Abstract
We aimed to investigate the validity of urinary N-terminal titin (TTN) fragment as a biomarker for limb-girdle muscular dystrophy LGMDR1-calpain 3 related. Thirteen LGMDR1 patients and eleven healthy controls were enrolled for the study. LGMDR1 patients had significantly increased urinary N-terminal titin fragment concentrations than age-matched controls. Even if urinary level of titin decreased with aging, it was still high in wheelchair bound patients. Thus, our findings indicate that urinary N-terminal titin fragment is a non-invasive measure of muscle damage in LGMDR1, which could be used in disease monitoring in clinical trials even in wheelchair-bound patients.
Introduction
Limb girdle muscular dystrophy R1 (LGMDR1) calpain3-related, is one of the most frequent recessive LGMD forms, globally accounting for approximately 30% of all recessive LGMD cases.1–4 Typical cases start with limb-girdle weakness in the second decade of life and become wheelchair-bound 20 years after clinical onset.5,6 Moreover, muscle degeneration continues and simple daily tasks become extremely difficult. To date, no effective treatments against the disease lacking calpain 3 have been developed.
The slow progression and the difficulty of assessing muscle strength in muscular dystrophy makes it hard monitoring therapy efficacy in a clinical trial. Biomarkers are crucial to objectively evaluate muscular dystrophies progression and monitoring therapeutic approaches. 7
Muscle breakdown or altered signaling pathways in myofibers release molecules into the blood stream that can be used as biomarkers in serum, plasma or urine samples. 8
Blood tests show that LGMDR1 patients experience a marked increase of their serum Creatin Kinase (CK) level (5–80 times increased) in the early stages of the disease, but it progressively decreases until it reaches normal levels in wheelchair-bound patients. 6 Previous studies in different muscle dystrophies have revealed variations in protein levels such as myomesin-3, 9 troponin10,11 and titin12–14 in different biofluids.
In skeletal muscles, calpain 3 binds to titin at multiple sites. The interaction in one of these sites, calpain 3-N2A, suppress calpain 3 autolysis and protects titin from proteolysis15,16
In this work, we measured urine N-ter titin/Cre concentrations in LGMDR1 patients compared to healthy controls and established that it is a useful and easily available biomarker for future clinical trials.
Methods
Urine titin and creatinine in human samples
Human samples from LGMDR1 patients and age-matched healthy controls were provided by the “Donostia University Hospital, Spain” after informed consent from each participant, who voluntary signed prior to the collection of samples. Samples were stored at −80 °C until the assay was performed. The mean age (average ± SD) of LGMDR1 patients (n = 13) and healthy controls (n = 11) were 29.92 ± 15.33 (ranging between 13 and 62) and 30.91 ± 15.75 (ranging between 14 and 61) years old, respectively. Functional stage of the dystrophy was graded according to North Star Assessment for Limb Girdle Type Muscular Dystrophies (NSAD). 17 Patients’ mutations and clinical information are detailed in Table 1. Urine titin was measured using Human Titin-N Fragment Assay Kit-ELISA system (Cat#: 27,900, IBL) according to the manufacturer's instructions. To adjust the concentration of urine titin within the quantitative range of the assay kit (46.88–3.000 pmol/L), urine samples from LGMDR1 patients and healthy volunteers were diluted 1:500 and 1:50, respectively, using the dilution buffer contained in the assay kit.
Analyzed urine samples. North Star Assessment for Limb Girdle Type Muscular Dystrophies (NSAD) Functional scale.
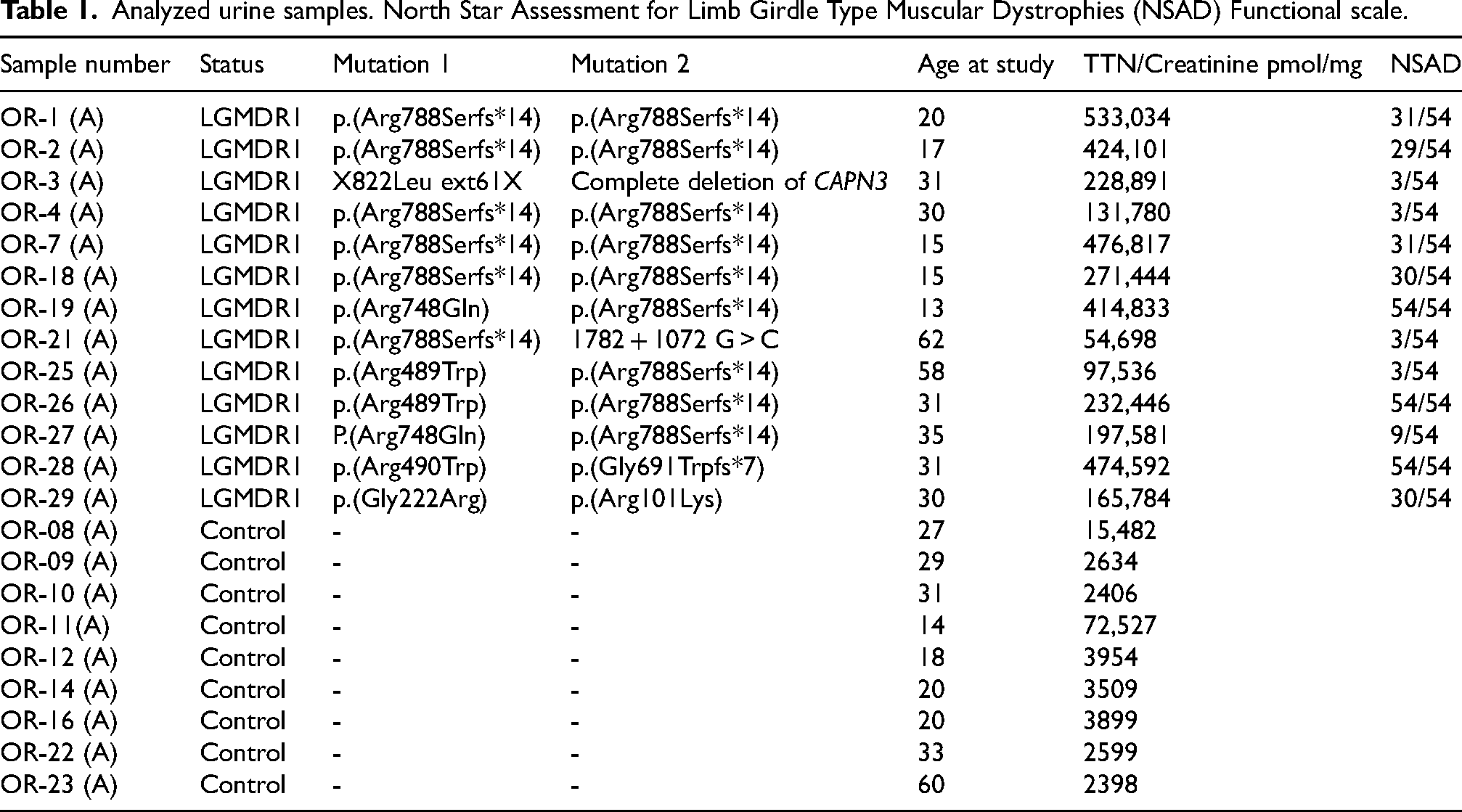
The creatinine colorimetric assay (Cayman Chemicals) was used for normalization. For urine creatinine determination, all samples were diluted 20 times in distilled water and measured following manufacturers’ instructions.
For control and patients N-ter titin concentrations, the statistical significance was assessed by an Unpaired T-test. Correlation of titin N-ter fragment concentration and age or functional scale in LGMDR1 patients was performed by Pearson correlation coefficient (r).
Results
The obtained results showed that LGMDR1 patients showed increased N-ter titin fragment level in urine samples. It was significantly higher in LGMDR1 patients with a mean ± SD of 284.90 ± 160.70 and 10.66 ± 20.86 pmol/mg Cre in LGMDR1 patients and healthy controls, respectively (Figure 1A).
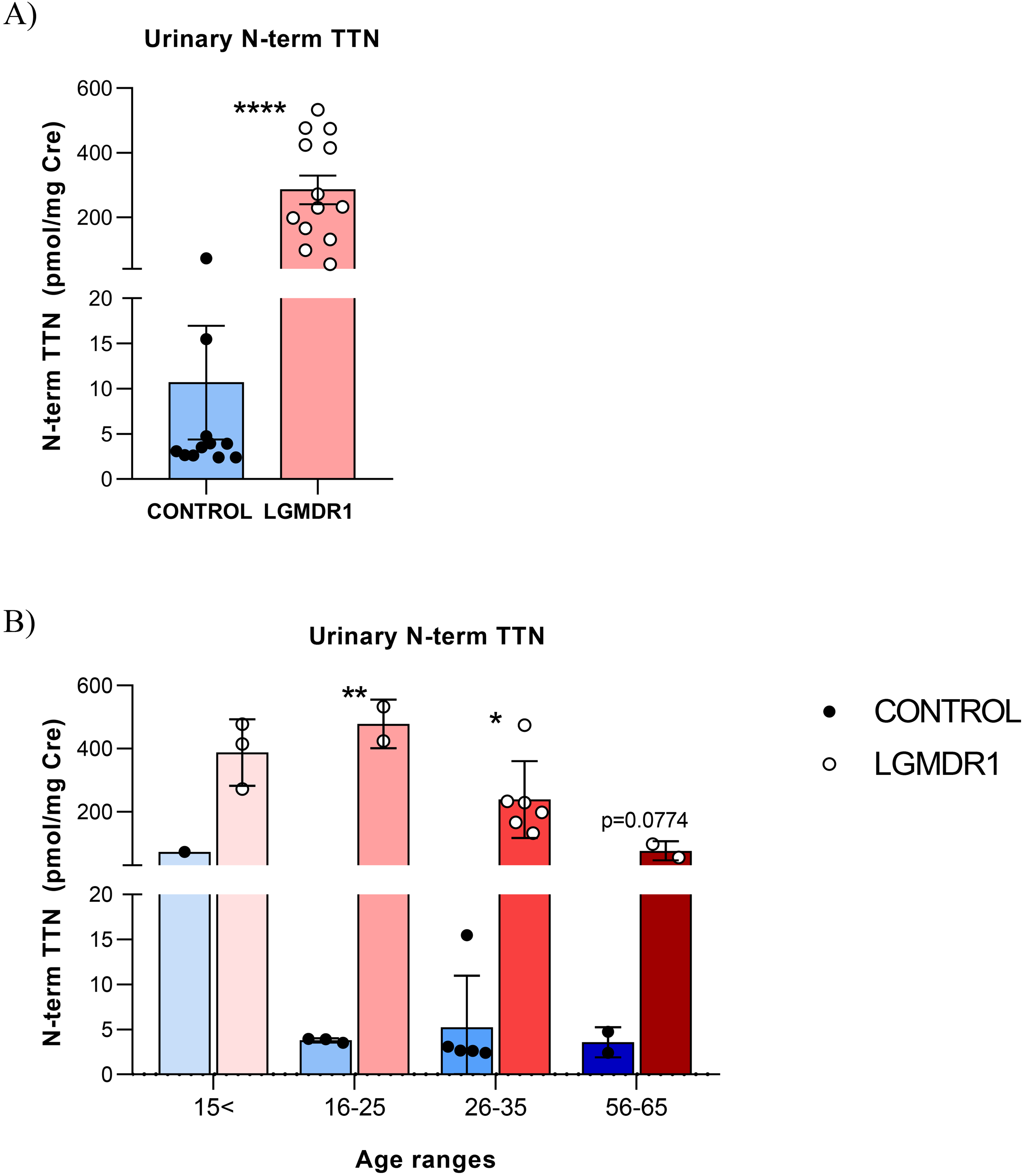
Urinary N-term TTN normalized concentrations in samples from HC and LGMDR1 patients. (A) Control and patients N-ter titin values and (B) samples grouped by age. Error bars represent Standard Deviation (SD). *p < 0.1, **p < 0.01, ****p < 0.0001.
Due to the observed wide distribution of the N-ter titin/Cre levels, mostly in patients, we split the samples according to age ranges. Differences were kept between the patients and controls in the different groups. In the youngest group, a young control (14 years-old) showed the highest N-ter titin/Cre concentration observed among controls. In the 16–25 and 26–35 years old group, the N-ter titin/Cre levels were significantly higher in patients (**p = 0,0013 and *p = 0,0117 respectively). The oldest group (56–65 years old) showed differences but do not reach statistical significance (Figure 1B).
The highest urine N-ter titin/Cre level was observed in the 16–25 year-old patients group, showing also the greatest difference between controls and LGMDR1 patients (a 126-fold higher). N-ter titin/Cre levels lightly decline with aging in LGMDR1 patients, although this reduction was not observed in healthy subjects (Figure 1B). In the most aged group, patients were not ambulant and they still showed significantly increased N-ter titin/Cre levels (more than 20-fold higher).
The correlation of the age with the N-ter titin/Cre concentration in controls (R2 = 0.1373) and patients (R2 = 0.5540; **p = 0.0035) was analysed (Figure 2A). We also analysed the N-ter titin/Cre level related to the phenotype severity in LGMDR1 patients
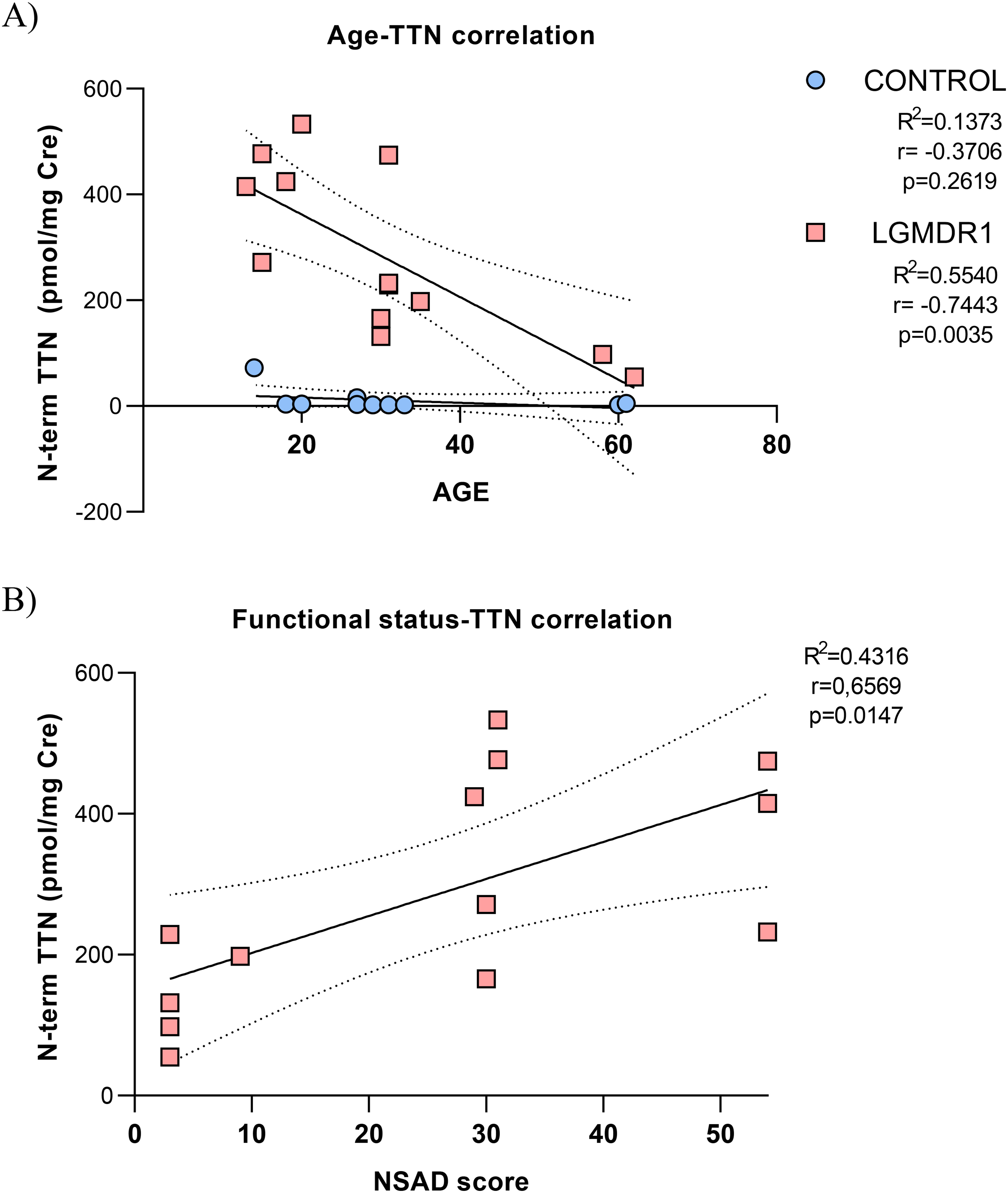
Correlation of titin N-ter fragment concentration and age or functional scale in LGMDR1 patients. A) Age/Urinary N-term TTN and B) phenotype severity/Urinary N-term TTN correlation analysis. **p < 0.01.
Finally, according to creatinine levels, patients showed a lower creatinine concentration that correlates with reduced muscle mass18,19 (Supplemental Material Figure S1A). Furthermore, urine N-term TTN without creatinine normalization also showed significant higher concentrations in LGMDR1 patients’ samples (Supplemental Material Figure S1B).
Discussion
Even if blood CK concentration was considered as a disease progression marker, it is unspecific since its increase has also been found after physical activity.20,21 Moreover, in LGMDR1 patients, serum CK level progressively decreases until it reaches normal levels when patients are wheelchair-bound. 6 This greatly limits its use as a biomarker in clinical trials where wheelchair-bound patients would participate. Thus, identifying a trustful biomarker for LGMDR1 was required for future clinical trials. The interaction of calpain 3 with the N2A region protects titin from proteolysis. 16 Therefore, as the lack of calpain 3 may affect the proteolysis rate of titin in LGMDR1 muscles, it was considered a good candidate to be used as a biomarker. A recent study reported that in two LGMDR1 patients N-terminal fragment of urine titin was approximately 100-fold higher than in controls. 22
It should be cautious when analyzing samples obtained from young people since we observed in a young control (14 years-old) the highest N-ter titin/Cre level (72.56 pmol/mg Cre). Nevertheless, the young age-matched patients showed much higher titin fragment levels (387.70 ± 105,30 pmol/mg Cre). The observed highest N-ter titin/Cre level in this control may be associated to the high protein turnover at growth, but further analysis of young teenagers are required. In any case, we underline the need of always including age-matched controls for the analysis.
In this study, a limitation of analyzing longitudinal data is acknowledged. Thus, analyzing the variation of the N-ter titin/Cre concentration during time would be required to establish a good correlation with the disease activity.
In a previous work, N-ter titin/Cre concentration was analysed and in ambulatory patients it was higher than in non-ambulatory patients in DMD patients. 12 However, in the study by Ishii and colleagues 14 in patients over 10 years old, there were no differences in urine N-ter titin/Cre between ambulant patients and non- ambulant patients. In wheelchair bound LGMDR1 patients, urine N-ter titin/Cre was also elevated. This is of great value for clinical trials since muscle recovery testing is limited in these cases.
Additionally, urinary N-terminal titin fragment has already been used to predict mortality in dilated cardiomyopathy, 23 as a novel biomarker for amyotrophic lateral sclerosis 24 and in idiopathic inflammatory myopathy. 25 This underlies the usefulness of this biomarker for the study of a great variety of diseases where muscles are affected. Furthermore, it has already been used as a biomarker for microdystrophin efficacy in a clinical trial. 8
Assessing the levels of urine biomarkers will enable, the regular sample collection and the valuation of changes when functional changes may be difficult to detect, establishing the efficacy of the treatment. The present study shows a good correlation between urinary N-ter titin/Cre with the phenotype severity and the age. Thus, urinary N-terminal titin fragment is considered as a useful marker for the disease follow up and to monitor the drug efficacy in a clinical trial in LGMDR1.
Supplemental Material
sj-docx-1-jnd-10.1177_22143602251323629 - Supplemental material for Urinary N-terminal titin fragment ascertained as biomarker in a small cohort of limb-girdle muscular dystrophy LGMDR1-calpain 3 related
Supplemental material, sj-docx-1-jnd-10.1177_22143602251323629 for Urinary N-terminal titin fragment ascertained as biomarker in a small cohort of limb-girdle muscular dystrophy LGMDR1-calpain 3 related by Andrea Valls, Cristina Ruiz-Roldán, Jenita Immanuel, Pilar Camaño, Juan José Poza, Roberto Fernández-Torrón, Adolfo López de Munain and Amets Sáenz in Journal of Neuromuscular Diseases
Footnotes
Acknowledgments
The authors would like to thank the patients, their families and the healthy controls. We would like to thank Patricia Garay-Albízuri for her contribution to score the functional status of the patients.
Funding statement
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study has been funded by Instituto de Salud Carlos III (ISCIII) through the project “PI21/00047” and co-funded by the European Union. It has been funded also by Department of Health of the Government of the Basque Country (project ref 2021111022 and 2024111043) and Association Française contre les Myopathies (AFM 24743). This work was, in part, supported by the Center for Networked Biomedical Research on Neurodegenerative Diseases (CIBERNED: CB06/05/1126 to Andrea Valls, Pilar Camaño, Adolfo López de Munain, and Amets Sáenz), GENE (Association of Neuromuscular diseases of Gipuzkoa).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Datasets/data availability statement
The data supporting the findings of this study are available within the article and/or its supplementary material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
