Abstract
Background and Purpose
The anti-obesity effects of vitalmelon extracts have been well verified in only adipogenesis and lipogenesis of adipocytes and high-fat diet-induced obesity mice except lipolysis. Therefore, we were investigated the novel function of edible vitalmelon fruit water extract (VW) on lipolysis.
Materials and Methods
The changes in the free glycerol release, cyclic adenosine monophosphate (cAMP)-dependent signaling pathway, and expression of β-oxidation-related regulators were analyzed in 3-isobutyl-1-methylxanthine, dexamethasone, and insulin (MDI)-stimulated 3T3-L1 adipocytes (MiSt-3T3 adipocytes) after a VW treatment.
Results
The successful differentiation of pre-adipocytes was confirmed with increased oil-red O (ORO)-stained lipid accumulation and PPARγ gene transcription. After maturation of adipocytes, the levels of free glycerol, intracellular cAMP, and hormone-sensitive lipase phosphorylation increased in a dose-dependent manner in the MDI+VW-treated group. In addition, the protein and mRNA levels of three β-oxidation-related genes (ACADs, ACO1, and CTP) were remarkably increased in the MiSt-3T3 adipocytes after VW treatment, while ATPCL phosphorylation was decreased. The changes in the expression levels of these proteins were accompanied by alteration on the expression levels of the PPARα transcription factors. The levels of PPARα protein phosphorylation and mRNA transcription in MiSt-3T3 adipocytes increased dose-dependently after the VW treatment.
Conclusion
These results show that VW can promote lipolysis by controlling the cAMP-mediated glycerol release pathway and β-oxidation in MiSt-3T3 adipocytes.
Introduction
Wild melons (Cucumis melo var. reticulatus) are commonly grown and used in the United States, Europe, and many Asian countries, including Korea, Japan, and China (Maietti et al., 2012). The pulp and fruit of this plant contain various bioactive compounds that are produced as secondary metabolites and are valuable for human health. Vitamins and β-carotene with strong antioxidant capacity were detected at high concentrations (36.7 mg/100 g and 2.02 mg/100 g) in the pulp of melon, but the total protein (840 mg/100 g) and lipids (190 mg/100 g) levels were very low (Gebhardt et al., 2008). In addition, the fruit of melon contains a significant level of bioactive polyphenols, which help prevent endothelium damage and cardiovascular disease, as well as potassium, an essential mineral, and electrolyte for maintaining physiological conditions (Koleckar et al., 2008; Lester, 2008; López et al., 2007). Furthermore, the concentration of bioactive compounds in melon extracts, including ascorbic acid, total flavonoids, total condensed tannins, total carotenoids, and total phenolics, were determined to compare the physiological state of plants in relation to the cultivar and seasonal variability (Maietti et al., 2012). The total phenolic and flavonoid contents were also detected in the hydroethanolic, hydromethanolic, and aqueous extracts of melon seeds and peel (Rolim et al., 2018). On the other hand, wild melon (C. melo var. reticulatus) is not cultivated for food because of its small size and bitterness; it is used mainly as a breeding material in Korea. Only C. melo var. makuwa Makino is well grown for food in many parts of Korea. The F1 hybrid produced by crossing C. melo var. reticulatus and C. melo var. makuwa Makino is commonly called vitalmelon and is registered with the Korea Research Institute of Bioscience and Biotechnology (KRIBB) under accession number KCTC14699BP (Guo et al., 2022).
The various parts (fruits, seeds, leaves, roots, and bark) of most melons have been reported to have anticancer, antimicrobial, anti-inflammatory, and anti-lipogenic activities (Guo et al., 2022; Maietti et al., 2012; Rolim et al., 2018). The cell proliferation of various types of cancer cells, including cervical adenocarcinoma, colorectal carcinoma, cervical carcinoma, and kidney carcinoma, was inhibited remarkably by some extracts of melon peels and seeds based on their antioxidant activity (Rolim et al., 2018). In addition, melons reduced the Salmonella Poona population significantly when applied to a chlorine dump tank after a levulinic acid (LV) treatment plus sodium dodecyl sulfate (SDS) (Webb et al., 2015). The anti-lipogenic effects of melon were observed in MiSt-3T3 adipocytes and a high-fat diet (HFD)-induced obese model. The enhanced glucose uptake, intracellular Tg accumulation, and PPARγ pathway in MiSt-3T3 adipocytes were alleviated by the vitalmelon fruit water extract (VW), while the increased levels of the weight of fat and liver, serum lipid profile, glucose regulators, and adipogenesis-related proteins were reduced under the same conditions (Guo et al., 2022). Despite this, no study has verified the efficacy and the action mechanism of the melon extract on lipolysis in differentiated adipocytes. Therefore, our study investigated the potential multidrug functions of VW extracts for their anti-obesity effects.
This study examined the lipolytic functions of VW and the mechanism involved in MiSt-3T3 adipocytes after a VW treatment for 2 days to determine its potential as a lipolysis stimulator.
Materials and Methods
Preparation of VW
The spray-dried form samples of VW were provided by Kwangdong Pharmaceutical Co. Ltd. (Seoul, Korea) after preparation, as described elsewhere (Guo et al., 2022). Briefly, the fruits of vitalmelon were harvested at 10 days of a fruiting setting based on the anthesis date because cucurbitacin A and B were detected at high concentrations (Guo et al., 2022). They were then freeze-dried and pulverized into powder. This powder was sifted with a 30-mesh pore-size sieve. The fruit powder (100 g) was extracted in an extractor with a 20-times volume (w/v) of dH2O at 80°C for 4 h. The filtration of extracted solutions was performed using Whatman No. 2 filter paper (Whatman International Ltd., Maidstone, England, UK). The final powder form sample (60.26 g) was harvested from the filtered solution using a spray dryer (Ilshin Lab Co. Ltd., Yangju, Korea) and stored in a freezer at –20°C before further analyses.
Determination of Bioactive Compounds in VW
The bioactive compounds in VW were analyzed by high-performance liquid chromatography (HPLC) (Agilent, Palo Alto, CA, USA) as described in a previous study (Guo et al., 2022). The VW samples were analyzed using the Luna C18 column (5 µm, 150 × 4.6 mm) under binary gradient elution (mobile phase consisted of 0.05% formic acid in distilled water (A) and methanol (B); gradient: 30% B in 0 min and 30–100% in 0–35 min, flow rate of 0.5 mL/min, and injected volume of 10 µL).
Culture and Differentiation of Adipocyte and Treatment of VW
Murine adipocytes 3T3-L1 were provided by the American Type Culture Collection (ATCC, Manassas, VA, USA). The Dulbecco Modified Eagle’s Medium (DMEM, Welgene, Gyeongsan, Korea) containing the essential compounds of appropriate dosage was used for cell culture.
The adipocytes were differentiated from pre-adipocytes as described in previous studies (Bae et al., 2020; Lee et al., 2021). Their differentiation was confirmed by oil-red O (ORO) staining analyses and enhancement of PPARγ expression. The lipolytic effects of VW were examined by classifying the differentiated 3T3-L1 adipocytes into five groups: No-treated group, dH2O-treated group (vehicle-treated group), low dosage (2 µg/mL) of VW-treated group (LVW-treated group), medium dosage (5 µg/mL) of VW-treated group (MVW-treated group), or high dosage (10 µg/mL) of VW-treated group (HVW-treated group). The VW treatment concentrations were determined based on the preliminary results of the cell viability for VW (Figure S1 in the supplemental material). Subsequently, VW-treated 3T3-L1 adipocytes were incubated for 2 days to ensure sufficient lipolysis effects based on the methods reported elsewhere (Castillo et al., 2019; Choi et al., 2023). Finally, these cells of the subset groups were harvested for further analysis.
ORO Staining Analysis
The accumulation of lipid droplets in 3T3-L1 adipocytes was determined using ORO staining analyses, as described elsewhere (Lee et al., 2021; Park et al., 2012). After staining with 0.5% ORO staining solution (Sigma–Aldrich Co.) for 30 min, the droplets that stained brown were detected using a microscope (Leica Microsystems, Wetzlar, Germany). Finally, the color intensity of droplets was determined using a VERSAmax plate reader (Molecular Devices, Sunnyvale, CA, USA) at 510 nm.
Quantitative Reverse Transcription-Polymerase Chain Reaction (RT- qPCR) Analysis
The transcription levels of the PPARα, PPARγ, and CPT genes were evaluated by RT-qPCR, as described elsewhere (Jeong et al., 2011). After synthesizing the cDNA template, the PCR products were quantified using 2× Power SYBR Green (Toyobo Co., Osaka, Japan) and the specific primer sequences (Table S1 in the supplemental material) under the specific condition as described in previous study (Jeong et al., 2011). The expression levels of each gene were evaluated with the threshold cycle (Ct) as described in a previous study (Livak & Schmittgen, 2001).
Western Blot Analysis
The expression level of each protein was evaluated using a Western blot analysis, as described in previous studies (Bae et al., 2020; Lee et al., 2021). Approximately 30 µg of the total cellular proteins were separated on 4–20% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and transferred onto nitrocellulose membrane (NC) membranes. Each protein on the membrane was detected using specific primary antibodies (Table S2 in the supplemental material) and horseradish peroxidase (HRP)-conjugated secondary antibody (Invitrogen). The chemiluminescence signals derived from the enzymatic reaction of HRP were evaluated using FluorChem®FC2 analyzer (Alpha Innotech Co., San Leandro, CA, USA).
Enzyme-linked Immunosorbent Assay (ELISA)
The concentration of cyclic adenosine monophosphate (cAMP) in 3T3-L1 adipocytes was analyzed using a cAMP ELISA kit (Cell Biolabs Inc., San Diego, CA, USA) according to the manufacturer’s guidelines. The total cell lysates were mixed with AP-conjugate and cAMP complete antibody. After several washes, the pNpp substrate was then added to each well containing this mixture and incubated at room temperature for 1 h. Finally, the absorbance of each sample was determined at 540 nm using a Vmax plate reader (Molecular Devices) after adding the stop solution.
Determination of Released Free Glycerol
The level of free glycerol was determined using a Cell-based Glycerol Assay Kit (Abcam Com., Cambridge, UK), as described elsewhere (Bae et al., 2020). After collecting the cell culture supernatants from 3T3-L1 adipocytes of each group, they (25 µL) were mixed with free glycerol reagent (100 µL) for 15 min. Their absorbance was determined at 540 nm using a Vmax plate reader (Molecular Devices). Finally, the following equation was used for the calculation of free glycerol concentration:
Free glycerol (µg/mL) = A540 – (y-intercept)/slope
Statistical Analysis
The statistical significance between each group was determined using one-way analysis of variance (ANOVA) (SPSS for Windows, Chicago, IL, USA) followed by a Tukey’s post hoc t-test. The data are represented as the means ± SD. Significance was given when p value was less than 0.05.
Results
Determination of Bioactive Compounds in VW
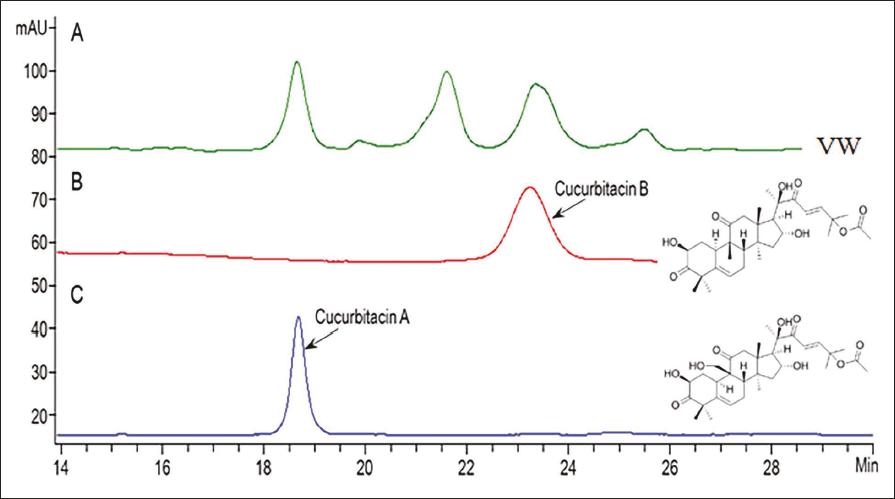
First, the bioactive compounds in VW were identified by HPLC. Two bioactive compounds, cucurbitacin A and B, were detected in VW, and their contents were approximately 34 µg/g and 2.5 µg/g dry weight, respectively (Figure 1).
Confirmation of Successful Differentiation of 3T3-L1 Adipocytes
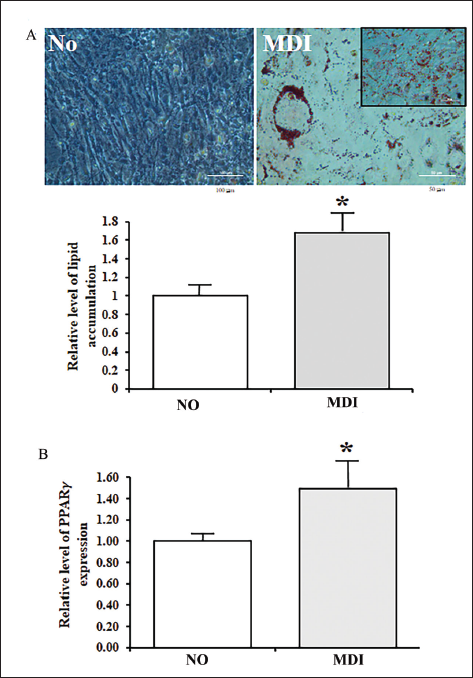
The changes in the lipid accumulation and PPARγ expression in MiSt-3T3 adipocytes were determined to confirm the successful differentiation of 3T3-L1 adipocytes after the 3-isobutyl-1-methylxanthine, dexamethasone, and insulin (MDI) treatment. The number of lipid droplets stained with ORO increased significantly after the MDI treatment compared to the No-treated group (Figure 2A). These changes were completely reflected in the level of PPARγ mRNA expression. They were enhanced in the MDI-treated group when compared to the No-treated group (Figure 2B). These results suggest that the MDI treatment can induce the differentiation of 3T3-L1 adipocytes. In addition, these cells are suitable for analyzing the lipolytic effects of VW.
Effects of VW on the Release of Free Glycerol from MiSt-3T3 Adipocytes
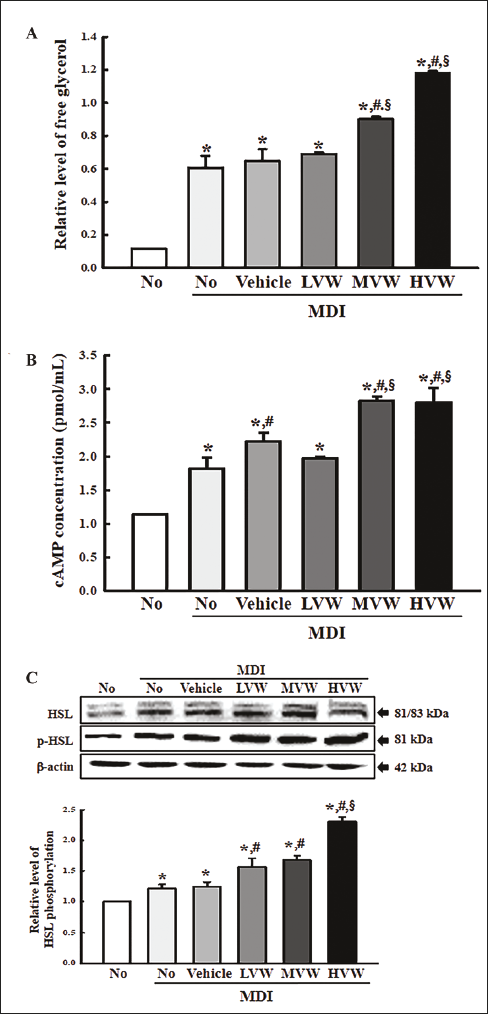
The level of free glycerol released in MiSt-3T3 adipocytes treated with VW was measured to determine if VW is linked to lipolytic activity. The level of free glycerol in the culture supernatant was significantly higher in the MDI+VW-treated group than in the control, even though the MDI+LVW-treated group was maintained (Figure 3A). Therefore, VW may have lipolytic effects in MiSt-3T3 adipocytes.
Effects of VW on the cAMP Concentration and Their Downstream Signaling Pathway in MiSt-3T3 Adipocytes
The cAMP concentration and the expression of key proteins on their signaling pathway were measured in MiSt-3T3 adipocytes treated with VW for 48 h to evaluate if the stimulatory effects of VW on the free glycerol release were accompanied by changes in the cAMP concentration and their downstream signaling pathway. The cAMP concentration increased remarkably in most MDI+VW-treated groups except for the MDI+LVW-treated group when compared to the control (Figure 3B). A significant increase in the VW-treated group was observed in the phosphorylation of the hormone-sensitive lipase (HSL) proteins. Their levels were remarkably enhanced in a concentration-dependent pattern after the VW treatment (Figure 3C). Therefore, the stimulatory effects of VW on glycerol release may be associated with the activation of the cAMP-mediated signaling pathway.
Effects of VW on β-oxidation of Lipid in Differentiated 3T3-L1 Adipocytes
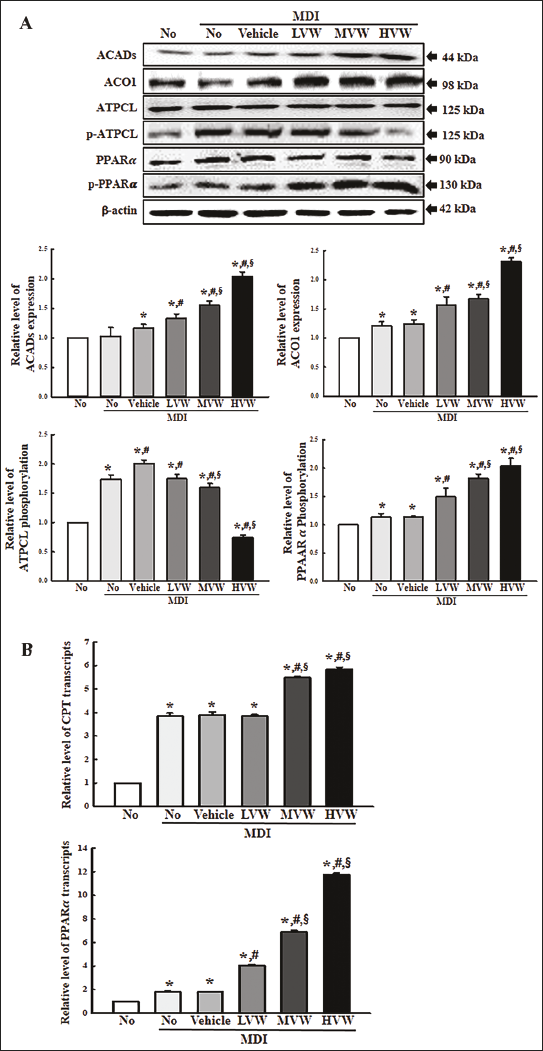
Finally, this study examined whether the lipolytic effects of VW were accompanied by stimulating the β-oxidation of lipids. The changes in the levels of β-oxidation-related markers, including ACADs, ACO, ATPCL, and p-ATPCL proteins, and their transcription factors, including PPARα and p-PPARα proteins, were analyzed in MiSt-3T3 adipocytes treated with VW for 48 h. Among the three β-oxidation-related proteins, the expression levels of ACADs and ACO proteins in the MDI+VW-treated group enhanced remarkably in a concentration-dependent pattern compared to the control (Figure 4A). On the other hand, the phosphorylation of ATPCL showed an opposite pattern to the above β-oxidation-related proteins. The protein level of ATPCL in the MDI+VW-treated group decreased significantly in a concentration-dependent pattern compared to the control (Figure 4A). In addition, a similar expression pattern was detected on the expression level of their transcription factor, which transcriptionally controlled the β-oxidation related genes. The phosphorylation level of PPARα protein in the MDI+VW-treated group increased remarkably in a concentration-dependent pattern compared to the control (Figure 4A). Furthermore, the changes in the protein levels of the transcription factor were reflected in the mRNA level (Figure 4B). In contrast, the mRNA level of the first transfer enzyme (CPT) for β-oxidation was enhanced after the VW treatment (Figure 4B). The results of the present study show that VW treatment may associate with stimulating β-oxidation in 3T3-L1 adipocytes.
Discussion
Fat metabolism can be classified into two main stages: (a) adipogenesis and lipogenesis (fat accumulation) and (b) lipolysis (fat degradation) (Rosen & Spiegelman, 2006). During these processes, many regulator genes and enzymes, including PPARγ, SREBP-1, ATGL, and HSL, play a key role in the balance between fat synthesis and breakdown (Kersten, 2001; Moseti et al., 2016). Based on the mechanism, the development of anti-obesity drugs affecting fat metabolism is underway to suppress lipogenesis and promote lipolysis (Rodgers et al., 2012). This study examined whether VW has lipolytic activity in differentiated adipocytes. These results provide evidence that VW can promote the lipolysis of MiSt-3T3 adipocytes by regulating the release of glycerol and β-oxidation.
The cAMP-dependent signaling pathway is the best mechanism for mediating lipolysis. cAMP production is associated with the G protein-coupled receptor in the plasma membrane and stimulated by adenylate cyclase, while the degradation of cAMP is mediated by phosphodiesterase (Carmen & Víctor, 2006; Egan et al., 1992). The resulting enhancement of the cAMP concentration induces the activation of cAMP-dependent protein kinase (PKA), which then phosphorylates HSL, perilipin, and adipocyte lipid-binding protein (ALPB) (Chaves et al., 2011). These proteins promote the release of free fatty acid and glycerol through the breakdown of triglycerides and diglycerides (Frühbeck et al., 2014). The analyses for the cAMP-dependent signaling pathways have been used widely to evaluate the lipolytic effects of several natural products. α-cubebenol and α-cubebenol isolated from Schisandra chinensis increase the concentration of cAMP, expression of adenylyl cyclase, and the activation of their downstream signaling pathway in MiSt-3T3 adipocytes (Bae et al., 2020; Lee et al., 2021). In addition, similar responses were observed in the MiSt-3T3 adipocytes after Citrus aurantium extracts, Emblica officinalis and Hordeum vulgare L. mixture, Toona sinensis extract, and an ice plant extract (Campitelli et al., 2020; Drira et al., 2016; Hsu et al., 2003; Park et al., 2021). This study measured the changes in the cAMP-dependent signaling pathway to evaluate the lipolytic effects of VW. The cAMP concentration and HSL phosphorylation were increased in the MiSt-3T3 adipocytes after the VW treatment. These results were like previous studies investigating the lipolytic activity of several natural products and bioactive compounds.
Fatty acid β-oxidation is a breakdown process of fatty acid to generate energy in various tissues (Lopaschuk, 2016). This process involves four major enzymes: acyl-CoA dehydrogenase (ACADs), enoyl-CoA hydratase (ECH), hydroxy acyl-CoA dehydrogenase (HADH), and ketoacyl-CoA thiolase (KAT) (Schreurs et al., 2010). Several factors tightly regulate the levels of these enzymes during the transcriptional and posttranscriptional steps (Fatehi-Hassanabad & Chan, 2005). In particular, PGC-1α and PPARs are some of the most well-known transcription factors, but each tissue has a specific type of transcription factor (Huss & Kelly, 2004). Based on the above scientific knowledge, the lipolytic activity of natural products was evaluated by measuring the expression levels of the proteins related to fatty acid β-oxidation. Immature Citrus sunki peel extract induced a decrease in acetyl-CoA carboxylase (ACC) phosphorylation in C57BL/6 mice with HFD-induced obese, whereas the ethanol extract of Smilax china L. enhanced the activities of carnitine palmitoyltransferase 1 (CTP1) and acyl-CoA oxidase (ACO) in HFD-induced obese ICR mice (Kang et al., 2012; Yang et al., 2019). Similar responses were detected in obese mice after treatment with the Allium hookeri root extract, Cirsium brevicaule A. GRAY leaf (CL) extract, and Hibiscus rosa sinensis flower extract (Inafuku et al., 2013; Kim et al., 2019; Lingesh et al., 2019). Furthermore, the β-oxidation-stimulating effects of natural products detected in obese mice were observed in mature 3T3-L1 adipocytes. The ethyl acetate extract of the H. rosa sinensis flower and extract of A. hookeri root enhanced the expression levels of some factors regulating fatty acid oxidation in mature 3T3-L1 cells (Kim et al., 2019; Lingesh et al., 2019). The present study analyzed the expression levels of β-oxidation-related enzymes and their transcription factors in MiSt-3T3 adipocytes after the VW treatment. The expression levels of most of these factors, except ATPCL, increased in a concentration-dependent pattern in MDI+VW-treated cells. Therefore, VW has high potential as a novel natural product with lipolysis-promoting effects.
Conclusion
The present study investigated the novel therapeutic functions and action mechanism of VW on lipolysis of mature adipocytes. In mature adipocytes, the VW treatment increases the release levels of free glycerol by regulating cAMP concentration and phosphorylation of HSL, whereas it recovers the expression levels of the β-oxidation-related proteins by regulating the PPARα. The results show that VW stimulates lipolysis in mature 3T3-L1 adipocytes. Nevertheless, more studies will be required on the mechanisms for their action in laboratory animals.
Summary
Functional foods and their derived products are used widely to treat various chronic diseases, including diabetes, obesity, dyslipidemia, arteriosclerosis, and hypertension. The fruit of melon has attracted considerable attention for its potential for obesity treatment because it contains a significant level of various bioactive polyphenols. As part of this study, the mechanism of action on anti-lipogenesis and anti-adipogenesis effects of melons had been well studied in MiSt-3T3 adipocytes and HFD-induced obese models. This study examined the novel function of edible VW on lipolysis and the molecular mechanism in MDI-stimulated 3T3-L1 adipocytes. VW has a prominent effect against key regulators, including free glycerol, intracellular cAMP, and HSL phosphorylation on the cAMP-mediated glycerol release pathway. Furthermore, VW exhibits β-oxidation related genes, such as ACADs, ACO1, and CTP, which stimulate the β-oxidation of fat. Therefore, VW might be a prominent therapeutic agent to improve obesity.
Abbreviations
ACADs: Acyl-CoA dehydrogenase; ACC: Acetyl-CoA carboxylase; ALPB: Adipocyte lipid-binding protein; CL: Cirsium brevicaule A. GRAY leaf; DMEM: Dulbecco Modified Eagle’s Medium; HRP: Horseradish peroxidase; LV: Levulinic acid; ORO: Oil-red O; PKA: Protein kinase; KRIBB: Korea Research Institute of Bioscience and Biotechnology; RT-qPCR: Quantitative reverse transcription-polymerase chain reaction; SDS-PAGE: Sodium dodecyl sulfate–polyacrylamide gel electrophoresis; VW: Vitalmelon fruit water extract.
Footnotes
Acknowledgments
The authors wish to thank Dr So Hae Park, Department of Biomaterials Science (BK21 FOUR Program) in Pusan National University, for helping interpret the results of the bioactive compounds analysis.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grants funded by the Korean government (MSIT) (NRF-2021R1I1A3044431). Also, this is study was also supported by the BK21 FOUR Program through the National Research Foundation of Korea (NRF), funded by the Ministry of Education, Korea (F22YY8109033).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
