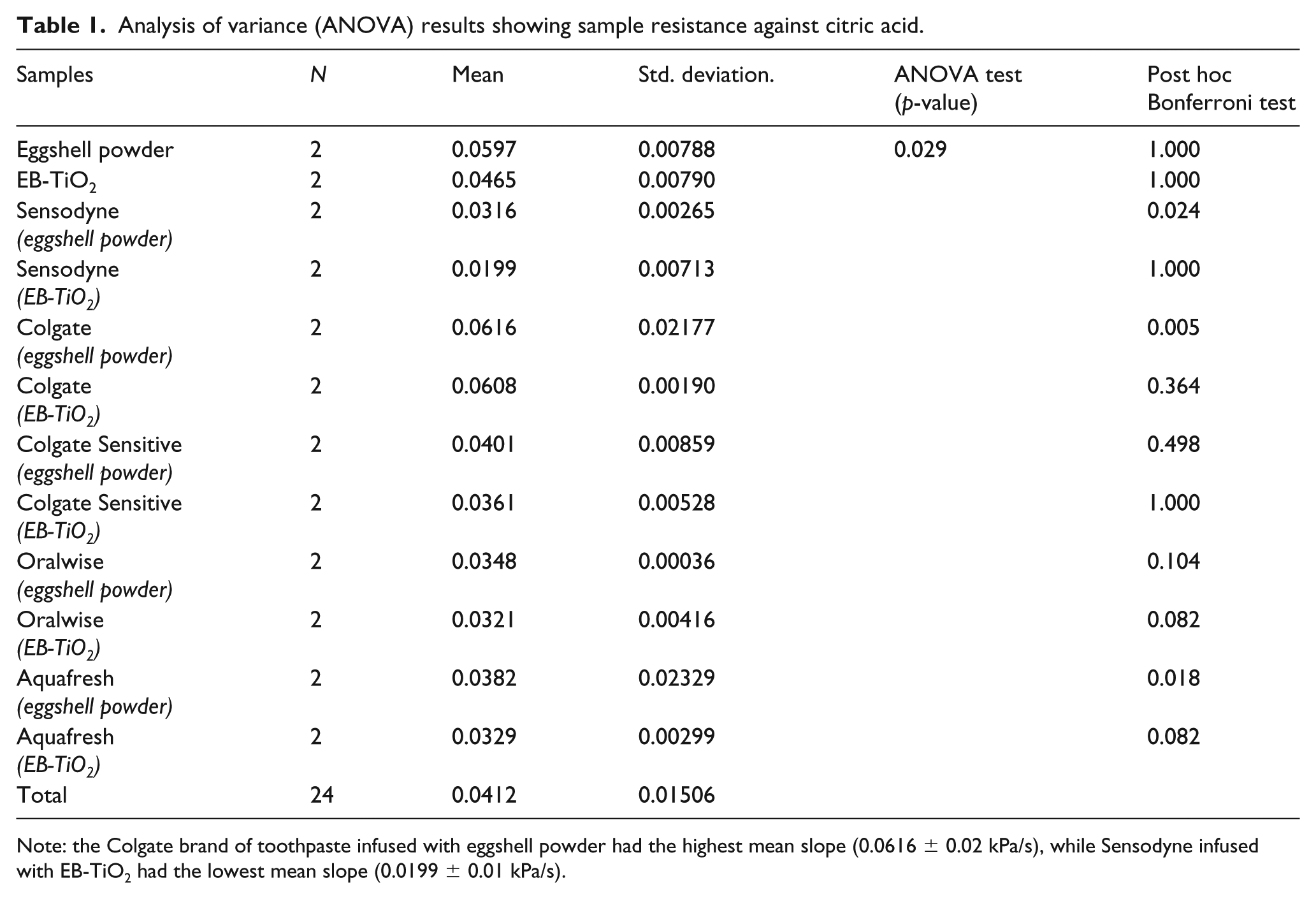
Note: the Colgate brand of toothpaste infused with eggshell powder had the highest mean slope (0.0616 ± 0.02 kPa/s), while Sensodyne infused with EB-TiO2 had the lowest mean slope (0.0199 ± 0.01 kPa/s).
Discussion
Calcium carbonate-based material has been extensively studied for its buffering properties, particularly for the treatment of gastro-esophageal reflux disease and peptic ulcers. 41 The calcium carbonate (CaCO3) constituent of EB-TiO2 (Figure 1(a)) suggests that it can effectively buffer acidic solutions. The buffering activity for the eggshell powder, EB-TiO2, and the Colgate brand of toothpaste at pH values of 2, 4 and 5, respectively, were much more effective than the other tested commercial toothpastes (Figure 5). The enhanced buffering property of the carbonate-based products is attributed to their higher solubility in acidic solutions. This supports the argument of Raliya et al. 41 that the higher solubility of calcium carbonate-based material results in a greater concentration of ions in solution. Consequently, this leads to a higher buffering activity as the faster dissolution rate increases the pH quickly, thereby expanding the horizon of neutralization.
Equally important, the buffering activities of eggshell powder and EB-TiO2 were comparable to each other. It can therefore be gathered that the modification of eggshell powder with titanium dioxide does not adversely affect the carbonate structure of EB-TiO2. From a dental oral health perspective, and corroborating with Macri, 42 the buffering property of EB-TiO2 is particularly important, as it can increase the pH of an acidic environment where demineralization is likely to occur and offer a high bioavailability of calcium.
The acid-resistant properties of EB-TiO2 were evaluated in vitro using a pressure sensor. The mean average slope (kPa/s) was measured and used for statistical analysis. On the basis of the research data, the stated hypothesis was accepted. The experimental results indicate that the addition of EB-TiO2 to commercial toothpastes statistically affected the acid resistance when exposed to citric acid (P < 0.05).
Overall, the kPa/s values measured in the brands of toothpastes added with EB-TiO2 were consistently below those with eggshell powder alone (Table 1). In light of the differences measured in the acid-resistant properties between EB-TiO2 and eggshell powder, it can be assumed that this difference is related to the presence of TiO2. According to García-Valverde et al., 43 titanium dioxide is reported to have good acid-resistant characteristics. The presence of TiO2 on the surface of the eggshell powder (Figure 2(b)) confirms that EB-TiO2 will be effective against acidic attacks. This finding supports Tao et al. 35 in that modification of calcium carbonate-based material with titanium dioxide is more likely to produce a higher resistance to acids. Eggshell powder should therefore be modified with titanium dioxide for reducing erosive attacks.
Conclusions
The prominent feature of this study revealed the successful modification of EB-TiO2 through ball-milling. The FTIR and XRD results confirmed TiO2 coating on the surface of eggshell powder. The TEM revealed spherical-shaped particles with a particle size of 13 nm. Conclusively, this paper showed that EB-TiO2 had good buffering and acid-resistant characteristics. From an oral health perspective, using EB-TiO2 in toothpaste formulation will provide low cost, affordable, feasible, and sustainable products that are needed to improve the quality of oral health globally.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of South Africa (No. 104824).
