Abstract
Healthcare reform has changed the manner of delivery of health services forever. One of the areas of greatest impact has been in clinical laboratory services. While the cost of health care overall has risen steadily over the past 5 years, the actual cost of a Chem 7 panel in our laboratory, for example, has decreased by 31%. This example of cost control is undoubtedly typical of the focus which clinical laboratories have placed on cost containment in recent years.
Clinical laboratories have responded to the imperative to decrease operating costs by several initiatives, including (1) consolidation of multiple laboratory sites within a given institution, (2) reevaluation of breadth of test menus, (3) discontinuance of internal offering of low volume tests, (4) adoption of more automated instrumentation, (5) entering into cooperative relationships with other laboratories [partnerships, networking, regionalization], and (6) adopting manufacturing industry approaches to laboratory automation.
The Dept. of Pathology/Clinical Laboratories at Duke University Medical Center have been involved extensively in such productivity improvements. Over the past five years, we have implemented activities in initiatives (1) through (4), and more limited in (5). These actions have provided us with the opportunity to see billed revenues increase by 38%, while operating costs have increased by only 17% (Figure 1). While recovered revenues have become significantly less than billed revenues, this favorable revenue/expense ratio has allowed us to remain as a profit center for the institution.
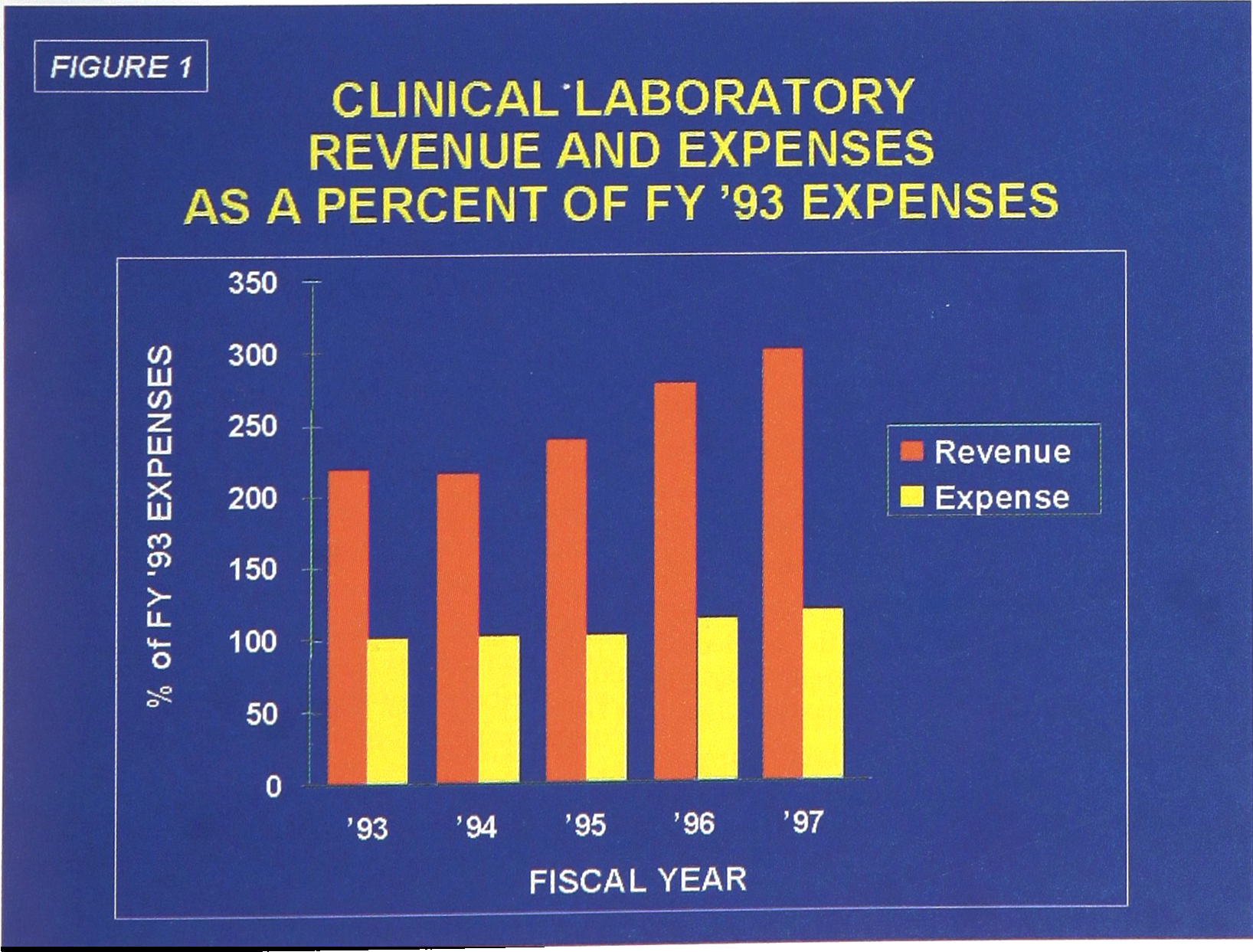
Clinical Laboratory Revenue and Expenses
LAB REDESIGN PROJECT
In an effort to assure that we continue our productivity improvement efforts, for the past two years we have directed our attention at a more in-depth review of our laboratory structure and operations, in a laboratory-wide redesign project. Using a project team with representation from our Department, the Medical Center, Hospital Administration, Procurement and the Project Engineering Office, an in-depth assessment of six potential approaches to implementation of laboratory redesign were undertaken: internal reorganization, partnerships, joint ventures, risk sharing agreements, networking, and outsourcing.
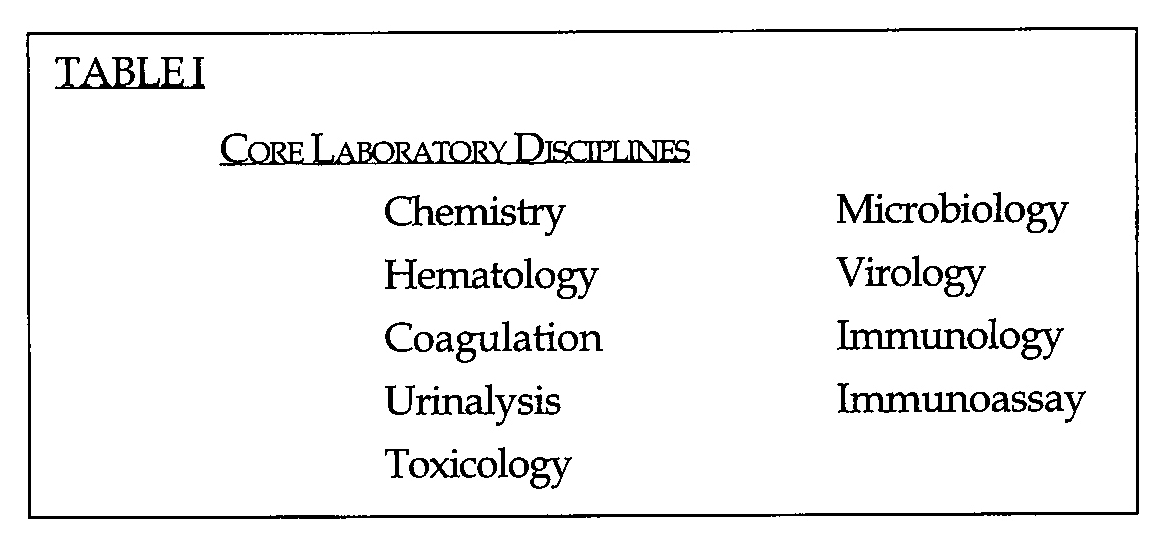
The outcome of the project team's efforts was to move ahead with a significant reorganization of the method of performing clinical laboratory medicine at Duke. It was recognized that certain laboratory services would be required to remain intact as STAT or specialty services. The remaining core laboratory service lines (Table I) should be:
Brought together as an “FLAT” Lab (immediate turnaround time—TAT of < 60min for 80% of specimens), Function as a “Laboratory Without Walls”, with maximum cross training and flexible assignment of personnel, Provided with the benefits of TLA (total laboratory automation) in a phased implementation approach, Supported by a central client service desk to handle all laboratory inquiries, Positioned to provide optimal support for the medical center's continuing regional outreach initiatives, and Retain the “Centers of Excellence” laboratory organization model [1]
Core Laboratory Disciplines
TLA IMPLEMENTATION PLAN
It was determined early in the planning process that the TLA project should be implemented in several phases, because (1) the current space would need to be renovated and new equipment installed while the laboratory continued 24 hr/day operations, (2) the expected labor savings could more fully justify the capital investment in several segments as test volumes increased, and (3) the impact on continuing operations and personnel would be lessened.
Consequently a three phase implementation approach was developed:
Phase I — Installation of Automated Front End Processing System Phase II — Selection and Acquisition of Six Automated Instrument Platforms Phase III — Installation of Specimen Carrier and Remaining TLA System
Specimen flow for the four functions of the Automated Front End Processing System are diagrammed in Figure 2. A conceptualized diagram of all three phases in shown in Figure 3. Note the test disciplines represented by the six instrument systems to be selected for Phase II. It is expected that these six analytical platforms will provide 80% of all test requirements in the ITAT lab. Support for a continuing requirement for manual tests is also provided.
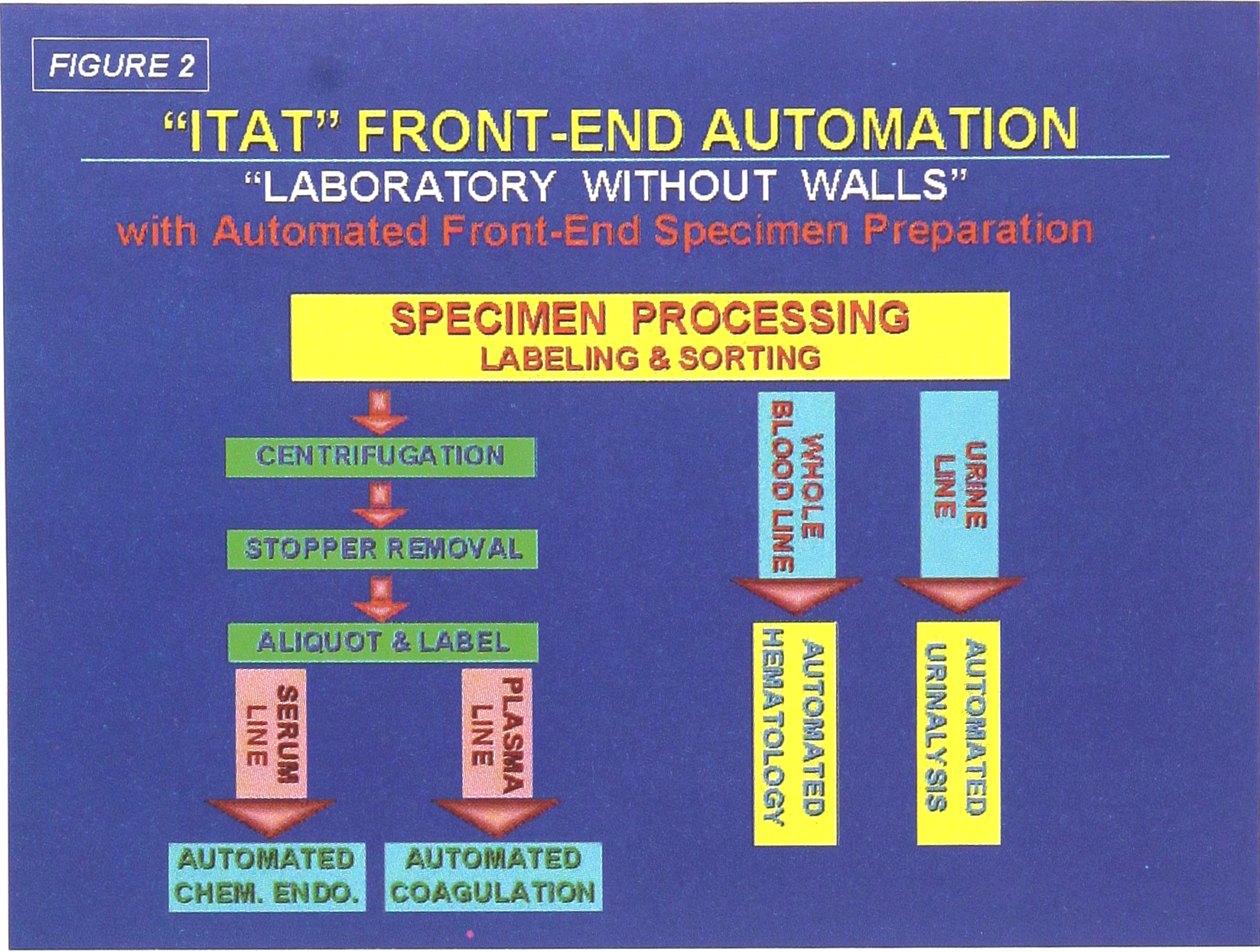
“ITAT” Front End Automation
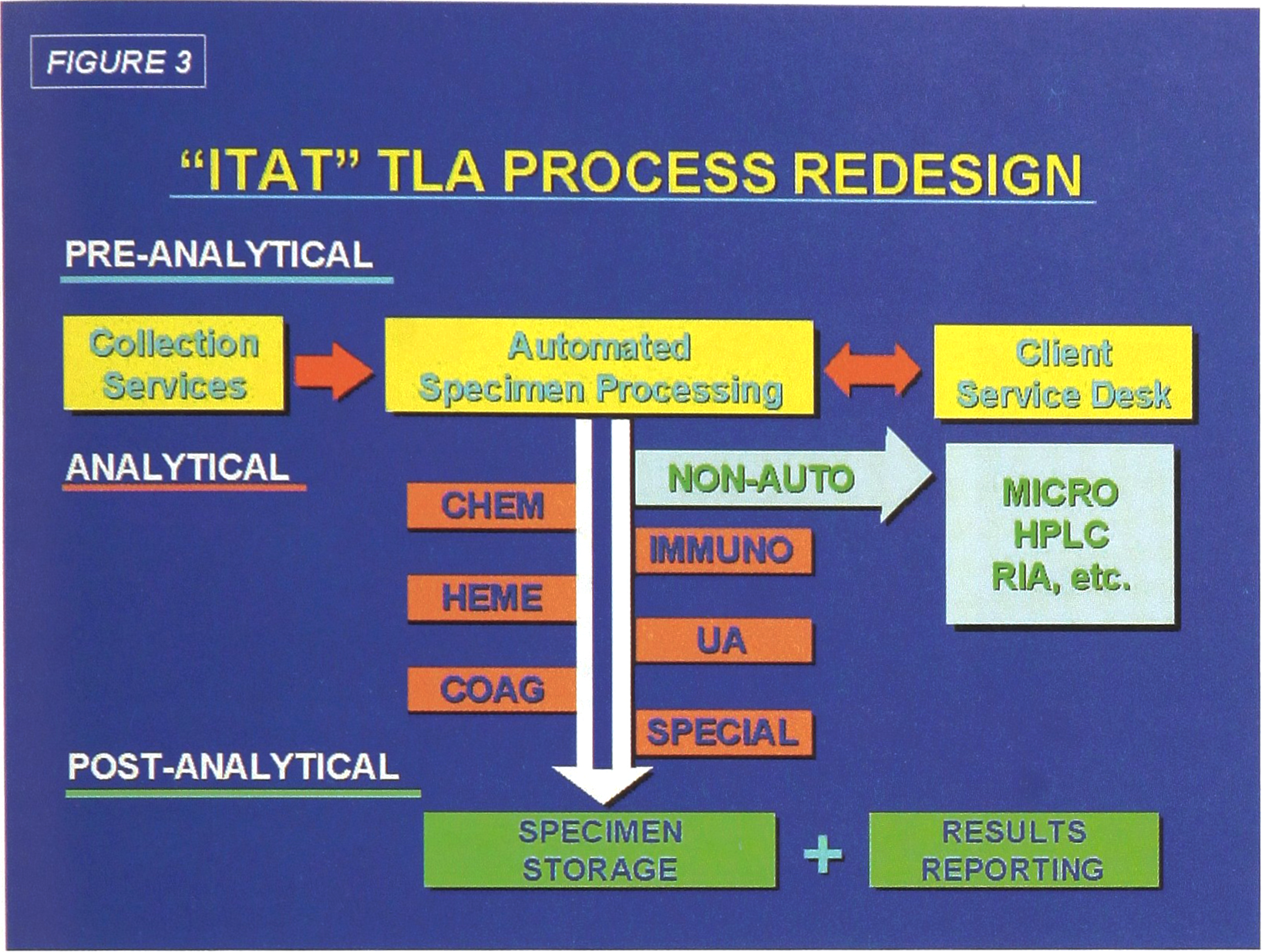
“ITAT” TLA Process Redesign
PROJECT JUSTIFICATION
It has long been recognized that the major manual labor components in the total analytical process, including those at Duke, are the pre-analytical steps involved in specimen collection (acquisition, transport) and specimen processing (receiving, accessioning, centrifugation, aliquotting, sorting). The subsequent steps of analysis and result reporting have been made available by instrument and information system vendors, respectfully, for several years. Likewise, these steps are minor labor components in the Duke laboratory system.
The major focus for justification of TLA was in the reduction in manual labor requirements in the planned ITAT functions. By careful audit of all laboratory processes, we determined that approximately 53% of the total workload is currently devoted to pre-analytical steps. By automation of front-end processing (which requires barcoding of all specimens prior to accessioning), front-end processing drops to 21% of the total labor requirements.
Upon completion of the total system, analytical and post-analytical labor requirements are also reduced (Figure 4). Full implementation of the TLA system will allow for some 67% of the present assigned labor, covered by the ITAT functions, to become available to support the expanded outreach efforts of the laboratories.
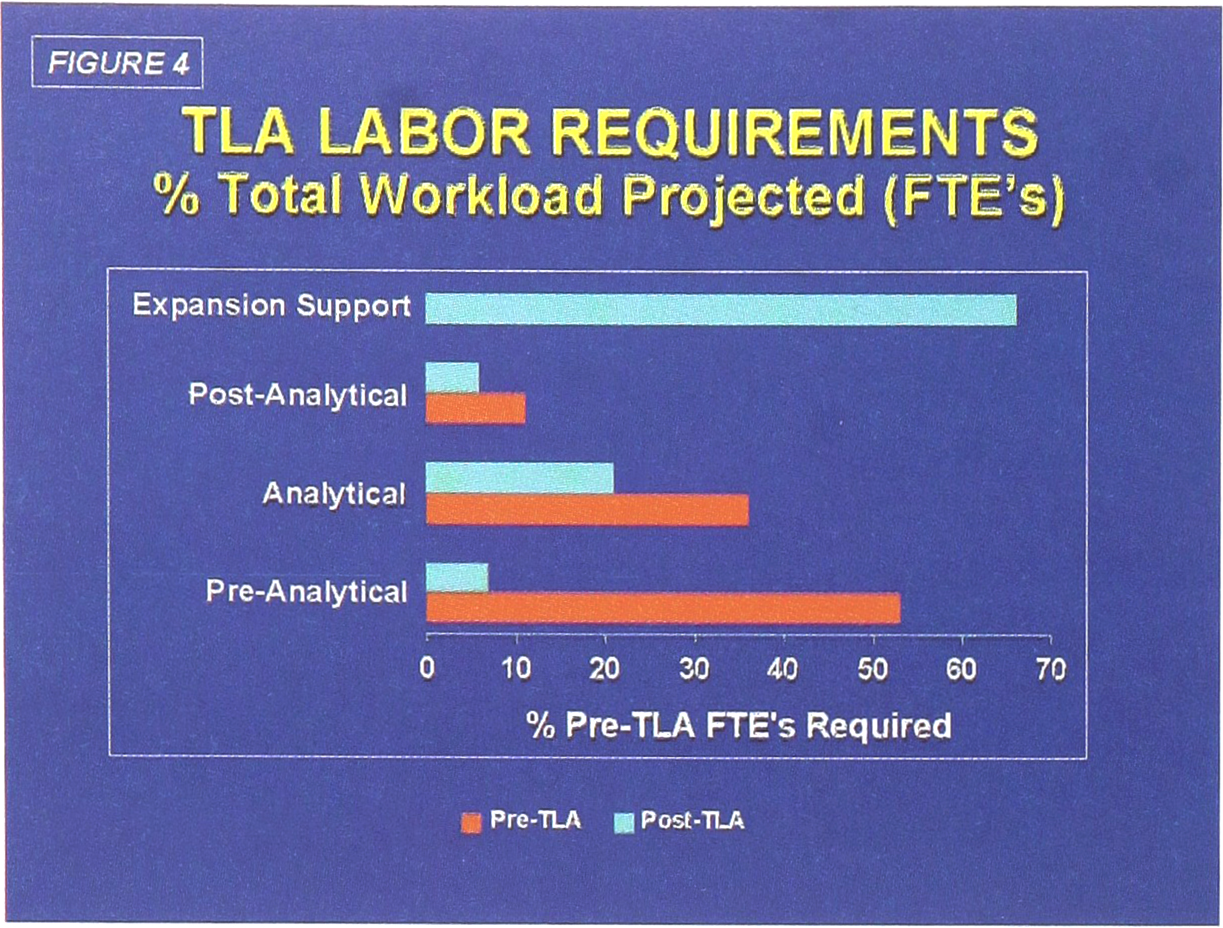
TLA Labor Requirements
The most dramatic labor reductions, as expected, will be in front-end processing (Pre-analytical), where the manual labor requirements drop by 87%. A further 52% reduction is projected to be achieved from automated specimen loading (Analytical) onto the six major laboratory instrument systems (chemistry, hematology, immunoassay, coagulation, urinalysis, special chemistry). Automated removal and storage of specimens (Post-analytical) account for another 44% decrease in labor (Figure 4). The overall distribution of the projected impact of TLA on labor requirements is shown in Figure 5.
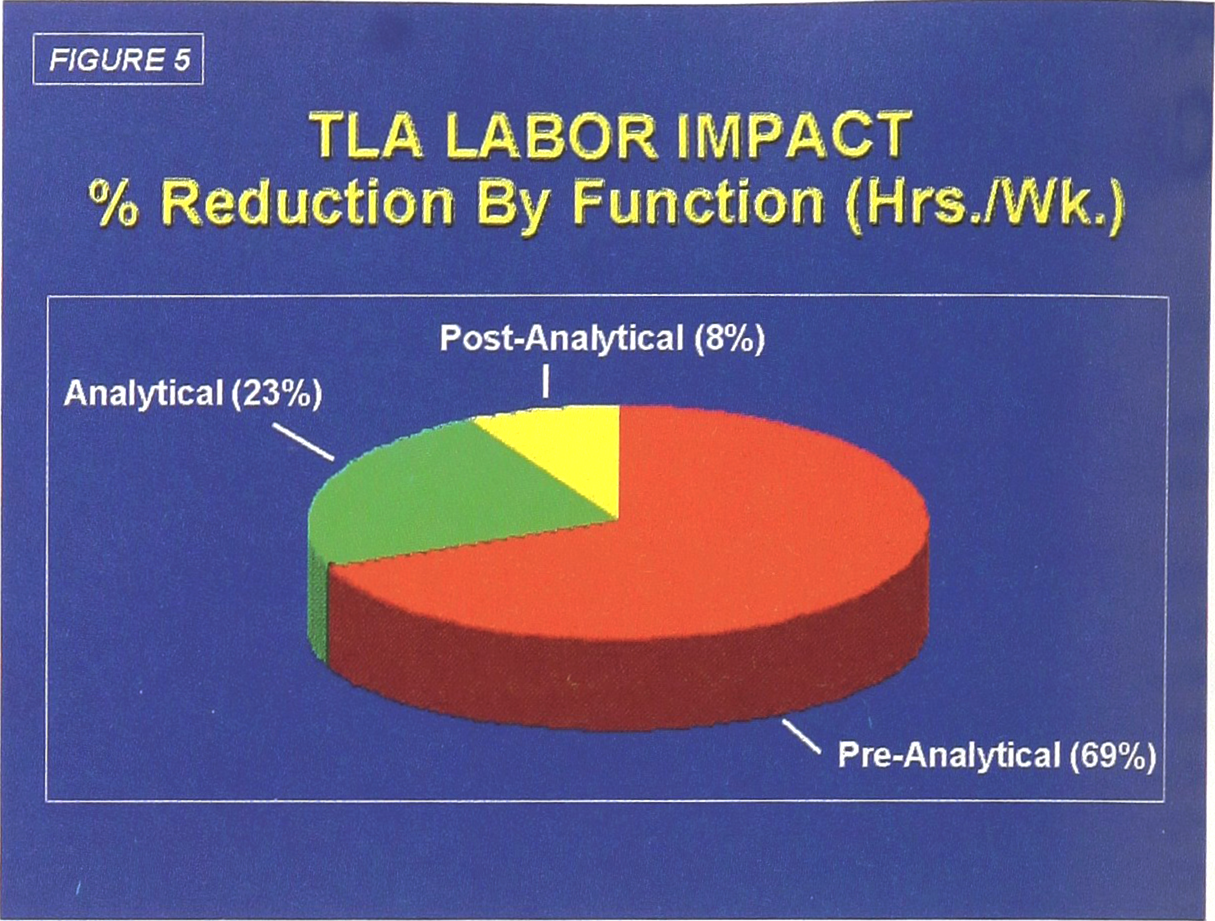
TLA Labor Impact
TLA VENDOR SELECTION
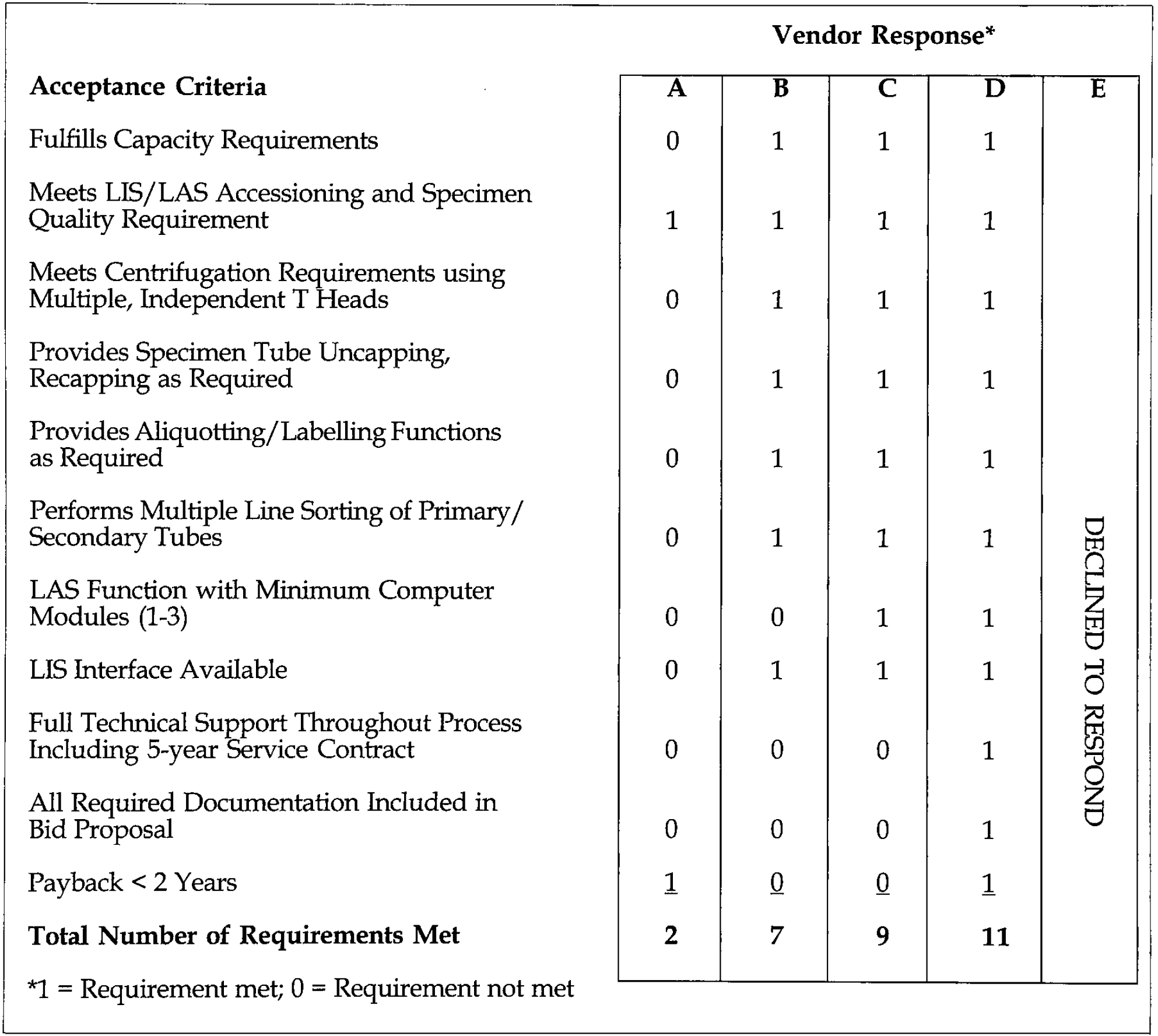
Once the project recommendations had been accepted by senior administration, it became necessary to select a vendor and envision the placement of the TLA system within the existing ITAT laboratory structure. A list of performance characteristics were developed (Table II), acceptance criteria were prepared (Table III) and formal bids were solicited from the major North American TLA vendors Table IV).
TLA Performance Characteristics

TLA ACCEPTANCE CRITERIA AND VENDOR RESPONSE
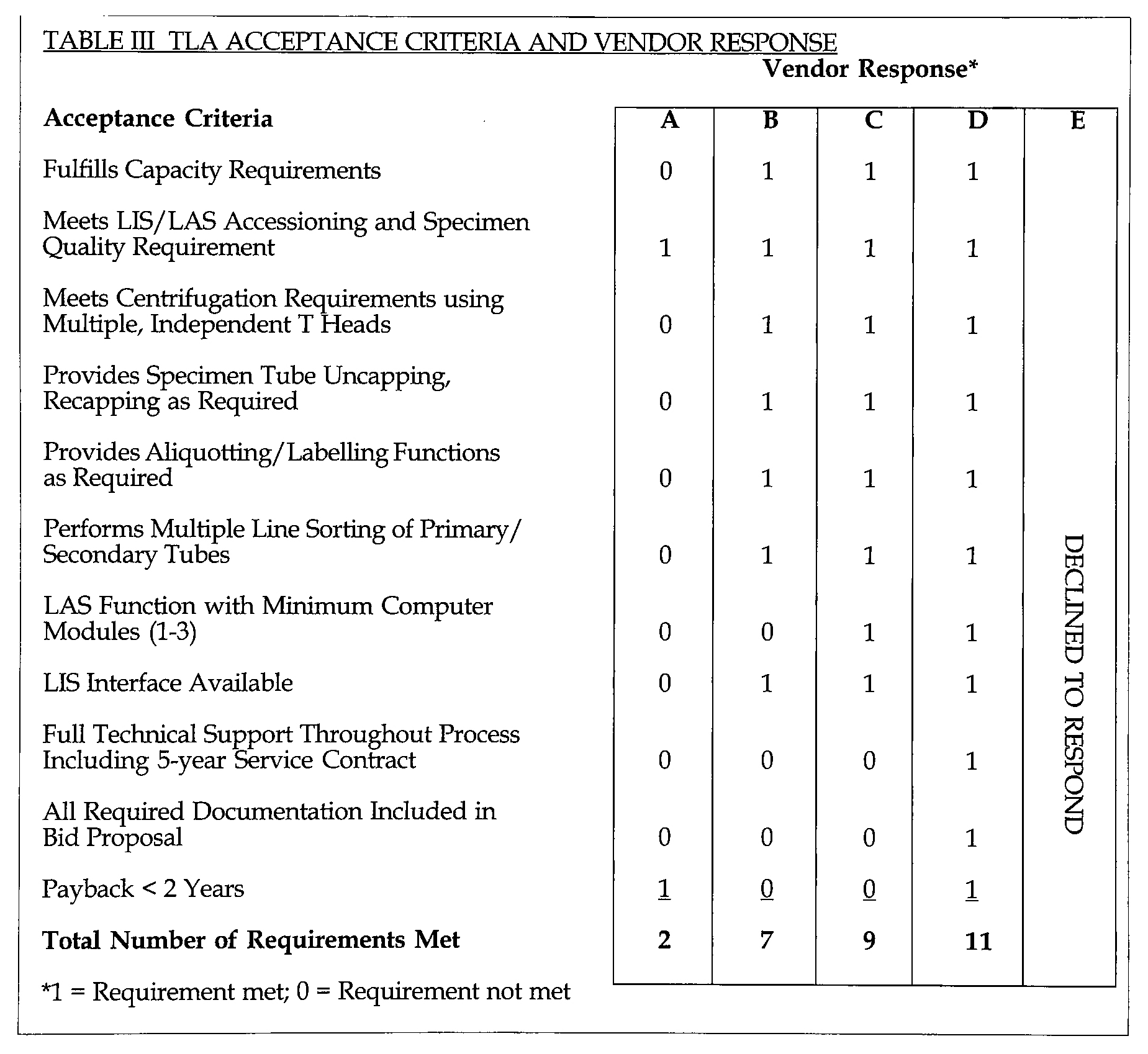
1 = Requirement met; 0 = Requirement not met
North American TLA Vendors

Vendor responses were then evaluated against the requirements (Tables II, III) and scored for acceptability. Keyed vendor scores are summarized in Table III. The impartial manner in which the selection process was conducted provided a clear decision to select vendor D, Labotix Automation, Inc. as the vendor for the project.
The Labotix proposal met all project requirements and was bid at a price projected to be recovered in less than two years from labor cost reductions.
Unique features of the Labotix proposal included:
Simple, flexible specimen transport system Single LAS computer unit compatible with our LIS Accommodation of all specimen tubes utilized in our system in individual handlers Accommodation of all anticipated automated test instruments and specimen tube carriers Minimum number of individual robotic systems On-board automated refrigerated specimen storage Flexible Components Placement Competitive Price
CONCLUSION
The decision to undertake a major clinical laboratory redesign project is becoming more prevalent. In our experience, we have learned that such a decision needs to be taken very seriously, and to be successful must be initiated only after preparing a carefully designed project plan, identifying the key components to assure successful completion of the plan, and careful selection of the automated equipment, to best meet the requirements of the project.
Furthermore, such a decision to consider automation in the laboratory, while prudent, should be only one of several redesign initiatives which should be explored.
The industrial engineering support of Argent Consulting Services, Inc., Oklahoma City, OK, is gratefully acknowledged.