Abstract
Background
This study aimed to determine the physicochemical characteristics of an eggshell-based dental abrasive material.
Methods
The eggshell powder abrasive material (EPAM) was synthesized by ball milling eggshell powder and surfactants. Fourier transform infrared (FTIR) spectroscopy, X-ray diffraction (XRD), transmission electron microscopy (TEM) and a laser diffraction particle size analyzer (PSA) were used to characterize EPAM. In addition, the abrasive characteristics of EPAM were evaluated by comparison using poly(methyl methacrylate) (PMMA) resins. Surface roughness (Ra) was measured using a profilometer.
Results
The FTIR spectroscopy and XRD analysis confirmed that the carbonate product was primarily calcite (97.3%) with traces of graphite 2H (1.3%) and thenardite (1.4%). The TEM imagery revealed irregular particles in EPAM. The PSA analysis of the particle size distribution showed EPAM to be a superfine powder (0.3 μm to 50 nm). In addition, the 50-nm EPAM (Ra = 0.04 μm) measured the lowest Ra value when compared with pumice (Ra = 0.08 μm).
Conclusions
The salient features of this study indicate that EPAM can naturally replace calcite, which is generally mined and used as a dental abrasive material. In addition, and regarding the abrasive characteristics of EPAM in reducing the surface roughness of PMMA resin specimens, this study conclusively showed that EPAM effectively reduces the surface roughness below the threshold limit value of 0.2 μm. Potentially, EPAM could reduce waste disposal problems while enabling an economic benefit from using eggshell waste material.
Keywords
Introduction
Eggshells are solid by-product waste materials of aviculture and domestic kitchen waste. In most countries, eggshells are mainly disposed of in landfills without further treatment, thereby adversely affecting the environment (1–3). To reduce this, attempts have been made to use eggshell waste materials in the production of value-added products. For instance, Abdulrahman et al (4) used calcium carbonate derived from eggshells to produce hydroxyapatite, which is predominantly used in bone and dental treatments. They argued that eggshell-based hydroxyapatite and nanohydroxyapatite could reduce the cost of treatment in bone repair or replacement, and concomitantly reduce waste and protect the environment. To obtain value-added products while establishing better waste management, it is important to achieve sustainable economic development and management of the environment. Eggshell is a natural bioceramic composite with a unique chemical composition of inorganic and organic compounds. Eggshell together with the membrane is composed of high degrees of inorganic components (∼95% calcium carbonate in the form of calcite) and ∼5% of organic components (type X collagen, sulphated polysaccharides) (5–7). The global estimate for egg production is 65.5 million metric tons per year, with Asia leading the growth in world output (8). Notwithstanding this, the projected total production of eggs in South Africa is 133.9 million per week (9). Apart from an eggshell being lightweight, crushable and porous, it weighs approximately 11% of the total mass (ca. 60 g) of an egg (6, 7, 10, 11). This together with it being cheap, abundantly available and renewable makes eggshells a valuable waste material, one that can be used for various dental applications.
In the last decade, eggshells have reportedly been used as an additive for animal feed and human nutrition (12–14), coating pigments for inkjet printing paper (15), a natural absorbent for heavy metals (14, 16–20) and a dye-effluent removers (14, 21–24), and as biofillers for polymer-based composites (10, 14, 25–29). Recently, there has been an increased interest in using eggshell powder for abrasive purposes. Henuset (30) and Brand-Garnys (31), for example, reportedly used eggshells to manufacture abrasive paper, abrading wheels, bands and disks, and cosmetics. Chen (32), on the other hand, revealed that eggshells of particle size between 0.1 μm to 10 nm produced an ultra-fine powder, which made them useful in toothpastes and cosmetics. There has, however, been limited research on using eggshell powder as an abrasive polishing material for dental applications. This study therefore aimed to characterize an eggshell powder abrasive material (EPAM) as an alternative material to pumice in the polishing of dental appliances. The formulated null hypothesis was that EPAM is not a suitable alternative to pumice as it does not reduce the surface roughness of poly(methyl methacrylate) (PMMA) specimens.
Materials and Methods
Preparation of EPAM
Eggshells collected from food outlets were washed to remove impurities. Disinfection then followed by storing the eggshells in a diluted solution of household sodium hypochlorite for 6 hours. Subsequently, eggshells were vacuum dried for ±6–9 minutes at 250°C and crushed to a powder using a blender. Linear alkyl benzene sulfonate (LAS; 0.06 kg) was added to the eggshell powder (1.26 kg) to improve its solubility in water. The mixture was then ball milled to a fine powder, following 2 steps: In the first step, 6 g of the powder was placed in a 500-mL stainless steel jar (inner diameter of 100 mm), together with 10 stainless steel balls of 10-mm diameter, and dry-milled in a planetary ball mill (Retsch® PM 100) at 400 rpm for 50 minutes. Subsequently, the fine powder obtained from step 1 was further milled with 10 stainless steel balls of 1-mm diameter for an additional 50 minutes.
Methods of Characterization
Fourier transform infrared spectroscopy analysis
The infrared spectra for Fourier transform infrared (FTIR) spectroscopy were measured using a Perkin Elmer Universal ATR spectrometer to identify the structure and the functional group constituents of EPAM.
X-ray diffraction analysis
X-ray diffraction (XRD) analysis was performed to observe the possible changes in crystallinity between the eggshell powder (without anionic surfactant) and EPAM. The XRD patterns were recorded using a diffractometer (PANalytical-Empyrean instrument; Co radiation 1.54056 A) and analyzed between 0–90° (20). The voltage, current and pass time used were 40 kV, 40 mA and 1 second, respectively.
Dissolution and Solubility Test
The solubility and dissolution rate of EPAM were determined using a shake flask method. This involved adding 3 g of EPAM in 50-mL distilled water contained in a volumetric flask, which was then stirred for 24 hours at room temperature. Subsequently, the mixture was filtered using 0.5-μm filter paper to separate the slurry from the solvent. After filtration, the calcium content in the filtrate was analyzed using inductively coupled plasma optical emission spectroscopy (ICP-OES) (ICPE-9000; Shimadza).
Transmission Electron Microscopic Analysis
A transmission electron microscope (TEM) was used to observe the particle size, shape and distribution of EPAM. Very small quantities of EPAM were dispersed in 10-mL ethanol and sonicated at 10 kV for 10 minutes. Subsequently, thin cross-sections of cryomicrotomed specimens were prepared using a Leica microtome (South Africa) and placed on carbon copper grids. Analysis was conducted using a TEM (Philips CM 120 model) at 120 kV.
Laser Diffraction Particle Size Analyzer
The particle size distribution of EPAM was measured using a HORIBA Laser Particle Size Analyzer (LA-950). The EPAM powders were dispersed in isopropanol and used in the funnel of the LA-950 analyzer. The form of distribution was set at manual mode. Particle size distribution was calculated on the volume basis. The refractory indexes of EPAM and isopropanol were kept at 1.660 and 1.378, respectively.
Mechanical Evaluation of the Abrasive Characteristics of EPAM
The abrasive characteristics of EPAM were evaluated by comparison using PMMA resin specimens, which is the material commonly used in the fabrication of dental appliances (33). It was assumed that if EPAM matched the abrasive characteristics of pumice, which is the abrasive material commonly used in the polishing of dental appliances (34), then it would be a suitable alternative material.
Preparation of PMMA Specimens
A custom stainless steel mould (50 x 20 x 3 mm) was used to prepare 20 PMMA specimens. All specimens were polymerized according to the manufacturer's recommendation (Vertex-Dental B.V., The Netherlands). After curing, specimens were trimmed with a tungsten carbide bur (Cross-cut, coarse – ISO No. 500104237065; Bredent GmbH and Co KG) at 18,000 rpm. Prior to final polishing, all specimens were smoothed using an abrasive paper (CC768 Silicon Carbide; Deer Abrasives, Ridgefield, NJ, USA). Thereafter, specimens were polished with EPAM slurry and Navajo® pumice slurry with particle size of 15 μm (control) for 2 minutes. It must be noted that each sample group had 10 specimens.
Surface roughness analysis
The surface roughness (Ra) values of the PMMA specimens was analyzed using a Wintrace surface analysis system (Taylor Hobson®). The profilometer was calibrated with a cutoff filter of 0.8 mm, together with an evaluation length of 4.00 mm and a range of 5.1 μm. Five Ra value measurements of surface roughness were performed for each specimen; the mean average was used for the statistical analysis.
Results
Characterization of EPAM
Following the successful modification of eggshell powder through ball milling, the FTIR spectra of eggshell powder and EPAM in Figure 1 revealed several bands from 4,000 cm-1 to 400 cm-1. There are prominent absorption peaks of carbonates at 1,405.50 cm-1, which is associated with the carbonyl group. The absorption band aspect of the FTIR spectra show the differences between the eggshell powder and EPAM. In contrast to eggshell powder (Fig. 1A), EPAM (Fig. 1B) shows a specific band around 1,000–1,200 cm-1. This corresponds to the alkyl chain of surfactant and further confirms the surface modification of EPAM with the LAS.
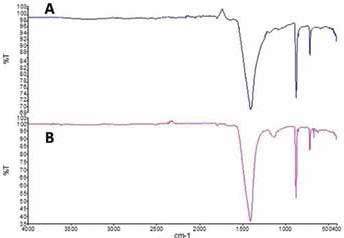
Fourier transform infrared (FTIR) spectra of eggshell powder (A) and eggshell powder abrasive material (EPAM) (B).
In Figure 2, the XRD patterns for eggshell powder and EPAM show characteristic diffraction peaks with diffraction angles at 27.5°, 34.5°, 42.5°, 46.5°, 50.5°, 56°, 57°, 67°, 68° and 72°. The characteristic peak marked at around 34.5° (20) indicates that both eggshell powder and EPAM contain a thermodynamically stable calcite crystalline structure, which is similar to calcium carbonate. The XRD pattern in Figure 2 further revealed differences in the mineral content of eggshell powder and EPAM. The eggshell powder contains calcite, apatite and graphite 2H, whereas EPAM has calcite, graphite and thenardite (Tab. I). As illustrated in Figure 3, the amount of calcium released into distilled water was higher for the bulk eggshell (26.67 mg/L) than the EPAM (6.67 mg/L).
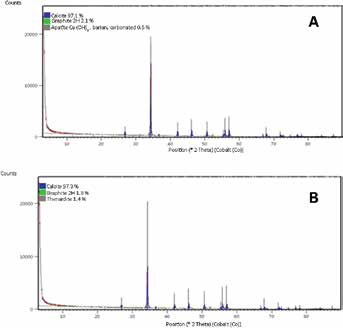
X-ray diffraction (XRD) patterns of eggshell powder (A) and eggshell powder abrasive material (EPAM) (B) showing mineral content.
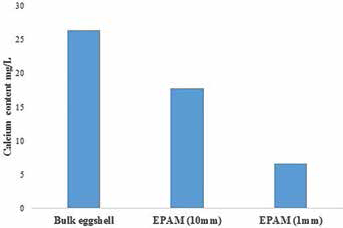
Calcium content levels of bulk eggshell and eggshell powder abrasive material (EPAM) in distilled water.
Mineralogical composition of eggshell powder and EPAM, as determined by XRD analysis
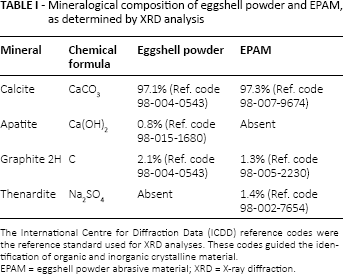
The International Centre for Diffraction Data (ICDD) reference codes were the reference standard used for XRD analyses. These codes guided the identification of organic and inorganic crystalline material.
EPAM = eggshell powder abrasive material; XRD = X-ray diffraction.
The TEM images in Figure 4 reveal that EPAM particles are irregular in shape, with an angular structure.
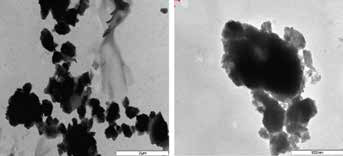
Transmission electron microscopy (TEM) images of eggshell powder abrasive material (EPAM), showing particle shapes.
Figures 5 illustrates the particle size distribution of EPAM in a histogram and S-curve. The average mean size of the 100-mm ball milled EPAM is 0.3 μm, whereas the 1-mm ball milled EPAM is 50 nm.
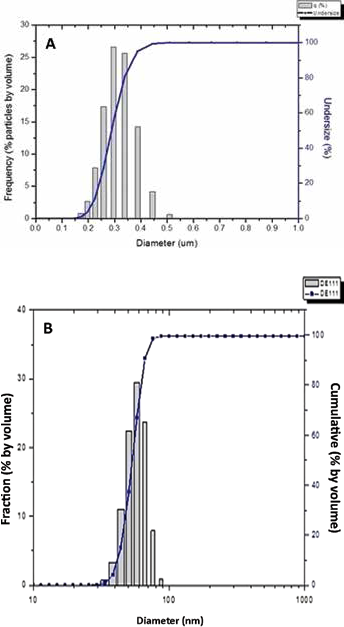
Different particle size distributions of eggshell powder abrasive material (EPAM): fine particles (A); nano particles (B).
Abrasive Characteristics of EPAM
Figure 6 illustrates the surface roughness values of the PMMA specimens at each abrasive application stage – that is – grinding the specimens with a tungsten carbide bur, smoothing with silicon carbide, polishing the specimens with EPAM (fine and nanorange) and polishing the specimens using pumice. In contrast to those polished with pumice, specimens polished with EPAM showed improved polished surfaces, with mean Ra values of 0.04 and 0.06 μm.
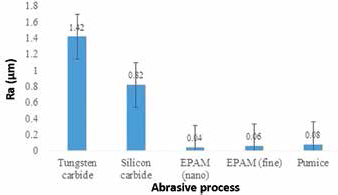
Mean differences in surface roughness reductions at each processing stage: grinding the specimens with a tungsten carbide bur, smoothing with silicon carbide, polishing the specimens with eggshell powder abrasive material (EPAM) (fine and nanorange) and polishing the specimens using pumice. Whiskers show standard deviations.
Discussion
Calcium carbonate is used as a mildly abrasive paste to polish tooth enamel, gold foil, amalgam and plastic materials (35). The hydrophilic property of calcium carbonate (CaCO3) as a constituent of eggshell, however, has limited its use as a denture polisher. A critical point deserving mention is that the common practice when polishing dentures is to mix the abrasive powder with water to form a slurry. The presence of sodium sulfate molecule (So32-) coating on the surface of EPAM (Fig. 1B) confirmed that it mixed with distilled water to form slurry. Eggshell powder should therefore be modified to mix with water when polishing acrylic dentures. Moreover, the distinct calcium carbonate patterns in Figure 2 suggest that both materials have high levels of carbonate. This is different from mined and precipitated calcium carbonates, which usually have undesirable concentrations of other elements (31).
Equally significant, the prominent absorption peaks of carbonates at 1,405.50 cm-1 (Fig. 1) support the argument of Mosaddegh and Hassankhani (36), who claimed that carbonate-based materials are commonly detected by the broad stretching frequency of the C=O in carbonate ions at around 1,416 cm-1. The reported absorption peak of calcite at 712.76 cm-1 and 873.53 cm1 is consistent with the results in the work of several authors (10, 28, 37–40), particularly that the asymmetric stretching, out-of-plane bending and in-plane bending vibration modes are due to the presence of CO32- molecules. It can therefore be gathered that the ball milling process does not adversely affect the carbonate structure of EPAM. Generally, thenardite occurs naturally as an anhydrous sodium sulfate and is known to provide good stability and high solubility in water (41). Significantly, thenardite was present in EPAM (Fig. 2), and this could be attributed to the formation of sodium sulfate (Na2SO4). This compound may have formed by the surface modification of EPAM with LAS (42). Equally important, the lesser amount of calcium (6.67 mg/L) measured in distilled water further supports LAS forming a coating on the EPAM surfaces. This finding is consistent with those of Barhoum et al (43). LAS coating was also more effective when the particle size of EPAM was reduced from fine (17.8 mg/L) to nano-sized (6.67 mg/L).
From a dental laboratory perspective, and consistent with reports by O'Brien (44), the angularity of EPAM (Fig. 4) is more likely to produce more highly polished surfaces on dental prostheses. The different Ra values between the fine and nano-sized EPAM (Fig. 6) further support the argument of Bassam et al (45) that abrasive materials of smaller particle sizes create newly formed and sharper particles faster during the abrasion process. Consequently, the surface roughness of dentures is reduced rapidly. Importantly, the null hypothesis in this present study was rejected, as the Ra values of EPAM were below the acceptable threshold limit value of 0.2 μm (46, 47), which confirmed that EPAM effectively reduced surface roughness. Arguably, and contrary to the common understanding that harder abrasive materials such as pumice produce smoother polished surfaces (35, 45), the reported surface roughness value (Ra = 0.04 and 0.06 μm) suggests that EPAM can produce smoother polished surfaces. Moreover, the inclusion of LAS in EPAM tightly holds the EPAM particles together, thereby increasing the abrasion rate and subsequently the smoothness of the denture surfaces.
Conclusion
The key findings of this study showed that EPAM is a pure calcite with traces of graphite 2H and thenardite. Notably, this study conclusively showed that EPAM effectively reduces the surface roughness to below the threshold limit value of 0.2 μm, thereby confirming that 50-nm and 0.3-μm particle sizes provide clinically acceptable polished surfaces. From an environmental sustainability and management perspective, using the EPAM to polish removable dental appliances strengthens the economic benefits associated with using natural waste material, which is high on the global agenda for a greener environment. This is an area for further research.
Footnotes
Financial support: The authors acknowledge the financial support for this study from the National Research Foundation of South Africa (No. 104824).
Conflict of interest: None.
