Abstract
Obesity-associated adipocyte dysfunction promotes chronic inflammation, insulin resistance, and dysregulated lipolysis, contributing to metabolic disorders. This study addresses the unclear mechanisms of gamma-aminobutyric acid (GABA) in adipocytes by investigating its coordinated effects on three core metabolic pathways: inflammation, insulin resistance, and lipolysis. We utilized a palmitic acid (PA)-induced adipocyte dysfunction model in 3T3-L1 cells. Results showed that 800 µM PA for 24 hours induced insulin resistance and increased IL-6 and TNF-α secretion in mature 3T3-L1 adipocytes (Day 9). GABA treatment (500–1,000 µM) for nine days effectively prevented PA-induced insulin resistance, as insulin-stimulated glucose uptake was significantly improved. These high doses of GABA also reduced PA-induced IL-6 and TNF-α release. Regarding lipolysis, PA directly increased glycerol release and weakened insulin’s ability to suppress lipolysis. GABA (250–1,000 µM) dose-dependently reduced glycerol release and restored insulin’s anti-lipolytic effect in PA-treated adipocytes. GABA’s anti-inflammatory activity was further confirmed in RAW macrophages, where its dose-dependently inhibited nitric oxide (NO) production induced by 1 µg/mL lipopolysaccharide (LPS). However, its inhibitory effect on NO release in adipocytes was unclear, likely due to the low NO levels in LPS-treated adipocytes. In conclusion, although only high concentrations of GABA attenuated PA-induced adipocyte dysfunction through coordinated regulation of insulin signaling, inflammation, and lipolysis in cultured cells, these findings warrant further investigation into the potential of GABA and/or GABA receptor agonists in obesity-related metabolic dysfunction.
Introduction
Obesity is a major global public health concern, substantially increasing the risk of insulin resistance, type 2 diabetes mellitus (T2DM), and cardiovascular diseases. 1 It is characterized by excessive fat accumulation and metabolic dysfunction driven by hypertrophic adipocytes, which secrete free fatty acids (FFAs) and pro-inflammatory cytokines, including interleukin-6 (IL-6) and tumor necrosis factor-alpha (TNF-α).2–5 These mediators impair adipocyte function and recruit immune cells, establishing chronic low-grade inflammation that exacerbates insulin resistance and contributes to obesity-related complications.6,7
In addition to inflammation, dysregulated lipolysis—the hydrolysis of triglycerides into glycerol and FFAs—plays a critical role in metabolic disturbances. 8 Excessive lipolysis increases circulating FFAs, promotes ectopic fat deposition, and amplifies inflammatory responses, linking adipocyte dysfunction to systemic metabolic disorders. 8 Therefore, regulation of both inflammation and lipolysis is central to maintaining adipocyte health and metabolic homeostasis.
Natural bioactive compounds have attracted attention for mitigating obesity-related dysfunctions due to their favorable safety profiles. Gamma-aminobutyric acid (GABA), a non-protein four-carbon amino acid abundant in vegetables, fruits, and fermented foods, exhibits anti-inflammatory properties by reducing TNF-α and IL-6 production and modulating immune responses through GABAA receptor activation.9–13 GABA also improves glucose tolerance, enhances insulin sensitivity in high-fat diet-fed mice, and prevents pancreatic islet inflammation (insulitis), highlighting its therapeutic potential for metabolic disorders.4,14
Despite these findings, the precise mechanisms by which GABA modulates adipocyte inflammation, insulin resistance, and lipolysis remain unclear. Understanding these pathways is essential for developing effective strategies against obesity-related complications. In this study, we employed a palmitic acid (PA)-induced adipocyte dysfunction model to investigate the effects of GABA on adipose inflammation, insulin resistance, and lipolysis. Specifically, we examined its impact on pro-inflammatory cytokine secretion (IL-6 and TNF-α), its inhibitory action on LPS-induced nitric oxide (NO) production in macrophages and adipocytes, and its regulatory effects on lipolysis, a key process closely associated with adipocyte dysfunction and metabolic disease.
Materials and Methods
Chemicals
GABA, PA, 3-isobutyl-1-methylxanthine (IBMX), dexamethasone, Oil Red O, isopropanol, MTT, and insulin were obtained from Sigma-Aldrich (St. Louis, MO, USA). DMEM, calf bovine serum (CBS), fetal bovine serum (FBS), trypsin–EDTA, and penicillin–streptomycin were sourced from Gibco (Carlsbad, CA, USA). The fluorescent glucose analog 2-NBDG was purchased from Invitrogen™ (Thermo Fisher Scientific, Waltham, MA, USA). ELISA kits for murine IL-6 (Cat. No. 900-T50) and TNF-α (Cat. No. 900-T54) were supplied by PeproTech→ (Thermo Fisher Scientific, Waltham, MA, USA).
3T3-L1 Cell Culture and Differentiation
Mouse 3T3-L1 pre-adipocytes (RCB1124) were obtained from the RIKEN BRC (Saitama, Japan) and maintained in high-glucose DMEM with 10% CBS and antibiotics at 37°C with 5% CO₂. Differentiation was induced by culturing cells for three days in high-glucose DMEM containing 10% FBS, 1 µM dexamethasone, 0.5 mM IBMX, and 10 µg/mL insulin. Cells were then placed in high-glucose DMEM with 10% FBS and 10 µg/mL insulin and maintained for nine days, with medium changed every three days.
Cell Treatments
PA was solubilized in 100% ethanol (75 mM) in a heated, stirred water bath at 90°C for 15 minutes. PA containing culture medium was prepared by mixing PA solution and 5% fatty acid-free BSA in high-glucose DMEM, and incubated at 60°C for 30 minutes under shaking to ensure PA-BSA conjugation (~1:1) before being added to 3T3-L1 adipocytes. 5% fatty acid-free BSA medium was used as vehicle control. GABA (125–1,000 μM) was freshly dissolved in sterile water, and sterile water was used as vehicle control. For the treatment, 3T3-L1 adipocytes were continuously exposed to GABA for nine days covering differentiation and lipogenesis periods (GABA-containing medium refreshed every three days). On day 9, cells were washed with PBS and exposed to 800 µM PA containing culture medium for 24 hours in the presence of GABA at tested concentration.
Cell Viability Determination
Cell viability was assessed using the MTT assay. 3T3-L1 cells were seeded at a density of 5 × 10³ cells per well in 96-well plates. Following differentiation, cells were exposed to GABA (0–1,000 μM) for nine days. MTT (0.5 mg/mL) was added for the final two hours, formazan was dissolved in 200 μL DMSO, and absorbance was read at 595 nm.
Determination of Glucose Uptake in Adipocytes
Glucose uptake in adipocytes was determined via a 2-NBDG fluorescent assay using 3T3-L1 cells in black 96-well plates. Cells were treated with varied GABA concentrations (125–1,000 μM) for nine days and cells were exposed to 800 µM PA (in 5% fatty acid-free BSA) for 24 hours. Cells were washed with PBS, incubated overnight in glucose-free and serum-free DMEM, and incubated with 2-NBDG (25 µM) in the absence and presence of insulin (1 µg/mL) for four hours, and fluorescence was measured at 485/535 nm.
Determination of Pro-inflammatory Cytokines by ELISA
Cells were treated with varying concentrations of GABA (250–1,000 μM) for durations specific to each experiment. The culture medium was collected to quantify pro-inflammatory cytokines, including IL-6 and TNF-α, using commercial ELISA kits. All assays were conducted following the manufacturer’s instructions provided with the kits.
Determination of Lipolysis Via Glycerol Release from Adipocytes
After cell treatment, cells were washed and further incubated for four hours in serum-free culture medium with or without insulin in the presence of PA and/or GABA at tested concentration. Isoproterenol (Iso) at 1 μM was used as a positive control as glycerol release inducer. Glycerol levels were quantified using an adipolysis assay kit based on a coupled enzymatic reaction measured colorimetrically at 570 nm.
Statistical Analysis
All data are expressed as mean ± standard deviation (SD) from at least three independent experiments (n = 6). Statistical analyses were performed using one-way analysis of variance (ANOVA) followed by LSD (least significant difference) post hoc tests. A p value of ≤ .05 was considered statistically significant (*).
Results
Effect of GABA on PA-induced Insulin Resistance in 3T3-L1 Adipocytes
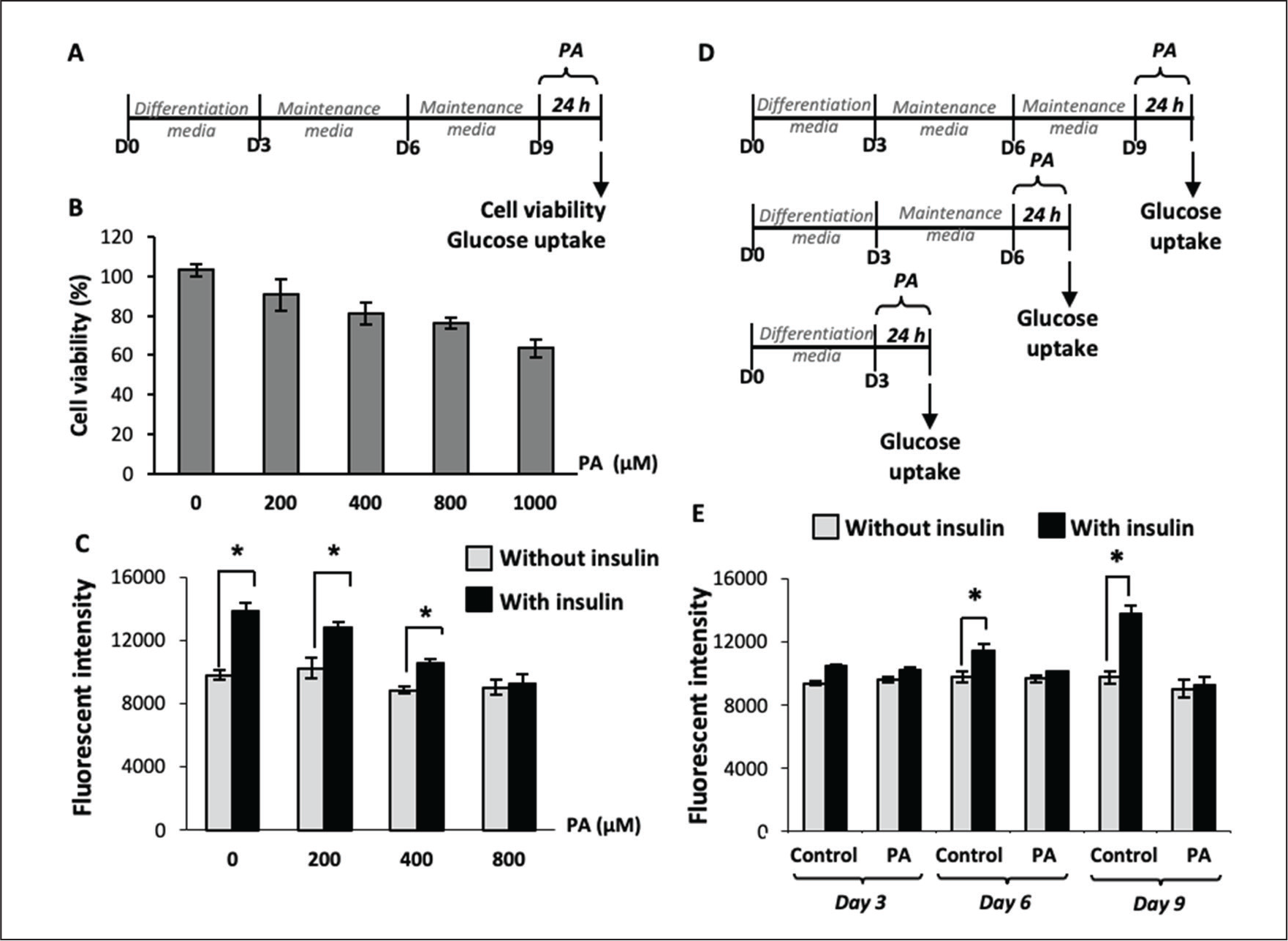
3T3-L1 adipocytes showed different levels of insulin responsiveness depending on their age in culture, indicating a time-dependent effect. Adipocytes at days 3, 6, and 9 displayed progressively stronger insulin-stimulated glucose uptake (Figure 1E). Mature adipocytes at day 9 were therefore chosen for further experiments on PA-induced adipocyte dysfunction. PA at concentrations of 200–1,000 µM caused slight reductions in viability as determined by the MTT assay (Figure 1B). PA impaired insulin-stimulated glucose uptake in a dose-dependent manner, with 800 µM completely eliminating insulin responsiveness (Figure 1C). Therefore, 800 µM PA was chosen for modeling insulin resistance. Day 9 adipocytes treated with 800 µM PA were used as the insulin-resistant model for evaluating the effects of GABA.
PA Induced Insulin Resistance in Mature 3T3-L1 Adipocytes. Schematic of the Cell Treatment Protocols (A) and (D). Mature 3T3-L1 Adipocytes (Day 9) were Treated with Varying Concentrations of PA for 24 h Prior to Cell Viability by MTT Assay (B) and Glucose Uptake Assay (C). The Glucose Uptake Assay was Conducted in the Presence and Absence of Insulin. Glucose Uptake was Assessed in Adipocytes at Days 3, 6, and 9 of Culture Following Treatment with 800 µM PA, with Untreated Cells (Medium Only) Serving as the Control (E). Data are Mean ± SD (n = 6). * p ≤ .05 Compared Between Groups.
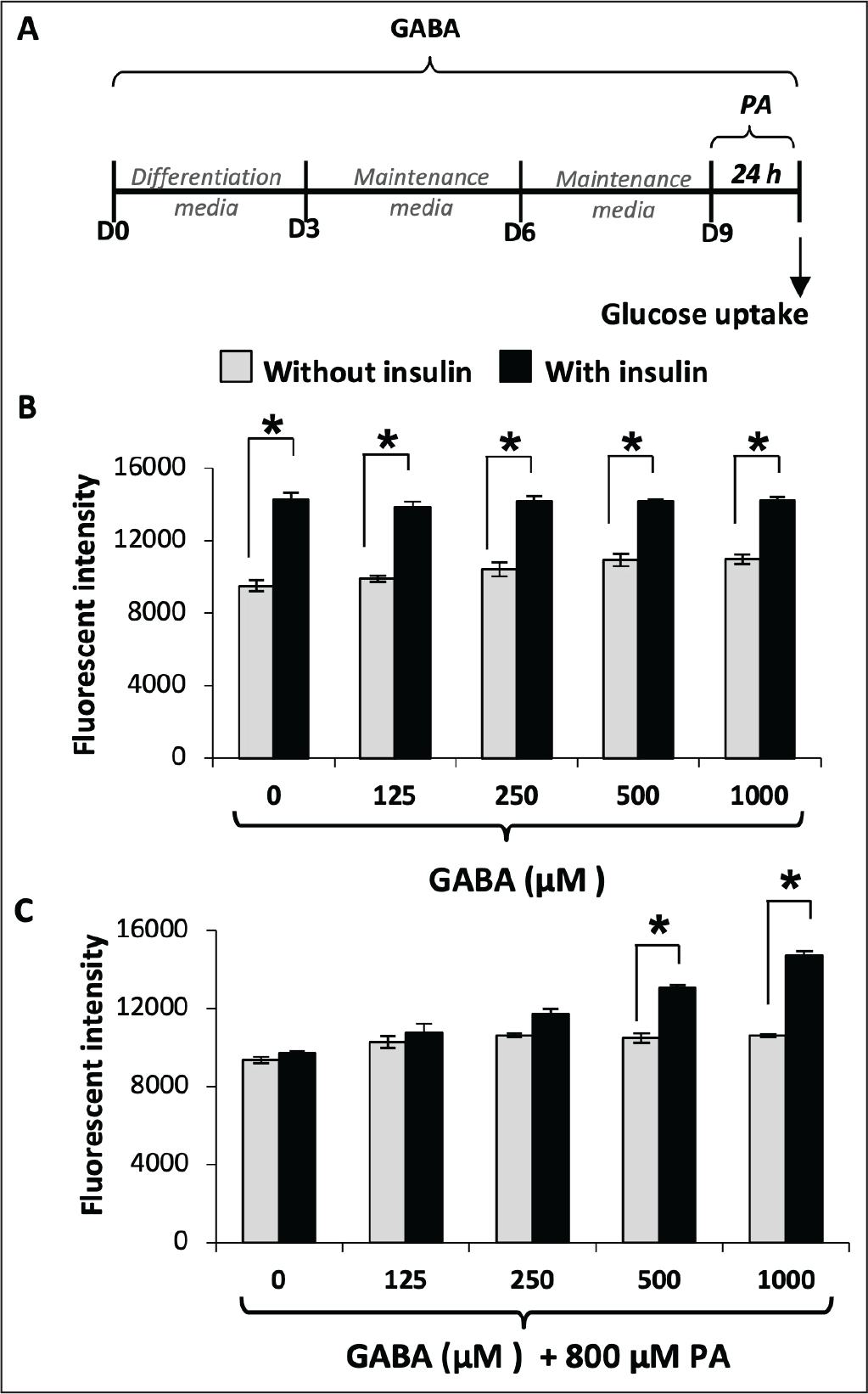
Pretreating 3T3-L1 adipocytes with GABA for nine days did not affect basal glucose uptake or their normal insulin response (Figure 2B). In PA-treated adipocytes, however, GABA dose-dependently improved insulin-stimulated glucose uptake, increasing it by approximately 25% at 500 µM and 40% at 1,000 µM (Figure 2C). These results showed that GABA enhanced insulin sensitivity in PA-induced insulin-resistant adipocytes without altering basal glucose uptake.
Effect of GABA on PA-induced Insulin Resistance in Mature 3T3-L1 Adipocytes. (A) Schematic of the Protocol. (B) Adipocytes were Cultured with Various Concentrations of GABA (Medium Refreshed Every Three Days Until Day 9) (C) and then Exposed to 800 µM PA for 24 h. The Glucose Uptake Assay was Conducted in the Presence and Absence of Insulin. Data are Mean ± SD (n = 6). *p ≤ .05, Compared Between Groups.
Effect of GABA on IL-6 and TNF-α Release in PA-induced Insulin Resistance 3T3-L1 Adipocytes
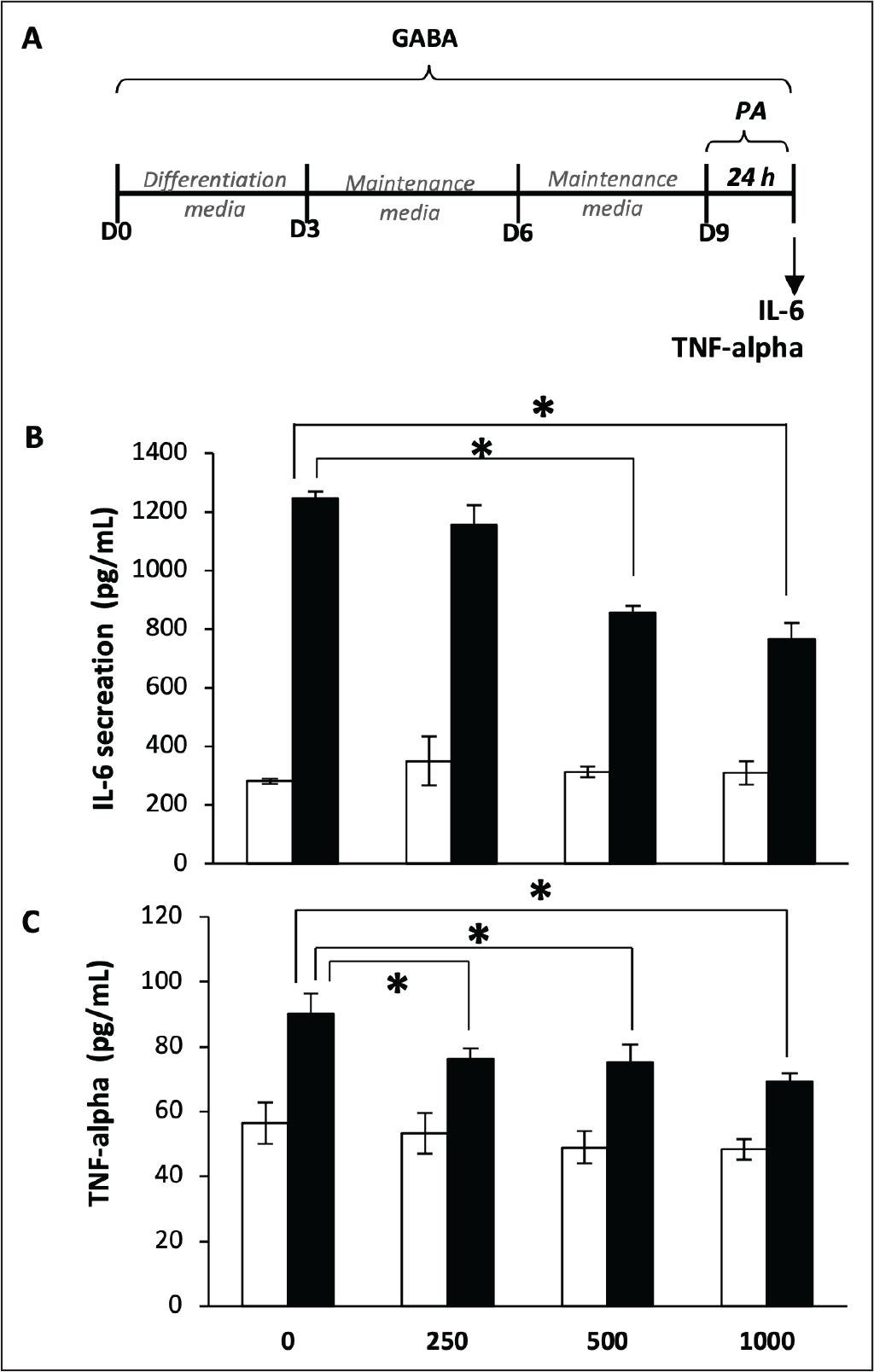
IL-6 and TNF-α secretion was measured in mature 3T3-L1 adipocytes (Day 9) after 24 h of treatment with 800 µM PA (Figure 3A). PA significantly increased the release of both cytokines, while GABA alone had no effect at any concentration tested (Figures 3B and 3C). In PA-treated adipocytes, however, GABA dose-dependently reduced IL-6 and TNF-α secretion, with significant inhibition observed at 500 µM and 1,000 µM. Although GABA did not fully restore cytokine levels to baseline, it clearly demonstrated anti-inflammatory activity under PA-induced lipotoxic conditions. Overall, these findings indicate that mature adipocytes produce inflammatory cytokines in response to lipotoxicity, and GABA can suppress this inflammatory response.
Effect of GABA on IL-6 and TNF-α Release in PA-induced Insulin Resistance Mature 3T3-L1 Adipocytes. (A) Schematic of the Protocol. Adipocytes were Cultured with Various Concentrations of GABA (Medium Refreshed Every Three Days Until Day 9) in the Absence and Presence of 800 µM PA. After 24 Hours PA Treatment, Culture Medium were Collected to Determine (B) IL-6 and (C) TNF-α using ELISA. Data are Mean ± SD (n = 6). * p ≤ .05 Compared with PA-treated Cells without GABA.
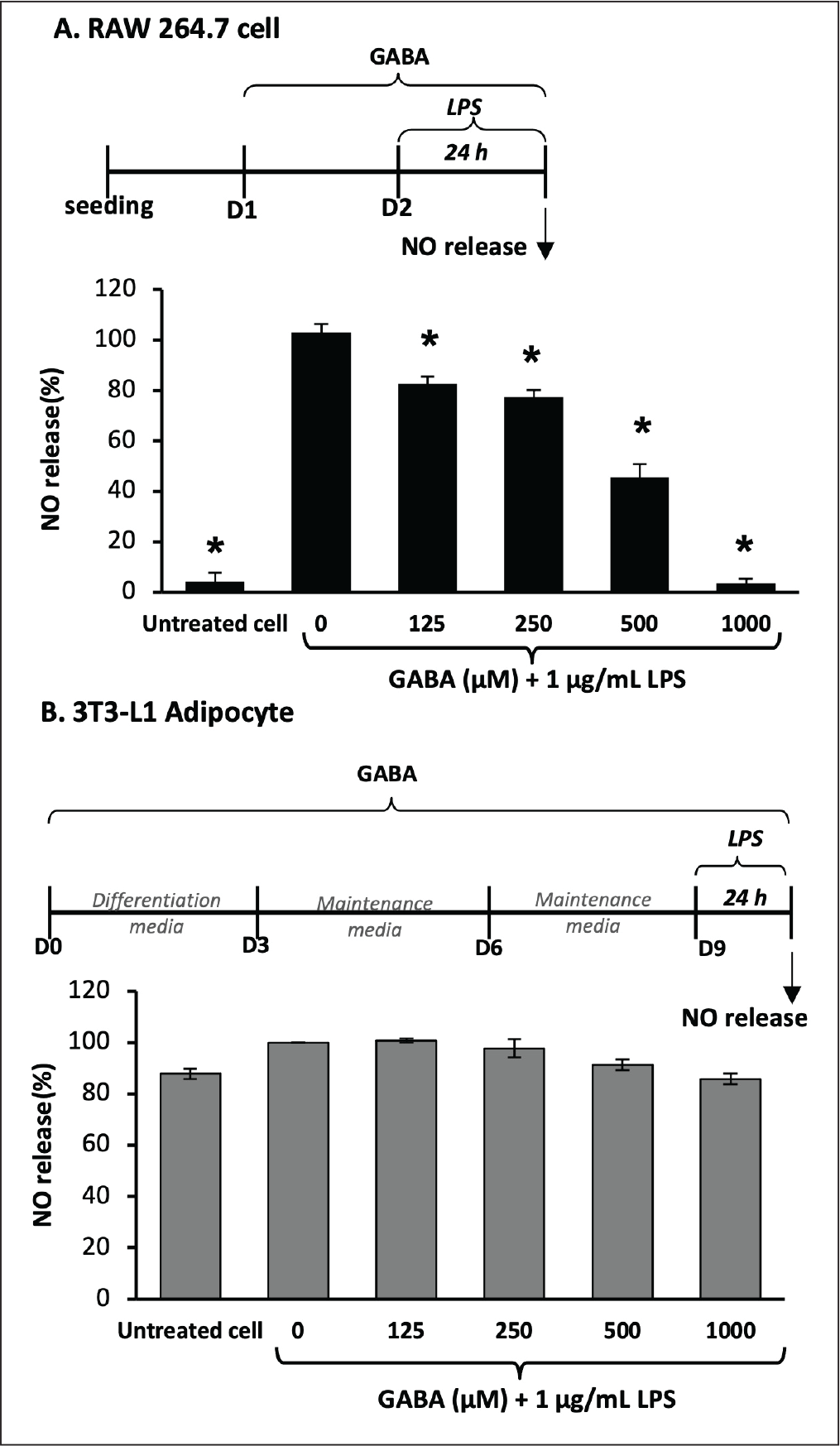
Effect of GABA on NO Synthesis
NO release was measured in RAW264.7 cells (Figure 4A) and mature 3T3-L1 adipocytes (Figure 4B). LPS (1 μg/mL) markedly increased NO secretion in RAW264.7 cells but induced only a minimal response in mature 3T3-L1 adipocytes. In RAW264.7 cells, GABA significantly reduced LPS-stimulated NO production in a dose-dependent manner, with 1,000 μM producing the greatest reduction (96%). In contrast, GABA had no measurable effect on LPS-treated 3T3-L1 adipocytes, likely because LPS induced only low levels of NO in these cells. These results suggest that the inflammatory responses of different cell types are dependent on the specific stimuli to which they are exposed.
Effect of GABA on Nitric Oxide (NO) Release in RAW264.7 and 3T3-L1 Adipocytes. (A) RAW264.7 Cells were Pretreated with GABA for 24 Hours and then Stimulated with 1 µg/mL LPS for 24 Hours; NO Levels in the Medium were Measured. (B) 3T3-L1 Adipocytes were Cultured with GABA Until Day 9 and Exposed to 1 µg/mL LPS for 24 Hours; NO Levels were Quantified. Data are Mean ± SD (n = 6). * p ≤ .05 Compared with LPS-Treated Cells (no GABA).
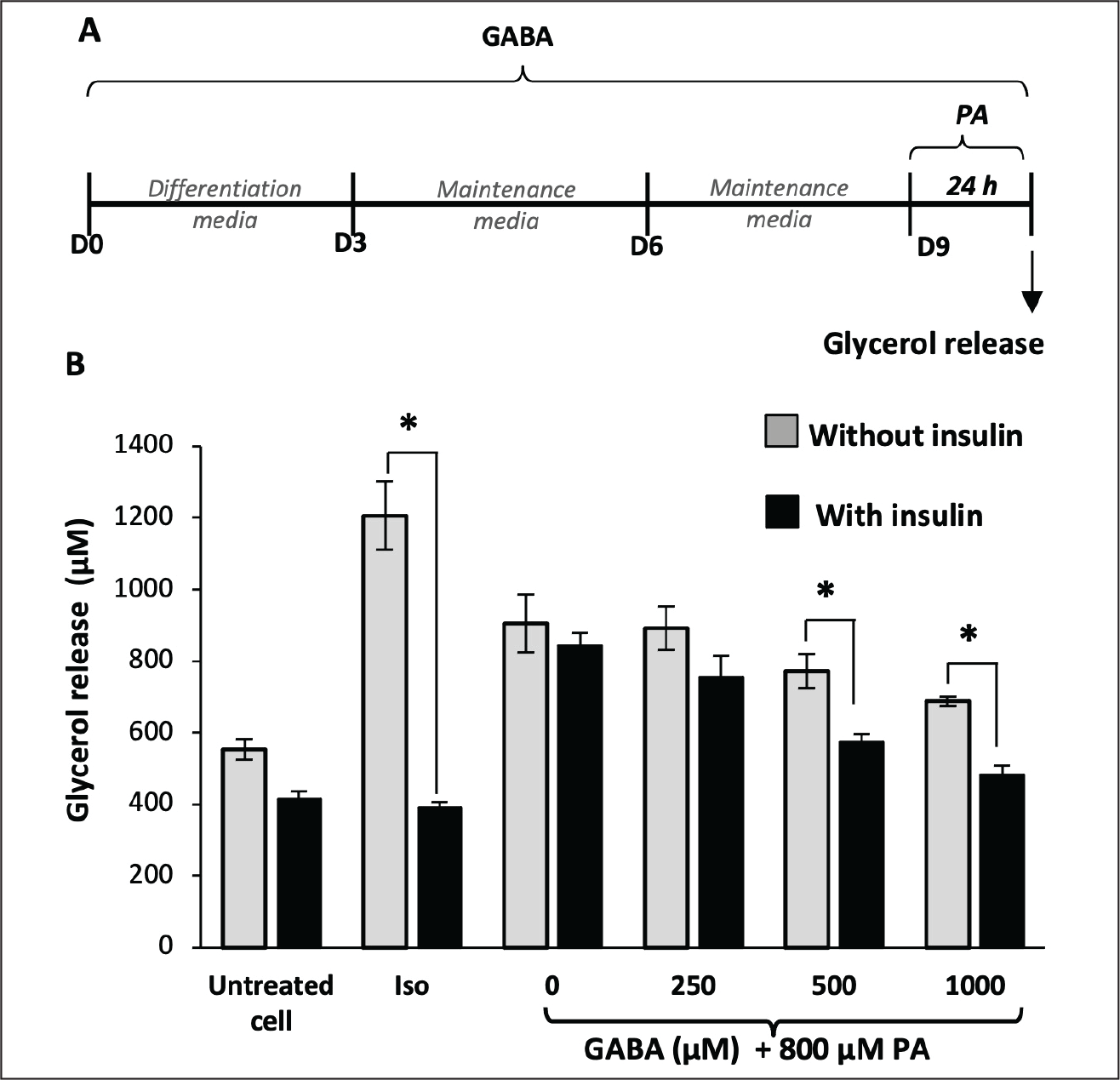
Effect of GABA on Lipolysis of Mature 3T3-L1 Adipocytes
Lipolysis was evaluated by measuring glycerol release from mature 3T3-L1 adipocytes (Day 9). Isoproterenol (Iso, 1 μM) strongly increased glycerol release, confirming its ability to stimulate triacylglycerol (TAG) breakdown (Figure 5B). In normal adipocytes, insulin reduced glycerol release in both control and Iso-treated cells, indicating its suppressive effect on lipolysis. In contrast, PA (800 μM) increased glycerol release with or without insulin, suggesting that PA impaired insulin’s ability to inhibit lipolysis. In PA-treated cells, GABA reduced glycerol release in a dose-dependent manner and restored insulin’s lipolysis-suppressing effect (Figure 5B). These results indicate that GABA improved insulin sensitivity in PA-induced insulin-resistant adipocytes.
Effect of GABA on Glycerol Release from Mature 3T3-L1 Adipocytes. (A) Schematic of the Protocol. Adipocytes were Cultured with (A) GABA (Medium Refreshed Every Three Days Until Day 9) and Exposed to (C) 800 µM PA (24 Hours). Cells were then Treated with and without Insulin for Four Hours before Determination of Glycerol Level. 1 µM Isoproterenol (Iso) was Used as Glycerol Release Inducer. Data are Mean ± SD (n = 6). * p ≤ .05 Compared Between Groups.
Discussions
Obesity promotes metabolic dysfunction via insulin resistance, chronic inflammation, and excess lipolysis-derived fatty acids from pathologic adipocytes.14,15 This study investigates the mechanisms of GABA in adipocytes by examining its effects on inflammation, insulin resistance, and lipolysis. This study employed PA-induced 3T3-L1 adipocyte dysfunction models, and we found that PA induced insulin resistance, inflammation, and increased lipolysis. GABA reversed these effects by improving insulin signaling, lowering cytokine secretion, restoring insulin’s anti-lipolytic action, and suppressing LPS-stimulated NO in macrophages.
Previous studies have shown that PA impairs insulin-stimulated glucose uptake 16 and elevates pro-inflammatory cytokines such as TNF-α and IL-6,17,18 in agreement with our findings. The PA concentration used in this study (800 µM) is within the physiological range. Although evidence regarding toxic plasma FFAs concentrations in humans is limited, plasma palmitic acid levels in a diverse cohort of healthy young Canadian adults (n = 826) have been reported to range from 300 µM to 4,100 µM. 19 In this study, GABA mitigated PA-induced adipocyte dysfunction by improving insulin-stimulated glucose uptake and suppressing IL-6 and TNF-α release. These observations are consistent with previous reports describing the anti-inflammatory actions of GABA in HFD-fed mice and skeletal muscle.7,20 Evidence indicates that adipocytes express both GABAA and GABAB receptors,3,21 although their functional roles in these cells have not been clearly established. Evidence from other studies indicates that GABAA receptor activation inhibits inflammatory diseases, and these receptors are particularly central to GABA’s anti-inflammatory and metabolic effects.3,9–11 Furthermore, GABAB receptors promote leptin mRNA expression through an unidentified mechanism related to leptin gene transactivation in adipocytes, 21 supporting adipose tissue mass homeostasis. 22 Thus, GABA receptor activation may influence adipocyte dysfunction, inflammation, and obesity, but current evidence remains limited, underscoring the need for more comprehensive investigation.
Excessive lipolysis elevates circulating FFAs, drives ectopic fat deposition, and intensifies inflammation, contributing to systemic metabolic dysfunction. 8 In obesity, proinflammatory cytokines such as TNF-α and IL-6 impair insulin signaling and stimulate lipolysis,23–25 further elevating basal lipolytic activity and insulin resistance. Thus, controlling inflammation and lipolysis is critical for maintaining adipocyte and metabolic homeostasis. We show that lipolysis was assessed by measuring glycerol release from mature 3T3-L1 adipocytes (Day 9). In normal adipocytes, insulin suppressed lipolysis, consistent with previous reports showing that insulin inhibits both basal and catecholamine-stimulated lipolysis. 23 Increased lipolysis in dysfunctional adipocytes elevates circulating FFAs, promoting lipotoxicity and insulin resistance.23,26
Similarly, PA exposure augmented lipolysis in 3T3-L1 adipocytes and impaired insulin-mediated suppression of glycerol release. GABA lowered glycerol release and restored insulin’s suppression of lipolysis in PA-treated adipocytes, indicating improved insulin sensitivity. This is consistent with prior evidence that GABA enhances insulin sensitivity in high-fat-diet mice 2 and with evidence from animal studies using GABA-enriched germinated brown rice.27,28 Germinated brown rice reduced adipogenesis markers including fatty acid synthase (FAS), and increased expression of lipolytic enzymes such as hormone-sensitive lipase (HSL) and adipose triglyceride lipase (ATGL) in adipose tissue of high-fat diet-induced obese mice. 27 In mature adipocytes, our previous study showed that GABA promotes lipolysis. 13 This provides further insight into GABA’s lipolytic effects in both normal and insulin-resistant adipocytes.
The β3-adrenoceptor primarily promotes lipolysis in adipose tissue. 29 Beta-blockers also inhibited the actions of both Iso and GABA, suggesting an interaction between GABA and beta-adrenergic receptors in adipocyte fat metabolism. 13 GABA-mediated lipolysis further requires both GABAA and GABAB receptors, as blocking either suppresses this response. 13 Nevertheless, the crosstalk between GABAergic and adrenergic receptors in lipolysis remains unclear due to limited studies.
Our findings reveal clear cell-type differences in NO regulation. LPS strongly induced NO in RAW264.7 macrophages but elicited only minimal NO in adipocytes. GABA reduced LPS-induced NO in macrophages but showed no effect in adipocytes due to their low NO levels. In addition, LPS primarily signals through toll-like receptor 4 (TLR4), which is abundant in leukocytes but also present on several non-immune cells, including adipocytes, hepatocytes, and muscle cells.30–32 Because FFAs share structural similarity with the LPS-binding region, it has been proposed that FFAs can also bind to and activate TLR4. In our experiments, 3T3-L1 adipocytes responded more strongly to PA than to LPS, suggesting that their TLR4 may be more effectively engaged by FFAs. TLR4 activation is known to play a key role in adipose tissue inflammation,33,34 which supports this observation. These observations suggest that inflammatory signaling varies depending on both the cell type and the stimulus involved. They also indicate that GABA influences NO pathways in immune cells and adipose cells through different mechanisms. 35
This study focused on functional and physiological endpoints, including glucose uptake, cytokine secretion, lipolysis, and NO production, without investigating intracellular signaling pathways at the molecular level. Moreover, adipose tissue in vivo is highly complex, consisting not only of adipocytes but also macrophages, endothelial cells, and fibroblasts within the stromal vascular fraction. In the present study, 3T3-L1 adipocytes were examined in isolation to characterize the cell-specific effects of GABA. While this approach helps clarify individual cellular responses, it does not fully reflect the multicellular interactions or systemic metabolic regulation present in vivo. In addition, both 3T3-L1 adipocytes and RAW264.7 macrophages are of murine origin, and their signaling characteristics may differ from those of human adipose tissue. Despite these limitations, these models provide valuable insights into fundamental cellular mechanisms underlying adipocyte dysfunction and inflammation. Furthermore, the effective concentrations of GABA observed in this study (500–1,000 µM) are relatively high and may not be achievable in humans. Nevertheless, these preclinical findings provide insight into the potential mechanisms of GABA action, possibly mediated through specific GABA receptor subtypes. Accordingly, the development or evaluation of more potent GABA receptor agonists may represent a promising direction for future research.
Conclusion
GABA improves insulin sensitivity and reduces inflammation in PA-induced insulin-resistant 3T3-L1 adipocytes, enhancing insulin-stimulated glucose uptake and lowering IL-6 and TNF-α. It also differentially affects lipolysis in PA-treated cells, reducing PA-induced glycerol release and restoring insulin’s anti-lipolytic effect highlighting its protective role against PA-induced adipocyte dysfunction. Additionally, GABA selectively inhibits LPS-induced NO production in RAW264.7 without affecting 3T3-L1 adipocytes.
Footnotes
Acknowledgements
This work was supported by Naresuan University (NU) and the National Science, Research and Innovation Fund (NSRF). Grant number: R2567B066. The authors gratefully acknowledge The Center of Excellence for Innovation in Chemistry (PERCH-CIC), Faculty of Pharmaceutical Sciences, Naresuan University, Thailand, for the support.
Authors’ Contribution
All authors made substantial contributions to conception and design, acquisition of data, or analysis and interpretation of data; took part in drafting the article or revising it critically for important intellectual content; agreed to submit to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. All the authors are eligible to be author as per the International Committee of Medical Journal Editors (ICMJE) requirements/guidelines.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Data Availability
All the data is available with the authors and shall be provided upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approvals
This study does not involve experiments on animals or human subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Naresuan University (NU), and National Science, Research and Innovation Fund (NSRF). Grant number; R2567B066.
Use of Artificial Intelligence-assisted Technology
The authors declare that they have not used artificial intelligence (AI)-tools for writing and editing of the manuscript, and no images were manipulated using AI.
Informed Consent
Not applicable.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of the publisher, the editors, and the reviewers. This journal remains neutral with regard to jurisdictional claims in published institutional affiliation.
